Abstract
Feline peripheral nerve sheath tumors are uncommonly reported, and their clinical behavior has not been well documented. Fifty-nine peripheral nerve sheath tumors were collected from 53 cats. All of the tumors involved skin, subcutis, skeletal muscle, and/or mucous membranes. Histologically, the tumors were composed of compact to loosely arranged streams and fascicles of spindled cells with eosinophilic, often wavy cytoplasmic processes; small to occasionally moderate amounts of collagenous to myxoid matrix; and nuclear palisading. Immunohistochemically, all tumors were positive for vimentin and S-100 protein, 44 of 59 were positive for glial fibrillary acidic protein (GFAP), and all were negative for muscle specific actin. The tumors fell into 3 histologic categories: 34 benign tumors with Antoni A areas that were S-100 protein and GFAP positive, 9 benign tumors that lacked Antoni A areas and were S-100 protein positive and GFAP negative, and 16 tumors with features of malignancy. Seventy-five percent of these cases involved the head, neck, or limbs. Recurrent tumors were submitted or tumors were reported to have recurred in 9 cases. Tumor recurrence was reported for all 3 of the histologic subtypes. None was documented as having metastasized.
Introduction
Peripheral nerve sheath tumors (PNSTs) are neoplasms that arise from Schwann cells, perineurial cells, and intraneural fibroblasts and can be subdivided based on morphologic features and benignancy versus malignancy into schwannoma (previously called neurilemoma or neurinoma), neurofibroma, and malignant PNST. Primarily because of uncertain histogenesis in veterinary medicine, PNSTs are simply divided into benign and malignant variants in the most recent edition of the WHO International Histological Classification of Tumors of Domestic Animals.8
The published information on PNSTs in cats is limited (Tables 1, 2), with only 10 peer-reviewed articles1, 5–7, 11, 12, 17, 19, 20, 22 spanning 31 years. Each describes either a single case or a series of 7 or fewer cases. Seven of these reports include information regarding biologic behavior,1, 5, 6, 11, 12, 19, 22 and only 5 include immunohistochemistry.5, 11, 17, 19, 20 Two veterinary textbooks2, 4 provide some summarized information regarding PNSTs in the collections of the books' authors, and a third textbook18 briefly describes a case of perispinal feline PNST.
Summary of peripheral nerve sheath tumors (PNSTs) in the peer-reviewed literature. ∗
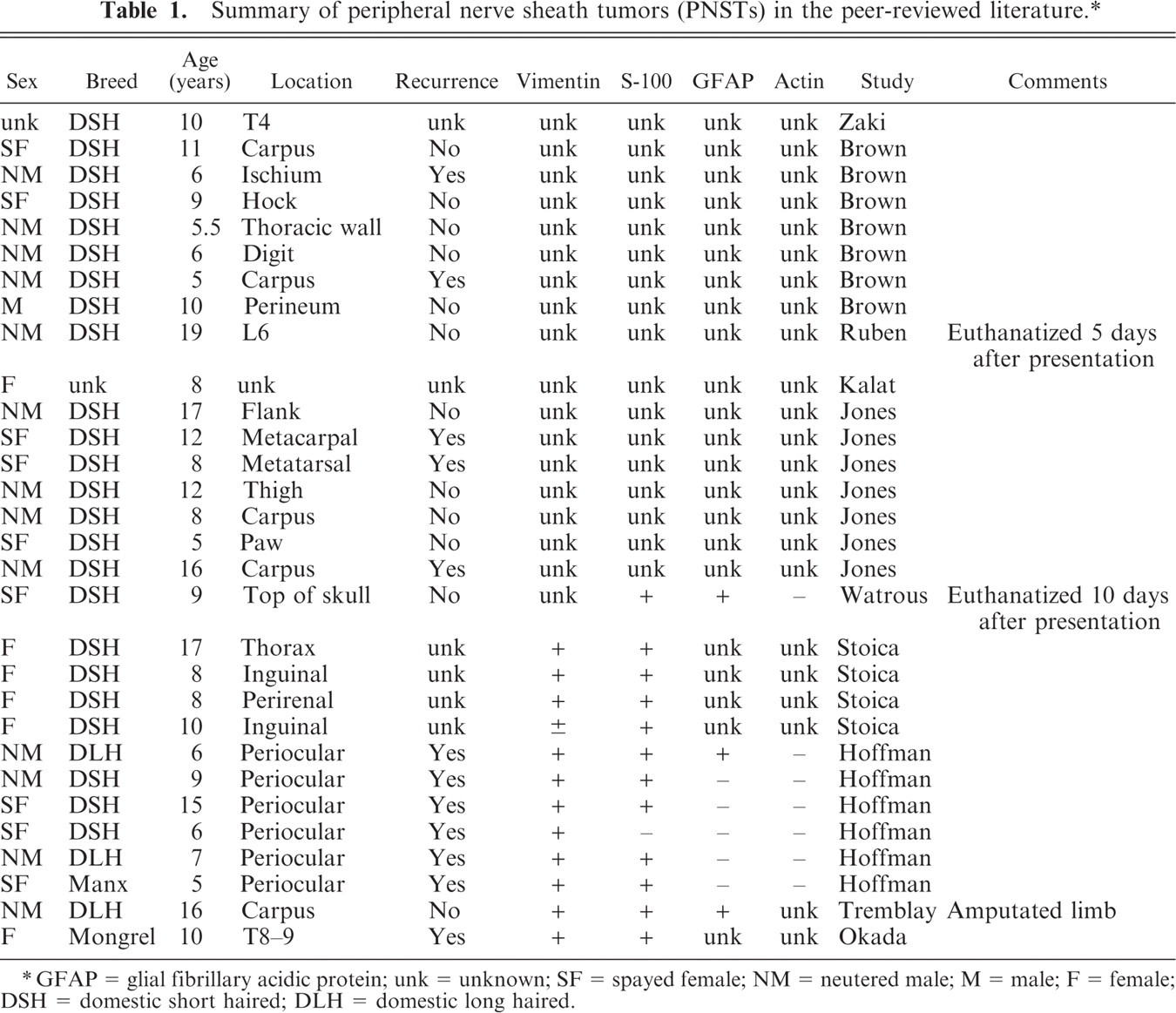
GFAP = glial fibrillary acidic protein; unk = unknown; SF = spayed female; NM = neutered male; M = male; F = female; DSH = domestic short haired; DLH = domestic long haired.
Summary of feline PNSTs in textbooks. ∗

SF = spayed female; NM = neutered male; M = male; F = female; PNSTs = peripheral nerve sheath tumors.
A study of a larger number of immunohistochemically confirmed cases could provide better evaluation of the spectrum of histomorphologic presentations and a more accurate description of biologic behavior while facilitating future investigations of potential therapies. This study describes the histologic, immunohistochemical, and clinical features of 59 PNSTs that were collected from 53 cats and compares the findings with those reported in the literature.
Materials and Methods
Feline tumors diagnosed as PNSTs or spindle cell tumors/sarcomas suspected to be PNST were selected by 2 veterinary pathologists (FYS and Tom Lipscomb) from a subset of case specimens submitted to the veterinary diagnostic laboratory of Marshfield Laboratories between June 2003 and March 2008. An additional case from 2002 was retrieved after it was identified as the initial biopsy of one of the cases collected between 2003 and 2008. The slides were reviewed by all authors.
All tissues had been fixed in 10% neutral buffered formalin, embedded in paraffin by conventional methods, processed routinely, sectioned at 4–6 μ, and stained with hematoxylin and eosin.
Immunohistochemistry was performed using Ventana Autostainers (Benchmark or XT platforms, Ventana Medical System, Inc.) with diaminobenzidine chromogen and Harris hematoxylin counterstain. Antibodies used included those for vimentin (prediluted, monoclonal: V9), S-100 protein (1:4,800, polyclonal rabbit anti-S-100), glial fibrillary acidic protein (GFAP) (1:8,000, polyclonal), neural fibrillary protein (1:500, monoclonal: 2F11), and muscle specific actin (1:200, monoclonal: 1A4). Nonimmune mouse and rabbit serum (1:10,000) were used in place of antibody for negative controls. Feline brain, adrenal gland, small intestine, haired skin, lymph node, and skeletal muscle served as external controls.
Clinical information was obtained from accompanying biopsy submission forms, follow-up questionnaires, and/or telephone conversations with the attending clinician, veterinary technician, or owner.
Results
Fifty-nine PNSTs were collected from 53 cats. Grossly, the tumors were generally described by the histology technicians who processed them as semifirm to firm, off-white to tan, glistening, sessile masses that were sometimes ulcerated (11 of 59) and/or lobulated (7 of 59).
Selected histologic and immunohistochemical findings are summarized in Tables 3 and 4. Histologically, the tumors involved the dermis and/or subcutis in 49 cats; the mucosa and submucosa of the tongue in 2 cats; the dermis, submucosa, and mucosa of a lip in 1; and the mucosa of a lip of another. Most (34) tumors were fairly well circumscribed, exhibiting limited tissue invasion. Twenty-one tumors exhibited more diffuse tissue infiltration, often surrounding and separating adipocytes in a honeycomb growth pattern and/or demonstrating invasion of skeletal muscle fibers. Some abutted the overlying epidermis. Fourteen tumors demonstrated a plexiform growth pattern, affecting multiple nerve fascicles and appearing as variably sized, often elongate nodules in histologic section (Fig 1). In 4 cases, only portions of the tumors with very limited or no tumor margins were submitted, precluding proper evaluation of invasive growth.
Summary of histologic and immunohistochemical findings in 43 histologically benign feline PNSTs. ∗
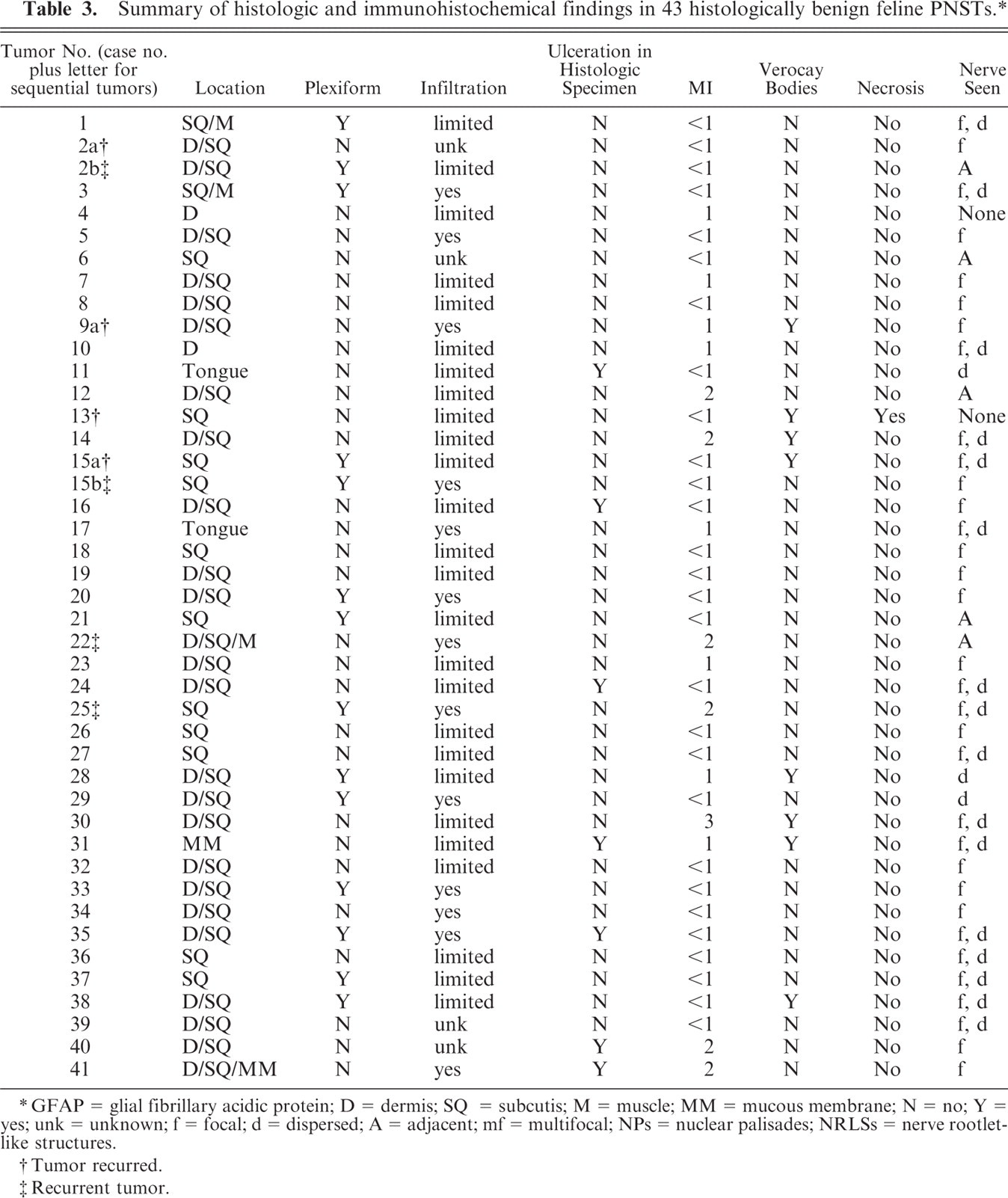
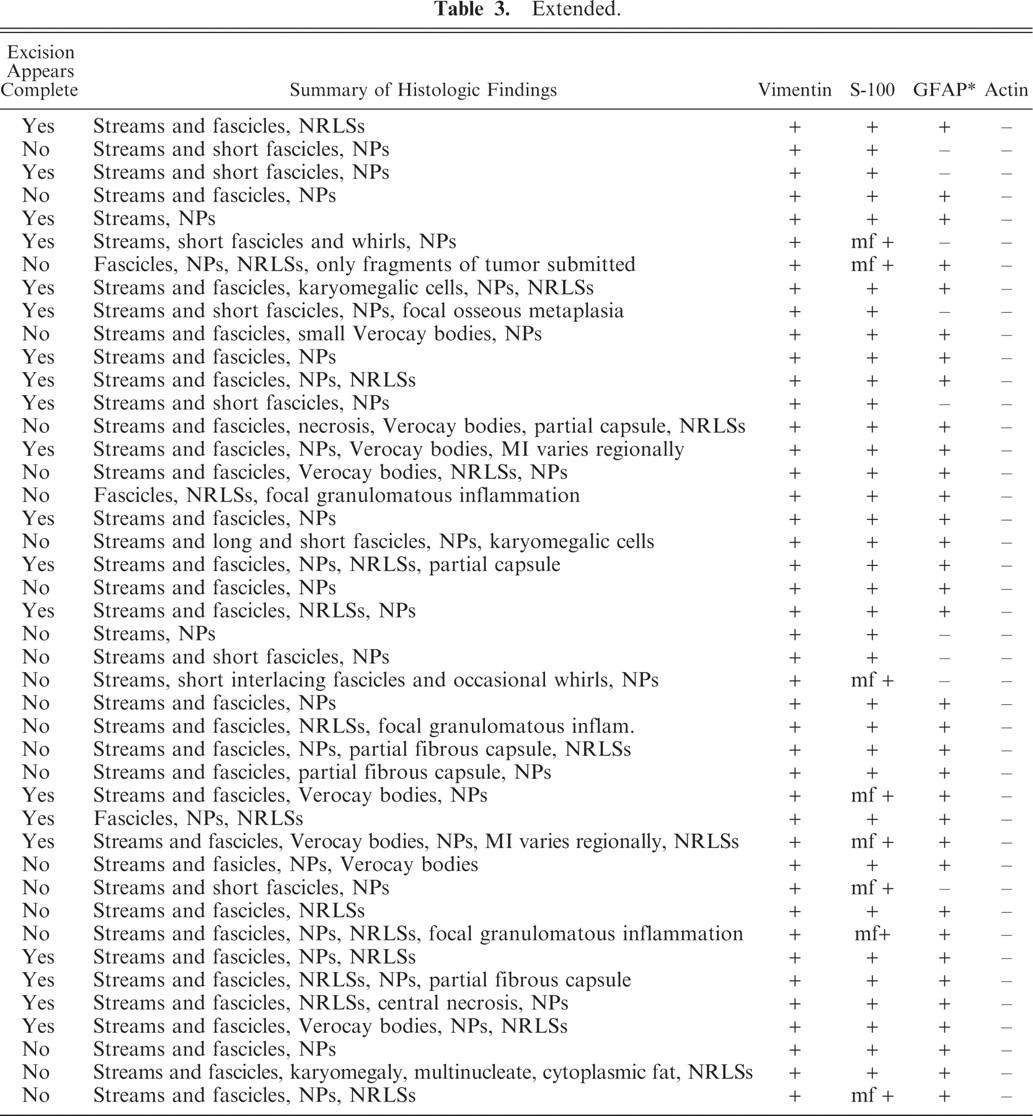
GFAP = glial fibrillary acidic protein; D = dermis; SQ = subcutis; M = muscle; MM = mucous membrane; N = no; Y = yes; unk = unknown; f = focal; d = dispersed; A = adjacent; mf = multifocal; NPs = nuclear palisades; NRLSs = nerve rootlet-like structures.
Tumor recurred.
Recurrent tumor.
Extended.
Summary of histologic and immunohistochemical findings in 16 histologically malignant female PNSTs. ∗
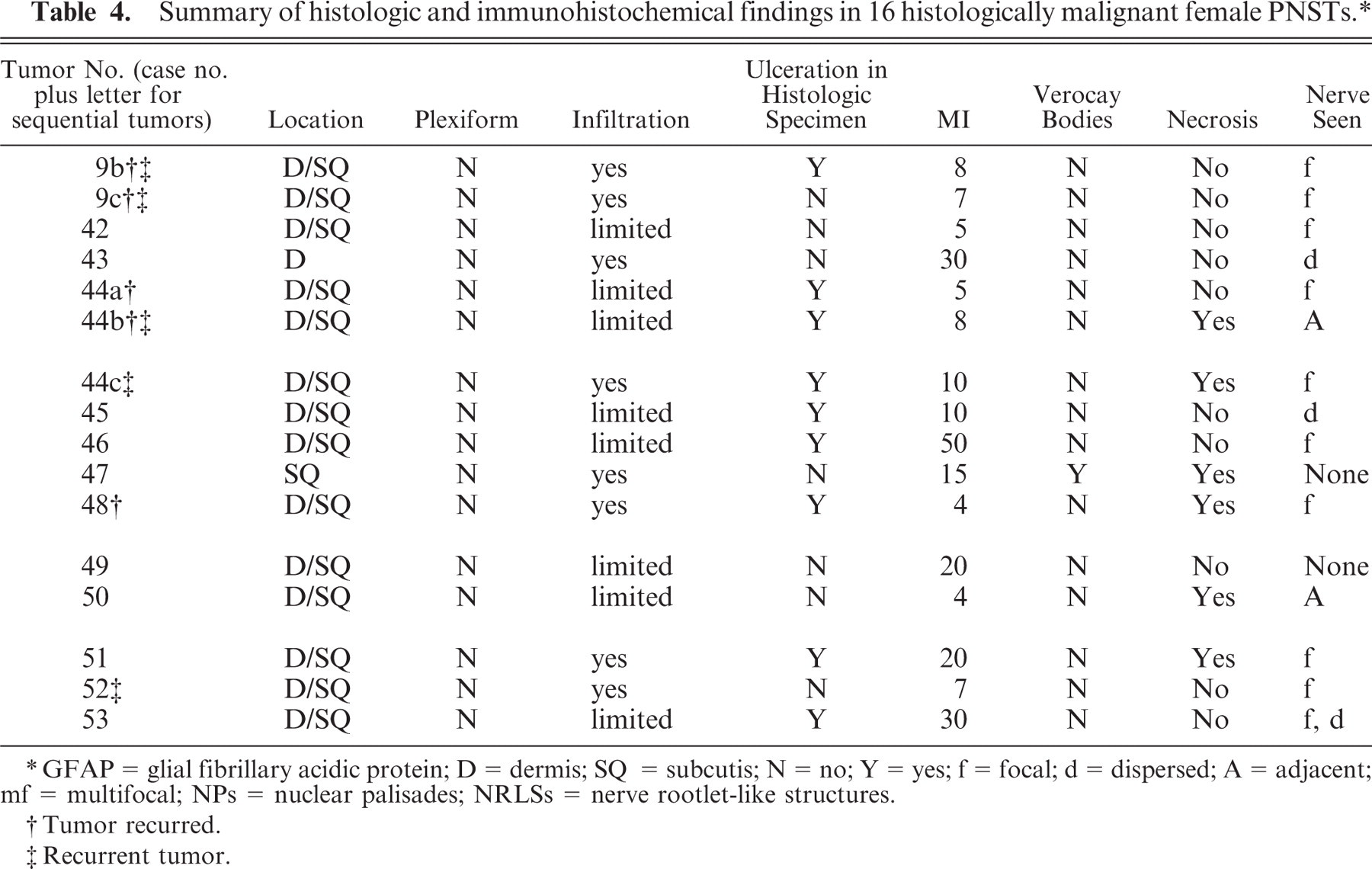
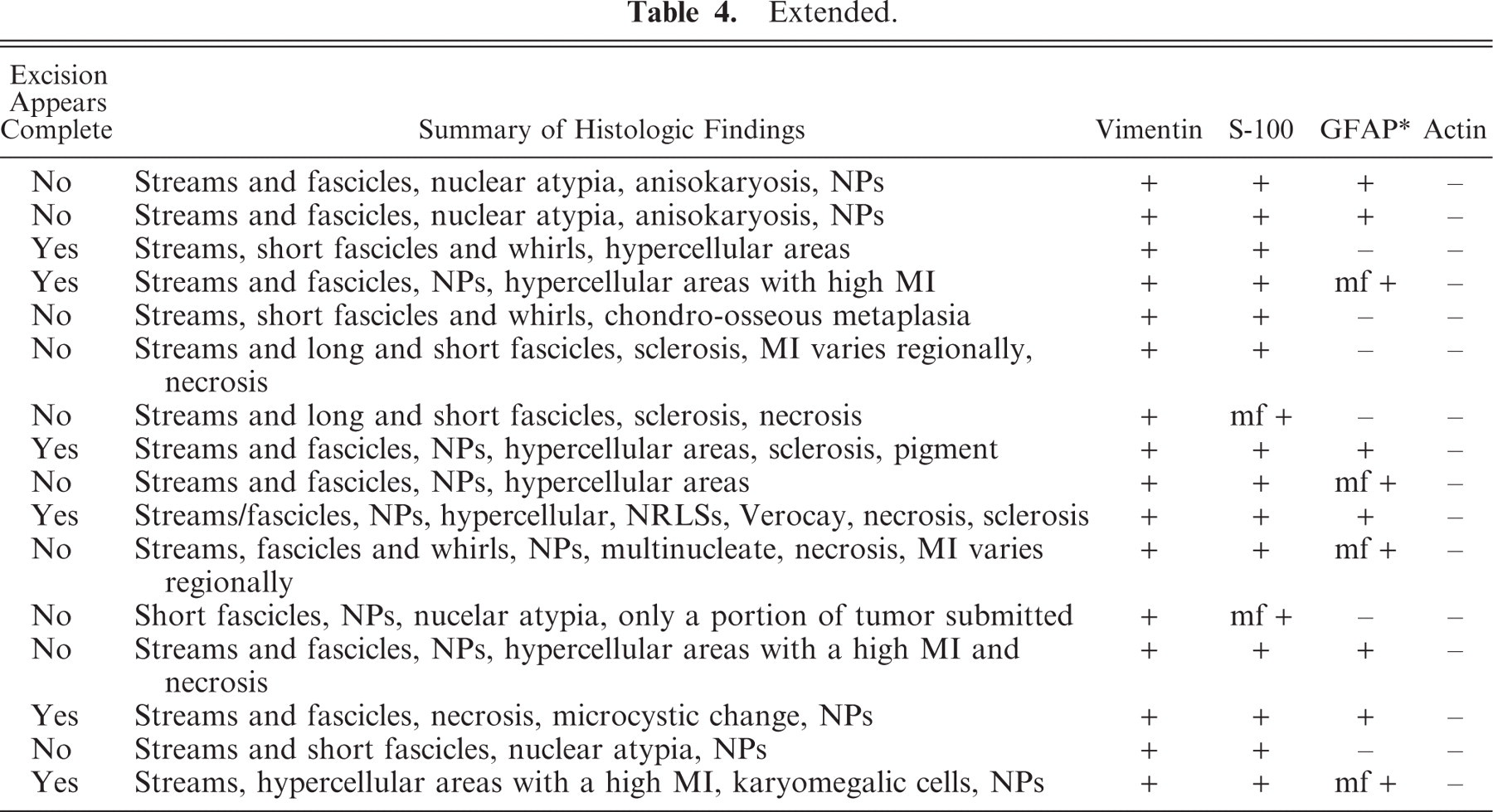
GFAP = glial fibrillary acidic protein; D = dermis; SQ = subcutis; N = no; Y = yes; f = focal; d = dispersed; A = adjacent; mf = multifocal; NPs = nuclear palisades; NRLSs = nerve rootlet-like structures.
Tumor recurred.
Recurrent tumor.
Extended.
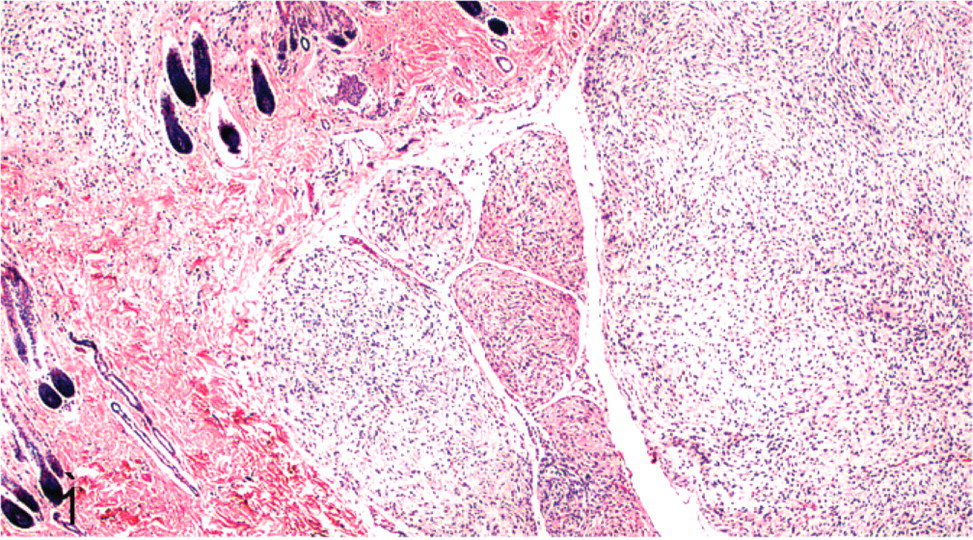
Subcutis; cat No. 2. Peripheral nerve sheath tumor involving multiple nerve fascicles resulting in a plexiform growth pattern. HE.
The neoplasms were characterized by compact to loosely arranged streams and fascicles of spindled cells that had wavy (Fig. 2) to delicate cobweb-like cytoplasmic processes (Fig. 3). Neoplastic cells were multifocally separated by small to occasionally moderate amounts of myxoid to collagenous matrix. Variably prominent nuclear palisading was present, and some (9 of 59) had rows of nuclei separated by anuclear zones of eosinophilic fibrillar material (Verocay bodies) (Fig. 4). Most tumors had areas composed of long fascicles of compact cells that intersected each other at sharp angles (Fig. 5). Many had thin bundles of neoplastic cells with closely packed nuclei that were reminiscent of nerve rootlets (Fig. 6). These nerve rootlet-like structures were frequently separated by small amounts of myxoid matrix. Some tumors had nodular Antoni A areas (see description in the Discussion) that were adjacent to or surrounded by Antoni B or neurofibromatous areas (see description in the Discussion) (Fig. 7). Occasionally, areas of central, closely packed, wavy cells surrounded by more myxoid neoplastic tissue were present in the intraneural or plexiform tumors. Few to many individual mast cells were scattered throughout the tumors, and occasional aggregates of small numbers of lymphocytes and plasma cells were often present. Additional findings seen in a minority of tumors included areas of sclerosis, a partial fibrous capsule, karyomegalic neoplastic cells (Fig. 8), “tactile differentiation” in which the tumor cells were aggregated into structures reminiscent of meissnerian corpuscles (Fig. 9), foci of tumor necrosis, areas of hypercellularity, osseous differentiation, and pigmentation. The mitotic index, that is, the number of mitoses per 10 high-power fields, was usually low. Thirty-five had a mitotic index equal to or less than 1. Forty-three had a mitotic index of less than 4. Of the 16 tumors that had 4 or more mitoses per 10 high-power fields (Fig. 10), all but 1 (No. 44a) had other histologic features of malignancy, that is, hypercellularity, significant nuclear atypia, and/or tumor necrosis. In the histologic sections, 16 tumors were ulcerated, and only 25 appeared to be completely excised.
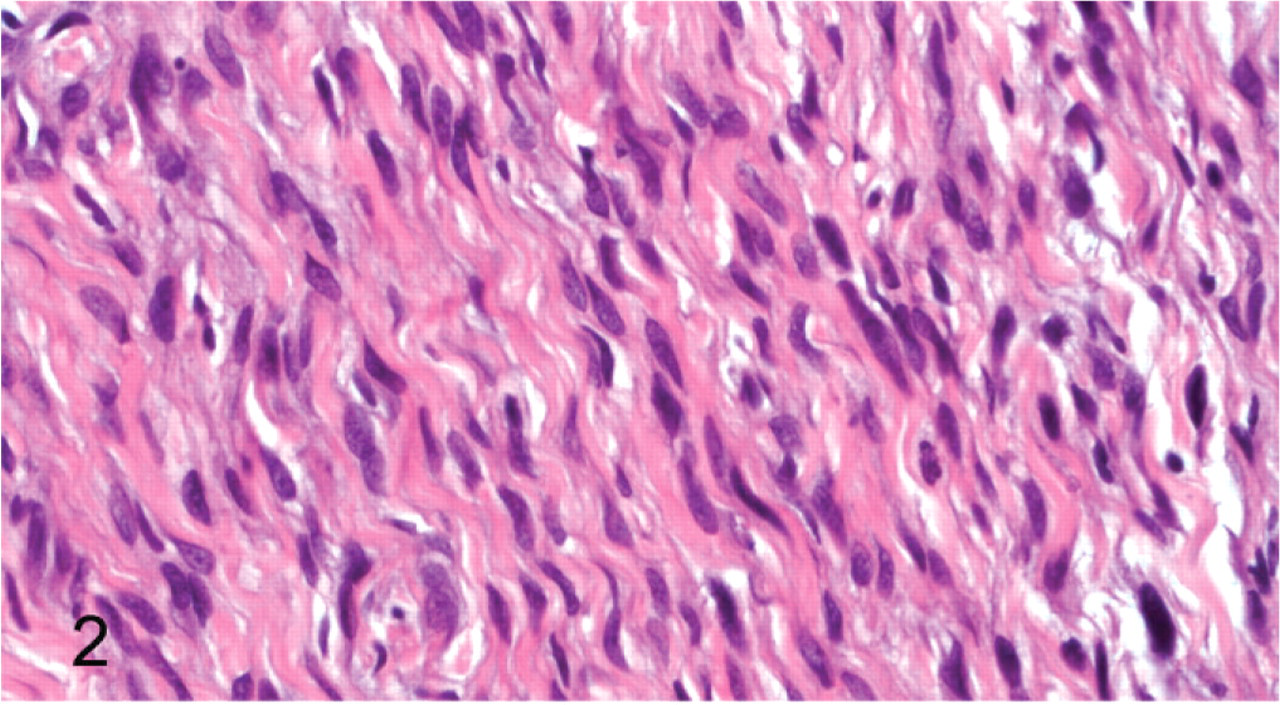
Peripheral nerve sheath tumor; cat No. 27. Neoplastic spindle cells with wavy eosinophilic cytoplasmic processes. HE.
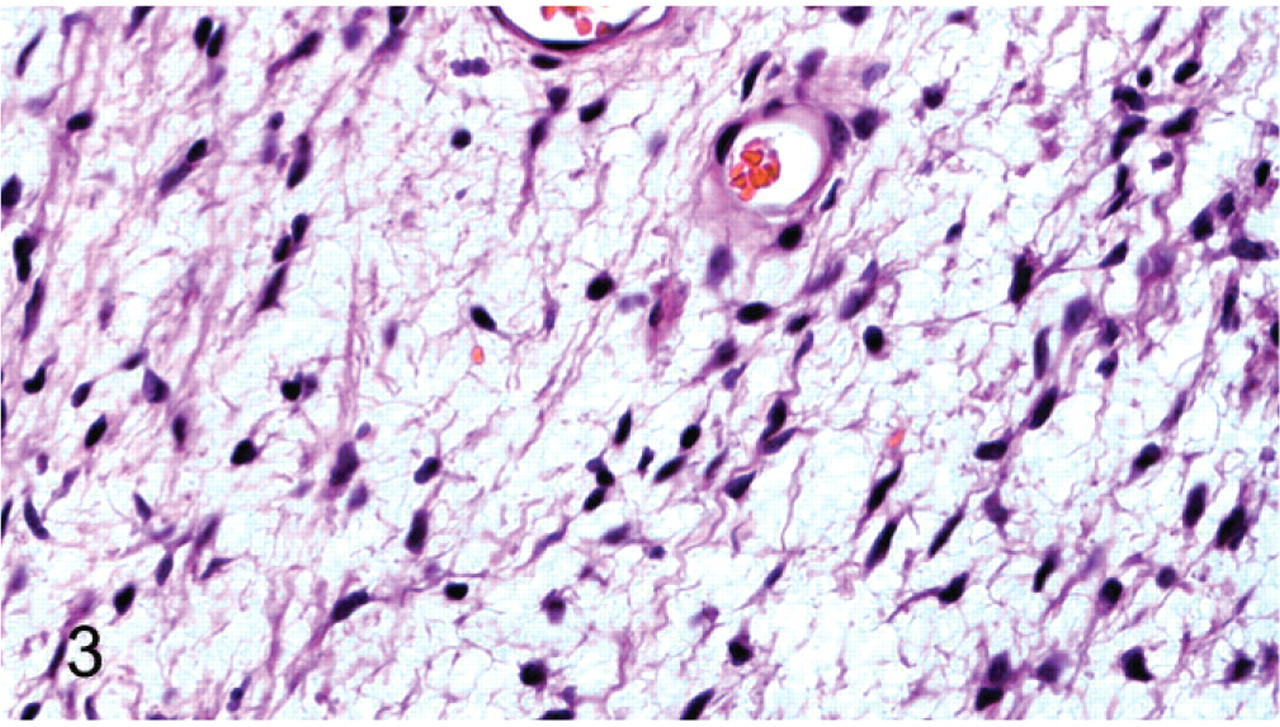
Peripheral nerve sheath tumor; cat No. 14. Loosely arranged neoplastic cells with delicate cobweb-like processes. HE.
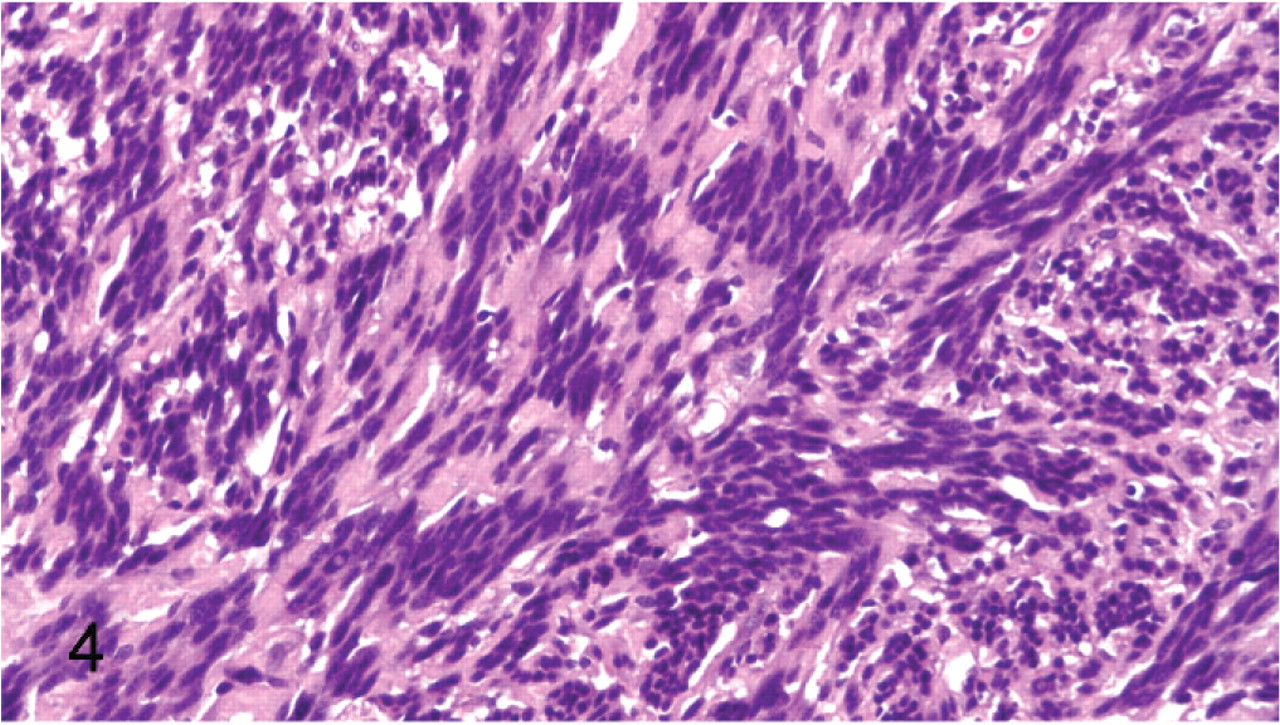
Peripheral nerve sheath tumor; cat No 13. Rows of nuclei separated by anuclear eosinophilic fibrillar material (Verocay bodies). HE.
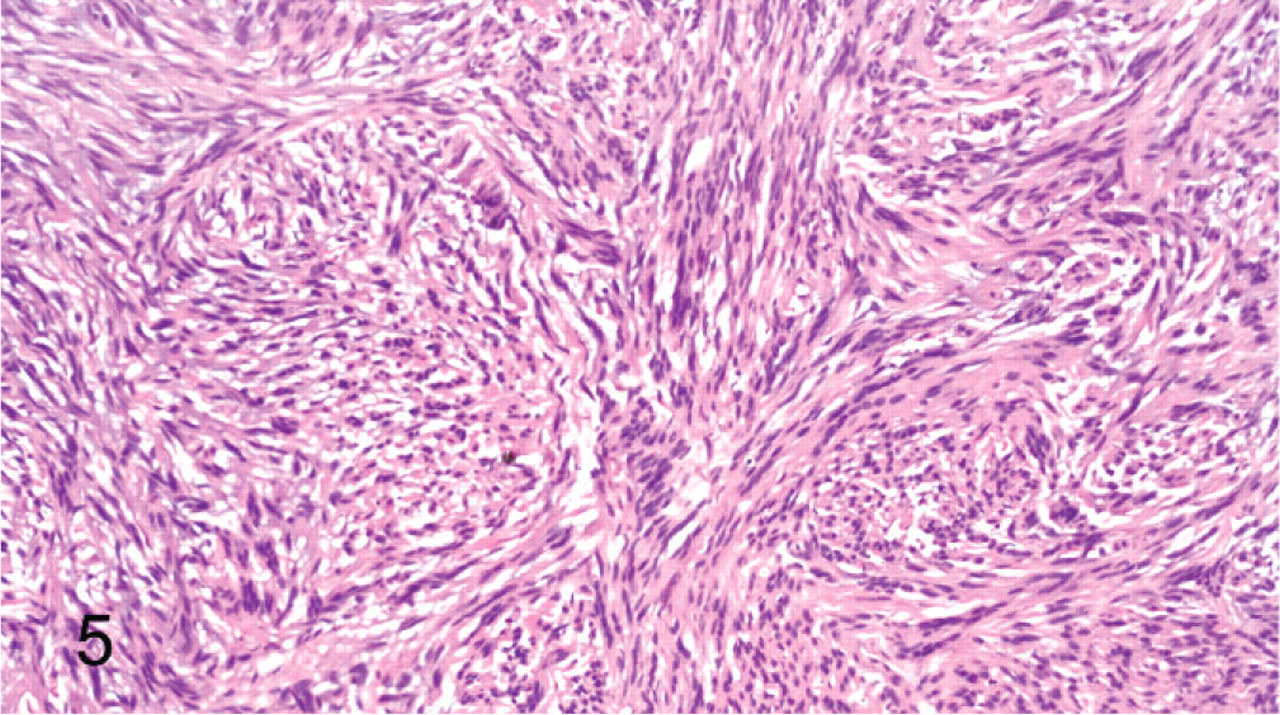
Peripheral nerve sheath tumor; cat No. 28. Long fascicles of compact spindled cells intersect at sharp, almost 90° angles. HE.
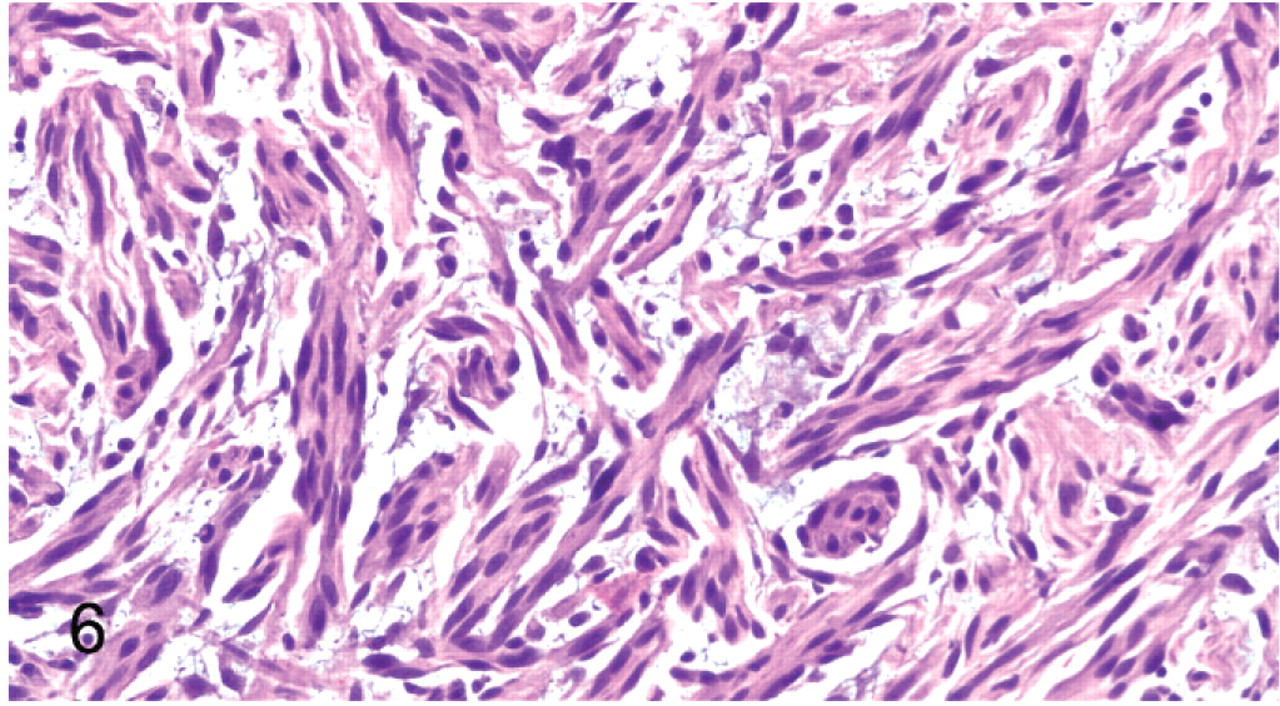
Peripheral nerve sheath tumor; cat No. 11. Thin fascicles of spindled cells with clumped nuclei reminiscent of nerve rootlets. These nerve rootlet-like structures are surrounded and separated by small amounts of myxoid matrix. HE.
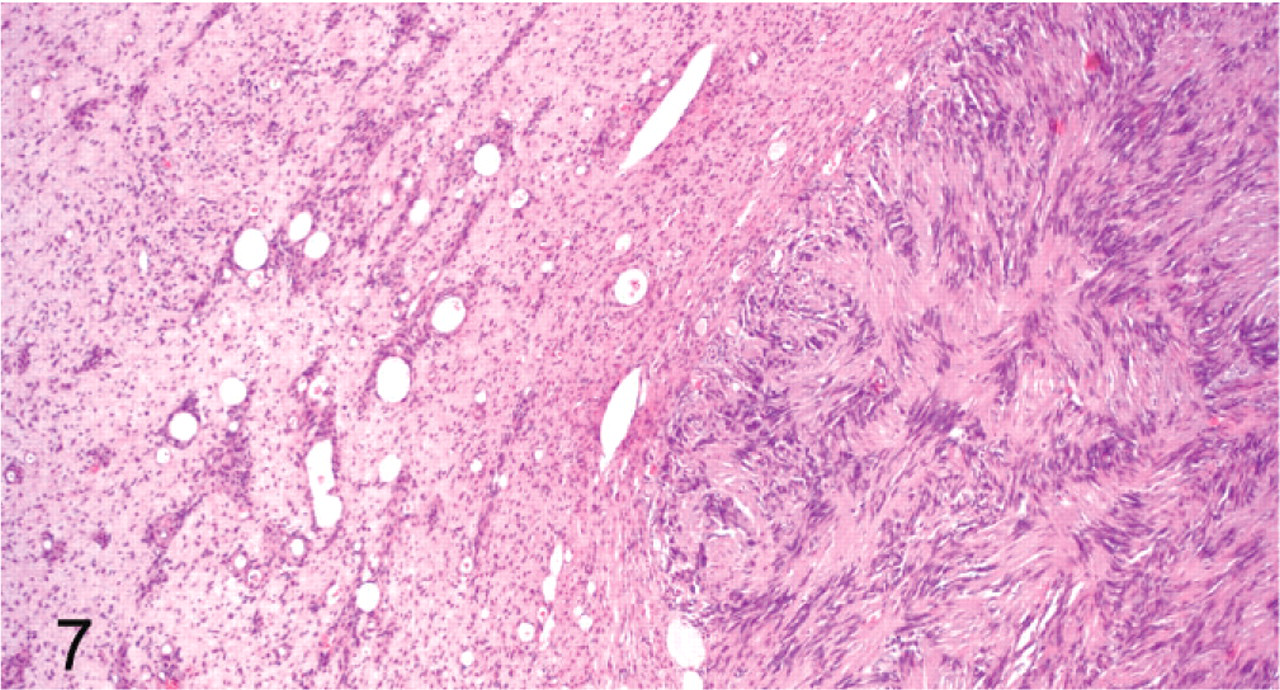
Peripheral nerve sheath tumor; cat No. 14. Antoni A area with Verocay bodies (right side of the field) surrounded by an area of neurofibromatous differentiation (left side of the field). HE.
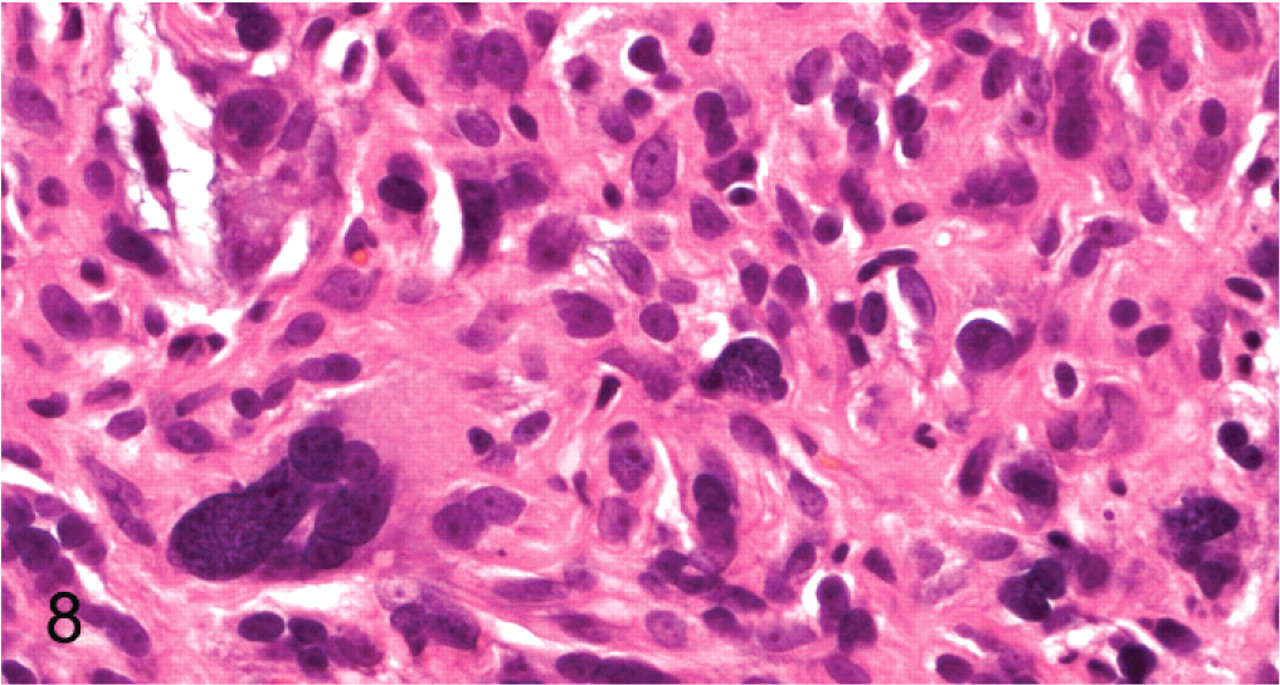
Peripheral nerve sheath tumor; cat No. 40. Karyomegalic, pleomorphic and hyperchromatic nuclei (“ancient” change). HE.
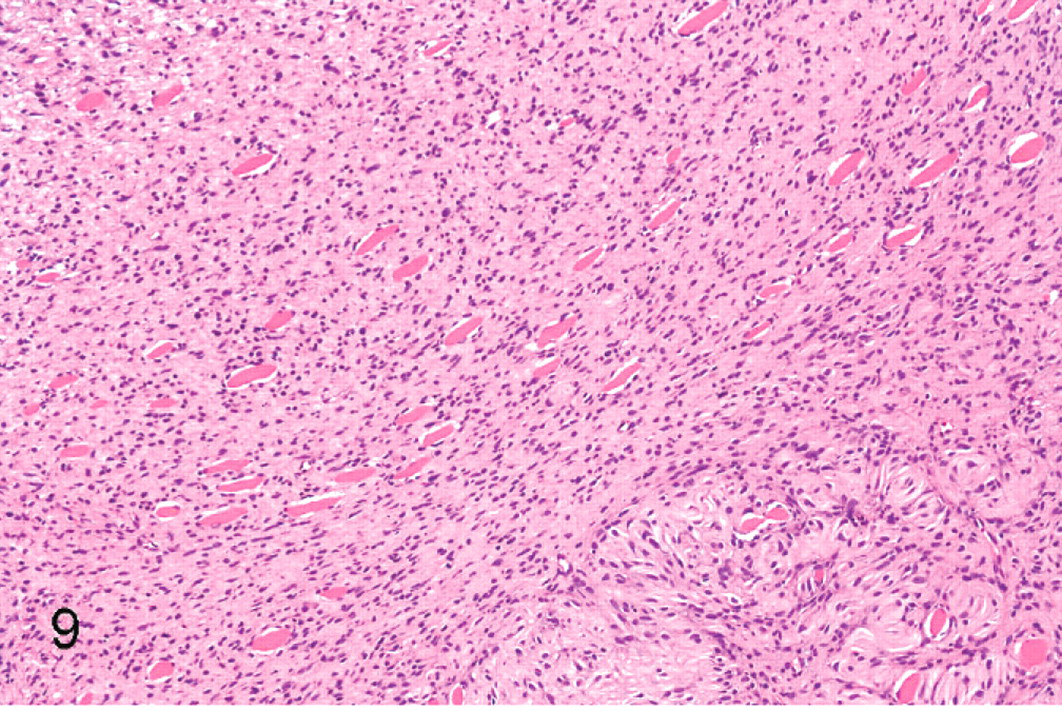
Peripheral nerve sheath tumor; cat No. 17. Spindled neoplastic cells surround and separate myofibers. Two myofibers are indicated by arrowheads. Pseudo-meissnerian corpuscles in the lower right corner. HE.
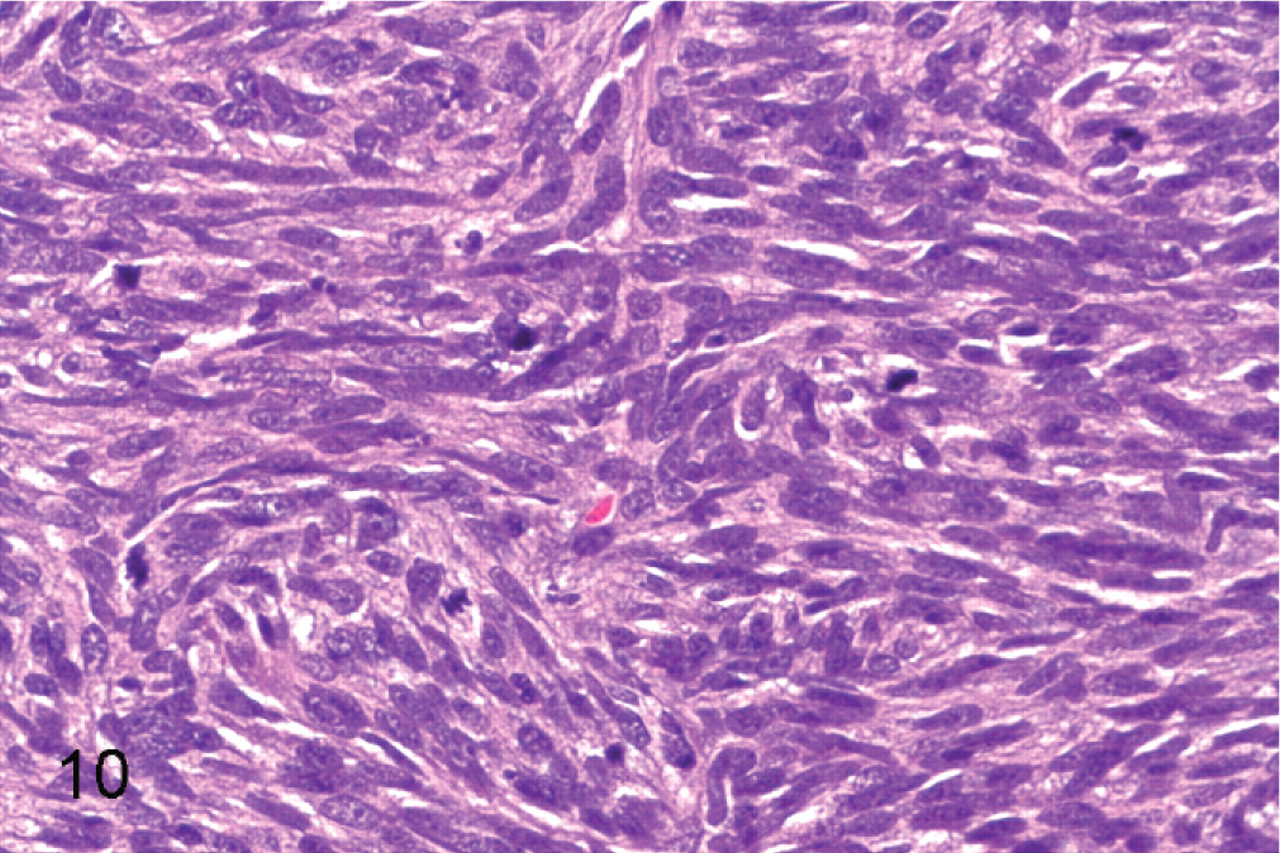
Malignant peripheral nerve sheath tumor; cat No. 53. The histologic features of malignancy in this field include hypercellularity and multiple mitotic figures. HE.
A subgroup of tumors (12 of 59), tumor Nos. 2a, 2b, 5, 8, 12, 21, 22, 23, 32, 42, 49, and 52, was composed of streams of often loosely arranged spindled cells (Fig. 11) and/or short interlacing fascicles (Fig. 12). These tumors lacked Antoni A areas.
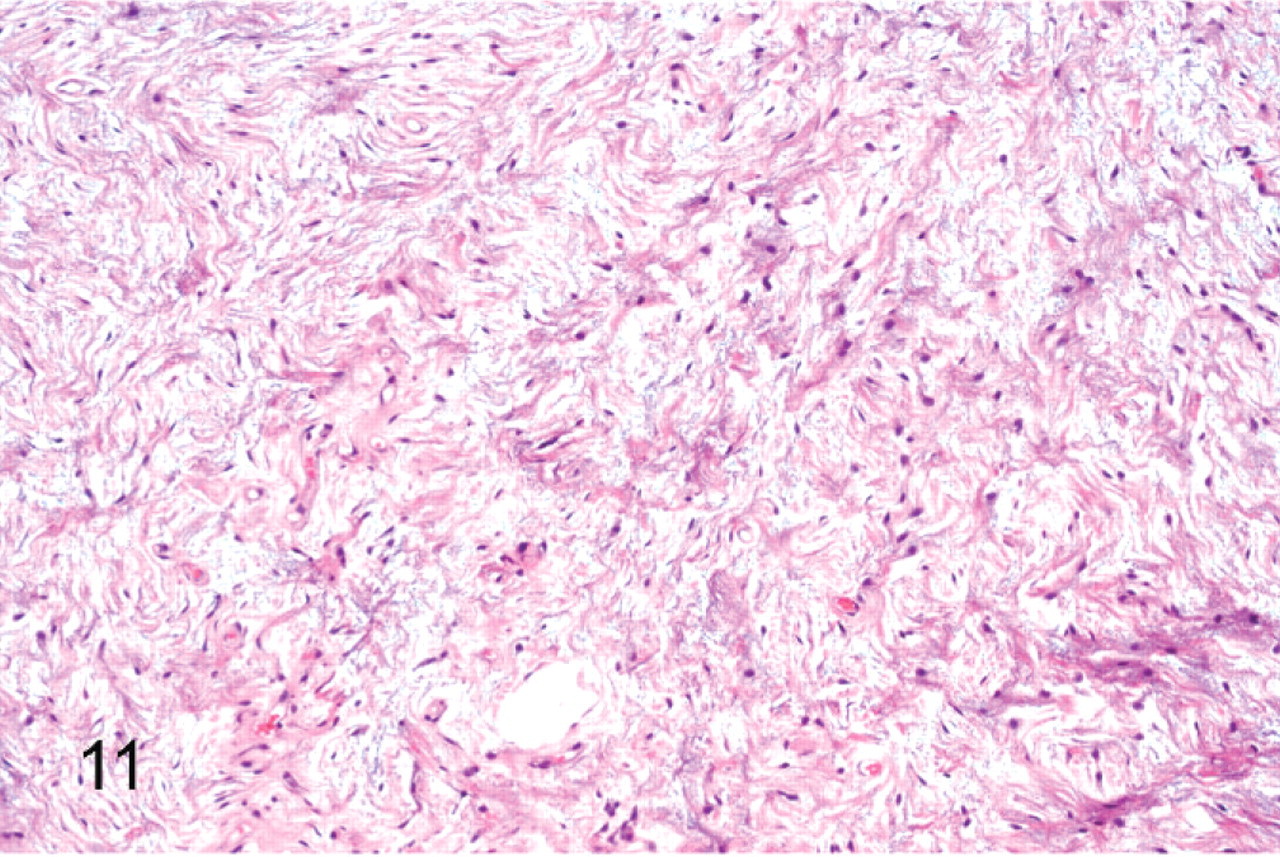
Peripheral nerve sheath tumor; cat No. 21. Loosely arranged spindled cells with myxoid matrix and an absence of Antoni A areas. HE.
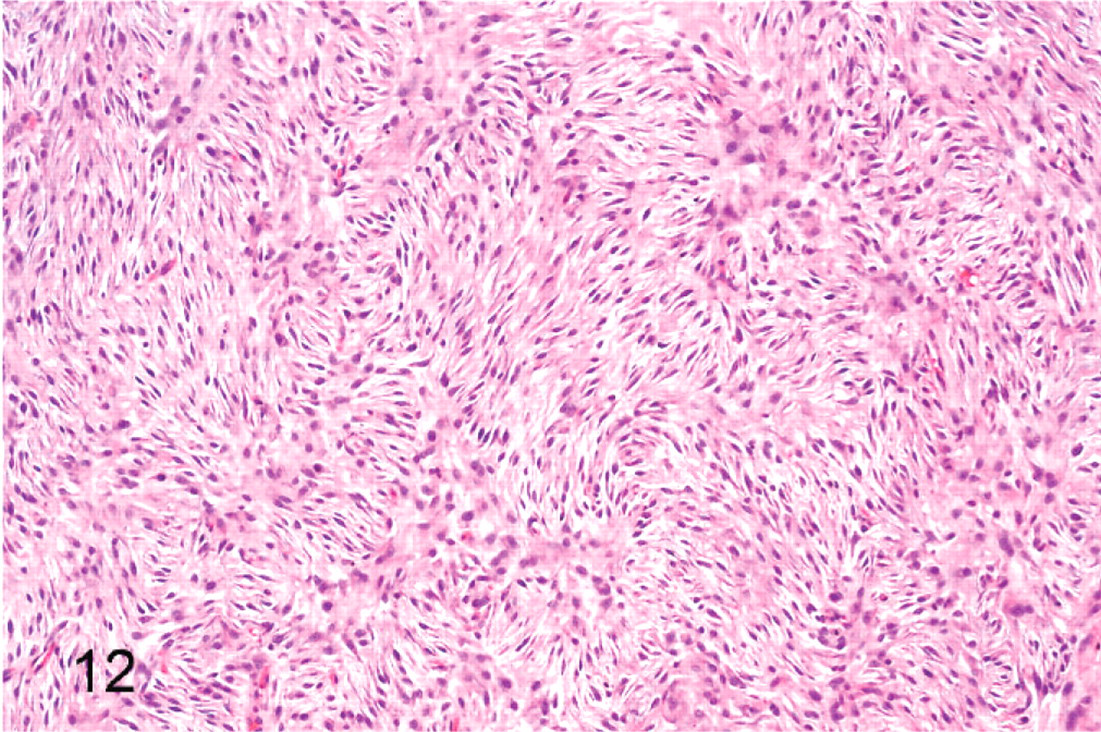
Peripheral nerve sheath tumor; cat No. 52. Spindled cells arranged in short interlacing fascicles with an absence of Antoni A areas. HE.
Immunohistochemically, all tumors were diffusely and strongly positive for vimentin. All were moderately to strongly positive for S-100 protein, 49 diffusely and 10 multifocally (Fig. 13). Forty were diffusely positive, and 4 were multifocally positive for GFAP (Fig. 14). All tumors in the subgroup that were composed of streams and/or short fascicles without Antoni A areas were negative for GFAP. All tumors were negative for muscle-specific actin. Neural fibrillary protein–positive axons were identified in or adjacent to 55 of the tumors. The axons within tumors were present focally (entrapped nerve) and/or in a dispersed pattern (residual neurites).
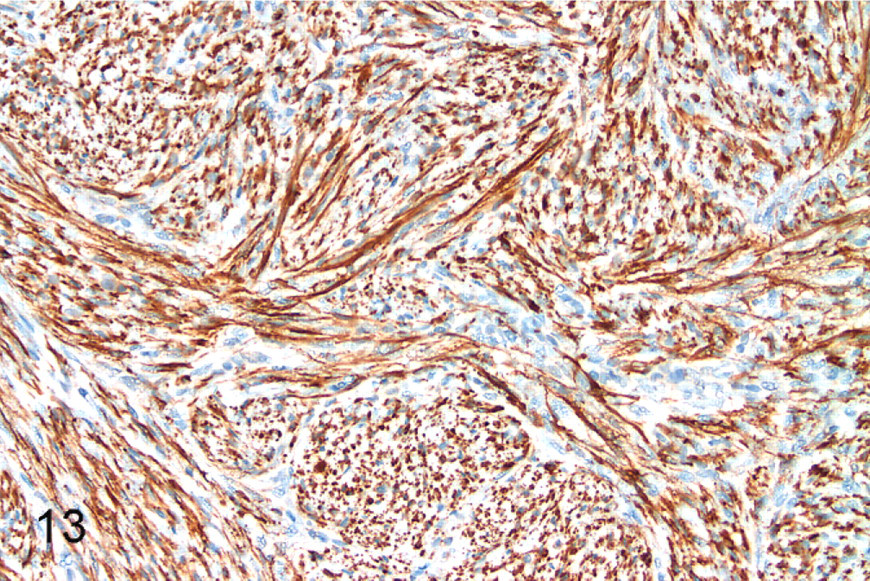
Peripheral nerve sheath tumor; cat No. 7. Diffuse, strong cytoplasmic and multifocally nuclear staining for S-100 protein. Diaminobenzidine staining, hematoxylin counterstain.
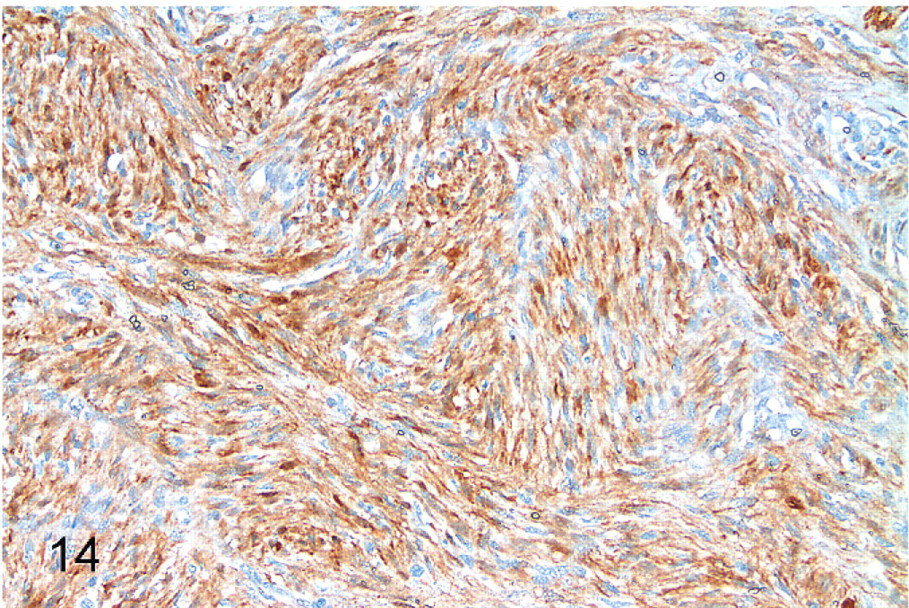
Peripheral nerve sheath tumor; cat No. 7. Diffuse, strong cytoplasmic staining for glial fibrillary acidic protein. Diaminobenzidine staining, hematoxylin counterstain.
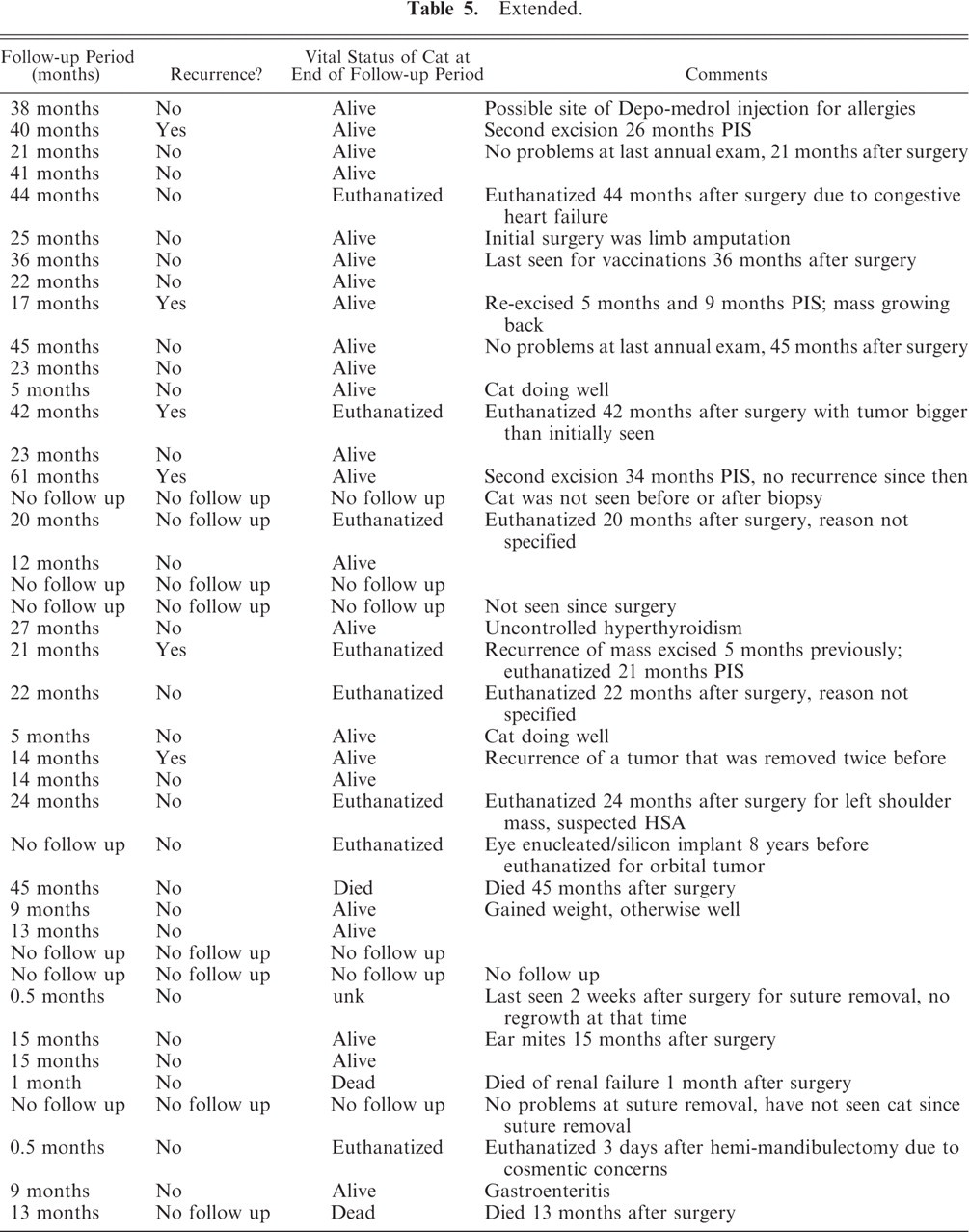
The clinical and follow-up information is summarized in Tables 5 and 6. There were 30 domestic short-haired cats, 6 domestic long-haired cats, 3 domestic medium-haired cats, 4 Siamese cats, 2 Maine Coon cats, 1 Russian Blue cat, 1 Himalayan cat, 1 Persian cat, and 5 cats of unknown breed. Twenty-nine (55%) were neutered males, 23 (43%) were spayed females, and 1 (2%) was an intact male. The age at initial surgery ranged from 3 years to 19 years; the mean age was 10.75 years and the median age was 10 years. Twenty-six cats had tumors on the head and neck, including 7 from the lip area, 2 from the tongue, and 6 periocular. Fourteen had tumors of the limbs, including 9 from the paws. Six cats had a tumor(s) on the back (including 3 in the shoulder area), 1 on the left side of the thorax, 1 on the left side of the abdomen, 1 on the right side, 1 on the ventral abdomen, 1 near the tail base, and 2 on the tail. Seventy-five percent involved the head, neck, or limbs.
Clinical information on 41 cats with histologically benign PNSTs. ∗
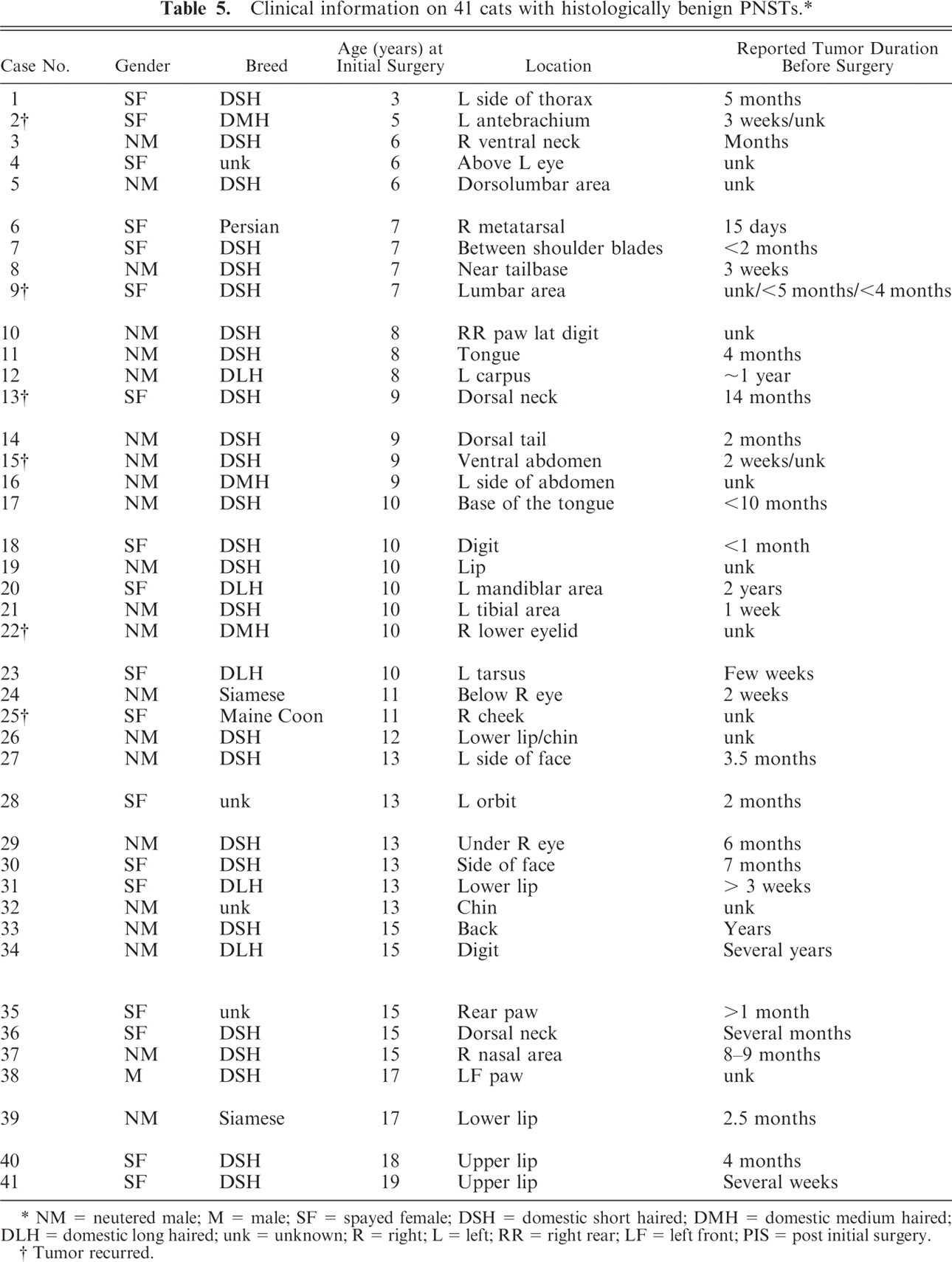
NM = neutered male; M = male; SF = spayed female; DSH = domestic short haired; DMH = domestic medium haired; DLH = domestic long haired; unk = unknown; R = right; L = left; RR = right rear; LF = left front; PIS = post initial surgery.
Tumor recurred.
Extended.
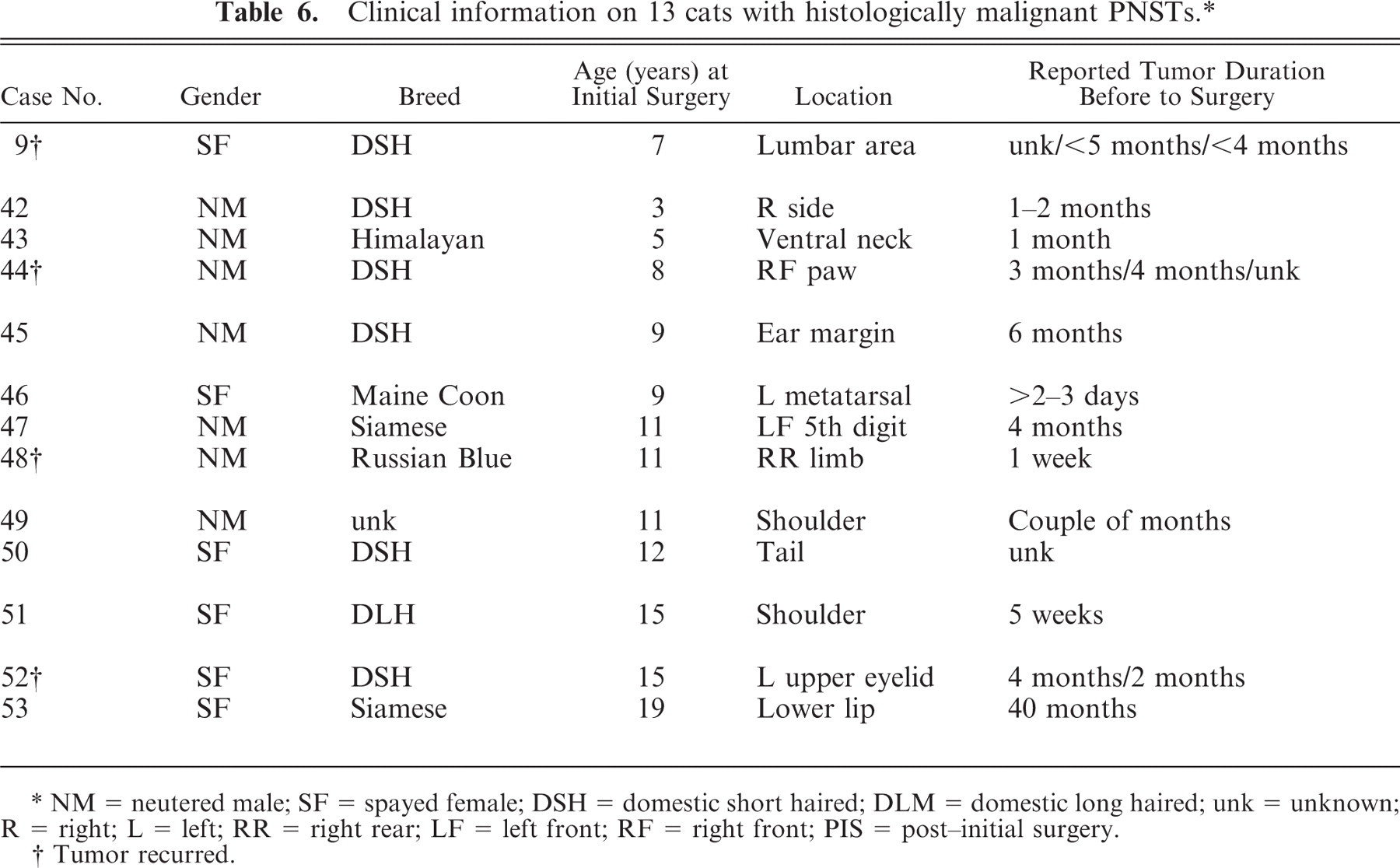
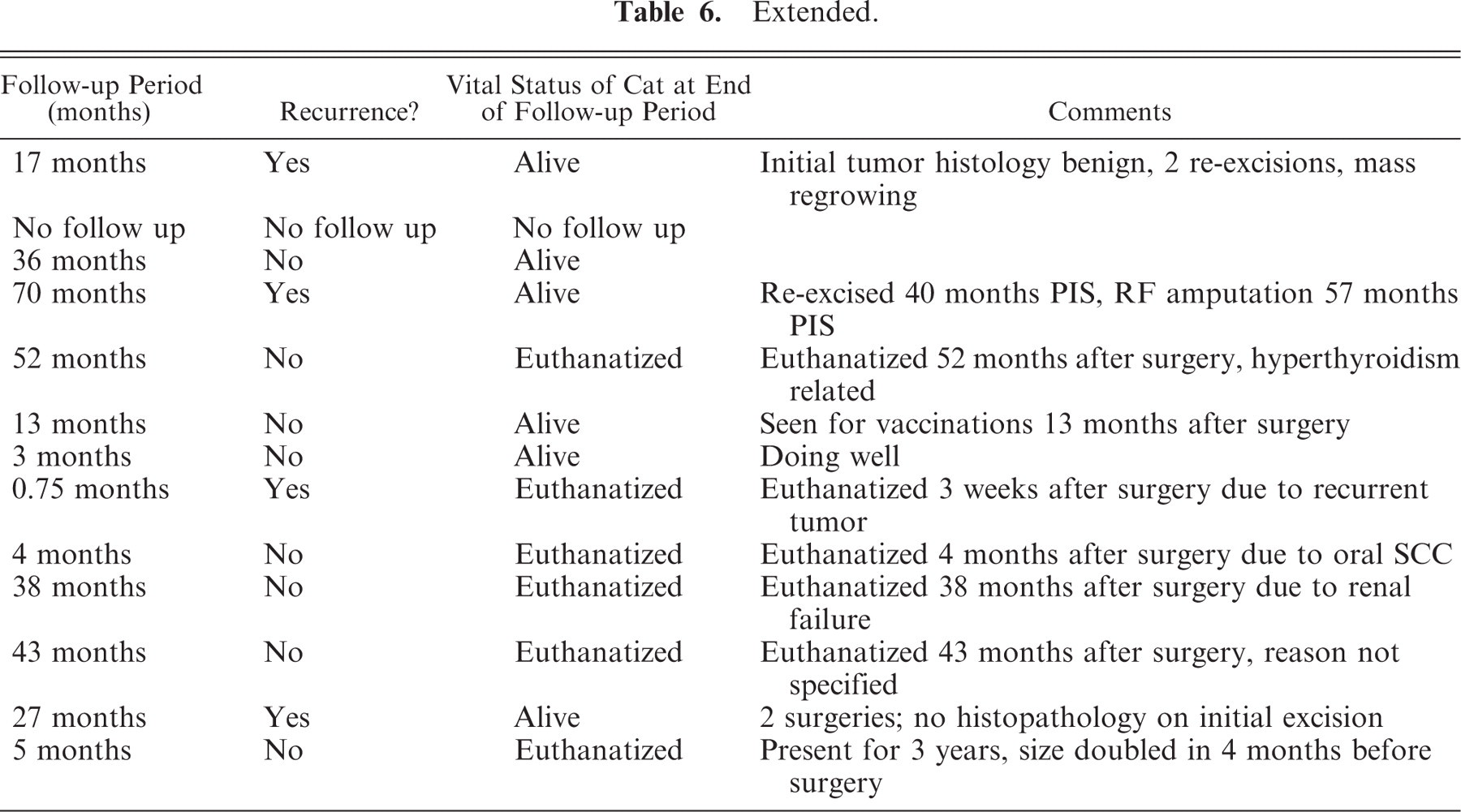
Clinical information on 13 cats with histologically malignant PNSTs. ∗
NM = neutered male; SF = spayed female; DSH = domestic short haired; DLM = domestic long haired; unk = unknown; R = right; L = left; RR = right rear; LF = left front; RF = right front; PIS = post-initial surgery.
Tumor recurred.
Extended.
When the tumors were divided into histologically benign and malignant groups, the clinical information for each group was similar. The age range for each group was 3 to 19 years, and the sex distributions for each group were nearly identical. The mean and median ages for the benign group were 10.78 years and 10 years, respectively; for the malignant group, they were 10.38 years and 11 years, respectively. Thirty-two (78%) of the benign group and 8 (62%) of the malignant group were located on the head, neck, or limbs. However, cats with histologically malignant tumors were more likely to be purebred (5 of 13, or 38%) than cats that developed histologically benign tumors (5 of 41, or 10%).
Clinical follow-up information was obtained in 45 cases. The amount of information available varied from case to case, and the follow-up period ranged from 2 weeks to 70 months. The mean follow-up period was 23.4 months, and the median was 21 months. Forty-one cases had follow-up periods of at least 3 months.
Treatment included surgical excision and/or amputation in all cases. No other treatments were reported. Of the 45 cases for which follow-up information was available, 9 (20%) had local tumor recurrence; 3 of these recurred locally twice. In cat No. 9, the initial tumor was histologically benign and the 2 recurrent tumors had histologic features of malignancy. Therefore, tumors from cat No. 9 are listed in both Tables 3 and 4 and cat No. 9 is listed in both Tables 5 and 6. Metastasis was not documented in any of the cases. Thirteen cats were euthanatized 2 weeks to 52 months (median = 21.5 months, mean = 22.6 months) after surgery for various reasons. One cat was euthanatized and the tumor submitted postmortem.
Discussion
In human medicine, PNSTs are divided into schwannomas, neurofibromas, and malignant peripheral nerve sheath tumors (MPNSTs), because each subgroup has a different set of clinicopathologic features.13–15 Schwannomas occur most commonly on the head, neck, and flexor surfaces of the extremities. They are noninvasive and usually encapsulated. The associated nerve is generally located at the periphery of the tumor, because the tumor arises from and is entirely or almost entirely composed of Schwann cells. Neoplastic Schwann cells are arranged in 2 characteristic patterns, Antoni A and B, in various proportions. The cellular Antoni A region is characterized by Schwann cells aligned in compact streams, fascicles, and occasionally whirls, whereas the hypocellular Antoni B region consists of loosely arranged admixture of Schwann, fibroblast, and perineurial cells in a myxoid matrix (recapitulating normal nerve histology and mimicking neurofibroma) with cobweb-like cytoplasmic proccesses.21 Nuclear clusters, palisades, and double rows of nuclei separated by a zone of eosinophilic cell processes (Verocay bodies) are common in Antoni A areas. The vessels are often hyalinized. Mitotic activity and degenerative atypia, that is, marked nuclear pleomorphism and hyperchromasia seen in longstanding or “ancient” tumors, are acceptable in schwannoma; necrosis is absent. Immunohistochemically, these tumors are uniformly S-100 protein positive and can be GFAP positive. Schwannomas exceedingly rarely undergo malignant transformation to an epithelioid malignancy.10 Schwannomas can be plexiform and multiple. A majority of schwannomas are associated with aberrations of chromosome 22. Most are sporadic, but acoustic schwannomas are associated with neurofibromatosis type 2.15
Neurofibromas, on the other hand, have no site predilection, arise directly from the center of nerves, and extend into surrounding soft tissue. They are composed of widely separated cells (a mixture of Schwann, perineurial-like, and fibroblastic cells) embedded in myxoid to collagenous matrix, with central higher cellularity and peripheral hypocellularity. These tumors surround and separate nerve fibers of the parent nerve, expanding the nerve. Tumor nuclei are smaller than those in schwannomas, and the cytoplasmic processes are generally inapparent in hematoxylin and eosin–stained sections. Although neurofibromas are also positive for S-100 protein, unlike schwannomas, the proportion of immunoreactive cells is variable and the staining is not uniform because S-100 protein only stains Schwann cells, one cellular component of this cellular milieu. Neurofibromas may be sporadic and, in the setting of neurofibromatosis type 1 (von Recklinghausen's disease), have potential to undergo malignant transformation. Clues to early malignant transformation include increased cellularity with cytologic atypia, fascicular growth pattern, and mitotic activity; the last feature is not acceptable in benign neurofibroma in humans.13
In humans, MPNSTs, including tumors that were previously called neurofibrosarcomas and malignant schwannomas, are malignant tumors arising from a peripheral nerve or neurofibroma. These usually occur in large or medium-sized nerves, that is, sciatic nerve, brachial plexus, and paraspinal nerves. Histologically, MPNSTs resemble other soft-tissue sarcomas and one or more of the following criteria are used to confirm the nerve sheath differentiation/origin: presence within a nerve, transition from a benign PNST, immunohistochemical and/or ultrastructural features, association with neurofibromatosis type 1. Most MPNSTs are high grade and have a mitotic index of at least 4. Necrosis is often present in a characteristic geographic pattern, sparing perivascular rings of viable tumor. S-100 protein positivity is usually patchy and varies in intensity, and MPNSTs can stain variably with GFAP. The prognosis for individuals with MPNSTs is poor. These tumors are prone to recurrence and have metastatic potential.14
PNSTs have also been reported in cats, dogs, cattle, rats, and horses.3, 9, 16, 18 These tumors have been subclassified as neurinomas, neurilemmomas, schwannoma, neurofibromas, and neurofibrosarcomas based on their presumed cell(s) of origin. In the dog, 2 groups of tumors have been referred to as PNSTs, 1 occurring in the cranial and spinal nerves and 1 occurring in the skin and subcutis.3, 9 Many of the first group are consistent with MPNSTs and have metastatic potential.18 Those occurring in the skin and subcutis of dogs are usually of uncertain histogenesis and are referred to by some as hemangiopericytoma.3 Palisading, as seen in classic PNSTs, is usually absent.3 These tumors tend to recur, but they rarely metastasize.3 PNSTs in cattle are usually incidental findings at slaughter and tend to involve the autonomic nervous system.18 Multiple nerves can be affected, and this condition in cattle has been referred to as neurofibromatosis, but the histologic features are more consistent with schwannoma.18 Spontaneous and experimentally induced schwannomas have been described in rats.18 Schwannomas with a high incidence of recurrence after surgery have also been reported in the skin and/or subcutis of horses, but misdiagnosis of sarcoids with schwannomatous-like areas is possible.16 Although PNSTs may abut the overlying epidermis, nuclei oriented perpendicular to the epidermis and long, thin rete ridges characteristic of sarcoids are not present.
The PNSTs in the cats of this study were identified histologically by the presence of spindled cells arranged in streams and fascicles, wavy to delicate cobweb-like cytoplasmic processes, and nuclear palisades. The immunohistochemical results support the histologic diagnosis of PNST, that is, all the tumors were S-100 protein positive and muscle-specific actin negative and most (44 of 59) were GFAP positive. Of the previously published cases of feline PNSTs in which immunohistochemistry was performed,5, 11, 17, 19, 20 12 of 12 were vimentin positive, 13 of 14 were S-100 protein positive, 2 of 7 were GFAP positive, and 7 of 7 were negative for smooth muscle actin.
Although feline PNSTs have some features seen in schwannomas, neurofibromas, and MPNSTs, they do not fit well into the subcategories used in human medicine. Although most of the feline tumors have Antoni A areas with nuclear palisades and/or Verocay bodies consistent with schwannoma, very few have a fibrous capsule and many are infiltrative. There is no consistent relationship of the tumor to the associated nerve. Several tumors have areas of both schwannomatous and neurofibromatous histomorphology, and several of the MPNSTs appear to have arisen in tumors with schwannomatous differentiation. Therefore, the more general term of PNST is preferred for these feline tumors. Multiple neurofibromas and malignant transformation from tumors with neurofibromatous differentiation, characteristics of neurofibromatosis, were not found and have not been reported in cats.
The feline PNSTs in this study can be divided histologically into benign tumors with Antoni A areas that are S-100 protein and GFAP positive, benign tumors that lack Antoni A areas and are S-100 protein positive and GFAP negative, and tumors with features of malignancy, that is, mitotic index greater than or equal to 4, nuclear atypia, tumor necrosis, and areas of high cellularity, that are S-100 protein positive and GFAP variable. Recognition of these subtypes may aid in identification of feline PNSTs.
There were 29 neutered males (55%), 1 intact male (2%), and 23 spayed females (43%) in this study. Of the other cases in which the sex was reported in the peer-reviewed literature1, 5–7, 11, 12, 17, 19–21 and 2 textbooks,2, 4 28 of 104 (27%) were spayed females, 31 of 104 (30%) were intact females, 25 of 104 (24%) were neutered males, and 20 of 104 (19%) were intact males. In this study, many different cat breeds were represented. Most cats were domestic short haired (30 of 48, or 63%), but 38% of the histologically malignant tumors were in purebred cats versus 10% of the histologically benign tumors. Of the cases in which the breed affected was reported in the previous literature,1, 5, 6, 11, 12, 17, 19, 20, 22 24 of 28 (86%) were domestic short haired, 3 of 28 (11%) were domestic long haired, and 1 of 28 (4%) was a Manx. Most tumors in this study were on the head (42%) and the limbs (26%). This is similar to what has been reported in the literature,∗ in which 30 of 90 (33%) tumors for which the location was given were located on the head and neck; 42 of 90 (47%) on the limbs; 7 of 90 (8%) on the thorax; 2 of 90 (2%) on the back; 1 (1%) each on the tail, flank, and perirenal, perineal, and ischial areas; and in which 4 of 90 (4%) were perispinal and 2 of 90 (2%) were inguinal.
In this study, 9 of the 45 (20%) cats for which follow-up information was available had tumor recurrence. In another case (cat No. 34), there was granulomatous inflammation adjacent to the tumor, raising the possibility that this too was a recurrent tumor. Tumor recurrence was reported for all 3 of the histologic subtypes. In 6 cases that had recurrence, the initial and/or recurrent tumor had a mitotic index of 2 or less. The other 3 had initial mitotic indices of 4–7. Therefore, the mitotic index alone is not predictive of potential for recurrence. Of the cases in the literature for which follow-up information was given,1, 2, 4–6, 11, 12, 19, 20 16 of 41 (39%) were reported to recur, in some cases up to 6 times.5
Sixteen of the PNSTs from 13 cats in this study were considered histologically malignant. Although both histologically benign and histologically malignant tumors recurred, 4 of the 43 (14%) histologically benign tumors recurred and 5 of the 16 (31%) histologically malignant tumors recurred, suggesting that feline PNSTs with malignant histologic features are more likely to recur. No feline PNST, histologically malignant or otherwise, in this study or elsewhere in the literature,1, 2, 4–7, 11, 12, 17–20, 22 has been documented to metastasize, with follow-up times as great as 5.8 years.
Although the published reports and textbooks suggest that feline PNSTs are rare, they may be underdiagnosed due to uncertainty of histogenesis. The presence of wavy cytoplasm, nuclear palisades, long fascicles intersecting at sharp angles, nerve rootlet-like structures, Verocay bodies, and/or S-100 protein with or without GFAP-positivity can help identify many of these tumors. Additionally, there is a subset that lacks Antoni A areas but has wavy cytoplasm, nuclear palisades, and often short interlacing fascicles. A third subset has histologic features of malignancy. Regardless of the histologic subset, they have potential for local recurrence but have not been documented to metastasize. Additional studies to further investigate the cellular composition of and potential therapies for these tumors are needed.
Footnotes
∗ References 1, 2, 4–6, 11, 12, 17–19, 20, 22.
Acknowledgements
We thank all the submitting veterinarians and owners for their willingness to contribute to the study. We thank Dr. T. P. Lipscomb for providing cases and reviewing the manuscript. Ms. L. Zimmerman of Marshfield Laboratories, Marshfield, Wisconsin, provided invaluable logistic support. This paper was supported in part by the American Registry of Pathology. T. O. Johnson is a Lieutenant Colonel and P. R. Facemire is a Major in the US Army. The opinions and assertions contained herein are the private views of the authors and are not to be construed as official or as reflecting the views of the Department of the Army or Department of Defense.
