Abstract
Plasma adrenocorticotropic hormone (ACTH) concentration is used in the diagnosis of pituitary pars intermedia dysfunction (PPID) in horses. We enrolled 10 horses, 5 PPID-positive and 5 PPID-negative, in our study, September 20–22, 2016. On day 0, 5 mL of whole blood was collected into each of 6 EDTA tubes and immediately placed in a refrigerator at 7°C. One tube was centrifuged within 15 min of collection, followed by centrifugation of one tube from each horse at 4, 8, 12, 24, and 36 h following collection. At each time, centrifuged plasma was pipetted into 1.5-mL polypropylene tubes and stored at −80°C. None of the plasma samples were turbid, hemolyzed, or icteric. Plasma was shipped frozen with cold packs overnight to the Animal Health Diagnostic Center of Cornell University (Ithaca, NY) for analysis. The percent change from baseline (PCFB) was reported to standardize the data given that baseline values differed. The mean PCFB was 2.8 (95% confidence interval: –2.9%, 7.0%). Neither refrigeration of whole blood for up to 36 h prior to centrifugation nor freezing affected plasma ACTH concentrations significantly.
Pituitary pars intermedia dysfunction (PPID) is considered the most common endocrinologic disorder of aged horses.1,7,12 The diagnosis of PPID is typically based on history, clinical signs, and demonstration of endocrinologic dysfunction via assay results. 2 In addition to clinical signs, the laboratory test used most commonly by veterinary practitioners in the diagnosis of PPID is measurement of basal plasma adrenocorticotropic hormone (ACTH) concentration. 2 Determination of basal plasma ACTH concentration has been the most practical test for PPID, likely given the ease of blood collection and the availability of seasonal reference intervals that allow for testing at any time of the year.4,12
Multiple recommendations exist concerning the in vitro stability of ACTH. Recommendations state that samples should be chilled within 3 h of collection.3,4 This can be done by placing samples in ice water, a refrigerator, or insulated cooler promptly after collection (https://ahdc.vet.cornell.edu/docs/Equine_Cushings_Tests.pdf). Human and equine plasma samples with an ACTH concentration in the normal range undergo significant reduction by 24 h if kept at 21–24°C.5,9 In healthy horses, no significant interactions were identified when the stability of ACTH in whole blood stored at 4°C was determined over 72 h. 9 In the United States, a discordance exists between laboratory recommendations concerning the time lapse between sample collection and centrifugation; a range of 2–4 h from sample collection is advised. Many equine ambulatory practitioners do not have the ability to centrifuge the sample within 2–4 h. Based on this dilemma, we evaluated the stability of ACTH in refrigerated blood samples from PPID and healthy non-PPID horses with increasing time from collection to centrifugation.
Our study was performed in accordance with the policies of the Institutional Animal Care and Use Committee of Middle Tennessee State University (Murfreesboro, TN) under protocol 17-2002. Five PPID-positive and 5 PPID-negative horses (status based on ACTH concentrations and clinical signs) were enrolled in the study in September. On day 0, 5 mL of whole blood was collected from each horse into each of 6 EDTA tubes and immediately chilled in a refrigerator at 7°C. One tube was centrifuged within 15 min of collection (time 0) followed by centrifugation of one tube from each horse at 4, 8, 12, 24, and 36 h after collection. At each time, blood was centrifuged and plasma was transferred into snap-top, 1-5 mL polypropylene tubes and stored at −80°C. None of the plasma samples were turbid, hemolyzed, or icteric. Plasma was shipped frozen with cold packs overnight to the Animal Health Diagnostic Center, Cornell University (Ithaca, NY). Samples were batch-analyzed for plasma ACTH concentration by chemiluminescent immunoassay that had been validated previously for horses.
8
The percent change from baseline (PCFB) was reported to standardize the data given that baseline values differed. Mean PCFB, standard deviation, and 95% confidence intervals (CI) were reported. The null hypothesis was that there was no time effect on plasma ACTH concentrations, and this was tested with a mixed-model, repeated-measures methodology with “subject” as a random effect.
The absolute changes over time revealed no consistent pattern of variation (Fig. 1). The mean PCFB was 2.8 (95% CI: –2.9%, 7.0%). There was not enough evidence of significant time effect from the repeated-measures model (
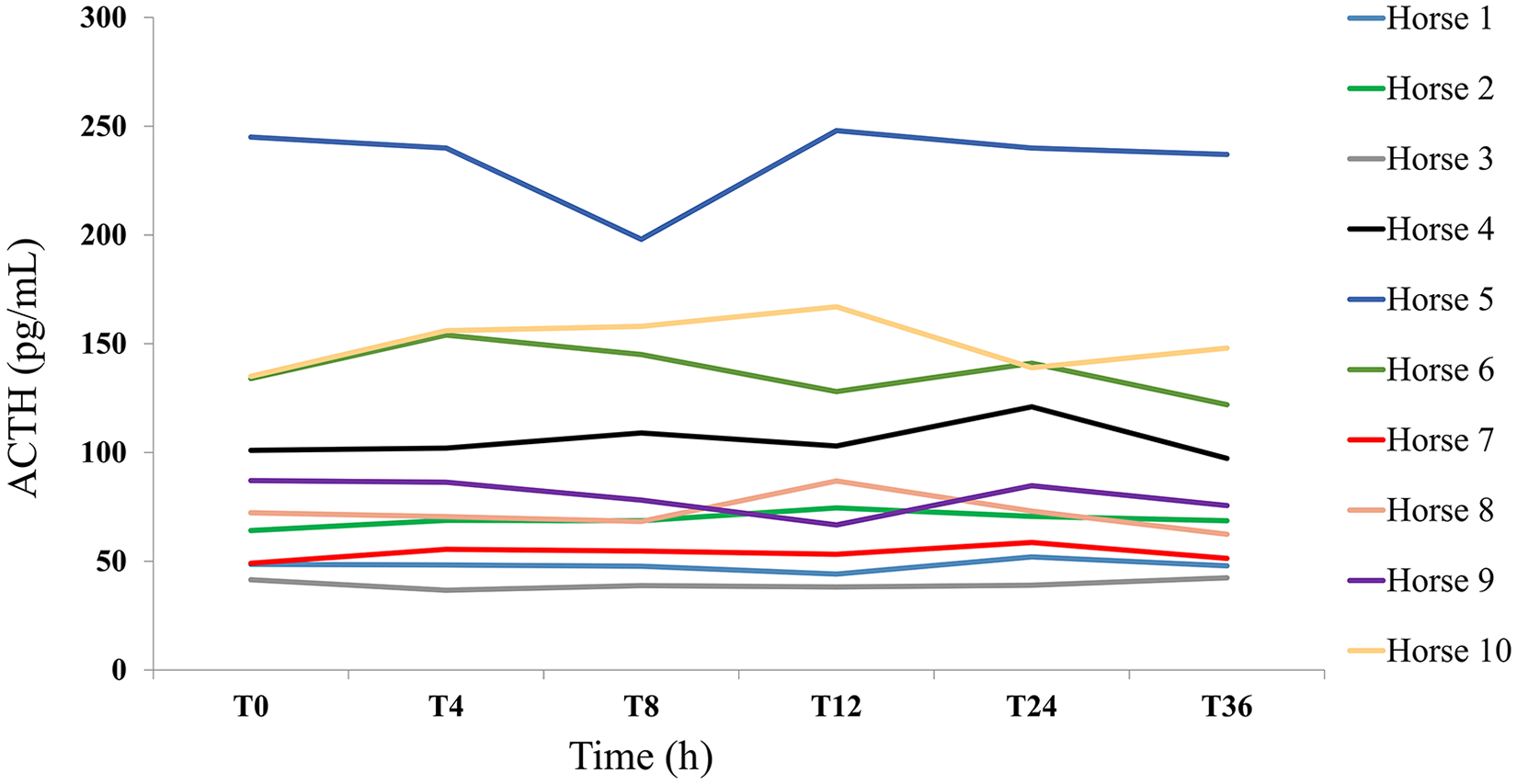
Absolute changes in adrenocorticotropic hormone (ACTH) concentration from baseline, in pg/mL, in blood samples maintained at 7°C. Lines represent the ACTH concentration over time (0–36 h; T0–T36) for each horse.
In human samples, hemolysis results in a marked reduction in ACTH in plasma samples, but overall, the mechanism for degradation of ACTH has been considered largely unknown and has been attributed primarily to proteolytic degradation.5,6 The addition of
Our study demonstrated that refrigeration of whole blood for up to 36 h prior to centrifugation and freezing did not significantly affect plasma ACTH concentrations. Laboratory results alone should not be utilized to classify a horse as PPID-positive or -negative. History and clinical signs in conjunction with laboratory results should always be utilized for the diagnosis of PPID. Although we included both PPID-negative and -positive horses in our study, further investigations could be conducted in a larger population of PPID-negative and -positive horses, particularly to include more horses with equivocal results. If equivocal results are obtained in clinical practice, further testing has been recommended using the thyrotropin-releasing hormone stimulation procedure.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was funded through Boehringer Ingelheim Vetmedica (Duluth, GA) and The John C. Miller Chair of Equine Reproduction at Middle Tennessee State University (Murfreesboro, TN).
