Abstract
Neoplastic cells and stem cells share several phenotypic characteristics. Recently, numerous studies have identified adult stem cells that have been hypothesized to be the cellular origin for cancer in several tissues. Oct4 has been consistently associated with pluripotent or stemlike cells, and it is hypothesized that Oct4 is necessary for the maintenance of pluripotency. We hypothesize that Oct4-positive cells are present in all canine neoplasms and that these subpopulations of neoplastic cells might represent “cancer stem” cells. To test this hypothesis, 83 canine neoplasms representing 21 neoplastic diseases were evaluated for Oct4 expression using immunohistochemistry. The results of this study showed that all tumors included in this study contained a subpopulation of Oct4-positive cells, although the proportion of Oct4-positive cells and the intensity of immunoreactivity varied both within and between tumor types. Subpopulations of Oct4-positive cells identified in these tumors are likely to represent “cancer stem” cells and therefore might be responsible for the maintenance and propagation of the tumors. If these cells represent cancer stem cells, and are therefore responsible for the maintenance and growth of the neoplastic cellular population, then these cells should serve as relevant therapeutic targets and offer the greatest potential for curative treatment.
Oct4 is a member of the POU (pit, oct, unc) family of transcription factors. 15, 20, 29– 31 The POU family of transcription factors is characterized by a bipartite DNA binding domain, which consists of a POU homeodomain and a POU-specific domain that is unique for each family member and binds to an octamer consensus sequence of ATGCAAAT. 11, 31 In the mouse, Oct4 is primarily expressed during embryogenesis, where it has been shown to be developmentally regulated. 6, 22, 28, 29 At the time of fertilization, low levels of maternally derived Oct4 are present. 22, 28, 31, 37 At the 8-cell stage of embryogenesis, Oct4 expression is upregulated, and this upregulation is maintained in all cells through the compaction of the morula. 22, 37 During blastocyst formation, high levels of Oct4 expression are maintained in the inner cell mass, 22, 28, 31 which contains the pluripotent embryonic stem cells that will develop into the embryo proper, 21 but Oct4 expression is downregulated as the surrounding cells differentiate into trophectoderm. 22, 28, 31 During later stages of blastocyst differentiation, the inner cellular mass becomes divided into the epiblast, which contains pluripotent cells that will later differentiate into the embryo proper, and the hypoblast, which differentiates into extraembryonic endoderm. 21 During endodermal differentiation of the hypoblast, these cells transiently upregulate Oct4 expression, followed by downregulation of Oct4. 22 Oct4 expression is maintained in the pluripotent cells of the epiblast until gastulation, at which time Oct4 expression is confined to the primordial germ cells. 28, 31 In the adult, Oct4 expression remains at low levels in the oocyte 28, 31 and in the spermatogonia A, but not in subsequent stages of spermatogenesis, 24 and Oct4 expression has been seen in the progenitor cell population of some adult human tissues with high turnover, such as the skin. 33 Additionally, recent work by Tai et al. has described Oct4 expression in various multipotent human adult stem cells but not in their differentiated descendants. 33
The pattern of Oct4 expression in embryogenesis, namely, the restriction of Oct4 to the pluripotent stem cell population, has led to the hypothesis that Oct4 plays a critical role in the maintenance of pluripotency and that alteration in Oct4 expression is the deciding point as to whether cells will differentiate. 19, 28, 31 Experimentally, Niwa et al. have shown that quantitative changes in Oct4 expression define embryonic stem cells' fate. In their studies, Niwa et al. showed that less than a 2-fold increase in Oct4 expression causes embryonic stem cells to differentiate into primitive endoderm and mesoderm, and downregulation or loss of Oct4 expression leads to trophectoderm differentiation. 19 Taken together, these studies and its unique pattern of gene expression during embryogenesis place Oct4 as a master regulator of pluripotency in stem cells.
Neoplastic cells and stem cells share many phenotypic similarities, which has led to their direct comparison on multiple occasions. 7, 23, 25, 27 These shared phenotypic characteristics of prolonged self-renewal or immortality, high proliferative capacity, and the propensity for tissue migration (invasion/metastasis), have led to the hypothesis that many types of cancer might be derived from adult stem cells that possess and facilitate the accumulation of genetic and epigenetic alterations that either partially or completely blocked the differentiation of these cells. 25, 33, 34 Additionally, in highly proliferative/high-turnover tissues, such as the gastrointestinal tract or the liver, stem or progenitor cells might be the only cells with a sufficient lifespan that would allow for the accumulation of somatic mutations, chromosomal alterations, or epigenetic changes that would be necessary for neoplastic transformation. 23, 27 Recently, restricted populations of tumorigenic neoplastic cells have been identified in several types of cancer, including leukemia, breast cancer, brain tumors, and bone sarcomas. 1, 8, 32, 36 These tumorigenic cells are characterized by a propensity for continued cellular proliferation, the ability to form new tumors when transplanted into immunocompromised mice, and the potential for multilineage differentiation. The phenotypic characteristics of these cells and their ability to repropagate tumors when transplanted into mice has led to the hypothesis that these distinct populations of “stemlike” cells are responsible for the maintenance of the respective tumor, thereby further supporting the stem cell theory of cancer.
Oct4 expression in cancer has been best characterized in germ cell tumors; namely, seminomas, embryonal carcinomas, dysgerminomas, and in the premalignant component of testicular germ cell tumors. 2, 3, 5, 9, 14, 16 Reports of Oct4 expression in other neoplastic diseases has varied between studies, which likely reflects a variation in the methods of Oct4 detection. 2, 3, 5, 8, 9, 13, 14, 16, 17, 26 However, Oct4 expression has been described in carcinomas of the kidney, lung cancer, chondrosarcoma, osteosarcoma, and breast cancer. 5, 8, 13, 16, 26 Experimental studies have also demonstrated a potential role for Oct4 in tumorigenesis. It has been shown that embryonic stem cells xenografted into mice form mixed teratomas. 4 Recent work by Gidekel et al. has shown that ectopic Oct4 overexpression increases the malignant phenotype of these tumors. 9 Additionally, ectopic Oct4 expression in a transgenic mouse model has been shown to lead to epithelial dysplasia, which is thought to be due to an expansion of the progenitor cell population in the skin and the gastrointestinal tract. 12 Together, these studies further suggest a potential role for Oct4 in tumorigenesis.
Consistent with the discovery of Oct4 expression in multiple tissue-specific adult stem cell lines, if stem cells are the neoplastic progenitor cells that are responsible for tumorigenesis, a population of Oct4-positive cells that represents “cancer stem” cells should be present in most if not all neoplasms. 33 Spontaneous neoplastic diseases commonly occur in dogs, and these cancers share many phenotypic and biologic characteristics with their human counterparts. Due to the similarities between canine and human neoplasms and the similar environment in which dogs and humans live, spontaneous canine neoplasms may serve as excellent in vivo and in vitro model systems for studying tumor biology and as a model system for novel chemotherapeutic clinical trials. 10, 35 Our laboratory is interested in using spontaneous canine neoplasms as models for human neoplastic diseases; therefore, the goal of this study was to evaluate Oct4 expression in a series of canine neoplasms in order to test the hypothesis that Oct4-positive cells, which might represent cancer stem cells, are present in all canine neoplasms. In order to test this hypothesis, we evaluated Oct4 expression in a series of 83 formalin-fixed, paraffin-embedded tumors, which represent 21 distinct neoplastic diseases, using immunohistochemical staining with light microscopic evaluation by a single pathologist (JDW). The results of this study reveal the universal presence of Oct4-positive cells in all tumors tested, suggesting that these Oct4-positive cells are excellent candidates for the putative “cancer stem” cells in these neoplasia.
Materials and Methods
Case selection and tissue samples
A total of 126 archival, formalin-fixed, paraffin-embedded tissues from 126 dogs were included in this study. All tumors included in this study were previously submitted to the Diagnostic Center for Population and Animal Health at Michigan State University for routine histologic evaluation. Routine 5-μm hematoxylin and eosin sections were recut from each tumor in order to confirm the diagnosis and to ensure adequate material was available for immunohistochemical evaluation. A total of 43 of the 126 cases were unsuitable for inclusion in the study due to inadequate tissue or inadequate numbers of neoplastic cells in the tissue section available for evaluation, as well as excessive pigment deposition or large areas of necrosis that would have confounded the evaluation of the immunoreaction. The 83 remaining tumors that were included in this study represented 21 distinct neoplastic diseases (Table 1).
Number and histologic classification of tumors included in this study.∗

∗A total of 83 tumors representing 21 distinct tumor samples were included in this study.
Oct4 immunohistochemistry
Five-micrometer sections of formalin-fixed, paraffin-embedded tissues were prepared for immunohistochemical staining. Sections were deparaffinized in xylene, rehydrated in graded alcohol, and rinsed in distilled water. In order to neutralize endogenous peroxidases, sections were incubated in 3% hydrogen peroxide for 5 minutes and subsequently rinsed in distilled water. Sections were incubated in a steamer for 20 minutes in a citrate buffer antigen retrieval solution (Dako Cytomation, Carpinteria, CA) and cooled for 20 minutes for antigen retrieval. Nonspecific antigen binding was blocked by incubating slides with a protein-blocking agent (Dako Cytomation) for 10 minutes. Using an autostainer, slides were incubated for 60 minutes with mouse anti-human Oct4 monoclonal antibodies (Santa Cruz Biotechnology, Santa Cruz, CA) at a 1 : 100 dilution. A linked strepavidin-biotin immunoperoxidase system was used for immunolabeling. The reaction was visualized with a 3,3′-diaminobenzidine substrate. Canine mast cell tumors, which have been previously shown to express Oct4, 33 were included in each run as a positive control. Negative controls were identical to positive controls, except that incubation with primary antibodies was replaced with buffer.
Immunohistochemical evaluation
All tumors were evaluated for Oct4 expression using light microscopy at 100× and 400× magnification. Any Oct4 expression in the nuclei of the neoplastic cell population was considered positive. Occasional immunopositive cells were seen in adjacent normal tissue, which were considered to represent normal adult tissue-derived progenitor cells, as previously isolated from several normal adult human tissues. 33
Results
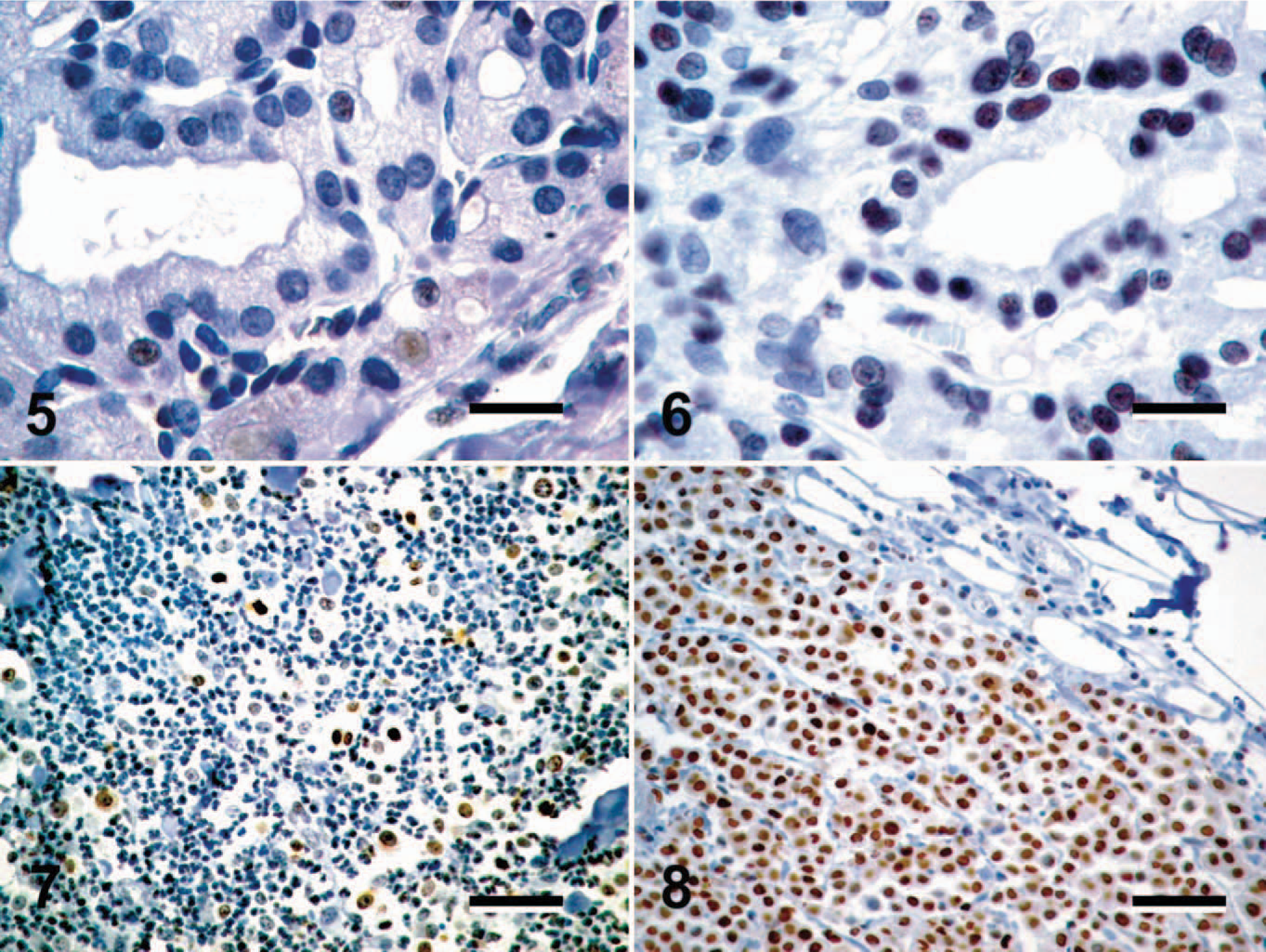
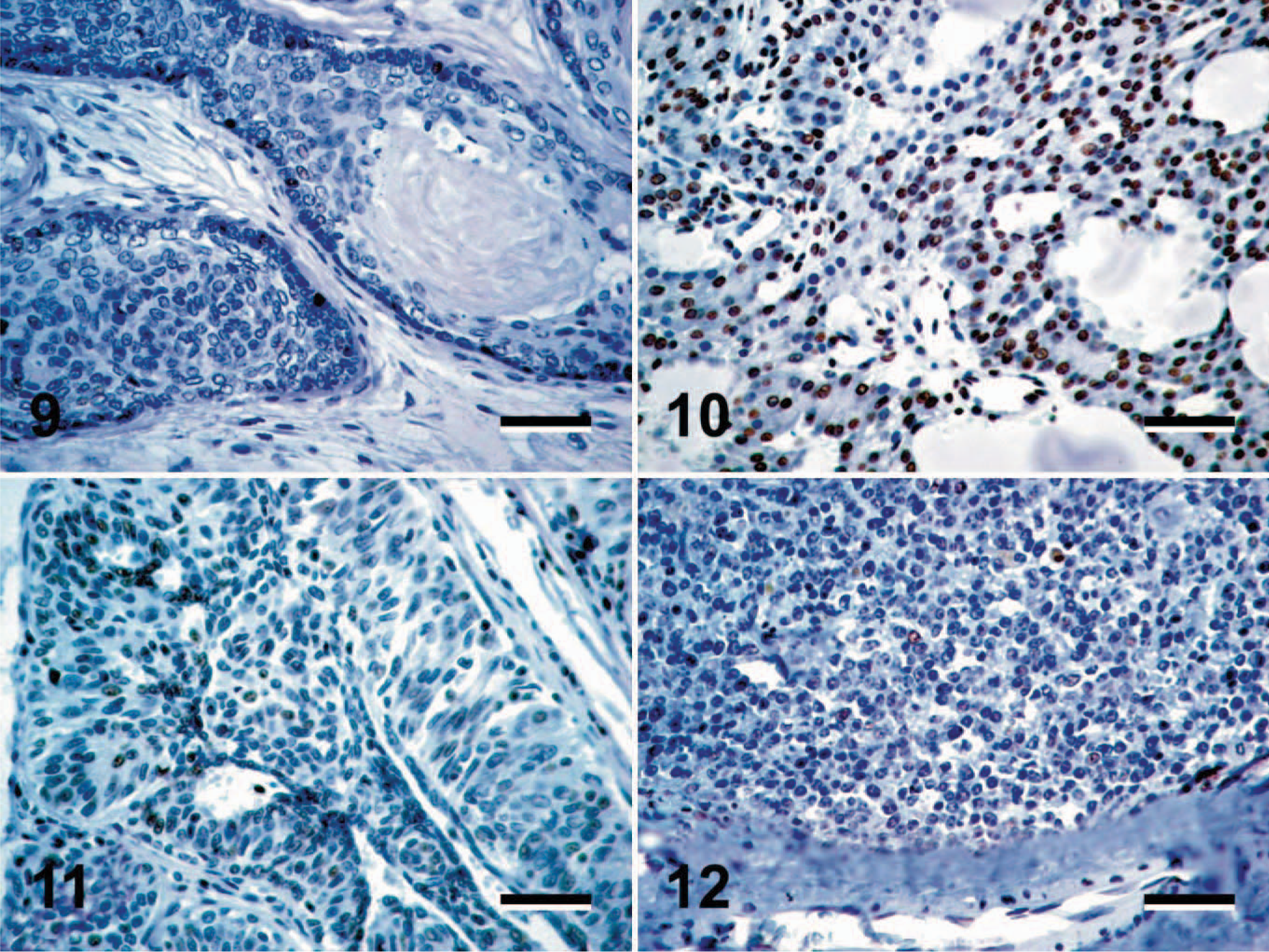
All of the 83 tumors included in this study expressed Oct4 in a subpopulation of neoplastic cells (Figs. 1–12). The pattern of Oct4 expression varied both in the intensity of nuclear staining and in the proportion of Oct4-positive cells within a tumor. This variation ranged from faint to intense nuclear staining, as well as from rare randomly scattered cells that accounted for less than 1% of the neoplastic cell population to diffuse staining of greater than 90% of the neoplastic cells (Figs. 1–8). In the majority of tumors, Oct4-positive cells were randomly scattered throughout the tumor, with no obvious pattern of distribution (Figs. 1–12). Occasional immunopositive cells were also found in the surrounding normal tissue in multiple tissues. In several tissues, these immunopositive cells were in locations consistent with tissue progenitor cells (data not shown), and as such were considered to be adult tissue-derived stem cells, similar to those previously isolated and described. 33 The degree of immunopositivity in adjacent nonneoplastic tissues varied between tissues. Correlations between the immunopositivity in neoplastic cells and adjacent normal tissues were not observed.
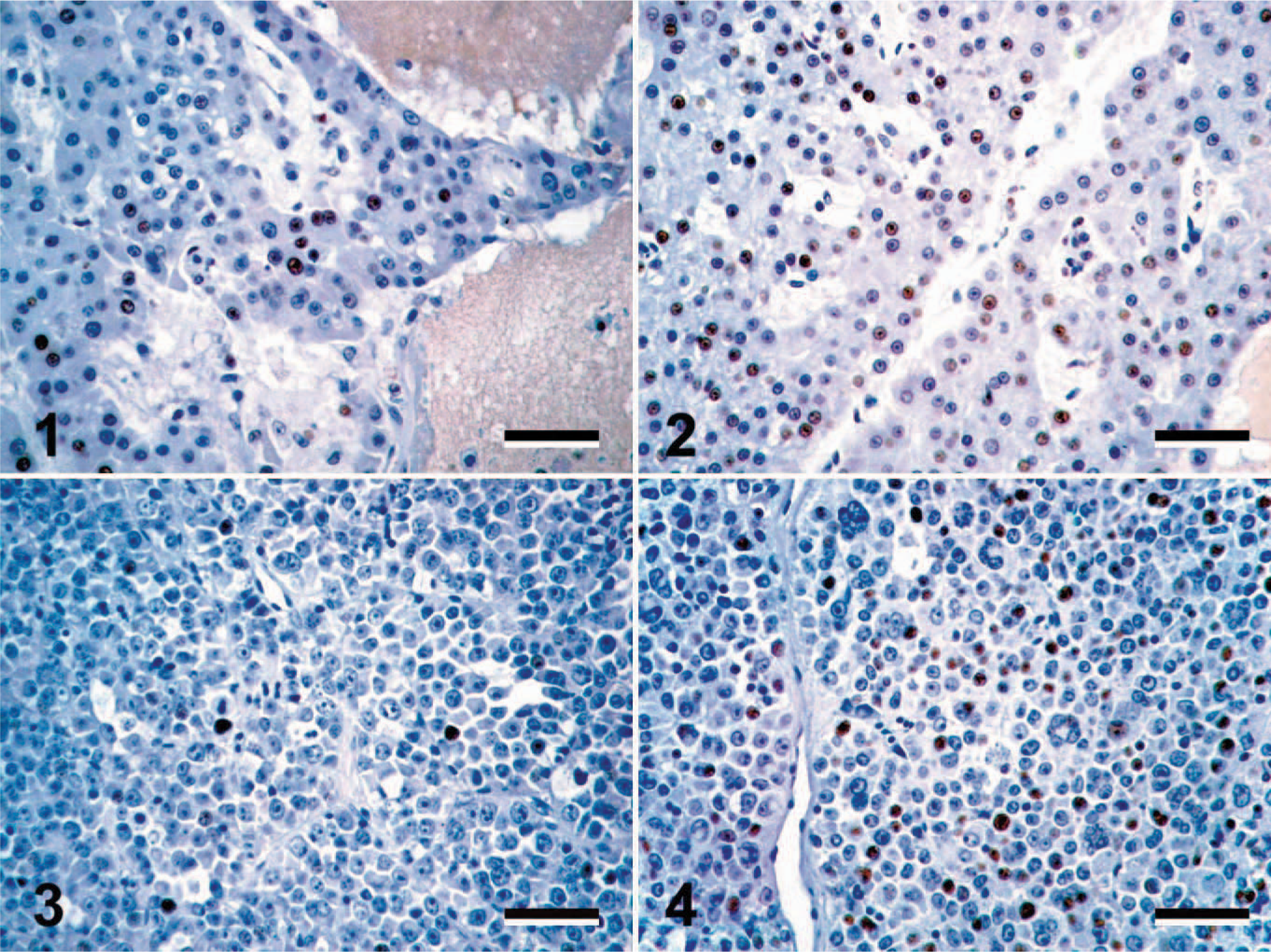
Intertumor and intratumor variation in Oct4 expression in spontaneous canine neoplasms. Low (Figs. 1, 3) and high (Figs. 2, 4) Oct4 expression in canine hepatocellular carcinomas (
Oct4 expression in canine thyroid carcinoma and mast cell tumor. Thyroid carcinomas (
Oct4 expression in spontaneous canine cancers squamous cell carcinoma. Oct4-positive cells (brown nuclear precipitate) were identified in every tumor included in the study. Immunohistochemical staining with mouse anti-human Oct4 monoclonal antibody visualized with the substrate 3,3′-diaminobenzidine with hematoxylin counterstain. Bar = 80 μm.
Due to our laboratory's interest in canine cutaneous mast cell tumors (MCTs), 21 of the 83 tumors evaluated were canine MCTs. Interestingly, the highest levels of Oct4 expression were seen in canine MCTs, with greater than 80% of the neoplastic cell population expressing Oct4 in 7 of 21 MCTs evaluated, and 6 of 21 MCTs containing approximately 40% to 60% Oct4-positive cells (Figs. 7, 8).
Discussion
Oct4-expressing subpopulations of neoplastic cells were identified in every tumor included in this study. The proportion and distribution of Oct4-positive cells varied both within and between tumor types; however, these cells were seen consistently within all of the tumors. Based on the association of Oct4 with stem cells and the maintenance of an undifferentiated state and pluripotency, 19, 28, 31, 33 we hypothesize that these subpopulations of Oct4-positive cells might represent subpopulations of “cancer stem” cells or progenitor cells that are responsible for the maintenance and endless proliferative capacity of the respective neoplasm.
The variation in Oct4 expression within and between tumor types most likely represents variation in the cellular differentiation of neoplastic subpopulations. In cancer, tumors are made up of clonal subpopulations of neoplastic cells that are originally derived from a single cell. Within a given tumor, each of these clonal subpopulations might be blocked at unique stages of cellular differentiation, leading to phenotypic variations. 25, 34 Therefore, depending on the degree of differentiation and the point at which differentiation is blocked, some cells that are blocked early in cellular differentiation might maintain high levels of Oct4, whereas other cells that are able to partially differentiate might do so to a degree that Oct4 is downregulated. In some tumors, most daughter cells might undergo partial differentiation, which results in Oct4 downregulation in the majority of cells, whereas only the true cancer stem cell population, which is responsible for the long-term maintenance of the cancer, continues to express Oct4. The latter might account for the rare randomly scattered Oct4-positive cells seen in many of the tumors examined.
Previous studies have reported variable Oct4 expression in human tumors, often finding little to no Oct4 expression in the majority of tumors. 2, 3, 5, 8, 9, 13, 14, 16, 17, 26 The discrepancy in many of these studies is likely due to variation in the methods of Oct4 detection, as Oct4 has been most consistently identified on the mRNA level using reverse transcriptase–polymerase chain reaction, 5, 13, 26 but less frequently on the protein level. 2, 9, 16, 26 The results of this study strikingly contrast many of these previous studies in that Oct4 protein was identified in every tumor evaluated in this study. 2, 9, 16 In several of the tumors included in this study, Oct4-positive cells were randomly scattered, accounting for less than 10% of the neoplastic cell population, and often expressed low levels of Oct4. In these cases, identification of Oct4-positive cells required meticulous evaluation of the entire tumor sections at 400× magnification, and these cells could be easily missed at lower magnifications or if only “representative” samples of the tumors were evaluated, as might be the case when tissue microarrays are used. 9, 16 Additionally, it has become a common practice that tumors are only considered to express a protein when greater than 10% of their neoplastic cell population expresses the given protein. Under this criterion, small but significant populations of Oct4-positive cells might have been overlooked in previous studies. Therefore, depending on the methods used for immunohistochemical evaluation and the criteria used to consider a tumor positive for Oct4, several tumors might have been considered Oct4 negative when, in fact, Oct4-positive cells were present within the neoplastic tissue. Additionally, variation in the antibodies and immunolabeling conditions might have further resulted in discrepancies between studies.
Since its discovery, Oct4 has been consistently associated with the maintenance of an undifferentiated state and pluripotency. 20, 28, 30, 31, 33 Additionally, Oct4 knockout mice have also demonstrated the importance of Oct4 in the maintenance of pluripotency, as these mice have an embryonic lethal phenotype during blastocyst formation due to a loss of inner cell mass pluripotency and trophectodermal differentiation of these cells. 18 Together, these data demonstrate the role of Oct4 in the maintenance of an undifferentiated state and pluripotency and suggest that cells that express Oct4 should represent stem cells. In light of this, the subpopulations of Oct4-positive cells identified in the tumors evaluated in this study are likely to represent subpopulations of “cancer stem” cells, and therefore might be responsible for the maintenance and propagation of the respective tumor. These cells might serve as a rational therapeutic target, allowing for the targeting of the cellular pool that is responsible for the continuing proliferative potential of the tumor. The greatest hope for definitively treating cancer is the elimination of the source of new neoplastic cells. One potential therapeutic target in these cells is Oct4, as the feasibility of this approach is evidenced by the fact that downregulation of Oct4 in stem cell transplant-derived teratomas and in conditional transgenic mice results in cellular differentiation and, thereby, the elimination of the proliferative capacity and neoplastic phenotype of precancerous and cancerous cells. 9, 12
The identification of Oct4-positive cells in a series of canine neoplasms and the recent characterization of Oct4 expression in human adult stem cells from various tissues 33 support the hypothesis that adult stem cells are the neoplastic progenitor cells in cancer. However, further studies are needed to better characterize these cells and to define their role in tumorigenesis. At present, little is known as to which cells are the true progenitor cells for cancer and how these progenitors might be targeted in order to improve the therapeutic response to a given tumor. If these Oct4-positive cells truly represent cancer stem cells, they might serve as the best target for eliminating many forms of cancer.
Footnotes
Acknowledgements
We would like to acknowledge the immunohistochemistry section of the Diagnostic Center for Population and Animal Health at Michigan State University for its technical assistance. During the course of this study, J.D.W. was funded by the NIH T-32 postdoctoral training grant number RR17189.
