Abstract
Functional evaluation of the pars intermedia (PI) is required for the early diagnosis of equine pituitary PI dysfunction (PPID), yet most assays target the hypothalamic-pituitary-adrenal axis, which regulates the pars anterior. In contrast, the PI is regulated by dopaminergic tone from hypothalamic neurons. Loss of dopaminergic inhibition is hypothesized to cause the PI hypertrophy and hyperplasia that result in the clinical manifestations of PPID. Domperidone, a dopamine receptor antagonist, should exacerbate the loss of dopaminergic inhibition in horses with PPID and increase the release of endogenous adrenocorticotrophic hormone (eACTH) by PI melanotrophs. To test this, plasma eACTH concentration was determined in horses with or without clinical signs of PPID at 0, 4, and 8 hours after oral administration of 3.3 mg domperidone/kg. Pituitary glands were evaluated postmortem by histologic grading and morphometry. In the 33 horses, median age, plasma ACTH concentration 8 hours after domperidone, and PI area in median sagittal sections were associated with histologic grade as follows: pituitary grade 1 (normal), n = 3, 7.5 years, 20.0 pg/ml, 0.16 cm2; grade 2 (focal hypertrophy or hyperplasia), n = 9, 14.5 years, 27.1 pg/ml, 0.27 cm2; grade 3 (diffuse adenomatous hyperplasia), n = 5, 21.0 years, 64.4 pg/ml, 0.48 cm2; grade 4 (microadenomas), n = 12, 23.3 years, 128.0 pg/ml, 0.87 cm2; grade 5 (adenoma), n = 4, 24.9 years, 720.5 pg/ml, 2.1 cm2. Results suggest that horses with pituitary histologic grade ≥3 respond to domperidone with increased plasma ACTH concentration.
Keywords
Pituitary pars intermedia dysfunction (PPID), also known as equine Cushing's disease, is the most commonly diagnosed equine endocrinopathy. 25 Affected horses may have hirsutism, laminitis, muscle atrophy, abnormal adiposity, weight loss, polyuria and polydypsia, infertility, and increased susceptibility to infection. 1,11,18,19,25 Hirsutism is considered almost pathognomonic for PPID and strongly predictive of pars intermedia (PI) adenoma (or microadenomas). 14,25 However, pathologic evaluation of the equine PI is complicated, because hypertrophy, hyperplasia, or adenomas in the PI (and less commonly in pars anterior) were documented in equids with and without clinical signs of PPID. 7,11,14,22,28 The clinical diagnosis of PPID is straightforward in horses with hirsutism but difficult in horses with early disease or less-specific clinical signs.
The lack of a sensitive and specific test to detect PPID in horses without hirsutism has prompted the development of multiple assays. 13,19,25 The dexamethasone suppression test is commonly used but may have low sensitivity. 12 The combined dexamethasone suppression/thyrotropin-releasing hormone stimulation test resulted in higher sensitivity than with either test alone. 14 The dexamethasone suppression test is also subject to false-positive responses (failure to suppress plasma cortisol levels), especially in the autumn. 12 The plasma concentration of α-melanocyte-stimulating hormone (α-MSH) is also elevated in the autumn in horses and ponies. 17 The dexamethasone suppression test depends on negative feedback in the hypothalamic-pituitary-adrenal axis, in which glucocorticoids suppress adrenocorticotrophic hormone (ACTH) release from the pituitary pars anterior; the PI is not subject to this negative feedback. 4,6 In equine PPID, melanotrophs in the PI synthesize excess proopiomelanocortin (POMC) and release excess α-MSH and β-endorphins, as well as relatively less, but still excessive, ACTH. 21,23 Horses with PPID fail to suppress plasma cortisol concentration after dexamethasone stimulation, because PI melanotrophs do not express glucocorticoid receptors 3 and thus are not under the control of the hypothalamic-pituitary-adrenal axis. In the PI of the rat, dopaminergic hypothalamic neurons inhibit POMC synthesis, and release by binding dopamine D2 receptors on melanotrophs. 29 Tissue dopamine concentration is decreased in PI adenoma; the associated loss of dopaminergic inhibition is believed to cause equine PPID. 21 Treatment with dopamine or its agonists results in decreased concentration of POMC-derived peptides and may alleviate signs of PPID. 23 Currently, the dopamine agonist pergolide is considered the treatment of choice for equine PPID. 25
A test that is based on dopaminergic regulation of the PI, rather than the hypothalamic-pituitary-adrenal axis, is needed to evaluate the PI function in horses without hirsutism. Domperidone, a synthetic benzimidazole that is approved for treating endophyte-associated agalactia in mares, 10 specifically blocks peripheral dopamine D2 receptors. 5,8 In the PI, this releases melanotrophs to express POMC-derived peptides, including α-MSH, β-endorphin, corticotrophin-like intermediate peptide (CLIP), and ACTH. 29 Domperidone was selected as the dopamine antagonist for investigation in this study, because it is marketed for use in the horse, has a wide margin of safety, and does not cross the blood-brain barrier. We hypothesized that domperidone treatment would have little effect on plasma ACTH concentration in horses with normal PI function but that the same treatment of horses with PPID would exacerbate the loss of dopaminergic inhibition of PI melanotrophs and result in increased plasma ACTH concentration.
Postmortem histologic evaluation of pituitary glands has been used to validate clinical diagnostic tests for PPID, 14 although a recent study revealed only moderate concordance among pathologists or correlation between pituitary lesions and antemortem test results. 19 The purposes of the study reported here were to determine the effect of domperidone administration on plasma ACTH concentration in horses with or without clinical signs of PPID and to correlate the plasma ACTH response to domperidone with postmortem histologic and morphometric features of the pituitary gland.
Material and Methods
Test horses were donated to Purdue University by private owners for various reasons. Horses were kept in box stalls in the large animal teaching hospital during initial assessment, then were moved to the veterinary teaching farm, and maintained on pasture or dry lot with free access to grass hay and water. Horses were included in this study if they were mature and scheduled for euthanasia, with sufficient time for acclimation and testing. Ponies were excluded. Animals were classified by 2 authors (LPJ and JES) upon entry into the study as PPID suspects if they had hirsutism or a history of not shedding properly or abnormal fat distribution. All procedures were approved by the institutional animal care and use committee.
Domperidone-provoked plasma endogenous ACTH (eACTH) assays were performed from September 2005 to December 2006. Horses were weighed and moved into box stalls with free access to water and hay 48 hours before testing. Domperidone paste (Equidone, 11% domperidone w/v, Equi-Tox, Inc., Central, SC) was administered per os at a dose of 3.3 mg/kg body weight at 8:00 a.m. Blood was collected in silicon-coated ethylenediamine tetraacetic acid (EDTA) tubes 0, 4, and 8 hours after domperidone administration. Plasma was harvested and stored within 20 minutes in a −80°C freezer. Plasma ACTH concentration was measured by chemiluminescent immunoassay (Immulite ACTH, Diagnostic Products Corporation Cirrus, Inc., Los Angeles, CA), which was validated for use in the horse in our laboratory. The laboratory reference range for normal equine plasma eACTH concentration is 10–59 pg/ml.
Within 1 month after domperidone administration, horses were euthanized with intravenous pentobarbital and were submitted for postmortem examination. For horse Nos. 1–9, materials were retrieved for histomorphometric evaluation from archived paraffin blocks and histologic sections. For horse Nos. 10–33, pituitary glands were dissected at postmortem, weighed, and prepared for histologic processing by 1 of 2 authors (IDP and MAM). The entire pituitary gland, slices of each adrenal gland, brain including hypothalamus, and other selected tissues were fixed in neutral-buffered 10% formalin. After fixation, the pituitary gland was bisected through the median sagittal plane. A parallel cut was then made on either side of the midline to create 2 approximately 3-mm-thick slices for histologic processing. The remaining pituitary tissue was trimmed to create right and left paramedian slices. Tissues were embedded in paraffin so that histologic sections were cut from the medial face of each slice.
Histologic sections stained with hematoxylin and eosin (HE) were examined and graded independently by 2 authors (IDP and MAM), without knowledge of ACTH assay results. Any discordance in grading was resolved by arbitration before ACTH assay results were revealed. Pituitary glands were graded histologically as 1 (within normal limits), 2 (focal or multifocal PI hypertrophy or hyperplasia), 3 (diffuse PI adenomatous hyperplasia), 4 (PI adenomatous hyperplasia with microadenomas [1- to 5-mm diameter]), or 5 (adenoma [>5-mm diameter] in PI or pars anterior). Although grades were based on the presence or absence of the above-mentioned features, pituitary sections were also evaluated for PI cytologic features, the presence of colloid-filled follicles or cysts (≥1 mm in diameter) in PI or pars anterior, the definition of PI border with pars nervosa and pars anterior, hyperplasia or hypertrophy of the pars anterior, and degenerative changes in pars nervosa.
For image analysis, median sagittal histologic sections of the pituitary gland were scanned digitally at high resolution (1,200 dpi) in red-green-blue color and saved as TIFF files; a millimeter scale was included in each scanned image for spatial calibration. By using a magnetic lasso (Adobe Photoshop Elements 2.0, San Jose, CA), pars nervosa, PI, and pars anterior were selected separately; pixels in each selection were painted with a contrasting color. The area of the PI, pars anterior, and pars nervosa in median sagittal sections (both right and left face of the bisected pituitary gland) was calculated with a public domain Java image processing program (Image J, National Institutes of Health, Bethesda, MD [http://rsb.info.nih.gov/ij/]) and recorded for each gland as the mean of right and left measurements. Continuous variables were described in summary statistics as mean or median and range. The Spearman rank correlation coefficient for nonparametric data was used to quantify the strength of association between variables. Results were considered statistically significant when P < .05.
Results
Clinical findings
Signalment, body weight, pituitary grade, history, clinical diagnoses, and PPID classification of the 33 horses are presented in Table 1. Horses ranged in age from 3 to 35 years; mean, 19.0 years; median, 20.0 years. Only 3 horses were younger than 10 years. Breeds included 9 Quarter Horses; 6 Thoroughbreds; 5 American Saddlebreds; 5 Appaloosas; 4 Arabians; and 1 each of Paint, Paso Fino, Warmblood, and Albino. There were 16 geldings, 16 mares (none lactating; one 60-day pregnancy), and 1 stallion. Body weight of the 33 horses ranged from 342 to 599 kg; mean, 472.8 kg; median, 477 kg. Lameness, including 11 cases of laminitis, was a major clinical problem in 21 horses, making it the most commonly recorded clinical sign (and most common reason for donation of the horse).
Signalment, body weight, pituitary grade, and clinical diagnoses.
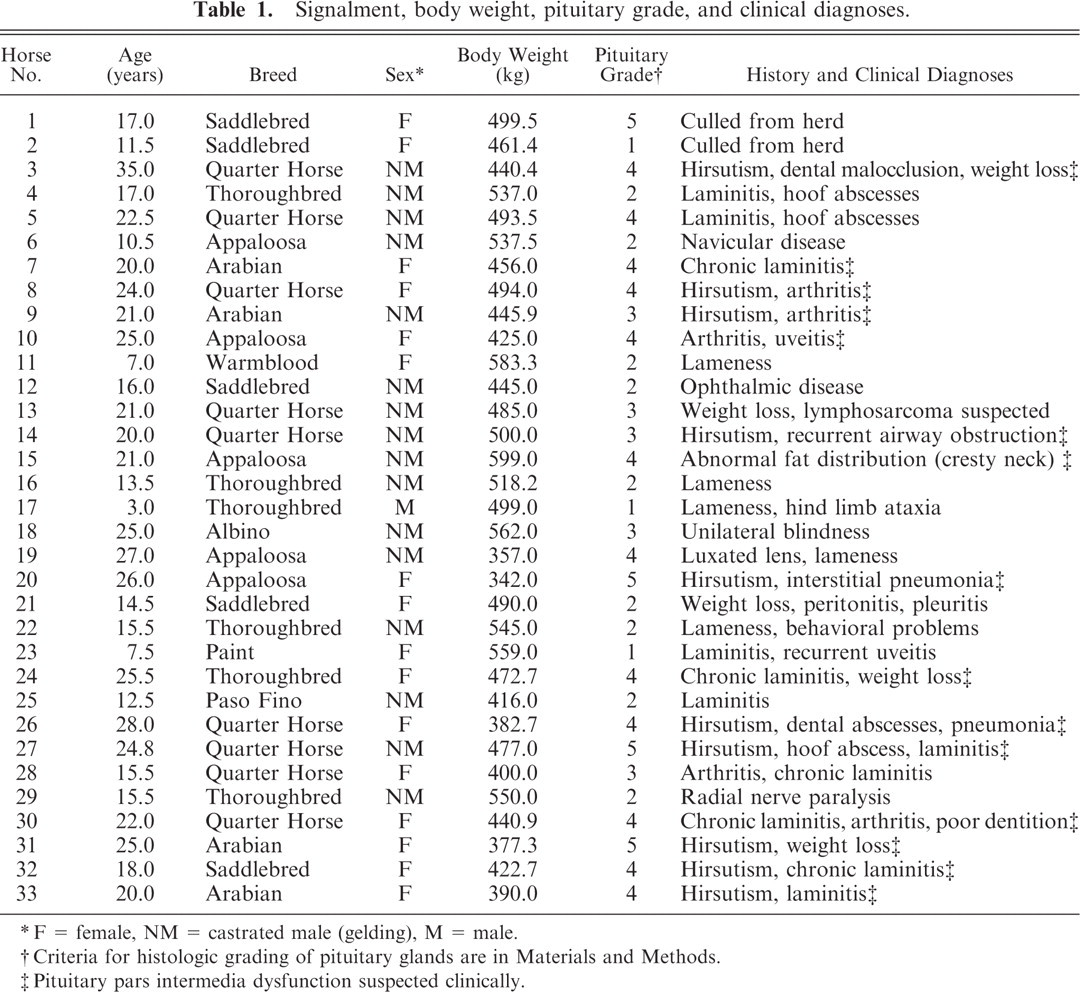
∗ F = female, NM = castrated male (gelding), M = male.
† Criteria for histologic grading of pituitary glands are in Materials and Methods.
‡ Pituitary pars intermedia dysfunction suspected clinically.
The 15 horses for which PPID was suspected on the basis of clinical signs had pituitary histologic grades of 3 (2 horses), 4 (10 horses), or 5 (PI adenoma in 3 horses). The 18 horses without overt signs of PPID had pituitary histologic grades of 1 (3 horses), 2 (9 horses), 3 (3 horses), 4 (2 horses), and 5 (the 1 horse with an adenoma in the pars anterior). Grading was based mainly on histologic features of the PI and examination of median sagittal sections, except for horse No. 1, which had an adenoma in the pars anterior that was best visualized in paramedian section.
Histologic findings
Of the 33 pituitary glands evaluated, 3 were classified histologically as grade 1, 9 as grade 2, 5 as grade 3, 12 as grade 4, and 4 as grade 5 (Table 1). In median sagittal section, grade 1 pituitary glands were elliptical (Fig. 1). The PI appeared as a narrow band of regular thickness, sharply demarcated from pars nervosa and pars anterior, and comprised packets of polyhedral cells without hyperplasia or hypertrophy. A few colloid-filled follicles were formed by PI cells in 2 glands. Cystic follicles (luminal diameter ≥1 mm) were not observed. Mild patchy hyperplasia of chromophobes in the pars anterior was noted in 1 horse.
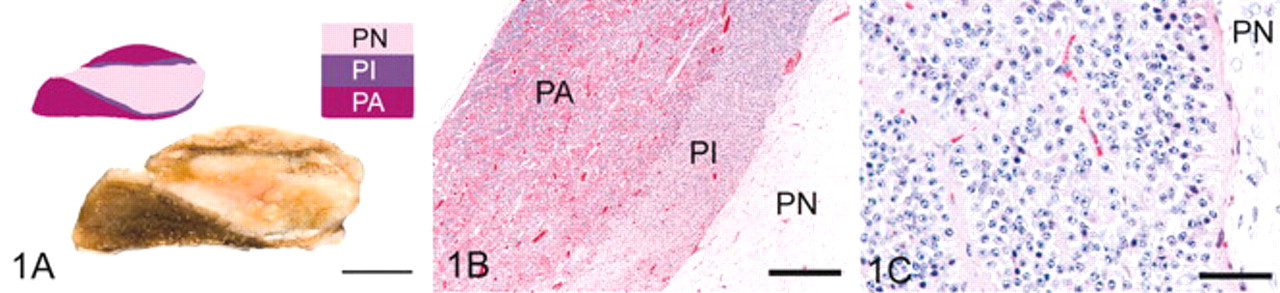
Pituitary gland, grade 1; horse.
In grade 2 pituitary glands (Fig. 2), focal or multifocal hypertrophy or hyperplasia resulted in uneven thickness of the PI. The interface between PI and pars nervosa was focally obscured in 7 of 9 horses; the pars anterior was minimally compressed in 1 horse. Hypertrophied PI cells had larger nuclei and more abundant pale cytoplasm than adjacent cells. Eight of 9 grade 2 pituitary glands had a few to moderate number of follicles in the PI; in 6 of these horses, some follicles were cystic (≥1 mm in diameter).
Pituitary gland, grade 2; horse.
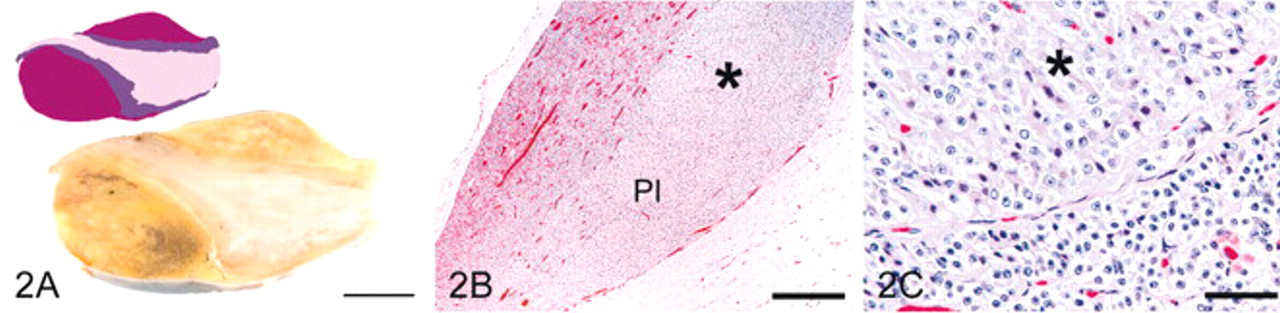
In grade 3 pituitary glands (Fig. 3), diffuse hyperplasia resulted in irregular thickening throughout the PI with compression of the pars anterior and an irregular and indistinct border, with both pars anterior and pars nervosa. PI cells had larger nuclei than those of grade 1 pituitary glands. Follicles and cysts were consistently present and sometimes numerous in the PI.
Pituitary gland, grade 3; horse.
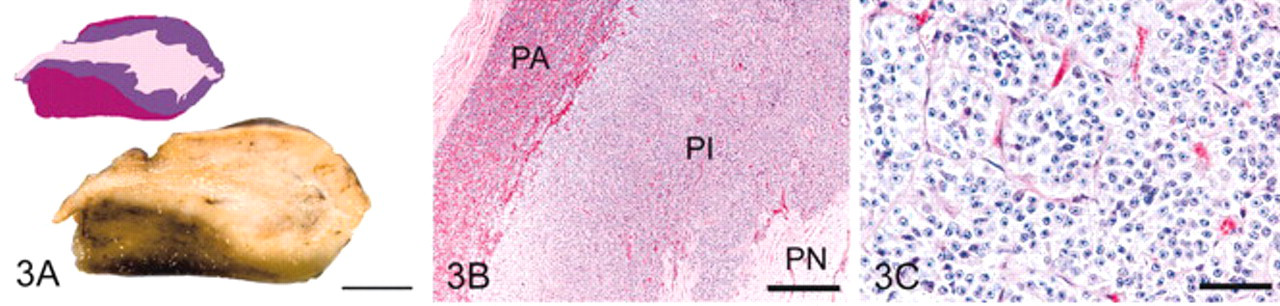
Grade 4 pituitary glands (Fig. 4) had features of grade 3 pituitary glands plus the presence of discrete PI nodules 1–5 mm in diameter (microadenomas). Cells within the microadenomas were pyriform with enlarged nuclei, ample eosinophilic or amphophilic cytoplasm, and distinct cell borders. Follicles were consistently present, sometimes numerous, and focally cystic in all but 1 horse.
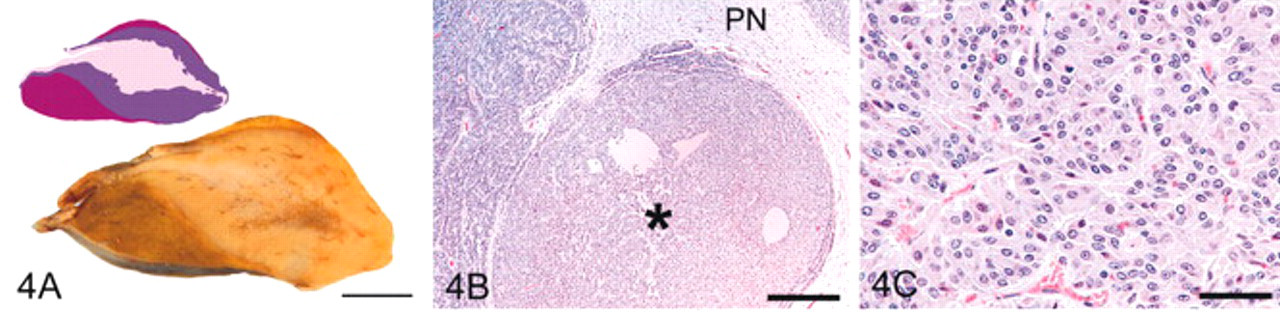
Pituitary gland, grade 4; horse.
Four pituitary glands were classified as grade 5 by the presence of an adenoma. In horse No. 1, an adenoma, 8 mm in diameter, was in the pars anterior; only diffuse hyperplasia was evident in the PI. Each of the other 3 horses had an adenoma that exceeded 1 cm in diameter in the PI. In horse No. 20 (Fig. 5), the PI adenoma formed a roughly spherical, 1.4-cm diameter, pale gray mass that compressed pars nervosa and pars anterior. The PI adenoma in horse No. 27 was an irregularly shaped, multifocally necrotic, and hemorrhagic, about 2.5-cm-diameter mass that occupied most of the median sagittal section. The PI adenoma in horse No. 31 was about 2.1 cm in diameter and histologically similar to that in horse No. 20. Follicles were present in the PI of all 4 horses and focally cystic in the 3 horses with PI adenoma.
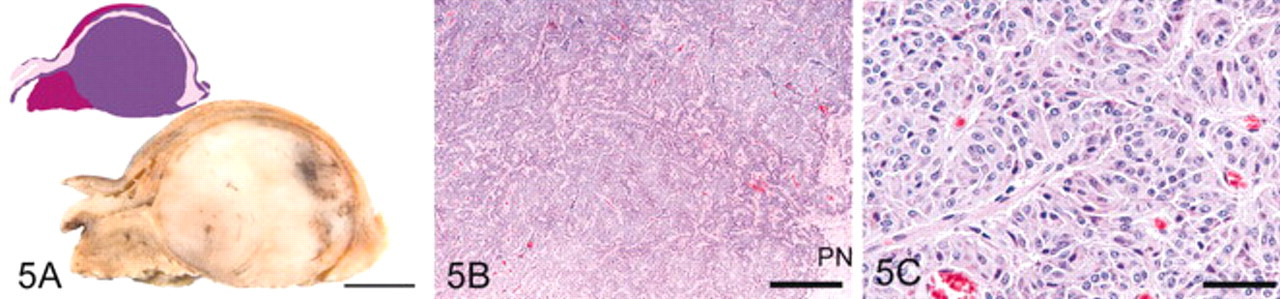
Pituitary gland, grade 5; horse No. 20.
In the pars nervosa, axonal swellings (spheroids), pigment accumulation, hemorrhage, and gliosis were observed in some horses, but a strong correlation between histologic changes in the pars nervosa and histologic grade was not evident. However, degenerative changes in the pars nervosa were particularly severe and accompanied by necrosis in horse No. 3 (the grade 4 horse with the largest PI area measurement) and horse No. 27 (the horse with the largest PI adenoma).
Histologic changes in the pars anterior that neither contributed to nor correlated with the histologic grade included follicles and cysts in some horses in all grades except grade 1. Focal hyperplasia of chromophobic cells in the pars anterior was noted in 1 of 3 grade 1 pituitary glands and in most glands in grades 2–5. Chromophobe hyperplasia was moderate to marked in 5 glands (grades 2, 4, and 5); however, because it was not a criterion for grading, unless it resulted in formation of a nodule larger than 5 mm in diameter, it did not correlate with histologic grade.
The hypothalamus was examined histologically in 24 horses (Nos. 10–33); lesions were observed only in horse No. 13, in which multicentric lymphosarcoma involved the brain, and, in horse No. 27, in which the 15-g pituitary gland compressed the hypothalamus. Adrenal gland was examined histologically in 32 horses (all except horse No. 21); horse No. 24 had an adrenocortical adenoma; other adrenal glands were within normal limits.
Correlation of clinical parameters with histologic grade
The mean age of horses with pituitary grade 1 was 7.3 years (median, 7.5 years); horses with grade 2, 13.6 years (median, 14.5 years); horses with grade 3, 20.5 years (median, 21.0 years); horses with grade 4, 24.0 years (median, 23.3 years); horses with grade 5, 23.2 years (median, 24.9 years). The age of the horses correlated strongly and positively with pituitary grade (rho = 0.791; P < .0001). Body weight tended to decrease with pituitary grades higher than 2 and correlated negatively with pituitary grade (P = .002). Horses with hirsutism had pituitary grades of 3, 4, or 5, but hirsutism was not apparent in all horses with pituitary grade ≥3. Other clinical signs were not obviously associated with pituitary grade. An association between breed or sex and pituitary grade was not evident.
Correlation of ACTH assay results and pituitary morphometry with histologic grade
Test date, plasma ACTH concentration before and after domperidone administration, pituitary weight, pituitary height, and pituitary area in histologic section are presented in Table 2, in which data are sorted by pituitary histologic grade and PI area in median sagittal section. One horse was tested in each of the months of January, February, and March; 4, 4, and 5 were tested in April, May, and June, respectively; 1, 0, and 9 were tested in July, August, and September, respectively; 4, 1, and 3 were tested in October, November, and December, respectively. No correlation between month of testing and other variables was evident.
Test date, post-domperidone plasma adrenocorticotrophic hormone concentration, and pituitary morphometry sorted by histologic grade and pars intermedia area.
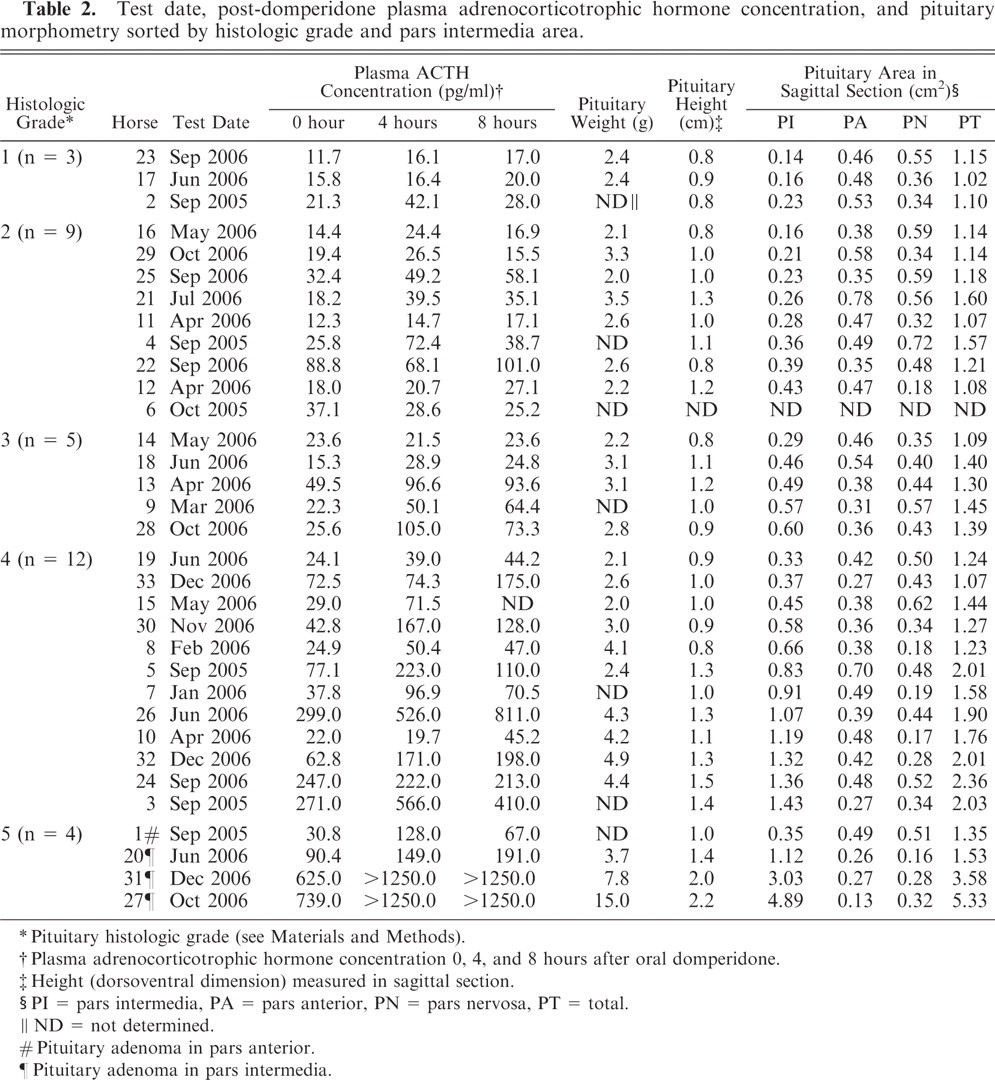
∗ Pituitary histologic grade (see Materials and Methods).
† Plasma adrenocorticotrophic hormone concentration 0, 4, and 8 hours after oral domperidone.
‡ Height (dorsoventral dimension) measured in sagittal section.
§ PI = pars intermedia, PA = pars anterior, PN = pars nervosa, PT = total.
∥ ND = not determined.
# Pituitary adenoma in pars anterior.
¶ Pituitary adenoma in pars intermedia.
Pituitary weight was recorded for all but 7 horses and tended to increase with increasing pituitary grade (P = .001), but there was considerable overlap in the range of values. The length (rostral to caudal) of most pituitary glands was nearly equal to the width of the gland (data not shown); neither length nor width correlated strongly with pituitary grade. Height (dorsal-to-ventral dimension, measured in sagittal section) of pituitary glands varied more than length or width and correlated positively with pituitary grade (P = .002); however, the range in values overlapped among groups.
The area of the pituitary gland, especially that of the PI, in median sagittal histologic section, correlated strongly and positively with histologic grade (rho = 0.794; P ≤ .0001). The area of the PI also correlated strongly and positively with ACTH concentration at 0 hour (rho = 0.757), 4 hours (rho = 0.757), and 8 hours (rho = 0.766) after domperidone administration (P ≤ .0001). Mean (median) PI area was 0.18 (0.16) cm2 in grade 1 horses, 0.29 (0.27) cm2 in grade 2 horses, 0.48 (0.48) cm2 in grade 3 horses, 0.88 (0.87) cm2 in grade 4 horses, and 2.3 (2.1) cm2 in grade 5 horses. Although the area of the pars anterior increased because of hyperplasia in some horses, the area of pars anterior and pars nervosa tended to decrease as PI area increased and correlated negatively with histologic grade (P = 0.03). The area of the pituitary gland in horse No. 6 was not measured, because the gland had not been sectioned through the median sagittal plane.
Plasma eACTH concentration at 0 hour (resting or baseline), 4 hours, and 8 hours after domperidone administration correlated positively with histologic grade (P ≤ .0001). This correlation was strongest at 8 hours after domperidone administration (rho = 0.761). In the 3 horses with grade 1 pituitary glands, mean (median) ACTH concentration at 0, 4, and 8 hours was 16.3 (15.8), 24.9 (16.4), and 21.7 (20.0), pg/ml, respectively. In 1 horse (No. 2), plasma ACTH concentration 4 hours after domperidone was twice the baseline concentration but diminished to 1.3 times baseline concentration by 8 hours. The median 4 hour and 8 hour to baseline ACTH concentration ratios were 1.4 and 1.3, respectively. Plasma ACTH concentration never exceeded the reference range of 10–59 pg/ml for horses with grade 1 pituitary glands.
In the 9 horses with grade 2 pituitary glands, mean (median) ACTH concentration at 0, 4, and 8 hours was 29.6 (19.4), 38.2 (28.6), and 37.2 (27.1) pg/ml, respectively. One horse, No. 22, had a baseline ACTH concentration >59 pg/ml but less than twofold increase after domperidone. In 2 horses (Nos. 4 and 21), plasma ACTH concentration more than doubled 4 hours after domperidone but diminished by 8 hours; this resulted in ACTH concentration >59 pg/ml in only 1 of the 2 (horse No. 4). Mean (median) 4 hour and 8 hour to resting ACTH concentration ratios were 1.5 (1.4) and 1.3 (1.4), respectively.
In the 5 horses with grade 3 pituitary glands, mean (median) ACTH concentration at 0, 4, and 8 hours was 27.3 (23.6), 60.4 (50.1), and 55.9 (64.4) pg/ml, respectively. None had an elevated baseline ACTH concentration, but, in 3 horses, the ACTH concentration exceeded the reference range after domperidone administration. In horse Nos. 9, 13, and 28, the plasma ACTH concentration was twofold or higher than baseline 4 hours after domperidone; in horse Nos. 9 and 28, it had nearly tripled by 8 hours. The mean (median) 4 hour and 8 hour to resting ACTH concentration ratios were 2.2 (2.0) and 2.0 (1.9), respectively.
In the 12 horses with grade 4 pituitary glands, mean (median) ACTH concentration at 0, 4, and 8 hours was 100.8 (52.8), 185.6 (132.0), and 204.7 (128.0) pg/ml, respectively. Six horses (Nos. 3, 5, 24, 26, 32, and 33) had a resting ACTH concentration >59 pg/ml. In horse Nos. 3 and 5, ACTH concentration more than doubled 4 hours after domperidone but diminished to a <twofold increase by 8 hours. The ACTH concentration in horse Nos. 26 and 33 had more than doubled by 8 hours after domperidone. In horse No. 32, the ACTH concentration had more than doubled by 4 hours and more than tripled by 8 hours after domperidone. In horse 24, baseline ACTH concentration was elevated but did not increase further after domperidone. In horses 7, 8, 10, 15, and 30, ACTH concentration was <59 pg/ml before domperidone administration but at least doubled at 4 or 8 hours after domperidone. ACTH concentration in horse No. 19 never doubled and never exceeded the reference range. Mean (median) 4 hour and 8 hour to resting ACTH concentration ratios were 2.1 (2.1) and 2.1 (1.9), respectively.
The grade 5 horses comprised the most diverse cohort. Mean (median) ACTH concentration at 0, 4, and 8 hours was 371.3 (357.7), 694.3 (699.5), and 689.5 (720.5) pg/ml, respectively. In the horse with a pars anterior adenoma (No. 1), resting ACTH concentration was <59 pg/ml but increased 4.2-fold at 4 hours after domperidone and was still 2.2 times the resting concentration at 8 hours. All 3 horses with PI adenomas (Nos. 20, 27, and 31) had elevated baseline ACTH concentration. Plasma ACTH concentration doubled by 8 hours after domperidone in horse No. 20. In horses No. 27 and No. 31, ACTH concentration exceeded the assay detection limit (1250 pg/ml) at 4 and 8 hours after domperidone.
Discussion
As in previous studies, 14,28 the age of the horses correlated strongly with pituitary histologic grade. On histologic examination, normal pituitary glands (grade 1) with a small PI were found in horses <12 years old; these horses had a minimal ACTH release in response to domperidone. Most 10- to 20-year-old horses had only focal or multifocal hypertrophy and hyperplasia of the PI (grade 2) with a ≤twofold increase in the PI area; these changes may be considered normal aging changes and were not associated with marked ACTH release after domperidone. In contrast, most study horses older than 20 years had pituitary adenomatous hyperplasia, microadenomas, or adenoma (grades 3, 4, or 5) and approached a threefold or greater increase in PI area and a twofold or greater increase in plasma ACTH concentration after domperidone, which exceeded the laboratory reference range.
The histologic finding of adenomatous hyperplasia, microadenomas, or adenoma in the pituitary PI has been considered the gold standard for the diagnosis of equine pituitary PI dysfunction. 19 Hypertrophy or proliferation of PI melanotrophs is thought to result in increased POMC production. 21,23,30 In this study of donated horses with and without clinical signs of PPID, a few horses had histologically normal pituitary glands (grade 1) and a few had pituitary adenomas (grade 5), but most horses had focal hypertrophy or hyperplasia, diffuse adenomatous hyperplasia, or microadenomas. Similar pathologic findings were previously described in horses and ponies with or without clinical evidence of PPID. 7,11,14,22,28 Adherence to the grading scheme used in this study resulted in good concordance between pathologists and a strong correlation with domperidone-evoked plasma ACTH concentrations. The scheme allowed ranking of hyperplasia in pituitary glands based on the presence or absence of certain histologic features, but measurement of PI area in histologic sections provided a continuous variable and more objective means to quantify the degree of hyperplasia. PI area measurements not only corroborated grading results but allowed ranking within grades and correlated strongly with domperidone-evoked plasma ACTH assay results. The presence of only focal hypertrophy or hyperplasia (grade 2) had little effect on the cross-sectional area of the PI, and grade 2 horses usually had minimal change in plasma ACTH concentration after domperidone administration. In contrast, diffuse adenomatous hyperplasia (grade 3) or the presence of microadenomas (grade 4) in the PI was usually associated with a threefold to fourfold increase in PI cross-sectional area. Only 1 grade 3 horse (the one with the smallest PI area) and 1 grade 4 horse (the one with large PI area and >fourfold elevation of baseline plasma ACTH concentration) failed to respond to domperidone with increased plasma ACTH concentration. The other 4 grade 3 horses experienced at least a 1.9-fold increase in ACTH concentration after domperidone administration. Of the remaining 11 horses with grade 4 pituitary glands, one responded to domperidone administration with a 1.8-fold increase in plasma ACTH concentration; the others had greater than twofold increases. Within histologic grades, ACTH concentration tended to increase with PI area. The largest PI area in grade 3 horses was 0.6 cm, and all grade 3 horses had normal baseline ACTH concentration. Four of the grade 4 horses had PI area smaller than 0.6 cm; 3 of the 4 had normal baseline ACTH concentration. In contrast, 4 grade 4 horses had PI area larger than that of the grade 5 horse with the smallest PI adenoma. Three of those 4 horses also had either higher baseline or domperidone-provoked concentration of ACTH. Though few in number, the grade 5 cohort had considerable variation in postdomperidone plasma ACTH concentration and in PI cross-sectional area. The 1 horse with a pars anterior adenoma had only diffuse microadenomatous hyperplasia of the PI, which may explain why its plasma ACTH concentration and PI cross-sectional area measurements fell within the range of values for these variables in grade 3 horses. The other 3 grade 5 horses had PI adenomas that varied considerably in size. The horse with the smallest PI adenoma had plasma ACTH concentration and PI area measurements within the range for these variables in grade 4 horses, whereas the 2 horses with large adenomas had marked increases in PI area measurements and in resting and domperidone-provoked plasma ACTH concentration. This variation in adenoma size and ACTH response to domperidone in the grade 5 horses plus the tendency for PI area in grade 4 horses to increase with increased resting or domperidone-provoked plasma ACTH concentration emphasize the importance of morphometry in conjunction with histologic grading to validate clinical tests of pituitary function.
In this study, consensus in grading between the 2 pathologists was high, although there was discordance between grading and the original histologic diagnosis in the 9 archived cases. This is not surprising, because the original diagnosis of pituitary lesions in the 9 archived specimens was not based on the grading scheme used in this study. Discordance among pathologists in interpretation of pituitary lesions in early PPID has been recognized and stresses the need for standardized classification of normal and pathologic changes in the pituitary gland of aging equids. 19
Pituitary weight, dimensions, and histologic features were evaluated in 100 Dutch Warmblood horses without overt clinical PPID and were compared with those in 19 Dutch Warmbloods and 17 ponies with PI adenoma. 28 Histologic lesions in the PI and pars distalis (pars anterior) in the 100 horses without overt PPID were graded as 0, no significant lesions; 1, single or multiple cysts; 2, focal or multifocal hyperplasia; 3, single or multiple microadenomas (<0.5-cm diameter); and 4, adenoma. Histologic grading in the current study was based in part on the Dutch Warmblood study, 28 with a few differences: 1) In this study, single or multiple cysts were noted in pituitary glands with all degrees of PI hyperplasia and were not a criterion for grading; 2) focal or multifocal hyperplasia (our grade 2/5) was distinguished from diffuse hyperplasia (our grade 3/5) of the PI; 3) less emphasis was allotted to changes in the pars anterior. In the van der Kolk study, the prevalence of pituitary lesions in horses without clinical signs of PPID increased with increasing age and 5 of 69 mares had pituitary adenoma. 28 In another study, 5 of 165 Thoroughbred mares without clinical evidence of PPID had PI adenomas. 22 In the study reported here, all 3 horses with PI adenomas had hirsutism; 2 were mares and 1 was a gelding.
The vast majority of equine pituitary adenomas develop in the PI, so the presence of a pars anterior adenoma in the current study was unexpected and not anticipated in the development of the grading scheme. However, in a study of 42 donated horses, 17 of which had adenomas or microadenomas in the PI, one also had a microadenoma in the pars anterior. 14 Milder hyperplastic changes were also common in the pars anterior in this study, but multinodular hyperplasia of the equine pars anterior has been recognized as a common but nonfunctional lesion in older horses, 15 and histologic grading in the current study was based on changes in the PI except for the 1 horse with a pars anterior adenoma. Lesions in the pars anterior of other horses in this study did not correlate strongly with pituitary histologic grade, ACTH assay results, or clinical evidence of PPID.
Degenerative changes were observed in the pars nervosa of some horses but were not strongly associated with histomorphometric features or ACTH concentration. Hypothalamic degeneration was not found in horses in this study with 2 exceptions: 1) a horse with multicentric lymphoma that involved the brain (but not the pituitary gland), and 2) the horse with the largest PI adenoma, which compressed the hypothalamus. Although degeneration or loss of dopaminergic hypothalamic neurons has been sought as the reason for dysregulation of PI melanotrophs, hypothalamic lesions were not detected in another study of horses with pituitary PI hyperplasia or adenoma. 7
Only 1 horse in this study had an adrenocortical adenoma; adrenal glands were unremarkable in the other 32 horses. Adrenocortical hyperplasia was also rare in other pathologic studies of horses with PI hyperplasia or adenomas. 7,15 Therefore, equine pituitary PI dysfunction is considered a more appropriate term than equine Cushing's disease (or hyperadrenocorticism), because the equine syndrome is distinct in clinical, biochemical, and pathologic features from canine or human Cushing's disease. 7,13,16,25 In addition, the term “dysfunction” is more appropriate clinically than “adenoma,” because biochemical changes in response to hormone receptor modulation may occur without actual adenoma formation in horses with PI hyperplasia.
Because mature horses were selected and because many donated horses were old, the age of the population in this study was skewed toward geriatric horses. Nevertheless, age was the clinical variable with the strongest correlation to pituitary histologic grade. In a study of 100 horses without clinical signs of PPID, the prevalence of lesions in the PI and pars anterior increased with increasing age, except in pregnant or lactating mares, which had the heaviest pituitary glands and in which the apparent predilection for PI lesions was attributed to the influence of ovarian hormones. 28 The investigators concluded that pituitary PI lesions without associated clinical disease were common in older horses, especially mares. Although female predilection for pituitary lesions or PPID was suggested, 15 other studies have not corroborated this. 14,25 Except for a 60-day pregnancy in 1 grade 1 horse, no pregnant or lactating mares were included in our study; sex-based predilection for pituitary lesions was not apparent. Breed was not obviously associated with pituitary histologic grade, PI area, or plasma ACTH concentration.
Although lameness, particularly chronic laminitis, 11 is a frequent clinical sign in horses with PPID and was noted in the history of half of the grade 4 horses in this study, laminitis was noted in only 1 grade 3 and 1 grade 5 horse and did not correlate strongly with either plasma ACTH concentration or pituitary histomorphometric features. However, hirsutism is considered almost pathognomonic for PPID and, though not consistently present, was limited to animals with pituitary grade ≥3 and was obvious in all 3 horses with PI adenoma.
Pituitary weight tended to increase with increasing pituitary grade but was an insensitive indicator of PI size. Morphometric determination of the pituitary area in histologic sections had the advantages of specifically quantifying the PI, and strong correlation with both histologic grade and plasma ACTH response to domperidone. Furthermore, provided the pituitary gland is sectioned through the midline sagittal plane, the determination of PI area can be performed retrospectively in HE-stained sections.
Of the 3-dimensional measurements of the equine pituitary gland, height may be the most useful to distinguish horses with PPID from normal horses but fell within the normal reference range in 18% of mares and 25% of geldings with PI adenoma. 28 Estimates of pituitary height by computed tomography were less accurate than estimates of pituitary length or width when compared with measurements made at postmortem examination. 20 This suggests that noninvasive determination of pituitary height would be difficult. In the study reported here, pituitary height correlated with pituitary grade; however, height of the gland did not always change proportionally with PI area and overlapped considerably among histologic grades.
Concordance between pathologists in this study was high, because the grading scheme was based on the presence or absence of defined criteria. Image analysis was performed after histologic grading to quantify pituitary hyperplasia and demonstrated the range in the degree of PI hyperplasia in each pituitary grade with overlap between grades. Area measurement provides an objective quantification of hyperplastic changes in the PI to corroborate histologic grading and to validate antemortem tests of PI function.
The dexamethasone suppression test is commonly used for the diagnosis of PPID but may have a low sensitivity in horses without obvious clinical signs, such as hirsutism, 14 and false-positive reactions, especially in autumn. 12,14 Corticotrophs, the predominant cell of the pars anterior, respond to hypothalamic arginine vasopressin and corticotrophin releasing hormone by release of POMC, which undergoes post-translational proteolysis, mainly into ACTH, β-lipotrophin, and β-endorphin. 23,26,30 In contrast, melanotrophs are the predominant cell type of the PI. Melanotrophs also produce POMC but do not respond directly to negative feedback in the hypothalamic-pituitary-adrenal axis, 6 and most of the POMC produced in the PI is proteolytically cleaved into ACTH then further cleaved into α-MSH, β-endorphin, and CLIP, with little ACTH remaining. 2,23,26 Most cells in the normal or hyperplastic equine PI are strongly immunoreactive for POMC, α-MSH, and β-endorphin, with weaker reactivity for ACTH. 2,7,15,22,25 Bioactive ACTH is scanty in the PI of healthy equids, because it is further processed into α-MSH and bio-inactive CLIP; however, in PI adenomas, although bioactive ACTH concentration is not elevated, the total tumor ACTH content is greater than that in normal whole pituitary gland. 24 The fact that immunoreactive ACTH concentration is elevated in the tumors and in the plasma of horses with PI adenomas indicates that some fraction of the ACTH is bio-inactive. 23,24,30
The current study was performed to evaluate the usefulness of measuring domperidone-evoked plasma ACTH concentration in horses without hirsutism or other obvious clinical signs of advanced PPID. Horses were tested in all 4 seasons, but the effect of season on either resting plasma ACTH concentration or ACTH response to domperidone administration was not evaluated. Pituitary glands were evaluated postmortem to determine the relationship between domperidone-evoked ACTH concentration and pituitary histomorphometric features. Both histologic grade and the area of the pituitary PI in sagittal section correlated positively and strongly with changes in plasma ACTH concentration in response to domperidone administration.
Horses with PPID often have increased ACTH concentration in the PI and in plasma, but the use of the resting plasma ACTH concentration to diagnose PPID has met with variable success. 9,11,25 An assay of domperidone-evoked changes in plasma ACTH concentration is a novel test focused on the dopaminergic regulation of the pituitary PI. In this study, resting ACTH levels were used mainly as the baseline to quantify the response to domperidone. Resting ACTH concentration was above the laboratory reference range in only 1 of 12 horses with pituitary histologic grade 1 or 2 and that grade 2 horse had minimal increase after domperidone administration. Grade 1 or 2 is considered to be within normal limits, so the elevated resting ACTH concentration would be considered a false positive. Grade 3 pituitary glands have diffuse PI adenomatous hyperplasia, which resulted in at least a 1.9-fold increase in ACTH concentration after domperidone in 4 of 5 horses. However, all 5 grade 3 horses had a resting plasma ACTH concentration within the laboratory reference range. Thus, resting ACTH concentration was an insensitive test to detect horses with mild PI lesions but was more likely to be elevated in horses with more severe PI lesions. Six of 12 horses with PI microadenomas (grade 4) and 3 of 4 horses with adenoma (grade 5) had an elevated resting ACTH concentration. Other studies reported a normal resting ACTH concentration in horses with clinical signs of PPID. 11,14 The observation that resting plasma ACTH concentrations were within normal limits in all grade 3 horses and half the grade 4 horses in the current study supports the idea that an evocative agent is needed to distinguish affected from normal equids and is reminiscent of other evocative endocrine assays, such as the human chorionic gonadotrophin test to distinguish cryptorchid stallions from geldings 27 or the combined dexamethasone suppression/thyrotropin-releasing hormone stimulation test for PI adenoma. 14
In summary, results of this study suggest that the domperidone-provoked ACTH assay is promising for the diagnosis of PPID and will be particularly useful in detecting horses with mild PI lesions that have not developed hirsutism or other obvious clinical signs of PPID. Domperidone is commercially available for use in horses and can be administered orally in paste formulation with completion of blood collection for the ACTH assay in 8 hours. The test should be initiated at a standard time to avoid diurnal variation in plasma ACTH concentration; plasma should be promptly harvested and frozen for accurate results. Further studies are needed to evaluate any seasonal effect on ACTH response to domperidone, to determine the effect of repeated testing, and to compare the ACTH response in domperidone-treated horses with that in vehicle-treated control horses. Functional assays should be validated by standardized postmortem histomorphometric evaluation of the pituitary gland. The pituitary grading scheme used in this study is easily performed in HE-stained sagittal tissue sections and correlated well with functional assessment of the PI.
Footnotes
Acknowledgements
We thank Jessica Brooks for assistance with image analysis; technical staff of the Clinical Pathology Laboratory, School of Veterinary Medicine, for plasma ACTH assays; pathologists, pathology residents, and histotechnicians of the Animal Disease Diagnostic Laboratory, Purdue University, for assistance in tissue collection and histologic slide preparation. This work was supported in part by the American College of Veterinary Internal Medicine (ACVIM) and the American Quarter Horse Association. Dr. Pardo's work was supported by a scholarship from Pfizer, Inc., and the American College of Veterinary Pathologists (ACVP). Portions of this study were presented at the 2006 annual meetings of the ACVIM and ACVP.
