Abstract
A 7-year-old dairy sheep suffering from chronic loss of weight without diarrhea or anorexia was euthanized after failing to respond to any treatment (antibiotic and antiparasitic). The main findings at the necropsy of this animal were multifocal miliary nodules in several organs, mainly in the Peyer’s patches of the small intestine, and a segmental thickening of the jejunal wall. Histologic examination of the samples taken at the necropsy showed a multifocal chronic granulomatous inflammation, with mineralization and caseous necrosis at the core of the larger granulomas and scarce intrahistiocytic acid-fast bacilli consistent with a disseminated digestive tuberculosis. Polymerase chain reaction and bacteriological culture from these samples confirmed Mycobacterium avium subsp avium to be the etiologic agent of this infection. Histologically, the cause of the segmental thickening of the jejunal wall was found to be a small intestine adenocarcinoma, which in some areas coexisted with the granulomatous lesion.
A 7-year-old Churra ewe with poor body condition and a clinical history of chronic weight loss, in the absence of diarrhea or anorexia, was humanely euthanatized by an overdose of intravenous barbiturate because of unresponsiveness to symptomatic treatment (ivermectin and marbofloxacin). The ewe belonged to an intensively managed flock for milk production of approximately 1500 sheep and was kept indoors most part of the year, although occasional periods of daily grazing were allowed during the dry period of the animals. This was a sporadic case, and the health of the flock was considered to be good, above the average, by both the farmer and the veterinary clinician who treated the animal.
During the necropsy, the most striking finding was the presence inside the gut of multiple gray-yellowish miliary nodules, visible through the intestinal wall even before opening it (Fig. 1), and distributed in discrete groups throughout the length of the small intestine. When the mucosa of the intestine was exposed, it was clear that these lesions affected, with variable intensity, all the jejunal Peyer’s patches. Besides the small intestine, these nodules were also found disseminated in the lungs, liver, and enlarged mediastinal and mesenteric lymph nodes.
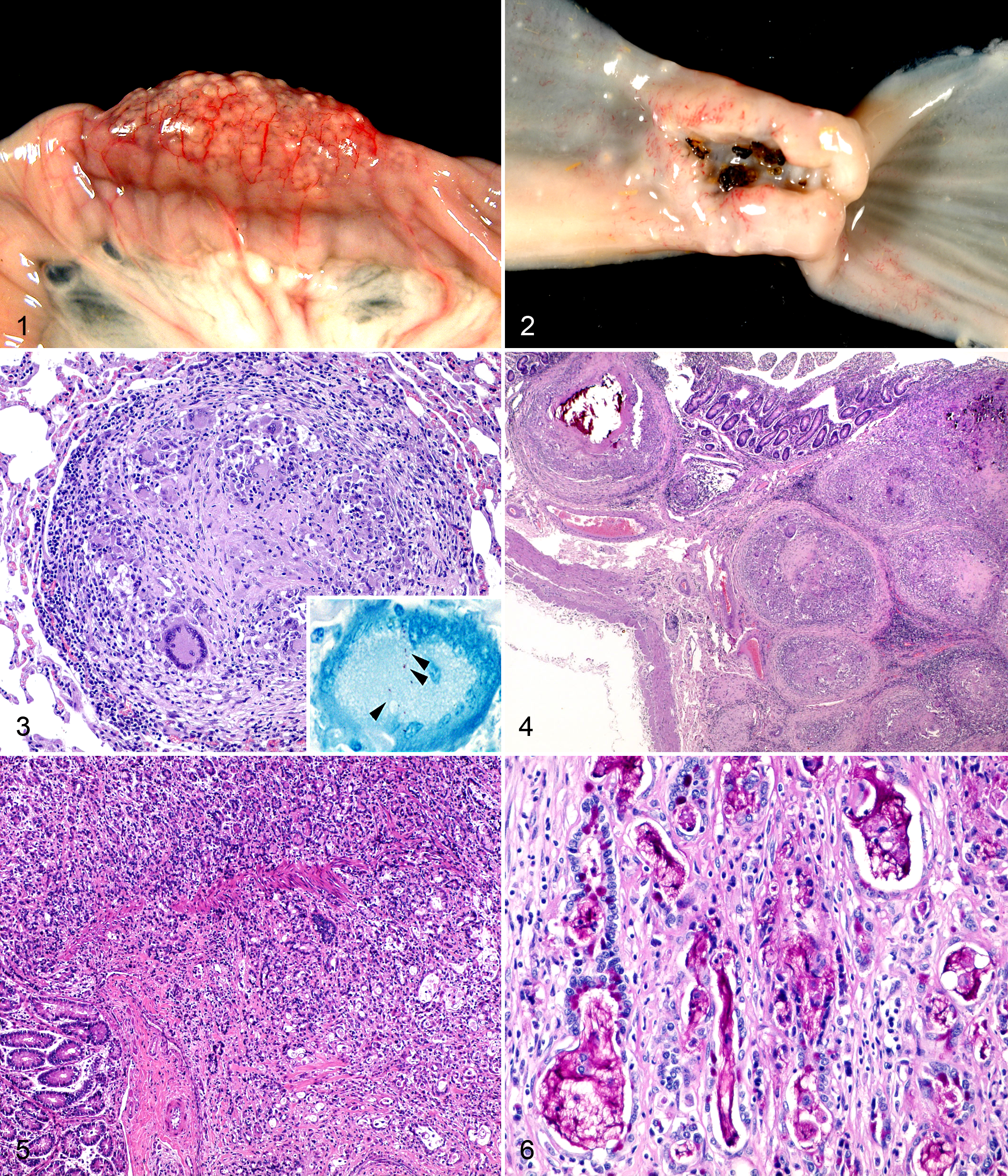
There was also a segmental dilation of several intestinal loops at the middle area of the jejunum. The intestinal wall of this area was pale and uniformly enlarged, and it showed fibrous adherences of the serosa between the affected loops. When longitudinally opened, this area showed an annular stenosis encircling the intestine and caused by the focal overgrowth of firm, white, fibrous-rich tissue at the anterior extreme of the segment (Fig. 2). The wall of the intestine cranial to the narrowing showed no gross abnormalities other than the nodules at the Peyer’s patches already described.
Differential Diagnoses
Chronic emaciation in an adult sheep can be caused by several conditions. When coughing or breathing distress is present, a chronic bacterial or parasitic pneumonia, or ovine pulmonary adenocarcinoma, could be suspected. In older sheep, especially in those grazing, dentition problems, such as broken mouth, could force the animal to stop eating or reduce the amount of ingested food. However, none of these clinical signs were observed in the animal.
In the northwest of Spain, where most of the sheep flocks are intensively managed for milk production, with a highly controlled diet, slow diseases, such as maedi-visna or paratuberculosis, are the most common causes for chronic loss of weight without diarrhea or anorexia. Gastrointestinal parasites or hepatic trematodes could also cause chronic wasting disease in sheep, especially in those cases of antihelmintic resistance. Besides these infectious diseases, any slow-growing malignant tumors would also cause chronic emaciation.
The necropsy of this animal was highly informative and showed clear lesions, mainly in the intestine, but also affecting other viscera. The multifocal miliary nodules present in the intestine, lung, and liver were suggestive of the hematogenous spread of a chronic infection causing a granulomatous inflammation, such as mycobacteriosis, fungal (aspergillosis), or parasitic (Oesophagostomum spp) infections, although the latter was not considered probable due to geographical distribution of the parasite (not common, although present, in Europe) and the localization of the nodules (in oesophagostomiasis, they are randomly distributed in the intestine rather than focused on the Peyer’s patches). The segmental thickening of the intestinal wall at the jejunum and the focal stenosis at the cranial end of this lesion were highly suggestive of an intestinal adenocarcinoma, a relatively frequent tumor of sheep.
Microscopic Findings
The histologic analysis of the miliary nodules showed them to be granulomas surrounded by connective tissue and formed by abundant Langhans giant cells and epithelioid macrophages, with few neutrophils and lymphocytes at the periphery (Fig. 3). At the core of the largest granulomas, there was caseous necrosis and mineralization. Ziehl-Neelsen stain at selected sections showed very few acid-fast bacilli within the giant cells of the granulomas (Fig. 3). These granulomas were mainly present in the small intestine and mainly located at the Peyer’s patches, where they almost entirely replaced the lymphoid tissue. From the submucosa, they spread to the lamina propria and muscular layers, even to the serosa (Fig. 4). When lesions at the intestine were present in areas not related to lymphoid tissue, these appeared as isolated small granulomas or giant cells within the lamina propria. Granulomas similar to those observed at the Peyer’s patches were found at the cortical area of mediastinal and mesenteric lymph nodes, liver, lung (Fig. 3), spleen, kidney, and mammary gland. In the kidney, there was a pale, eosinophilic, homogeneous deposit, positive to Congo Red stain (amyloid), in the tubulointerstitium and a number of glomerular tufts that replaced a portion, of variable size, of the normal glomerular architecture and produced tubular atrophy.
Histologic examination of the samples from the pale, fibrous annular thickening of the jejunum showed a poorly demarcated, tightly packed infiltrative mass formed by epithelial cells arranged in rows, tubules, or glandular, seldom cystic, structures and supported by an abundant fibrous stromal tissue. The neoplastic cells infiltrated all the layers of intestinal wall and were moderately differentiated, large, and polyhedral, with variable distinct borders and a large round nucleus with loose stippled chromatin (Fig. 5). These cells showed low pleomorphism, and rare mitotic figures per high-power field were found. Some of these cells showed a signet-ring morphology where the nucleus was found flattened and displaced to the periphery by 1 or few variably sized clear, foamy vacuoles that were shown to contain mucin by the periodic acid-Schiff (PAS) stain. Tubules, glandular acini, and cystic structures often contained cellular debris and variable amounts of a foamy, amphophilic material, positive to PAS stain (mucin) (Fig. 6). In some areas, probably of Peyer’s patches, the neoplastic cells intermingled with numerous granulomas, causing a complete loss of the normal architecture of the tissue (Suppl. Fig. S1).
Laboratory Findings
Fresh samples from mesenteric lymph node and lung were homogenized and processed for mycobacterial culture. After 8 weeks of incubation, some colonies were observed in tubes of Coletsos medium. Ziehl-Neelsen staining of colony smears showed typical acid-fast bacilli. No growth was observed for 16 weeks in any of the Löwentein-Jensen and Herrold medium tubes.
Molecular methods were used to identify the colonies obtained. Although the Mycobacterium bovis–Mycobacterium tuberculosis complex specific polymerase chain reaction (PCR) was negative, the PCR–restriction endonuclease analysis confirmed the identity as a M. avium subspecies different from paratuberculosis that was definitely confirmed as avium subspecies by the PCR-positive result for IS901.
Diagnosis
The histopathologic diagnosis was severe, multifocal, chronic granulomatous enteritis and lymphadenitis with intrahistiocytic acid-fast bacilli, consistent with digestive tuberculosis, moderate renal amyloidosis, and small intestine adenocarcinoma (SIA).
As a result of the hematogenous spread of the mycobacteria, there was also mild to moderate, multifocal, chronic granulomatous hepatitis, pneumonia, splenitis, and mastitis with intrahistiocytic acid-fast bacilli, consistent with disseminated tuberculosis.
Final confirmation and definitive etiological diagnosis of M. avium subsp avium infection was supported by bacterial culture and endonuclease analysis of the PCR amplification product.
Discussion
The 3 conditions diagnosed in this animal (ie, avian tuberculosis, amyloidosis, and small intestine adenocarcinoma) are considered to be of rare, or relatively rare, occurrence in sheep. The singularity of this case is that, besides being rare, the 3 of them were simultaneously diagnosed in the same animal.
M. avium subsp avium is an ubiquitous opportunist pathogen that often causes disease in birds but only seldom in mammals. 14 Small ruminants are considered to be resistant to avian tuberculosis, particularly sheep, although few cases of natural and experimental disease have been described. 2 The distribution of the lesions in this animal, specifically the clear predilection for the Peyer’s patches, is very rare when compared with previous descriptions of avian tuberculosis in mammals where lesions are found in the thoracic cavity and lymph nodes only. 14 In this case, such frequent occurrence of lesions within the intestine would suggest a digestive route of infection, as it has been shown to occur under experimental conditions, 2 and is regarded as a common route of infection in mammals. 4 These histologic features of the inflammatory reaction (ie, fibrous encapsulation and tendency toward caseous necrosis and mineralization) are not considered to be frequent in M. avium infections, 14 although they have been previously described in avian tuberculosis of sheep and goats. 1,2 This may suggest variations in the inflammatory reaction depending on the species or an intense reaction against an infection not common in these species. The scarcity of acid-fast bacilli within the lesions and the granulomatous response seen in this animal, especially the tendency toward encapsulation, caseous necrosis, and mineralization, are features associated with a better containment of tuberculosis progression. 4 Amyloid deposition in sheep is mainly secondary to a chronic inflammatory disease or neoplasia. Primary amyloidosis, although the most common form of systemic amyloidosis in humans, is very rare in domestic animals, having not been reported affecting sheep. 15 Moreover, amyloid deposition in the kidney is a common finding in domestic animals suffering from a chronic inflammatory disease, such as tuberculosis. 11
Bearing in mind that SIA is the most frequent noninfectious neoplasia in sheep 6 and that it has been already reported in this region of Spain, 13 it is likely that the diagnosis of these 2 rare conditions in the same animal is a coincidence. However, the etiology of SIA is not clear yet, and several hypotheses, such genetic predisposition or carcinogens ingestion, have been proposed to explain such high prevalence of this neoplasia. 6,10 Infectious agents has been proposed to be a significant carcinogen to humans, 12 but previous studies have failed to find a relation between SIA. 9 However, some studies have discussed the possible relation between the occurrence of colon carcinoma in humans, a neoplasia similar to ovine SIA, 8 and infectious diseases. 3 Inflammation promotes a highly reactive cellular microenvironment capable of causing genomic instability and DNA mutations. Thus, the longer the inflammation persists, such as in chronic inflammatory diseases, the higher the probability for this mutation to occur. These changes could then lead to the initiation and promotion of cancer. 7
On the other hand, patients with cancer show increased susceptibility to infections due to several factors, such as disturbances to host immunity, tissue necrosis, or mechanical obstructions. 5 It seems logical to hypothesize as well that, notwithstanding any of those factors, the poor body condition or malnutrition showed by this animal, and usually present in sheep with SIA, 6,13 could have favored the establishment of infectious conditions or the reemergence of such infections from a latent, controlled state because of an inadequate immune status of the host. It is possible then that the disseminated tuberculosis found in this sheep could have appeared as an opportunistic infectious consequence of SIA.
Footnotes
Acknowledgements
Thanks are due to Gloria Belver and Jaime Reyero for technical assistance and also to the farmer and veterinary clinician concerned for their assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: J. Benavides is supported by CSIC through the JAE-Doc program, financed in part by the European Social Fund.
