Abstract
An 8-y-old castrated male, outdoor European shorthair cat was presented with a history of hindlimb weakness and paralysis. Disease progression was continuous from the onset; deep algesia disappeared at the final stage. Radiography of the vertebral column was unremarkable; along with patient history and physical examination results, magnetic resonance imaging suggested inflammatory lesions in the spinal cord, although neoplasia could not be ruled out. Feline leukemia virus (FeLV) positivity was confirmed by a serum ELISA prior to euthanasia. Upon postmortem examination, hemorrhages were present in the spinal cord at the level of vertebrae T7-8. Histologic and immunohistochemical analysis revealed primary diffuse large B-cell lymphoma of the spinal cord with multifocal myelomalacia and hemorrhages. To determine the presence of a pathogen within the lesion, we developed a novel in situ hybridization protocol for FeLV (RNAscope). The reaction revealed large amounts of FeLV viral RNA in the tumor cells.
In cats, ~ 90% of hematopoietic tumors are lymphomas, arising from lymphoid tissues rather than from bone marrow. 16 Typically lymphomas develop in the lymph nodes (nodal forms), although sometimes they arise from other lymphoid centers of the body (extranodal forms). The latter category includes lymphomas of the central nervous system, eye, and respiratory and gastrointestinal tracts.
In cats, ~ 50% of all lymphomas are alimentary, arising from extranodal mucosa-associated lymphoid tissue (MALT lymphomas). Most of these alimentary lymphomas have a T-cell phenotype 1 and can be small-cell, large-cell, or granulated type.8,17 Feline leukemia virus (FeLV)-associated lymphomas (excluding thymic forms) typically have B-cell phenotype. Given the widespread use of vaccination against FeLV, alimentary B-cell lymphomas have become less common (~ 30% of all enteric lymphomas).11,16,19 Also in association with vaccination, the organ involvement of lymphomas in general has shifted from young cats with the mediastinal type to older cats with extranodal types.16,19
Although enteric lymphomas are commonly of T-cell origin in cats, almost 90% of all respiratory tract lymphomas tend to be B-cell type. More than half of these cases are also FeLV positive. 10 In the nervous system, most lymphomas are metastatic, but rarely, primary or multicentric lymphomas also occur. 12
FeLV (
The syndrome most frequently associated with FeLV-induced lymphoma is characterized by discrete tumors and/or diffuse infiltration of organs by neoplastic lymphoid cells.
16
Three forms of lymphoma have been described in FeLV-infected cats: 1)
An 8-y-old castrated male, outdoor European shorthair cat was presented to a veterinary clinic with a 3-wk history of hindlimb weakness and paralysis. Disease progression was continuous, and, at the final stage, deep pain sensation disappeared. Radiography of the vertebral column was within normal limits, and magnetic resonance imaging suggested myelitis, but neoplasia could not be ruled out. Prednisolone treatment for 1 wk did not improve the condition of the animal. The cat was FeLV seropositive (Witness FeLV-FIV test; Zoetis, Parsippany-Troy Hills, NJ) before euthanasia, which was performed with T-61 (MSD, Kenilworth, NJ) under general anesthesia.
Gross postmortem inspection revealed pallor, generalized peripheral lymphadenomegaly, mild splenomegaly, and a hemorrhagic lesion in the spinal cord at the level of vertebrae T7-8. Other organs were unremarkable.
A pool of organs (mesenteric lymph node, bone marrow, liver, spleen) was tested by PCR to confirm FeLV infection, 15 with the forward and reverse primers 5′-AACAGCAGAAGTTTCAAGGCC-3′ and 5′-TTATAGCAGAAAGCGGCG-3′, respectively. The primers amplified early-stage products (proviral DNA) of the reverse transcription. Sanger sequencing of the amplicons and subsequent analysis of the sequence data confirmed the specificity of the positive reaction.
Representative samples of spinal cord and all major organs (spleen, liver, kidney, bone marrow, stomach, small and large intestines, lung, heart, brain; submandibular, axillary, mesenteric, and popliteal lymph nodes) were fixed in 10% neutral-buffered formalin for 24 h at room temperature. Following routine processing for histopathology, additional sections were cut at 5 µm and mounted on SuperFrost Plus slides (Thermo Fisher Scientific, Waltham, MA) for immunohistochemistry (IHC) and in situ hybridization (ISH). Histologic evaluation of routine hematoxylin and eosin (H&E)-stained sections of the spinal cord revealed multifocal-to-coalescing proliferation of neoplastic, immature, round, immunoblastic lymphoid cells in the white and gray matter. There was moderate anisocytosis and anisokaryosis; mitotic count of 35–40 per 2.37 mm2 (ten 400× fields); concurrent, severe, multifocal myelomalacia (Fig. 1); and small hemorrhages in the adjacent areas. The immature lymphoid cells had nuclei that were twice the diameter of red blood cells, with some nuclei having shallow nuclear indentations. Some intermediate-sized lymphoid cells were also present with nuclei 1.5× the diameter of red blood cells. Single and prominent nucleoli were centrally located. These neoplastic cells had small-to-moderate amounts of basophilic cytoplasm. A few small, mature lymphocytes were also present (Fig. 1).
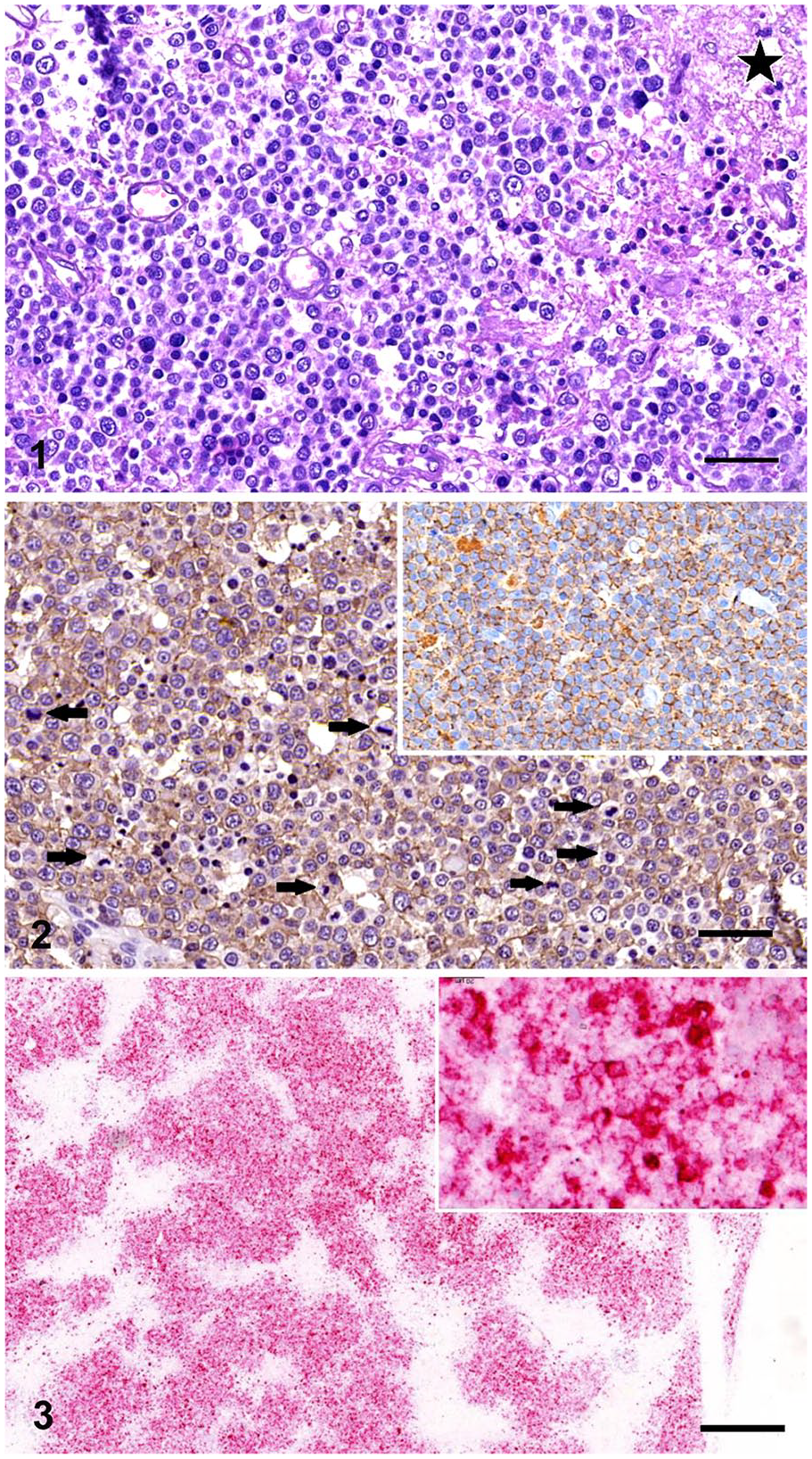
B-cell lymphoma in the spinal cord of a cat.
Based on histologic examination of the other tissue samples, we excluded extraneural involvement or involvement of other spinal regions. Incidental findings included mild congestion in the spleen, presumably as a result of euthanasia, and mild lymphoid reactivity in examined lymph nodes. We diagnosed primary extranodal nervous system lymphoma, involving the thoracic spinal cord.
To further define the immunophenotype of the neoplastic lymphoid cells, we performed IHC with polyclonal rabbit anti-human CD20 antibody (Thermo Fisher Scientific) and polyclonal rabbit anti-human CD3 antibody (Dako, Kyoto, Japan) markers, according to the manufacturers’ instructions. Positive and negative controls were run beside sections from the spinal cord. The immunostaining expression patterns proved that the tumor was of B-cell type, with intense CD20 positivity in the cell membrane and cytoplasm of neoplastic cells (Fig. 2). CD3-positive T-cells (presumably reactive lymphocytes) were present only in small numbers, hence we ruled out B-cell–rich T-cell lymphoma and T-cell–rich diffuse large B-cell lymphoma. A final diagnosis of high-grade, diffuse, predominantly immunoblastic, large B-cell lymphoma was made.
Given that the FeLV status of the cat was positive, a novel method of RNA-based ISH (RNAscope), based on the detection of messenger RNA molecules in tissue samples, 18 was used to detect actively replicating FeLV in tissue samples. The reaction steps of ISH (RNAscope 2.5 high definition [HD] red kit; Advanced Cell Diagnostics, Newark, CA) were performed according to the manufacturer’s recommendations. Briefly, sections were deparaffinized in xylene and rehydrated through a series of alcohols. Slides were incubated for 10 min in hydrogen peroxide at room temperature, exposed to retrieval buffer for 15 min at 95–102°C, and treated with protease at 40°C for 30 min. Hybridization with the target RNA-specific oligonucleotide probes for FeLV and controls was done for 2 h at 40°C. RNAscope probes have a Z shape, and they are designed such that 2 probes attach next to each other on the target RNA. In the next step, pre-amplifiers hybridize to the upper part of the Z probes, but only when 2 probes are hybridized next to each other. Amplifiers then attach to the multiple binding sites on the preamplifiers. Labeled probes containing a red chromogenic enzyme bind to multiple binding sites on the amplifiers. Gill II modified hematoxylin stain was used for counterstaining. Each single targeted RNA transcript molecule appeared as a distinct dot of red chromogen precipitate.
We used RNAscope Probe-V-FELV-env (catalog 491001) as the target probe, binding to region 41–1281 (
In the ISH-stained slides, all feline tissues showed a positive reaction for the PPIB reference gene and a negative reaction for the
Our research group successfully developed an RNA-based ISH assay (RNAscope) to detect FeLV RNA. The method was validated on numerous FeLV-positive and -negative control samples. Application of the RNAscope to our case of high-grade diffuse large B-cell lymphoma in the spinal cord indicates that the lymphoma was presumably induced by FeLV infection. Although FeLV RNAscope examination showed viral RNA-containing cells in the spleen and bone marrow as well, such large amounts of viral RNA in the tumor cells strongly suggests oncogenic activity of FeLV. The probe is available from Advanced Cell Diagnostics, and it can be used as a robust method to detect FeLV RNA in formalin-fixed, paraffin-embedded (FFPE) samples even in retrospective studies. Advantages of this ISH method are high specificity, sensitivity, and ease of use. One of the main differences, and a major advantage, of RNAscope compared to traditional ISH is that RNAscope does not require an RNA-free environment. RNAscope could also be used for diagnostic purposes, in which only FFPE tissues and no clinical samples are available; however, the price of the assay may be too high for routine use, especially when compared to PCR and ELISA. RNAscope has been described for detection of RNA in various fields of research,6,7,9,13 and our study shows its applicability in the case of an oncogenic retrovirus.
Footnotes
Acknowledgements
We thank Renáta Pop and Kitti Schönhardt for providing help in histology work.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grant 12190-4/2017/FEKUTSTRAT of the Hungarian Ministry of Human Capacities. Gy. Balka was supported by the János Bolyai Scholarship of the Hungarian Academy of Sciences.
