Abstract
During fibrosis, the extracellular matrix (ECM) is continuously remodeled and increases in volume due to the production of various proteins. We studied the distribution of tenascin-C (TN-C) and the correlation of TN-C with the necro-inflammatory activity and expression of alpha–smooth muscle actin (α-SMA), cytokeratin 7 (CK7), and CD3+ T-lymphocytes in canine chronic hepatitis. This was analyzed using immunohistochemistry and semiquantitative scoring. We used 3 groups (n = 19) of dogs: group 1 (n = 5) with neonatal hepatitis/lobular dissecting hepatitis (NH/LDH), group 2 (n = 8) with chronic hepatitis/cirrhosis (CH/CIRR), and group 3 (n = 6) consisting of healthy animals. In normal livers, TN-C was localized in Disse's space and around bile ducts and blood vessels. In CH/CIRR livers, TN-C was localized at the periphery of the regenerating nodules and was conspicuous in the bridging fibrous bands. In NH/LDH, TN-C was diffusely distributed along the reticular fibers that dissected between single cells or groups of hepatocytes. α-SMA in the normal hepatic parenchyma showed an irregular distribution along the perisinusoidal linings. In other groups, α-SMA was increased in fibrotic septa and perisinusoidal linings. In normal livers, CK7 was positive in bile ducts. In other groups, CK7-expressing cells were conspicuous in the portal-parenchymal interface, the periphery of the regenerative nodules, and the degenerated parenchyma. The pattern of CD3+ lymphocytes was inversely proportional to that of TN-C. These results also showed that TN-C is strongly correlated with increased fibrotic stage, inflammatory activity, and expression of CK7 and α-SMA. TN-C, CK7, and CD3 expression did not differ between diagnostic groups.
Regardless of etiology, hepatic fibrosis is a common outcome of liver injury in both man and dog. Fibrosis leading to the development of cirrhosis is a major cause of both morbidity and mortality in man. 2, 13 Despite modern diagnostic techniques, the etiology of most cases of canine liver fibrosis remains unknown. 5 Several infectious and noninfectious causes have been suggested for chronic liver disease of dogs. These include canine adenovirus type 1, Leptospira spp., canine acidophil cell hepatitis virus, copper accumulation, drug administration, alpha-1-antitrypsin anomaly, and autoimmunity. 4, 12, 14, 18, 27, 33, 43, 47 However, a recent study failed to identify an infectious agent as the underlying etiology of chronic canine hepatitis. 6 Common histologic features of canine chronic hepatitis are hepatocellular necrosis and apoptosis, inflammation, and progressive fibrosis. 5
In fibrosis, the extracellular matrix (ECM) is continuously being remodeled. 37 An essential feature of fibrosis is an increase and disarray of many ECM proteins. 14 Secretion of cytokines, synthesis of matrix proteins, and inhibition of the activity of serine proteinase and matrix metalloproteinases leads to progressive scar formation. 2 Tenascin-C (TN-C) is one of the ECM proteins that has both adhesive and antiadhesive activities. 2, 8, 10, 29, 37 It is a disulfide-linked oligomer with subunits that range between 190 and 300 kd. 29 In embryonic development, TN-C is present in several developing tissues and at the cellular borders where epithelial/mesenchymal transformation occurs in organs such as the gut, kidneys, and mammary glands. 28, 29, 38 In adult tissue, TN-C expression is absent or much reduced. However, expression is enhanced during processes involving ECM remodeling and cell migration, such as in tumor growth, fibrosis, and wound healing. 8, 37
In the present study, we investigated the immunohistochemical localization of TN-C in livers of healthy dogs and dogs with 2 different types of hepatitis, namely chronic hepatitis/cirrhosis (CH/CIRR) and neonatal hepatitis/lobular dissecting hepatitis (NH/LDH). The relationship between hepatocellular degeneration and necrosis, inflammatory activity, fibrotic stage, TN-C, alpha–smooth muscle actin (α-SMA), cytokeratin 7 (CK7), and CD3 expression was analyzed using a semiquantitative scoring system. Most of the ECM proteins in fibrotic liver are known to be produced by activated hepatic stellate cells (HSC) and other fibroblasts. The correlation between TN-C and α-SMA in various locations of the liver tissue sections was also evaluated to identify the producing cell type.
Materials and Methods
Animals
Liver samples were obtained from biopsy or necropsy cases submitted to the Division of Pathology, Faculty of Veterinary Medicine. Cases of moderate-to-severe CH/CIRR (n = 8) and NH/LDH (n = 5) were selected on the basis of World Small Animal Veterinary Association (WSAVA). 52 The livers of the CH/CIRR group were characterized by broad fibrotic septa and regenerative parenchymal nodules, and the livers of the NH/LDH group were characterized by necrosis, weak-to-severe fibrosis, and disruption of the lobular architecture. 3, 50, 52 Six normal liver samples were obtained from dogs used in research projects approved by the responsible ethical committees for the use of experimental animals and client-owned animals according to Dutch legislation and were used as a control group. Animals were recategorized into 4 subcategories on the basis of their fibrotic stage (stage 0–3) to evaluate the expression of TN-C, α-SMA, CK7 and CD3, and necro-inflammatory activity at different stages of fibrosis (Table 1a). Included were 11 females and 8 males of various breeds with a mean age of 5.7 (±3.6) years.
Mean scores of evaluated criteria at different stage of fibrosis in canine chronic hepatitis.
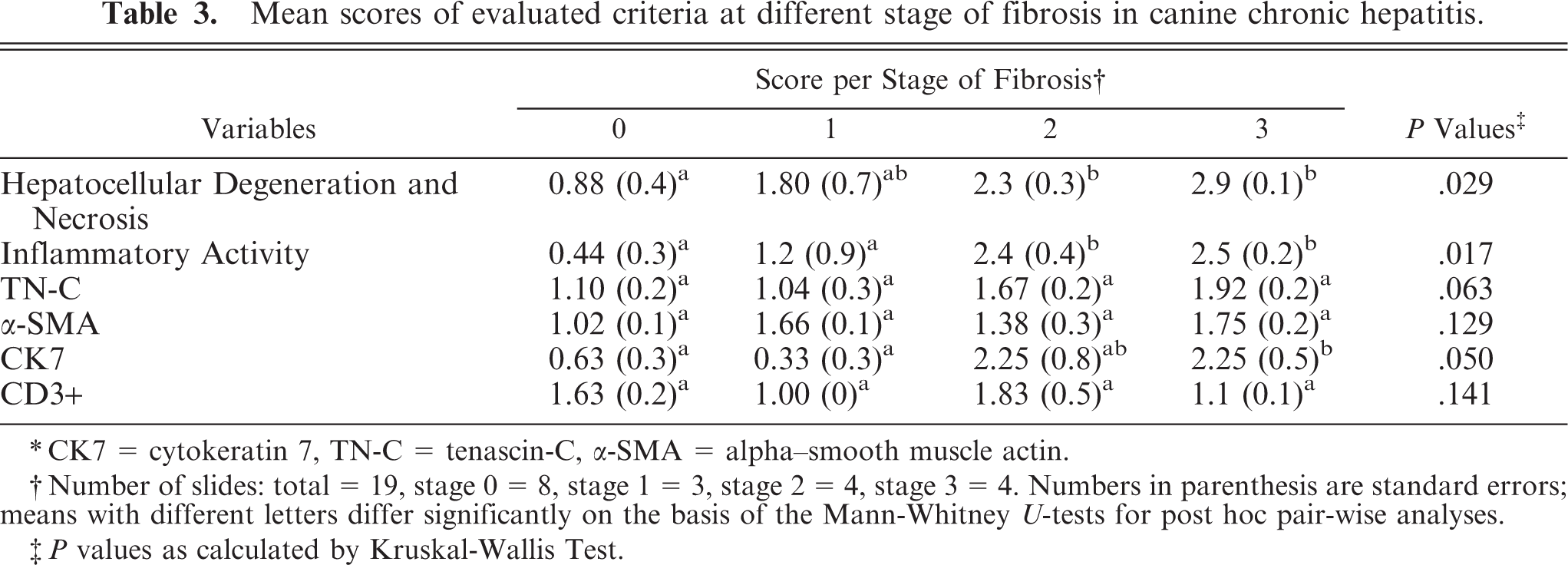
∗CK7 = cytokeratin 7, TN-C = tenascin-C, α-SMA = alpha–smooth muscle actin.
†Number of slides: total = 19, stage 0 = 8, stage 1 = 3, stage 2 = 4, stage 3 = 4. Numbers in parenthesis are standard errors; means with different letters differ significantly on the basis of the Mann-Whitney U-tests for post hoc pair-wise analyses.
‡P values as calculated by Kruskal-Wallis Test.
Histopathology
Liver specimens were fixed in 10% neutral buffered formalin and paraffin-embedded. Slides (3 μm) were routinely stained with HE and Masson trichrome to evaluate the necro-inflammatory activity and fibrotic stage.
Hepatocellular degeneration was evaluated by the presence of intracellular hepatocytic microvacuoles or ballooning degeneration and pyknotic nuclei (Fig. 1), and necrosis, by cellular shrinkage combined with karyorrhexis.
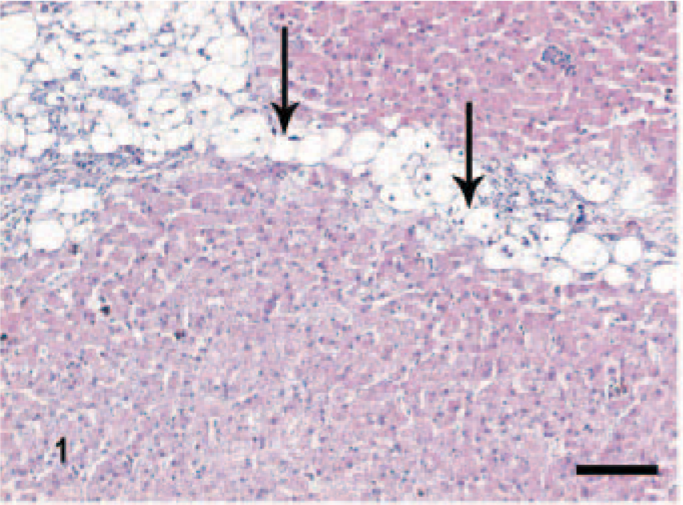
Liver; dog, chronic hepatitis/cirrhosis group. Bridging (portal-portal) hepatocellular ballooning degeneration and necrosis (arrows). HE stain. Bar = 200 μm.
Immunohistochemistry
ABC peroxidase or Envision techniques of immunohistochemical analysis were performed for α-SMA, CD3, CK7, and TN-C. Sections (3 μm) mounted on poly-L lysine–coated slides were deparaffinized and hydrated in decreasing concentrations of alcohol (100–70%). Endogenous peroxidase activity was blocked by 0.3% hydrogen peroxide (H2O2) in methanol for 30 minutes at room temperature (RT). Antigen retrieval was performed for TN-C, CK7, and CD3. TN-C sections were incubated overnight at 37°C with 0.5-U/ml chondroitinase ABC (Sigma-Aldrich Inc., USA) in 0.05-M tris(hydroxymethyl)aminomethane (Tris)/0.06-M Na-acetate, pH 7.8, to unmask the TN-C from chondroitin sulphate 15 and subsequently treated with 0.1% trypsin in 0.05 Tris/hydrochloride buffer, pH 7.8. CK7 sections were treated with proteinase-K (S3020; DAKO Corporation, Carpinteria, CA, USA) for 40 minutes at RT. CD3 sections were heated in 10-mM citrate, pH 6.0, in a microwave oven (765 W, 600 ml, 10 minutes). After washing with phosphate buffered saline (PBS)/Tween-20 (0.01-M PBS, 0.1% Tween-20) for TN-C, α-SMA, and CD3 and with Tris/Tween-20 (0.01-M Tris buffer, 0.1% Tween-20) for CK7, we blocked background staining by incubating the sections with normal horse serum (TN-C and α-SMA) or normal goat serum (CK7 and CD3) at 1 : 10 dilution for 30 minutes at RT. Sections were incubated overnight (4°C) with the primary antibodies (anti-TN-C, 4F10TT, at 1 : 800 dilutions in PBS/1% BSA, bought from IBL Co., Japan; anti-α-SMA, clone 1A4, at 1 : 1,200 dilutions in PBS, bought from BioGenex; anti-CK7, OV-TL, at 1 : 25 dilutions in Tris-buffered saline/1% BSA, bought from DAKO; rabbit polyclonal anti-CD3 antibodies at 1 : 300 dilutions in PBS, bought from DAKO).
After washing, we incubated TN-C and α-SMA sections in horse anti-mouse biotin (Vector Laboratories, Burlingame, CA, USA) diluted 1 : 125 for 45 minutes, and CD3 sections were incubated in goat anti-rabbit biotin diluted at 1 : 250 for 30 minutes at RT. CK7 sections were incubated with Envision (DAKO, K4001, goat anti-mouse) for 45 minutes at RT. After washing, we incubated sections for TN-C, α-SMA, and CD3 in avidin–biotin–peroxidase complex (Vector Laboratories). The color was developed in 3-3′-diaminobenzidine and H2O2 0.02% in Tris buffer, pH 7.8, for TN-C and α-SMA and Vector NovaRed (Vector Laboratories) for CK7 and CD3. The sections were counterstained with 10% Mayer's hematoxylin. After sections were washed in tap water for 10 minutes, they were dehydrated in increasing concentrations of alcohols (70–100%). Negative controls were incubated in PBS instead of the primary antibodies. Sections from mammary tumors with previously demonstrated TN-C expression were used as positive controls for TN-C. Smooth muscle within the blood vessel walls was used as internal positive controls for α-SMA. Bile ducts were used as internal positive controls for CK7. Normal canine lymph nodes were used as positive controls for CD3 immunohistochemical staining.
Semiquantitative analyses
Semiquantitative scoring for each parameter was performed independently by 2 of the authors (GAM, HN). The scoring method was based on the system used by Boisclair et al. 5 with the modifications as described in Tables 1a, 1b.
Criteria used in immunohistochemical semiquantitative scoring of canine chronic hepatitis.

∗CK7 = cytokeratin 7, TN-C = tenascin-C, α-SMA = alpha–smooth muscle actin.
Criteria used in semiquantitative scoring for canine chronic hepatitis.
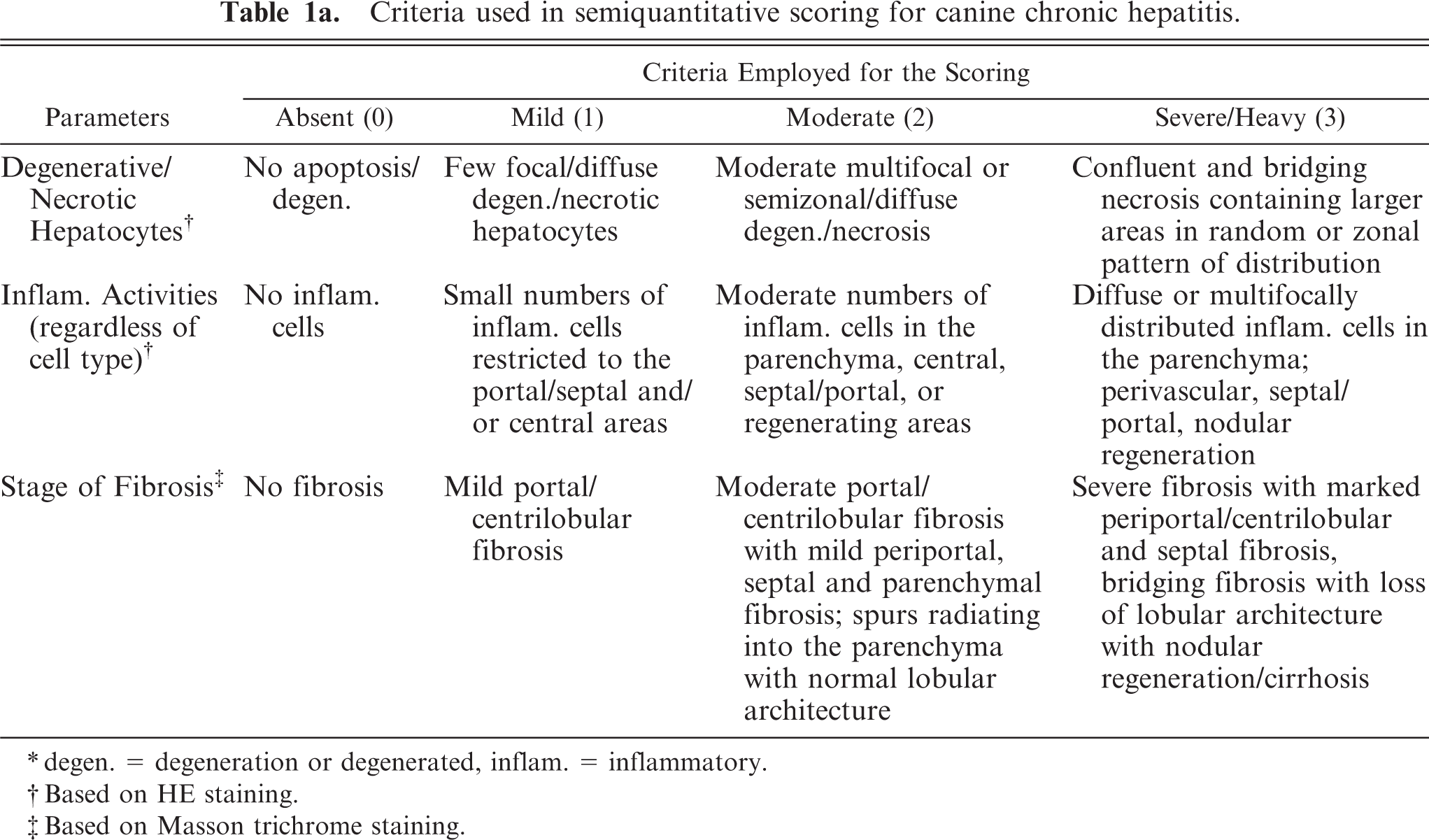
∗degen. = degeneration or degenerated, inflam. = inflammatory.
†Based on HE staining.
‡Based on Masson trichrome staining.
Statistical analysis
The scoring results between different groups were analyzed using the Kruskal-Wallis H-test. Mann-Whitney U-tests for post hoc pair-wise analyses were used to identify the specific group demonstrating significant difference. Possible associations between variables were verified by the Spearman correlation test. A nominal significance level of P < .05, P < .01, and P < .001 was used for the analysis.
Results
Histopathology: HE
Hepatocellular degeneration, necrosis, and inflammatory activity
The definition of the scores of these parameters, based on HE staining, is given in Table 1a. In normal liver sections, hepatocellular degeneration, inflammation, and necrosis were absent. In the CH/CIRR group, there was extensive, often bridging hepatocellular degeneration and necrosis (Fig. 1). The parenchyma was divided by fibrotic septa, which encompassed regenerative nodules. These septa were mildly or moderately infiltrated with lymphocytes and plasma cells. In 1 dog, massive infiltration of neutrophils with lymphocytes and plasma cells occurred without remarkable hepatocellular degeneration.
Dogs in the NH/LDH group had diffusely or focally disseminated degenerate hepatocytes (Fig. 2) with mild-to-moderate lymphocytic inflammation, which infiltrated along the fibrotic septa. However, 1 dog had massive infiltration with both lymphoid cells and neutrophils. The hepatic parenchyma was separated into individual cells or small groups of cells by thin fibrotic septa throughout (Fig. 2).
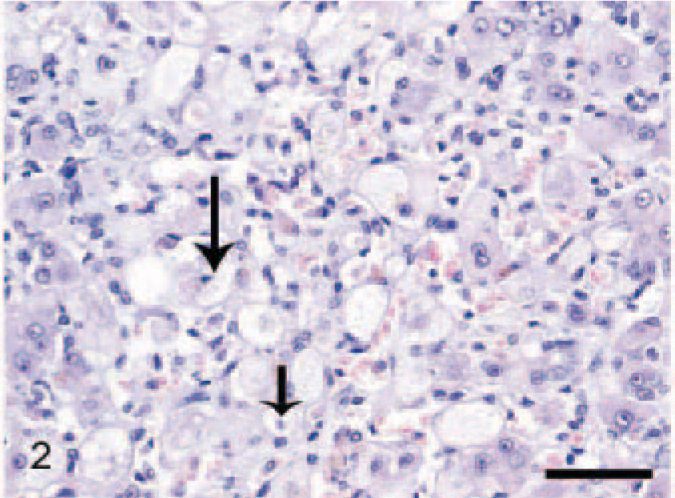
Liver; dog, neonatal hepatitis/lobular dissecting hepatitis group. Degenerated hepatocytes are subdivided into small groups or individual cells by fine fibrous septa throughout the parenchyma. Necrosis is present (arrows), HE stain. Bar = 100 μm.
Fibrotic stage
The definition of the fibrosis scores, based on Masson trichrome staining, is given in Table 1a. The control dogs (n = 6) showed no signs of fibrosis. Among the chronic CH/CIRR dogs (n = 8), 3 dogs showed the third stage of fibrosis; 3 dogs, the second stage; 1, the first stage; and the other showed no fibrosis. In NH/LDH dogs (n = 5), 1 dog showed the third fibrotic stage; 2 dogs, the second stage; 1 showed the first stage; and the other showed no fibrosis. In livers of dogs in both hepatitis categories, which are based on HE staining, fibrosis intensity might vary from absent to severe.
Immunohistochemistry
TN-C
In normal liver, moderate discontinuous reactivity was observed in Disse's space (between the endothelial lining of the sinusoid and hepatocytes) (Figs. 3, 4), randomly distributed around a few bile ducts and blood vessels and under Glisson's capsule. These findings were consistent in all control livers.
Same animal as in Fig. 3 at a higher magnification. The positive tenascin-C signal is localized in Disse's space. Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 50 μm.
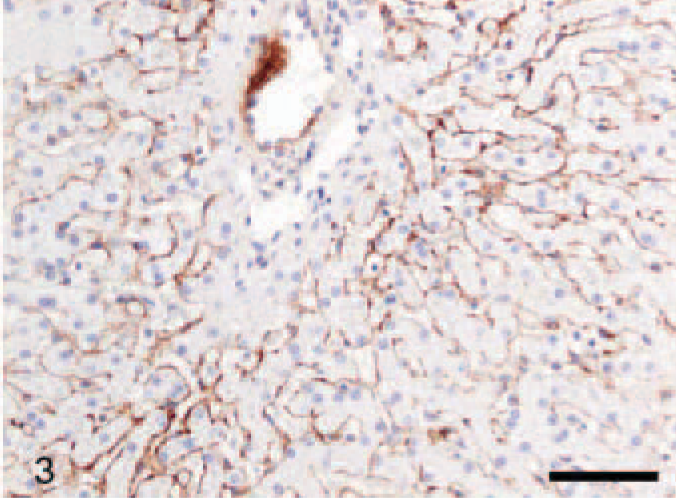
Liver; dog, control. Labeled for tenascin-C. Moderate perisinusoidal positive reaction, mainly in zone 2 of hepatic acinus. Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 100 μm.
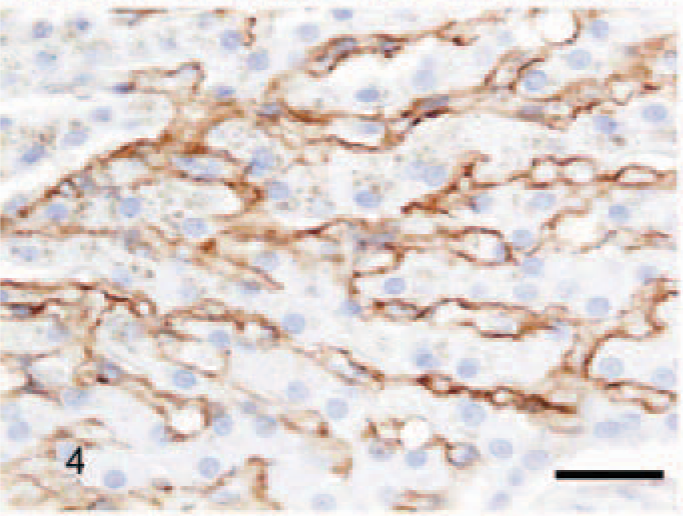
In CH/CIRR livers, intense positivity for TN-C was shown in the fibrotic areas bordering the regenerative nodules (Fig. 5) and in the fibrous bands invading into the parenchyma. Generally, a prominent signal was present in Disse's space in the pre-existing parenchyma outside the regenerative nodules, often associated with ballooning degeneration (Fig. 6) and at random around a few bile ducts and blood vessels. However, in regenerative nodules, reactivity was very scant or absent. In NH/LDH dogs, the pattern of positivity was associated with the reticulin and collagen fibers dissecting individual cells or groups of cells. However, the signal in Disse's space and around the bile ducts and blood vessels was decreased when compared with that in the controls.
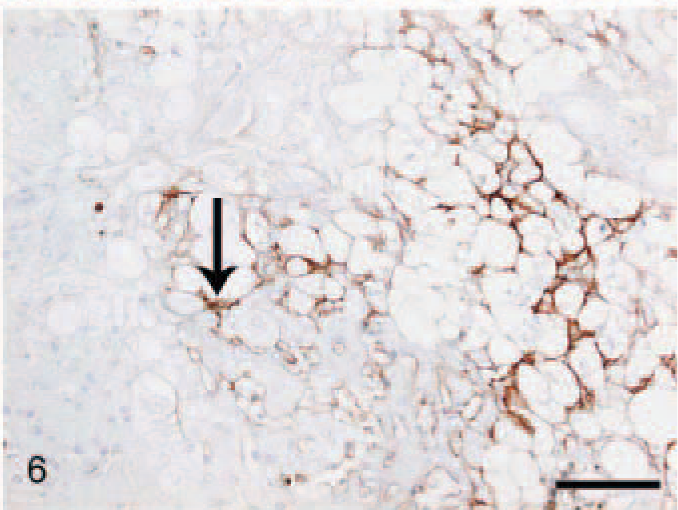
Liver; dog, chronic hepatitis/cirrhosis group. Labeled for tenascin-C. Moderate positive reaction in Disse's space (arrow) associated with hepatocellular ballooning degeneration. Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 100 μm.
Liver; dog, chronic hepatitis/cirrhosis group. Labeled for tenascin-C. Intense positive staining in the fibrous stroma (arrow) surrounding a negatively staining regenerative nodule (arrowhead). Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 200 μm.
α-SMA
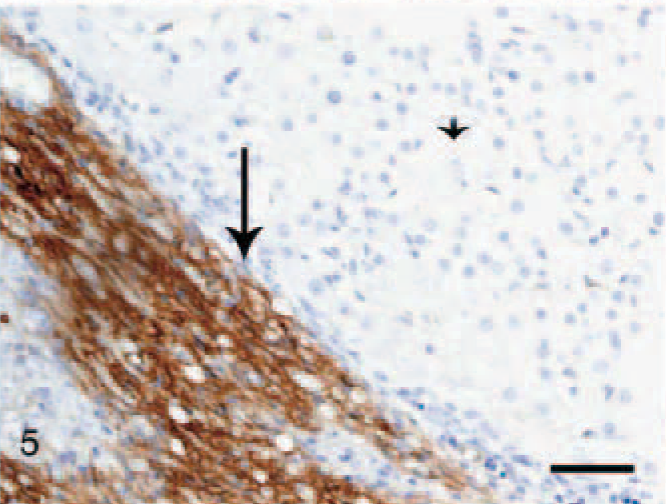
In the normal liver, a slightly irregular (1–3 μm wide) moderate staining was seen in the perisinusoidal spaces throughout the hepatic parenchyma, interpreted as being present in HSC (Fig. 7). In fibrotic livers (both CH/CIRR and NH/LDH), the perisinusoidal signal was enhanced in intensity and surface area, irregularly distributed among the pre-existent lobules (Fig. 8). In the regenerative nodules, the signal was reduced or absent. Livers with moderate-to-severe fibrosis showed numerous α-SMA-positive cells in the portal triads, fibrotic septa, and stroma surrounding the regenerative nodules, interpreted as activated myofibroblasts. In addition, the NH/LDH livers showed numerous α-SMA-positive cells associated with the reticulin and collagen fibers dissecting individual or groups of hepatocytes.
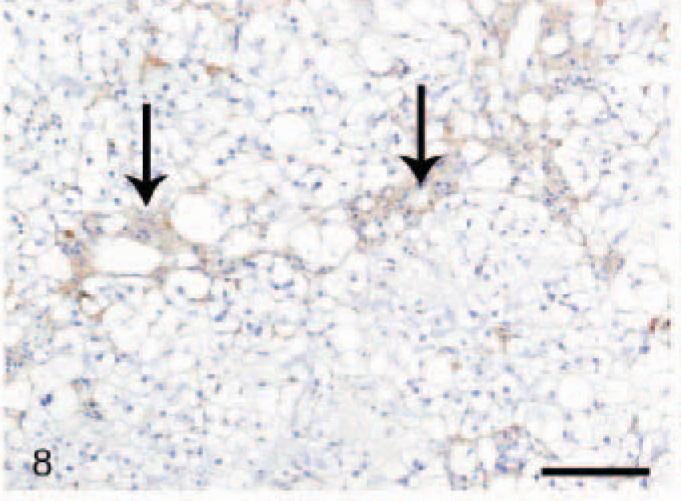
Liver; dog, chronic hepatitis/cirrhosis group. Labeled for alpha–smooth muscle actin. The perisinusoidal signal is enhanced in intensity and surface area (arrows), depending on the number of activated portal myofibroblasts and activated (parenchymal) hepatic stellate cells. Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 100 μm.
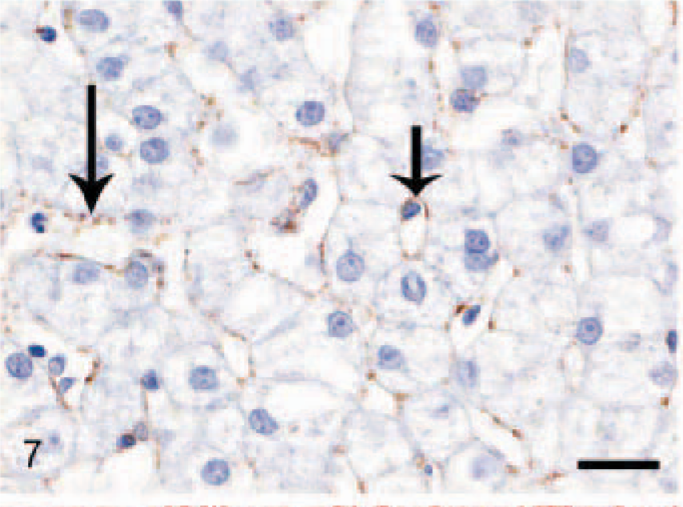
Liver; dog, control. Labeled for alpha–smooth muscle actin. Reactive perisinusoidal hepatic stellate cells produce a thin irregular positive band lining the sinusoids (arrows). Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 20 μm.
CK7
Normal livers usually contained less than 3 bile duct cross-sections per portal triad. CK7+ cells were present mostly in the bile ducts and rarely in the portal-parenchymal interface. In the cirrhotic sections, CK7+ ductular proliferation was conspicuous in the portal areas, especially at the portal-parenchymal interface with some local infiltration into the surrounding parenchyma (Fig. 9). Extensive CK7+ ductular proliferation was also observed in fibrotic septa and extending into the periphery of the regenerative nodules (Fig. 10). In 3 of the CH/CIRR livers, clusters of CK7+ cells were diffusely distributed among the sinusoids, associated with marked hepatocellular degeneration. In the NH/LDH group, CK7+ cells were present in the portal areas, the portal-parenchymal interface, and the parenchyma (Fig. 11). Positive cells, individually or in primitive ducts, were diffusely distributed in the parenchyma, associated with reticular fibers of the smallest fibrotic septa.
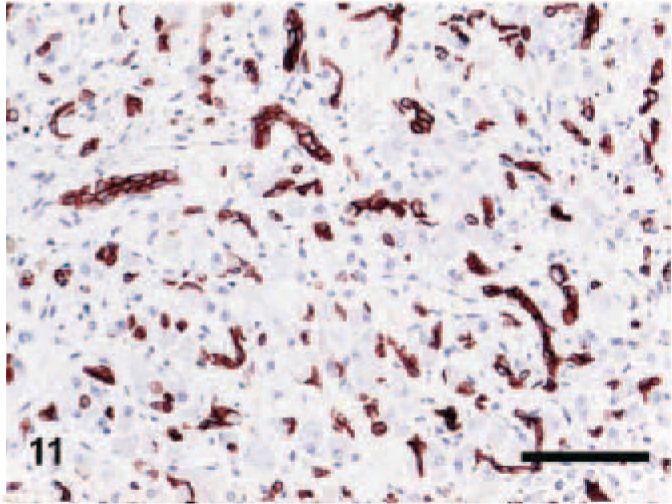
Liver; dog, neonatal hepatitis/lobular dissecting hepatitis group. Labeled for cytokeratin 7 (CK7). CK7-positive cells in the parenchyma. Envision method, counterstained with Mayer's hematoxylin. Bar = 100 μm.
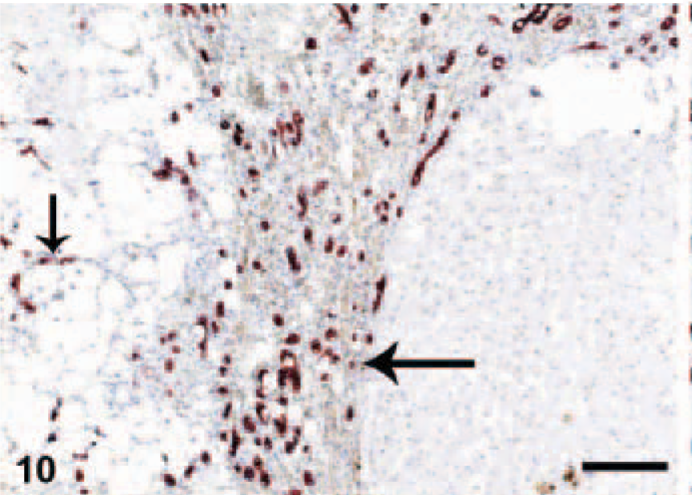
Liver; dog, chronic hepatitis/cirrhosis group. Labeled for cytokeratin 7. Abundant positive reaction in ductular proliferations in a thick fibrous septum along the periphery of the regenerative nodule (arrow) and in degenerated parenchyma (arrowhead). Envision method, counterstained with Mayer's hematoxylin. Bar = 200 μm.
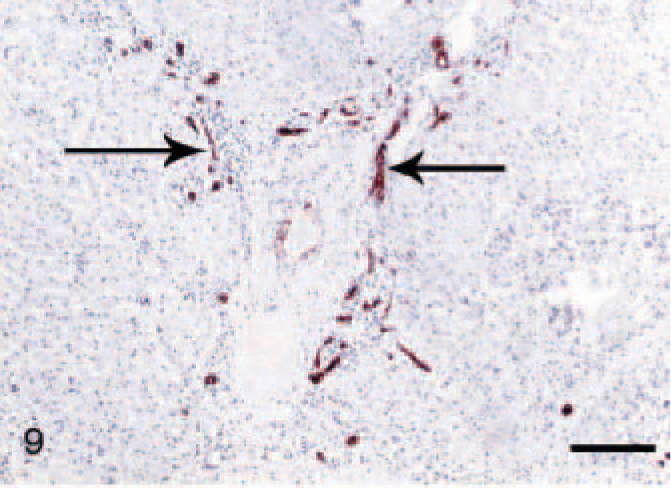
Liver; dog, chronic hepatitis/cirrhosis group. Labeled for cytokeratin 7. Ductular proliferation (arrows) was conspicuous in the portal areas and fibrous septa, particularly at the portal-parenchymal interface. Envision method, counterstained with Mayer's hematoxylin. Bar = 200 μm.
CD3
Moderate-to-high numbers of CD3+ T-lymphocytes were observed in 6 out of 8 CH/CIRR dogs and in 1 of the dogs in the NH/LDH group (n = 5), while all controls were negative (n = 6). Some dogs in both CH/CIRR and NH/LDH groups showed no CD3+ T-lymphocytes. These were associated with moderate-to-severe infiltrations primarily of neutrophils and plasma cells in the portal and periportal areas. The pattern and distribution of CD3+ cells was inversely proportional to TN-C reactivity (Fig. 12).
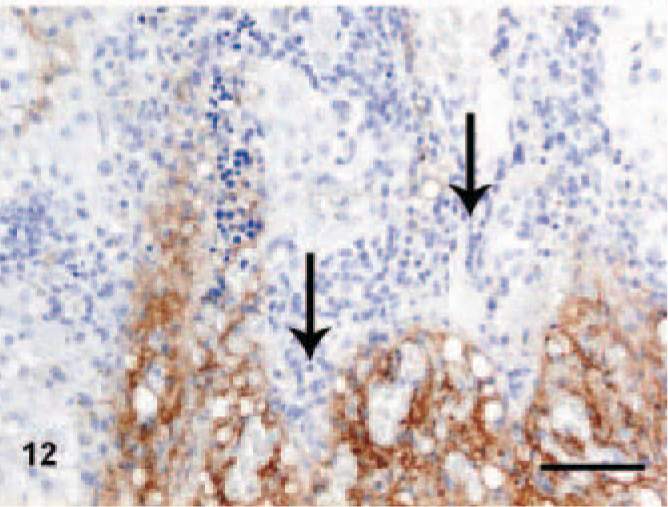
Liver; dog, chronic hepatitis/cirrhosis group. Labeled with tenascin-C (TN-C). TN-C reaction is low or absent in areas with severe lymphocytic infiltrations (arrows). Avidin–biotin–peroxidase method, counterstained with Mayer's hematoxylin. Bar = 100 μm.
Semiquantitative scoring and statistical analysis
Comparison of scores among the groups of dogs
Hepatocellular degeneration and necrosis, inflammatory activity, fibrotic stage, and α-SMA expression showed significant differences in their mean scores among the 3 groups. Fibrotic stage, TN-C, α-SMA, and CK7 showed higher mean scores in the CH/CIRR group compared with the NH/LDH group; however, significance was seen only for α-SMA. Inflammatory activity and hepatocellular degeneration and necrosis scores were higher in the NH/LDH group, although this finding was not statistically significant (Table 2).
Mean scores of evaluated criteria in canine chronic hepatitis.
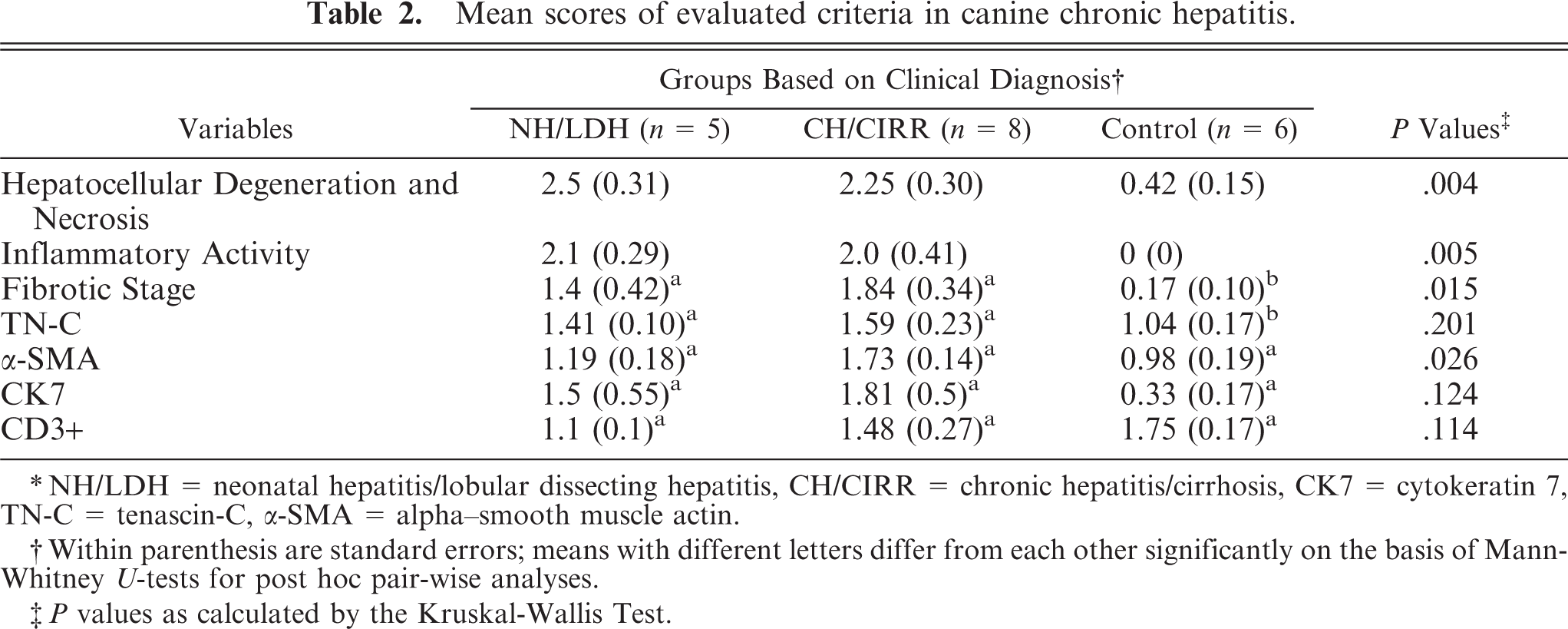
∗NH/LDH = neonatal hepatitis/lobular dissecting hepatitis, CH/CIRR = chronic hepatitis/cirrhosis, CK7 = cytokeratin 7, TN-C = tenascin-C, α-SMA = alpha–smooth muscle actin.
†Within parenthesis are standard errors; means with different letters differ from each other significantly on the basis of Mann-Whitney U-tests for post hoc pair-wise analyses.
‡P values as calculated by the Kruskal-Wallis Test.
Comparison of scores among dogs by fibrotic stage
The overall pattern and degree of hepatocellular degeneration and necrosis increased with the stage of fibrosis (Table 3). Significant (P < .05) increase was only observed in dogs with stage 2 and 3 of fibrosis compared with the control dogs. Also, inflammatory activity was significantly higher (P < .05) in these stages. Hepatic TN-C level increased concurrently with the fibrotic stage, although this increase was not significant (P > .05). The number of α-SMA+ mesenchymal cells was higher in dogs with the first and third stage of fibrosis compared with the controls and those with the second stage of fibrosis. The signal for CK7 was significantly higher (P < .05) in dogs with stage 3 of fibrosis compared with the control group. Although CK7 scores in stage 2 had high variability, the overall magnitude of CK7+ ductular proliferation was similar in the second and third stage of fibrosis. The number of CD3+ T-lymphocytes was relatively higher in stage 2 and lower in stage 1, but the difference was not significant.
Relationship among variables
Highly significant correlations (P < .001) were found between the stage of fibrosis, inflammatory activity, and hepatocellular degeneration and necrosis (Table 4). The fibrotic stage was also positively correlated significantly with all variables, except CD3. Moreover, TN-C also positively correlated not only with the stage of fibrosis but also with inflammatory activity, CK7, and α-SMA scores. Also, CK7 reactivity significantly correlated with the stage of fibrosis, inflammatory activity, hepatocellular degeneration and necrosis, and TN-C scores. CD3 scores significantly correlated with hepatocellular degeneration and necrosis (for values, see Table 4).
Correlations between evaluated criteria analyzed by the Spearman rho test.
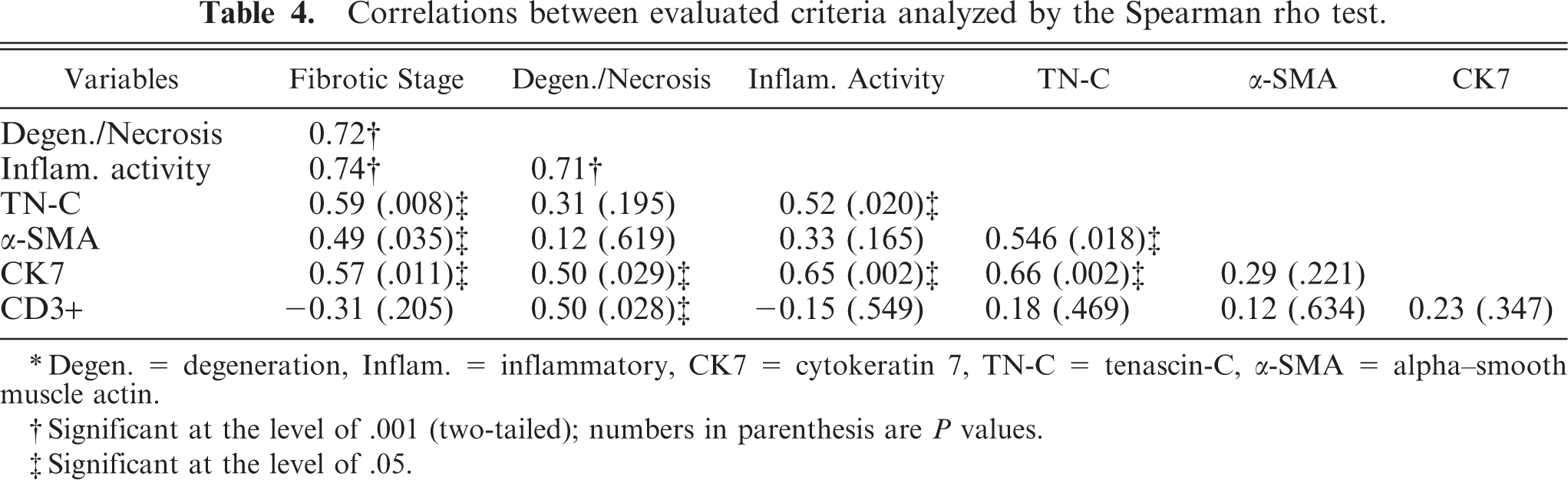
∗Degen. = degeneration, Inflam. = inflammatory, CK7 = cytokeratin 7, TN-C = tenascin-C, α-SMA = alpha–smooth muscle actin.
†Significant at the level of .001 (two-tailed); numbers in parenthesis are P values.
‡Significant at the level of .05.
Relationship between TN-C and α-SMA per location
The mean scores of TN-C and α-SMA across all dogs regardless of the group showed a statistically significant correlation (P < .05, r = 0.536, Table 4). There was a highly positive correlation (P < .001) at the perisinusoidal lining in normal dogs and in the stroma encompassing the regenerative nodules in CH/CIRR dogs. However, when the scores in various locations (sinusoids, septa/portal triads, portal-parenchymal interface, and stroma around the regenerative nodules) were summarized and analyzed, they showed nonsignificant positive correlations (r = 0.382, P > .05) (Table 5). Although not statistically significant, positive correlations were also observed in the perisinusoidal linings of the hyperplastic nodules, in areas where degenerated cells predominated, in the portal-parenchymal interface, in fibrotic septa, and in portal triads excluding blood vessels.
Localizations and mean scores of TN-C and α-SMA expressions irrespective of the stages of fibrosis or hepatitis group.
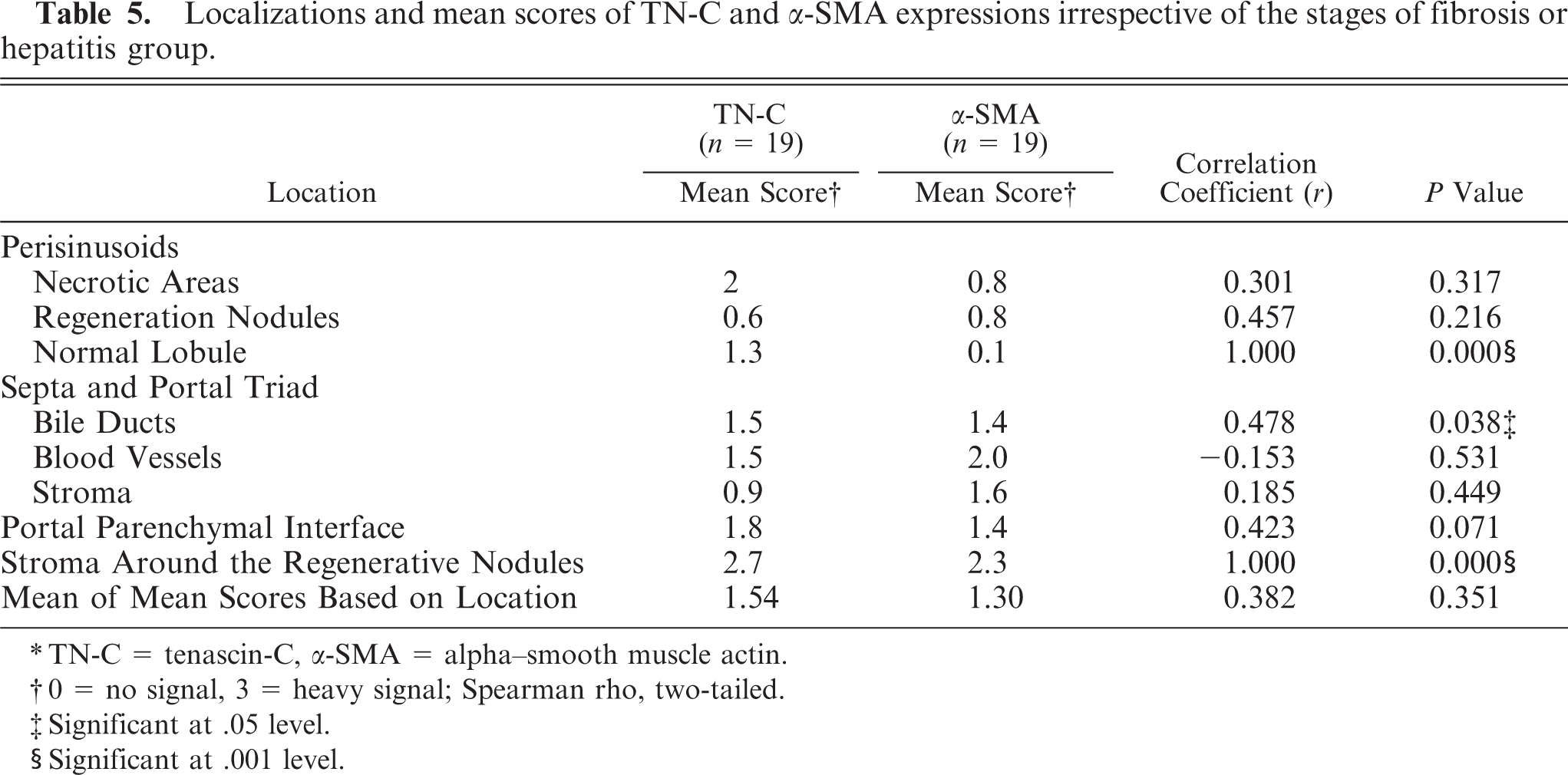
∗TN-C = tenascin-C, α-SMA = alpha–smooth muscle actin.
†0 = no signal, 3 = heavy signal; Spearman rho, two-tailed.
‡Significant at .05 level.
§Significant at .001 level.
Discussion
Histologic and immunopathologic investigations have led to remarkable progress in the understanding of chronic liver diseases in both humans and in rat models. 1, 5 However, the etiology and pathogenesis of canine chronic liver disease are still poorly understood. ECM proteins play an important role in the progression of hepatic fibrosis. 44, 51, 55 The ECM protein TN-C is expressed in normal and fibrotic livers of humans and rats. 19, 35, 48, 49, 53 TN-C serum level is reported to have a strong correlation with the progression of the fibrosis. 54 In canine hepatitis, there is lack of knowledge of the contribution of ECM proteins to the progression of hepatic fibrosis. In the present study, we defined the presence and localization of TN-C and the correlation with the stage of fibrosis, hepatocellular degeneration and necrosis, inflammatory activity, and expression of α-SMA, CK7, and CD3 in canine liver.
Hepatitis grade is indicated by the degree of necro-inflammatory activity, while the stage of hepatitis is reflected by the degree of fibrosis. 16, 30– 32, 34, 40, 42, 44, 45, 51, 55 In our study of canine chronic hepatitis, necro-inflammatory activity showed a close positive relation with fibrosis, and thus hepatitis grade increased with hepatitis stage. This does not hold true for hepatitis in general, for the included cases were chronic hepatitis with neutrophilic inflammatory reactions. Confluent or bridging patterns of hepatocellular degeneration and necrosis were predominant in the selected CH/CIRR dogs. Bridging necrosis indicates progression of the disease and a poor prognosis. 11 When CH/CIRR was compared with NH/LDH, both inflammatory activity and degeneration and necrosis showed a nonsignificant decrease of mean scores. This suggests that the NH/LDH dogs selected might have had a higher hepatitis grade. Severe inflammation has been described as a feature of NH/LDH. 3, 50
In this study, the fibrosis in NH/LDH dogs was diffusely distributed throughout the parenchyma, surrounding individual cells, or groups of hepatocytes dissecting the lobular architecture. Conversely, in CH/CIRR, fibrotic septa radiated from the portal and/or central areas into the parenchyma. In cirrhotic dogs, the architecture was disrupted by broad bridging fibrous septa and regenerative nodular hyperplasia of hepatocytes. These findings are in concordance with previous studies. 3, 7, 46, 50 Most CH/CIRR dogs showed moderate-to-severe fibrosis, while those with NH/LDH showed only moderate fibrosis (Table 2). The increase in the amount of fibrous tissue can be explained by the activation, migration, and accumulation of HSC in the parenchyma and myofibroblasts at the other locations. These activated cells deposit many ECM proteins in the inflamed areas. 17, 36 Also, activated HSC synthesize tissue inhibitors of metalloproteinase-1 and 2, which inhibit interstitial collagenase activity and thus contribute to the accumulation of ECM proteins. 5, 9, 16
In the healthy canine liver, TN-C was located in Disse's space, around the bile ducts and blood vessels, and under Glisson's capsule. This may be related to the TN-C-expressing cells, most likely HSC in Disse's space and myofibroblasts in the portal areas, walls of central veins, and, occasionally, in the parenchyma. 30, 32, 45 The localization of TN-C that we found in the normal canine liver is in agreement with that reported for man and rat. 19, 35, 48, 49, 53 In contrast to rat and man, quiescent canine HSC express α-SMA (Fig. 7). 25 During hepatic fibrogenesis, HSC in man and rat increase in number and differentiate into myofibroblast-like cells with marked expression of α-SMA. 16, 36 In the presented cases, canine stellate cells and other hepatic myofibroblasts showed dramatic increase in α-SMA expression. This means that in the dog, the presence of activated HSC is not indicated by the presence of α-SMA expression but by the increased α-SMA production, since α-SMA is expressed in healthy liver tissue.
Both TN-C and α-SMA expression increased as the stage of fibrosis increased. This may be the result of continuous activation of HSC and other myofibroblasts during hepatitis and synthesis of large amounts of ECM proteins, including TN-C. A significant relationship (P < .05) between TN-C and α-SMA was obtained when their correlation was analyzed without considering the locations. This probably indicates a concurrent upregulation of these proteins in the producing cells. However, when their correlation was analyzed per location (Table 5), the relation was nonsignificant (P > .05). The lack of significance may be the result of continuous activation of HSC and other myofibroblasts in the liver and their response to tissue injury. 45 TN-C and α-SMA positivity around the bile ducts and blood vessels confirms that myofibroblast cells are not uniquely confined to the perisinusoidal areas. These findings indicate that HSC and other myofibroblasts contribute to fibrosis in the canine liver, which is in agreement with findings in rat and man. 32, 45
The demonstration of a considerable number of CK7-positive bile ductular cells in the portal-parenchymal interface of the fibrotic livers is consistent with previous studies in man. 39, 40 A possible explanation for their presence could be cholangiocytic metaplasia of periportal hepatocytes and/or proliferation of the local progenitor cell compartment in the canals of Hering. 39, 40 The strong correlation of CK7 with TN-C may indicate the role of the latter in the proliferation and migration of the progenitor cells. 29 Ductular proliferation also increased with the stage of fibrosis in this study. Several factors may contribute to this: cytokine induction by HGF and TNF-like weak inducer of apoptosis (TWEAK) or possible compression exerted on the bile ducts by the fibrotic septa. 5, 21, 26, 42
CD3+ T-lymphocytes were not found consistently in all the groups of dogs in the current study. This can be an indication for immunologic reaction evoked by the unknown etiologic agent. Our results suggest the possibility of cell-mediated immunity in canine chronic hepatitis. 5 Interestingly, in the dogs with intense CD3 positivity, the signal for TN-C deposition was weak (Fig. 12) compared with similar areas in the same patient without CD3+ cells. This has also been reported in tumors. 31 Two mutually enhancing factors may play a role: TN-C can suppress CD3-mediated T-cell activation, 22– 24, 34, 41 while activated CD3+ T-lymphocytes induce plasminogen-dependent proteolysis of TN-C by secreting u-PA. 20 Both may have a negative impact on the progression of fibrosis.
Summarizing the relationship of parameters of hepatocellular degeneration and necrosis, one may conclude that inflammatory activity, fibrotic stage, and α-SMA showed significant differences, while the expression of TN-C, CK7, and CD3 did not differ between diagnostic groups. When dogs were reclassified on the basis of fibrosis, hepatocellular degeneration, and necrosis, inflammatory activity and CK7 expression showed significance, while TN-C, α-SMA, and CD3 did not.
Footnotes
Acknowledgements
This work was part of the master dissertation sponsored by the Netherlands Organization for International Cooperation for Higher Education. We thank Ms. Anne Marie van Ederen and Mr. Ronald Kisjes, who assisted this work directly or indirectly.
