Abstract
Invasive micropapillary carcinoma (IMC) is a variant of infiltrating ductal carcinoma of the breast associated with poor outcome. In this study, we report 16 carcinomas of the feline mammary gland displaying histologic features that correspond to IMC of the breast in women. The clinicopathologic findings, overall survival time, disease-free survival time, and nuclear DNA content of these cats were compared with 65 more common invasive mammary carcinomas (other feline mammary carcinoma [FMC]) of nonspecified type. IMC was associated with larger tumor size, higher histologic grade (P < .0001), deeper muscle invasion (P = .004), and more frequent lymphovascular invasion and nodal metastases (P = .009 and P = .001, respectively) than other FMCs. The aneuploid pattern was more frequent in IMC lesions. IMCs were also associated with lower survival rates. In summary, all cases of feline IMC were associated with clinicopathologic features of high biologic aggressiveness and should be classified as independent histologic types of FMC.
Keywords
Introduction
Carcinomas are the most common neoplasias of the feline mammary gland, representing 80–90% of cases. They have high biologic aggressiveness and poor prognosis 10, 13, 18 and are an important cause of mortality, especially in middle-age and elderly females. 11 Mammary carcinomas of female cats have several types of histologic patterns, a mixture of which is found in the same tumor. The World Health Organization (WHO) 13 classification of feline mammary carcinomas (FMCs) is a descriptive histologic classification that is difficult to match due to the high number of patterns present in one lesion. 10 Most studies have not found associations between histologic type and prognosis. 2, 4, 10 In most cases, looking for the histologic patterns may be futile. However, there are some cases for which unique histology does indicate a more or less aggressive behavior. The micropapillary invasive pattern is a distinct histologic pattern that we observed in 16 female cat tumors. In addition to histology, they also presented with DNA content, behavior, and survival rates that justify the independent classification of invasive micropapillary carcinoma (IMC).
Materials and Methods
Eighty-one feline IMCs surgically resected from 73 queens and observed in the Histopathology Laboratory of Trás-os-Montes e Alto Douro University were selected for this study. Tissues were fixed in 10% buffered formalin and embedded in paraffin. Three-micrometer–thick sections were stained with hematoxylin and eosin (HE). All cases were reviewed and reclassified independently by two pathologists to detect micropapillary growth patterns without knowledge of the clinical outcome. We defined as IMCs all carcinomas with more than 50% infiltrating micropapillary pattern. All cases were graded according to the Elston and Ellis 6 scoring system and staged according to the WHO clinical staging system TNM for feline mammary tumors. 18 To confirm the presence of myoepithelial cells and to evaluate the tumor growth fraction, additional tumor sections were immunostained with a panel of antibodies using the streptavidin-biotin-peroxidase method. The markers used were cytokeratin (CK) 14 (1:20; NCL-LL 002; Novocastra Laboratories Ltd., Newcastle Upon Tyne, UK), p63 (1:150; 4A4; Neomarkers, Fremont, CA), calponin (1:400; CALP, Dako Corp, Carpinteria, CA), and antigen Ki-67 (1:100; Mib-1; Dako Corp). Immunohistochemical data were evaluated as positive (+) or negative (−) for p63 (nuclear) and CK14 and calponin (cytoplasmic positivity). Mitotic and Ki-67 indices were assessed in 8 to 10 representative areas at the periphery of the tumors, in the most mitotically active areas, or areas that presented the highest Mib-1 positivity at high-power magnification (40×). The numbers of immunopositive cells per 1,000 cells examined were expressed as percentages.
Automated image analysis was performed on 6-μm sections cut from the paraffin block used for histologic analysis. Slides were stained with the Feulgen DNA staining kit (Cell Analysis Systems, Elmhurst, IL), and analysis was performed using a CAS 200 image analyzer (Becton-Dickinson, San Jose, CA). In each lesion, a minimum of 100 nonoverlapping and well-preserved tumor nuclei and 20 to 30 normal lymphocytes (used as internal reference diploid cells) were measured. The resultant DNA histograms were analyzed and classified as diploid or aneuploid according to previously described methods. 14 The 5c exceeding rate (5cER), defined as the percentage of tumor nuclei with DNA content above 5n, was also evaluated. 20
Statistical analysis was performed using SPSS software (version 11.5; SPSS Inc., Chicago, IL). The numeric parameters were tested by analysis of variance, Kruskal-Wallis, or Mann-Whitney tests. Other clinicopathologic factors, including nuclear or histologic grade, were evaluated by a κ2 or Fisher test (2-sided). The follow-up was performed by the referring surgeons for a 2-year period. Overall survival time was defined as the period between surgery and death due to cancer. Queens that died of other causes were censored at the time of death. Disease-free survival (DFS) was defined as the period of time between surgery and recurrent or metastatic disease. Survival rate was calculated by the Kaplan-Meier method, and statistical significance was examined using log-rank test. P < .05 was considered statistically significant.
Results
The micropapillary pattern, a common architectural pattern in feline mammary carcinomas, was observed in 70 of 81 (86.4%) analyzed invasive carcinomas. With respect to the established proportion of the micropapillary component, 16 (19.7%) carcinomas were classified as IMCs because more than 50% of these tumors had an infiltrating micropapillary pattern.
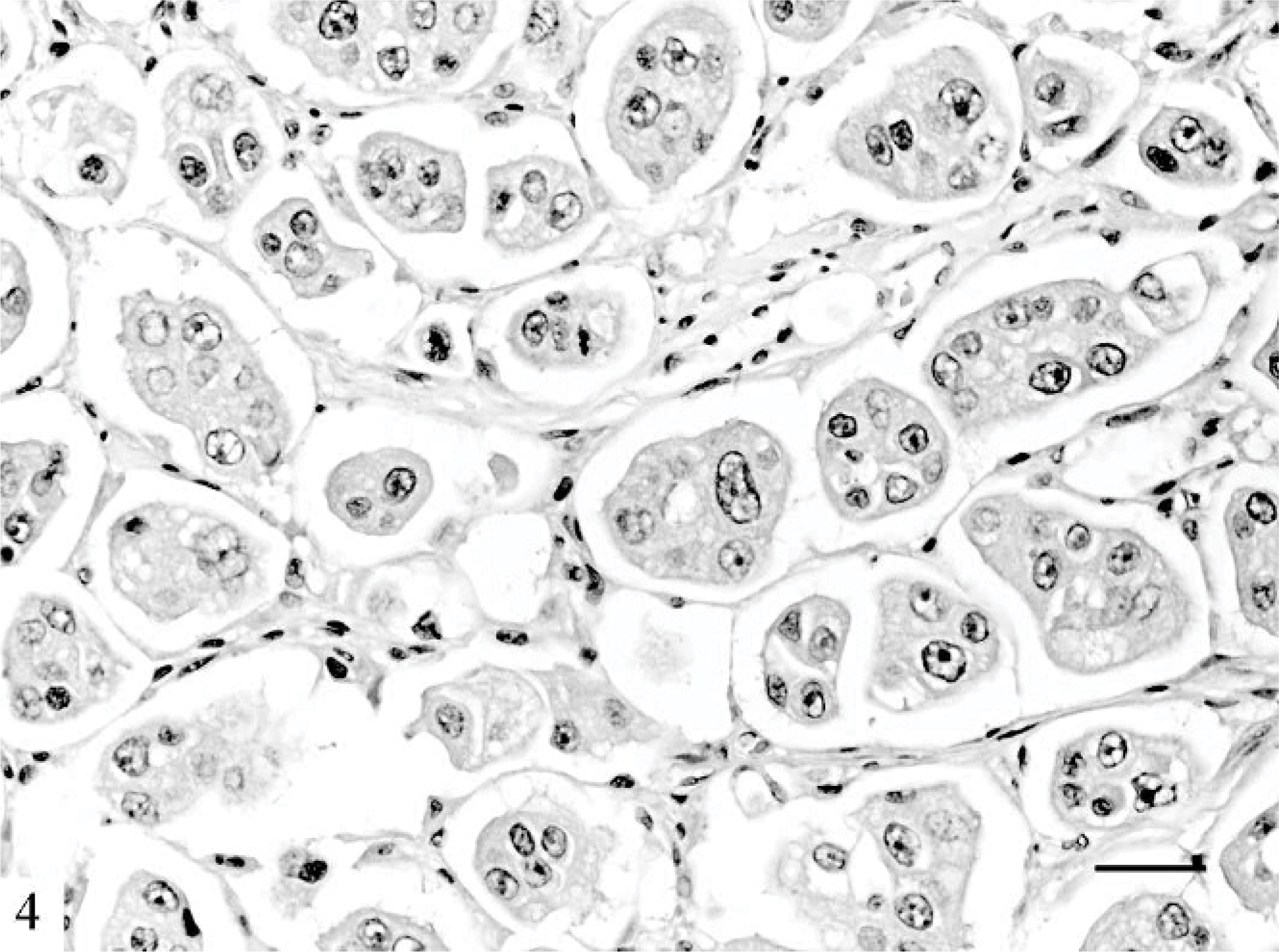
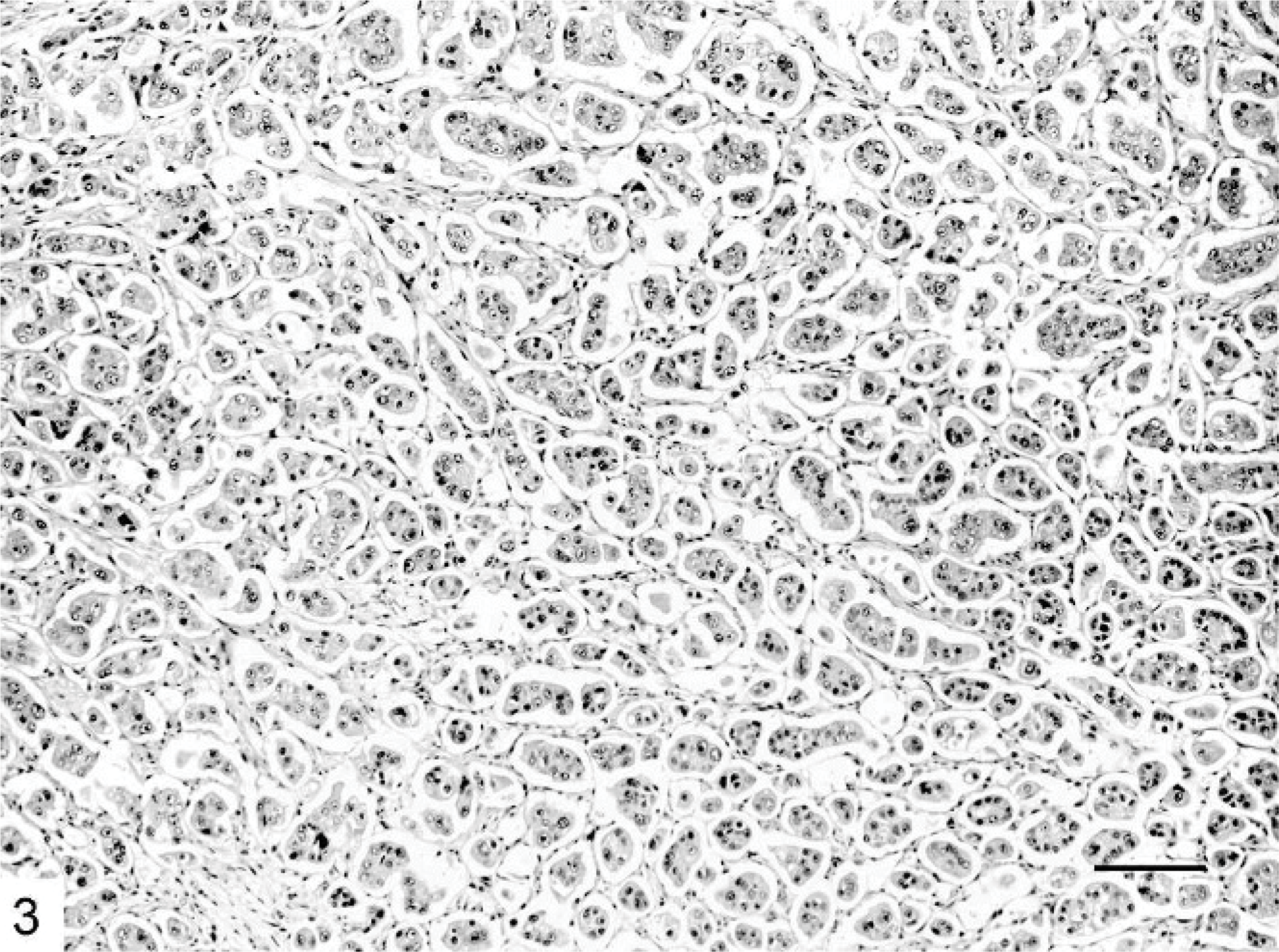
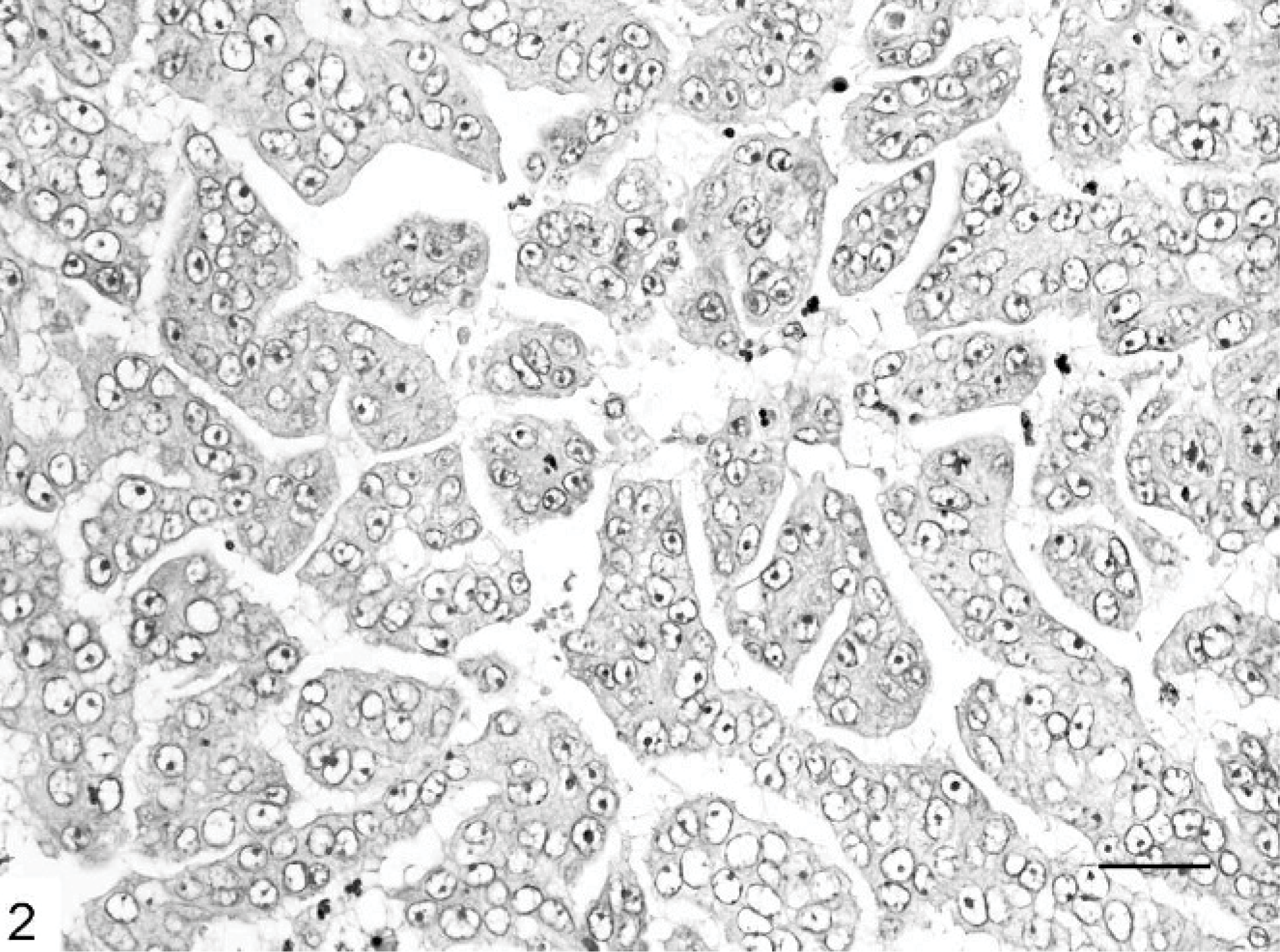
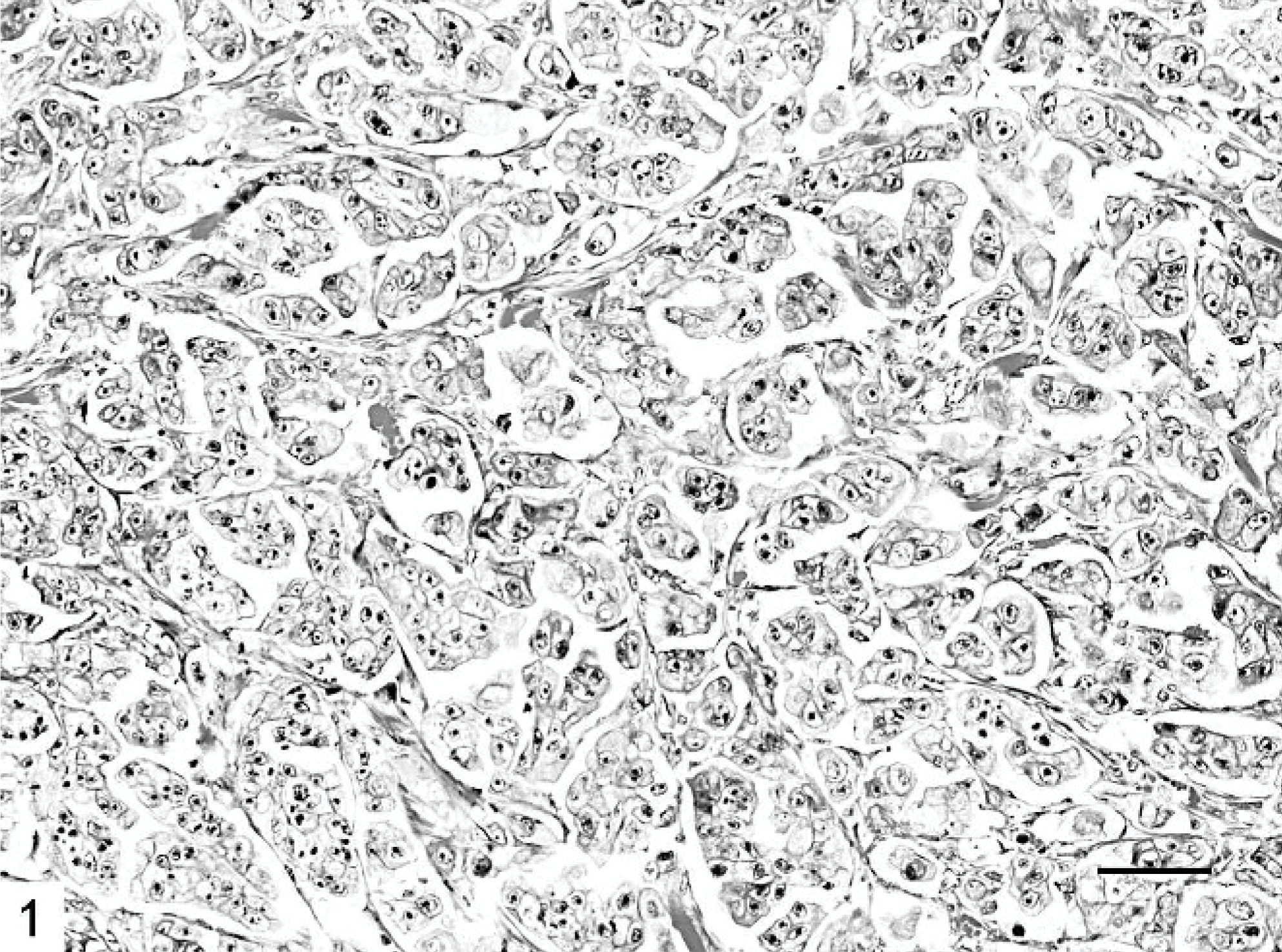
Microscopically, IMC lesions were mainly composed of atypical cells arranged in small tufts and avascular papillae devoid of fibrovascular cores (Figs. 1, 2) or in cell clusters floating in clear empty spaces, mimicking extensive lymphatic invasion (Fig. 3). Tumor cells, cuboidal or polygonal in shape, had eosinophilic or amphophilic abundant cytoplasm and vesicular pleomorphic nuclei with prominent nucleoli. The cell clusters in the lacunar spaces had solid or tubular configurations and were arranged so that the polarities of the cells were reversed, with abundant cytoplasm facing the clear spaces on high-power magnification (Fig. 4). These spaces were not lined by endothelial cells. In most cases, stroma was fine reticular fibrocollagenous and lacked desmoplasia.
Mammary gland; cat. The cell clusters are arranged so that the polarity of the cells is reversed. HE. Bar = 30 μm.
Mammary gland; cat. Micropapillary carcinoma showing neoplastic cell clusters floating in a spongy loose stroma. HE. Bar = 120 μm.
Mammary gland; cat. Most papillae are abortive and lack true fibrovascular cores. HE. Bar = 30 μm.
Mammary gland; cat. Micropapillary carcinoma composed of small papillary clusters lined by polygonal cells showing high-grade nuclei. HE. Bar = 60 μm.
Tumors with pure micropapillary patterns were fairly unusual (1/16), and more frequently (15/16) the infiltrating micropapillary areas were seen in association with solid areas and small papillary areas. Micropapillary carcinoma cells massively invaded the thoracic or abdominal muscle (12/16) and lymphatic vessels (16/16). The micropapillary pattern was preserved in the emboli. Myoepithelial cells were not identifiable in this tumor type using HE-stained slides or basal/myoepithelial markers.
The clinicopathologic features of IMCs and other FMCs are summarized in Tables 1 and 2. The DNA ploidy pattern was determined in 7 IMCs and 65 other FMCs (Table 1). There were no significant differences between IMCs and other FMCs with respect to the queens' ages. The ages for IMCs and other FMCs ranged from 7 to 15 years (mean age, 11.6 ± 2.7 years) and from 6 to 19 years (mean age, 11.6 ± 3.1 years), respectively. IMC cases were associated with larger tumor sizes, although this difference did not achieve statistical significance. IMC lesions varied from 1 to 7 cm, whereas other carcinomas varied from 0.55 to 5.5 cm. IMCs were associated with higher nuclear grade (P = .001) and histologic grade (P < .0001) than other more common invasive carcinomas of nonspecified type. All IMC cases were high-grade carcinomas (grade III), whereas 3 (4.6%) other FMCs were classified as grade I, 36 (55.4%) as grade II, and 26 (40.0%) as grade III. Micropapillary carcinomas were mostly high-stage lesions, although this difference did not achieve statistical significance.
Clinicopathologic features and nuclear DNA content of feline mammary IMCs compared with other FMCs of nonspecified type.
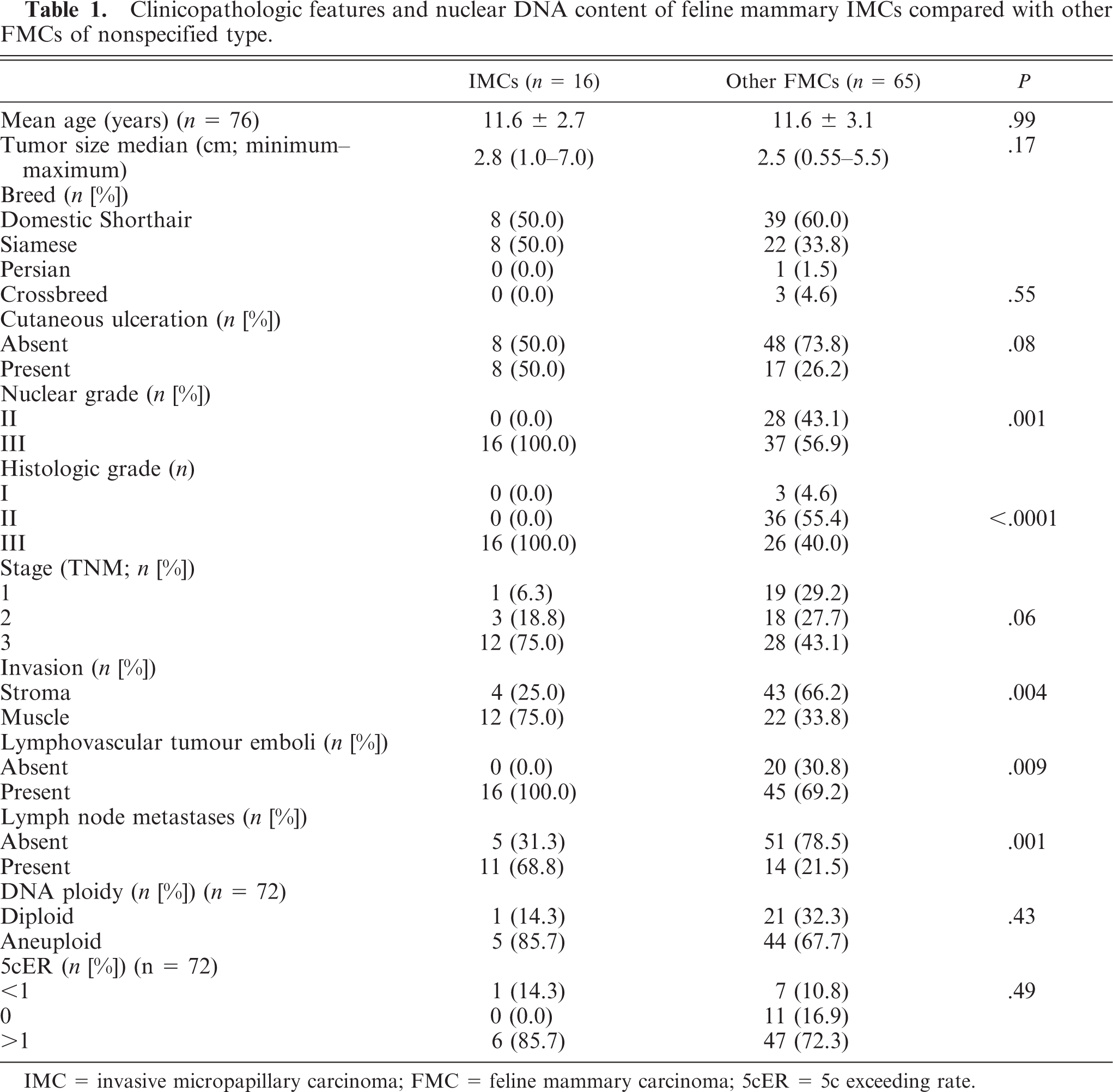
IMC = invasive micropapillary carcinoma; FMC = feline mammary carcinoma; 5cER = 5c exceeding rate.
Comparison of mitotic and Ki-67 rates between IMCs and other FMCs.

IMC = invasive micropapillary carcinoma; FMC = feline mammary carcinoma.
Corrected survival rate in cats with available follow-up data.∗
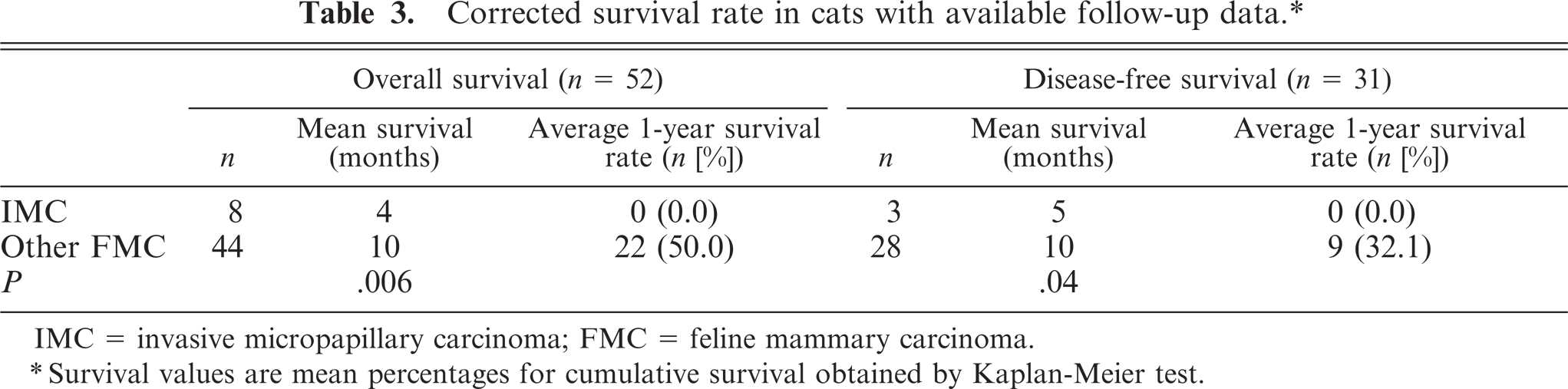
IMC = invasive micropapillary carcinoma; FMC = feline mammary carcinoma.
∗Survival values are mean percentages for cumulative survival obtained by Kaplan-Meier test.
Deep-muscle invasion (P = .004), lymphovascular tumor emboli (P = .009), and lymph node metastases (P = .001) were more common in IMCs. Metastases showed the same arrangement found in the primary tumors (Fig. 5): a pure micropapillary or mixed solid micropapillary pattern. No significant differences were observed in mitotic and Ki-67 labeling indices between the 2 tumor groups featured in the study. Carcinomas in situ with pure micropapillary or micropapillary and solid cribriform patterns were observed in 10 IMCs (62.5%) and in 20 (30.8%) other FMCs.
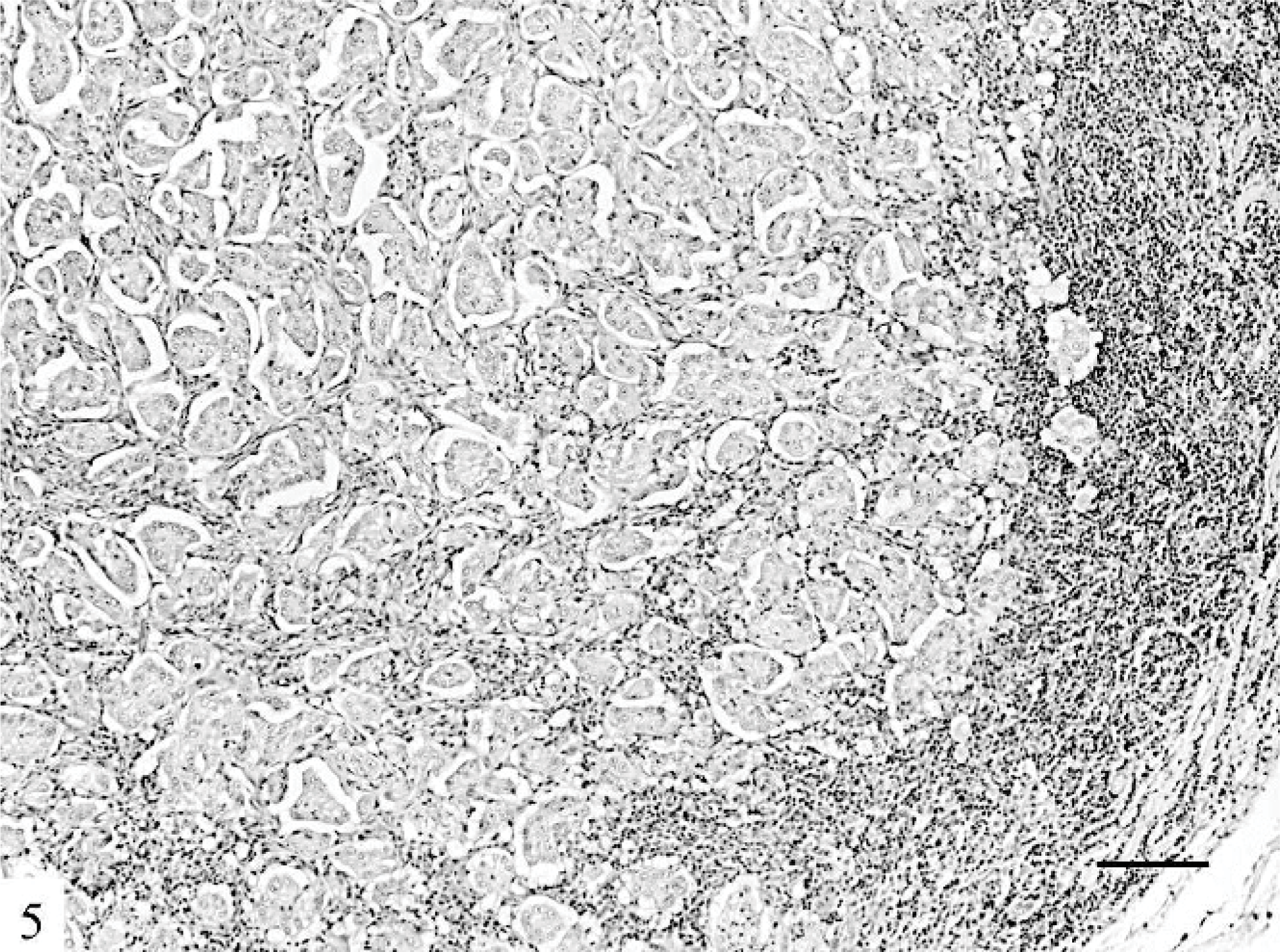
Lymph node; cat. The lymph node metastases show micropapillary patterns. HE. Bar = 120 μm.
With regard to DNA image analysis, no significant differences between IMCs and other FMCs were observed. However, the aneuploid pattern frequency and the percentage of true aneuploid cells with DNA content higher than 5c were higher in the IMC group.
Follow-up data is summarized in Table 3. Overall survival data was available in 8 queens diagnosed with IMC and 44 queens diagnosed with other FMCs of nonspecified type. Kaplan-Meier survival curves revealed that queens with nonmicropapillary tumors had better survival rates than those with IMCs. All queens presenting IMC lesions died or were euthanized due to oncologic disease in a 9-month period after surgery. The median overall survival was 4 months for IMCs, whereas it was 10 months for queens presenting with other FMCs (P = .006; Fig. 6).
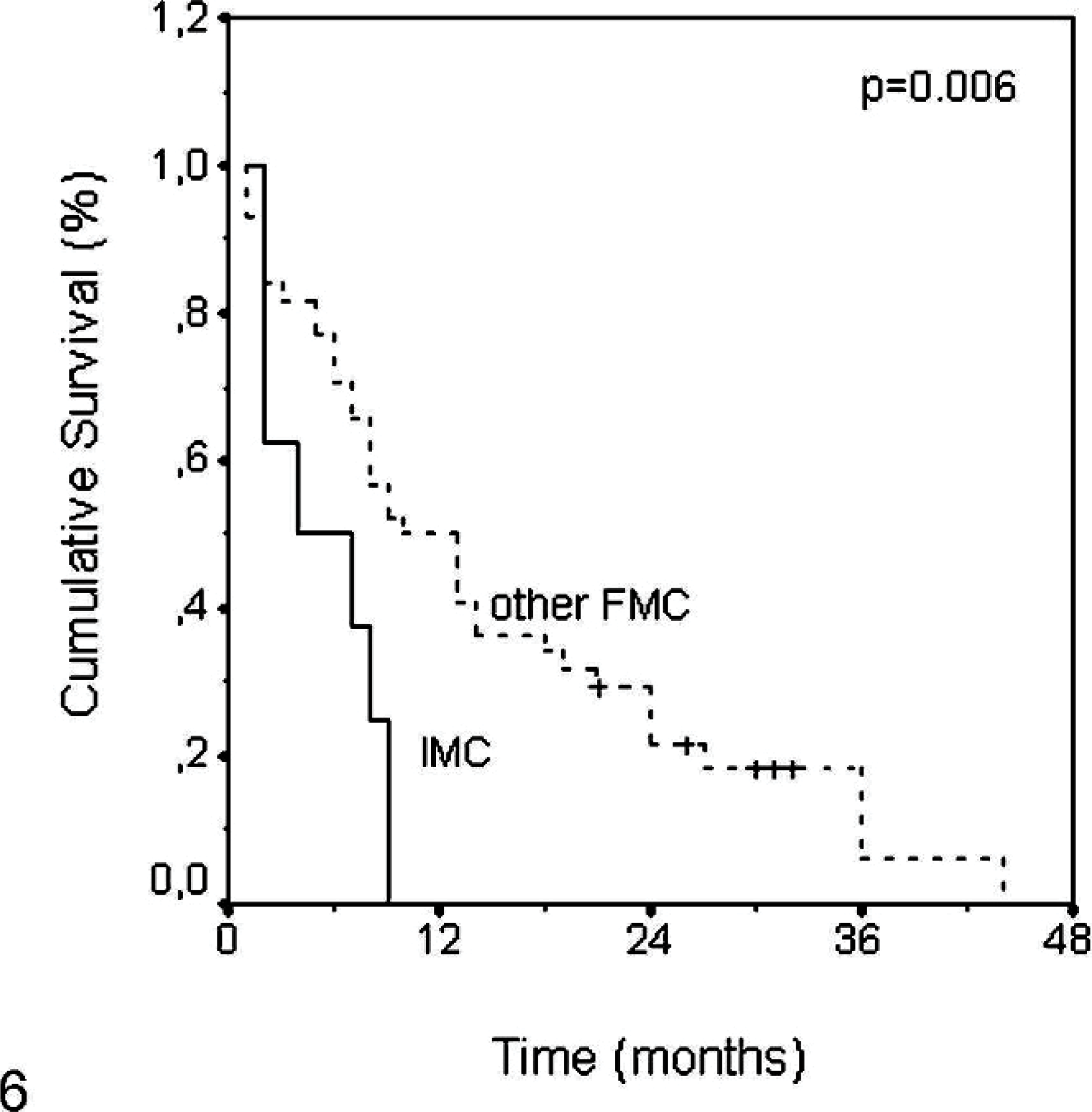
Comparison of overall survival time between invasive micropapillary carcinoma and other more common feline mammary carcinomas.
Data concerning DFS were available in 7 queens presenting with IMCs: 4 (57.1%) queens were censored from the study because they were not free of oncologic disease after mastectomy. All of the other 3 queens developed disease within a 5-month period. On the other hand, just 2 of 30 queens presenting with other FMCs showed signs of metastatic disease shortly after surgery and were censored from analysis. The median DFS in this group was 10 months (P = .04; Fig. 7).
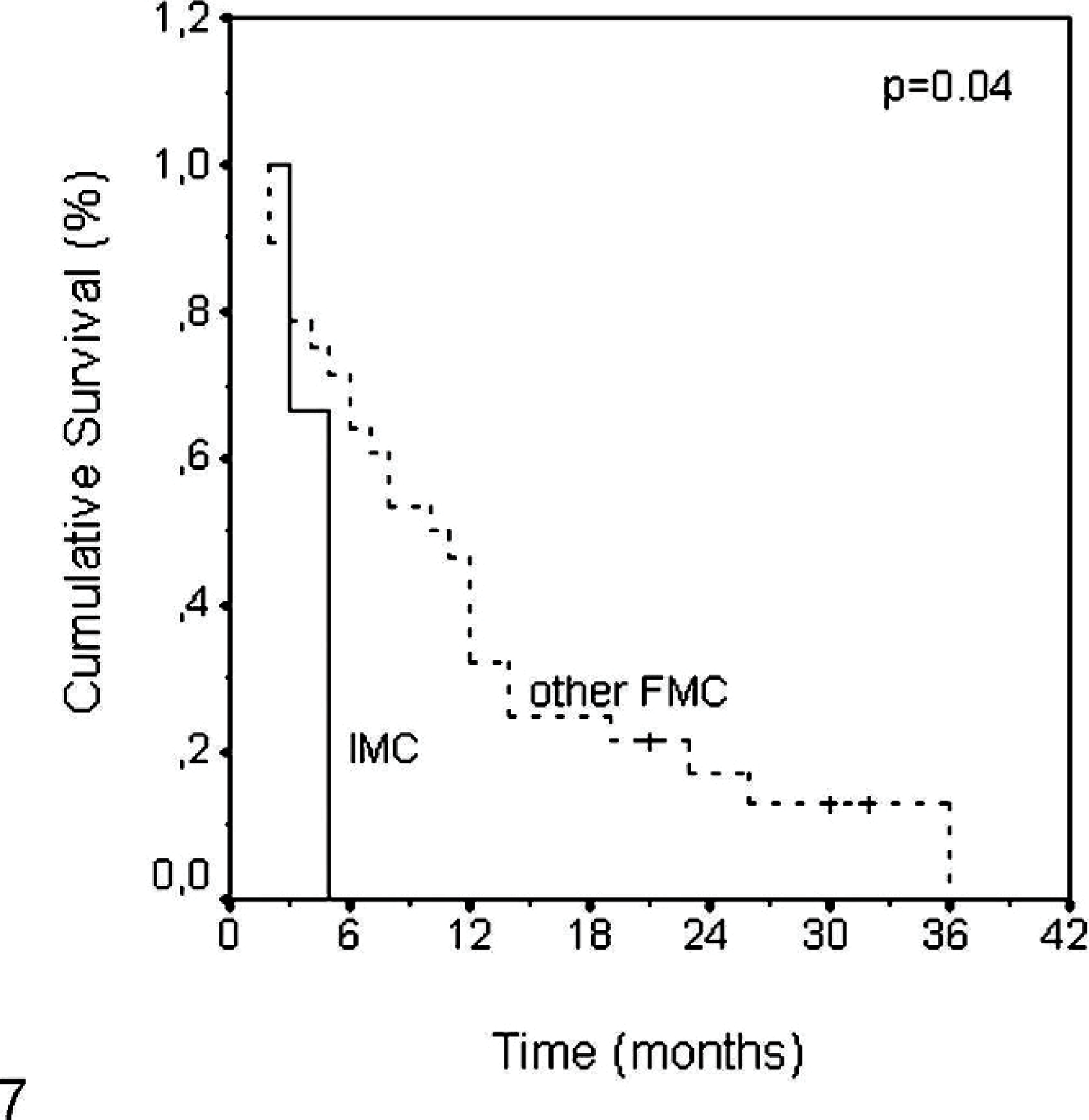
Comparison of disease-free survival between invasive micropapillary carcinoma and other mammary carcinomas.
Discussion
Mammary neoplasias are the third most frequent neoplasias in cats, accounting for 12% of all feline tumors and for 17% of all neoplasias of queens. 12, 18 Although the WHO classification of FMCs is based on morphologic criteria, 13 it does not recognize the micropapillary pattern, which is included in the spectrum of papillary tumors. However, the presence of papillae devoid of stromal fibrovascular cores was first described in FMCs by Carpenter et al., 2 and there is a report of 2 cases in canine mammary glands. 3
IMCs are defined as carcinomas composed of small papillary structures without stroma or small clusters of tumor cells lying within an artifactually created clear space simulating vascular channels. 3, 7, 19, 22, 23,24 IMC was first reported in women's breasts as a distinctive variant of infiltrating ductal carcinoma. Subsequent studies have demonstrated that this tumor, with its unique histologic features, has a high incidence of lymphatic invasion and lymph node metastases on clinical presentation, resulting in poor outcome. 19 Recently in humans, carcinomas demonstrating histologic features similar to IMC of the breast have been reported to occur in various organs, including urinary bladder, lung, 7, 16, 19 ureter, salivary gland, colon, 19 and ovary. 7, 16 All cases were associated with poor prognosis. 7, 16, 19
All the cases reported in this series have similar morphologic features to cases observed in women's breast tumors. 7, 8, 15, 16, 23, 24 They were characterized by small micropapillary structures or cell aggregates floating in clear spaces, mimicking vascular invasion. These feline IMCs were associated with larger tumor sizes and with higher histologic grade and stage than nonmicropapillary carcinomas; our data also confirmed the lymphotropic nature of IMCs. Nodal involvement is a significant feature of this tumor, and in this series, 10/17 (58.8%) IMC tumors were associated with lymph node metastases, whereas just 14/65 (21.5%) other FMCs presented lymph node metastases. All cases reported here had aggressive clinical behavior and shortened overall survival times than nonmicropapillary invasive tumors. DFS was also low.
The association among aneuploidy, 5cER, and IMC also agrees with reports that associate abnormal DNA content 5, 17, 21 and 5cER with increased biologic aggressiveness. 1, 9, 20, 21
As reported above, the micropapillary pattern is frequently observed in FMCs; foci of the micropapillary pattern, less than 50% of the tumor area, were detected in 53/65 (81.5%) other FMCs. Because this tumor type is an aggressive variant of carcinoma irrespective of the extent of IMC 7, 16 and its site of origin, 23 the poor prognosis classically associated with feline carcinomas may be linked to this growth pattern. Although speculative, this fact deserves more investigation.
In summary, this report described some clinical and pathologic features of 16 cats with histologic features that correspond to IMCs of the mammary gland in women. To the best of our knowledge, this is the first report describing the clinicopathologic features of this tumor type in feline mammary glands.
Conclusion
All cases of feline IMC of the mammary gland herein reported were associated with clinicopathologic features of high biologic aggressiveness and low survival rate and should be classified as an independent histologic type of FMC. These results also suggest that FMC, on the basis of histologic and clinical outcome, is a good animal model to study women's breast cancer, especially IMC.
Footnotes
Acknowledgements
We thank Mrs. Lígia Bento for excellent technical assistance. This work was supported by a PhD grant from the EU/Fundo Social Europeu, reference 2/5.3/PRODEP/2001.
