Abstract
Around 1.6% of sheep in New Zealand develop small-intestinal adenocarcinomas. These neoplasms typically develop widespread metastases. The common development of these neoplasms and their subsequent behavior suggests that sheep could be a useful animal model of human colonic cancer. However, for an animal model of human disease to be relevant, similar genetic mutations should be present. Genetic mutations within human colonic cancers frequently result in expression of cycloxygenase-2 (COX-2), loss of membranous expression of β-catenin and E-cadherin, and accumulation of p53 protein within the neoplastic cells. Immunohistochemistry was used to investigate the presence of these 4 proteins within 26 ovine intestinal adenocarcinomas. Loss of membranous β-catenin reactivity was observed in 14 of 26 ovine intestinal adenocarcinomas (54%). The loss of membranous β-catenin reactivity was accompanied by cytoplasmic and nuclear reactivity in 2 neoplasms. Loss of E-cadherin was observed within 8 of 26 neoplasms (31%). Neoplastic cell expression of COX-2 was observed in 12 of 26 neoplasms (46%), whereas cells within 3 of 26 neoplasms (11%) contained visible p53 protein. In conclusion, all 4 proteins that commonly have altered expression in human colonic cancers were also altered in a proportion of the ovine intestinal adenocarcinomas. These results provide additional evidence that sheep could be useful for the study of human colonic cancer.
Intestinal epithelial neoplasms represented 11% of all new human cancers in the USA in 2001. 12 Seventy percent of these tumors developed within the colon, 26% in the rectum, and 4% in the small intestine. 12 Almost all neoplasms were adenocarcinomas, 13 and 97% of human intestinal neoplasms are considered sporadic. In 2001, intestinal adenocarcinomas were the second most frequent cause of cancer mortality within the USA. 12
Genetic mutations in neoplastic cells can result in protein loss or accumulation. Altered cellular protein concentrations can have undesirable effects, including increased proliferation and dissemination of the neoplastic cells. Among the most important proteins to become dysregulated in colon cancer cells are β-catenin, E-cadherin, cycloxygenase-2 (COX-2), and p53 protein. 3, 18, 35 Beta-catenin can accumulate within the cell because of mutations within either the adenomatous polyposis coli (APC) gene or the β-catenin gene. 21 Beta-catenin is a component of the WNT signalling pathway, 4 and increased protein concentrations promote transcription of genes important in regulating the cell cycle. 3, 4 E-cadherin is a transmembrane protein that forms intercellular bonds between epithelial cells. 39 Loss of E-cadherin promotes neoplasm dedifferentiation, invasion, and metastasis. 10, 39 Expression of COX-2 is common within colonic neoplasms, 14 and evidence suggests that therapeutic enzyme inhibition reduces cancer development and progression. 26 The mechanisms by which COX-2 expression influences neoplastic cell behavior are currently unresolved. 35, 40 Mutations within the p53 protein gene are common in all types of human neoplasms. 9 The p53 protein plays key regulatory roles in cell cycling and apoptosis pathways. 1
About 1.6% of sheep in New Zealand spontaneously develop small-intestinal adenocarcinomas. 7 These tumors rapidly metastasize to local lymph nodes before, in most cases, developing widespread metastases. 24 Because ovine intestinal adenocarcinomas have some histologic and behavioral similarities to human colonic tumors, sheep may provide a large animal model to investigate treatment options of advanced human colonic neoplasia. This study investigated expression of β-catenin, E-cadherin, COX-2, and p53 by ovine small-intestinal adenocarcinoma cells. The expression of these 4 proteins by ovine cells was compared with the reported protein expression in human colonic adenocarcinoma cells, as well as expression within other colon cancer animal models.
Materials and Methods
Sample collection
Twenty-six ovine intestinal adenocarcinomas were detected by gross examination of viscera at a slaughterhouse. When a tumor was detected, the complete intestinal tract was collected. Adjacent lymph nodes, mesentery, and serosal surfaces from the affected animal were then examined for evidence of metastatic spread. All samples were collected, fixed in 10% buffered formalin for 24 hours, and then routinely processed for histologic evaluation.
Immunohistochemistry
Sections from all neoplasms were cut at a thickness of 5 μm, deparaffinized in xylene, rehydrated in graded ethanol, and rinsed in distilled water. Immunohistochemical staining was achieved by using a routine protocol. 37 Briefly, endogenous peroxidases were neutralized with 3% hydrogen peroxide for 5 minutes, followed by rinsing for 5 minutes in distilled water. Antigen retrieval was achieved by incubating slides in an antigen retrieval solution in a steamer for 20 minutes. Nonspecific immunoglobulin binding was blocked by incubation of slides for 10 minutes with a protein-blocking agent (Dako, Carpinteria, CA) before application of the primary antibody. By using an autostainer, slides were incubated for 30 minutes with a rabbit polyclonal anti-β-catenin antibody (Abcam Inc., Cambridge, MA) at a dilution of 1°100, a mouse monoclonal anti-E-cadherin (Transduction Lab, Lexington, KY, USA) at a dilution of 1°200, a rabbit polyclonal anti-cycloxygenase-2 (COX-2, Cayman, Ann Arbor, MI) at a dilution of 1°100, and a rabbit polyclonal anti-p53 (Signet Laboratories, Dedham, MA) at a dilution of 1°100, respectively. A streptavidin-immunoperoxidase staining procedure (Dako, Carpinteria, CA) was used for immunolabeling. The immunoreaction was “visualized” with a 3,3′-diaminobenzidine substrate (Dako). Sections were counterstained with Mayer's hematoxylin. Non-neoplastic intestinal epithelial cells within the sections were used as positive controls for β-catenin and E-cadherin, whereas adjacent non-neoplastic intestinal epithelial cells were used as negative controls for COX-2 and p53 protein. In addition, canine neoplasms with known antigen expression were used as positive immunohistochemical controls. Negative immunohistochemical controls were colon carcinomas treated identically as routine sections, with 20-minute antigen retrieval and 10-minute protein blocking, except the 30-minute incubation with primary antibodies was replaced with a 30-minute incubation with buffer.
Morphometry
Immunohistochemical expression by the neoplastic cells was assessed histologically. Each neoplasm was examined, and the percentage of neoplastic cells expressing each protein was estimated. Each tumor was assessed a score of 0 to 3. A score of 0 indicated that no altered protein expression was visible within the neoplastic cells. A score of 1 was assigned to tumors in which approximately 5% had altered protein expression, whereas a score of 2 was assigned if 10% of the neoplastic cells had altered expression. A score of 3 was assigned to tumors in which more than 10% of the cells had altered expression. Each tumor was scored 5 times, with good repeatability within the neoplasm scores. One hundred neoplastic cells were assessed for altered protein expression within tumors that were assigned a score of 3.
Statistical analysis
Correlations between the expression of the 4 proteins, as well as relations between abnormal protein expression and the presence of distant metastases were investigated by using analysis of variance techniques and linear regression models by using SPSS for Windows (SPSS Inc, Chicago, IL).
Results
All neoplasms developed within the jejunum and appeared as poorly defined firm white masses. All tumors followed a consistent pattern of growth and spread. The neoplastic cells markedly expanded the mucosa and the submucosa, as well as extending through the muscularis layer into the serosa. From the serosa, the tumors infiltrated the mesentery and extended within mesenteric adipose and lymphatics toward the adjacent mesenteric lymph node. The size of the neoplasms ranged from 0.5 to 7 cm. The appearance and the pattern of growth of ovine intestinal adenocarcinomas has been described. 24
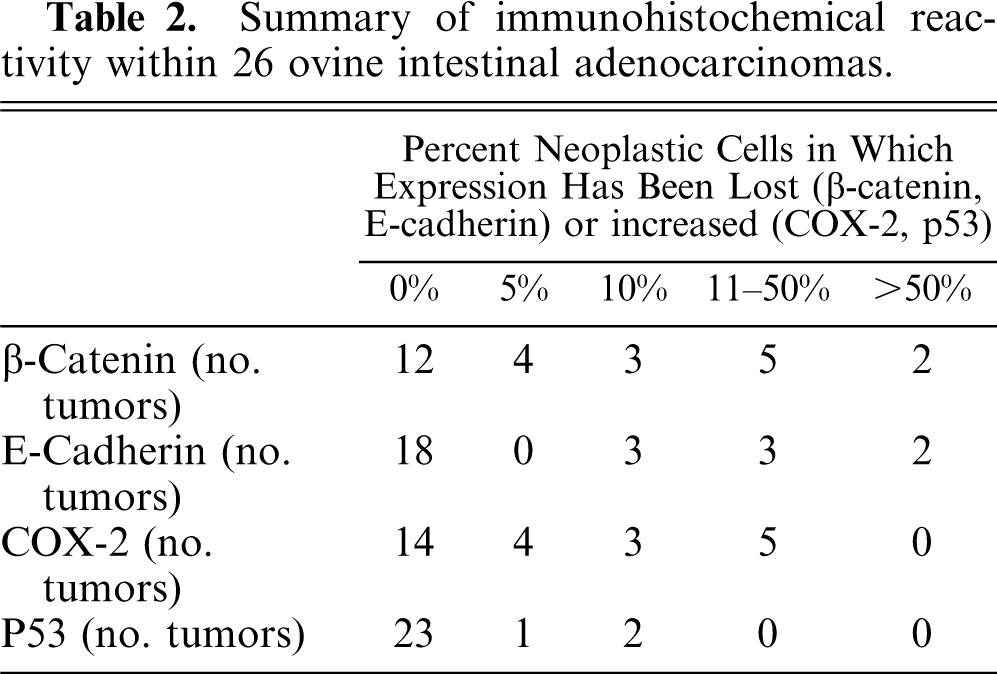
The results of the examination of individual tumors are included in Table 1. Non-neoplastic epithelial cells reacted with antibodies against β-catenin. This reactivity was intense and confined to the cell membranes (Fig. 1). Loss of membranous β-catenin was visible in 5% of the neoplastic cells in 4 neoplasms, whereas 10% of the neoplastic cells within 3 tumors had reduced staining. Between 11 and 50% of the neoplastic cells had lost membranous reactivity in 5 neoplasms, whereas 90 and 100% of the cells in tumor Nos. 19 and 24, respectively, had lost β-catenin expression (Table 2). Two tumors contained cells that had lost membranous β-catenin reactivity with concurrent cytoplasmic and nuclear β-catenin reactivity. This staining pattern was visible in 80% of the neoplastic cells without membranous β-catenin reactivity in tumor 12 and 60% of the neoplastic cells without membranous β-catenin reactivity in tumor 19 (Fig. 2). In total, neoplastic cells within 14 of the 26 ovine small-intestinal adenocarcinomas had reduced membranous β-catenin reactivity (Table 3). In some neoplasms, a higher proportion of cells with reduced membranous β-catenin reactivity were visible within the submucosa. However, this was not observed consistently and examination of larger numbers of neoplasms may be required to investigate the pattern of altered β-catenin expression within the neoplastic cells.
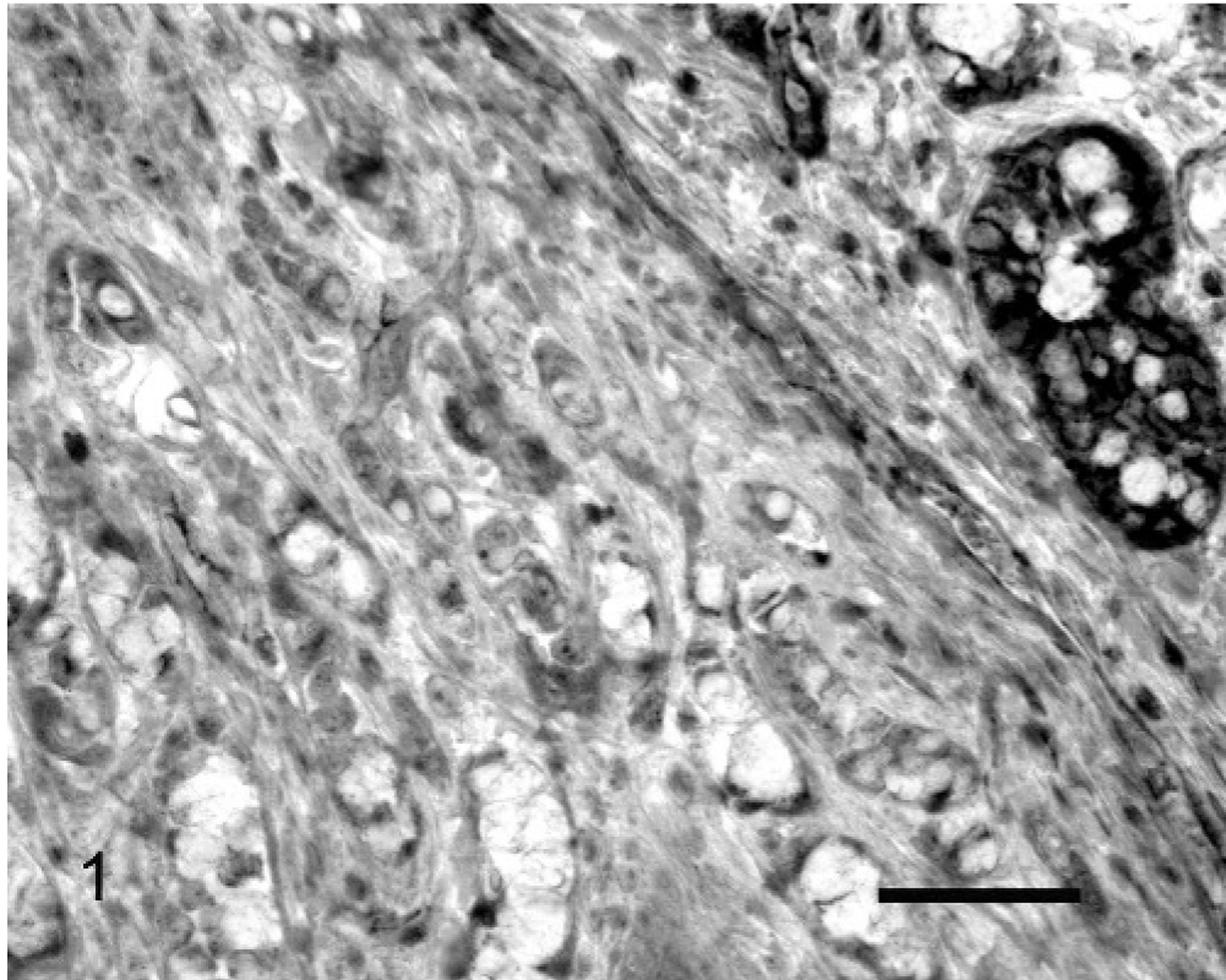
Small-intestinal adenocarcinoma; Sheep No. 24. Neoplastic intestinal epithelial cells did not react with antibodies against β-catenin. Residual non-neoplastic intestinal epithelial cells exhibit strong membranous reactivity (
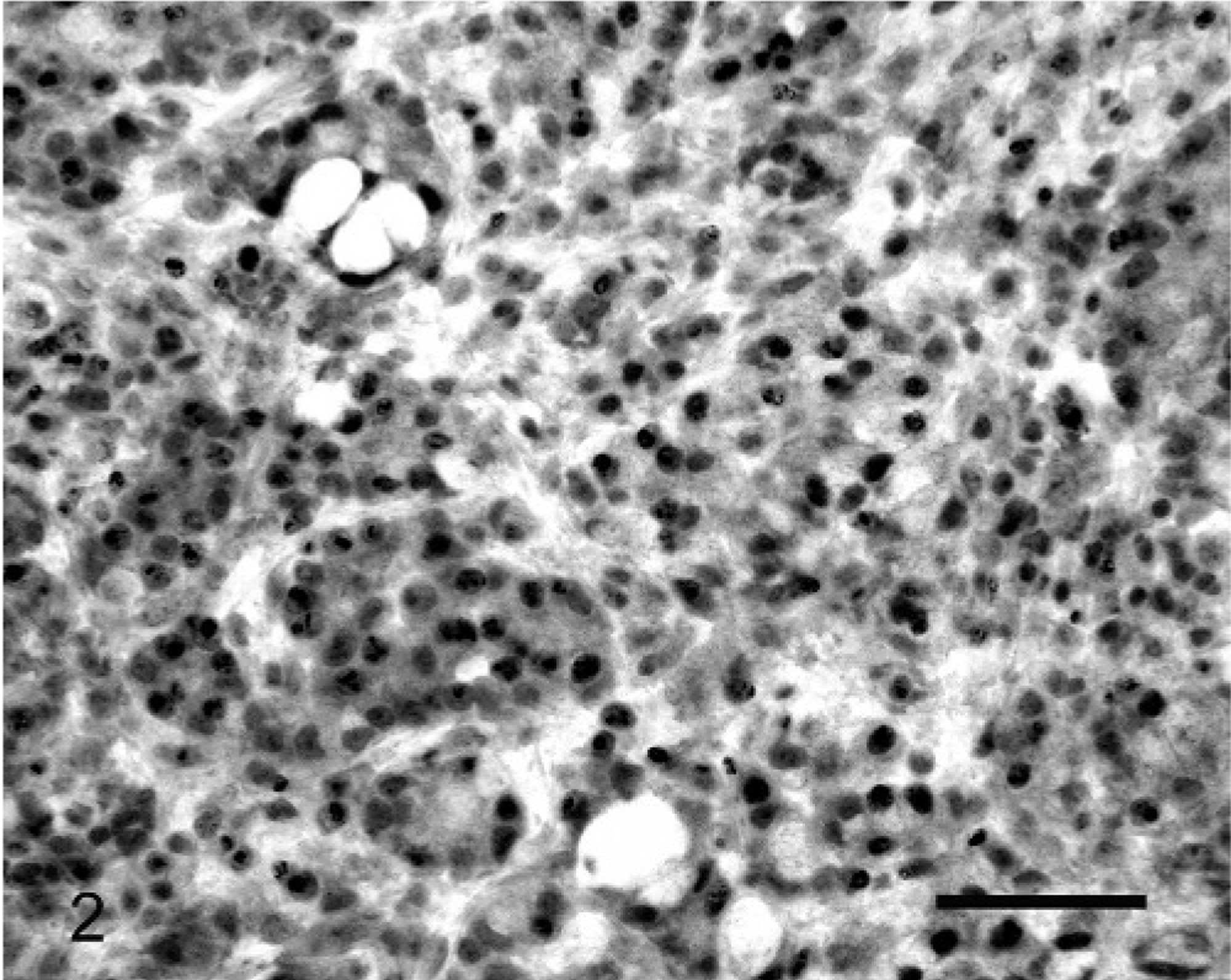
Small-intestinal adenocarcinoma; Sheep No. 19. Neoplastic cells showed strong nuclear reactivity with antibodies against β-catenin. Consistent weak cytoplasmic reactivity was also visible within the cell population. Membranous β-catenin reactivity has been lost within the cell population. Streptavidin-biotin-peroxidase complex system and Mayer's hematoxylin. Bar = 35 μm.
Immunohistochemical reactivity and metastases of ovine intestinal adenocarcinomas.
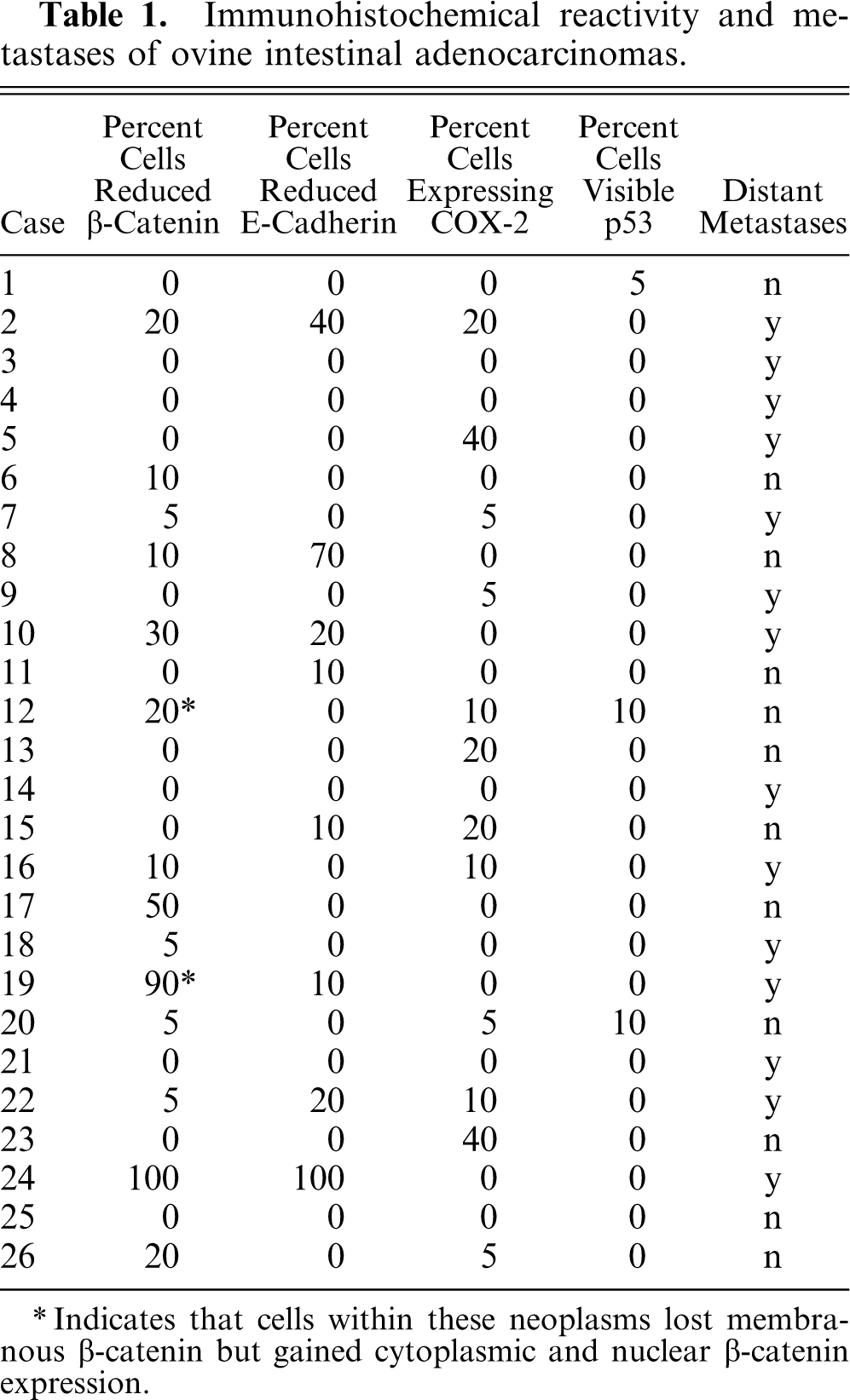
∗ Indicates that cells within these neoplasms lost membranous β-catenin but gained cytoplasmic and nuclear β-catenin expression.
Summary of immunohistochemical reactivity within 26 ovine intestinal adenocarcinomas.
Comparison of immunohistochemically-detectable alterations in cell protein expression within ovine intestinal and human colonic neoplasia.

The use of antibodies against E-cadherin revealed intense membranous reactivity in normal intestinal epithelial cells (Fig. 3). Neoplastic cells without E-cadherin reactivity were observed within 8 ovine small-intestinal adenocarcinomas. Loss of E-cadherin was visible in 10% of the neoplastic cells within 3 tumors, 11 to 50% of the cells in 3 tumors, and 70 and 100% of the neoplastic cells in tumor Nos. 8 and 24, respectively. No cytoplasmic or nuclear E-cadherin was observed in any of the neoplastic cells. Cells with reduced E-cadherin expression did not appear localized to either the superficial or deep aspects of the neoplasms. Loss of membranous E-cadherin reactivity was significantly correlated with loss of membranous β-catenin reactivity (R2 = 0.32, P = .003).
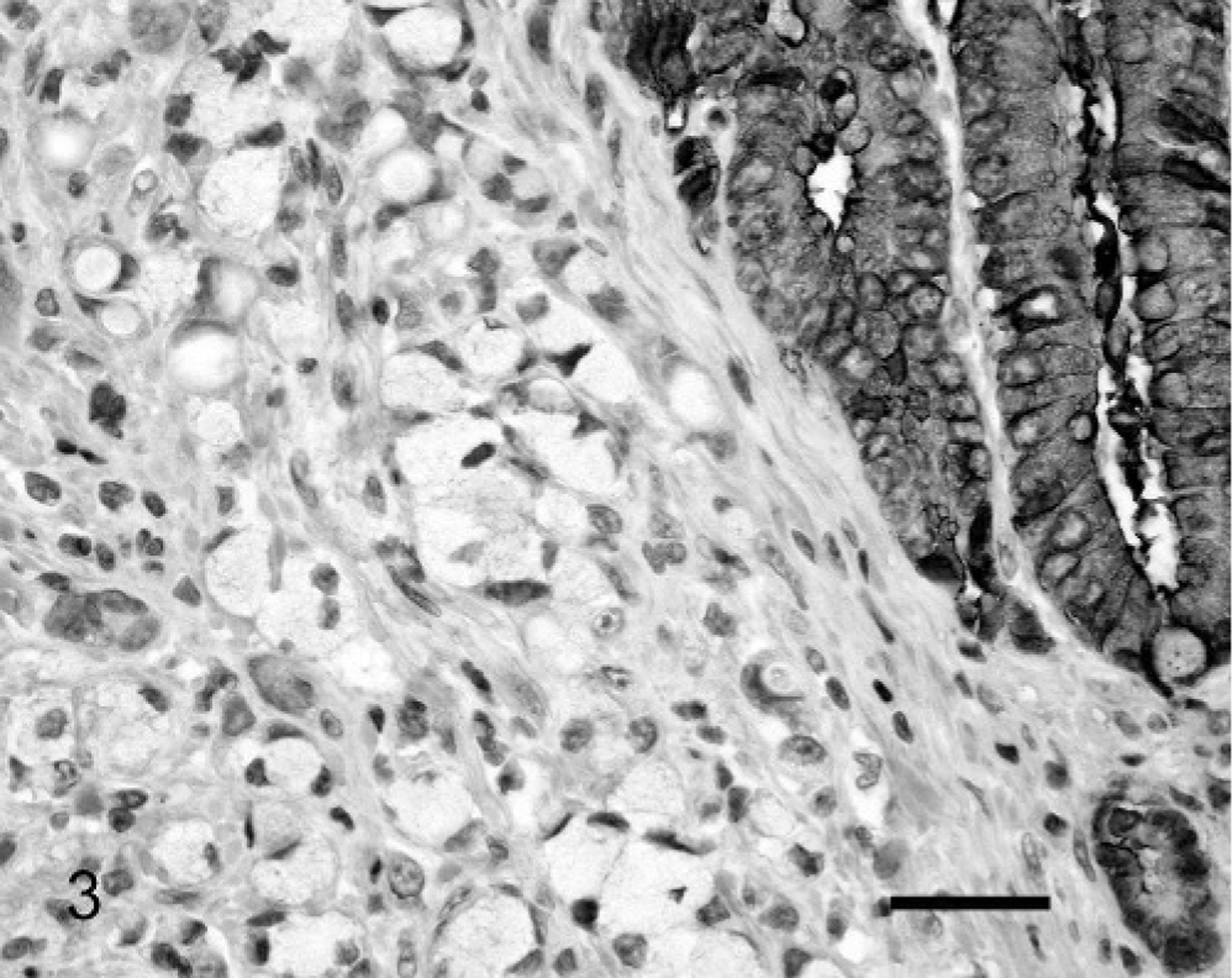
Small-intestinal adenocarcinoma; Sheep No. 24. Neoplastic intestinal epithelial cells did not react with antibodies against E-cadherin. Residual non-neoplastic intestinal epithelial cells exhibit strong membranous reactivity (top right). Streptavidin-biotin-peroxidase complex system and Mayer's hematoxylin. Bar = 25 μm.
Expression of COX-2 was consistently present within stromal cells within the lamina propria immediately underlying areas of ulceration. These cells were mononuclear, although they could not be definitively identified. Further investigation of these cells, as well as other components of the inflammatory response to the neoplasms, is being performed. Less consistently, COX-2 expression was observed within lymphocytes that were contained within well-defined submucosal lymphoid follicles. The development of lymphoid follicles appeared to be secondary to neoplasia-induced ulceration. Twelve of 26 ovine intestinal adenocarcinomas (46%) contained neoplastic epithelial cells that expressed COX-2 (Fig. 4). Of these, COX-2 was only observed in 5% of the neoplastic cells in 4 tumors, within 10% of the cells in 3 tumors, and within 11 and 50% of the cells within 5 tumors. Expression of COX-2 within these cells was confined to the cytoplasm. Cells expressing COX-2 did not appear localized to either the superficial or deep aspects of the neoplasms. Expression of COX-2 was not correlated with loss of β-catenin (R 2 = 0.06, P = .23) or E-cadherin reactivity (R 2 = 0.014, P = .56).
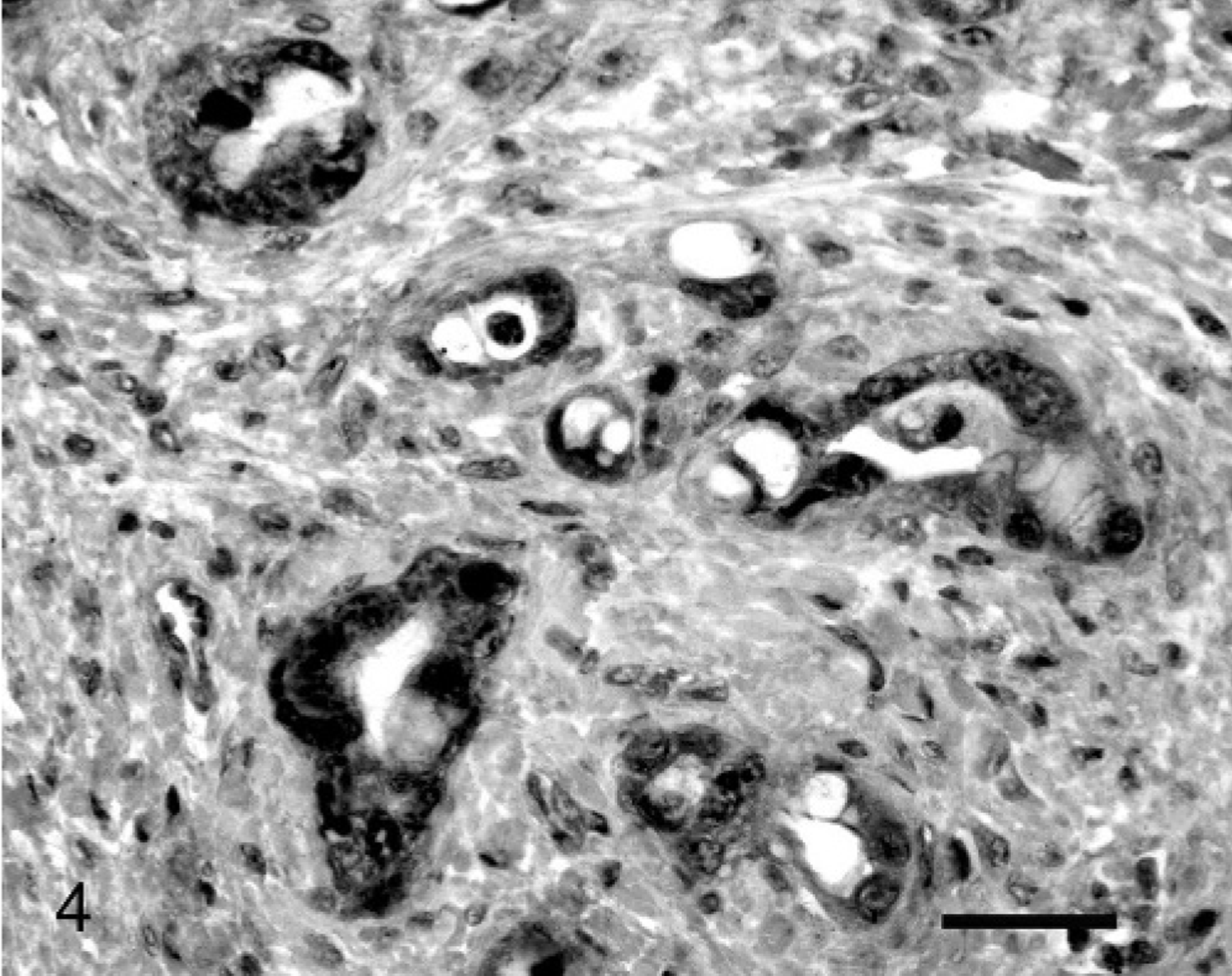
Small-intestinal adenocarcinoma; Sheep No. 5. Cytoplasmic reactivity with antibodies against COX-2 was visible within the neoplastic cell population. Streptavidin-biotin-peroxidase complex system and Mayer's hematoxylin. Bar = 27 μm.
Non-neoplastic tissue did not contain immunohistochemically visible p53 protein. However, cells with mild to moderate nuclear p53 protein immunostaining were visible within 3 (11%) of the neoplasms (Fig. 5). The number of cells in which p53 was visible was generally small, with only 5% of the neoplastic cells containing visible p53 protein in 1 neoplasm and 10% of cells containing p53 protein in 2 tumors. Because the numbers of cells visible that expressed p53 were small, no relation between protein expression and the location of the cell within the tumor could be determined. The presence of visible p53 protein was not correlated with COX-2 expression (R 2 = 0.07, P = .69) or with loss of expression of either β-catenin (R 2 = 0.03, P = .41) or E-cadherin (R 2 = 0.25, P = .44).
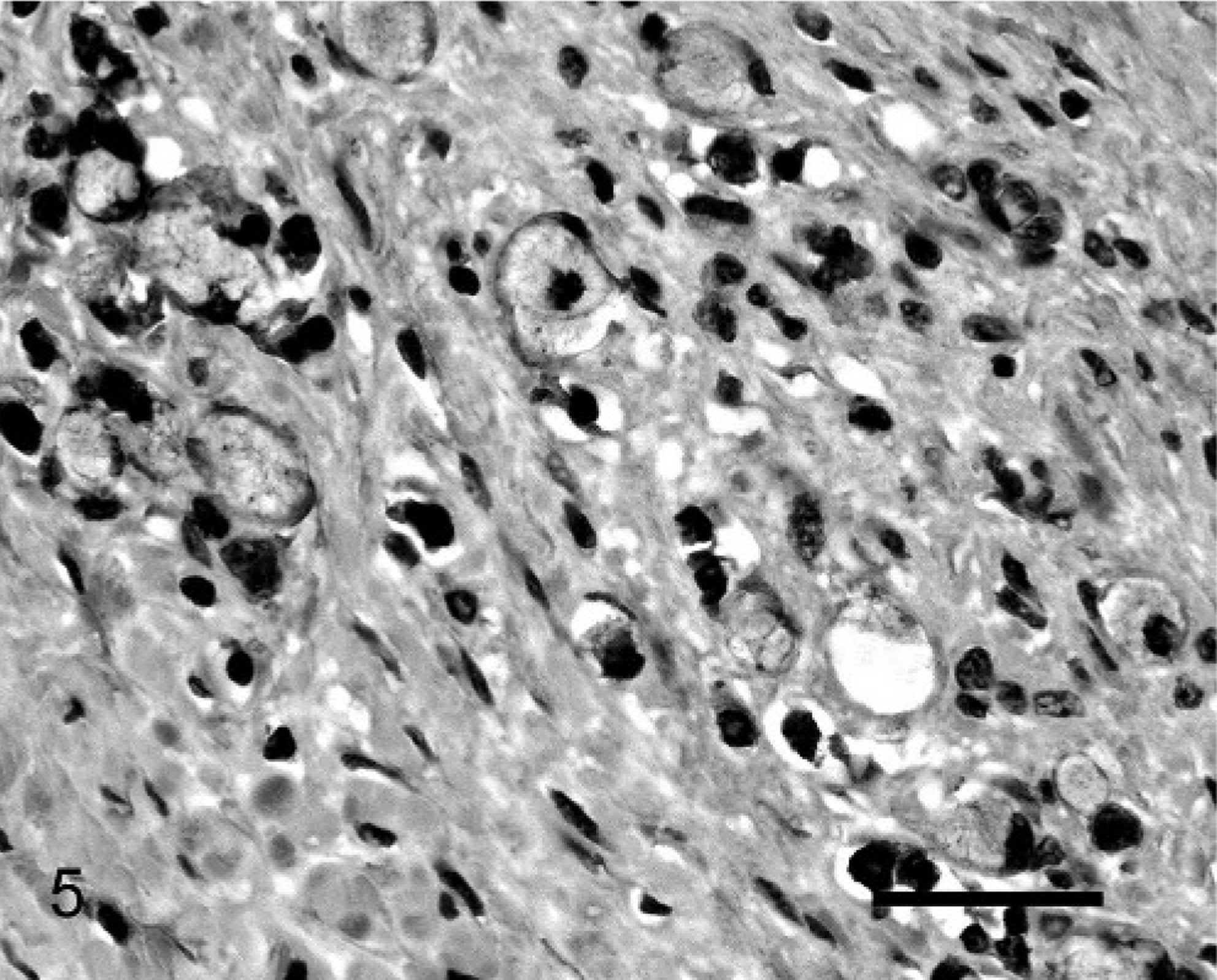
Small-intestinal adenocarcinoma; Sheep No. 20. Nuclear reactivity with antibodies against p53 protein was visible within the neoplastic cell population. Streptavidin-biotin-peroxidase complex system and Mayer's hematoxylin. Bar = 35 μm.
Metastatic spread to the adjacent mesenteric lymph node was identified in 22 of the 26 intestinal adenocarcinomas. Distant metastases were detected in 14 of 26 samples (54%). These metastases were most common within the peritoneal cavity but were also present within lymph nodes other than the mesenteric node adjacent to the neoplasm and the liver. There were no significant differences in loss of membranous immunostaining of β-catenin (P = .63) or E-cadherin (P = .88) or expression of COX-2 (P = .55) or p53 protein accumulation (P = .10) between tumors that developed distant metastases and those that did not.
Discussion
Loss of membranous β-catenin reactivity was visible in neoplastic cells in 54% of the ovine neoplasms. In comparison, immunohistochemistry revealed loss of membranous β-catenin reactivity in approximately 84% of human colonic adenocarcinomas. 15 A study of canine intestinal carcinomas revealed cells with abnormal β-catenin immunostaining in 5 of 19 tumors (26%). 22 Loss of membranous β-catenin was detected by using polymerase chain reaction analysis in APC-defective mice and in around 80% of azoxymethane-induced rodent neoplasms. 33
As well as anchoring E-cadherin to the cytoskeleton, β-catenin is also a component of the Wnt signalling pathway. 4 In a normal cell, β-catenin is rapidly degraded by the APC protein. 3 However, mutations within either APC or β-catenin genes can prevent β-catenin degradation. 3 This results in protein accumulation within the cell, nuclear entry of β-catenin, and activation of the Wnt signalling pathway. 3, 4 Products of this pathway include c-myc (a growth promoter), cyclin D1 (a cell-cycle regulator), and survivin (an apoptosis inhibitor). 4 Inherited APC gene mutations cause familial adenomatous polyposis in which large numbers of neoplasms develop within the colon. 21 In addition, mutations within the APC gene are present within 80% of sporadic colonic cancers. 4 Of the sporadic human colonic cancers that do not have APC gene mutations, about half have mutations of the β-catenin gene. 32
In a normal cell, β-catenin links the transmembrane protein E-cadherin to the cytoskeleton. 3 This function of β-catenin is responsible for its normal association with the cell membrane. However, cellular accumulation of β-catenin disrupts E-cadherin binding so that the localization of β-catenin to the membrane is lost. 3 Therefore, cellular β-catenin accumulation is expected to appear immunohistochemically as loss of membranous β-catenin, with increased cytoplasmic and nuclear β-catenin reactivity. This pattern has been reported in people, 2 dogs, 22 and mice. 34 In the examined ovine neoplasms, loss of membranous β-catenin with increased cytoplasmic and nuclear β-catenin reactivity was only visible in 2 tumors. It is considered possible that these 2 neoplasms contained cells with APC or β-catenin mutations that prevented cellular β-catenin degradation. In contrast, 12 ovine neoplasms contained cells that had lost membranous β-catenin but did not have visible cytoplasmic or nuclear β-catenin. It is possible that this staining pattern is more consistent with mutations within the β-catenin gene, preventing the production of an immunohistochemically reactive protein.
Loss of E-cadherin reactivity was visible in cells within 8 of 26 ovine small-intestinal adenocarcinomas (31%). Immunohistochemically detectable loss of E-cadherin has been reported in 44 and 31% of intestinal adenocarcinomas in people 10 and dogs, 22 respectively. Loss of immunohistochemically detectable membranous E-cadherin was reported in APC-mutant 30 but not chemically induced mouse intestinal tumors. 34
E-cadherin maintains adhesion between epithelial cells. 10 Loss of E-cadherin reduces cellular adhesion, enabling invasion and metastases by the neoplastic cells. 16 In addition, E-cadherin inhibits cell cycling so that reduced E-cadherin transcription promotes cellular proliferation. 39 In human colon-cancer cells, loss of E-cadherin often develops because of inhibition of gene transcription caused by increased cellular β-catenin concentrations. 5, 19 In the ovine intestinal adenocarcinoma cells, loss of E-cadherin expression was correlated with loss of membranous β-catenin expression. However, because increases in cellular β-catenin concentrations were rare within the ovine neoplastic cells, it appears that altered β-catenin expression reduced E-cadherin by a different mechanism. Within the cell, β-catenin anchors E-cadherin to the cytoskeleton. 3 Therefore, it is hypothesized that loss of functional β-catenin with the ovine neoplastic cells may have resulted in dissociation of E-cadherin from the cell membrane. Neoplasms Nos. 11 and 15 contained cells that had lost membranous E-cadherin but had immunohistochemically normal β-catenin reactivity. This could indicate that mutations within the E-cadherin gene may have been present in these 2 cases.
Neoplastic cell expression of COX-2 was visible within 12 of 26 of the ovine small-intestinal adenocarcinomas (46%). In addition, most sections contained stromal cells that expressed COX-2. However, this stromal expression appeared related to inflammation because of ulceration and necrosis rather than because of the presence of neoplastic cells. Immunohistochemically detectable COX-2 expression has been reported within 83% of human 14 and 47% of canine 23 intestinal adenocarcinomas. In human colon cancers, COX-2 expression has been reported both in the neoplastic epithelial cells, as well as in surrounding inflammatory cells, vascular endothelial cells, and fibroblasts. 27 Likewise, both stromal cell and neoplastic cell COX-2 expression was detected in canine intestinal carcinomas. 23 Expression of COX-2 is frequently reported in APC-mutant and carcinogen-induced rodent models of colorectal neoplasia. 28, 40 However, despite numerous experiments investigating the effects of COX-2 inhibitors in these models, there remains some uncertainty regarding which cell populations express COX-2. 17, 40
Epidemiologic studies suggest that inhibition of COX enzymes results in a 30 to 50% decrease in colon cancer risk. 40 In addition, inhibiting COX enzymes causes regression of colorectal adenomas in people with familial adenomatous polyposis. 36 Furthermore, COX enzyme inhibition reduced tumor development, progression, and metastases in most, but not all, rodent studies. 8, 35 The mechanism of COX-2 induction within neoplastic cells is not fully resolved. Unlike COX-1, which is found constitutively in tissues, COX-2 expression is induced by a variety of mediators, including inflammatory mediators, insulin-like growth factors, ras, and β-catenin. 35 Once expressed by a cell, COX-2 has been reported to promote cell proliferation, suppress apoptosis, promote angiogenesis, and inhibit the host antitumor immune response. 35, 40 It is hypothesized that these affects are mediated by increased prostaglandin E2 production. 35, 40 There was no correlation between COX-2 expression and β-catenin dysfunction within the ovine adenocarcinomas. This suggests that neoplastic cell COX-2 expression was induced independently of β-catenin.
Nuclear p53 protein was visible within cells in 11% of the ovine small-intestinal adenocarcinomas. Unlike wild-type p53 protein, mutated p53 protein has a long half-life within the cell, 9 so that immunohistochemically visible p53 protein is suggestive of mutations within the p53 gene. 9 Mutations within the p53 gene have been reported within approximately 50% of human colonic cancers 18 and 16 22 and 41% 38 of canine intestinal carcinomas. Mutations within the p53 gene are rare within chemically induced rat 31 or APC-mutant mouse 20 colorectal cancer models; however, intestinal neoplasia can be chemically induced in p53-mutant mice. 11
Missense mutations in the p53 gene are found in approximately half of all human neoplasms. 1 The p53 protein maintains genetic stability within a cell by regulating cell-cycle arrest, apoptosis, genetic integrity, and deoxyribonucleic acid repair. 1 In the ovine adenocarcinomas, immunohistochemically detectable p53 protein was not correlated with COX-2 expression or with loss of β-catenin or E-cadherin. This suggests that nuclear p53 accumulation developed independently of dysfunction of the other 3 proteins examined in this study.
Human studies of colorectal cancer have found that cellular accumulation of β-catenin 2 or loss of E-cadherin 10 increases the risk of developing metastatic disease. Because of inconsistent data, it is currently uncertain whether COX-2 35 or p53 25 mutations influence the development of metastatic disease. Alteration of none of the 4 proteins examined in this study was predictive of distant metastases. However, it is unlikely that the number of samples used in this study was sufficient to investigate this correlation.
Abnormal expression of β-catenin is reported within 84% of human colorectal carcinomas 15 and abnormal β-catenin expression is considered a key component of human colorectal carcinogenesis. 3 Although altered β-catenin expression was observed in 54% of the ovine neoplasms, only 27% of the neoplasms contained altered expression within more than 10% of the neoplastic cells. Likewise, rates of altered E-cadherin, COX-2, and p53 protein expression were lower than those reported in humans. The reason for these lower rates is uncertain but could be because of less frequent mutations within the ovine neoplastic cells, less sensitive methods of detection, or sampling of less advanced lesions. If lower rates of mutations are present within the ovine neoplasms, this may detract from their use as an animal model.
Slaughterhouse studies of normal older sheep in New Zealand reveal small-intestinal adenocarcinoma incidence rates of 1.6%. 29 When older, unthrifty sheep are selected, 7% are expected to have intestinal adenocarcinomas. 6 Ovine intestinal adenocarcinomas rapidly metastasize to the mesenteric lymph nodes and then widely within the body. 24 The histologic appearance and biological behavior of these neoplasms show some similarities to colonic cancer in people. 24 To the investigators' knowledge, there is no currently used animal model of human colonic cancer that spontaneously develops tumors that consistently develop metastases. It is hypothesized that sheep may provide a large-animal model of spontaneous metastatic intestinal cancer. Such a model may allow evaluation of new chemotherapeutics, surgical techniques, or novel treatments, e.g., immunotherapy. However, for an animal model of human cancer to be useful, particularly when considering response to treatments, similar genetic defects should be present within the neoplastic cells of both species. Abnormal expression of all 4 proteins investigated in this study was identified within the ovine intestinal adenocarcinoma cells. All 4 exhibited abnormalities at rates comparable, albeit lower, with those reported in human colonic cancers. The results of this study provide additional evidence that sheep may be an appropriate model of human colonic cancer.
In conclusion, 26 ovine intestinal adenocarcinomas were investigated for altered protein expression by using immunohistochemistry. The 4 proteins examined in this study were selected because of their frequent abnormal expression in human colonic neoplasms. Loss of membranous β-catenin was observed in 54% of tumors, E-cadherin was lost in 31%, COX-2 was expressed in 46%, and p53 protein was visible in 11% of neoplasms. While these rates are generally lower than corresponding rates reported in human colonic neoplasms, they suggest that similar alterations in protein expression may be present in both human colonic and ovine small-intestinal adenocarcinomas. The presence of similar altered protein expression within neoplasms from both species suggests that sheep may provide a suitable model to investigate novel treatments of advanced human colonic cancer.
