Abstract
Sheep in New Zealand develop small intestinal adenocarcinomas more frequently than sheep elsewhere in the world. This high rate of neoplasm development could be due to a genetic predisposition or due to an environmental carcinogen. Differentiation between a genetic and an environmental factor is important as, if an environmental carcinogen is present, people could be exposed directly or by consuming sheep meat. In humans, germline defects in the mismatch repair (MMR) genes cause hereditary nonpolyposis colorectal cancer (HNPCC). Affected people are predisposed to neoplasm development, most commonly colonic adenocarcinomas. It was hypothesized that MMR defects are common within the New Zealand sheep flock, and these defects predispose New Zealand sheep to intestinal neoplasia. To investigate this, immunohistochemistry was used to evaluate the expression of the MMR proteins MSH2, MSH6, MLH1, and PMS2 within 49 ovine intestinal adenocarcinomas. Neoplastic cells within all sheep tumors expressed MSH2, MSH6, and MLH1. Expression of PMS2 could not be assessed, most likely because of insufficient affinity of the anti-human PMS2 antibody to ovine PMS2. The consistent expression of MSH2, MSH6, and MLH1 within the ovine intestinal adenocarcinomas does not support the hypothesis that defects in the MMR genes are common in New Zealand sheep.
Ovine small intestinal adenocarcinomas (SIAs) have behavioral, histologic, and immunohistochemical similarities to human colonic adenocarcinomas. 11,12 Ovine SIAs are common in New Zealand with rates of tumor development 8 times higher in sheep from some regions than sheep from other countries. 17 The reasons for this high rate of neoplasm development are not known, but SIAs are more common within some sheep breeds 17 suggesting a genetic predisposition.
There are two main genetic abnormalities that predispose humans to colonic cancer. 10 First, familial adenomatous polyposis is caused by defects in the adenomatous polyposis coli (APC) gene. 5 Affected individuals develop 100–1,500 colonic polyps that progress to adenocarcinoma in almost all cases. 10 Second, hereditary nonpolyposis colorectal cancer (HNPCC) is caused by defects within the DNA mismatch repair (MMR) genes. 1,3,14 Affected individuals often develop colonic cancer at a young age without previously developing multiple colonic polyps. 1 As human MMR defects also cause reproductive, stomach, small intestine, liver, and other extracolonic malignancies, this disease is also known as Lynch Syndrome. 1,14
The aim of this study was to investigate whether sheep in New Zealand are genetically predisposed to SIA development. A defect within the APC gene was considered unlikely as few sheep with SIAs have multiple polyps. 11 However, it was considered possible that MMR defects could be common in the New Zealand sheep flock. A high frequency of germline MMR defects could then explain the high SIA rates observed in sheep in New Zealand. To investigate this, immunohistochemistry was used to detect expression of the MMR genes, MSH2, MSH6, MLH1, and PMS2, by ovine SIA cells.
Forty-nine ovine intestinal tumors were detected by gross examination of viscera at a slaughter-house. All 49 samples came from the Manawatu region, a geographic location previously reported to have a high prevalence of SIAs. 17 Three intestinal samples from animals with Mycobacteria avium subs paratuberculosis were used as nonneoplastic controls. All samples were fixed in 10% buffered formalin for between 3 days and 1 week and embedded in paraffin. Sections for immunohistochemistry were cut at a thickness of 5 μm, deparaffinized in xylene, rehydrated in graded ethanol, and rinsed in distilled water. Endogenous peroxidases were neutralized with 1% hydrogen peroxide in methanol for 15 minutes followed by rinsing for 5 minutes in distilled water. Antigen retrieval was achieved by incubating slides in antigen retrieval solution (ethylenediamine tetra-acetic acid [EDTA]/Tris buffer pH 9.0) in a water bath for 40 minutes with 20-minute cool down. Using an autostainer, slides were incubated for 30 minutes with a mouse anti-human MLH1 antibody at a dilution of 1 : 50, a mouse anti-human MSH2 antibody at a dilution of 1 : 100, a mouse anti-human MSH6 at a dilution of 1 : 50, and a mouse anti-human PMS2 at a dilution of 1 : 50, respectively (mouse anti-human antibodies MLH1, MSH2, MSH6, and PMS2; BD Biosciences, San Jose, CA). An EnVision horseradish peroxidase (HRP)-labeled polymer immunoperoxidase staining procedure (Dako, Carpinteria, CA) was used for immunolabeling. The immunoreaction was “visualized” with 3,3′-diaminobenzidine substrate (Dako). Sections were counterstained with Harris' hematoxylin. Nonneoplastic intestinal epithelial cells, lymphocytes, and stromal cells were used as positive controls. Negative controls were treated identically as routine sections except that the 30-minute incubation with primary antibodies was replaced with 30-minute incubation with buffer.
Histology confirmed that all 49 tumors were SIAs. None of the tumors contained significant quantities of mucin or tumor-infiltrating lymphocytes. Immunohistochemistry revealed diffuse consistent intranuclear expression of MSH2, MSH6, and MLH1 by internal positive controls as well as neoplastic intestinal epithelial cells within all 49 samples (Figs. 1, 2). The consistent MMR expression by the SIA cells suggests neoplasm development was not due to inherited germline MSH2, MSH6, or MLH1 mutations. No reactivity to PMS2 was observed in internal positive controls from either sheep with neoplastic disease or sheep without neoplastic disease. Attempts to evaluate expression of PMS2 were only performed in 3 SIAs because of the failure to demonstrate PMS2 expression in any sample.
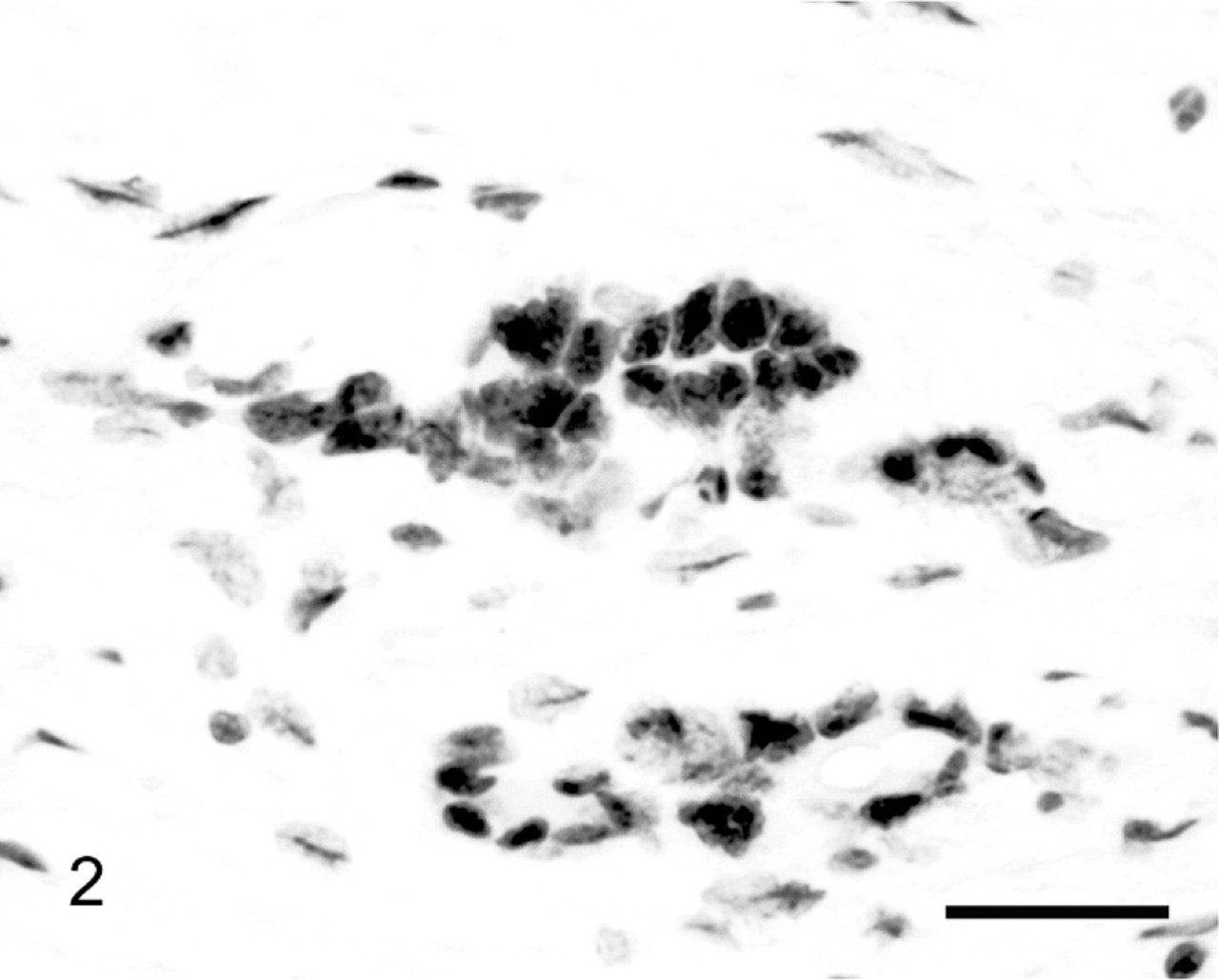
Small intestinal adenocarcinoma; sheep. Neoplastic cells showed nuclear reactivity with antibodies against MLH1. EnVision horseradish peroxidase–labeled polymer immunoperoxidase and Harris' hematoxylin. Bar = 20 μm.
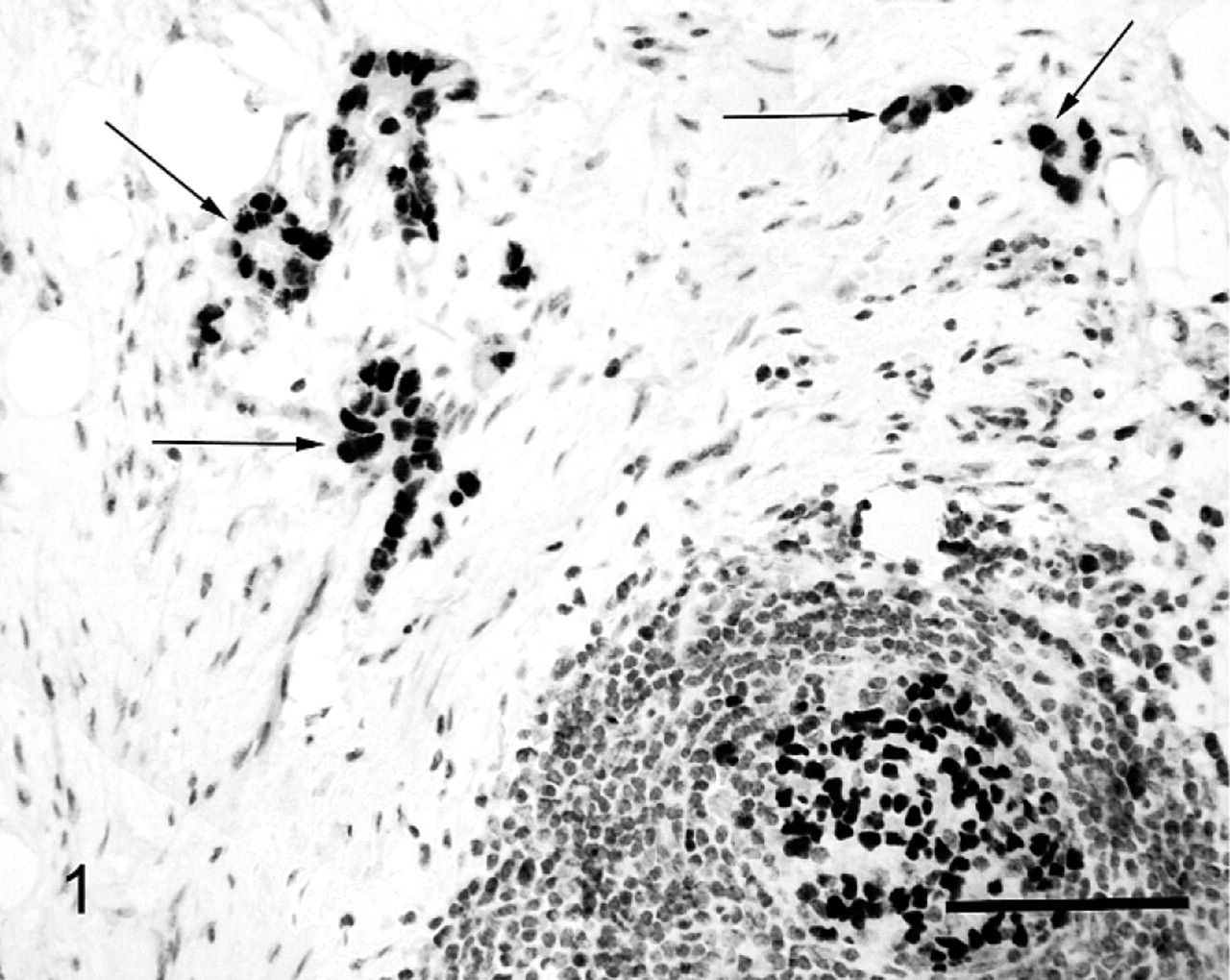
Small intestinal adenocarcinoma; sheep. Neoplastic cells (arrows) and lymphoid follicular cells (bottom right) showed strong nuclear reactivity with antibodies against human MSH6. EnVision horseradish peroxidase–labeled polymer immunoperoxidase and Harris' haematoxylin. Bar = 42 μm.
Loss of genetic stability within a cell can be due to chromosomal or microsatellite instability (MIN) mutations. 2 Chromosomal instability mutations result in gains or losses of large sections of chromosomes owing to dysfunction of proteins regulating DNA replication or mitotic-spindle checkpoints. 2,8 MIN mutations result in subtle changes within the DNA sequence. 8 Microsatellites are short repeating portions of DNA that contain the same number of base pairs within every cell in the body. 1 Microsatellites are particularly vulnerable to base/base mismatches or small insertion/deletion loops during DNA replication. 3 Such replication errors lead to microsatellites of varying lengths within the body (microsatellite instability). 1,3 The MMR system prevents MIN mutations and MMR-deficient cells have 100–1,000 times more MIN mutations than normal cells. 1–3 The mammalian MMR system contains 4 main proteins. These 4 proteins form 2 heterodimers. The heterodimer MutSα consists of MSH2 and MSH6 and detects mismatches within the DNA. 4 On detecting a mismatch, MutSα associates with another heterodimer, MutLα. 4 MutLα consists of MLH1 and PMS2 and couples mismatch recognition with the downstream mismatch repair processes. 4
HNPCC causes 5 to 10% of human colorectal cancers and is due to inherited germline mutations that inactivate one MMR gene allele. 1 Loss of one allele does not impair MMR, and neoplastic disease does not develop until somatic inactivation of the wild-type allele occurs, usually around the 4th decade of life. 1 Loss of normal expression of MSH2 is observed in 40% of patients with HNPCC, loss of MLH1 in 39%, loss of MSH6 in 15%, and loss of PMS2 in 6%. 20 A person with HNPCC has a lifetime colorectal cancer risk of 50 to 80%, compared with 5% within the general population. 1 Immunohistochemical examination of a neoplasm from a person with HNPCC reveals normal MMR expression in nonneoplastic cells, but no MMR expression in neoplastic cells. 19
In the present study, expression of MSH2, MSH6, and MLH1 was visible in all ovine SIA cells. Defects in these 3 genes account for 94% of human HNPCC cases. This suggests that inherited defects within the MMR genes resulting in an HNPCC-like syndrome in sheep is unlikely to be the cause of the high rate of ovine SIA in New Zealand. However, it is possible that the ovine SIAs contained MMR proteins that had retained their immunoreactivity, but are no longer functional. Alternatively, it is also possible that sheep may have defects within a currently unrecognized MMR component. Finally, as PMS2 could not be detected within any ovine tissues, an inherited PMS2 dysfunction predisposing to SIA cannot be excluded. Loss of MMR function within a tumor results in the development of high MSI. 1–3 Investigation of MSI within these tumors is planned as an indirect measure of MMR activity.
Dysfunction of MMR is present in 15% of non-HNPCC human colon cancers. 8 Therefore it was surprising that loss of MMR expression was not detected within any ovine SIAs. Histologic features of MMR-deficient colon cancers include a high frequency of mucinous subtypes and large numbers of tumor-infiltrating lymphocytes 9 ; neither of these features was visible within any of the ovine SIAs. It is uncertain why MMR dysfunction appears to be rare within ovine SIAs, but selection of ovine SIAs with these histologic features may enable identification of MMR-deficient tumors.
Expression of PMS2 was not detected immunohistochemically within any ovine cell including those from animals without neoplastic disease. The complete lack of detectable PMS2 expression was considered most likely to be because the antibody against human PMS2 protein that was used in this study had a low affinity for ovine PMS2 protein. In addition, inconsistent fixation times have been previously reported to impair immunohistochemical detection of PMS2 expression. 7 The ovine samples were fixed in formalin for at least 3 days, which could have further reduced the antibody affinity. Alternatively, it is possible that sheep do not express PMS2. However, the MMR system is highly conserved within bacteria, yeast, and mammalian cells 3 and this is considered less likely. The possibility that all 6 examined sheep had PMS2 defects was considered. However, the lack of PMS2 expression by nonneoplastic cells would indicate that all 6 tested sheep had homozygous PMS2 mutations. Although the results cannot exclude this possibility, such a high frequency of PMS2 mutations within the sheep population was considered unlikely.
The observation that sheep in New Zealand develop markedly higher rates of SIAs than sheep elsewhere in the world indicates that there is an underlying genetic or environmental predisposing factor. Previous studies have not provided evidence to support APC defects, 11 and the present study does not support MMR defects as the cause of the high rate of SIAs in New Zealand sheep. Supporting an environmental carcinogen, epidemiologic studies have reported links between neoplasm development and exposure to phenoxy and picolinic acid herbicides 13 and high stocking rates. 17 In addition, the different rates of neoplasm development within different areas of New Zealand 17 could suggest different levels of exposure to an environmental carcinogen.
People in New Zealand have the highest rate of colorectal cancer in the world. 6,15 While genetic factors are probably contributory, studies in other countries show that immigrants tend to develop the colon cancer rate of their adopted countries, suggesting environmental factors are more important. 16 High fat diets have been found to be a strong risk factor for colorectal cancer. 18 However, colorectal cancer rates in New Zealand are higher than rates in other countries that consume similar, or higher, levels of fat. 6,15 The unexplainably high rate of colorectal cancer in New Zealand may suggest that both people and sheep are exposed to an environmental carcinogen that promotes intestinal neoplasia. People could be exposed directly or by eating sheep meat. To the authors' knowledge, other New Zealand domestic species, including ruminants, do not have an elevated incidence of intestinal neoplasia.
While mice with MMR defects have been developed, 3 this is the first time that MMR expression has been evaluated within a spontaneous neoplasm from a non-human species. The results of this study suggest that some MMR proteins may be sufficiently conserved in non-human species to allow immunohistochemical detection using antibodies against human MMR proteins.
In conclusion, the results of this study do not support the hypothesis that the high rate of SIAs observed in New Zealand sheep is due to a high frequency of inherited MMR defects. The results of this study confirm that some of the MMR proteins are highly conserved between species and suggest anti-human MMR antibodies should allow evaluation of MMR expression in non-human species.
