Abstract
Accumulating evidence supporting the cancer stem cell (CSC) hypothesis is based on the finding that tumors contain a small population of self-renewing cells that generate differentiated progeny and thereby contribute to tumor heterogeneity. CSCs are reported to exist in several human cancers, yet only a few reports demonstrate the existence of CSCs in primary lung cancer in dogs. In this study, the authors established a cancer cell line derived from a canine primary lung adenocarcinoma and identified a side population (SP) of cells that displayed drug-resistant features. To confirm the characteristics of these SP cells, the authors investigated the tumorigenicity of the cells in vivo by using a nude mouse xenograft model. Only 100 SP cells were able to give rise to new tumors, giving a 10-fold enrichment over the main population (MP) of cells, suggesting that these cells have the cancer-initiating ability of CSCs. Further studies characterizing CSCs in canine lung adenocarcinoma might contribute to the elucidation of the mechanisms of tumorigenesis and to the establishment of novel therapeutic strategies.
Primary lung cancer is markedly less common in dogs than in humans and accounts for approximately 1.2% of all canine tumors. 9 Little is known about the characteristics of canine lung cancer, in particular cancer stem cells (CSCs). Accumulating evidence supporting the CSC hypothesis is based on the identification of a small population of self-renewing cells in tumors that generate differentiated progeny by asymmetric division. 18 Numerous investigators have provided evidence that CSCs exist in various human tumors such as hepatocellular caricinoma, 21 brain tumor, 12 breast cancer, 7 melanoma, 10 prostate tumor, 3 gastroenterological cancer, 5 lung cancer, 6 and bone sarcoma. 8 It is reported that CSCs of canine tumors exist in osteosarcoma, 20 glioblastoma, 13 and mammary gland tumor 2 .
To identify and characterize CSCs, cell surface molecules as several makers, such as CD44,7,3,18 CD99, 18 CD133,16,18,21 and Oct4, 19 were investigated. In contrast, a subpopulation that effluxes the DNA-binding dye Hoechst 33342 out of the cell membrane through an adenosine triphosphate–binding cassette (ABC) transporter was recognized as a hematopoietic stem cell population. 4 This cell population expressing the ABC transporter was defined as side population (SP) cells, which were distinguished from cells of the other population (non-SP or main population [MP]). Recent studies demonstrated that SP cells could be characterized as CSCs in primary tissues and tumor cell lines.5–8
In this study, we established a line of canine adenocarcinoma cells derived from a canine primary lung adenocarcinoma. From these cells, we isolated SP cells using the Hoechst 33342 dye-based side population technique that is based on an important property relating to multidrug resistance that CSCs have more active ABC transporter compared with MP. 18 We found that the SP cells displayed higher tumorigenic potential than non-SP cells in a nude mouse xenograft model in vivo. These results suggest that a small subpopulation of SP cells have CSC-like properties.
Methods
Establishment of the Cell Line
A 10-year-old male Pointer weighing 18.8 kg underwent surgical resection of a lung tumor, diagnosed histopathologically as lung adenocarcinoma (Fig. 1). After surgery, an accumulation of carcinomatous pleural effusion was observed, and a sample was removed for primary cultivation of tumor cells. Cells were cultured in Dulbecco’s modified eagle’s medium (DMEM) (Sigma-Aldrich, St Louis, MO) supplemented with 10% fetal bovine serum, 1% nonessential amino acids solution (Invitrogen, Carlsbad, CA), 100 U/ml penicillin, 100 μg/ml streptomycin, and 0.29 mg/ml L-glutamine (Invitrogen) in a humidified atmosphere with 5% CO2 at 37°C. When the cells were approximately 95% confluent, they were subcultured at a 1:3 dilution repeatedly for 50 passages over 7 months and were designated as CLAC cells. CLAC cells subcultured over 50 passages were used in all experiments.
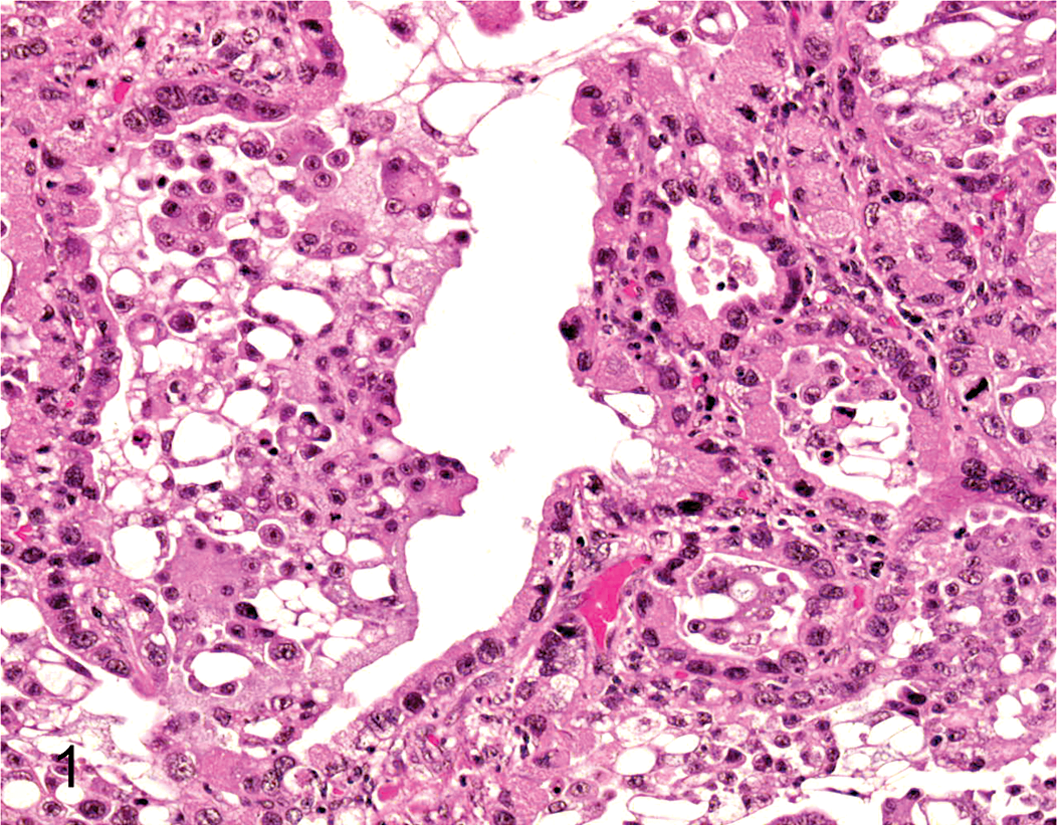
Adenocarcinoma, lung; dog. Histopathology of the original tumor. HE.
Growth Curve
The growth curve and doubling time of the cells were examined. Cells were plated at a density of 5.0 × 105 cells/well in 6-well dishes with the medium and counted at days 1, 2, 4, 6, and 8 with a cytometer. Doubling time was determined using a semilogarithmic graph.
Xenotransplantation for Assessment of Tumorigenesis
Inbred, female BALB/c nu/nu mice (20-22 g, 7 weeks of age) were obtained from Japan SLC, Inc. (Shizuoka, Japan). All procedures were performed in compliance with the guidelines of the Animal Research Committee of Azabu University. Cells were resuspended at 1 × 101 to 1 × 106 cells per 50 μl of phosphate-buffered saline (PBS), and following the addition of 50 μl of Matrigel (BD Biosciences, Bedford, MA) they were injected subcutaneously into the dorsal region of the mice. After implantation, the tumor volume (tumor volume = length × [width]2 × 0.5) was measured in situ periodically for up to 8 weeks. At the end of the 8-week assessment period, the xenografted tumors were harvested and immediately fixed with 10% buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin (HE).
Identification of the Side Population
Cell suspensions were labeled with the dye Hoechst 33342 (Cambrex Bio Science Walkersville Inc., MD) using the methods described by Goodell et al 4 with some modifications. Briefly, the cells were trypsinized and resuspended in prewarmed DMEM supplemented with 5% fetal bovine serum (FBS) at a concentration of 1 × 106 cells/ml. Hoechst 33342 was added to a final concentration of 5.0 μg/ml in the presence or absence of verapamil (150 μg/ml; Sigma-Aldrich), an inhibitor of the ABC transporter. The cells were incubated at 37°C for 90 minutes with continuous shaking and then washed with ice-cold PBS containing 5% FBS. The cells were centrifuged at 4°C and then resuspended in ice-cold PBS, 5% FBS. Flow cytometry and cell sorting were performed using FACSVantage SE Diva (BD Biosciences, Bedford, MA). Hoechst 33342 was excited at 357 nm, and its fluorescence was analyzed using dual wavelengths (blue, 402-446 nm; red, 650-670 nm).
Results
Morphological and Growth Characteristics
The CLAC cells showed a spindle-shaped morphology and grew in a monolayer without contact inhibition (Fig. 2). The population doubling time of the cells was approximately 51 hours (Fig. 3).
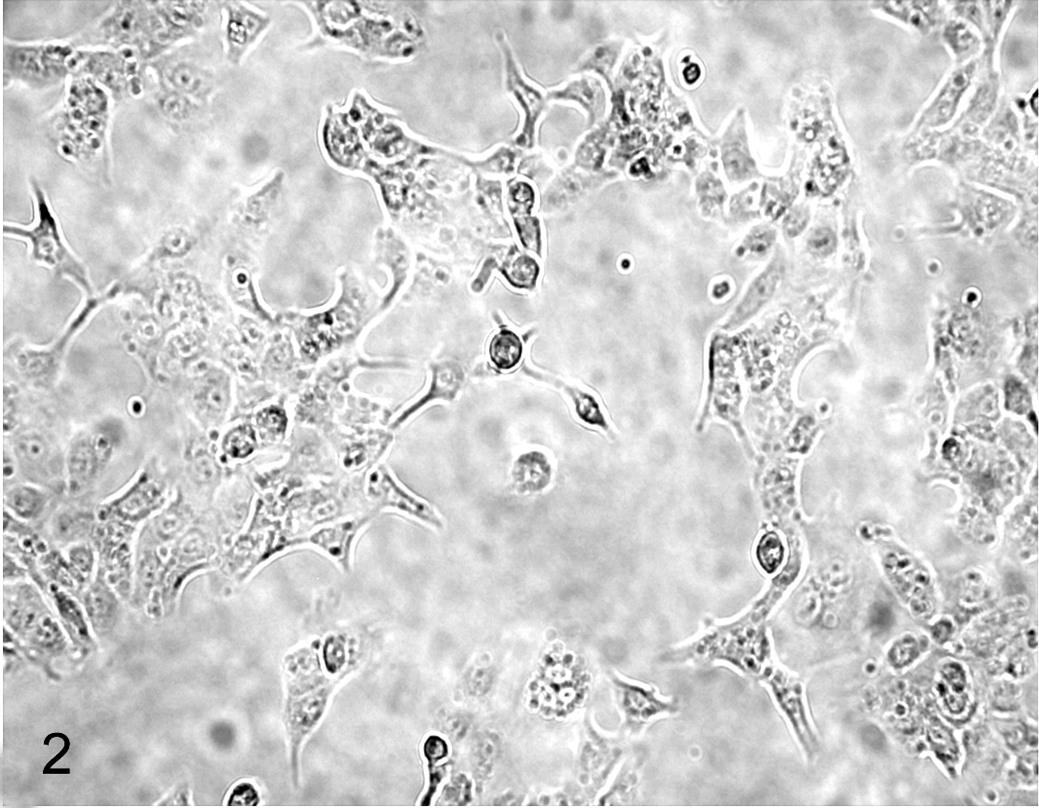
Microscopy of CLAC cell line.
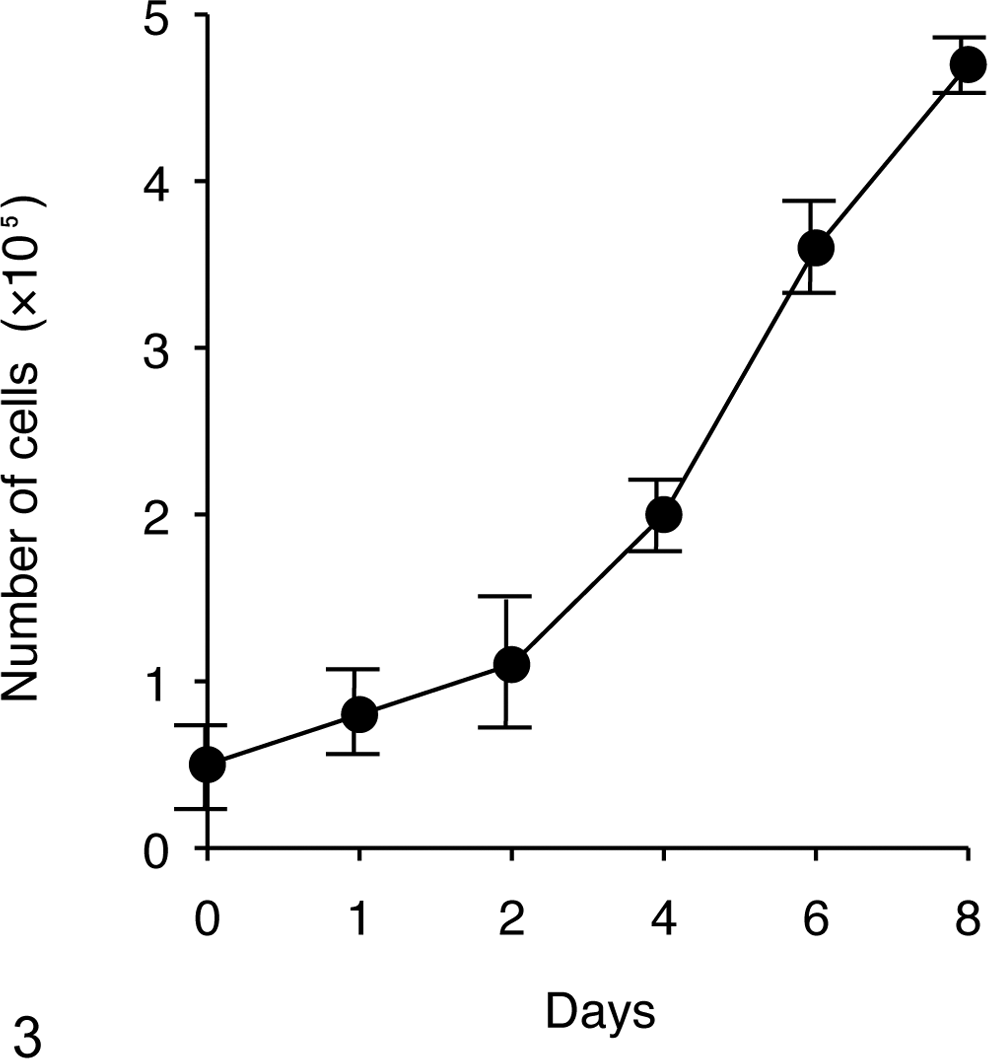
Growth curve of the CLAC cells (mean ± SD).
Xenotransplantation
Tumors approximately 250 mm 3 in size were formed in 4 of the 6 nude mice by 40 days after inoculation with 106 CLAC cells (Fig. 4). Subcutaneous tumors were diagnosed histopathologically as adenocarcinoma (Fig. 5), and were almost identical to the primary tumor. There were no metastatic lesions and/or tumors in any organs examined (brain, lung, heart, spleen, liver, intestine, urinary bladder, and bone marrow).
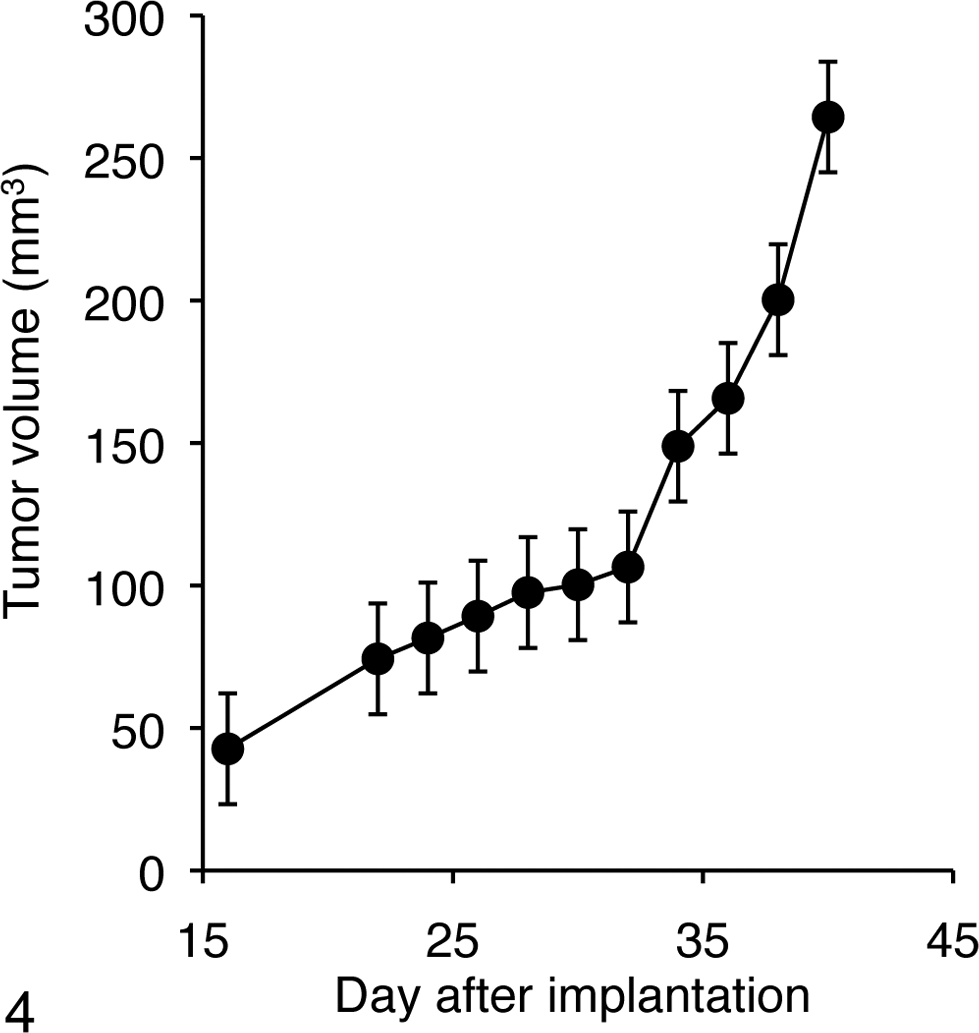
Tumor growth after inoculation with 106 CLAC cells in nude mice (mean ± SD).
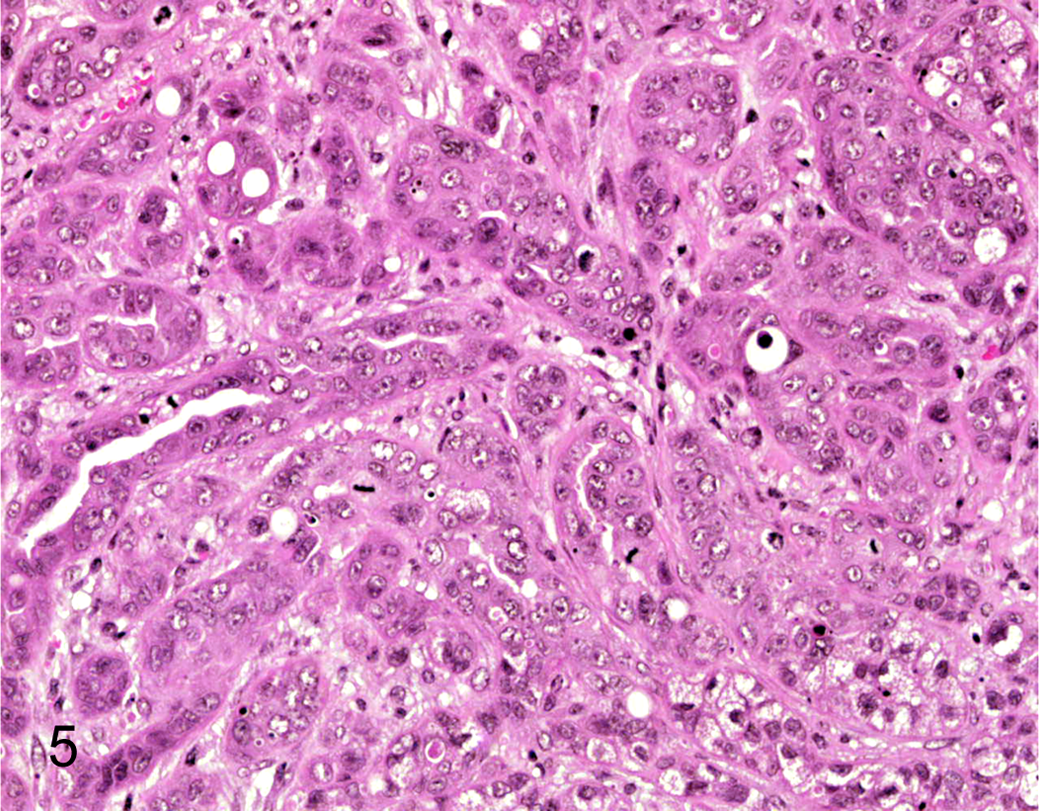
Histopathology of the xenografted tumor derived from bulk CLAC cells. HE.
Identification of SP Cells in CLAC
It is well known that a subpopulation (SP) of cells that effluxes the DNA-binding dye Hoechst 33342 out of the cell membrane through an ABC transporter was recognized as CSCs.5–8 To detect CSCs, we tried to identify the SP cells in CLAC. As shown in Figure 6, the CLAC cell line included 0.075% SP cells. The percentage of SP cells was markedly diminished after treatment with verapamil, an inhibitor of the pumps that exclude Hoechst dye. These results indicated that since the effluxes of Hoechst 33342 out of SP cells could be inhibited by verapamil, these cells possessed efflux activity with ABC transporter dependence, which therefore this population truly represented SP cells.
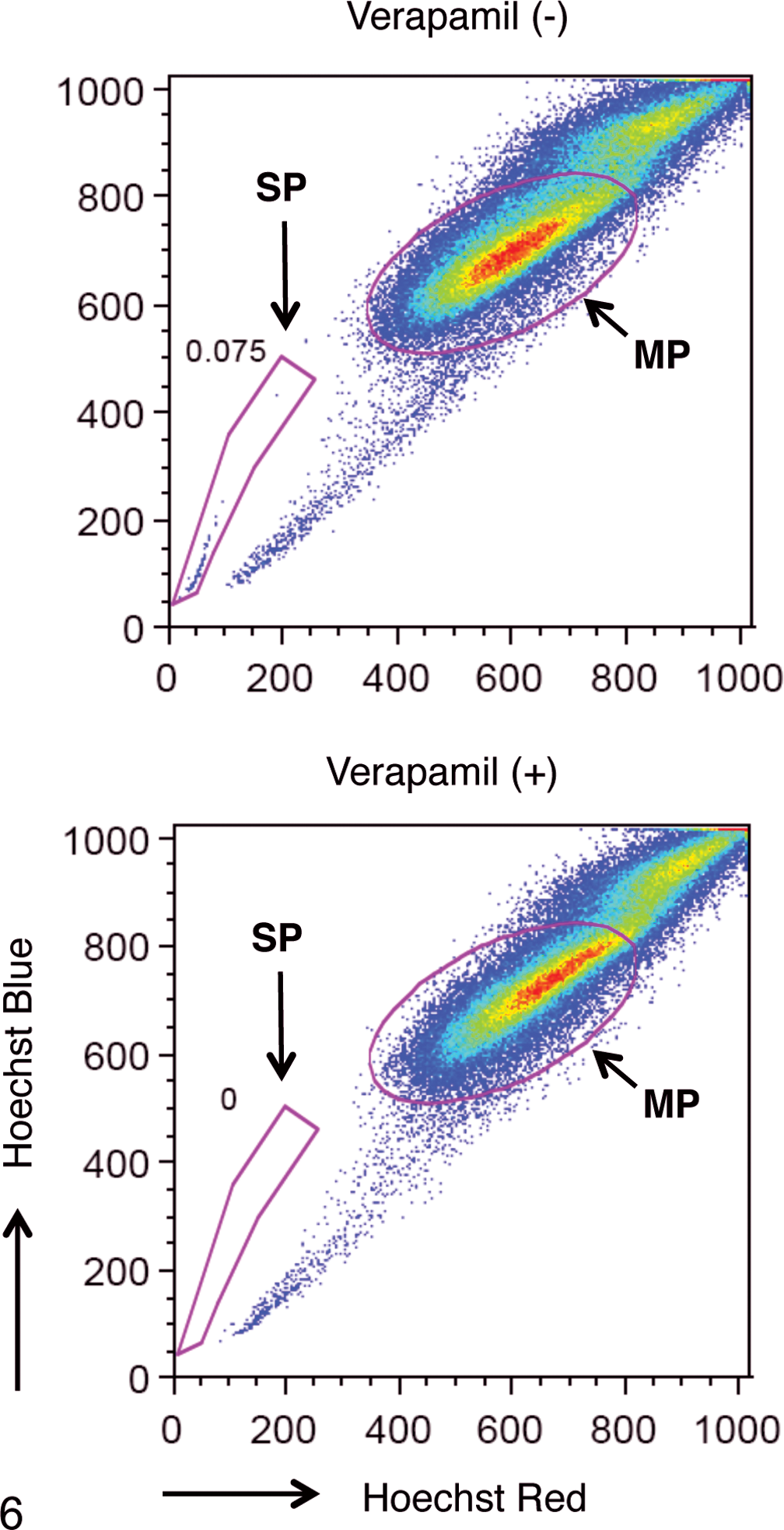
Detection of side population (SP) cells in CLAC. The populations of SP cells of CLAC in the presence (+) of or absence (–) of verapamil are shown. SP cells are marked by red dotted polygon. Main population (MP) marked by red oval.
In Vivo Growth Characteristics of CLAC SP and MP Cells
To address the in vivo tumorigenicity of CLAC SP and MP cells, 1 × 101 to 1 × 103 SP and MP cells sorted from CLAC were injected into nude mice. To rule out the effects of Hoechst toxicity, we routinely checked viability of the cells by staining with trypan blue after cell sorting. Almost all of the MP and SP cells were viable. Solid tumors were observed in 2 of 4 mice 8 weeks after they were injected with 1 × 102 SP cells and solid tumors were also observed in all 4 mice injected with 1 × 103 SP cells (Table 1). In addition, the tumors generated from 1 × 103 SP cells were larger in volume than the tumors generated from 1 × 102 SP cells. In contrast, tumors did not appear in any of the 4 mice at 8 weeks after they were injected with 1 × 102 MP cells, but tumors did appear in 2 of 4 mice injected with 1 × 103 MP cells. Hence, the CLAC SP cell population was significantly enriched with tumorigenic cells by ~10-fold compared with MP cells. The subcutaneous tumor derived from the SP and MP cells was diagnosed histopathologically as adenocarcinoma (Figs. 7, 8) and was nearly identical to that of the primary tumor. These data support the hypothesis that SP cells have a high cancer-initiating ability, similar to cancer stem-like cells.
Tumorigenesis of SP and MP Cells in Nude Mice a
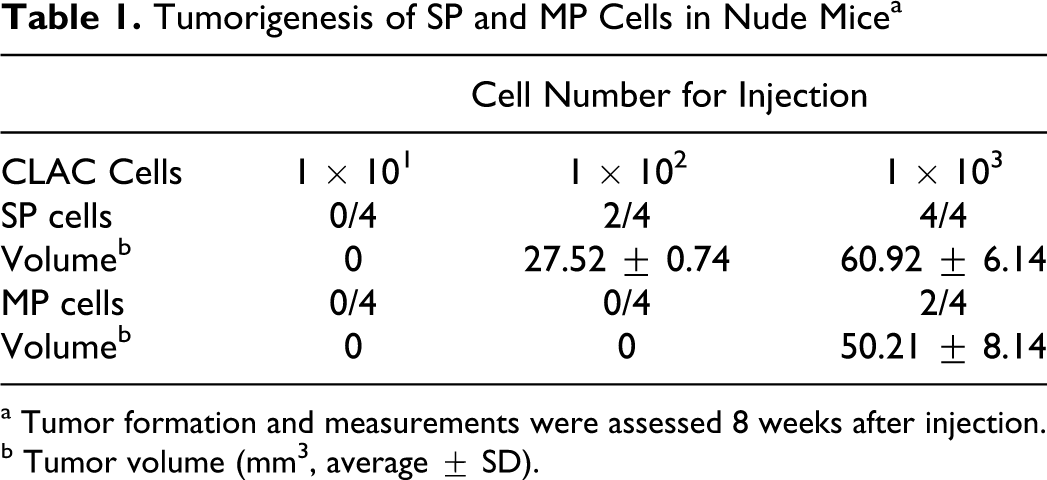
a Tumor formation and measurements were assessed 8 weeks after injection.
b Tumor volume (mm3, average ± SD).
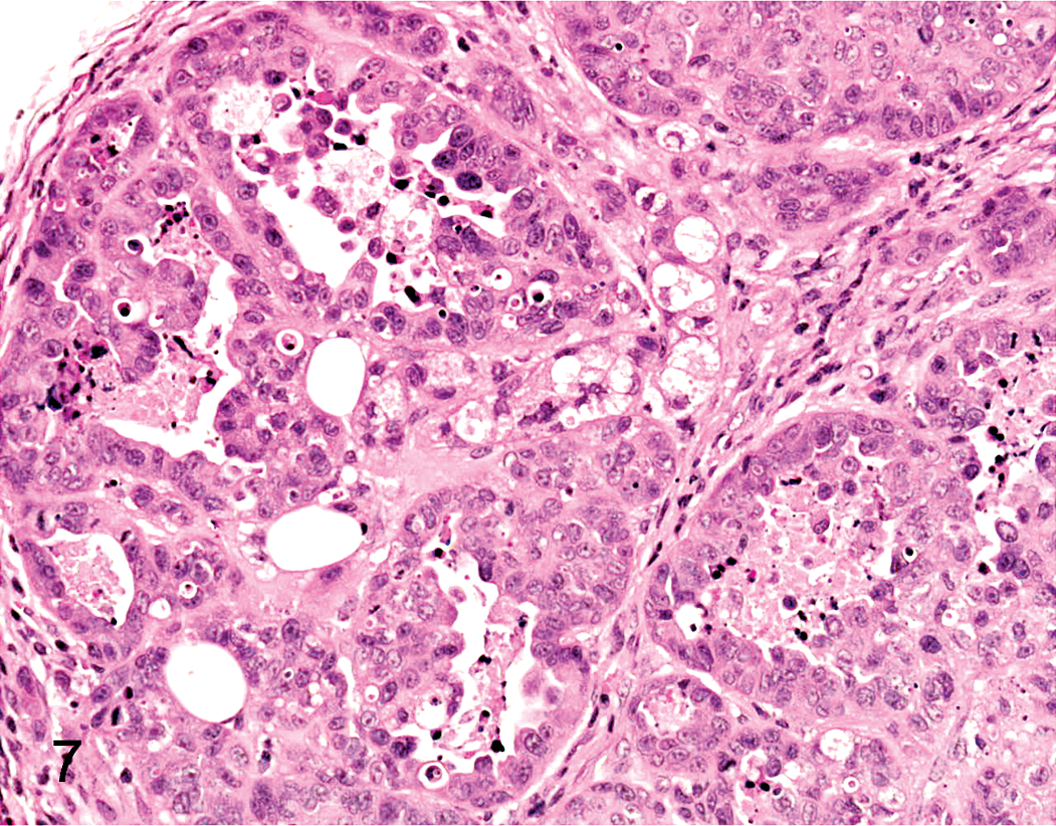
Histopathology of the xenografted tumor derived from SP cells in CLAC cells. HE.
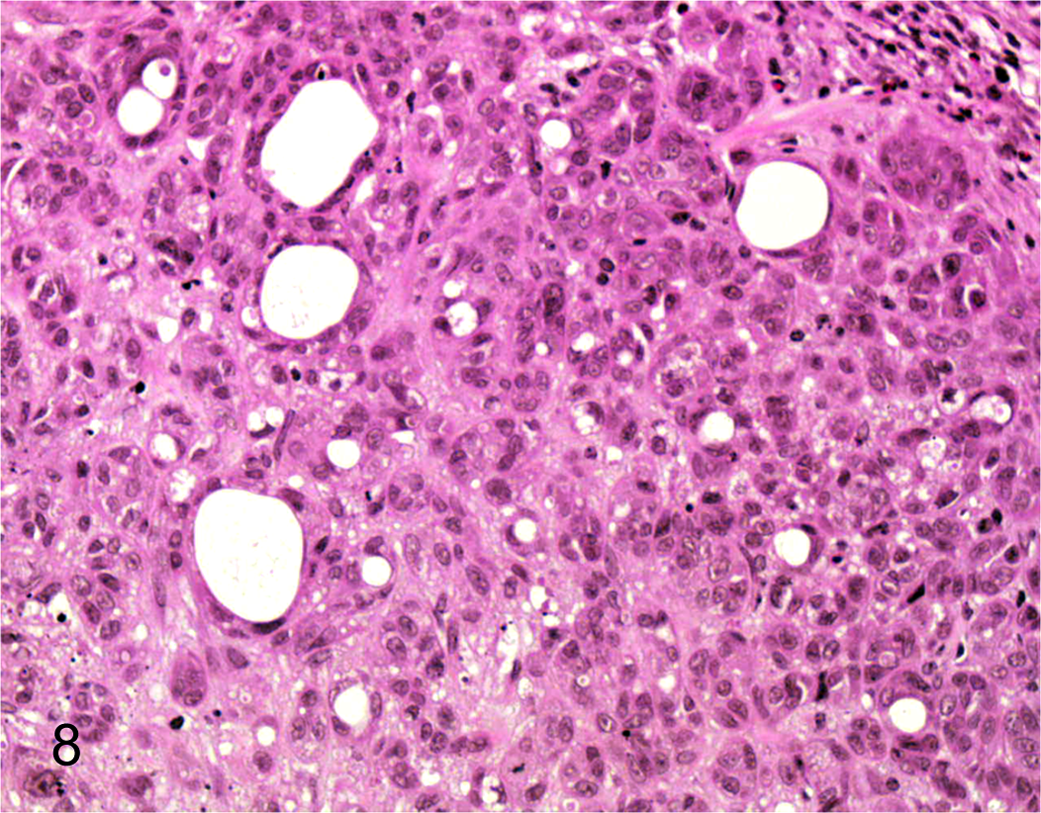
Histopathology of the xenografted tumor derived from MP cells in CLAC cells. HE.
Discussion
In the case of human lung cancer, many studies have established lung cancer cell lines as a useful model for cancer research. However, little is known about the establishment of lung cancer cell lines from canine primary lung adenocarcinoma. In this study, we established a lung adenocarcinoma cell line derived from canine lung cancer and injected the cells into a nude mouse xenograft model. We detected high (10-fold) tumorigenic potential of SP cells compared with MP cells in vivo. This suggests that a tumor hierarchy between SP and MP cell populations might exist in CLAC and that a subpopulation of CLAC SP cells has CSC-like properties. We observed 0.07% SP cells in CLAC, which is a smaller proportion than has previously been reported with tumor cell lines, with 1.5 to 6.1% noted in 6 human lung cancer cell lines, 6 0.75 to 7.77% in human bone sarcoma cell lines, and 19.1% in a mouse ovarian cancer cell line. 14 Primary cancer cells tend to have fewer SP cells, with 0.047 to 0.69% being reported in primary ascites obtained from ovarian cancer patients, 14 a proportion comparable to what we observed in our CLAC tumor cell line. The significance of the low number of SP cells in our cell line is unclear.
In this study, we detected an approximately 10-fold higher tumorigenic potential of SP cells than MP cells in vivo using a nude mouse xenograft model. SP cells from human lung adenocarcinoma cell lines A549, H441, and H460 have been reported to give rise to tumors with as little as 1 × 103, 5 × 103, and 1 × 104 cells, respectively. 6 In the present study, only 100 SP cells gave rise to new tumors, suggesting that these cells have the cancer-initiating ability of CSCs. Even though human CSCs have been identified using the Hoechst 33342 dye-based side population technique, little is known about canine CSCs in lung cancer.11,1 Several attempts to characterize CSCs using cell surface marker have been reported, in which CD1333,12,16,21 and Oct415,17 were reported to be candidates for CSC-specific markers. The expression of Oct4 has been reported in 83 canine neoplasms. 19 Thus, further studies that characterize CSCs in canine lung adenocarcinoma will contribute to the elucidation of the mechanisms of tumorigenesis including CSCs-specific molecular expression and to the establishment of novel therapeutic strategies.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This study was partially supported by a project grant (Creative Research Project, 2009) awarded by the Azabu University Research Services Division.
