Abstract
Animal models are useful tools to study etiology, progress, and new treatments of disease and are an approximation of human disease for experimental study. Intracardiac injection of the human estrogen-independent breast cancer cell line MDA-MB-231 in nude mice is a well-characterized animal model of bone metastasis mainly used to study new treatments for late-stage breast cancer. According to the published literature, this model should produce radiologically distinguishable bone tumors within 17 days after injection. Mice should develop complications such as cachexia, paraplegia, and morbidity within 28 days and require euthanasia within 35 days after injection. We report a study in which injection of MDA-MB-231 cell line led to brain rather than bone metastasis. Unexpected alterations in biological behavior are an important confounding variable in the use of tumor cell lines, and the occurrence and cause of such variants is poorly documented.
Bone is the preferred site for metastasis of many human cancers, particularly those of breast, prostate, thyroid, and lung. Bone metastasis causes intractable pain; skeletal damage, such as pathologic fracture and spinal column compression; and hypercalcemia of malignancy. Many animal models have been developed to study the preferential spread of cancer cells to bone. One of these, intracardiac injection of the human estrogen independent breast cancer cell line MDA-MB-231 into the left ventricle of BALB/c nude mice, is well-characterized 1, 8 and has been used to evaluate new treatment and prevention strategies for breast cancer metastasis. 4, 6 Arterial distribution of tumor cells follows left ventricular injection with direct spread of cancer cells through the systemic circulation to bone and visceral organs, including brain, adrenal glands, and ovaries. 12 With this model, mice develop radiologically distinguishable bone lesions within 17 days after injection (AI).
MDA-MB-231 (HTB-26 clone) cells were obtained from the American Type Culture Collection (ATCC) (Manassas, Virginia, USA). The vial received from the ATCC was held in the vapor phase of liquid nitrogen for 5 days and then cultured in a humidified atmosphere of 5% CO2 in air at 37°C in Dulbecco's Minimal Essential Medium/F12 (DMEM/F12) supplemented with 10% fetal bovine serum and 1% penicillin-streptomycin solution (all purchased from Invitrogen/GibcoBRL, Edmonton, Alberta, Canada). Cell aliquots were cultured for 5 days with 3 expansions. An aliquot was then used immediately for the study reported herein, and 3 additional aliquots were frozen at −70°C. In total the cells were cultured 3 passages beyond the reference culture, which is within the range of acceptable passages recommended by ATCC. The aliquot used for the experiment demonstrated >95% viability, as tested with trypan-blue exclusion.
Eight 3-week-old female BALB/cAnN.Cg-Foxn1 nu/Crl nude mice (strain code 194) were purchased from Charles River Laboratories (Saint-Constant, Quebec, Canada), housed according to the standards established by the Animal Care Committee of the Cross Cancer Institute and in accordance with the guidelines of the Canadian Council on Animal Care, and adapted to laboratory conditions for 2 weeks before the study.
Intracardiac injection was performed as described previously, 9 and weekly radiographic images of the mice were collected from days 28 to 60 (AI) using a Faxitron high-resolution digital x-ray instrument (Field Emission Corporation, McMinnville, Oregon, USA) at 20 kV with a 10-second exposure time. Upon termination of the study at day 60 each animal was subjected to gross and microscopic postmortem examination.
Mice maintained a normal growth rate up to 40 days AI. There was no significant difference in weight gain from control mice that had no injection or other treatment (data not presented). The mice were clinically normal during the first month of the study but then exhibited decreased activity, vocalized more commonly, lost their balance when placed on a glass surface, and appeared disoriented with normal handling. A few mice became paraplegic and/or developed head tilts and/or had a rapid decrease in body weight. In accordance with the animal care policy, animals exhibiting these signs were euthanatized. Most of the mice in the study (5/8; 63%) survived without severe clinical signs to an established end point of 60 days. Despite our expectation based on this animal model, radiographic images showed no evidence of skeletal metastases.
Postmortem examination did not reveal any gross abnormalities in any of the mice. The brain, lungs, heart, liver, spleen, kidneys, and adrenal glands were removed from all 8 mice, fixed in 10% neutral buffered formalin, and HE stained sections prepared for histopathologic examination. In addition, the femurs and humeri were removed from 4 mice and fixed, and HE sections were similarly prepared. Microscopic examination revealed masses of malignant tumor cells randomly located in the brain and adrenal glands of 6 of 8 mice. The morphology of the MDA-MB-231 cells was similar to that in other published reports using this cell line. 3 Metastases in the brain varied from large areas overrun by carcinoma cells (Fig. 1) to small clusters or individual malignant cells (Fig. 2). The larger masses tended to be located in the cerebellum, basal ganglia, and olfactory lobes, but tumor masses could be found anywhere in the brain. Malignant cells in the brain were variable in size and shape, and very large bizarre types were common. Where malignant cell masses were present in the adrenal glands, they tended to be located in the corticomedullary region and to grow outward into the cortex from this location. Tumor cells had two general morphologic appearances in the adrenal glands: a large epithelioid type and an elongated fibrocyte type. Malignant cells were not observed microscopically in sections of long bones from those mice selected for microscopic examination of the bone.
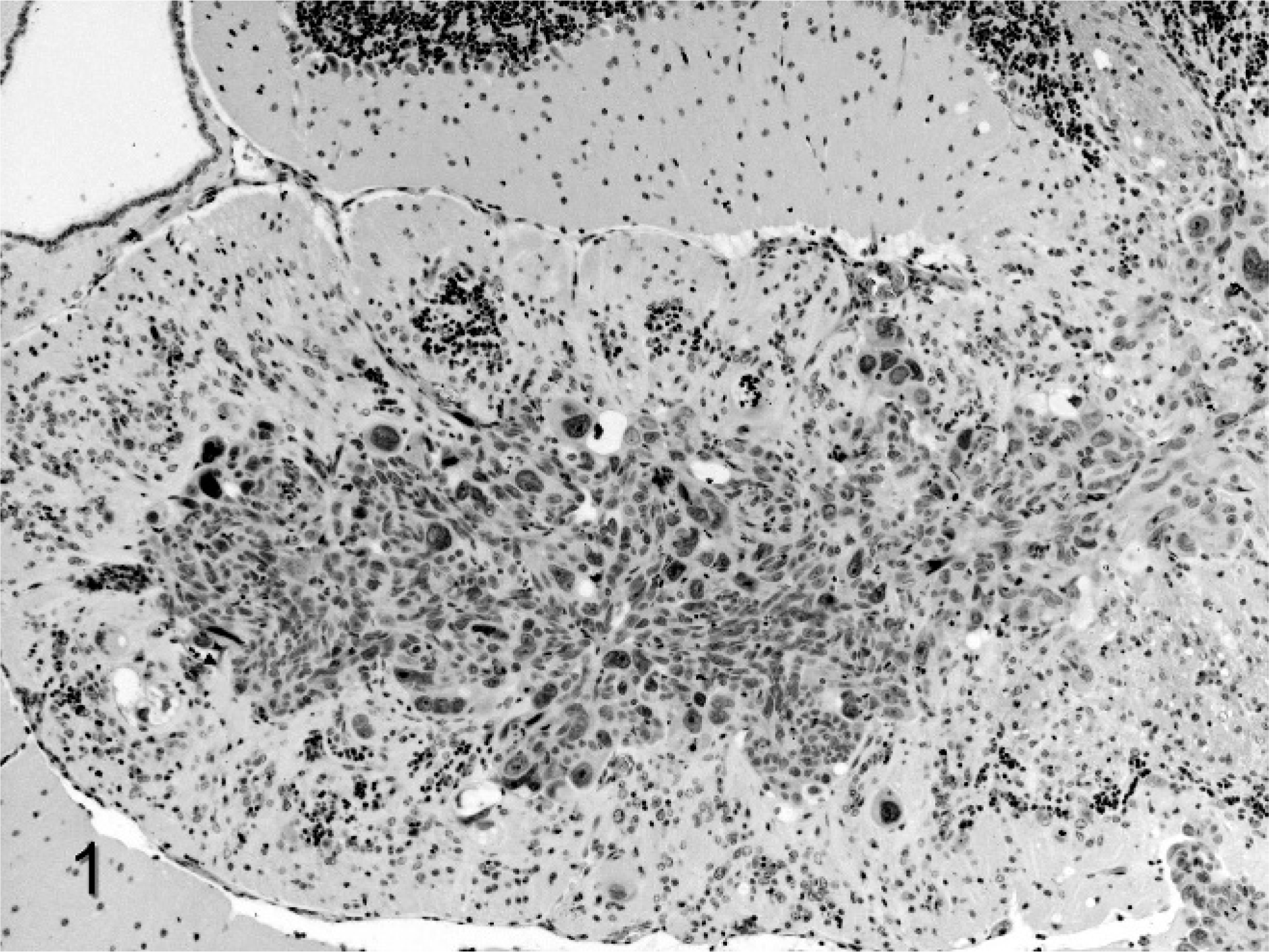
Large metastatic mass from the human estrogen–independent breast cancer cell line MDA-MB-231 in the cerebellum of female BALB/c nude mice. Extensive disruption of the cerebellar architecture accounts for the clinical signs. 100×.
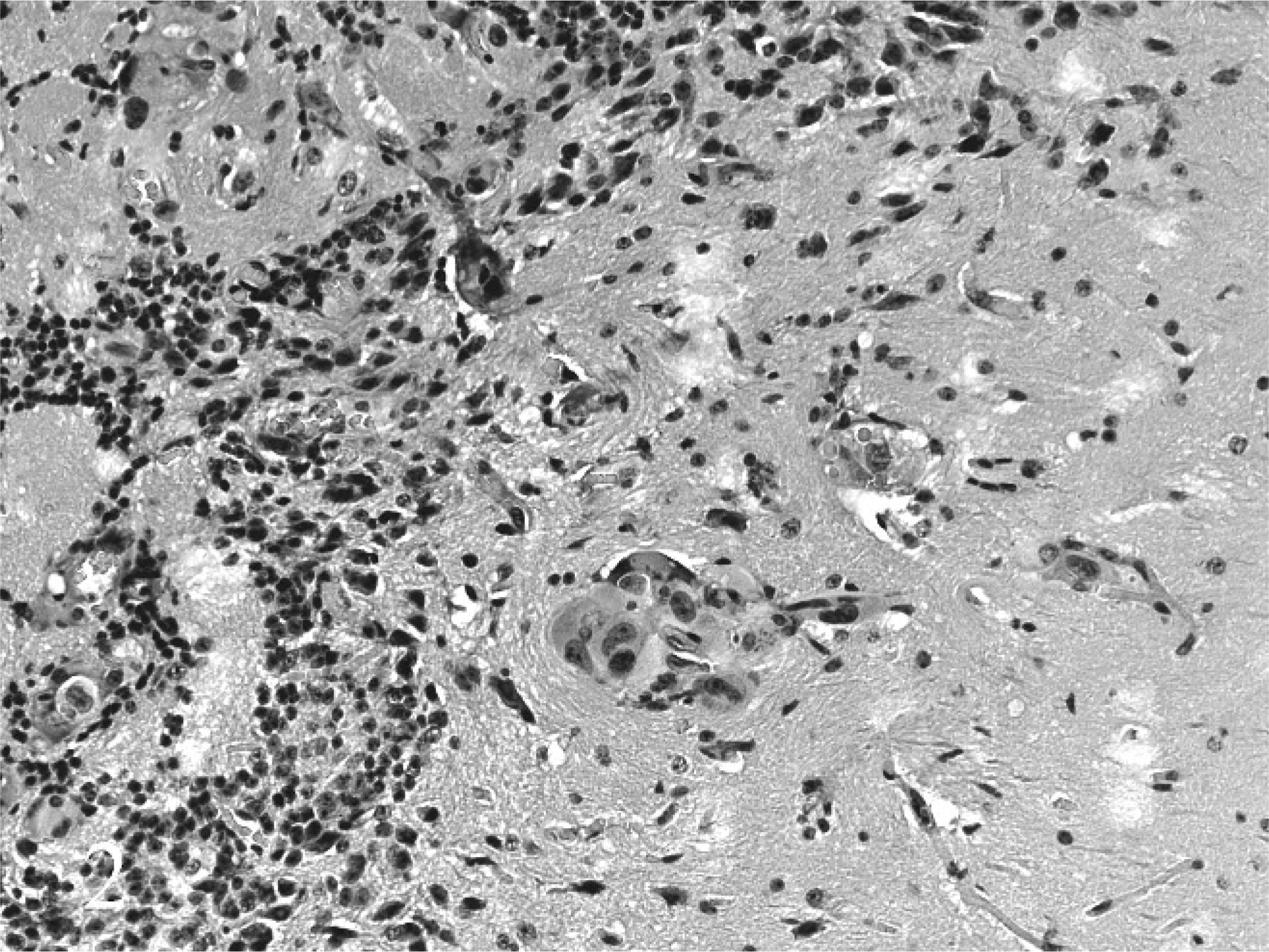
Micrometastases from the human estrogen–independent breast cancer cell line MDA-MB-231 in the olfactory area of female BALB/c nude mice. 200×.
Researchers generally expect cell lines transplanted into mice to behave in a consistent manner from experiment to experiment. This is particularly true for lines obtained from sources such as the ATCC. Commonly, when a cell line behaves differently from expected, researchers tend to question the quality of the animal used in the model rather than the cell line. However, in modern facilities with high standards of animal management and biosecurity, the animals are usually of consistent quality, and the nature of the cell line needs to be more critically assessed in these situations. 2
Clinical signs exhibited by the mice in this study, such as decreased activity and increased vocalization, could be mistaken for the effects of bone metastases in causing pain and/or damaging long bones. However, radiographic signs of metastases and the general weight loss reported for this model by other groups 9 were not present in this study. Such clinical signs as ataxia, head tilting, and loss of balance are not typical of mice with bone metastases. Therefore, the clinical presentation was not entirely typical for bone metastasis as described for this model. Microscopic metastases of the tumor cell line in the brain and adrenal glands, but not in any of the bones, is consistent with the clinical and radiographic findings. Localization of metastases in brain tissue and tissue of neural crest origin (adrenal medulla) in preference to bone is anomalous for this cell line and animal model.
Local microenvironment is thought to play an important role in explaining the predilection of tumor cells to preferentially grow in tissues such as bone rather than other tissues. 5 There is now also recognition that selection pressure is needed on cell lines to maintain their phenotypic metastatic patterns. 8 Divergence of sites of predilection for metastasis has been produced experimentally for the MDA-MB-231 cell line by serial passage in nude mice and selection for bone metastasis in one subline, brain metastasis in another, 12 and bone and adrenal in yet another. 10 However, in the situation reported in this article, it appears that spontaneous and rather sudden alteration occurred, resulting in immediate preferential metastatic colonization of brain rather than bone. Cells were obtained directly from the ATCC, handled according to ATCC guidelines, and used immediately after only 3 passages of the reference line. Mishandling of the cell line was unlikely the cause of such sudden change in this particular situation.
Although the etiology of metastatic predilection for neural tissue rather than bone has not been determined for the MDA-MB-231 cell line in BALB/c nude mice, two broad classes of factors govern the metastasis of malignant cells to a distant site: the local environment at the site of metastasis and the biological properties of the malignant cells. 1 Bone is a common site of metastatic osteolytic tumors from primary breast cancer in humans, and although the mechanism of metastatic colonization is complex, the site-specific mediators of tumor growth are well understood. Although breast and other tumors are known to metastasize to the brain, this is much less frequently encountered than bone. A number of models of mouse brain tumors have been developed, but relatively little is known about the factors involved in the development of brain metastases in animal models or in humans. 11 The blood-brain barrier presents an initial impediment to tumor cell colonization, and this barrier at the level of the brain microvasculature must be penetrated in order for metastases to develop. It has also been observed that the release of cytokines by glial cells contributes to the growth and metastatic potential of tumor cells within brain tissue. 7
The MDA-MB-231 tumor line has been a well-established model for metastatic bone tumors after intracardiac injection, even though the development of tumors in other tissues has been observed. 12 It is notable that the manipulation of this line in an attempt to develop clones with more specificity to bone or brain required multiple passages through mice where tumors colonizing the bone or brain were harvested, expanded, and reinjected into new mice. To develop 2 sublines with differing organ specificity in mice required 9 passages for the bone-specific subline and 6 passages for the brain subline, respectively. 12 The ability of this cell line to provide clones capable of metastasizing to different organs provides a clue to the observations made in our study but does not explain the dramatic loss of bone-targeting ability we observed. Subsequent to these observations we have successfully used the bone-specific clone MDA-MB-231BO developed by Yoneda et al. 12 in the BALB/c nude mouse model to reliably develop metastatic bone tumors.
Our observations suggest that the MDA-MB-231 cell line is capable of rapid transformation in addition to its previously observed heterogeneity in tissue targeting for metastatic growth. The cell line should be used with caution and with appropriate controls to ensure its utility as a model of bone metastases.
It is also necessary to recognize that as many as 18% of cell lines are not what they are thought to be. 2 The ATCC now does not distribute those lines that they know are contaminated or altered, but it does not keep a record of variances experienced by users (personal communication). The only source of such information is reports such as this in the scientific literature.
Footnotes
Acknowledgements
The authors acknowledge the Canadian Breast Cancer Foundation for their financial support of this work and the technical assistance of the animal care staff of the Cross Cancer Institute Vivarium.
