Abstract
CD44+/CD24– phenotype has been used to identify human and canine mammary cancer stem-like cells. In canine mammary tumors, CD44+/CD24– phenotype has been associated with high grade and lymph node infiltration. However, several studies have reported opposing results regarding the clinical significance of phenotypic groups formed by the combination of CD44 and CD24 in both human and canine mammary tumors. So far, no study has investigated the correlation between these phenotypes and survival in dogs. The aim of this study was to investigate the expression and distribution of CD44 and CD24 in canine mammary carcinomas and to correlate them with histological diagnosis and survival in a well-characterized cohort. Immunohistochemistry was performed in 96 mammary carcinomas with antibodies against CD44 and CD24. Expression of CD44+ and CD44+/CD24– phenotype was detected in 75 of 96 (78%) and 63 of 96 (65.6%) carcinomas, respectively. Their expression was associated with tumor type, occurring more often in tubular complex carcinomas than in solid carcinomas. CD44+/CD24– phenotype was associated with a better overall survival (
Mammary tumors are a major health issue in both women and bitches. Breast cancer is the most prevalent cancer and the first cause of cancer death in women worldwide, as well as the most common cancer in intact bitches. 15,36 It has been estimated that 1 in 8 women will develop breast cancer during their lifetime, while one-third of unspayed bitches will develop a mammary tumor by the age of 10. 21,57 Mammary neoplasms of both species share numerous clinical, genetic, and molecular similarities. 1,18,27,41,48 Canine mammary tumors, by their spontaneous occurrence, are hence representative of human breast cancer development, and accumulating data support the relevance of the canine model of mammary neoplasms as a valuable tool in comparative oncology.
Over the past decade, substantial evidence has been gathered assessing the presence of a subpopulation of tumor cells known as cancer stem cells (CSCs) in hematopoietic and solid tumors, including breast cancer. 4,10 CSCs would be the only cells able to regenerate and form a tumor and would be responsible for recurrences as they are resistant to conventional treatments such as radiotherapy or chemotherapy. 13,43,53 Targeting these cells represents a sine qua non condition for tumor eradication.
In women, the CD44+/CD24– phenotype has been considered a marker of breast CSCs. It has been demonstrated that as few as 200 cells presenting a CD44+/CD24– phenotype were able to form tumors after injection in the mammary fat pad of NOD/SCID mice when thousands of cells that did not present this phenotype were not able to give rise to a tumor. 4
CD44 represents a family of type I transmembrane glycoproteins; it is the receptor for hyaluronan but can also bind to collagen, fibronectin, or chondroitin sulfate. CD44 acts also as a signaling platform that integrates growth factor and cytokine signals to regulate cell adhesion, migration, proliferation, differentiation, and survival. 5,42,59 Histopathological studies of human breast cancer have associated CD44 expression with both favorable and unfavorable clinical outcomes. 3,8,31
CD24 is a mucin-like surface protein. CD24 can increase tumor cell proliferation, motility, and invasiveness as well as tumor growth and metastasis in vivo. 7,11,56 Its overexpression has been associated with increased aggressiveness of cancer, including breast, ovarian, lung, and prostate cancers. 8,17,28
The phenotype CD44+/CD24– was proposed as a prospective marker of breast CSCs in 2003. 4 Although CD44+/CD24– has been extensively used as a CSC marker in human breast cancer, the prognostic value of this phenotype in breast cancer is controversial. For example, the CD44+/CD24– phenotype has been reported to have a negative impact on long-term clinical outcome in patients with breast cancer, 50,55 have no prognostic value, 12,40 or even be associated with a favorable prognosis. 3,25 Other studies suggested that CD44+/CD24– breast cancer cells may not be associated with clinical outcome but may favor distant metastasis. 2
Clinical significance of the CD44+/CD24– phenotype in canine mammary tumors was reported by several groups. A CD44+/CD24– phenotype was described in 31% of 130 canine mammary tumors, with an advantage for grade II and III tumors and lymph node metastases. 34 Of 16 canine mammary tumors, 46% were CD44+/CD24–, although CD24 was not detected in any tissue and the phenotype CD44+/CD24– may in fact be a CD44+ phenotype. 6 Finally, 30% of 88 canine mammary tumors presented a CD44+/CD24– phenotype, which was statistically associated with higher grades. Furthermore, anaplastic carcinomas were observed more frequently in samples with a CD44+/CD24– phenotype but without reaching statistical significance. 22 However, tumor cohorts were not completely described, nor specificities of the antibodies against CD44 and CD24. Furthermore, the immunohistochemical scoring method varied among the studies.
The aims of the present study were (1) to perform specific CD44 and CD24 staining in a well-characterized cohort of 96 canine mammary carcinomas and (2) to determine whether the expression of CD44 and CD24 expression alone or in combination was related to clinicopathological parameters.
Material and Methods
Case Selection and Tumor Samples
The present study was based on a series of 96 cases of canine malignant mammary carcinomas and corresponding lymph nodes selected from the biobank of OCR (Oncovet Clinical Research, Parc Eurasanté Lille Métropole, France). Specimens were obtained from bitches from all ages and breeds, which underwent surgery between July 2011 and October 2015. All tissues were fixed in 10% neutral buffered formalin and embedded in paraffin wax. The study followed as far as possible the recommended guidelines for the conduct and evaluation of prognostic studies in veterinary oncology and more specifically on mammary neoplasms, with the inherent limitations of a retrospective study. 35,58 Dogs included in the study had either 1 single malignant mammary carcinoma or multiple mammary tumors with only 1 of them being malignant. Twelve cases of recurrences and 8 animals presenting more than 1 malignant tumor, whatever the subtype, were excluded because of the difficulty to determine which tumor would affect the follow-up. Thirteen carcinomas in situ, which may not be fully malignant, and 15 cases with insufficient clinical data were excluded.
Follow-up Data
The follow-up period was defined as 24 months between surgery and data collection. Clinicopathological parameters gathered for each tumor included histological type, grade, nodal status, tumor recurrence or distant metastasis, type of surgery (lumpectomy, regional mastectomy, unilateral mastectomy, or bilateral mastectomy), treatment modalities (no treatment after surgery, nonsteroidal anti-inflammatory drugs, nonsteroidal anti-inflammatory drugs plus chemotherapy,
Immunohistochemistry
Expression of CD24 and CD44 was assessed using immunohistochemistry (IHC) on 5-μm sections of the tumor, following the recommendations for IHC studies. 14 When several blocks were available for a single tumor, all archival hematoxylin and eosin–stained slides were reviewed by a veterinary anatomic pathologist (Q.P.), and 1 representative slide of each tumor was selected. The antibodies against CD44 and CD24 (Table 1) are directed against human epitopes, and cross-reaction with the canine tissues was assessed. CD44 and CD24 antibodies share respectively 82% and 81% sequence identity with the canine epitope (data not shown). The specificity of antibodies for canine CD44 and CD24 was partially confirmed by staining tissues known to express these markers—tonsils lymph follicles for CD24 and tonsils epithelium for CD44—and the accuracy of double immunostaining was assessed by comparing it with single immunostaining for CD44 and CD24 separately, as shown in Supplemental Figures S1 to S4. Finally, a negative control using isotype-matched immunoglobulin (Table 1) with the same protocol and at the same concentration as the antibodies was performed in canine tonsils (Suppl. Figs. S5–S8) and in samples of canine mammary carcinomas from all grades and histological types (data not shown). Double immunostaining for CD44 and CD24 was performed consecutively using a Discovery XT automated immunostaining device (Ventana Medical Systems, Roche, Meylan, France). Slides were deparaffinized and a short antigen retrieval using Cell Conditioning solution (CC1; Ventana Medical Systems) was performed (95°C—CC1 short). After incubation with primary antibody against CD24, slides were incubated during 4 minutes with biotin-free HRP multimer (DISCOVERY Ultramap anti-Rb HRP; Roche, Meylan, France) and an HRP-driven chromogen (DISCOVERY Purple Kit; Roche). Slides were then incubated with antibody against CD44 followed by incubation during 16 minutes with another biotin-free HRP multimer (DISCOVERY Ultramap anti-Rt HRP; Roche) and an HRP-driven chromogen (DISCOVERY ChromoMap DAB Kit; Roche). Slides were counterstained with a modified Mayer’s hematoxylin (Hematoxylin II; Roche) for 4 minutes and an aqueous solution of buffered lithium carbonate (Bluing Reagent; Roche) for 4 minutes. A negative control (in which primary antibody was omitted and replaced by reaction buffer) and 2 positive controls (canine tonsils and a mammary tumor tissue section known to express both markers) were included in each staining run.
Antibodies and Reaction Conditions for Immunohistochemical Labeling of Canine Mammary Tumors.
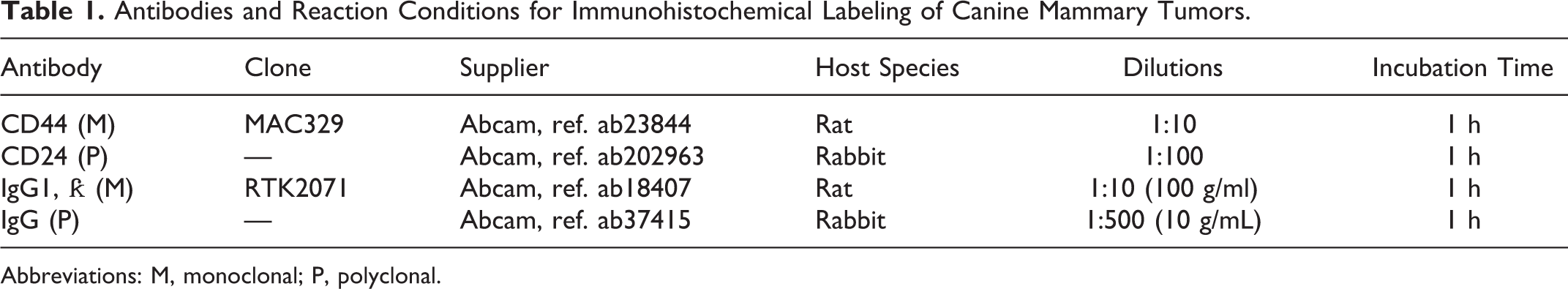
Abbreviations: M, monoclonal; P, polyclonal.
Immunohistochemical Evaluation
For each tumor, images of 10 medium-power cellular fields (20× objective) were captured using a Nikon Eclipse Ni microscope (Nikon, Tokyo, Japan). A consistent subset of CD24- and CD44-labeled slides were scored by 2 observers (B.R., Q.P.), including confirmation by an anatomic pathologist (Q.P.) to reach a scoring assessment consensus. Immunolabeling for both CD24 and CD44 was then scored by 1 observer (B.R.). All cases were scored without prior knowledge of the tumor pathology or patient outcome. On each field and for each marker, semiquantitative estimates of intensity of staining and percentage of positive cells were evaluated, as described in previous articles. 3,12 Proportion of cells stained was expressed by a score ranging from 0 to 3 (0 = none or <5%, 1 = 5%–25%, 2 = 25%–50%, 3 = >50%). Staining intensity was also expressed by a score ranging from 0 to 3 (0 = negative, 1 = weak, 2 = moderate, 3 = strong), as presented in Supplemental Figures S9 to S14. For each field, staining intensity (0–3) was multiplied by proportion (0–3) to obtain a staining score ranging from 0 to 9. Final scores for the tumor were obtained by adding the scores of the 10 fields, which led to a staining score ranging from 0 to 90. For the study of phenotypes measure by double immunostaining for CD44 and CD24, (CD44+/CD24–, CD44–/CD24+, CD44+/CD24+, CD44–/CD24–), the possible scores for the tumor were represented by the proportion of stained cells, ranging from 0 to 30 (scores of 0–3 summed over 10 fields). The threshold used to define positive or negative immunolabeling of tumors was a proportion of stained cells ≥1/30. Tumors were considered negative when the proportion of stained cells was 0/30, which corresponded to a complete lack of labeling.
Classification by Components
In addition to the frequently used classifications of canine mammary tumors according to grade and histological type, we also identified different cellular types and classified tumors into 3 different components. A luminal component corresponded to all structures related to a lumen (ductal, tubular, or papillary). The myoepithelial component corresponded to myoepithelial or myoepithelial-like (spindle-shaped) cells. Finally, a diffuse component corresponded to cells closely packed in solid areas or anaplastic areas. Every tumor could present with 1, 2, or 3 components.
Statistical Analysis
The quantitative variables were expressed by mean and standard deviation or by median and interquartile range. The normality of the distributions was checked graphically and by using the Shapiro-Wilk test. The qualitative variables were described using frequencies and percentages. The relationship between the expression of a phenotype or a marker and the types of component (luminal, diffuse, and myoepithelial) was estimated using generalized linear mixed models. Post hoc pairwise comparisons were performed in case of global significant difference, and a Bonferroni correction for multiple comparisons was applied. In case of sufficient sample size, comparison of the proportion of expression of a phenotype or a marker between groups of clinicopathological parameters was performed with χ2 tests. In case of expected counts lower than 5, Fisher exact tests were used. Staining scores of CD44 and CD24 alone or in combination were compared between grade and histological type using nonparametric Kruskal-Wallis tests, as well as between presence and absence of emboli, lymph node infiltration, tumor recurrences, and distant metastases using the nonparametric Mann-Whitney
Survival curves were estimated using the Kaplan-Meier method. Survival curves were compared between tumor groups expressing or not a marker or a phenotype using a log-rank test. Comparisons were further adjusted on age using a Cox proportional regression model; hazard ratios (HRs) were estimated as effect size.
Finally, we assessed the relationship between the score of CD24+/–/CD44+/– cells and overall survival in the Cox proportional hazard regression model by treating scores as continuous variables. Proportionality hazards and log-linearity assumptions were examined using Schoenfeld and martingale residual plots.
Data were analyzed using the SAS version 9.4 (SAS Institute, Cary, NC), and all statistical tests were performed with a 2-tailed α risk of .05.
Results
Animal Data and Histopathological Characteristics of Tumors
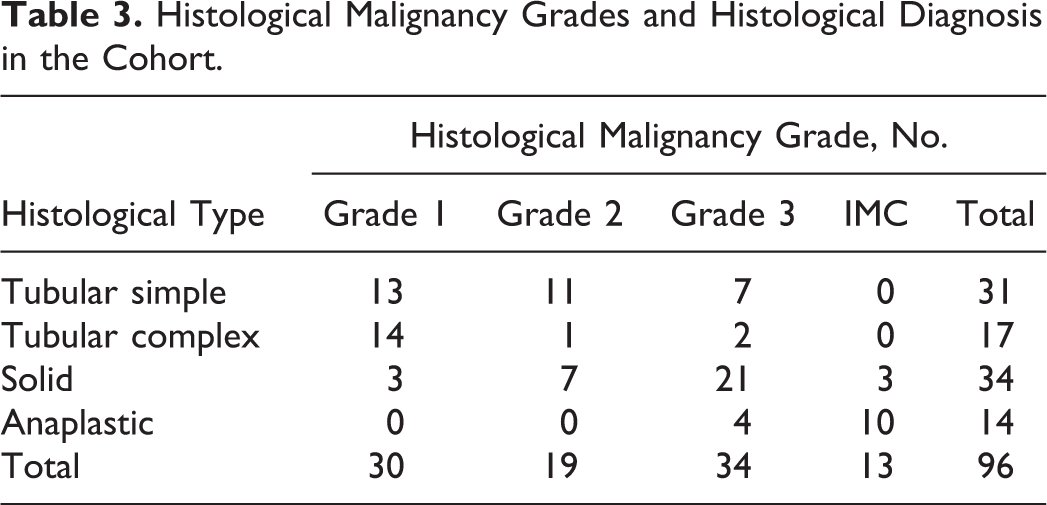
The study included 96 female dogs. Of those of known neuter status, at the time of diagnosis, most were intact: 67 of 88 (76%). Of 38 breeds represented, the most frequent were Yorkshire Terrier (13/96), Brittany Spaniels (7/96), and Shih Tzu (6/96). The mean age of dogs at the time of surgical removal of tumors was 10.1 ± 2.1 years (mean ± SD; range, 3.3–15.5 years of age). According to the Peña grading method, tumors were classified as grade I (30/96; 31%), grade II (21/96; 22%), or grade III (32/96; 33%). 46 Inflammatory mammary carcinomas represented 13 of 96 cases (14%). Tumors were histologically classified into subtypes according to the Goldschmidt classification of canine mammary tumors. 19 Our cohort was further classified into 4 major histological types—solid, tubular simple, tubular complex, and anaplastic (Table 2). 22,32,39 Tubular simple and tubular complex tumors are recognized to have the best prognosis, while solid and even more anaplastic tumors have a poor prognosis. 39,49 When tumors were heterogeneous (ie, solid and tubular or adenosquamous and tubular, etc), the solid component was thus privileged over the tubular one as the first is commonly associated with worse prognosis. Histological evaluation yielded 31 of 96 tubular simple carcinomas (32%), 17 of 96 tubular complex carcinomas (18%), 34 of 96 solid carcinomas (35%), and 14 of 96 anaplastic carcinomas (15%). Correspondence between grades and histological types is presented in Table 3.
Relationship Between Morphological Diagnosis and Histological Type of Malignant Canine Mammary Tumors.

Histological Malignancy Grades and Histological Diagnosis in the Cohort.
Tumors were also classified according to criteria of aggressiveness. Emboli were present in 29 of 96 cases (30%), including 16 grade III tumors and 13 inflammatory mammary carcinomas. Lymph nodes were available in 49 of 96 cases (51%), with confirmed infiltration by tumor cells in 17 of 49 cases (35%).
Tumors were finally classified in 3 groups according to the components. Seventy-five of 96 tumors (78.1%) presented a luminal component, 37 of 96 (38.5%) a myoepithelial component, and 68 of 96 (70.8%) a diffuse component.
The individual-animal data are available in Supplemental Table S1.
CD44 Expression
In almost all cases, CD44 staining was found to be membranous, either homogeneously on the whole membrane (notably on anaplastic cells) or on the basolateral domain of cells in the case of tubular or papillary structures (Fig. 1). In rare cases, cytoplasmic staining was observed in tumor cells bearing major anomalies like karyomegaly (Suppl. Fig. S15).
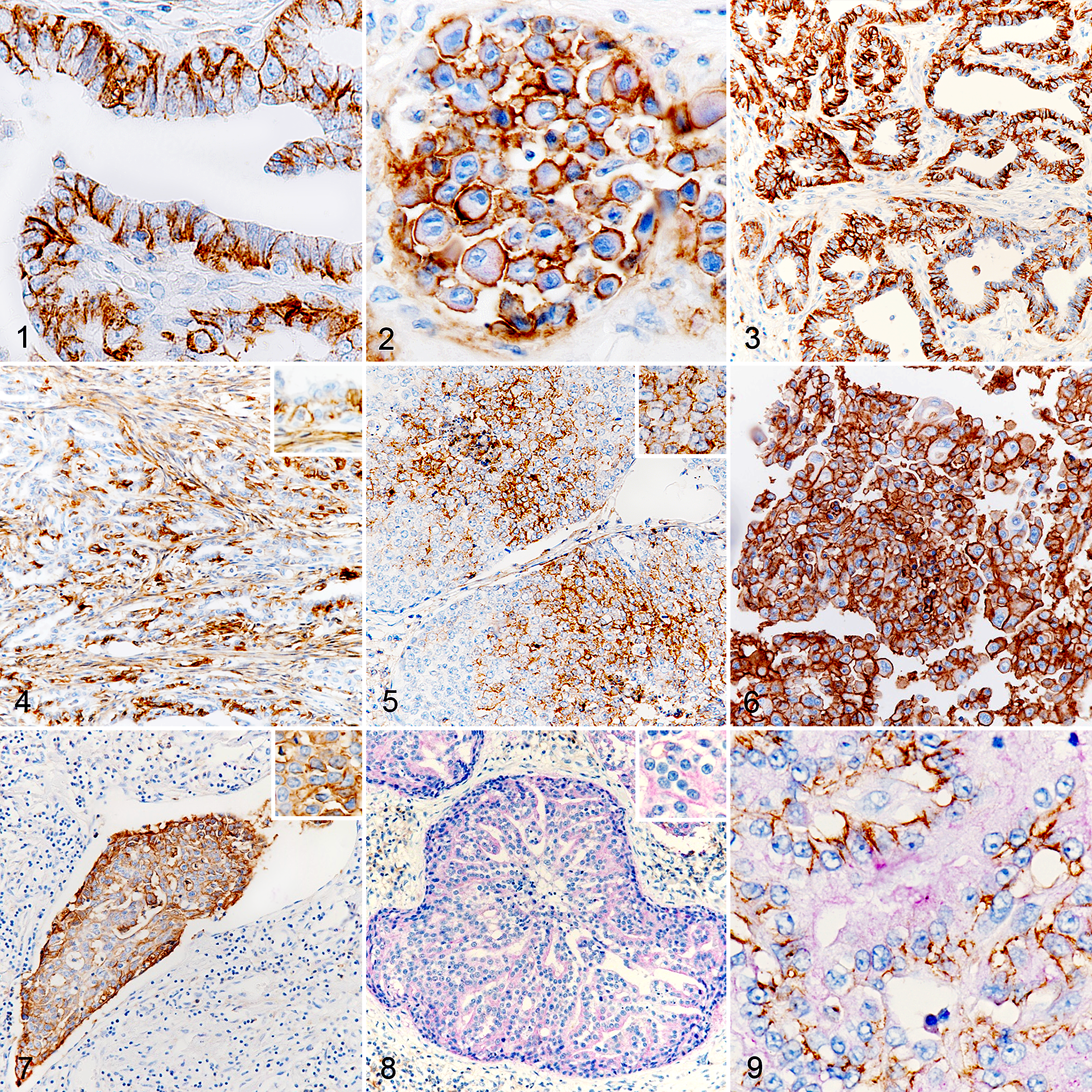
Malignant mammary carcinomas, dog. Immunohistochemistry for CD44/CD24. Examples of immunolabeling and associated phenotypes. CD44 stains brown; CD24 stains purple.
CD44 expression was significantly associated with the tumor component type (
Expression of CD44, CD24, CD44+/CD24–, and CD44–/CD24+ in Relation to the Type of Component.

a Group significantly different from myoepithelial.
b Group significantly different from diffuse.
c Group significantly different from luminal.
CD44 was expressed in 75 of 96 tumors (78%). CD44 expression (Table 5) was not significantly associated with tumor grade (
Expression of CD44, CD24, CD44+/CD24–, and CD44–/CD24+ in Relation to Clinicopathological Parameters.
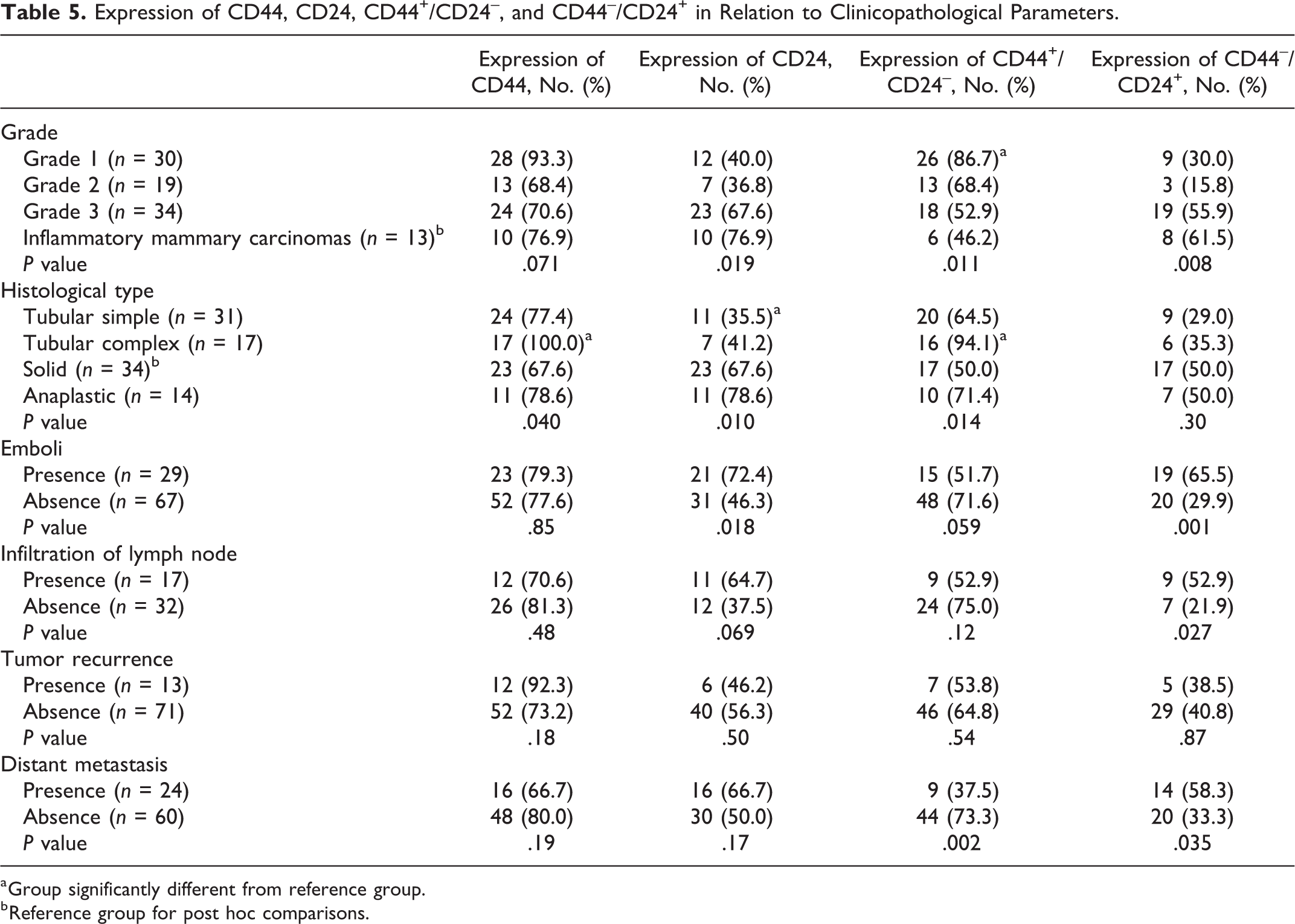
a Group significantly different from reference group.
b Reference group for post hoc comparisons.
Staining score for CD44 ranged from 0 to 90 (the maximum possible score, where virtually all cells were stained). Positive tumors presented a median of 43 (quartile 1 [Q1]: 13; quartile 3 [Q3]: 55.5). CD44 staining score (Table 6) was not significantly associated with higher tumor grade (
Staining Score of CD44, CD24, CD44+/CD24–, and CD44–/CD24+ in Relation to Clinicopathological Parameters.a
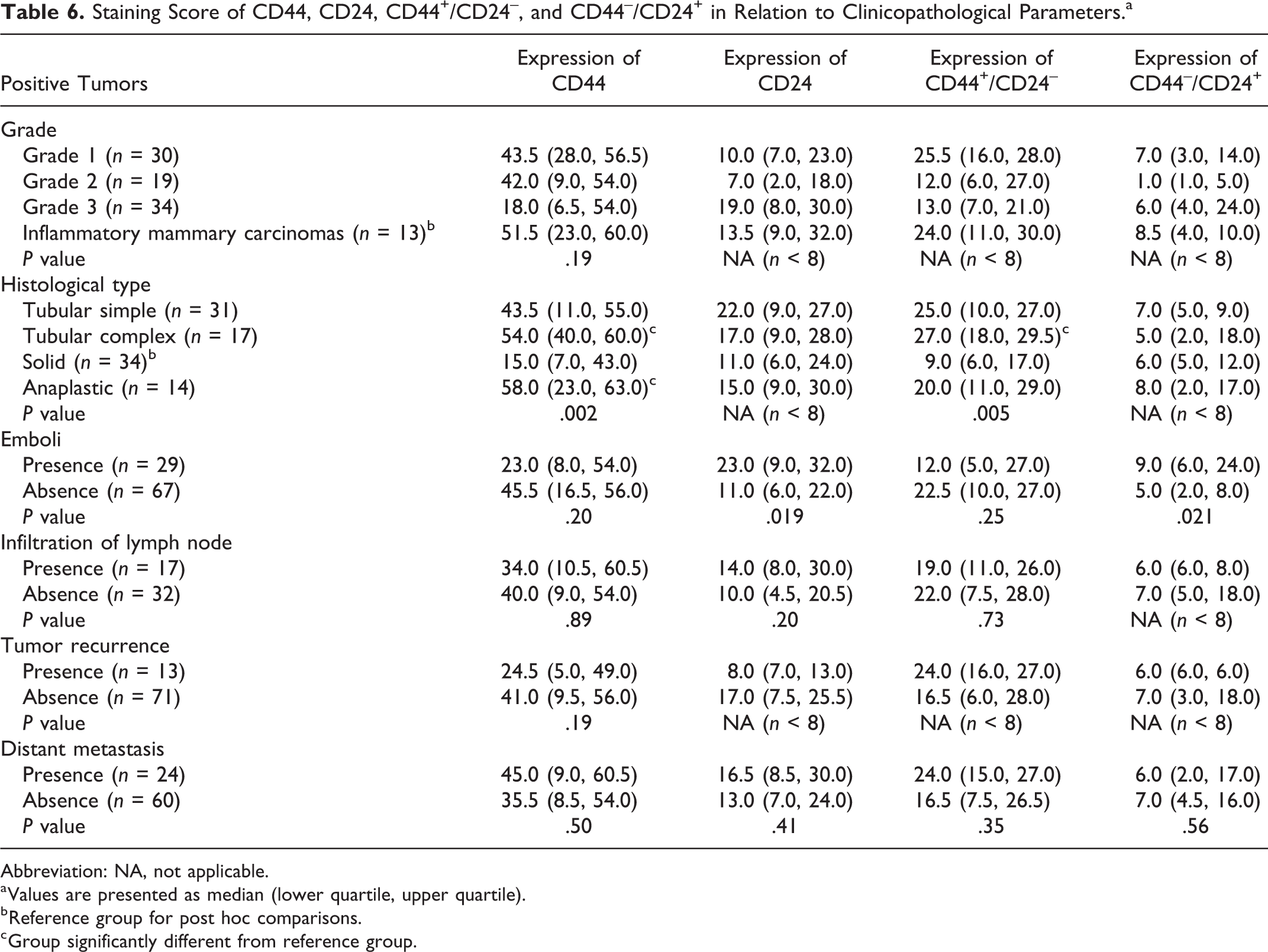
Abbreviation: NA, not applicable.
a Values are presented as median (lower quartile, upper quartile).
b Reference group for post hoc comparisons.
c Group significantly different from reference group.
CD24 Expression
CD24 staining was almost entirely cytoplasmic (Fig. 2). Apical staining was sometimes observed, notably on luminal surfaces, but was considered nonspecific due to staining of secretions (Suppl. Fig. S16).
Expression of CD24 was significantly dependent on the tumor component type (
CD24 was expressed in 52 of 96 tumors (54%). CD24 expression (Table 5) was significantly associated with higher tumor grade (
CD44+/CD24– Expression
CD44+/CD24– staining was significantly associated with the tumor component type (
CD44+/CD24– staining (Figs. 3–7) was observed in 63 of 96 tumors (65.6%) (Table 5). CD44+/CD24– was not associated with emboli (
CD44–/CD24+ Expression
The CD44–/CD24+ phenotype was associated with the tumor component type (
CD44–/CD24+ staining (Fig. 8; Table 5) was expressed in 39 of 96 tumors (40.6%). CD44–/CD24+ was not associated with histological type (
CD44+/CD24+ and CD44–/CD24– Expression
The CD44+/CD24+ phenotype (Fig. 9) was found in 39 of 96 tumors (40.6%) and the CD44–/CD24– phenotype in 74 of 96 tumors (77.1%). Expression of CD44+/CD24+ and CD44–/CD24– was not associated with grade (
Survival Study
Follow-up data were available for 84 of 96 cases (87.5%). These 84 cases were composed of 26 grade I (31%), 19 grade II (22.5%), 31 grade III (37%), and 8 inflammatory mammary carcinomas (9.5%). During the follow-up period, 13 animals relapsed and 24 animals died or were euthanized due to metastatic disease and/or local recurrence. Animals that died for reasons unrelated to cancer or that were still alive at the end of the 2-year follow-up period were censored as the tumor-related death event had not occurred. The median DFS time was 380 days and the median OS time was 417 days.
Survival was compared between subgroups regarding the presence of a particular marker/phenotype. In this cohort, CD44+/CD24– phenotype was significantly associated with better survival (
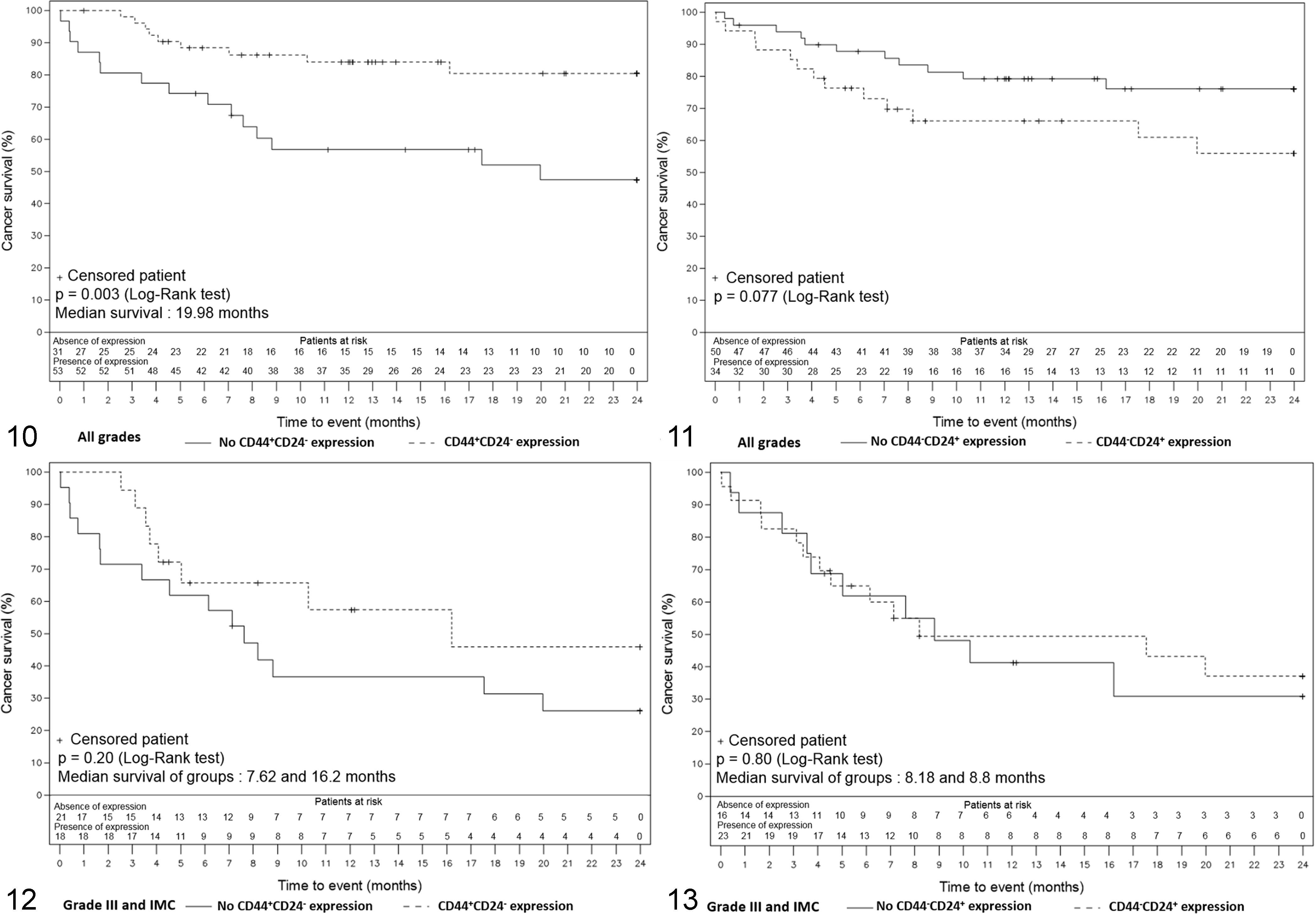
Kaplan-Meier overall survival curves representing the survival of the dogs included in the survival study according to time. The numbers under the x-axis correspond to the number of dogs alive at a certain time, in the presence or absence of the phenotype expression. IMC, inflammatory mammary carcinoma.
The cohort was then divided into 2 groups of different aggressiveness as defined by the grade to assess if CD44, CD24 alone or in combination could have an added prognostic value with regard to tumor grades. The “good prognosis” subgroup was composed of 45 animals, including all the grade I (26; 57.8%) and grade II tumors (19; 42.2%). During the follow-up period, only 1 of these animals died of reasons related to cancer. The “poor prognosis” subgroup was composed of 39 animals, including all grade III (31; 79.5%) and inflammatory mammary carcinomas (8; 20.5%). During the follow-up period, 23 of these animals died of reasons related to cancer. In this subgroup, the median DFS time was 249 days and the median OS time was 268 days. No marker or phenotype could be related to prognosis. Animals whose tumor exhibited a CD44+/CD24– phenotype showed a slight trend to better survival without reaching statistical significance (
Discussion
Since the use of CD44+/CD24– as a prospective marker of breast cancer stem cells, numerous studies have been performed to evaluate its clinical value in both human breast cancer and canine mammary tumors. 4 However, contradictory results have been obtained in different studies. In this study, we investigated the individual expression of CD44 and CD24 as well as their associated phenotypes in a well-characterized cohort of canine mammary carcinomas samples and associated them with tumor grade, histological type, presence of emboli, lymph node infiltration, recurrences, distant metastases, and survival. Our cohort allowed us to obtain a general overview of CD44 and CD24 expression in canine mammary carcinomas.
We found that presence and/or expression level (staining score) of CD44 did not show any correlation with poor prognosis parameters such as high grade, presence of emboli, or lymph node infiltration but was associated with histological type and especially complex tubular type. Concerning repartition of CD44 expression in mammary carcinomas, our results are consistent with those obtained in previous studies: frequent and abundant expression in anaplastic and tubular carcinomas, with basolateral expression in tubular carcinomas, and less frequent expression in solid carcinomas. 32,33,44
Interestingly, the expression of CD24 was associated with component (luminal and diffuse) and/or histological type and tumor aggressiveness parameters (high grade and presence of emboli). Moreover, expression of CD44–/CD24+ was also associated with numerous tumor aggressiveness parameters (grade, presence of emboli, infiltration of lymph node, distant metastasis). To our knowledge, we are the first to show that CD24 and CD44–/CD24+ phenotype are associated with poor clinicopathological parameters.
Our finding of CD24 as a potential poor prognosis factor is consistent with data obtained in human breast cancer. 8,17,29 CD24 overexpression has recently been reported to be an independent unfavorable prognostic factor in human breast cancer, especially for the luminal A and triple-negative breast cancer subtypes. 30 However, we did not find any association between CD24 and CD44–/CD24+ phenotype expression and survival, although the CD44–/CD24+ phenotype showed a trend toward an association with worse survival. This may be due to limitations of retrospective studies. For example, retrospective studies do not guarantee optimal or rigorous clinical follow-up. Clinical data could not be gathered for all cases, and some causes of deaths could not be related with certainty to cancer and had to be censored. Furthermore, in our study, no adjustment could be made regarding treatment modalities, as they were very heterogeneous among dogs, concerning both the type of surgery and adjuvant treatments. More specifically, chemotherapy modalities differed considerably both regarding the treatments used and the therapeutic plan such as the dates of beginning and end of treatments. Further studies with well-defined parameters of clinical follow-up are clearly needed.
Concerning the putative CSC phenotype CD44+/CD24–, our results demonstrated that CD44+/CD24– status does not show any relation with poor prognosis parameters (high grade, presence of emboli, or infiltrated lymph nodes) but is associated with histological type (like CD44 and CD44+/CD24+ expression) and absence of distant metastasis. Moreover, this phenotype is significantly associated with a better survival. However, previous studies have demonstrated an association of the CD44+/CD24– phenotype with high-grade canine carcinoma; the authors concluded that immunohistochemistry might be a reliable technique for the detection of CSC in dogs. 22,34 Discrepancies between our findings and the previous ones might be due to the size and the composition of different cohorts, the antibodies used for immunohistochemical analysis, and immunohistochemical scoring methods. For example, the aforementioned studies did not give a precise description of the cohort composition in terms of grade and histological type, which makes it hard to compare between studies. Moreover, the validation of antibody specificity in canine tissues was not detailed: in the study by Barbieri et al, 6 no tissue staining was obtained with the CD24 antibody, which raises doubt about the efficacy of this antibody to recognize the CD24 canine epitope and on the so-called CD44+CD24– phenotype. In the study by Magalhaes and colleagues, 34 only 100 stained or nonstained cells were counted to determine the number of stained cells. In the subgroup of mixed tumors, only epithelial neoplastic cells were counted, as they did not find labeling in mesenchymal and myoepithelial cells. Next, the study by Im and coworkers 22 did not specify the total number of analyzed cells, and the CD44+/CD24– phenotype was considered positive when more than 10% of tumor cells were labeled.
We found that the CD44+/CD24– phenotype was expressed in most tumors (65%), and high levels of expression were not compatible with a CSC signature. Supporting our findings, Blacking and colleagues 9 suggested that CD44 could rather be associated with proliferation than with a relevant CSC phenotype in canine mammary tumors. Alternatively, the high frequency of the CD44+/CD24– phenotype in our cohort could be explained by the predominant presence of some tumor subtypes. For example, the CD44+/CD24– phenotype is known to be enriched in the basal-like subgroup of breast cancer, generally considered a more aggressive subtype. 20,26,45 In addition, the CD44–/CD24+ phenotype was more often found in the HER2+ tumor subtype, another aggressive form of breast cancer. 20 As several works have demonstrated that the molecular classification of breast cancer can be also applied to canine mammary tumors, 1,18,23,24,47,54 it might be of interest to correlate the combined expression of CD44 and CD24 in different molecular subtypes of canine tumors.
In conclusion, our study shows for the first time that CD24 and the CD44–/CD24+ phenotype are associated with poor clinicopathological parameters in canine mammary tumors. Although further studies are needed, CD24 might be a potential marker of aggressiveness for canine mammary tumors. Moreover, CD44+/CD24– expression was associated with a better prognosis and occurred at a high frequency and high level. These features indicate that this phenotype is not suitable to detect CSCs in a cross-sectional study of canine mammary tumors that includes different subgroups. To more exactly determine the clinical value of CD44 and CD24, further studies should be done in a larger cohort, respecting standardized methods (cohort composition and histological characterization, antibody validation, immunohistochemical scoring methods, clinical follow-up parameters) and comprising sufficient numbers of molecular subtypes, as what has been done for human breast cancer. Finally, it will be also important to analyze the expression of other CSC markers like ALDH1A1, CD133, Sca-1, CD10, CD34, or CD49f in different subtypes of canine tumors. 16,37,38,51,52
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818813653 - CD44 and CD24 Expression and Prognostic Significance in Canine Mammary Tumors
Supplemental Material, DS1_VET_10.1177_0300985818813653 for CD44 and CD24 Expression and Prognostic Significance in Canine Mammary Tumors by Bernadette Rogez, Quentin Pascal, Audrey Bobillier, François Machuron, Chann Lagadec, Dominique Tierny, Xuefen Le Bourhis and Valérie Chopin in Veterinary Pathology
Supplemental Material
Supplemental Material, DS2_VET_10.1177_0300985818813653 - CD44 and CD24 Expression and Prognostic Significance in Canine Mammary Tumors
Supplemental Material, DS2_VET_10.1177_0300985818813653 for CD44 and CD24 Expression and Prognostic Significance in Canine Mammary Tumors by Bernadette Rogez, Quentin Pascal, Audrey Bobillier, François Machuron, Chann Lagadec, Dominique Tierny, Xuefen Le Bourhis and Valérie Chopin in Veterinary Pathology
Footnotes
Acknowledgements
We thank the staff of OCR (Oncovet Clinical Research) for their support and the biostatisticians of Lille University Hospital, particularly Hélène Behal and Julien Labreuche, for their time and implication. Special thanks are given to Tiffany Cheval and Emmanuel Bouchaert for their excellent technical assistance and to Agata Rybicka, Virginie Coste, and Thomas Bucher for their help in building and improving this study. Finally, we thank the referring veterinarians who gave us the clinical and follow-up data and the owners of the dogs included in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This study was supported by FEDER FSE Nord-Pas de Calais/GEFLUC Flandres Artois/SIRIC OncoLille and OCR (Oncovet Clinical Research).
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
