Abstract
An invasive micropapillary carcinoma (IMC) occurred in the buccal skin of an 18-year-old female cat. Histologically, the tumor had a honeycomb pattern characterized by clusters of neoplastic epithelial cells that were surrounded by empty clear spaces and lined with fibrocollagenous stroma. On immunohistochemistry, the neoplastic cells were positive for cytokeratin (clone CAM5.2; pancytokeratin, clone AE1/AE3) and carcinoembryonic antigen (CEA) but negative for cytokeratin 14, vimentin, S100, smooth muscle actin, and p63. The CEA-positive staining reaction was present along the outermost rim of the neoplastic cell clusters consistent with an “inside-out” immunoreactivity pattern. Examination of the tumor cells by electron microscopy revealed microvilli on the outermost rim of neoplastic cells that were directed toward the surrounding vacant space. Based on histomorphological characteristics, the neoplasm was defined as an IMC of “pure-type.” The location site and immunohistochemical features suggest the tumor was most likely derived from the apocrine sweat glands in the buccal skin.
Invasive micropapillary carcinoma (IMC) is a distinctive histologic variant of epithelial malignancy, which has been reported in human beings, and less frequently in veterinary medicine. In human beings, IMC was described initially in 1993 as a histologic variant of breast cancer, 12 and it was listed for the first time in the World Health Organization classification of breast tumors in 2003, where it is postulated to be a distinctive variant of infiltrating ductal carcinoma. 3 In human medicine, in 2004, IMC was reported in other sites, originating from the urinary bladder, lung, ureter, salivary gland, colon, and ovary. 8 In veterinary medicine, IMC has been reported in the mammary glands of cats and dogs. 1,4,10,11
Invasive micropapillary carcinomas are composed of small clusters of tumor cells lacking a fibrovascular core sitting within a clear space and supported by fine fibrous stroma. These small neoplastic clusters also sometimes form small pseudotubular structures in areas as well. Neoplastic cell clusters lie within empty spaces that are considered to be a retraction artifact caused by fixation because they are not seen in frozen sections and may reflect a phenotypic change within tumor cells. 14 Immunohistochemical staining with markers targeting epithelial membrane antigens such as epithelial membrane antigen (EMA) and carcinoembryonic antigen (CEA) often reveals linear, membranous staining along the external edges of clusters of tumor cells. In normal glands, these markers often line the apical or secretary edges of epithelial cells, thus this staining pattern is often referred to as an “inside-out” growth pattern. 6,14 In addition to its unique histological features, IMCs generally have a high incidence of lymphatic invasion and lymph node metastasis, which results in a poor prognosis. 6 This feline IMC was likely of cutaneous apocrine sweat gland origin based on immunohistochemical staining.
An 18-year-old, female, Japanese domestic cat was admitted to Apple Animal Hospital (Aomori City, Japan) for evaluation of a cutaneous mass located in the right cheek. The animal had no history of mammary tumors and apart from the buccal mass, no other abnormalities were observed on physical examination, complete hematology, and blood biochemistry. The mass, which had grown rapidly to approximately 2.1 cm × 1.8 cm × 0.6 cm during the month prior to admission, was surgically removed at the owner’s request. The cut surface of the neoplasm was gray–white in color. The patient died 16 months after surgery but unfortunately, a postmortem examination was not performed.
The excised mass was fixed in 10% neutral buffered formalin, dehydrated by alcohol, and embedded in paraffin wax. Three-micron serial sections were mounted on glass slides and separately stained with hematoxylin and eosin, alcian blue (AB) at pH 2.5, mucicarmine, and periodic acid–Schiff (PAS) reaction. The primary antibodies used in immunohistochemical staining were as follows: low molecular weight cytokeratin (clone CAM5.2 a ; prediluted), pancytokeratin (clone AE1/AE3 b ; dilution 200×), cytokeratin 14 (clone LL002 c ; dilution 50×), vimentin (clone V9 b ; dilution 100×), carcinoembryonic antigen (CEA; rabbit polyclonal b ; dilution 300×), α-smooth muscle actin (α-SMA; clone 1A4 b ; dilution 400×), S100 protein (rabbit polyclonal b ; dilution 1,500×), p63 (clone 4A4 d ; dilution 200×), von Willebrand factor (rabbit polyclonal b ; prediluted), and Ki-67 (clone MIB-1 b ; dilution 100×). After the reaction with the specific primary antibodies, the sections were incubated with biotinylated goat anti-mouse immunoglobulin (Ig)G or anti-rabbit IgG antibodies, b followed by peroxidase-conjugated streptavidin. b Finally, the reaction with each antigen was visualized following addition of diaminobenzidine tetrahydrochloride (DAB) and the slides counterstained with hematoxylin. Because the tumor was located in the dermis, the adjacent normal epidermis and adnexa served as internal controls. The Ki-67 indices were assessed by counting both Ki-67–positive and Ki-67–negative tumor cells in 10 fields at 400× magnification selected randomly, and expressed as a percentage. For electron microscopic examination, small pieces of the formalin-fixed neoplastic tissue were refixed in 1% osmium tetroxide in 0.2 M phosphate buffer, and embedded in resin. Using an electron microscope, ultrathin sections were examined after staining with uranyl acetate and lead citrate.
Histologically, the tumor was nonencapsulated, locally invasive, and expanded and effaced the dermis (Fig. 1A). The tumor was composed of many cell clusters that were unassociated with fibrovascular cores. The neoplastic cell clusters were surrounded by empty, clear spaces that were lined with fibrocollagenous stroma and some spindle-shaped stromal cells, altogether giving an impression of a honeycomb (Fig. 1B). The neoplastic cell clusters presented an image of micropapillae within clear spaces, with occasional pseudotubules caused by depletion of necrotic central cells. The neoplastic cells had round, ovoid, or constricted hyperchromatic nuclei, and eosinophilic cytoplasm (Fig. 1C). Mitotic figures were found at the rate of 2–3 per 10 fields (at 400× magnification). At deep edges of the tumor, neoplastic infiltrates invaded the cutaneous muscle (Fig. 1D) and were noted within adjacent lymphatic vessels (Fig. 1E). Staining with AB, mucicarmine, and PAS did not demonstrate any mucin within the clear spaces, nor within the cytoplasm of neoplastic cells.
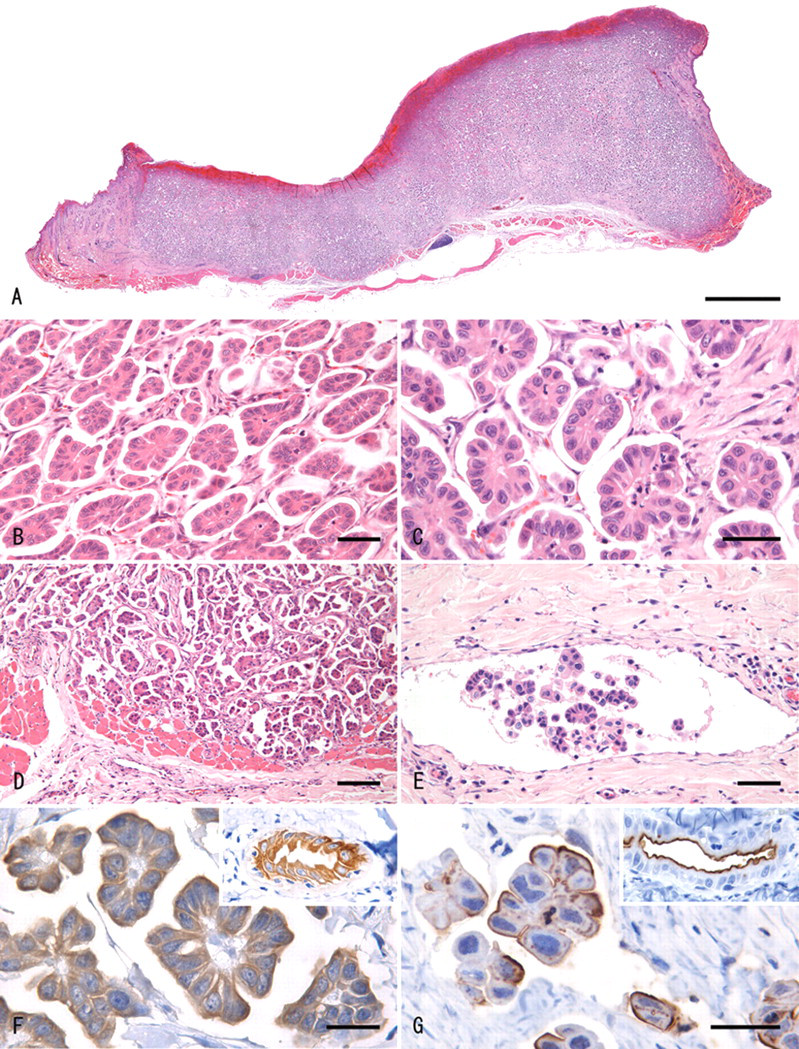
Haired skin (dermis); cat. A, the tumor effaced a focally extensive area of the dermis. Hematoxylin and eosin (HE). Bar = 2 mm. B, low-power view of the cutaneous neoplasm showing micropapillary small cell clusters surrounded by clear spaces. The latter were lined with delicate strands of fibrocollagenous stroma giving a honeycomb impression. HE. Bar = 50 µm. C, high-power view of the neoplasm in which each tumor cell cluster consists of cuboidal to columnar cells characterized by eosinophilic staining cytoplasm and vesicular nuclei. The papillary tumor clusters do not include a fibrovascular stalk. HE. Bar = 50 µm. D, tumor cells invading the underlying skeletal muscle. HE. Bar = 100 µm. E, lymphatic invasion by the neoplastic cells at the tumor periphery. HE. Bar = 50 µm. F, a positive immunoreaction for cytokeratin AE1/AE3 is present throughout the cytoplasm of the neoplastic cells. Inset: normal apocrine sweat gland shows a similar positive reaction. Cytokeratin AE1/AE3 immunohistochemistry with hematoxylin counter stain. Bar = 50 µm. G, strong linear positivity for carcinoembryonic antigen (CEA) is present at the periphery of the neoplastic cells facing the clear space. Inset: CEA positivity in a linear pattern is seen at the inner surface of normal apocrine grand. CEA immunohistochemistry with hematoxylin counter stain. Bar = 25 µm.
Immunohistochemically, the neoplastic cells were positive for cytokeratin CAM5.2, cytokeratin AE1/AE3 (Fig. 1F), and CEA, and were negative for cytokeratin 14, S100, α-SMA, p63, and vimentin. Cells that were immunoreactive for CEA formed rims external to the clusters of neoplastic cells (Fig. 1G). Spindle-shaped stromal cells were positive for vimentin and α-SMA, but negative for p63. Nonneoplastic apocrine sweat gland cells located in the area adjacent to the tumor were positive for cytokeratin CAM5.2, cytokeratin AE1/AE3, and CEA. Linear, membranous deposition of CEA was present along the luminal surface of epithelial cells lining adjacent apocrine sweat glands (Fig. 1G; inset). Myoepithelial cells were absent from the neoplastic cell clusters, as shown by negative immunostaining with cytokeratin 14, S100, α-SMA, and p63. There were no lining cells positive for von Willebrand factor at the outermost rim of clear spaces. The Ki-67 index of the neoplastic cells was 27.0%.
Ultrastructurally, the neoplastic cells had irregular nuclei with invaginations and relatively little cytoplasm that contained some mitochondria, endoplasmic reticulum, and Golgi complex. The neoplastic cells that comprised the outermost layer of the cell clusters lacked basement membrane at the basal surface and had a large number of microvilli directed toward the surrounding stroma (Fig. 2). Desmosome-like structures were at times seen between the neoplastic cells. The space around the neoplastic cell clusters was surrounded by stromal fibrous components, such as a variable volume of collagen fibrils and a few cell processes presumed to be from fibroblasts or myofibroblasts.
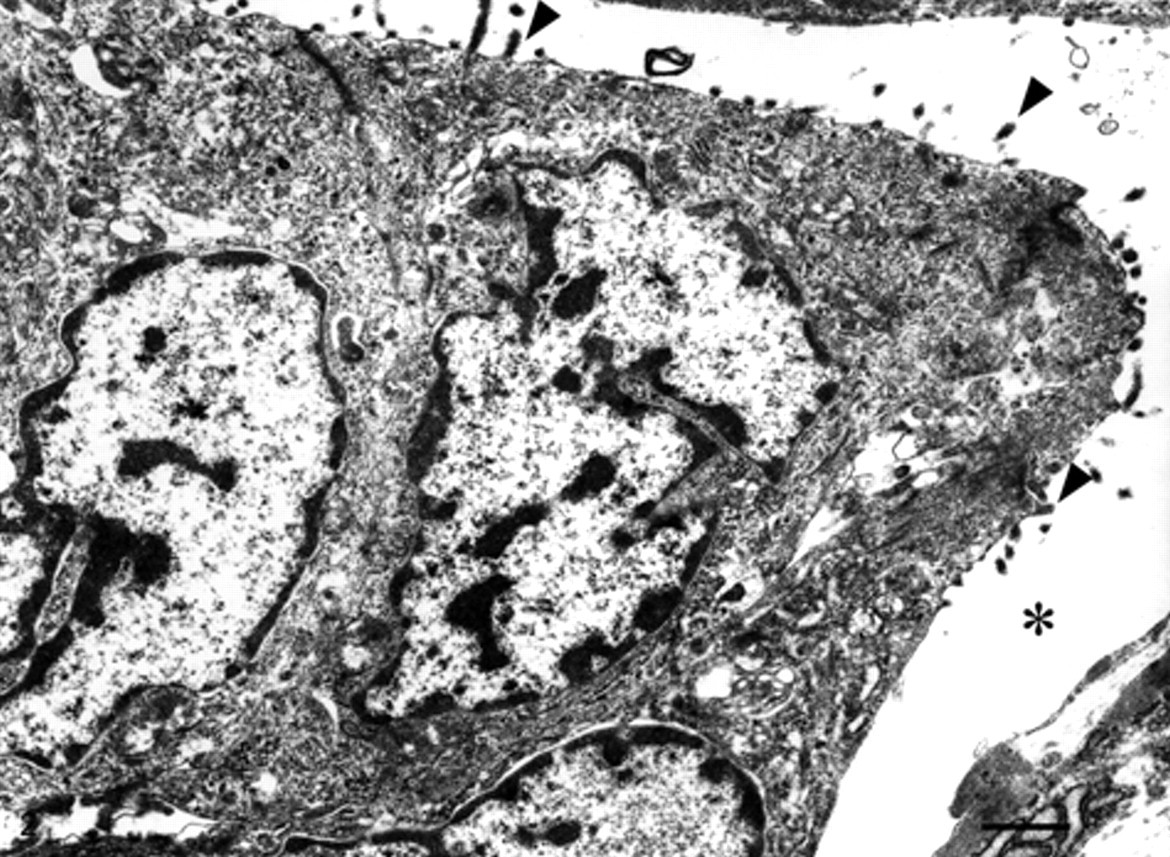
Electron microscopic view of the tumor cells showing microvilli (arrowheads) directed toward the clear space (asterisk). Bar = 1 µm.
The unusual growth pattern of IMC is also correlated with a high incidence of metastasis. In 2 previous studies of human breast IMC, the incidence of axillary node metastasis was 27 (100%) out of 27 cases and 20 (95%) out of 21 cases, respectively. 6,9 Similarly, in 16 feline cases of mammary IMC, the incidence of lymphatic invasion and lymph node metastasis, demonstrating the aggressive behavior of this tumor, was 16 (100%) and 11 (68.8%), respectively. 11 In the case herein reported, the aggressive nature of the tumor was exhibited by evidence of lymphatic invasion and rapid growth. Regardless of this potential for malignancy, the mitotic activity was not very high (Ki-67 index: 27.00%). In a report on mammary IMC in cats, the Ki-67 index (34.75%) was lower than that of other invasive mammary carcinomas of nonspecified type (41.10%). 11 This suggests that the neoplastic cells show high invasiveness even though they do not have high mitotic ability.
In most human and animal IMCs, tumors are composed of variable proportions of the neoplasm having this invasive micropapillary growth pattern interspersed with areas having more typical solid or tubulopapillary configurations. Tumors comprised entirely of neoplastic cells having IMC growth pattern (sometimes referred to as “pure-type” vs. mixed IMC) are rare. In a study based on data collected from 1,056 women with primary breast carcinoma, the incidence of all IMC variants was 11 (1.0%) and that of pure IMC was 6 (0.6%). 2 In a report based on 16 female cats with mammary IMCs, only 1 (6.3%) had a pure micropapillary pattern. 11 In an unrelated dog study, none of the 9 IMCs evaluated showed a pure micropapillary pattern. 4 Thus, the occurrence of a pure-type IMC seems to be rare. In human beings, it is known that the presence of the IMC pattern within the invasive breast carcinoma shows the unfavorable nature of the tumor, regardless of the proportion. 6
Invasive micropapillary carcinoma is different histologically from conventional tubulopapillary carcinoma, which is usually described in mammary and apocrine sweat glands, in that tubulopapillary carcinoma form a complex arborescent growth pattern within dilated cystic epithelial-lined spaces. In IMC, neoplastic cell clusters are devoid of fibrovascular cores and often display pseudotubules, due to foci of the central necrosis. Inverse polarity of these cell clusters, which is consistent with the characteristic inside-out growth pattern of IMC, was confirmed in the current case by the presence of a CEA-positive reaction or microvilli only on the cell surface directed outward toward the clear stroma spaces. The precise mechanism that underlies such a unique growth pattern remains unclear. In most IMCs, these cell clusters are interspersed with areas of more conventional tubulopapillary arrangement. It may be hypothesized that some of the neoplastic cells of the tubulopapillary type lose their polarity and instead develop an ability to invade tissue. Next, nonpolarized cells may penetrate the basement membrane and invade the fibrocollagenous stroma, and these may be transformed into the repolarized cells that form the cell clusters in clear spaces.
Based on the location of the mass and the immunohistochemical results that were positive for CAM5.2 and CEA, and negative for cytokeratin 14, the present tumor may have originated from the cutaneous apocrine sweat glands. In the skin of cats, epithelial cells of apocrine sweat glands are strongly positive for epithelium-specific markers such as CAM5.2 (cytokeratin 7/8; Walder EJ: 2000, Neoplasms of small adnexal keratinocytes: new developments, new questions, In: Proceedings of the 51st Meeting of the American College of Veterinary Pathologists, pp. 165–169, Amelia Island, FL). 5,13 This marker is also expressed in all well-differentiated apocrine sweat gland tumors. Keratinocytes in the basal and prickle cell layer, in the follicular outer root sheath, hair bulbs, and epithelial cells containing sebaceous glands, as well as the outer layer of the apocrine sweat glands, were positive for cytokeratin 14, but not for CEA. 7,9 In the current case, a CEA-positive reaction in the buccal skin was seen only along the luminal surface of the normal apocrine sweat glands and along the external rim of the neoplastic cell clusters within clear spaces. Furthermore, the site of the positive CEA reaction was ultramicroscopically shown to coincide with the location of the microvilli. In normal sweat glands, immunoelectron microscopic investigation shows that biosynthesis and processing of CEA occur in the endoplasmic reticulum and vesicles. Along the luminal surfaces, CEA as such represents an integral part of microvilli. 7 The results support the hypothesis that this tumor may originate from the apocrine sweat glands of the buccal skin. On the other hand, salivary gland carcinoma was ruled out since the growth was almost entirely restricted to the dermis. Unfortunately, the possibility of a skin metastasis from visceral carcinoma cannot be completely ruled out because a postmortem was not performed. However, this was thought less likely as the patient had neither clinical findings suggestive of the presence of another neoplasm, nor a history of previous surgery, such as mastectomy. To the authors’ knowledge, this is the first report of IMC most likely originating from the sweat glands in animals, including human beings.
Footnotes
Acknowledgements
The authors wish to thank Mr. Hirokazu Moriya for providing this specimen and the follow-up information, and are grateful to Mr. Akira Tanaka for his excellent technical assistance.
a.
BD, Franklin Lakes, NJ.
b.
Dako Denmark A/S, Glostrup, Denmark.
c.
BioGenex Laboratories Inc., San Ramon, CA.
d.
NeoMarkers Inc., Fremont, CA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
