Abstract
An 18-y-old female tufted deer (Elaphodus cephalophus) had a short history of chronic diarrhea, progressive weight loss, and hindlimb instability. Given the poor prognosis, the deer was euthanized and submitted for postmortem examination. The most significant gross finding was segmental and multinodular mural thickening of the proximal colon. On cut surface of the affected colonic segments, 0.5–2-cm diameter, intramural, multiloculated, cystic structures containing gray, translucent, gelatinous material elevated the edematous mucosa. Microscopically, the intramural cystic structures were filled with mucinous matrix admixed with foamy macrophages, and lined by discontinuous segments of well-differentiated columnar, pancytokeratin-positive epithelium with basilar nuclei. Multifocally, transition was observed from hyperplastic mucosal crypt epithelium to dysplastic or neoplastic columnar and flattened epithelium lining submucosal and serosal cysts. Cyst lumina were irregularly disrupted by polypoid ingrowths of collagenous tissue covered by attenuated epithelium. Based on these findings, we diagnosed a well-differentiated mucinous adenocarcinoma. Although intestinal adenocarcinomas have been described in humans and animals, they are considered uncommon in most domestic species, except for sheep, for which genetic and environmental factors appear to influence occurrence. Our report addresses the knowledge gap regarding intestinal adenocarcinomas affecting cervids and specifically the tufted deer, a less-studied, near-threatened Asian cervid.
An 18-y-old female tufted deer (Elaphodus cephalophus) from a zoologic collection had a history of diarrhea, hind limb instability, and weakness that lasted for several weeks. Given the poor prognosis, the deer was euthanized and submitted for autopsy to the California Animal Health and Food Safety Laboratory (CAHFS; San Bernardino Laboratory, UC Davis). The carcass was in a fair state of postmortem preservation and in poor nutritional condition, with generalized muscle wasting and serous atrophy of fat. Perianal fur was stained with dried green feces. The proximal loop of the ascending colon had two, 5–7-cm long segments of soft, multinodular, mural thickening (Fig. 1). On cut surface, and causing mural expansion in both segments, were 0.5–2-cm diameter, submucosal and serosal, sessile multiloculated cystic structures containing gelatinous, gray, translucent material (Fig. 2). Given the history of hindlimb weakness and instability, the hindlimbs were subjected to a detailed musculoskeletal examination, including major joints (stifle, hock), major peripheral nerve groups (sciatic, ischiatic), and musculature. The only findings in the hindquarters were deep, multifocal hemorrhages in the dorsal aspect of the left vastus lateralis and biceps femoris muscles, and diffuse dark-purple discoloration of left psoas major and minor muscles. The lumbosacral vertebral column was split sagittally from L1 to S5; no significant gross findings were detected.
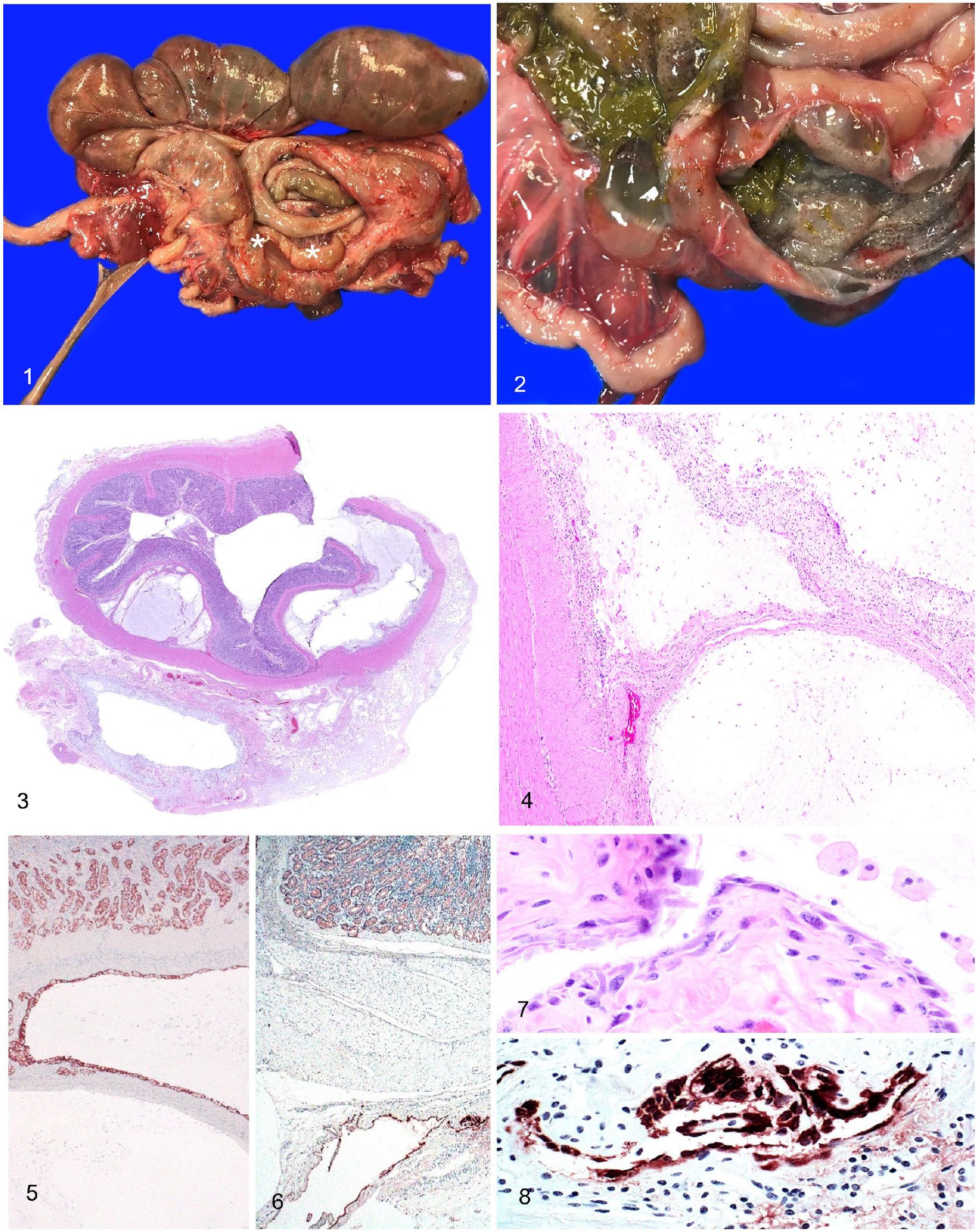
Colonic mucinous adenocarcinoma in a tufted deer.
Samples from brain, spinal cord, peripheral nerves (ischiatic, sciatic), trachea, thyroid gland, lung, heart, liver, spleen, kidneys, urinary bladder, adrenal glands, tongue, esophagus, reticulum, rumen, omasum, abomasum, intestine, diaphragm, and axial and appendicular musculature, including psoas major and minor muscles, were fixed in 10% neutral-buffered formalin for 24–72 h, and processed routinely to produce 4-μm thick sections stained with H&E. Additional histochemical stains (Alcian blue, periodic acid–Schiff [PAS]) and immunohistochemistry (IHC; pancytokeratin, human papillomavirus [Alphapapillomavirus]) were done according to CAHFS standard laboratory procedures.
Microscopically, the proximal portion of the colon was segmentally lined by hyperplastic to dysplastic mucosal epithelium. Additionally, the submucosa was multifocally expanded by numerous, discrete-to-interconnected cystic spaces containing mucinous material (Fig. 3). Some submucosal cysts were discontinuously lined by columnar epithelium with basal nuclei and markedly vacuolated cytoplasm, and, in at least one cyst, the epithelium appeared to be contiguous with the mucosal crypt epithelium through the muscularis mucosae. Infrequent connections were also observed between the submucosal and serosal cysts, via the focally disrupted tunica muscularis. The serosal cysts expanded the serosa multifocally (Figs. 3, 4). Positive immunolabeling to pancytokeratin IHC was observed within cells segmentally lining the submucosal cysts (Fig. 5) and fewer serosal cysts (Fig. 6). Single-to-layered aggregates of macrophages and multinucleate giant cells with abundant foamy cytoplasm floated within the cysts. The serosal cyst epithelium has a high nuclear:cytoplasmic ratio and mild anisocytosis and anisokaryosis (Figs. 7, 8). No mitotic figures were observed in 2.37-mm2 examined fields. The overlying, non-neoplastic mucosa was intact in all examined sections of the colon. IHC for human papillomavirus (cross-reactive with bovine papillomaviruses) on intestinal sections was negative. In addition, both the mucosal epithelium and cyst-lining epithelium stained positively with Alcian blue and PAS, indicating the presence of acid epithelial mucins, which are characteristic of the gastrointestinal (GI) tract epithelium.
Extraintestinal microscopic findings included mild intimal and medial vascular mineralization in the cerebellum, mild periglomerular and interstitial fibrosis and mild tubular degeneration with protein casts in the kidney, minimal Wallerian degeneration of the ischiatic and sciatic nerves, depletion of splenic peri-arteriolar lymphoid sheaths, and filamentous microorganisms in the stratum corneum of the ruminal epithelium. No significant microscopic abnormalities were observed in any of the other tissues examined.
Aerobic cultures of the small intestine, lung, and liver, and anaerobic cultures of the cecum were performed. Klebsiella pneumoniae was isolated in moderate and rare numbers from the small intestine and lungs, respectively. No bacteria were isolated by aerobic culture of liver or anaerobic culture of cecum. Salmonella sp. PCR performed on small intestinal contents and liver was negative. Mycobacterium avium subsp. paratuberculosis PCR performed on colonic contents was also negative. No endoparasites or parasite eggs were detected by the fecal flotation test.
Descriptions of spontaneous GI adenocarcinoma are fairly common in veterinary literature, but they are mostly limited to dogs and cats. Available reports of intestinal adenocarcinoma in small animals indicate a frequency of 8–10% of all tumors,12,24,39 surpassed only by lymphoma.11,51 The fairly high frequency of canine spontaneous intestinal neoplasia has even made colorectal tumors in this species suitable models for their human counterparts. 40 The uncommon occurrence of intestinal adenocarcinomas in other domestic species, including Vietnamese potbellied pigs10,25 and goats, 36 is also reported. The prevalence of intestinal tumors in horses and cattle, as a percentage of the total number of neoplastic diseases, has been reported to be <1% and 0.5–1.0%, respectively.31,34,43,46 In these species, intestinal adenocarcinomas are the most common non-lymphoid tumor.4,39,42,50
In contrast, sheep are the only domestic animal species in which a relatively higher frequency of intestinal neoplasia is reported. Intestinal adenocarcinoma is one of the top 3 most common ovine neoplasms (along with ovine pulmonary adenocarcinoma and enzootic nasal adenocarcinoma), with a prevalence of 2.9–6%.21,45 Small intestinal involvement in sheep is frequent and is strongly correlated with geographic region, given the high occurrence in New Zealand and Australia. Suggested predisposing factors for intestinal adenocarcinoma in sheep are old age, dietary carcinogens, pastures with heavy fertilizer and herbicide use, genetics (e.g., meat-producing breeds), oncogenic viruses, bracken fern (Pteridium aquilinum), and crested dog’s-tail (Cynosurus cristatus).26,30–32,45,46
There are a few reports of intestinal adenocarcinoma in wildlife species, mostly focused on nonhuman primates (rhesus macaques, common marmosets),29,47 snakes,19,41 amphibians,22,48 foxes, 8 seals, 28 opossums, 38 and fish,6,13 with a possible infectious etiology in fish. In nonhuman primates, dysplasia and carcinoma of the distal portion of the colon have been reported in captive cotton-topped tamarins (Saguinus leucopus). An association with environmental factors in captivity (e.g., brain-gut neuroimmune interactions) has been proposed, given that these conditions are virtually absent in the wild. 52 Generally, clinical signs of colonic adenocarcinoma in all species include large-bowel diarrhea, straining on defecation, hematochezia, and ascites secondary to serosal lymphatic obstruction by tumor emboli.15,35,44,46
Intestinal adenocarcinomas are frequently characterized by slowly evolving invasion of the intestinal wall beyond the muscularis mucosae by neoplastic epithelial cells, accompanied by a marked annular scirrhous reaction. Despite some of these neoplasms being well-differentiated, as in our case, intestinal adenocarcinomas are appropriately diagnosed as carcinomas given their transmural invasive behavior. 46 Ovine intestinal adenocarcinomas are markedly infiltrative and metastasize frequently to regional lymph nodes. 46 Regional lymph nodes in our case were grossly unremarkable; however, in the absence of histologic examination, metastasis to alimentary lymph nodes cannot be fully excluded. On the other hand, distant, extraintestinal metastasis and intra-abdominal dissemination corresponding to peritoneal carcinomatosis (as observed in clinically healthy sheep at slaughter) can be safely ruled out because there was no gross or histologic evidence of either.30,46
In our case, there was no significant scirrhous reaction surrounding the neoplasm. Although a scirrhous reaction is considered to be characteristic of intestinal adenocarcinomas, 46 this reaction is usually more marked in the small intestine than in the colon, and its occurrence is less common in well-differentiated tumors. 46 We speculate that the lack of extraintestinal metastasis and ascites in our case could be attributed simply to having a well-differentiated tumor that is much less likely to induce metastatic spread or blockage of lymphatic circulation compared to an annular scirrhous and highly invasive adenocarcinoma.
In our case, regional lymph nodes were grossly unremarkable; however, none were examined histologically, limiting our ability to rule out an early metastatic process. In dogs, survival is reduced in individuals with annular colorectal carcinomas compared to those with pedunculated polypoid carcinomas in the same location 46 ; perhaps the former have a greater degree of invasion, facilitating spread. In our case, the neoplasm occupied the entire circumference of the intestine, yet no extraintestinal metastases or ascites were observed. It is possible that the deer was euthanized before the development of metastatic spread.
Histologic patterns of intestinal adenocarcinoma include acinar, solid, papillary-tubular, mucinous, and mixed-type.18,35 All histologic types are considered malignant neoplasms, and no prognostic significance has been attributed to the histologic type.30,45 Mucinous intestinal adenocarcinoma occurs infrequently in humans and cats, with no published records in sheep or cattle.23,33,34,43
Intestinal adenocarcinoma has been induced experimentally and occurs naturally in immunosuppressed cattle with concurrent bovine papillomavirus 4 infection and exposure to bracken fern (Pteridium aquilinum), which contains the carcinogen ptaquiloside.7,46 Intestinal adenocarcinoma in any deer species was unreported until a novel intestinal adenocarcinoma syndrome was documented, including 33.5% mucinous subtype, involving the distal ileum, cecum, and proximal colon in 76 farmed sika deer after natural chronic exposure to bracken fern. 18 In the referenced study, 18 no extraintestinal masses were reported grossly, and a large proportion of the neoplasms were either well- or moderately well-differentiated, as in our case, with about half of the cases lacking histologic evidence of metastasis. Consumption of bracken fern is known to lead to urinary bladder hemorrhage and neoplasia and alimentary tract neoplasia in ruminants, 9 including squamous cell carcinoma of the upper alimentary tract in association with papillomatosis. In our case, IHC for papillomavirus was negative, there was no known exposure to bracken fern in the zoologic enclosure, and there was no clinical history indicating that other deer were affected.
Cellular senescence is a known risk factor for the development of neoplasia in vertebrates, given that both mature age and neoplasms share intrinsic cellular mechanisms such as genomic instability, altered nutrient sensing metabolism, and epigenetic changes. In contrast, there are also divergent mechanisms in neoplastic and aged cells; neoplastic cells have high energy consumption, and the ability to rapidly divide and maintain advantageous mutations, whereas senescent cells accumulate disadvantageous mutations and undergo telomere attrition. 1
Intestinal adenocarcinoma is considered a multifactorial, heterogeneous disease associated with numerous genetic and environmental factors and cell signaling pathways. The molecular pathogenesis of adenocarcinomas in the proximal (ascending) colon has been studied extensively in humans, unveiling various molecular mechanisms for tumor initiation, promotion, and progression identified by multiple mutations. 17 Colorectal adenocarcinomas of dogs, a commonly studied species in comparative pathology, and humans share common tumorigenesis features. These include the presence of an altered adenomatous polyposis coli protein (APC) encoded by the tumor suppressor gene Apc, an apparent aid in early tumor development, as well as β-catenin and cyclooxygenase-2 overexpression. 46 Future molecular characterization of cervid intestinal adenocarcinomas is needed to determine whether underlying tumorigenesis mechanisms parallel or differ from those of other species in the veterinary literature.
We considered various differential diagnoses for our case (Table 1), including, but not limited to, colitis cystica profunda (CCP). CCP is an uncommon intestinal condition described in various species, characterized by dilated mucous glands that protrude through the muscularis mucosae into the submucosa. No cause has been determined for CCP, but there is some speculation that the lesion may develop as a sequela to colitis (such as in swine dysentery), local damage to the muscularis mucosae, or herniation into the space left by an involuted submucosal lymphoid follicle.14,46 Although adenocarcinomas in the large intestine tend to be better differentiated and less scirrhous than their small intestine counterparts, 46 the scirrhous response that is often associated with intestinal adenocarcinomas was absent in our case—a feature that could support a diagnosis of CCP vs. neoplasia. However, we ruled out CCP based on the lack of significant dilation of mucosal glands and primary colitis, and the presence of significant involvement of the serosa represented by interconnected submucosal and serosal cysts.
Histopathologic characteristics of differential diagnoses considered for well-differentiated colonic adenocarcinoma in animals and humans.
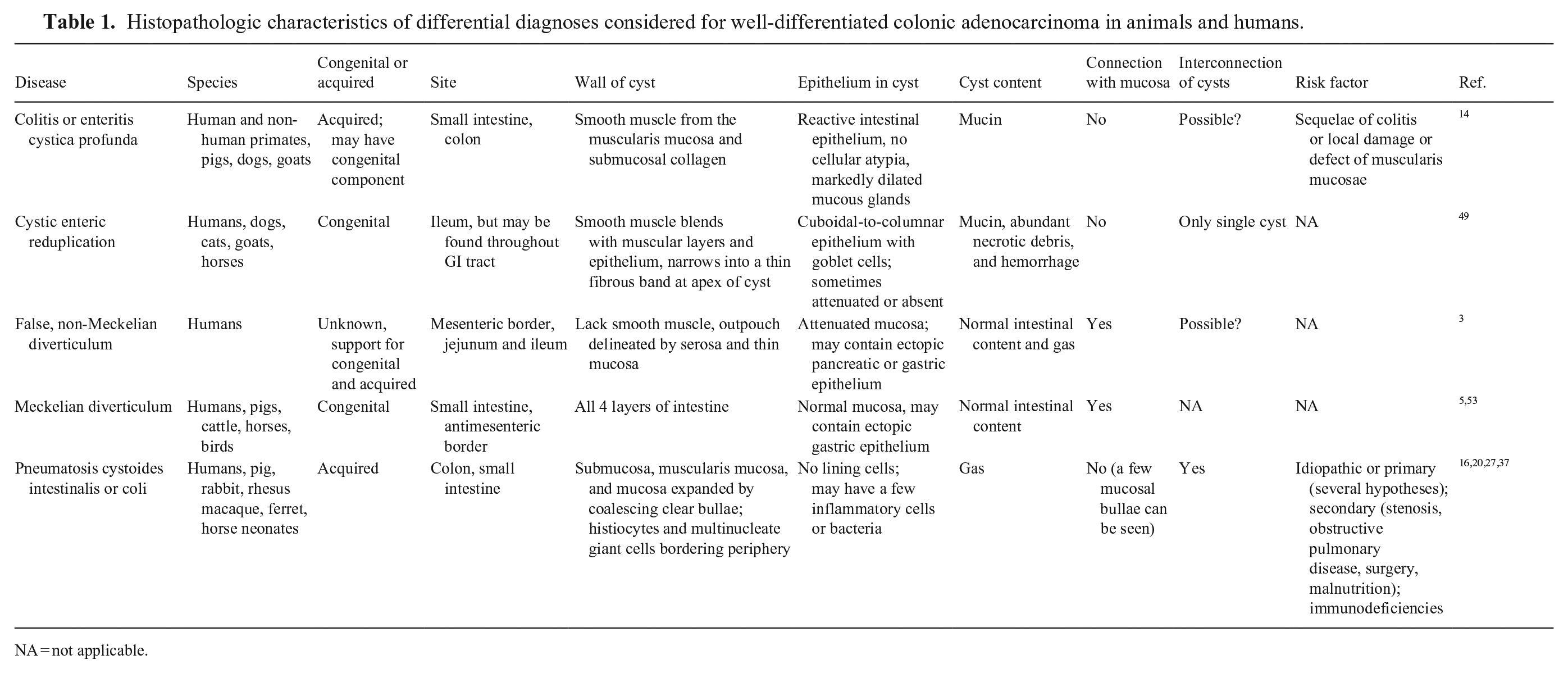
NA = not applicable.
Another diagnosis considered was cystic enteric reduplication (CER), a rare congenital malformation characterized by a blind pouch or cyst attached to the alimentary tract. CER is usually found in the small intestine but may affect other regions of the GI tract. CER is usually subclinical, but if located in the large intestine, diarrhea can develop. This condition was ruled out based on the lack of necrotic debris and hemorrhagic exudate filling the cysts, and the “outpouching” nature of CER versus the intramural-to-transmural cyst formation observed in cases of mucinous colonic adenocarcinoma. 49
Non-Meckelian diverticulum (NMD) is an intestinal outpouching reported in humans that occurs in segments in the small intestine without an association to the embryonic omphalomesenteric duct. 3 The most common clinical complaint is acute intestinal obstruction. Key pathology findings of NMD are lack of smooth muscle involvement, clear opening of the diverticula to the bowel lumen, and hypertrophic intestinal wall in adjacent, non-affected segments. Depending on the diameter of the diverticula, the walls can be “tissue paper” thin or have quite thick layers of serosa and mucosa in association with the larger and smaller diverticula, respectively. 3 NMD was ruled out based on the lack of involvement of the small intestine, the normal width of intestinal wall in non-affected segments, and the intramural nature of neoplastic cysts in our case, rather than layered intestinal outpouching.
Vitelline abnormalities such as persistence of the omphalomesenteric duct can lead to the formation of a Meckel diverticulum in the small intestine of herbivores. 2 The duct is normally resorbed during early fetal development but persists in certain waterfowl species.5,53 Meckel diverticulum was ruled out based on the lack of small intestinal involvement and absence of broad communication of the diverticulum with the intestinal lumen in our case. 53
Another condition of conjectural etiology that causes the formation of multiple thin-walled, cyst-like structures in the intestine is pneumatosis cystoides intestinalis or pneumatosis cystoides coli. These structures correspond to microvesicular, cystic, or diffuse emphysematous intramural bullae that contain gas and peripheral histiocytes and/or multinucleate giant cells. They lack both epithelium and mucinous content.16,20,27,37 Based on the lack of compatible morphologic characteristics, we ruled out pneumatosis cystoides coli.
We hope that future reports will help to unveil the prevalence, pathology, and epidemiology of colonic adenocarcinomas in deer species, with particular regard to determining the significance of intestinal adenocarcinoma in those species with decreasing population trends and risk of extinction.
Footnotes
Acknowledgements
We thank Juliann Beingesser and Jose Polanco for histopathology assistance.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
