Abstract
Cutaneous mast cell tumors (MCTs) are common canine neoplasms. Some dog breeds more frequently develop MCTs, suggesting a genetically mediated predisposition. In humans, the most common inherited cancer predisposition is caused by germline defects in the mismatch repair (MMR) genes. To investigate whether inherited defects in the MMR genes predispose some dogs to MCT development, MMR expression in 22 MCTs from young and predisposed breed dogs was compared with MMR expression in 22 MCTs from old dogs of non-MCT-predisposed breeds. MMR expression was investigated immunohistochemically using antibodies against MLH1, MSH2, and MSH6. Mast cells within all MCTs expressed MLH1, MSH2, and MSH6. There were no significant differences in the intensity of immunoreactivity or the percentage of cells expressing MMR proteins between MCTs from the 2 groups of dogs. There were no significant differences in MMR protein expression between grade II and grade III MCTs. These results do not support the hypothesis that inherited MMR defects predispose some dogs to MCT development.
Mast cell tumors (MCTs) are the most common potentially malignant cutaneous neoplasm of dogs. 13 Boxer dogs, as well as other breeds, are predisposed to the development of MCTs. 9, 25 This suggests that an inherited factor is present in some dogs that predisposes them to the development of an MCT. Although mutations within the c-KIT gene have been identified in some MCTs, 29 the mechanism for the inherited predisposition to MCT development is unclear.
As well as checking and repairing replicated DNA, the mismatch repair (MMR) system preserves genetic stability by multiple cellular interactions. 3 Studies suggest that all cells express MMR proteins with increased expression observed within replicating cells. 15, 30 Lynch syndrome is the most common inherited predisposition to cancer in humans. 11 It is caused by mutations in the MMR genes and results in more frequent development of neoplasia at a younger age. 1, 11 Intestinal adenocarcinomas are most common; however, people with MMR defects are also predisposed to skin cancer. 10 Cutaneous neoplasia commonly develops in mouse models of Lynch syndrome. 21, 22 Loss of MMR expression can also develop in sporadic skin tumors from people without Lynch syndrome, 26 and the presence of MMR defects is prognostically significant in some neoplasm types. 12, 27
Because of the more frequent development of MCTs in some dog breeds and the development of skin neoplasia in humans and mice with MMR gene mutations, it was hypothesized that inherited MMR gene defects predispose some dogs to MCT development. MMR expression within mast cells was detected immunohistochemically. To investigate possible inherited MMR defects, MMR expression within MCTs from young dogs of MCT-predisposed breeds, which should have high rates of MMR loss, was compared with MMR expression in MCTs from old dogs of mixed or nonpredisposed breeds, which should rarely have MMR loss. MMR expression within grade II MCTs was also compared with MMR expression in grade III MCTs as a preliminary investigation of a possible prognostic significance of MMR expression.
Canine cutaneous MCTs were identified by retrospective surveys of databases at New Zealand Veterinary Pathology Ltd. All neoplasms were routine diagnostic submissions from private veterinary clinics so that fixation conditions of the neoplasms were unknown. The diagnosis within the database was confirmed using histologic examination of a hematoxylin and eosin–stained section and graded using the criteria of Patnaik. 18 Neoplasms were included in the study if they were removed from a purebred dog fewer than 3 years of age, they were removed from a dog of a breed predisposed to develop MCTs and the dog was fewer than 6 years of age, or they were removed from a dog that was greater than 8 years of age that was either not of a MCT-predisposed breed or was a mixed breed dog.
For immunohistochemistry, sections were cut at a thickness of 5 µm, mounted on charged microscope slides, deparaffinized in xylene, rehydrated in graded ethanol, and rinsed in distilled water. Endogenous peroxidases were neutralized with 1% hydrogen peroxide in methanol for 15 minutes followed by rinsing for 5 minutes in distilled water. Antigen retrieval was achieved by incubating slides in epitope retrieval solution 1 (MSH2) or epitope retrieval solution 2 (MLH1, MSH6, and PMS2) (Leica Microsystems GmBH, Wetzlar, Germany) for 40 minutes with 20 minutes cool down. Slides were then incubated for 15 minutes with a mouse anti-human MLH1 antibody (BD Biosciences, San Jose, CA) at a dilution of 1 : 25, 30 minutes with a mouse anti-human MSH2 antibody (Leica Microsystems GmBH) at a dilution of 1 : 50, 15 minutes with mouse anti-human MSH6 antibody (BD Biosciences) at a dilution of 1 : 250, or 30 minutes with mouse anti-human PMS2 antibody (BD Biosciences) at a dilution of 1 : 500. Of the 4 antibodies used in this study, only the anti-MSH6 antibody has previously been reported to react with the canine MMR protein (BD Transduction Laboratories Technical Data Sheet). A Bond Refine Detection staining kit (Leica Microsystems GmBH) using 3,3′-diaminobenzidine substrate was used to visualize the immunoreaction, and sections were counterstained with hematoxylin. As with previous investigations of human skin neoplasms, 19 cells within surrounding normal epidermis were used as internal positive controls. The primary antibody was omitted in negative controls.
The intensity of the immunohistochemical reaction within the neoplastic cells was evaluated for each section. This was graded on a scale of 0 to 5, with 0 indicating no visible immunoreactivity and 5 indicating intense immunoreactivity. The percentage of mast cells with visible nuclear immunoreactivity was estimated by examining 5 different 400× fields within the neoplasm. Each MCT was assessed to see if immunoreactivity was distributed evenly within the neoplasm. All evaluations were performed by a single pathologist (JSM) who was unaware of the group of origin of the MCT. Differences between groups were investigated by analysis of variance using SPSS version 16 for Windows (SPSS Inc., Chicago, IL).
A total of 44 MCTs were included in the study. These were divided into 2 equal groups. Group 1 included MCTs from 10 Boxer dogs, 3 Golden Retrievers, 2 Weimaraners, a Labrador Retriever, a Staffordshire Bull Terrier, a German Shorthaired Pointer, a Shih Tzu, a Bichon Frise, a Border Collie, and a Doberman Pinscher. Included in this group were 1 grade I, 19 grade II, and 2 grade III MCTs. The average age of dogs in this group was 3.2 (range 0.5–5) years. Group 2 included MCTs from 16 mixed breed dogs, 2 Parson Russell Terriers, a Kelpie, a Vizsla, a Rhodesian Ridgeback, and a Rottweiler. Neoplasms included 19 grade II and 3 grade III MCTs. The average age of dogs in this group was 10.7 (range 9–14) years. The dogs in group 2 were significantly older than the dogs in group 1 (P < .001).
Results of the immunohistochemical evaluation are summarized in Table 1. Intranuclear expression of MLH1 was observed in nonneoplastic cells within all 44 samples. Nonneoplastic cells within the basal cell layer of the epidermis demonstrated the most intense immunoreactivity. The average intensity of the immunoreactivity was 2.4 within the 44 MCTs, with intensity ranging between grade 1 and grade 5 (Fig. 1). Overall, MLH1 expression was visible in 77% (range 30–100%) of the mast cells within the neoplasms. The intensity of the immunoreactivity was not significantly different between group 1 and group 2 MCTs. There were also no significant differences in intensity between grade II and grade III MCTs. There were no significant differences in the percentage of mast cells expressing MLH1 in group 1 MCTs and the percentage of cell expressing MLH1 in group 2 MCTs. There were also no significant differences in expression of MLH1 between cells from grade II and cells from grade III MCTs.
Summary of mismatch repair protein expression within mast cell tumors.*

All results are given as mean (1 standard deviation). Results with different letters are significantly different (P > .01).
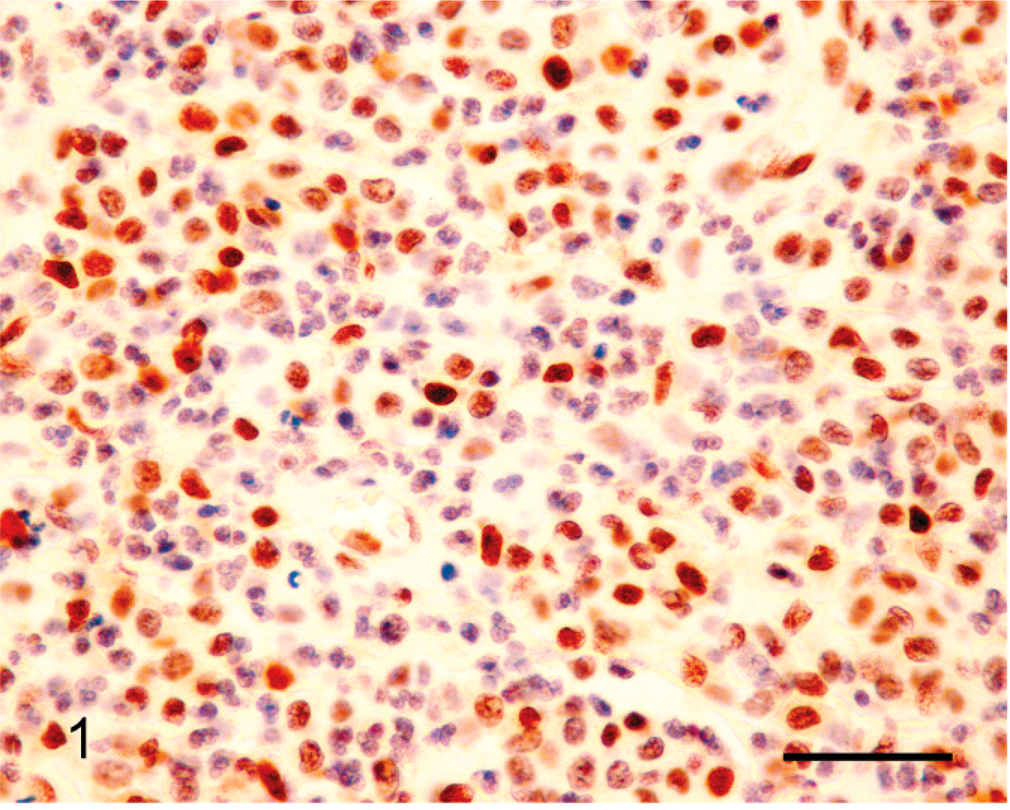
Canine grade II cutaneous mast cell tumor; approximately 70% of the mast cells within the neoplasm show nuclear reactivity to antibodies against MLH1. Note intense reactivity within the neoplastic cells. Bond Refine Detection staining kit. Bar = 30 µm.
Expression of MSH2 was not detected in internal controls within 2 sections, preventing evaluation of MMR expression within the neoplastic mast cells. Nonneoplastic cells within the remaining 42 sections showed a similar pattern of immunoreactivity to that observed using anti-MLH1 antibodies. Within mast cells in the 42 neoplasms, the average intensity of immunoreactivity using anti-MSH2 antibodies was 1.7 (range 1–3) (Fig. 2). This was significantly lower than the intensity observed using anti-MLH1 (P = .001) and anti-MSH6 antibodies (P < .001). Overall, 66% (range 40–90%) of mast cells within the 42 MCTs expressed MSH2, which was significantly lower than the percentage of cells expressing MLH1 (P = .006) and the percentage expressing MSH6 (P < .001). There were no significant differences in intensity of immunoreactivity or percentage of cells expressing MSH2 between group 1 and group 2 MCTs or between grade II and grade III neoplasms.
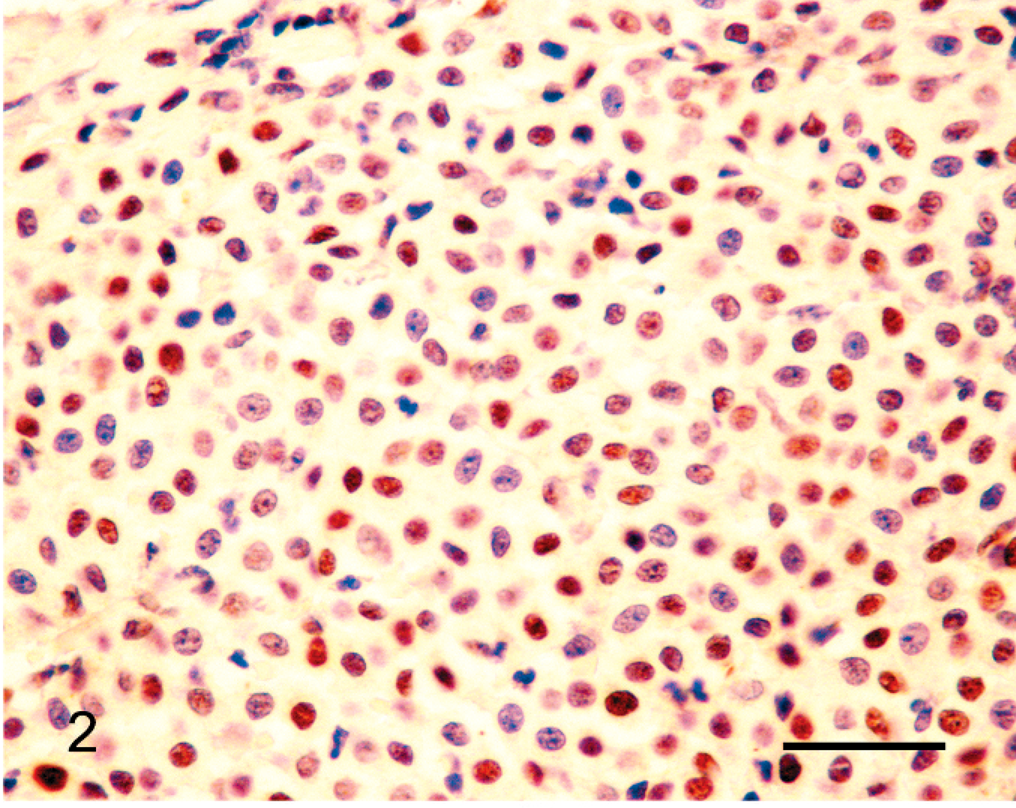
Canine grade II cutaneous mast cell tumor; approximately 80% of the mast cells within the neoplasm show nuclear reactivity to antibodies against MSH2. Note moderate intensity of reactivity within the cells. Bond Refine Detection staining kit. Bar = 30 µm.
Expression of MSH6 was not detected within positive controls in 1 sample. The pattern of MSH6 expression by nonneoplastic cells within the 43 sections demonstrating observable immunoreactivity was similar to that of anti-MLH1 and anti-MSH2 antibodies. Overall intensity of immunoreactivity of MSH6 was 3.3 (range 1–5) while 80% (range 50–100%) of the mast cells in the 43 neoplasms expressed MSH6 (Figs. 3, 4). Overall intensity of immunoreactivity to anti-MSH6 antibodies was significantly higher than intensity to anti-MLH1 antibodies (P = .001). There were no significant differences in intensity of immunoreactivity or percentage of cells expressing MSH6 between group 1 and group 2 MCTs or between grade II and grade III neoplasms.
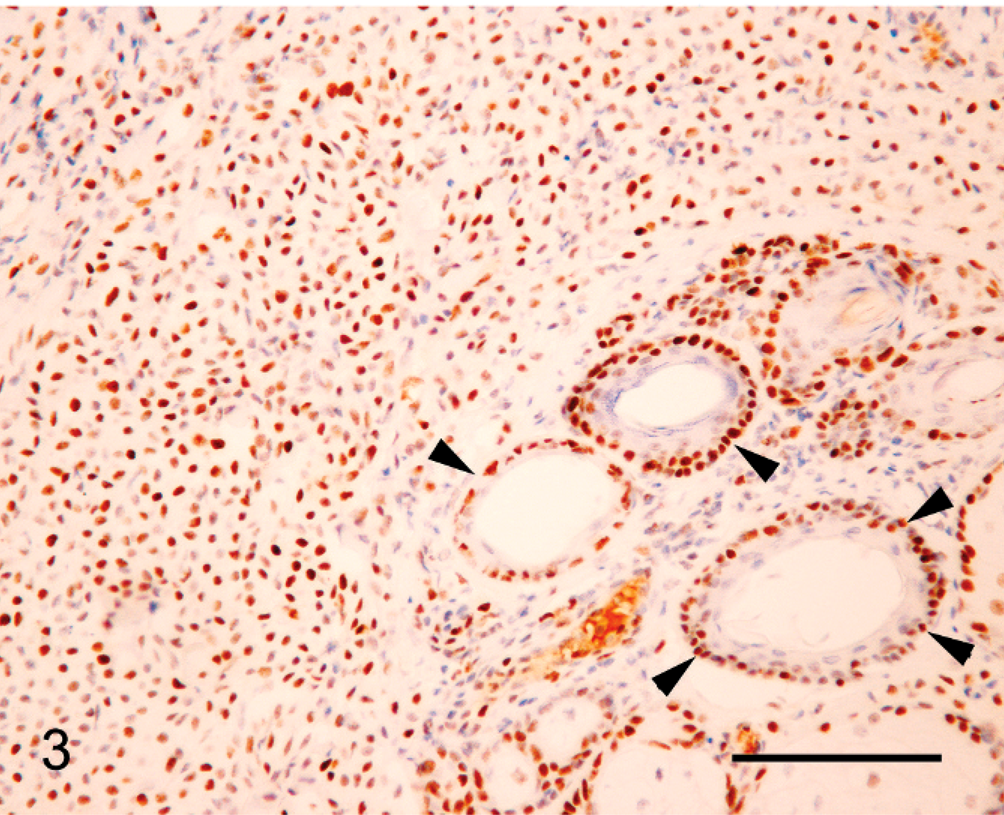
Canine grade II cutaneous mast cell tumor; mast cells within the neoplasm show consistent intense nuclear reactivity with antibodies against MSH6 (upper left). Note nuclear reactivity within basal cell nuclei within adjacent follicular epidermis (arrowheads). Bond Refine Detection staining kit. Bar = 85 µm.
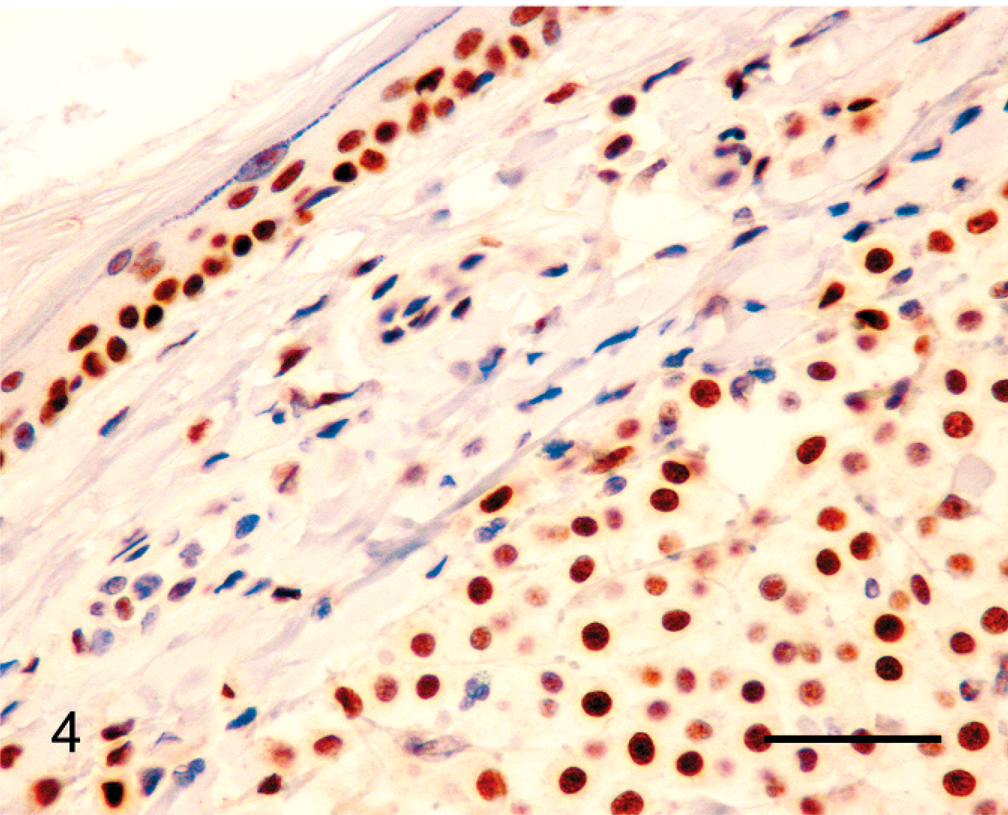
Canine grade II cutaneous mast cell tumor; mast cells within the neoplasm show nuclear reactivity with antibodies against MSH6 (lower right). Note nuclear reactivity in basal cells within adjacent surface epidermis (upper left). Bond Refine Detection staining kit. Bar = 30 µm.
Immunoreactivity within all neoplasms appeared evenly distributed throughout the tumors, and there was no evidence of differences in MMR expression as a result of tumor progression. The intensity of immunoreactivity of the neoplastic cells was generally similar to the intensity of the positive control cells. No expression of PMS2 was observed within 5 MCTs and no additional neoplasms were investigated for expression of this protein.
All 3 MMR proteins were detected immunohistochemically within all MCTs that were able to be assessed. Therefore, loss of MMR expression appears to be rare in canine MCTs. In humans, skin tumors in which altered MMR expression has been reported include cutaneous sebaceous gland neoplasms, keratoacanthomas, squamous cell carcinomas, basal cell carcinomas, dysplastic nevi, melanocytic nevi, and melanomas. 19, 24, 26, 33 Loss of MMR expression has not been reported in human mastocytosis. To the authors' knowledge, this is the first time MMR expression has been evaluated in a canine cutaneous neoplasm.
In the present study, MCTs were divided into 2 groups. Group 1 MCTs were from young dogs mainly of MCT-predisposed breeds. If inherited MMR gene defects predisposed these dogs to developing MCTs at an early age, loss of MMR expression should have been commonly observed within group 1 MCTs. Group 2 MCTs were from old dogs of breeds not predisposed to MCT development. These dogs were considered unlikely to have inherited MMR gene defects, and loss of MMR expression would be expected to be rare in group 2 MCTs. The failure to detect significant differences in MMR expression between the 2 groups of MCTs suggests that inherited MMR gene defects do not predispose dogs to MCT development. Alternatively it is possible that the MCTs contained MMR proteins that had retained their immunoreactivity but were no longer functional. Although this cannot be excluded, evidence from human colorectal cancers suggest most mutations within the MMR genes prevent protein expression and only a small fraction of MMR-deficient cells retain MMR immunoreactivity. 12 Additionally, it is possible that predisposed dogs may have defects within a currently unrecognized MMR component.
In humans, the inheritance of 1 functional and 1 mutated copy of a MMR gene results in Lynch syndrome. 11 Individuals with Lynch syndrome more frequently develop cancer and develop cancer at a younger age than the general population. 2 Although neoplasms of the gastrointestinal tract (most frequently hereditary nonpolypoid colorectal cancer) are most common in Lynch syndrome, affected individuals also are predisposed to developing reproductive tract, skin, brain, lymphoid, and urinary tract neoplasms. 1, 2, 10, 14 Muir-Torre syndrome is a subset of Lynch syndrome and is defined by the development of a cutaneous sebaceous neoplasm or keratoacanthoma and a second neoplasm within the Lynch syndrome spectrum. 19, 31 Recent evidence suggests that Lynch syndrome may also predispose humans to cutaneous melanomas. 20 Mice lacking MMR protein expression also develop neoplasia at a young age. Twenty-seven percent of mice with inactivated MSH6 genes developed skin or mammary gland neoplasms. 7 One mouse developed a cutaneous hemangioma, while another developed a pilomatricoma in a group of 31 mice with inactivated MLH1 genes. 21 Skin neoplasms, including squamous and sebaceous tumors, developed in 7% of mice lacking MSH2 expression. 22 Additionally, mice defective in MSH2 are predisposed to developing ultraviolet-induced skin neoplasia. 16
Mutations within the DNA of a cell can be subclassified as chromosomal or microsatellite instability mutations. 4 Microsatellite instability (MIN) mutations include base/base mismatches or small insertion/deletion loops during DNA replication and result in subtle changes to the DNA sequence. 5, 8 The MMR system prevents MIN mutations, and MMR-deficient cells have 100 to 1,000 times more MIN mutations than normal cells. 1, 4, 5 The mammalian MMR system contains 4 main proteins that form 2 heterodimers. 6 The heterodimer MutSα consists of MSH2 and MSH6 and detects mismatches within the DNA, while MutLα consists of MLH1 and PMS2 and couples mismatch recognition with the repair processes. 6 In humans with Lynch syndrome, loss of normal expression of MSH2 is observed in 40% of patients, loss of MLH1 in 39%, loss of MSH6 in 15%, and loss of PMS2 in 6%. 32 Immunohistochemical examination of a neoplasm from a person with Lynch syndrome reveals normal MMR expression in nonneoplastic cells but no MMR expression in neoplastic cells. 28
As well as recognizing and repairing DNA that has been mismatched during replication, the MMR system can also induce cell-cycle arrest and/or apoptosis in response to DNA damage. 3 These additional functions of the MMR proteins suggest that loss of MMR function within a neoplasm could promote progression and be prognostically significant. Loss of MSH2 expression was observed more commonly in higher grade human basal cell carcinomas and was correlated with neoplasm recurrence and patient death. 27 In contrast, failure to express MMR proteins by colorectal cancer cells was found to predict a more favorable clinical outcome. 12 In the present study, there were no significant differences in the expression of MMR proteins between grade II and grade III MCTs. However, investigation of a possible prognostic significance of MMR expression was limited by consistent expression of MMR proteins within the neoplasms and the small number of grade III neoplasms included in the study. Additional grade I and grade III MCTs need to be assessed before it can be definitively established that MMR expression does not predict prognosis.
Canine MCTs often have altered c-KIT expression. 29 Mutations in the c-KIT gene observed in MCTs include single nucleotide substitutions, deletion of a 6 base pair section of DNA, and tandem duplications of sections of DNA. 13, 23 Although is it possible that MMR dysfunction could predispose dog breeds to the development of these mutations, the results of the present study do not support this hypothesis. The causes of the increased development of MCTs within some dog breeds remain unresolved.
Immunoreactivity to anti-MSH2 antibodies was significantly less intense than that observed using antibodies against MLH1 and MSH6. The lower intensity of immunoreactivity using anti-MSH2 antibodies was consistent within both the neoplastic cells and the internal controls, suggesting the variation in intensity between the antibodies was caused by a lower affinity of the anti-MSH2 antibody to the canine protein. The lower affinity of the antibody was most likely the cause of the significantly lower proportion of neoplastic cells detectably expressing MSH2. No reactivity to PMS2 was observed in internal positive controls or neoplastic cells within 5 samples. This suggests that the anti-PMS2 antibody has a low affinity for the canine PMS2 protein. Antibodies against human PMS2 protein have been previously reported not to react with ovine PMS2 protein. 17
In conclusion, this is the first investigation of MMR protein expression by canine cells. Loss of MMR expression appears to be rare in canine cutaneous MCTs. The results of this study do not support the hypothesis that dogs can be predisposed to MCT development by inherited mutations within the MMR genes. Because of the infrequent loss of MMR expression within MCTs, MMR expression within the neoplastic cells appears unlikely to be a valuable prognostic indicator.
