Abstract
Kidney samples with interstitial nephritis from 26 pigs affected by postweaning multisystemic wasting syndrome (PMWS) were selected. A histologic evaluation was carried out to describe the type of inflammation and its relationship with viral load, as assessed by in situ hybridization (ISH). Of 26 cases, 10 revealed a tubulointerstitial, lymphoplasmacytic nephritis, 11 an interstitial granulomatous nephritis, and 5 both types of inflammation (mixed type). In 4 cases of granulomatous inflammation, the pattern was not classically nodular, and a population of macrophages and lymphocytes was present (interstitial lymphohistiocytic nephritis). ISH confirmed the presence of porcine circovirus type 2 (PCV2) nucleic acid in all cases. The epithelium of the renal tubules was the most constantly ISH-positive structure. In tubulointerstitial nephritis, the higher the number of positive inflammatory cells, the more severe the inflammation. The ISH reaction was more heterogeneous and unpredictable in granulomatous nephritis, with some epithelioid and giant cells positive by ISH. To quantify macrophages distributed in the three patterns of nephritis, immunohistochemical methods using anti-major histocompatibility complex II (anti-MHC-II) and anti-lysozyme antibodies were undertaken, and semiquantitative evaluation was carried out. MHC-II was mainly expressed by lymphocytes in tubulointerstitial nephritis, but did not always stain macrophages in cases of granulomatous (including lymphohistiocytic) nephritis; the anti-lysozyme antibody revealed macrophages when present in tissues. The amount of PCV2 nucleic acid was not apparently associated with the pattern of inflammation (tubulointerstitial or granulomatous). PCV2 load seems to reflect the severity of the lymphoplasmacytic inflammation but not that of granulomatous and lympho histiocytic types.
Porcine circovirus type 2 (PCV2) has been etiologically linked with different pathologic conditions. 4 The most studied one is postweaning multisystemic wasting syndrome (PMWS), which is clinically characterized by wasting and respiratory distress, and is pathologically defined by its lymphoid lesions, specifically, lymphocyte depletion and granulomatous infiltration. 19 Many organs besides lymphoid tissues can be damaged in PMWS-affected pigs; one of these is the kidney, in which interstitial nephritis has been described in about 40% of the diseased animals. 21 These lesions tend to vary widely in intensity, from very mild to severe damage and from multifocal to diffuse distribution. Histologic examination of these affected kidneys can reveal up to four main types of infiltrating cells, namely, lymphocytes, plasma cells, macrophages, and multinucleate giant cells, 6,18 in variable proportions. The described cases can be pathologically defined as lymphoplasmacytic to granulomatous nephritis.
Different types of cells of the interstitial inflammatory infiltrates or the specific structural compounds of the kidney can contain PCV2 in PMWS-affected pigs. Specifically, viral antigen and/or nucleic acid have been detected in the cytoplasm and/or nuclei of epithelial renal tubular, endothelial, monocyte-macrophage lineage, and multinucleate giant cells. 10,18 However, PCV2 is not always found in all cases of interstitial nephritis.
To date, no detailed pathologic or pathogenic studies have been focused on the occurrence of interstitial nephritis in PMWS-affected pigs and the role of PCV2 in the development of those lesions. Therefore, the objectives of present study were 1) to pathologically describe in detail the features of interstitial nephritis that occur in naturally occurring PMWS cases, 2) to ascertain if the type of renal inflammation is influenced by the viral load, and 3) to evaluate the type of inflammatory cells involved in nephritis lesions by immunohistochemical methods.
Materials and Methods
A total of 26 naturally occurring PMWS cases with lesions of interstitial nephritis were selected for this study. Pigs were aged 5–20 weeks, and they were submitted for necropsy to the Veterinary Schools of Bologna, Italy (n = 7), and Barcelona, Spain (n = 19). PMWS was diagnosed based on previously accepted diagnostic criteria; 20 selected pigs had wasting and respiratory distress, moderate to severe lymphocyte depletion and/or granulomatous inflammation of lymphoid tissues, and moderate to high amounts of PCV2 genome or antigen in the lymphoid lesions.
Kidney sections were fixed by immersion in 10% neutral-buffered formalin. Fixed samples were dehydrated, embedded in paraffin wax, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). Renal tissue was assessed for the type (lymphoplasmocytic, granulomatous, or mixed) and distribution (multifocal or diffuse in renal cortex, medulla, and/or pelvis) of inflammatory infiltrates.
PCV2 nucleic acid was detected in kidney slides by an in situ hybridization (ISH) technique using previously published methods and probe. 9,18 Sections from the lymph nodes of the same tested animals (diagnosed as PMWS) were used as positive controls for the ISH technique. Negative control consisted of a lymph node tissue from a pig with no lesions of PMWS and negative by a PCV2 polymerase chain reaction method. The presence of viral genome was graded in high (+++), moderate (++), or low (+) amounts for each kidney sample, following previously published semiquantitative methods. 15,17
Two different cell markers were used to classify different cell phenotypes present in the inflammatory lesions of selected kidneys by immunohistochemical methods. Major histocompatibility complex II (MHC-II) antigens, known to be expressed on antigen-presenting cells (APCs), resting B lymphocytes, and activated lymphocytes and macrophages, were identified with an anti–SLA-II-DQ (BL2H5) antibody. Macrophages also were detected with anti-human lysozyme antibody. 5 Sections were dewaxed in toluene and rehydrated in a graded alcohol series; blocking of endogenous peroxidase was made, respectively, in 3% H2O2 and 0.3% methanol, and antigen unmasking was made with pronase and protease XXIV; and preincubation with protein block serum (Dako, Carpinteria, CA) was carried out. Antibodies were used undiluted and at dilution 1 : 400 (anti-human lysozyme) and incubated overnight at 4°C. A labeled streptavidin-biotin-peroxidase detection method (Dako) was employed. As a chromogen, diaminobenzidine (0.05% for 10 minutes at room temperature) was used; sections were counterstained with Harris hematoxylin, dehydrated, and mounted with DPX. Sections of normal kidneys from specific pathogen free (SPF) pigs were used as controls. As negative controls, irrelevant primary antibody at the same dilution was used in substitution of the specific primary antibody. The amount of APC was graded as follows: no staining (−; no cells at 25× magnification), very low (+/−; 1–2 cells at 25×), low (+; 2–5 cells at 25×), moderate (++; 5–10 cells at 25×), and high (+++; more than 10 cells at 25×). The amount of macrophages stained with the antilysozyme antibody was semiquantitatively evaluated as follows: very low (+/−; 0–3 macrophages at 25× magnification), low (+; 4–24 macrophages at 25×), moderate (++; 25–50 macrophages at 25×), and high (+++; more than 50 macrophages at 25×).
Results
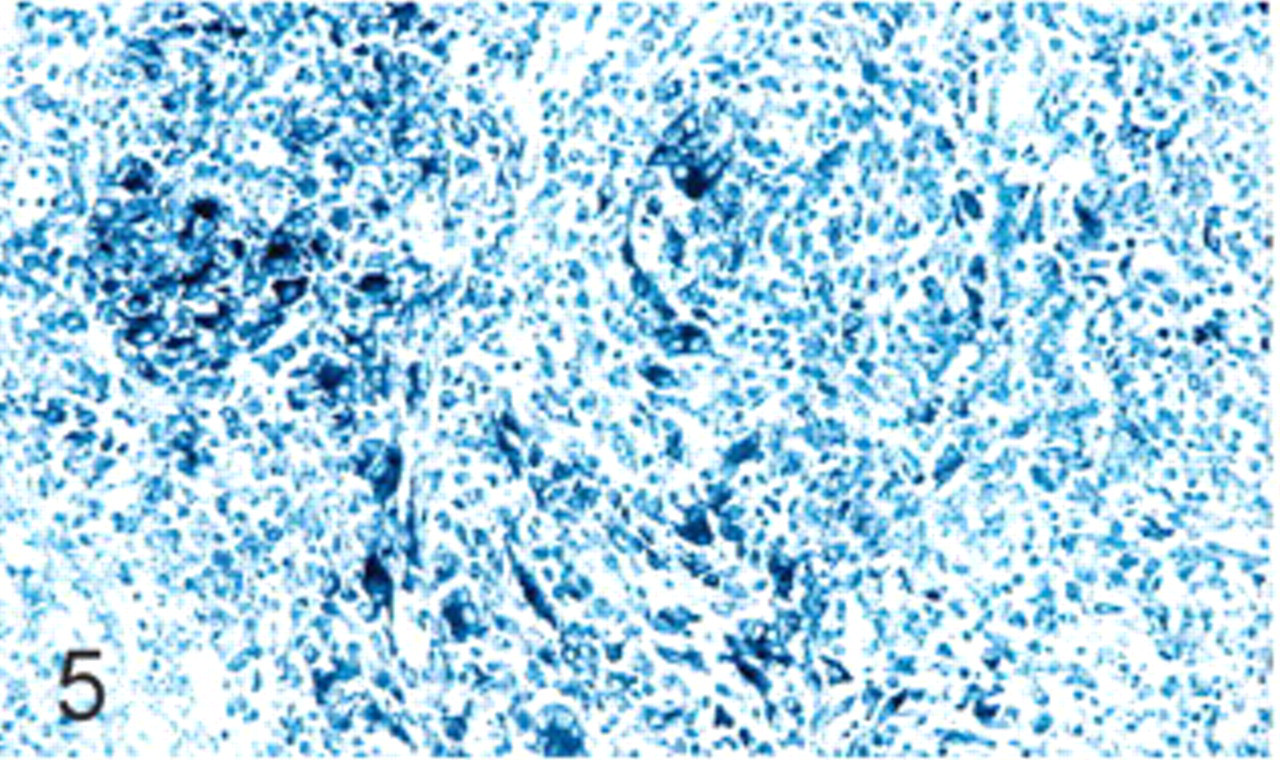
Results of histologic, immunohistochemical, and ISH studies are summarized in Table 1. When mild lesions were observed, they tended to be localized in deep cortex or corticomedullary junction of the kidney; moderate to severe lesions tended to have a multifocal to diffuse distribution. Interstitial nephritis cases were subclassified as lymphoplasmacytic (10 cases, 38.5%; Fig. 1), granulomatous (11 cases, 42.3%; Fig. 2), and mixed (5 cases, 19.2%). The latter group included lymphoplasmacytic as well as granulomatous inflammatory infiltrates in a similar amount. Lymphoplasmacytic inflammatory lesions were mild to moderate, whereas the granulomatous lesions were of moderate to severe intensity, usually with coalescent inflammatory foci. Granulomatous interstitial nephritis was characterized mainly by multiple nodular aggregations of regular and epithelioid-like macrophages and rare multinucleate giant cells. In four cases of granulomatous inflammation, the pattern was not classically nodular, and a mixed population of macrophages and lymphocytes was present (interstitial lymphohistiocytic nephritis; Fig. 3).
1. Results of histological classification, IHC and ISH∗
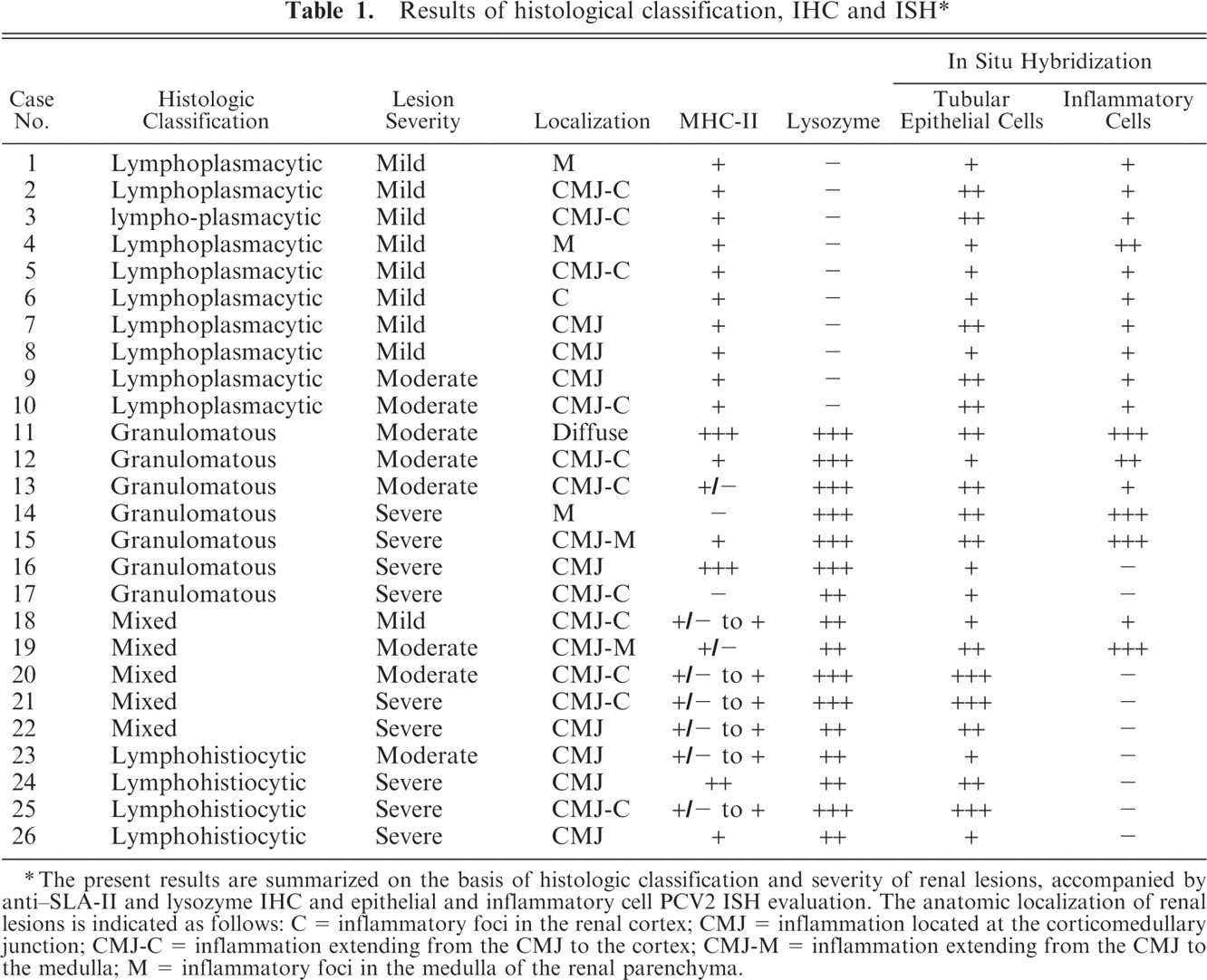
∗ The present results are summarized on the basis of histologic classification and severity of renal lesions, accompanied by anti–SLA-II and lysozyme IHC and epithelial and inflammatory cell PCV2 ISH evaluation. The anatomic localization of renal lesions is indicated as follows: C = inflammatory foci in the renal cortex; CMJ = inflammation located at the corticomedullary junction; CMJ-C = inflammation extending from the CMJ to the cortex; CMJ-M = inflammation extending from the CMJ to the medulla; M = inflammatory foci in the medulla of the renal parenchyma.
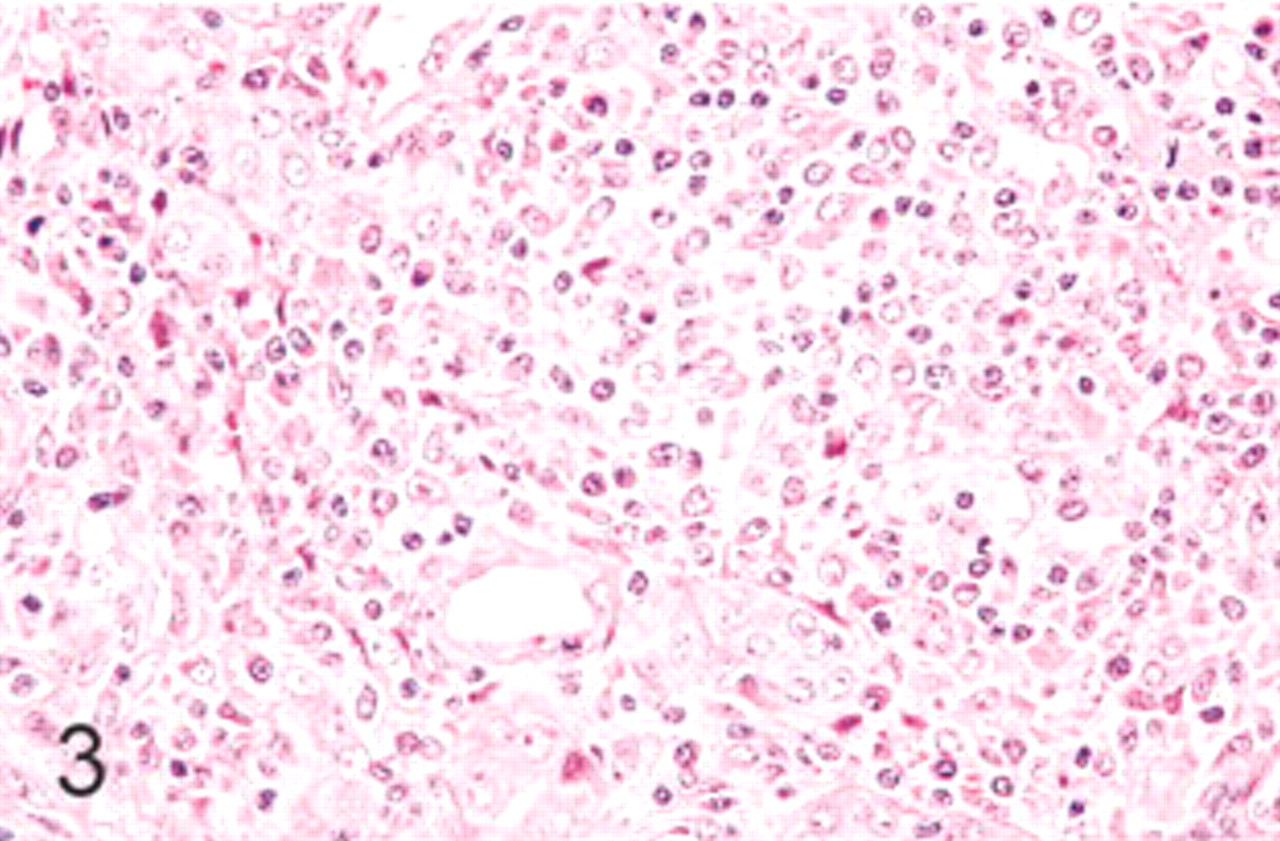
Kidney; swine. Lymphohistiocytic interstitial nephritis. HE.
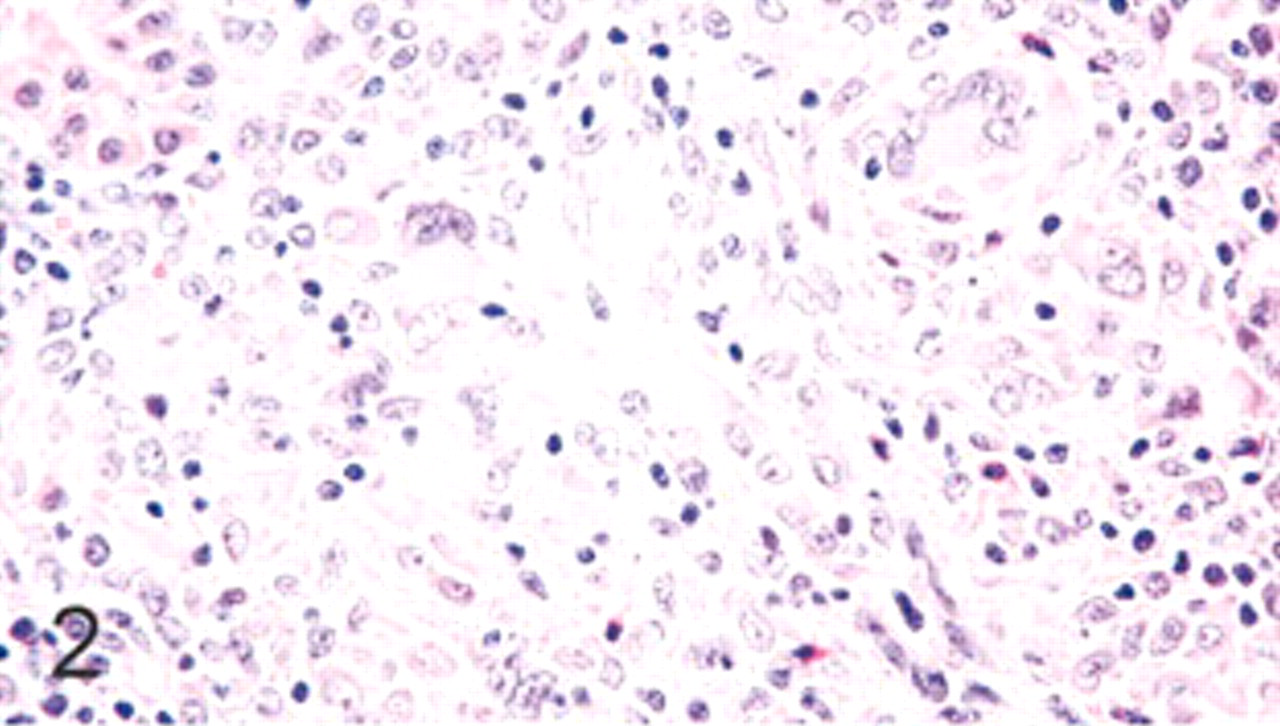
Kidney; swine. Granulomatous interstitial nephritis. HE.
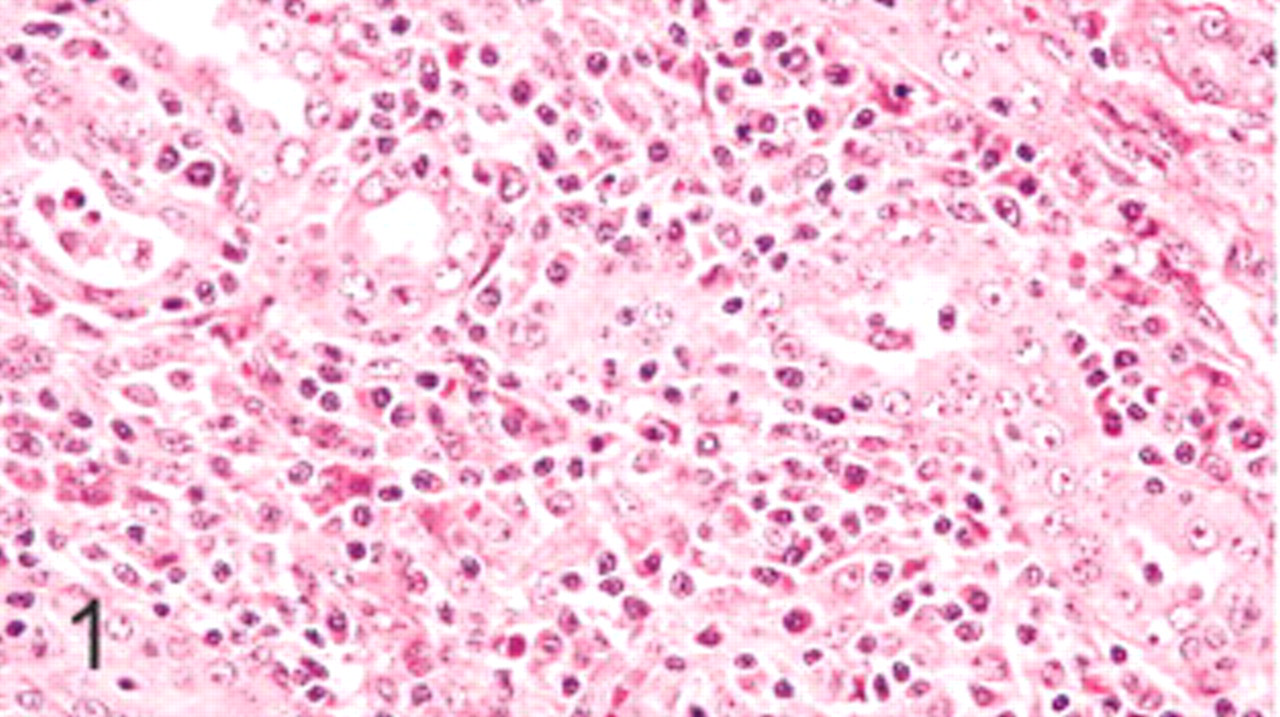
Kidney; swine. Lymphoplasmacytic interstitial nephritis. HE.
In all moderate to severe cases of interstitial nephritis, renal tubular epithelium showed degenerative changes characterized by hypereosinophilia of cytoplasm, pyknotic nuclei, and cell debris in the lumen. Regeneration of renal tubular epithelium was evident in those cases, with enlarged cells with pale cytoplasm, vesicular nucleus, and prominent nucleolus; sometimes, mitoses along the tubular wall were seen. No lesions were observed in glomeruli in any of the studied cases. Therefore, the involvement of tubules as well as the interstitium led us to classify the renal inflammatory lesions as a tubulointerstitial nephritis.
PCV2 nucleic acid distribution tended to follow the inflammatory pattern, with the corticomedullary junction the area with the highest amount of labeling in nearly all cases of nephritis. PCV2 nucleic acid was detected in the cytoplasm and/or nuclei of tubular epithelial cells as well as the cytoplasm (mainly) of inflammatory cells (lymphocytes and macrophages; Fig. 4). ISH positivity was detected also in cytoplasm and/or nuclei of intratubular desquamated cells and endothelial cells, and in the cytoplasm of mononuclear intravascular cells. Glomeruli were PCV2 ISH–negative in all cases. The amount of PCV2 in renal tubular epithelia was proportional to the intensity of the inflammatory reaction in lymphoplasmacytic nephritis; PCV2 nucleic acid was multifocally distributed in mild cases, localized mainly in the epithelium of collecting tubules, and the viral amount increased with the severity of lesions. As a general feature, PCV2 ISH labeling was more frequent in tubular epithelium than in inflammatory cells. Granulomatous and mixed nephritis showed a less predictive distribution of PCV2 genome, since 5 of these cases (4 granulomatous and 1 mixed) had a high amount of virus in inflammatory cells, and the rest (7 granulomatous and 4 mixed) had a low amount of (Fig. 5) or no PCV2 among cells of inflammatory infiltrates. The amount of viral genome in renal tubular epithelia was also variable in granulomatous and mixed nephritis.
Kidney; swine. Granulomatous interstitial nephritis. PCV2 nucleic acid in few macrophages in a granuloma. In situ hybridization, nitroblue tetrazolium staining, Fast Green counterstain.

Kidney; swine. Lymphoplasmacytic interstitial nephritis. PCV2 nucleic acid in the inflammatory infiltrate and in tubular epithelial cells. In situ hybridization, nitroblue tetrazolium staining, Fast Green counterstain.
The antibody to lysozyme stained the cytoplasm of rare granulocytes, epithelial cells of proximal convoluted renal tubules, and macrophages of granulomatous and mixed types of interstitial nephritis (n = 16, 61.5%; Fig. 6); this reactivity was generally strong and correlated with the number of macrophages present in the inflammatory infiltrates. Multinucleate giant cells contained also lysozyme in the cytoplasm (Fig. 7). No significant labeling was observed in cases of lymphoplasmacytic kidney inflammation.
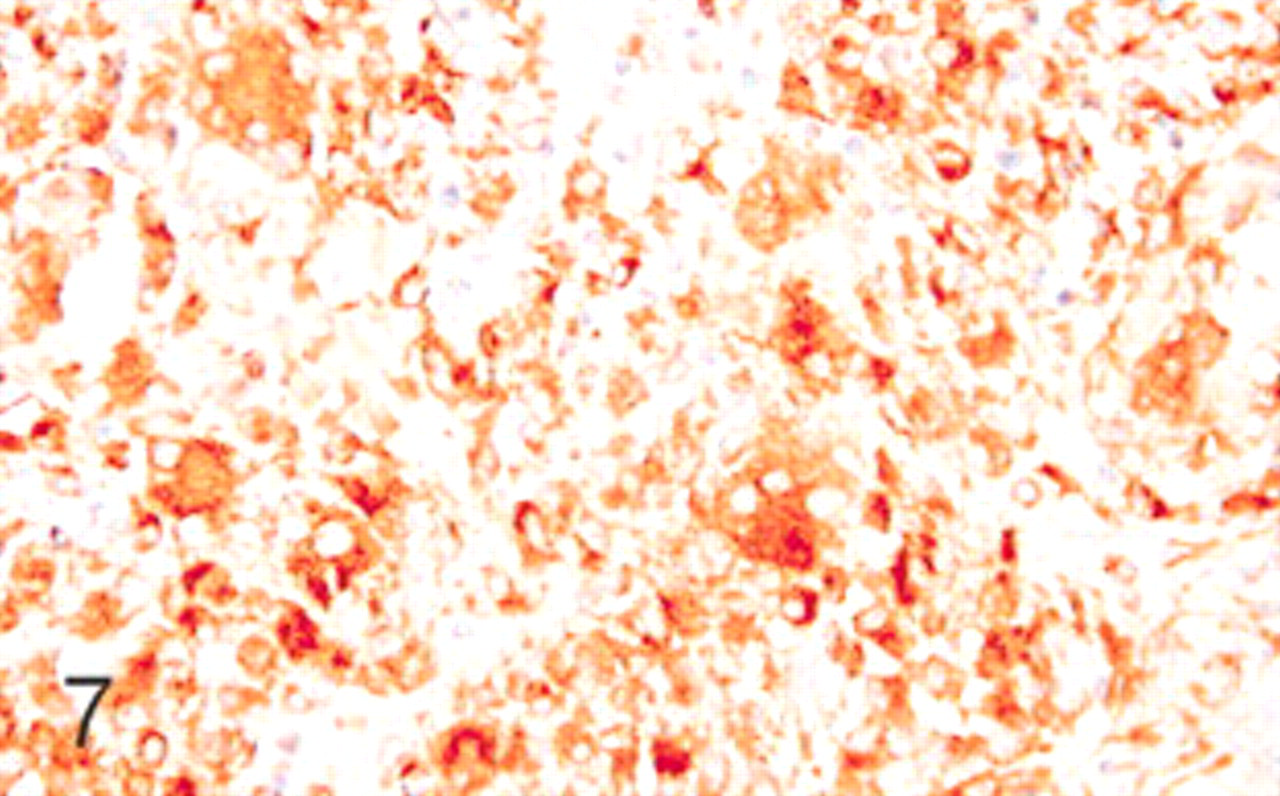
Kidney; swine. Granulomatous interstitial nephritis showing lysozyme positivity in most of the inflammatory cells. Immunohistochemistry for lysozyme, DAB staining, Harris hematoxylin counterstain.
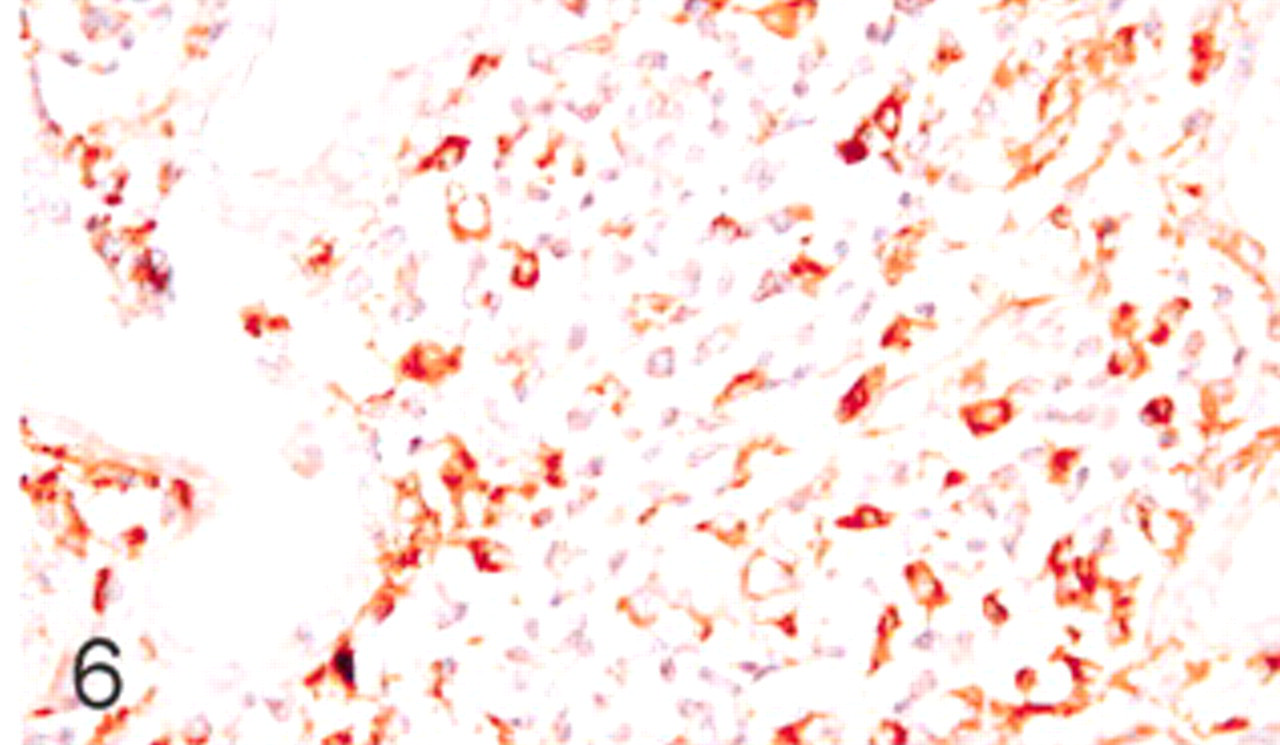
Kidney; swine. Lymphohistiocytic interstitial nephritis with numerous lysozyme positive inflammatory cells (histiocytes). Immunohistochemistry for lysozyme, DAB staining, Harris hematoxylin counterstain.
Positive BL2H5 cells were detected in 24 cases of nephritis (92.3%) and corresponded mainly to lymphocyte-like cells in all types of interstitial nephritis, generally in low to moderate numbers (Fig. 8). A variable proportion (low to high amounts) of macrophages and multinucleate giant cells of granulomatous and mixed inflammation types were BL2H5 positive, independently of the severity of the lesion. The two negative cases to BL2H5 antibody corresponded to granulomatous interstitial nephritis.
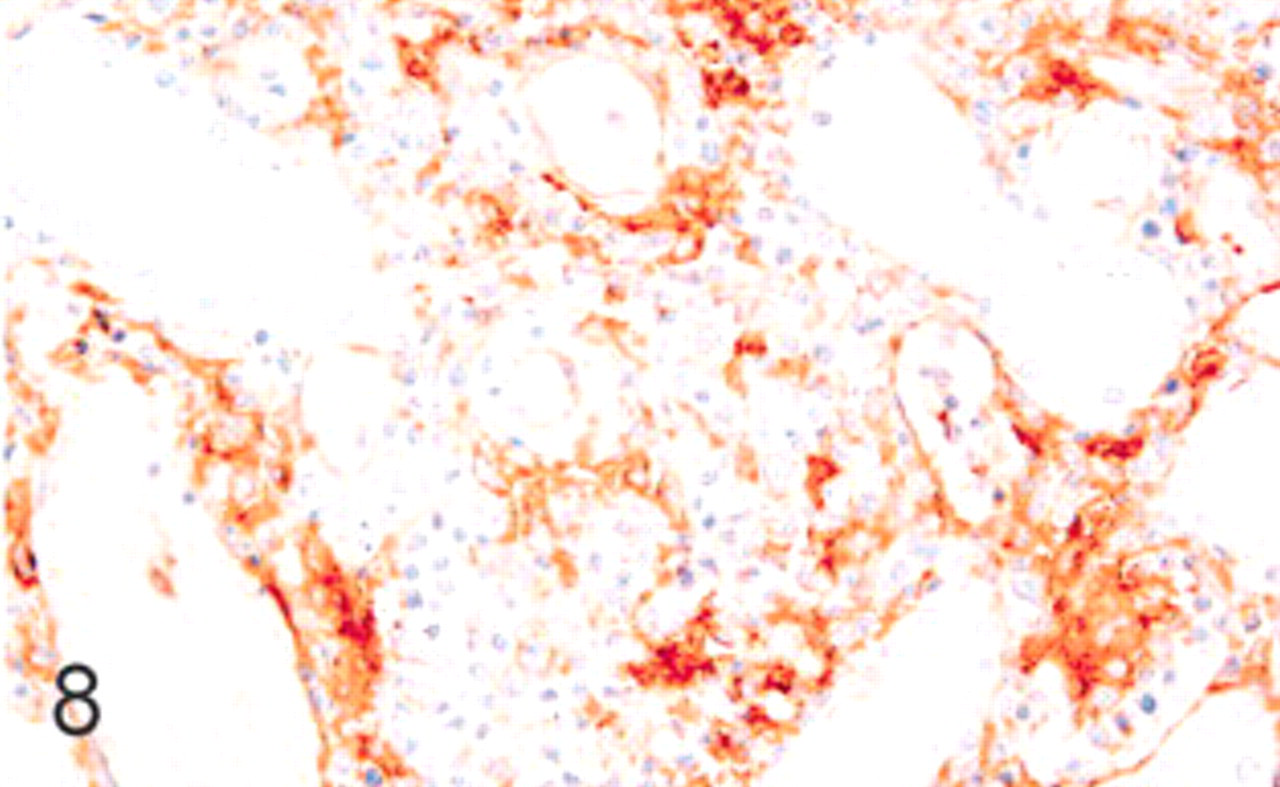
Kidney; swine. Lymphoplasmacytic nephritis with positive reaction in the inflammatory infiltrate. Immunohistochemistry for MHC-II, DAB staining, Harris hematoxylin counterstain.
Discussion
The present study further pathologically characterizes inflammatory lesions of the kidney that are observed in a proportion of PMWS-affected pigs. 21 The affection of interstitium as well as tubular epithelium, without affection of glomeruli, allowed us to classify the observed lesions as primary tubulointerstitial nephritis. 3 Two main types of tubulointerstitial nephritis lesions were described, which were characterized by the presence of lymphoplasmacytic and granulomatous inflammatory infiltrates or a mixture of them. The lymphoplasmacytic type of inflammation was usually of low severity, with low to moderate amounts of PCV2 in renal epithelial tubular cells and low numbers of PCV2-positive inflammatory cells, lack of significant numbers of lysozyme-containing cells, and low amounts of reactivity for MHC-II. The granulomatous type of tubulointerstitial nephritis was more heterogeneous and was characterized usually by moderate to severe lesional intensity and moderate to high numbers of cells containing lysozyme, very variable numbers of APCs (recognized by BL2H5 antibody), and variable numbers of inflammatory and tubular epithelial cells containing PCV2. Two cases of granulomatous nephritis did not react with the BL2H5 antibody; this was a surprising finding, which could be hypothetically explained by a PCV2-mediated decrease in activation or in expression of MHC-II molecules, as it has been demonstrated in lymphoid tissues. 13 The mixed tubulointerstitial nephritis pattern resembled the granulomatous one.
Although a trend was observed, no obvious apparent relationship was established between the severity of histologic lesions and the amount of PCV2 genome in the kidney. This is an apparently different feature than what is the hallmark in lymphoid tissues of PMWS-affected pigs: 5,18 the more severe the lesion, the higher the amount of PCV2. At this moment it is not possible to assess why only a proportion of PMWS-affected pigs develop renal lesions 21 and why two distinct inflammatory patterns of tubulointerstitial nephritis are seen in those pigs.
The functional unit of the kidney is composed of the nephron and the collecting tubules. 7 In the present study, both collecting tubules and some nephron sections (proximal and distal tubules) contained PCV2. In fact, the epithelial cells of the kidney revealed the most constantly positive structure by ISH, confirming these cells as kidney targets for PCV2 in PMWS cases. Since PCV2 genome was found in the cytoplasm and also in the nuclei of a proportion of renal tubular epithelial cells, this result strongly supports the idea that these cells support PCV2 replication. This fact would not be surprising, since the ability of PCV2 as well as PCV1 to replicate on primary and continuous kidney cell cultures is well known. 2
The presence of macrophages in the inflammatory infiltrates of the kidney in PMWS-affected pigs seems to be an inconstant feature in the present study, since cases of lymphoplasmacytic, tubulointerstitial nephritis did not apparently contain many cells with the morphology of macrophages. This situation, together with the variable amount of PCV2 within inflammatory lesions, may represent different pathogenic mechanisms of tubulointerstitial nephritis in PMWS. Two main potential mechanisms, not mutually exclusive, would be involved: 1) viral direct damage to the tubular renal epithelium and secondary inflammation, and 2) systemic inflammation with extension to the kidney. The former would explain the morphologic alterations seen in the renal tubuli associated with PCV2 infection and the presence of a dominant lymphoplamacytic inflammatory reaction close to the damaged tubuli. The latter would explain a predominant macrophage infiltration, as observed in many other tissues of PMWS-affected pigs, 6,18 probably due to a type IV hypersensitivity (cell-mediated) reaction, as has been suggested. 12
The lack of a consistent relationship between granulomatous inflammation and PCV2 presence can be considered relatively surprising, based on other studies in lymphoid lesions. 5,18 However, since hypersensitivity reactions are elicited by the individual reactivity to an antigen, 1 a severe granulomatous inflammation could take place independently from the amounts of virus in some subjects. On the other hand, it is possible to assume that besides PMWS, these pigs potentially suffered from other unrelated granulomatous inflammatory processes of the kidney. PCV1 is able to modulate MHC expression: upregulation of MHC-I and downregulation of MHC-II in alveolar macrophages. 16 Reduced expression of MHC-II molecules also revealed an important step in PMWS lymphadenopathy, 13 confirming its importance in PMWS lesions. The low MHC-II expression in macrophages of granulomatous and lymphohistiocytic nephritis would be related with this pathogenetic mechanism. The low level of MHC-II expression may also explain the low level of viral load, since it is known that macrophage activation is an important step in allowing PCV2 replication. 11
Finally, it must be emphasized also that tubulointerstitial nephritis can frequently occur in pigs without PMWS; 8,14 therefore, other potential causes of renal disease may have been contributory to the observed lesions in some animals.
