Abstract
A B-cell, Burkitt-type lymphoma, diffusely affecting the peripheral nerves and intramuscular nerve branches was diagnosed in a 4-year-old domestic shorthair cat with a chronic progressive history of flaccid tetraparesis and generalized muscle atrophy. There was no evidence of cranial nerve, central nervous system, radicular, bone marrow, splenic, or lymph node involvement. The cat tested negative for feline retroviruses and a wide variety of herpes viruses, including Epstein-Barr virus. The clinical manifestation of this case was similar to the chronic polyneuropathic variant of human diffuse neurolymphomatosis; a condition most commonly caused by an axonopathy resulting from infiltration of peripheral nerves with non-Hodgkin's lymphoma.
Neoplasms of the feline peripheral nervous system are uncommon, and malignant lymphomas resulting in peripheral nervous system complications are rare in both humans and cats. 1,3–5 Although malignant lymphoma is the most common tumor reported to secondarily invade the peripheral nerves of the cat, previous studies of cats with lymphomatous peripheral neuropathy primarily describe clinical manifestations of mononeuropathy, 4,13 brachial plexopathy, 3,12 or multiple cranial neuropathies. 1 Lymphomatous infiltration of the brachial plexus of the cat has been reported to occur as an isolated event and in regional association with malignant lymphoma of the spine. 3,12
Non-Hodgkin's lymphomas (NHL) are capable of causing a variety of peripheral neuropathic syndromes in humans. In people, peripheral neuropathies associated with NHL can be mediated through a variety of both direct and indirect mechanisms. The most common direct manifestation of lymphomatous neuropathy results from axonopathy caused by neoplastic infiltration of nerves. 6 Less frequently, a clinical neuropathy can be caused by the invasion of nontumorous lymphocytes into nerves, most often in conjunction with a systemic lymphoproliferative disorder. The widespread infiltration of malignant lymphoid cells into peripheral nerves causing a clinical neuropathy is classically defined as diffuse neurolymphomatosis (NL). 6,8 This report documents the clinical, histopathologic, and immunohistochemical features of a case of Burkitt-type B-cell lymphoma with generalized peripheral nervous system infiltration in a cat, and describes its similarities to diffuse NL in humans.
A 4-year-old, spayed female domestic shorthair cat presented for a 4-month history of weight loss, muscle wasting, and weakness. Initial abnormalities noted during clinical examination included proprioceptive ataxia of all limbs; flaccid, ambulatory tetraparesis with absent segmental spinal reflexes in all limbs; and diffuse appendicular skeletal muscle atrophy. Additional diagnostic evaluations and empirical treatments were declined, and the cat was discharged. Two months later the cat was evaluated for continual progression of signs. Clinical examination findings included flaccid tetraplegia, with the remaining abnormalities similar to those noted at the previous admission. Antemortem clinicopathologic evaluations were limited to the performance of a serum creatine kinase concentration and serologic testing for feline leukemia (FeLV) and feline immunodeficiency (FIV) viruses. The serum creatine kinase concentration was high (5,201 U/liter; reference range 111 to 1,213 U/liter), and an electromyographic evaluation revealed diffuse spontaneous activity in the appendicular muscles of both the right thoracic and pelvic limb. Serologic tests for FeLV and FIV were negative. The cat was euthanatized.
At necropsy, the most significant gross lesion was moderate generalized appendicular muscle atrophy. The kidneys had irregular capsular surfaces with extensive cortical loss and indistinct corticomedullary junctions. All other organs appeared grossly normal.
Tissues were harvested and fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, cut into 5-μm sections, and stained with hematoxylin and eosin (HE) for histopathologic examination. Representative sections of peripheral nerves were fixed in 10% neutral buffered formalin and routinely processed for immunohistochemistry. For immunohistochemical examination, sections of neoplastic tissue were deparaffinized and rehydrated by routine methods. 7 Antigen retrieval was accomplished by incubation of slides in antigen retrieval solution (Dako, Carpinteria, CA) in a steamer (Black & Decker, Towson, MD) for 20 minutes. Endogenous peroxidase was blocked for 15 minutes with 3% hydrogen peroxide. Nonspecific immunoglobulin binding was blocked by incubation of slides for 10 minutes with a protein-blocking agent (Dako) prior to application of the primary antibody. The latter was allowed to react for 30 minutes at room temperature. Sections were stained in a Dako autostainer (Dako) apparatus. The slides were incubated with a mouse monoclonal anti-CD79a antibody (B-cells, Dako, dilution of 1 : 100), and a rat monoclonal anti-CD3 antibody (T-cells, courtesy of Dr. Peter Moore, University of California, Davis, CA, dilution 1 : 10). A labeled streptavidin-biotin-immunoperoxidase staining procedure (Dako) was used for immunolabeling. The immunoreaction was visualized with 3,3′-diaminobenzidine substrate (Dako). Sections were counterstained with Mayer's hematoxylin. Positive immunohistochemical controls included a normal feline lymph node to which the appropriate antisera were added. For negative controls, the primary antibodies were replaced with homologous nonimmune sera.
Histologically, sections obtained from the triceps, suprascapularis, cranial tibial, and gastrocnemius muscles revealed similar, but variably severe, neuropathic changes consisting of angular myofiber atrophy and small grouped myofiber atrophy. In several instances, intramuscular nerve branches visible within muscle specimens were infiltrated or partially obliterated by a homogenous population of neoplastic lymphocytes (Fig. 1). Neoplastic lymphocytes were uniform with round to oval, intermediate-sized vesicular nuclei with 2 to 5 small nucleoli, thickening of nuclear membranes and parachromatin clearing and small amounts of cytoplasm. Neoplastic cells had a high mitotic index and apoptotic cells were common. 4
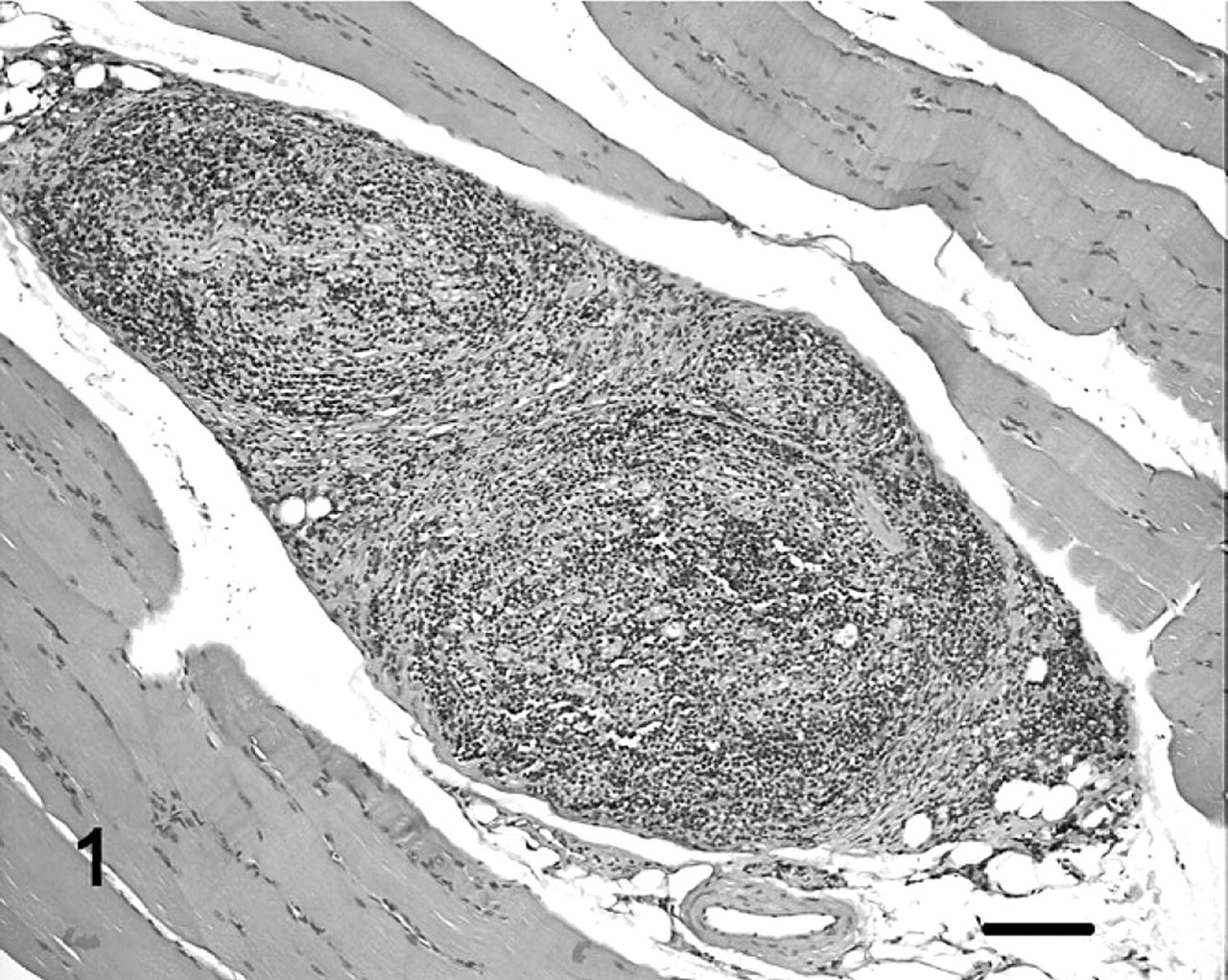
Skeletal muscle; cat. Intramuscular nerve fibers are infiltrated with neoplastic lymphocytes. HE. Bar = 250 μm.
The architecture of the sciatic nerve and multiple nerves of the brachial plexus were nearly completed obliterated by similar appearing neoplastic lymphocytes (Fig. 2a and 2b). Within the kidneys, there was diffuse, severe cortical loss with tubular atrophy and glomerulosclerosis. There was moderate interstitial nephritis characterized by multifocal lymphocytic aggregates within the cortex. No other significant microscopic findings were noted in other organs.
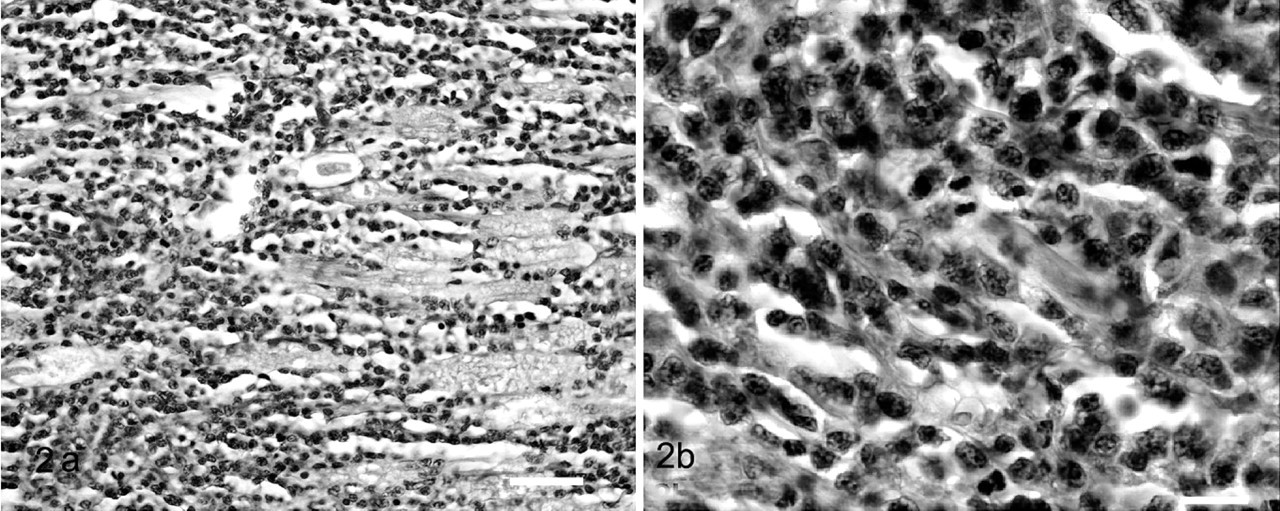
Sciatic nerve; cat.
Immunohistochemical evaluation revealed homogeneous positive staining of the lymphocytic infiltration within peripheral nerves with CD79a (Fig. 3). The vast majority of lymphoid cells in the kidneys stained positive with CD3 and a few cells were CD79a positive, a staining pattern consistent with the morphologic appearance of an interstitial lymphocytic nephritis. Immunohistochemistry for feline leukemia virus was negative. Based on the collective histopathologic and immunohistochemical results, a diagnosis of a B-cell, Burkitt-type lymphoma was made according to the World Health Organization International Histological Classification of Tumors of Domestic Animals. 15 However, the neoplasm described here is unusual in that it occurred without demonstrable lymph node, bone marrow, or central nervous system (CNS) involvement.
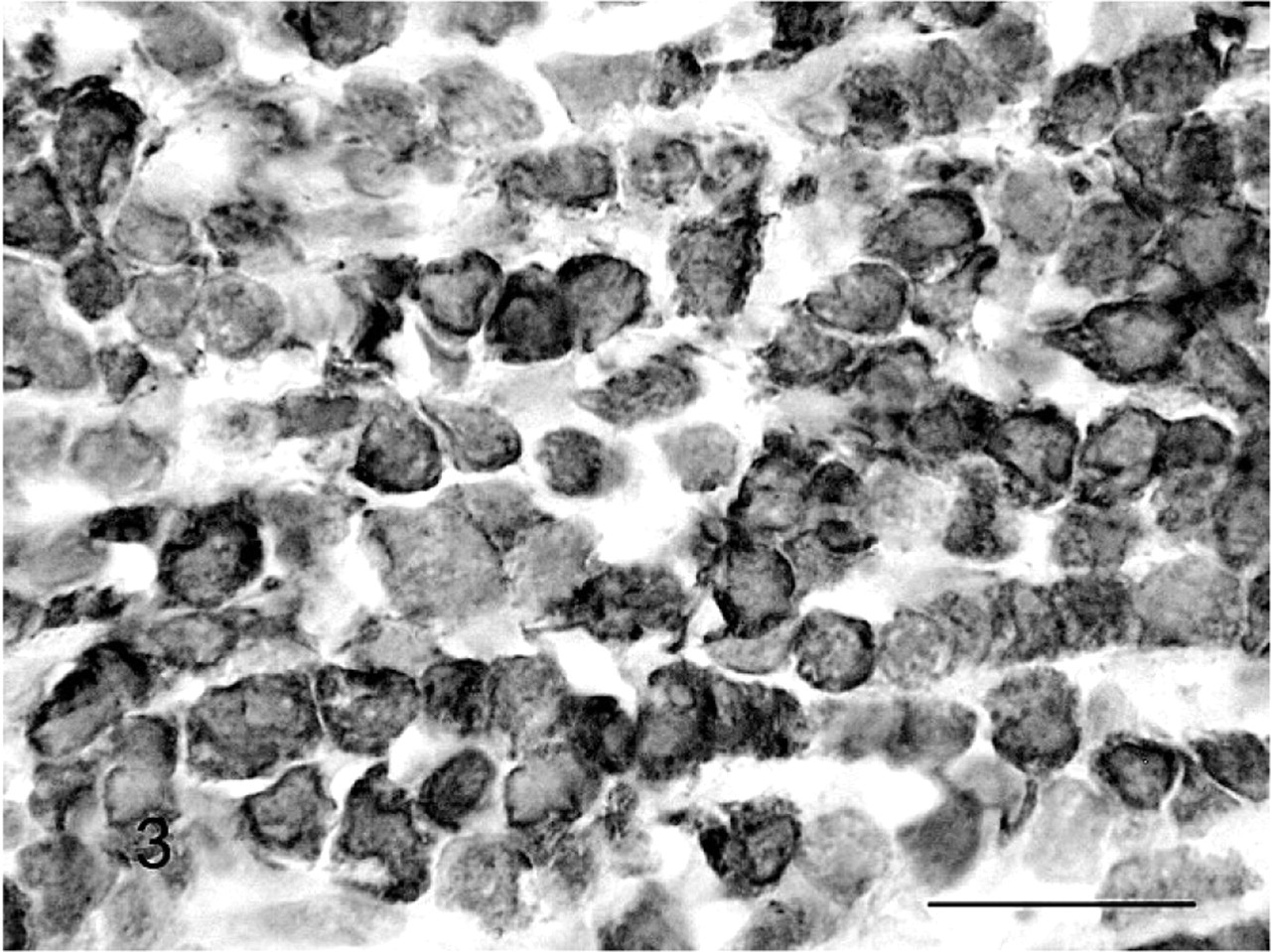
Peripheral nerve; cat. Neoplastic lymphocytes are positive for CD79a. Streptavidin immunoperoxidase. Mayer's hematoxylin counterstain. Bar = 10 μm.
A set of degenerate polymerase chain reaction (PCR) primers that amplify 21 species of herpesviruses (8 human and 13 animal viruses) including Epstein-Barr virus was used to detect the presence of herpesviral nucleic acid in sections of affected peripheral nerve. 16 Twenty five microliters of PCR mixture contained 5.0 μl (1–750 ng) of template DNA, 1 μM of each primer (5′-TGTAACTCGGTGTAYGGNTTYACNGGNGT-3′ and 5′-CACAGAGTCCGTRTCN CCRTADAT-3′), 80 μM (each) of deoxynucleoside triphosphate, 0.5 U of Taq polymerase (Invitrogen, Carlsbad, CA), 2.5 μl of 10× PCR buffer (200 mM Tris HCl [pH 8.4]) and 500 mM KCl (Invitrogen), and 2 mM MgCl2. PCR reaction was performed under mineral oil and cycled 45 times with 30 seconds of denaturation at 94°C, 1 minute of annealing at 46°C, and 1 minute of extension at 72°C. Negative controls were performed every 5 samples. Viral isolates were used as positive controls. PCR products were analyzed on a 2% agarose gel stained with ethidium bromide and detected under UV light. There was no evidence of Epstein-Barr virus or herpes viral DNA within sections of affected tissue.
Based on the clinical, gross, microscopic, and immunohistochemical findings, a diagnosis of a primary peripheral nervous system B-cell, Burkitt-type lymphoma was made in this cat. Although peripheral nervous system and CNS metastases, especially to the spinal cord, may occur in up to 50% of cats with malignant lymphoma, the case presented here differs in several ways from most previous reports of malignant lymphomas affecting the nervous system. 9,11 CNS metastases of extranodal malignant lymphoma originate most commonly from the kidneys and are usually associated with an infection with FeLV. 11 In this cat, there was no evidence of CNS lymphoma, and polyneuropathy was the only observed clinical manifestation of disease. In addition, the neoplasm was not associated with an identifiable, concurrent, oncoviral infection.
Historically, lymphomas affecting the central and peripheral nervous systems in several species, including humans, cats, and chickens were often associated with neurotrophic and oncoviral infections. 6,8–10 The most widely recognized peripheral nerve lymphoma in animals is Marek's disease, which is an endemic herpes-associated T-cell lymphoma of chickens. In addition, lymphomas affecting the nervous system of cats have historically been caused by retrovirus-mediated malignant transformation of T cells, and affected cats tested serologically positive for FeLV antigen. 2,4,9
The cat of this report was also evaluated for concurrent Epstein-Barr and other herpes viral infections because of their epidemiologic association with Burkitt-type lymphomas and other malignancies in humans. Epstein-Barr virus is a ubiquitous, orally transmitted, human herpesvirus that results in infection, which persists throughout the lifetime of the host. 14
Neurolymphomatosis is a heterogeneous clinical entity capable of causing a variety of clinical neurologic disturbances in humans, including mononeuropathy, cauda equina syndrome, plexopathy, asymmetrical regional neuropathies, polyradiculopathies, or a diffuse, subacute, or chronic progressive peripheral neuropathy. 6,8 The clinical and pathologic manifestations of disease displayed by the cat reported here most closely resemble the human, chronic and progressive polyneuropathic form of diffuse NL. 6
By definition, NL is characterized by lymphomatous infiltration of nerves, and definitive diagnosis requires histologic confirmation of infiltrative lymphomatous neuropathy by antemortem biopsy or autopsy. 6 Modern neuroimaging techniques have become useful in refining the need for pathologic confirmation of NL in people with previously diagnosed lymphoma; as in these cases, NL can often be tentatively diagnosed following identification of thickened or contrast-enhancing peripheral nerves or nerve roots on computed tomographic or magnetic resonance imaging studies. 6 However, a dilemma frequently encountered in humans, and further illustrated by the case presented here, is that NL may be the initial or only overt clinical manifestation of systemic lymphoma. 6 Thus, lymphoma should be considered as a differential diagnosis in cats with clinical signs of polyneuropathic disease. 5 This report also highlights that feline diffuse NL, although rare, may be one of the few conditions in which a definitive etiology for the polyneuropathic signs may be obtained from antemortem examination of fascicular nerve biopsy specimens, or skeletal muscle biopsies containing intramuscular nerve branches.
This case also demonstrates similarity to human NL in that it was caused by a B-cell NHL. The majority of cases of NL in people are associated with B-cell NHL, with only approximately 10% of human NL cases occurring with T-cell NHL. 4,6 The syndrome has rarely been documented with Hodgkin's lymphoma. 4 The clinical neurologic dysfunction that is seen in people with NL caused by B-cell NHL most commonly result from axonopathy caused by lymphomatous infiltration of nerves, which was the proposed primary mechanism of disease seen in the cat reported here. 4,6
Although an isolated case, based on the findings reported here, it appears that feline diffuse B-cell NL is clinically and histomorphologically similar to its human counterpart and should be considered a differential diagnosis for cats with clinical signs consistent with chronic, progressive polyneuropathy.
