Abstract
SOX2 is a major transcriptional regulator of stem cell pluripotency and self-renewability. Its expression in cancer stem cells from several different tumor types in humans and rodent models directly implicates SOX2 in tumorigenicity, metastasis, drug resistance, recurrence, and poor survival. Our objective was to investigate the expression of SOX2 in canine neoplasia. Immunohistochemistry for SOX2 was performed in sets of 10 archived formalin-fixed paraffin-embedded tissues from 45 distinct canine neoplasms. Normal expression of SOX2 was evaluated in a canine tissue microarray. Strong and diffuse SOX2 intranuclear immunolabeling was consistently found in the majority of ectodermal (13/15) and endodermal tumors (5/7). Negative, variable, or inconsistent SOX2 intranuclear immunolabeling was detected in the majority of mesodermal tumors (10/16) and in tumors with dual or uncertain origin (5/7). Although further studies are necessary to understand mechanistically how SOX2 contributes to the biology of each tumor type, this study demonstrates the expression of SOX2 in a wide variety of canine cancers. In the future, screening methods based on cellular plasticity and pluripotency biomarkers may provide avenues for the rational design of therapeutic strategies that target vulnerable signals upstream or downstream of SOX2 in different cancers, and possibly offer novel clinical applications for SOX2 as a prognostic indicator.
Keywords
The cancer stem cell (CSC) model proposes that, analogous to normal tissues, cancers have a hierarchical organization into clonally derived populations of heterogeneous cells with different cell phenotype and proliferative potentials. 6 This model suggests that the only cells able to self-renew and produce a heterogeneous tumor cell population are CSCs. These cells are especially prone to pathological reprogramming via reactivation of signaling pathways and genes that are essential for embryonic development and pluripotency. 16 Progenitor cells derived from normal stem cells lose their ability to divide upon achieving terminal differentiation after a certain number of divisions, but CSCs give rise to progeny with uncontrolled proliferative potential, containing multiple branches or subclones that evolve independently. 6 While various causes can lead to reprogramming—including genetic and epigenetic alterations or deregulated microenvironment—cancer initiation and progression seem to be deeply rooted in deregulated control of core embryonic stem cell pluripotency factors. 16
Defining CSC markers holds great promise for the development of CSC-targeted therapies that can be translated into the clinic to benefit patient survival. SOX2 is a major transcriptional regulator of stem cell pluripotency and self-renewability, is one of the most relevant markers of CSCs, and is a crucial regulator of tumorigenesis. 16,18 Small changes in the levels of SOX2 are proven to radically affect tumor cell behavior, and its expression in CSCs at both protein and RNA levels from at least 25 different tumors in humans and rodent models has been directly implicated in tumorigenicity, metastasis, drug resistance, recurrence, and poor survival. 20 The aberrant expression of SOX2 in distinct tumor types suggests that SOX2 may mastermind the tumor initiating potential of cells in these tumors. 18 Thus far, in the veterinary literature, the SOX2 gene was overexpressed in metastatic canine mammary tumors, 4 and isolation of primary canine osteosarcoma cell cultures proved that SOX2-positive cells exhibit distinctive sensitivity to drugs with potential to inhibit proliferation, metastasis, and self-renewal. 7
The existence of CSCs within heterogeneous tumors has profound implications for cancer therapy. Conventional chemotherapeutic and radiotherapeutic strategies often can efficiently eliminate the bulk of the tumor composed of intensively proliferating cancerous cells. However, in many instances, they leave behind residues of rarely dividing and therapy-resistant CSC populations, which subsequently may be responsible for promoting tumor regrowth. 6,8,10,18 Defining the molecular architecture of CSCs may therefore provide avenues for the rational design of cancer treatment strategies that, combined with conventional therapy, target both CSCs and bulk tumor cells. In veterinary medicine, very little is known about CSC niches in domestic animal species. As targeting SOX2 expression or its mode of action has the potential to improve clinical outcomes, recognizing alterations of SOX2 expression between normal and neoplastic tissues is an essential first step. Therefore, our objective was to investigate the expression of SOX2 in different types of canine neoplasia.
Materials and Methods
Ten samples from 45 distinct canine neoplasms embryologically originating from the 3 germ cell layers were assessed for SOX2 expression, for a total of 450 cases evaluated. For tumors in which a grading scheme is routinely used for diagnosis, only samples diagnosed with the highest grade were chosen: that is, mast cell tumors (grade III 12 /high grade 9 ), soft tissue sarcomas (grade III 2 ), and mammary tumors (grade III 13 ). Cases were retrieved from the Section of Anatomic Pathology archives at the New York State Animal Health Diagnostic Center. Normal expression of SOX2 was evaluated in a canine tissue microarray, which included brain, liver, lung, kidney, urinary bladder, heart muscle, skeletal muscle, aorta, haired skin, oral mucosa, esophagus, stomach, small intestine, large intestine, prostate gland, testicle, and lymph node, as well as in a set of normal tissues collected at necropsy from a dog including liver, lung, spleen, heart, kidney, thymus, stomach, pancreas, duodenum, jejunum, ileum, cecum, colon, mesenteric lymph node, cerebrum, cerebellum, and brainstem. Formalin-fixed paraffin-embedded tissues were used for immunohistochemistry against SOX2 (mouse monoclonal anti-SOX2 antibody, clone 20G5, Thermo Fisher Scientific, Catalog No. MA1-014, RRID AB_2536667), using the Leica Bond Max Automated Immunohistochemistry Staining System, according to the manufacturer’s instructions (Leica Microsystems). Briefly, tissues were sectioned at 5 μm, and deparaffinized with Bond Dewax Solution (Leica, Catalog No. AR9222). Pretreatment with heat-induced antigen retrieval was performed for 30 minutes using Tris/EDTA pH 9 (Bond Epitope Retrieval Solution 2, Leica, Catalog No. AR9640). Endogenous peroxidase activity was blocked with a 3% peroxide solution for 5 minutes (Leica, Catalog No. DS9800). The antibodies were diluted at 1:500 and applied to the slides for 15 minutes. Next, the biotin-free PowerVision poly-polymeric horseradish peroxidase anti-mouse (Leica, Catalog No. PV6114) IgG reagent was applied to the slides for 10 minutes. Tissues were then incubated with the Bond Polymer Refine Detection for 10 minutes (Leica, Catalog No. DS9800). Subsequently, tissues were developed with 3,3-diaminobenzidine (DAB) chromogen (Leica, Catalog No. DS9800) for 10 minutes. The slides were counterstained with hematoxylin (Leica, Catalog No. DS9390) for 5 minutes, dehydrated, cleared, and mounted. In the negative controls, isotype-matched antibodies replaced the primary antibodies. The immunolabeling intensity was classified subjectively as negative, weak, moderate, or strong, based on the degree of nuclear labeling in neoplastic cells evaluated via light microscopy. This correlates to no nuclear labeling (negative), faint nuclear labeling (weak), patchy nuclear labeling (moderate), and intense, diffuse nuclear labeling (strong). The immunolabeling distribution was further classified as multifocal, if immunopositive neoplastic cells were distributed in pockets throughout the tissue, or diffuse, if immunopositive cells were present uniformly throughout the tissue.
Results
In the normal control canine tissues, both in the microarray (Supplemental Fig. 1) and in the samples collected at necropsy, strong intranuclear SOX2 immunolabeling was diffusely identified in endothelial cells, basal squamous epithelium, gastric glandular epithelium, intestinal crypt epithelium (Supplemental Fig. 2), bronchial epithelium, bronchiolar epithelium, germ cells (Supplemental Fig. 3), and neural precursor cells. Weak to moderate intranuclear immunolabeling was multifocally detected in glial cells, biliary cells, hepatocytes, renal tubules (Supplemental Fig. 4), glomeruli, pneumocytes, urothelium, glandular epithelium, Sertoli cells, and Leydig cells (Supplemental Fig. 3). Lack of immunolabeling was found in leukocytes (Supplemental Fig. 5), skeletal (Supplemental Fig. 6) and smooth muscle, fibrous connective tissue, and neurons.
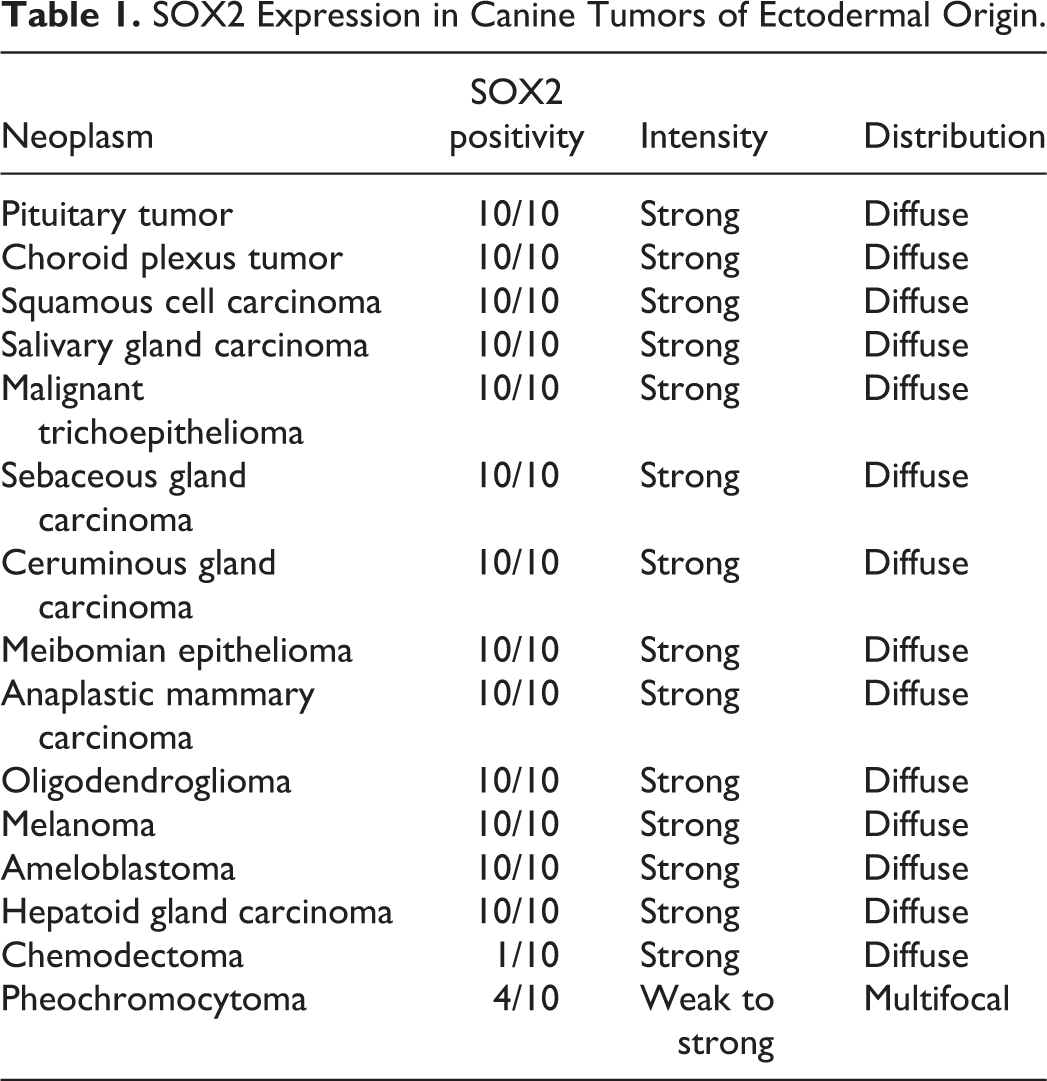
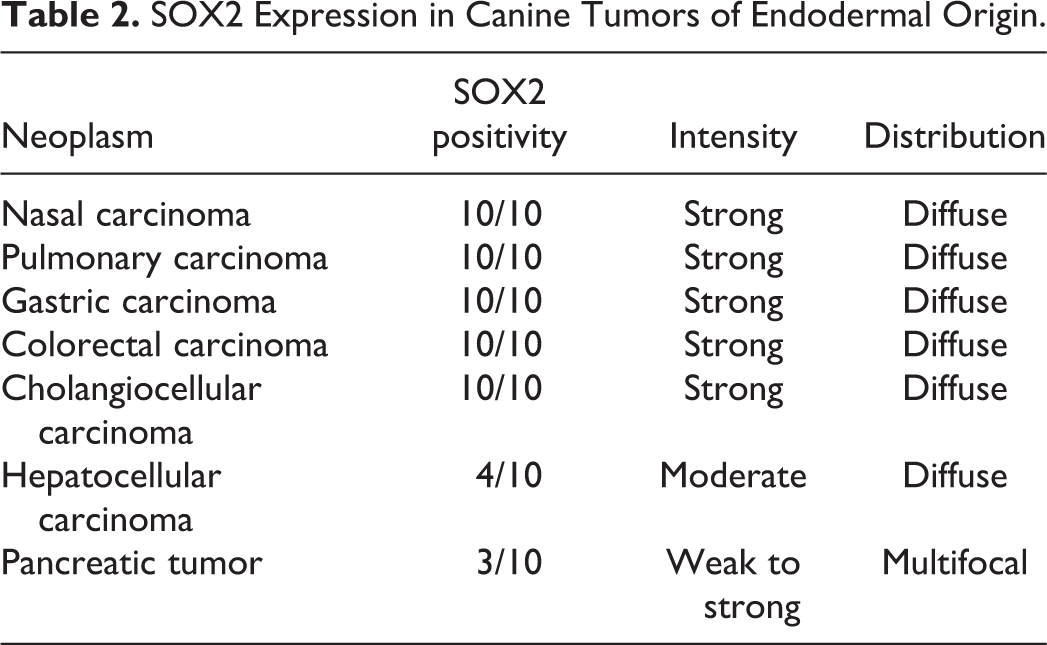
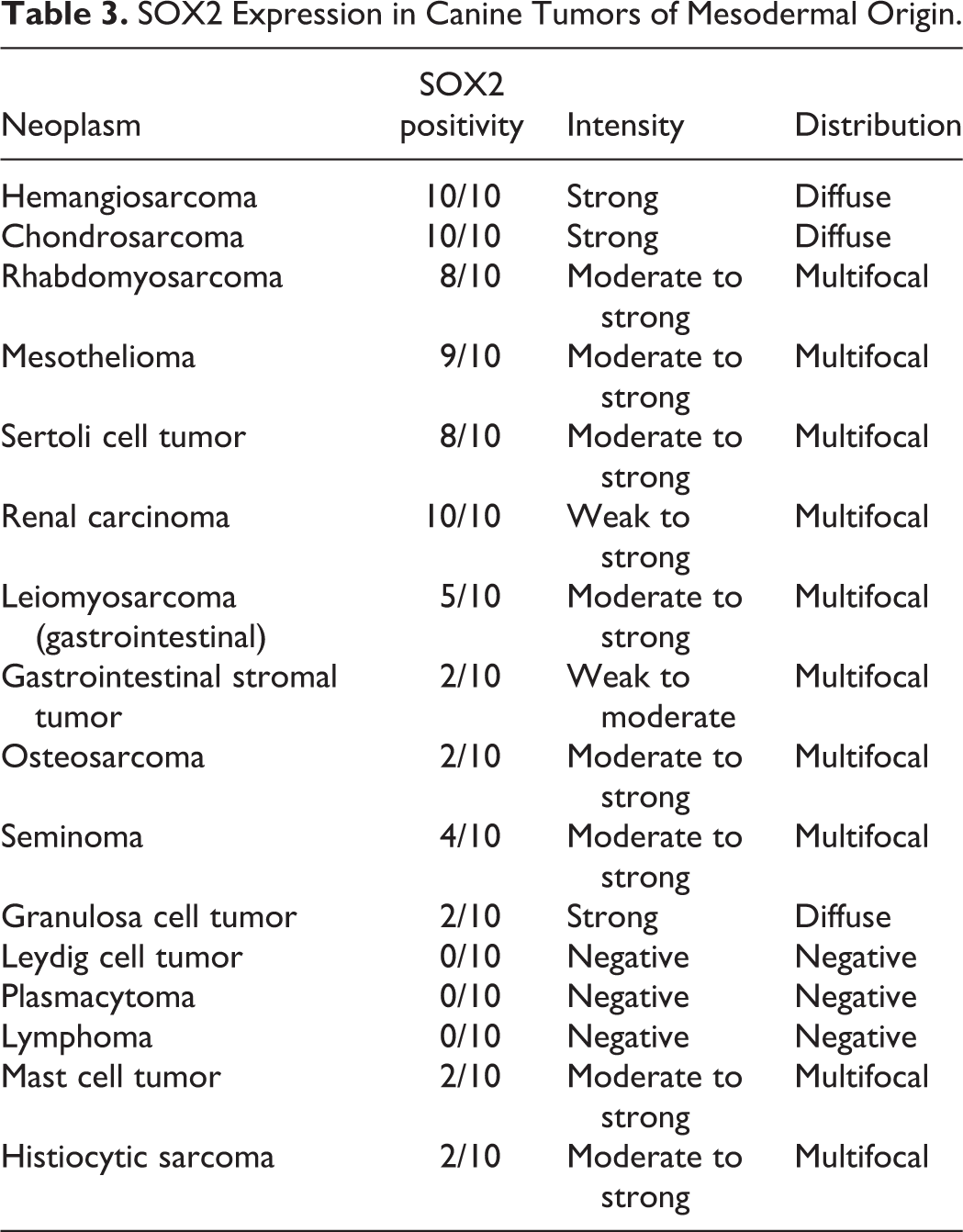
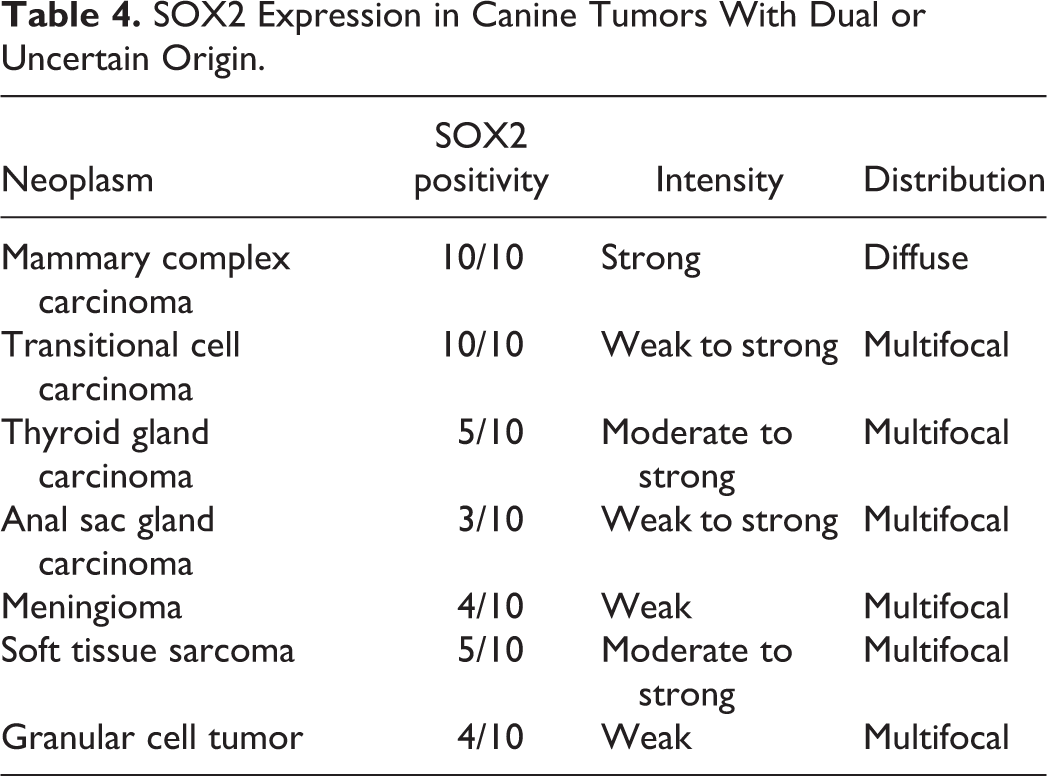
The SOX2 immunolabeling intensity and distribution in tumors of ectodermal, endodermal, mesodermal, and of dual or uncertain origin are detailed in Tables 1 to 4, respectively. In brief, strong and diffuse SOX2 intranuclear immunolabeling was consistently found in all evaluated oligodendrogliomas (Fig. 1), choroid plexus tumors (Fig. 2), as well as in all examined colorectal (Fig. 3), gastric (Fig. 4), and mammary complex (Fig. 5) carcinomas. Thyroid gland carcinomas (Fig. 6) had variable or inconsistent SOX2 intranuclear immunolabeling. Among sarcomas, all chondrosarcomas (Fig. 7) and most rhabdomyosarcomas (Fig. 8) had strong and diffuse SOX2 intranuclear immunolabeling, but this pattern was only found in half of the evaluated gastrointestinal leiomyosarcomas (Fig. 9) and soft tissue sarcomas (Fig. 10), whereas the other half had variably weak to negative intranuclear immunolabeling. As for round cell tumors, all evaluated plasmacytomas (Fig. 11) and lymphomas (Fig. 12) lacked SOX2 immunolabeling. However, 2 mast cell tumors (Fig. 13) and 2 histiocytic sarcomas (Fig. 14) had moderate to strong SOX2 intranuclear immunolabeling.
SOX2 Expression in Canine Tumors of Ectodermal Origin.
SOX2 Expression in Canine Tumors of Endodermal Origin.
SOX2 Expression in Canine Tumors of Mesodermal Origin.
SOX2 Expression in Canine Tumors With Dual or Uncertain Origin.
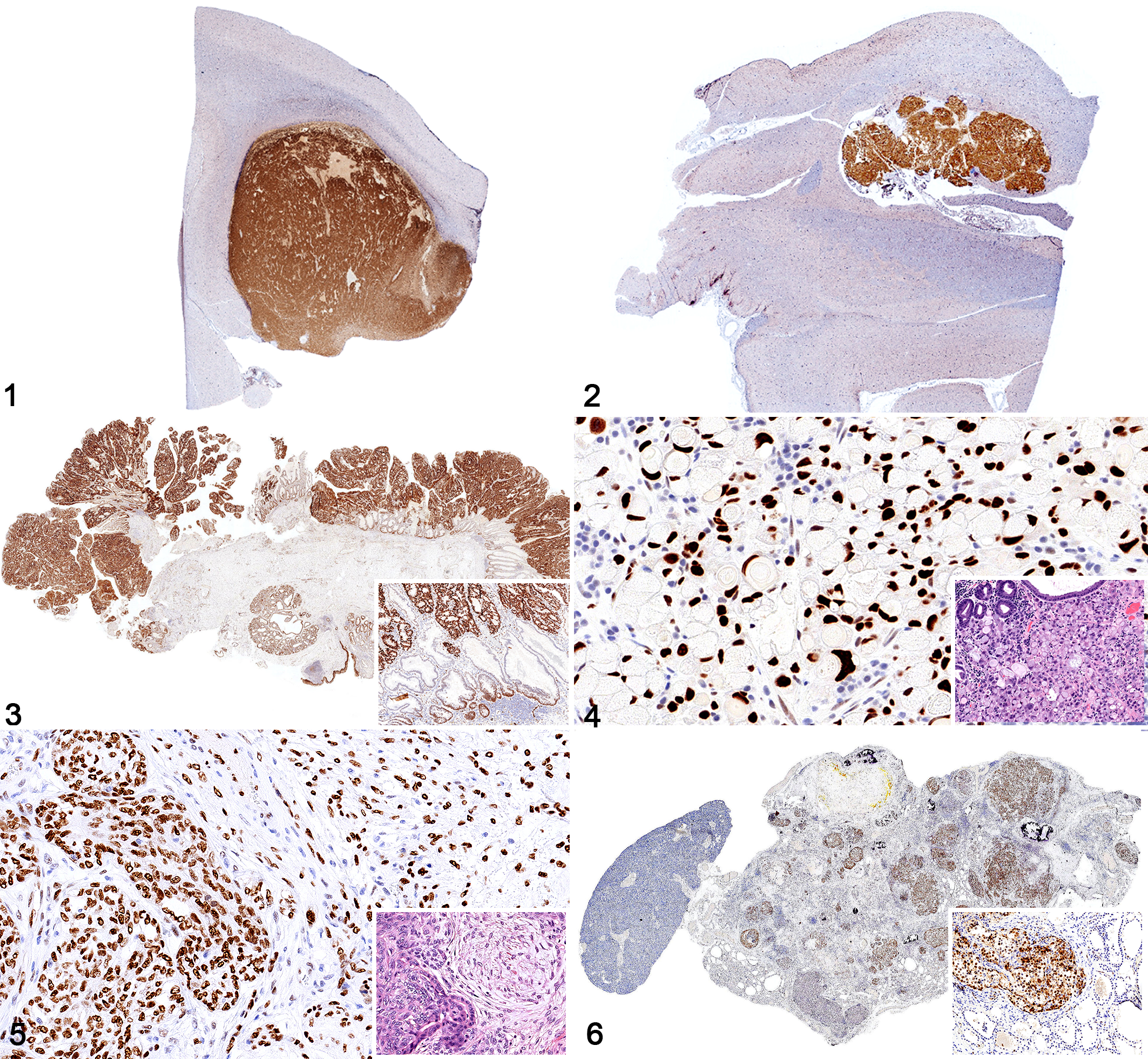
Glial tumors and carcinomas, dog. Immunohistochemistry for SOX2.
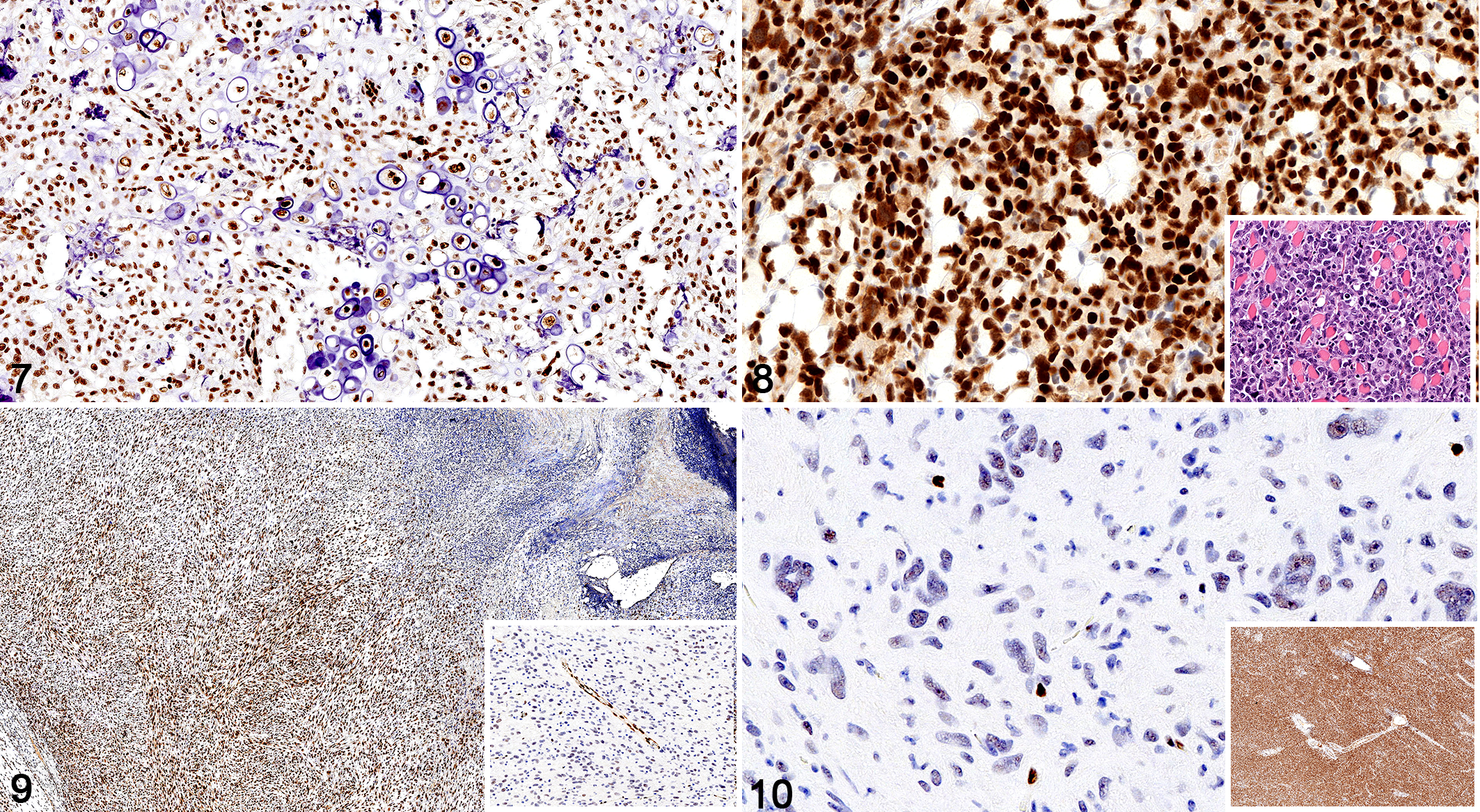
Sarcomas, dog, immunohistochemistry for SOX2.
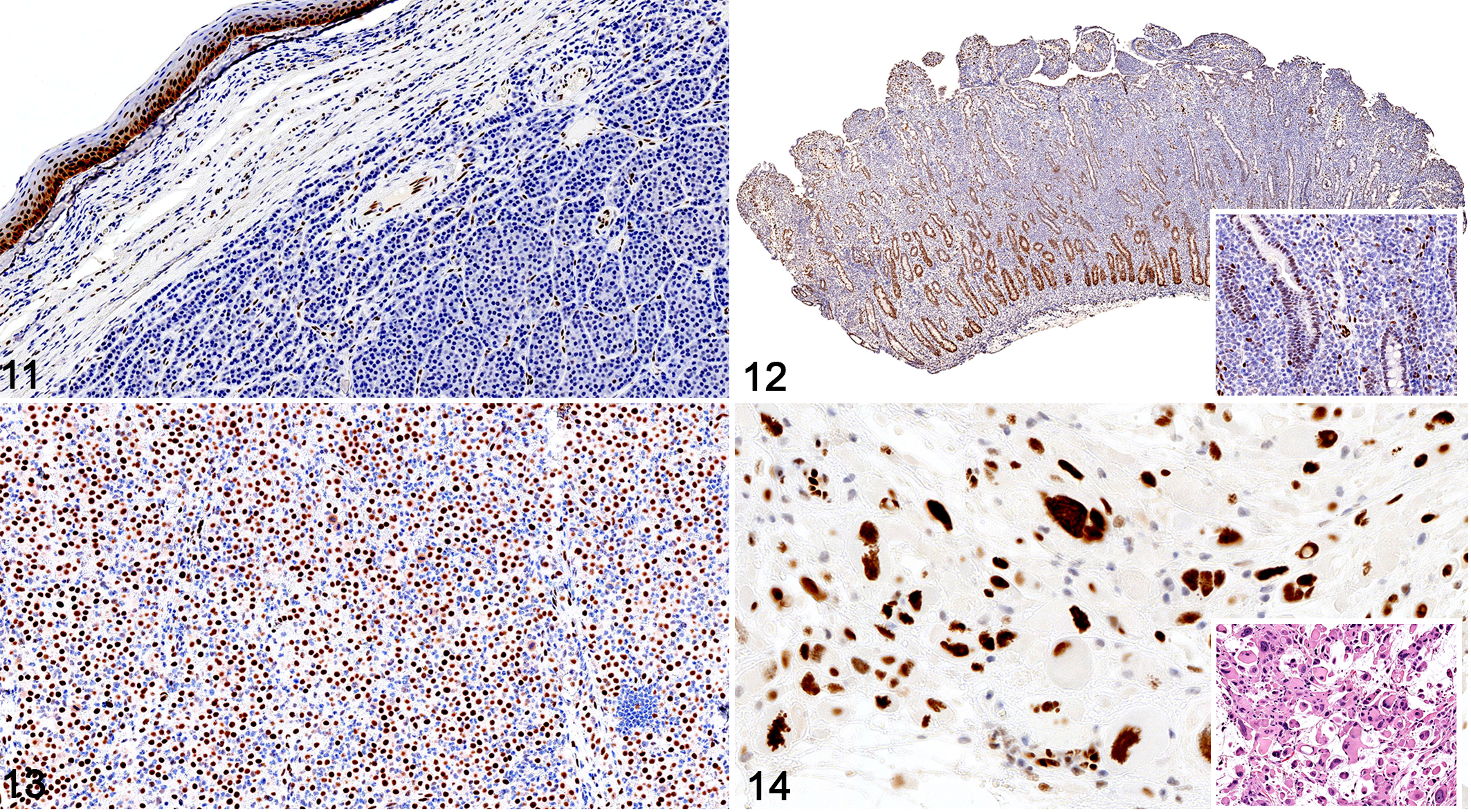
Round cell tumors, dog. Immunohistochemistry for SOX2.
Discussion
This study establishes SOX2 as a marker of many adult stem/progenitor cell types in the dog and reveals variable expression of SOX2 in different canine cancers. In the control samples, we identified SOX2 immunolabeling in several normal adult epithelial tissues, including the integumentary and mucosal squamous epithelium, glandular epithelium in the gastrointestinal tract, respiratory epithelium, testicular germ cells, and brain. These findings are similar overall to those previously reported in a systematic survey of SOX2 immunohistochemical expression in adult murine tissues. 1 Because SOX2 is considered a major transcriptional regulator that is commonly expressed in embryonic stem cells, fetal progenitors, and a number of adult stem cells, these results may point toward similarities between target genes and pathways that are crucial for molecular reprogramming and regulation of pluripotent and adult stem cells.
With regard to the neoplasms studied herein, our study revealed consistent, strong, and diffuse labeling for SOX2 in a wide variety of tumors (21/45), which suggests a potential role for SOX2 in these cancer types, including the majority of the evaluated tumors arising from the ectodermal (13/15) and endodermal (5/7) embryonal layers. Variable or multifocal SOX2 labeling, including in most mesodermal tumors (10/16), likely reflects a more heterogeneous population within this tumor group, such as the soft tissue sarcomas. Neoplasms with a consistent lack of SOX2 expression may utilize different pathways to malignancy, such as Leydig cell tumors and round cell tumors including all lymphomas and plasmacytomas, and the majority of histiocytic sarcomas and mast cell tumors.
SOX2 is one of the members of the core transcriptional network that guarantees the maintenance of stemness of embryonic and adult stem cell populations in lineages of all embryonic germ cell layers. 11,16 This network also includes the transcription factors OCT4 and Nanog—part of the so-called “Yamanaka factors” used to reprogram somatic cells into induced pluripotent stem cells. 17 Through this network, these 3 molecules cooperate functionally to promote the expression of pluripotency-associated genes and to suppress the expression of genes associated with differentiation. 11 During the reprogramming process, cells undergo global epigenetic remodeling, which resets their epigenome to a state similar to the one found in embryonic stem cells. 11,17 Interestingly, similarities found between the process of pluripotency reprogramming and tumorigenesis are striking. Both processes require the expression or activation of oncogenes, the inactivation of tumor suppressor genes, overriding of senescence and apoptotic barriers, extensive epigenetic changes, and a metabolic switch toward a glycolytic metabolism. 8 Out of these 3 Yamanaka factors, SOX2 seems to be the most prominent one associated with cancer. 16,18,20
Given its crucial role in stem cell maintenance and reprogramming toward pluripotency, it is not surprising that SOX2 has been demonstrated as essential for tumorigenicity and CSC self-renewal in many tumor types. 8,16,18,20 In sarcomagenesis studies, regardless of the level of many other potential CSC markers, only cell lines with upregulated SOX2 expression were found to be tumorigenic in NSG mice. 15 Remarkably, in studies of cutaneous squamous cell carcinomas in humans, conditional deletion of SOX2 from the epidermis dramatically delayed tumor onset and lesions induced by carcinogen exposure. 18 This evidence provides direct support for SOX2 as being functionally critical for conferring tumor initiation potential, and not merely behave as a bystander stemness marker. Accordingly, all squamous cell carcinomas in our study had strong and diffuse SOX2 intranuclear immunolabeling. In mice, SOX2 expression is detectable in embryonic neural progenitor cells, but also persists in adult neural stem cells and neural progenitor cells in the subventricular zone of the lateral ventricle, the subgranular zone of the hippocampus, and the ependyma of the adult central canal. 3 A similar distribution of SOX2-positive cells is found in dogs, where the subventricular regions are normally considered a primary niche for progenitor cells in the adult brain. 5 The stem cell origin of tumor cells aligned with this particular distribution may explain why the most frequent neuroanatomical localization of gliomas in dogs occurs in the periventricular regions of the brain. In line with the previously reported SOX2 gene overexpression in canine metastatic mammary tumors, 4 all evaluated anaplastic and complex mammary carcinomas from this study had strong and diffuse SOX2 intranuclear immunolabeling, including both epithelial and myoepithelial components in complex type cancers. As for pituitary tumors, neoplastic cells were consistently immunolabeled in all evaluated samples of this study. However, SOX2 expression was previously reported to be unsuitable as a clinical prognosticator in canine corticotroph pituitary adenomas, as there was no correlation between SOX2 immunolabeling and clinical parameters, such as size of the pituitary tumors, histological diagnosis, survival time, and disease-free interval. 19
While the effects of SOX2 can be highly context-dependent, the predominant protein overexpression or gene amplification of SOX2 has been demonstrated in many embryologically distinct cancer types in humans and rodent models. 20 Two factors are proposed to contribute to SOX2 upregulation: increased numbers of SOX2-positive cells in the tumor population and/or increased SOX2 levels in individual tumor cells. Regardless, the aberrant activation of SOX2 within a group of transformed cells might cause them to shift toward a CSC phenotype. 8,18 In our study, for instance, the lack of SOX2 expression in normal skeletal muscle cells, in contrast to the strong and diffuse SOX2 immunolabeling in most rhabdomyosarcomas, suggests that the gain of cellular plasticity via genetic reprogramming lining to the mechanisms used to control the pluripotent state might be essential in the process of malignant transformation. In these tumors, neoplastic cells might be recapitulating a developmental program that was normally present during embryonic myogenesis, but might now be reactivated in a very dysregulated manner. Likewise, in oligodendrogliomas, SOX2-positive cells may have reactivated a genetic program and followed an aberrant oligogenic differentiation that mirrored adult and developmental oligogenesis, thus reflecting the recapitulation of a developmental program in CSCs.
The effect exerted by SOX2 can vary depending on the cancer type, with different effects in different cell lineages based on its cellular and molecular interactions. Aside from cellular morphology and tumor histology, additional features of heterogeneity between tumors have been discovered, including variation in cell surface markers, genetic abnormalities, growth rates, and response to therapy. Even within the same tumor type, there is considerable variation in driver mutations and the same driver mutations can occur in different tumor types. 10 The high intertumor and intratumor heterogeneity makes it difficult to establish without functional testing whether a particular somatic polymorphism is a driver mutation or a passenger variant. Interestingly, the observation that SOX2 expression is not required for late stages of tumor development 8 suggests that, similar to pluripotency reprogramming, once reprogramming has occurred and the tumor fate has been primed, the initiating genetic events may no longer be necessary for the later stages of malignant development—therefore, the initiating lesion could become a passenger mutation. Although high SOX2 expression is often linked to cancer progression, the interplay between a multitude of factors, rather than a single molecule, determines the course and severity of cancer. While our study reveals robust presence of SOX2-immunolabeled neoplastic cells in late stage tumors, it remains unclear how mechanistically the SOX2 expression in CSCs differ from that of normal stem cells, and it is likely that not all SOX2-positive cancer cells are CSCs and vice versa. This homogeneous immunolabeling pattern might indicate SOX2 as a lineage-survival factor and not a distinguishing marker of the CSC compartment, as previously described in some epithelial tumors in which SOX2 expression coincides with chromosomal amplifications. 14
Intrinsic and extrinsic factors could have influenced the immunohistochemical results in some evaluated samples, including decalcification process (eg, in osteosarcomas), tumor subtype and hormonal functionality (eg, in pancreatic and thyroid gland tumors), and fixation or autolytic artifacts in any large masses. Variable immunolabeling intensity and distribution might also have been influenced by other unknown inherent tissue abnormalities. Of note, a single chemodectoma derived from a surgical biopsy sample exhibited strong and diffuse SOX2 immunolabeling (1/10), whereas all postmortem samples of chemodectomas (9/10) were diffusely negative. On the other hand, choroid plexus tumors, for instance, were evenly evaluated from biopsy (5/10) and necropsy (5/10) specimens, and no overt changes in the SOX2 immunolabeling pattern were noted. The sample size for each tumor category was limited by archive availability and financial constraints. In future studies, these pitfalls can be further assessed with a larger cohort of samples.
Based on the aberrant level of SOX2 in many types of cancer and the roles of promoting cancer progression in humans and rodent models, studying SOX2 may provide important insight into signal pathways associated with tumorigenesis and help guide cancer therapy in dogs. Screening methods based on cellular plasticity and pluripotency biomarkers may provide avenues for the rational design of therapeutic strategies that target vulnerable signals upstream or downstream of SOX2 in different cancers. The evidence of SOX2 as a functional CSC marker in different types of canine tumors provides a unique window of opportunity to dissect molecular signatures associated with CSCs and to interrogate differentiation programs that operate within these cells.
Supplemental Material
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820960130 - SOX2 Expression in Canine Neoplasia
Supplemental Material, sj-pdf-1-vet-10.1177_0300985820960130 for SOX2 Expression in Canine Neoplasia by Ileana C. Miranda and Andrew D. Miller in Veterinary Pathology
Footnotes
Acknowledgements
We thank the histopathology technicians for assistance with case processing and Jennifer Patterson for helping create the figure panels.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research presented herein was partially funded by a monetary award to one of the authors (ICM) at the 2018 Cornell University College of Veterinary Medicine Clinical Investigator’s Day.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
