Abstract
An 11-year-old Shetland Sheepdog was presented for exophthalmos caused by a locally extensive, poorly defined mass located behind the right eye. The primary orbital mass was identified by light microscopy and immunohistochemistry as a T-cell rich B-cell lymphoma (TCRBCL) composed predominantly of BLA.36- positive large neoplastic lymphoid cells admixed with fewer CD3- and CD79a-positive small lymphocytes. The dog was treated for lymphoma, but 6 months after presentation it was euthanatized for suspected hepatic and gastrointestinal metastasis. Gross findings revealed an enlarged liver with multiple well-demarcated, randomly distributed 0.1—1.5-cm white nodules, five firm white submucosal jejunal nodules, and ileocecal, mediastinal, and hilar lymphadenopathy. Metastatic liver lesions consisted of sheets of monomorphic large neoplastic lymphoid cells that effaced and expanded portal and centrilobular zones. These cells were morphologically similar to the large neoplastic cells of the original orbital tumor and were CD3-negative and variably BLA.36-positive, consistent with B-cell lineage. Similar cells comprised the jejunal nodules and effaced the lymph nodes. The progression of TCRBCL to a diffuse B-cell lymphoma in this case is consistent with reported human cases and has not been previously reported in the dog.
T-cell rich B-cell lymphoma (TCRBCL) is a histologic variant of the diffuse B-cell lymphoma group. The diffuse mixed cell lymphoma, a NCI Working Formulation Classification, has been reported in animals and could have represented TCRBCL previous to the common usage of immunohistochemistry for phenotyping. 20 TCRBCL has been reported only recently in the veterinary literature. 5 9 12 18 19 By definition, this class of tumor is characterized by a mixed cell population with large neoplastic B cells scattered within numerous small nonneoplastic T and B cells, which compose the background population of the mass. 16 The neoplastic B lymphocytes typically comprise <20% of the total population. Diagnosis of TCRBCL requires both morphologic and immunohistochemical examination of affected tissues.
In veterinary medicine, a limited number of cases of TCRBCL have been reported. Reported cases include a pig with multicentric lymphadenopathy, 19 cats with a single submandibular or cervical mass, 5 18 and horses with subcutaneous, pulmonary, lymph node, splenic, and/or hepatic masses. 9 12 Immunophenotypically, all of these tumors were characterized by a lymphoblastic population of neoplastic B lymphocytes within a background of small nonneoplastic T lymphocytes. In this report, we present a case of TCRBCL and its progression to a diffuse B-cell lymphoma in a dog.
An 8.6-kg, 11-year-old spayed female Shetland Sheepdog was presented to the University of Illinois Veterinary Medical Teaching Hospital (UIVMTH) for evaluation of exophthalmos of the right eye of 1-week duration. The dog was bright and alert with profound exophthalmos of the right eye and prolapse of the nictitans. The right eye was blind, deviated dorsolaterally, and did not retropulse normally into the orbit. In addition, the dog had a mildly enlarged right submandibular lymph node.
Initial diagnostic workup included a complete blood count (CBC), serum biochemical analysis, urinalysis, thoracic radiographs, and magnetic resonance imaging (MRI) with subsequent biopsies of the retrobulbar mass. Fine needle aspirate of the enlarged submandibular lymph node was obtained and consisted of nearly equal numbers of small and large lymphocytes, lesser numbers of plasma cells, and occasional macrophages. The cytologic impression was reactive lymphoid hyperplasia. Thoracic radiographs did not demonstrate any evidence of metastatic lung disease. Skull and right orbital MRI identified a 4–5-cm diffuse retrobulbar mass extending into the pterygoid muscles. The mass predominantly involved the medial muscle belly, with extension laterally and caudally to the region of the right ear. Multiple MRI guided tru-cut biopsies were obtained and fixed in 10% buffered formalin. Abdominal ultrasound did not demonstrate any evidence of abdominal metastasis. Results of CBC, serum biochemical analysis, and urinalysis revealed only a mature leukocytosis (18,900; reference values, 6,000–17,000 cells/μl).
The biopsies of the orbital mass were processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin (HE). The orbital mass contained a heterogeneous population of cells (Fig. 1). The primary cell population consisted of large cells (approximately 20 μm in diameter) characterized by a polygonal to spindyloid shape and a high nucleus: to cytoplasm ratio. These large cells were scattered throughout the mass but typically comprised 5–25% of the total cells. They had moderate amounts of lightly eosinophilic cytoplasm and they had pleomorphic nuclei (10–12 μm in diameter) with vesicular chromatin and one or two large nucleoli per nucleus. The nucleoli were variable in size and shape. Only these large neoplastic cells had mitotic figures (1–3 figures/400× field). The background cells were predominantly small nonneoplastic lymphocytes. Lesser numbers of eosinophils, neutrophils, and mast cells were also present. Sheets of both neoplastic and nonneoplastic cells were seen infiltrating collagen, muscle fibers, and lacrimal tissue. The preliminary morphologic diagnosis was a poorly differentiated round-cell tumor with a pleocellular inflammatory component.
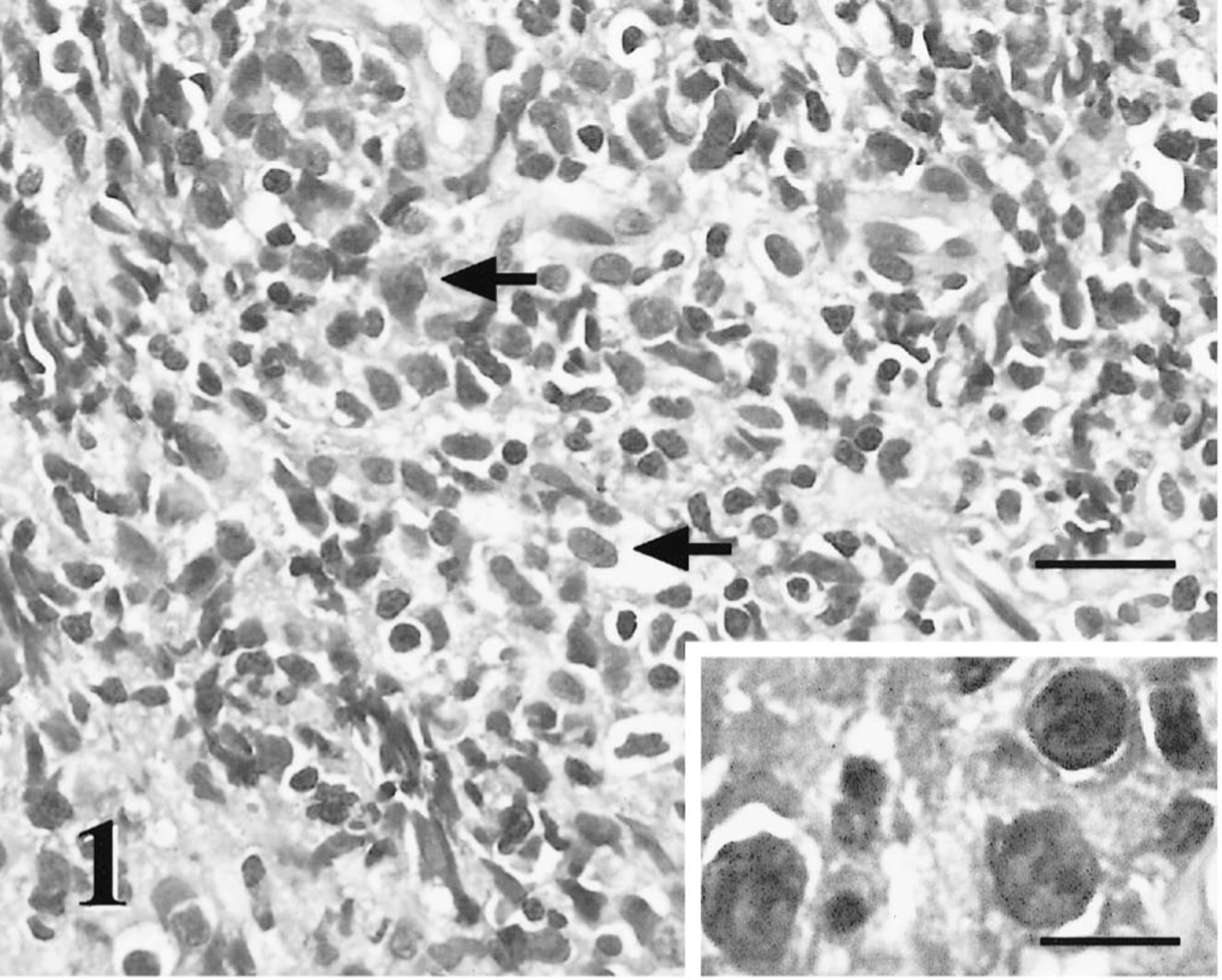
Orbital mass; dog. The orbital tissue is effaced by an infiltrative heterogeneous population of cells composed of large neoplastic lymphoid cells (arrows) within a predominant background of nonneoplastic small lymphocytes, neutrophils, and plasma cells. HE. Bar = 50 μm. Inset: The neoplastic cells have moderate amounts of cytoplasm, pleomorphic large round nuclei, and one to three large nucleoli. Bar = 15 μm.
Immunohistochemically, the neoplastic cells were immunophenotyped utilizing a basic indirect immunoperoxidase protocol. The protocol consisted of deparaffinization and rehydration of the sections through graded alcohol baths, endogenous peroxidase quenching with a methanol–hydrogen peroxidase solution, microwave antigen retrieval with citrate buffer (pH 6.0), a protein block, and incubation in the primary antibody. The sections were then incubated with a biotinylated secondary antibody, and this complex was labeled with streptavidin–horseradish peroxidase conjugate and identified with diaminobenzidine followed by Mayer's hematoxylin counterstain. The primary antibodies used were monoclonal antibodies anti-CD79a (clone HM57; Dako, Carpinteria, CA) and BLA.36 (BioGenex, San Ramon, CA) and a polyclonal anti-CD3 antibody (Dako). The larger neoplastic cells were CD3 and CD79a negative, cytokeratin negative, vimentin positive, s-100 positive, and neuron-specific enolase positive. Scattered neoplastic cells (5%) were BLA.36 positive (Figs. 2, 3). This staining pattern was most consistent with a B-lymphocyte phenotype. Most of the small nonneoplastic lymphocytes were cytokeratin negative, vimentin positive, and CD3 positive. This phenotype was most consistent with well-differentiated T lymphocytes. Small numbers of CD79a positive small nonneoplastic B lymphocytes were also present in the mass. The mixed population of cells and equivocal immunostaining of the large neoplastic cells made definitive diagnosis difficult. The microscopic diagnostic impression was that of a poorly differentiated round-cell tumor, most likely a mixed lymphoma.
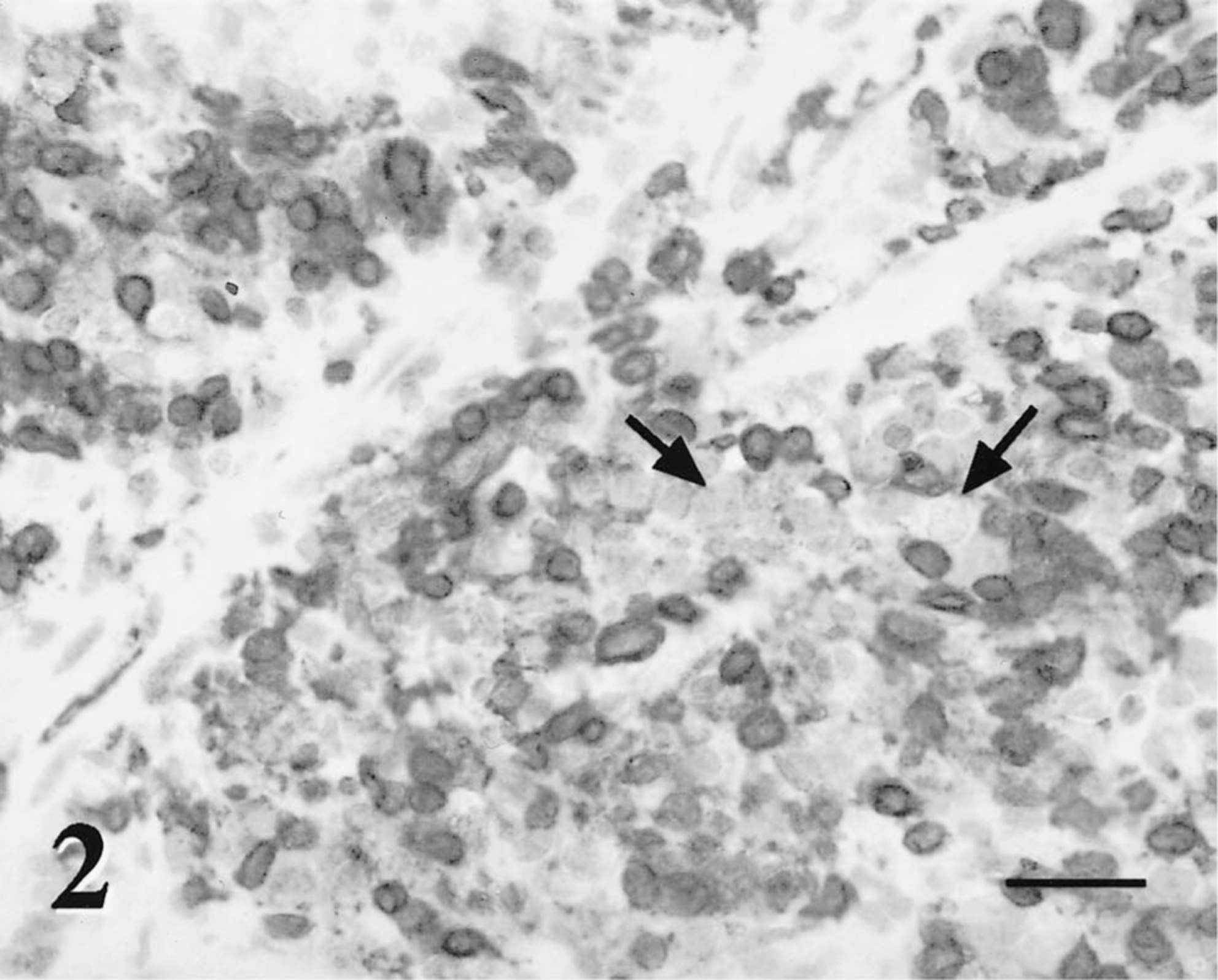
Orbital mass; dog. The large neoplastic lymphoid cells are negative for CD3 cell membrane antigen (arrows). The background small lymphocytes stain strongly positive for CD3 antigen. Avidin-biotin-peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 50 μm.
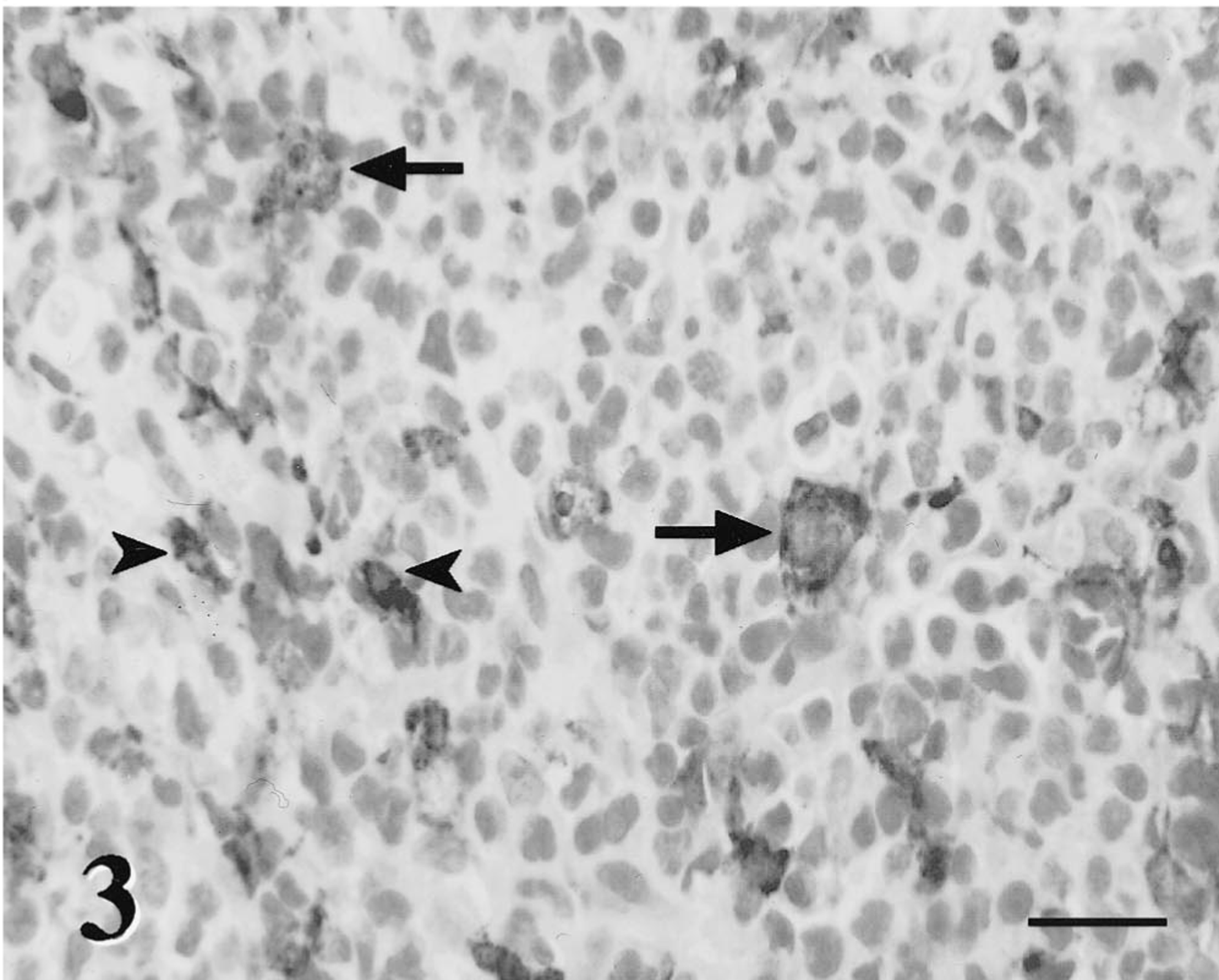
Orbital mass; dog. Scattered large neoplastic cells stain positive with BLA.36, a B-lymphocyte marker (arrow). Within the background, small plasma cells similarly stain positive with the B-cell marker BLA.36 (arrowheads). Avidin-biotin-peroxidase complex method, Mayers hematoxylin counterstain. Bar = 50 μm.
The dog received chemotherapy, and the exophthalmos was markedly improved 1 week later. Nineteen weeks after diagnosis, a CBC revealed a mild normocytic, normochromic anemia (hematocrit = 31.6%; reference values, 35–52%). Twenty-two weeks after diagnosis, the normocytic, normochromic anemia had worsened (hematocrit = 27.6%). Differential diagnosis for the anemia included anemia of chronic disease, chronic bleeding, or bone marrow suppression by chemotherapeutic agents, although no evidence of bone marrow suppression was found in other hematopoietic cell lines.
Approximately 6 months after initiation of chemotherapy, the dog began to exhibit vomition and diarrhea. The referring veterinarian palpated a left cranial abdominal mass. On exam at UIVMTH, the dog appeared slightly obtunded. Serum alkaline phosphatase level was mildly elevated (156; reference values, 12–110 U/liter). CBC showed progressive normocytic, normochromic anemia with a hematocrit of 24.4% and a leukocytosis with a neutrophilia, left shift, and a mild toxic changes to the neutrophils. Abdominal ultrasonic examination revealed a hypoechoic liver, multifocal small intestinal thickening, and mesenteric lymphadenopathy. Fine needle aspirates of the intestinal masses were obtained. Two separate sites had a similar monomorphic population of large immature lymphocytes with scattered red blood cells. The cytologic interpretation was lymphoma.
The dog was euthanatized due to progressive metastatic disease and submitted for necropsy 24 hours later in fair postmortem condition. Mucous membranes were pale. The liver was moderately enlarged with pale, friable parenchyma. The liver had numerous white 0.1–1.5-cm-diameter well-demarcated firm nodular masses randomly distributed throughout the liver, accounting for 40% of the hepatic parenchyma. The second quarter of the jejunum contained five large, irregularly shaped firm white, 2–4.5-cm-diameter nodules that extended from the submucosa. The associated mucosa and serosa were intact. The ileocecal, mediastinal, and hilar lymph nodes were moderately enlarged, firm and white, and nodal architecture was effaced. No gross or histologic evidence of orbital neoplasia was present in or around the right eye. There were no significant lesions in any other organs. Gross necropsy findings were consistent with metastatic neoplasia of the gastrointestinal tract, liver, and lymph nodes. Tissues collected at necropsy were processed as previously described. Histologically, the bone marrow consisted of 70% hematopoietic cells (all lineages present) and 30% adipocytes. The liver lesions were characterized by sheets of monomorphic large round cells centered on portal and centrilobular zones with locally extensive effacement of the hepatic parenchyma. The portal and centrilobular distribution suggested hematogenous metastasis (Fig. 4). These cells were morphologically similar to the large neoplastic lymphoid cells of the original orbital tumor but with no admixed populations of small nonneoplastic lymphocytes, eosinophils, neutrophils, or mast cells. The large neoplastic cells (approximately 20 μm in diameter) were polygonal to round with modest amounts of pale eosinophilic cytoplasm (high nucleus:to cytoplasm ratio). They had a single large round nucleus (10–12 μm in diameter) with open, coarsely clumped to vesicular chromatin and a thick nuclear membrane. One to three large nucleoli were centrally or peripherally located with impingement upon the nuclear membrane. Mitotic figures were common (2–5 figures/400× field). The large neoplastic cells were CD3 negative and variably BLA.36 and CD79a positive (20% of neoplastic population), suggestive of B-cell lineage (Figs. 5, 6). Similar cells were identified in the submucosal jejunal nodules and effacing the mesenteric lymph nodes.
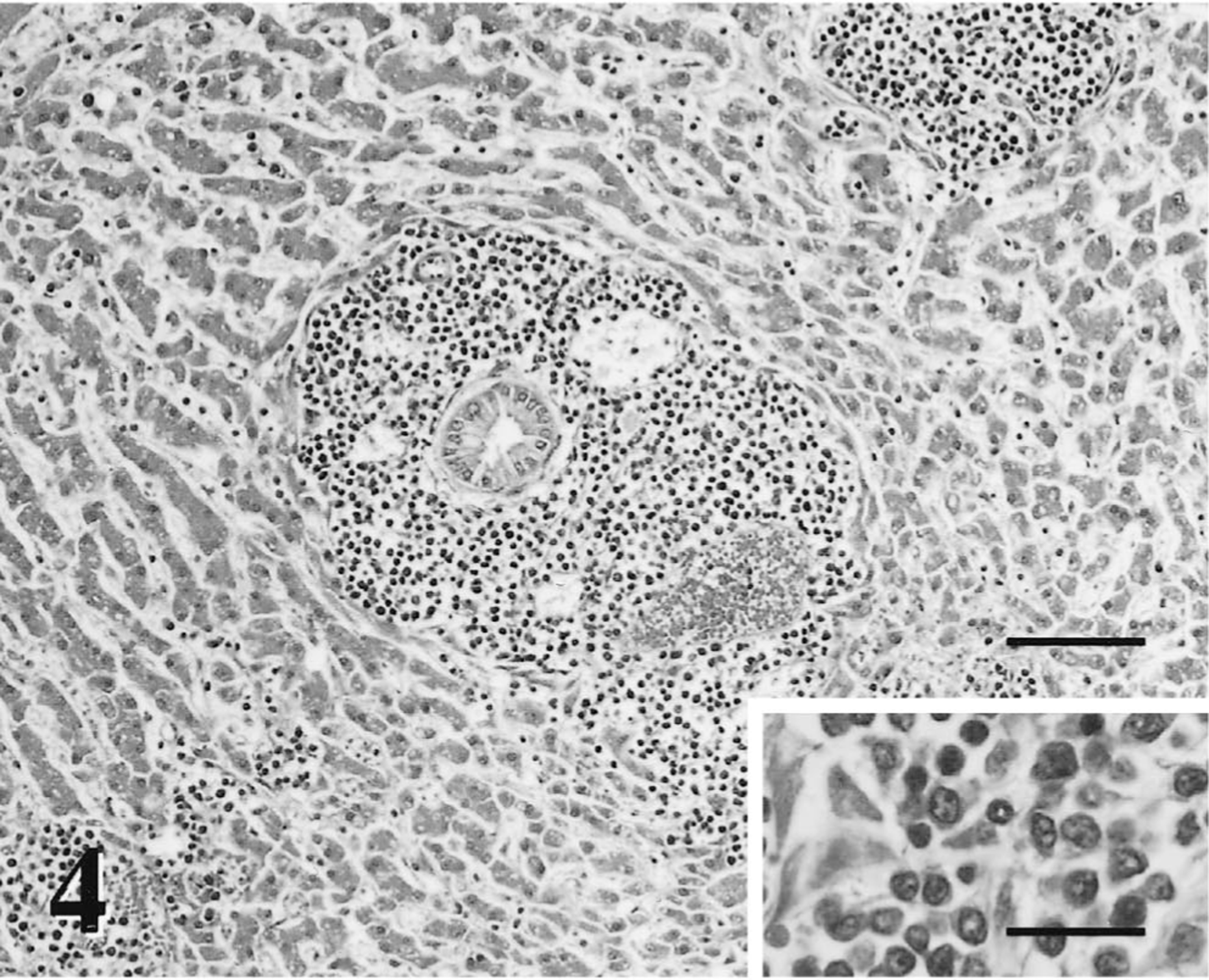
Liver; dog. Metastatic liver lesions are characterized by infiltrates of a homogenous population of large neoplastic lymphoid cells that expand the portal zones and locally efface adjacent hepatic parenchyma. HE. Bar = 150 μm. Inset: Cytomorphologically, the cells have moderate amounts of cytoplasm, pleomorphic large round nuclei, and one to three large nucleoli that differ in size and shape. Bar = 50 μm.
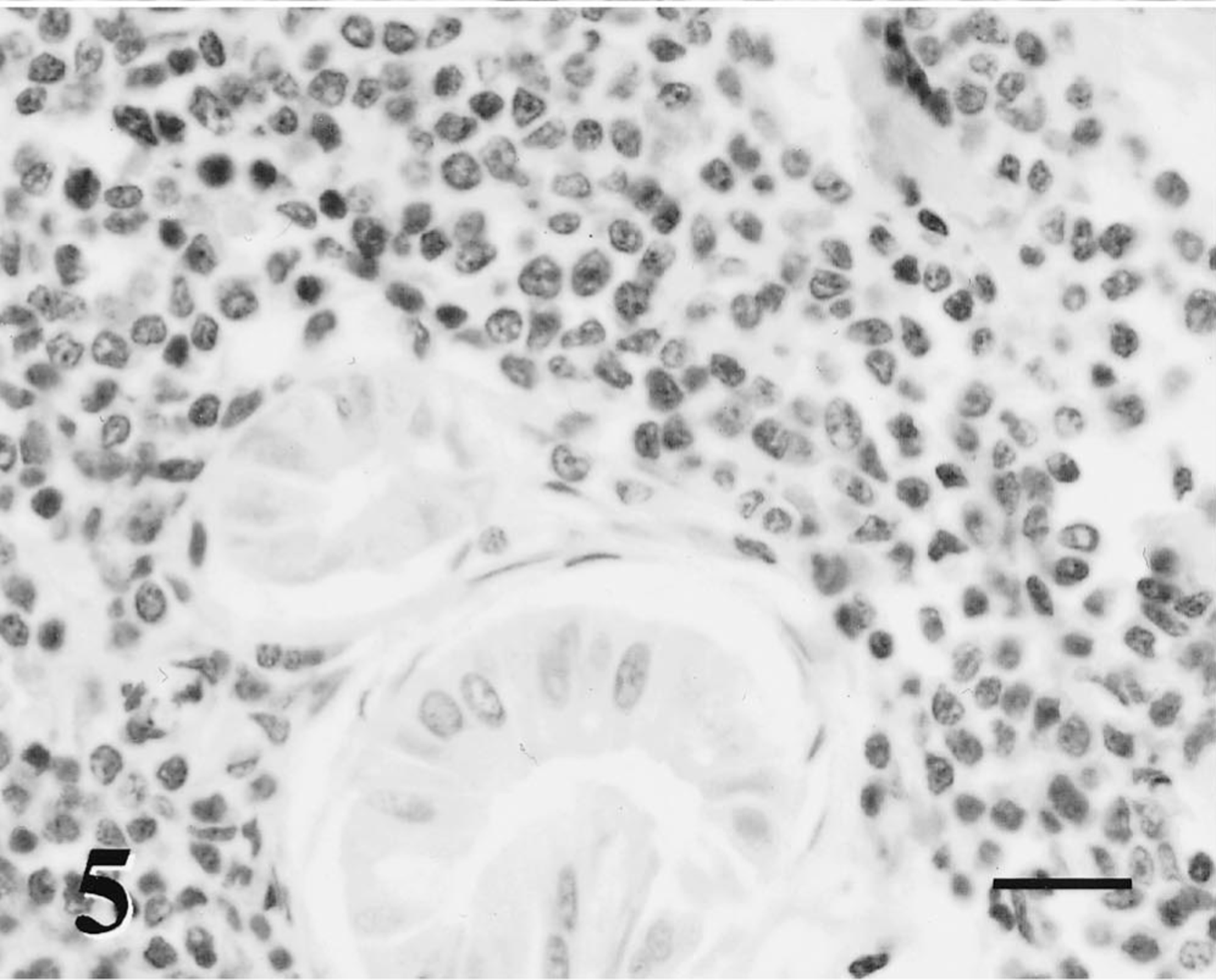
Liver; dog. Large neoplastic lymphoid cells that infiltrate periportal zones of the liver are diffusely negative for CD3 antigen. Avidin-biotin-peroxidase complex, Mayer's hematoxylin counterstain. Bar = 50 μm.
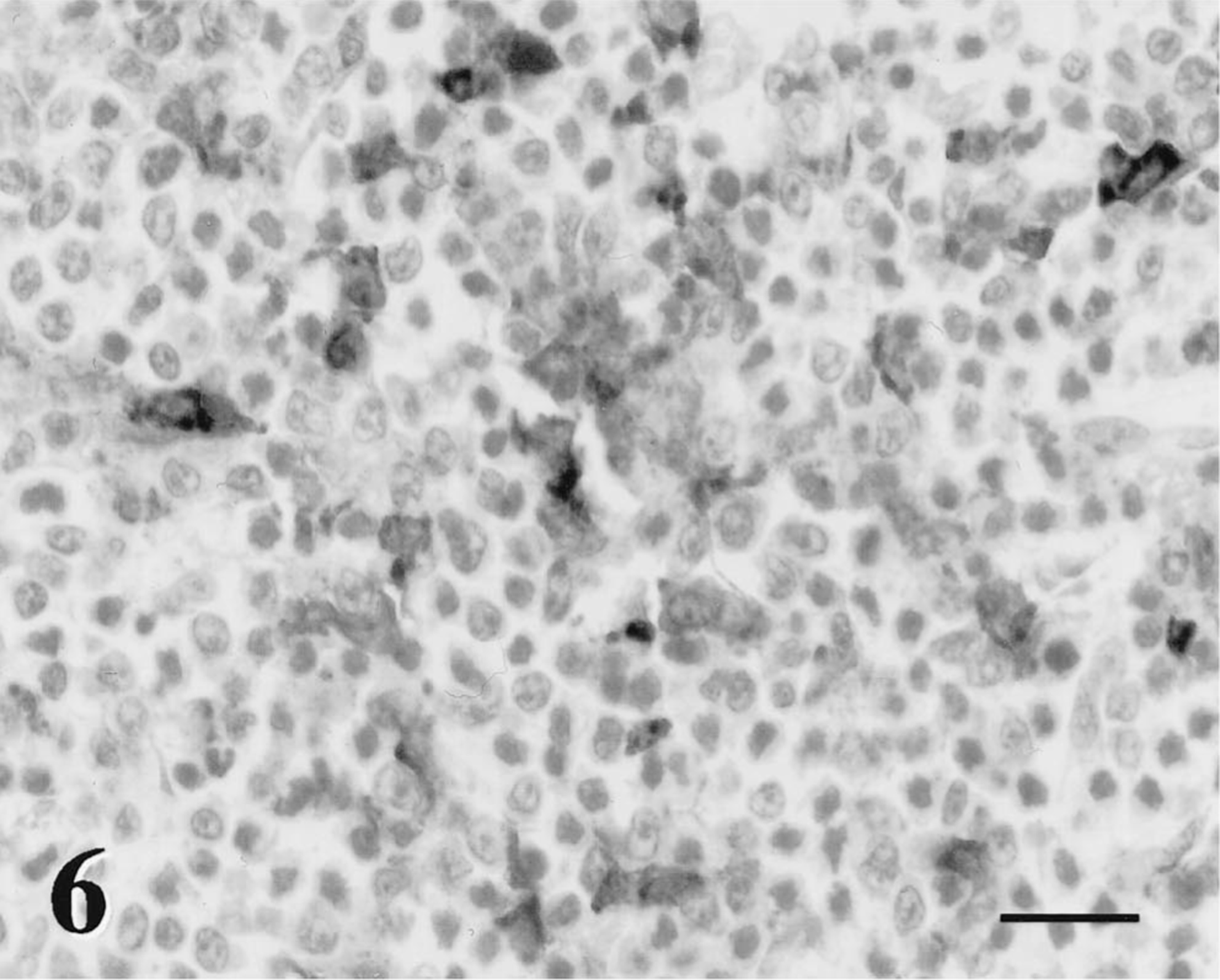
Liver; dog. Large neoplastic lymphoid cells infiltrate and efface the periportal zones and stain variably positive (20% of the neoplastic population) with BLA.36. All cells, both stained and unstained, have similar cytomorphologic characteristics. Avidin-biotin-peroxidase complex, Mayer's hematoxylin counterstain. Bar = 50 μm.
The gross and microscopic lesions in this case support a final diagnosis of primary orbital TCRBCL with metastasis to the liver, mesenteric lymph nodes, and jejunum. This case is unusual both for the primary orbital presentation of lymphoma and the diagnosis of TCRBCL with progression to a diffuse B-cell lymphoma after chemotherapy. Ocular involvement in cases of lymphoma is common in the dog. However, typical ocular manifestations are intraocular and usually in conjunction with other clinical signs. 8 14 The most frequent ocular finding associated with lymphoma is anterior uveitis. 8 14 In this case, the dog presented with classic signs of orbital neoplasia. This extranodal presentation of lymphoma is rare but has been documented previously in a dog. 15
The heterogeneous cell population of the original orbital biopsy sections made diagnosis difficult using morphologic criteria alone, which is typical of TCRBCL. By definition, TCRBCL, a variant of the diffuse large-cell lymphoma class, is characterized by large immunoblastic B cells occurring singly within a background of predominantly small, well-differentiated T lymphocytes. 2 3 6
TCRBCL was first suggested as a distinct entity in 1984 10 and later was described as a “diagnostic pitfall.” 16 The mixed nature of the cell population with a relative paucity of neoplastic B cells in a T-cell-rich background makes morphologic diagnosis difficult. The need to use both morphologic and immunophenotypic assessment for diagnosis has been emphasized in the human literature. Generally accepted morphologic diagnostic criteria include 1) diffuse tumor growth pattern, 2) large population of small nonneoplastic T lymphocytes, and 3) fewer numbers of neoplastic B lymphocytes. 1 7 13 16 Diagnostic confusion arises because TCRBCL shares morphologic characteristics with a number of other lymphocytic tumor types, including peripheral T-cell lymphoma, 13 16 lymphocytic predominant Hodgkin's disease, 13 and histiocyte-rich B-cell lymphoma. 7 With immunostaining, tumor cells of TCRBCL are typically CD20 positive, variably CD79a positive, and epithelial membrane antigen negative. The background population of small lymphocytes stain diffusely with CD3. 17
In veterinary medicine, a limited number of cases of TCRBCL have been reported to date, 5 9 12 18 including a pig with generalized lymphadenopathy. 19 Histologic examination of cells from this pig revealed a characteristic mixed population of lymphoid cells. The large neoplastic lymphoid cells had large centrally located nuclei with frequent mitotic figures (2–5 figures/400× field). The neoplastic lymphoid cells were CD79a positive, with 20–50% of these cells showing lambda type light chain restriction. The majority of the background cells were CD3 positive. 19
Similarly, TCRBCL has been identified in cats. 5 18 B-cell lymphoma was diagnosed in an older domestic shorthaired cat 18 after histologic examination of a mass in the parotid region revealed sheets of small round cells consistent with mature lymphocytes. Occasional large round to oval neoplastic cells were present in the background. Mitotic figures were seen in the population of large cells only. On immunostaining, these large cells were BLA.36 positive. Nearly all of the small lymphoid cells were CD3 positive, with only scattered small background lymphocytes being BLA.36 positive. These findings are consistent with TCRBCL in human beings, where neoplastic B cells exist in a mixed population of nonneoplastic small T and B lymphocytes. An additional eight cases of TCRBCL was reported in cats presented for masses in the cervical and submandibular regions. 5 Immunostaining results in these cats were similar to those previously reported for TCRBCL in the cat. 18
One case report of equine lymphoma and 11 cases in a series were identified as TCRBRL. 9 12 Nine of the 12 horses had subcutaneous lesions, which in four horses involved lung, lymph nodes, spleen, and/or liver. As in pigs and cats, the background cells were CD3 positive, and 10–60% of the cells were identified as large neoplastic B lymphocytes with BLA.36, mb-1 (leukocyte common antigen clone against CD45A), and B29 immunophenotype expression.
In the present case, initial biopsies of the retrobulbar mass had characteristics consistent with descriptions of TCRBCL in both human and veterinary literature. Histologic analysis of affected tissues revealed a diffuse predominant population of small nonneoplastic lymphoid cells with fewer large neoplastic lymphoid cells. These large neoplastic cells were morphologically suggestive of immunoblastic B lymphocytes. They were CD3 negative, and scattered cells (5%) were BLA.36 positive on immunostaining. All of the neoplastic cells had similar morphology, regardless of whether they were BLA.36 positive. The majority of the background population was composed of CD3-positive small T lymphocytes.
The rapid response to chemotherapy was consistent with the reported chemoresponsive nature of TCRBCL in humans 1 13 22 and of lymphoma in dogs. 4 11 21 Data regarding the biologic behavior, clinical response, and outcome of TCRBCL in veterinary medicine are limited at this time. TCRBCL has not been previously reported in dogs. Case reports in human literature have documented remission with early aggressive chemotherapy. In one case, 1 tumor morphology in recurrent TCRBCL changed from a mixed cell population to a diffuse B-cell population, and in one of the five cases of TCRBCL reviewed elsewhere, 16 transformation from a T-cell-rich pattern to a diffuse large B-cell neoplasia was revealed postmortem. In the dog of the present report, a similar change in tumor morphology was seen. Histologic examination of metastatic lesions observed at necropsy revealed sheets of monomorphic large neoplastic lymphoid cells that were morphologically similar to the neoplastic cells that comprised only a small fraction of the original orbital tumor. These cells stained CD3 negative and variably BLA.36 and CD79a positive (20% of neoplastic population), suggestive of B-cell lineage. This phenomenon of progression may be a reflection of altered host response or selection for resistant neoplastic B cells secondary to chemotherapy. 1 In this dog, the progression of TCRBCL to a more aggressive diffuse B-cell lymphoma is consistent with reported human cases and has not been previously reported in the dog.
