Abstract
A 13-y-old, spayed female dog had regenerative anemia, lymphopenia, hypoalbuminemia, and elevated hepatic biochemical parameters. Liver biopsy revealed hepatic peliosis (hepatic sinusoidal angiectasis), frequently associated with perisinusoidal fibrosis. The dog was seroreactive to Bartonella antigens by indirect fluorescent antibody assays, and quantitative PCR from blood identified Bartonella vinsonii subsp. berkhoffii genotype II. The dog was euthanized 9 mo later because of acute decompensation. Autopsy revealed icteric adipose tissues, end-stage liver, and abdominal effusion. Microscopically, there was marked mixed-cell chronic hepatitis with hepatocellular loss, nodular hepatocellular regeneration, and capillary proliferation. Retrospective molecular testing documented B. koehlerae and B. rochalimae DNA in the dog’s blood at 2 or more times during liver disease progression. B. koehlerae DNA was also amplified and sequenced from the autopsy sample of liver. Our case emphasizes that Bartonella infection may be associated with hepatic peliosis and end-stage liver in dogs and expands the spectrum of Bartonella species that potentially play a role in canine hepatic diseases.
A 13-y-old, 16.9 kg, spayed female Catahoula mixed-breed dog was examined by her primary care veterinarian in Texas on 2022 May 3 (day 1), for a yearly health assessment, which included CBC, serum biochemical profile and urinalysis. The dog lived in a suburban area with another dog. In contrast to up-to-date heartworm preventives and leptospirosis vaccine, administration of flea and tick preventives had not been documented. Past and current medical history included otitis externa, and atopic dermatitis treated regularly with Apoquel (Zoetis). Otherwise, physical examination was unremarkable. CBC (Suppl. Table 1) revealed mild normocytic, hypochromic, nonregenerative anemia. Serum biochemistry abnormalities (Suppl. Table 2) included hypoalbuminemia and elevation in alkaline phosphatase (ALP), alanine aminotransferase (ALT), and aspartate aminotransferase (AST) activities. Urine specific gravity was 1.025, with unremarkable dipstick and sediment examinations. A heartworm antigen ELISA, fecal flotation, and fecal antigen testing for ova and parasites were negative (Idexx). Claro otic solution (Elanco) and prednisone at 0.6 mg/kg/day for 10 d were dispensed to treat the otitis and atopic dermatitis, respectively.
Given the nonregenerative anemia and elevated liver enzyme activities, an abdominal ultrasound was performed on day 8, which identified a mottled hyperechoic liver with an irregular capsular surface. At day 16, a CBC demonstrated worsening anemia with regeneration, and lymphopenia. The dog had hypoproteinemia, worsening hypoalbuminemia, and an elevated symmetric dimethylarginine (SDMA) concentration. ALT and ALP activities remained elevated. After unsuccessful attempts of ultrasound-guided Tru-Cut hepatic biopsy, 2 surgical biopsies were obtained. Macroscopic evaluation of the liver revealed atrophy and fibrosis affecting all lobes, and red foci. Microscopic evaluation identified many dilated, blood-filled spaces lined by variably plump endothelial cells and affecting the 3 zones (Fig. 1; Suppl. Figs. 1A, 2). The dilated vessels were frequently associated with fibrosis in the perisinusoidal space (space of Disse), and hepatocytes were absent, atrophied, or had prominent cytoplasmic vacuolation (Fig. 1). These findings were consistent with hepatic peliosis (hepatic sinusoidal angiectasis); hemangiosarcoma was considered unlikely given the gross appearance of the liver and the lack of pleomorphism of the endothelial cells. An increase of α-smooth muscle actin (α-SMA)–positive cells, suggestive of activated hepatic stellate cells, was visible within the perisinusoidal fibrosis (Fig. 1C; Suppl. Table 3). Other findings included moderate extramedullary hematopoiesis (Suppl. Fig. 3A), moderate ductular reaction, uncommon porto-portal bridging fibrosis, hepatocellular glycogen accumulation (confirmed by periodic acid–Schiff stain), and dilated lymphatic vessels in the subcapsular region and portal tracts. Kupffer cells were hyperplastic, and frequently contained intracytoplasmic hemosiderin (confirmed by Perls Prussian blue stain; Suppl. Fig. 3B). Retrospectively, the Warthin–Starry stain was inconclusive, and Bartonella spp. were not identified with indirect immunofluorescence using an in-house polyclonal anti-Bartonella antibody or chromogenic RNAscope in situ hybridization (ISH) using a Bartonella 23S rRNA probe (Advanced Cell Diagnostics). Ursodiol (14.9 mg/kg q24h for 40 d), amoxicillin–clavulanate (14.9 mg/kg q12h for 7 d), and gabapentin (17.9 mg/kg q12h for 7 d) were dispensed at the time of hospital discharge.
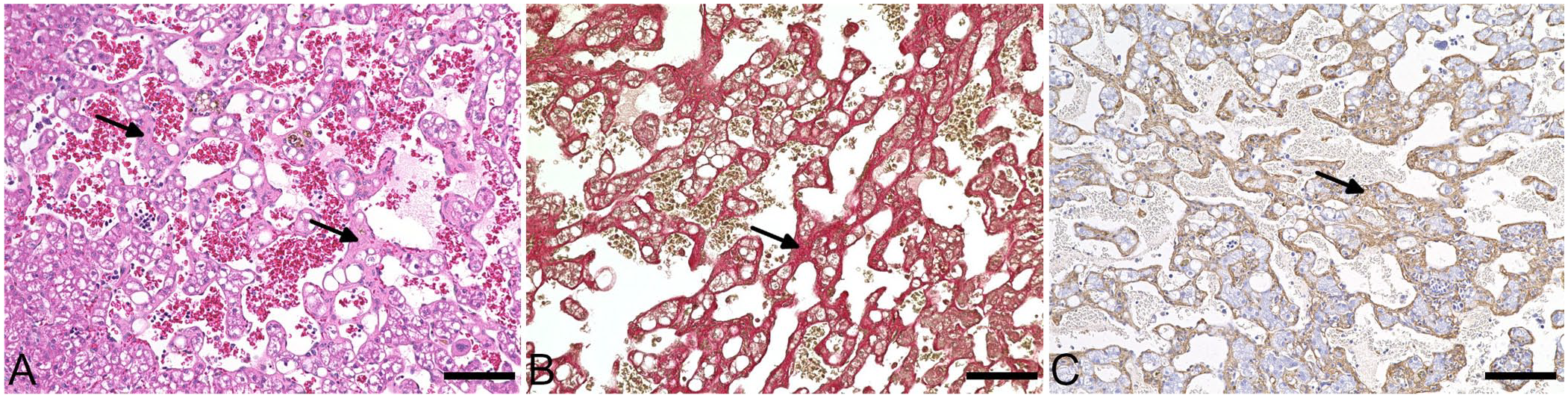
Peliosis hepatis (hepatic sinusoidal angiectasis) associated with perisinusoidal fibrosis in a dog. There are many dilated blood-filled spaces, and the perisinusoidal spaces (arrows) within the vascular lesions are expanded by fibrosis and an increase of α-smooth muscle actin (α-SMA)–positive cells.
Given a prior association of B. henselae (Bh) with peliosis hepatis in dogs and human patients,8,9 Bartonella quantitative real-time PCR (qPCR) targeting the 16S-23S rRNA gene intergenic spacer (ITS) region was performed (Galaxy Diagnostics). DNA was extracted from EDTA whole blood (QIAsymphony SP; Qiagen). B. vinsonii subsp. berkhoffii (Bvb) DNA was PCR-amplified and sequenced (Genewiz). Independent repeat testing by our laboratory confirmed the presence of Bvb genotype II (Table 1; Suppl. Table 4). Similar testing was performed from the formalin-fixed, paraffin-embedded (FFPE) liver biopsies; a Bartonella PCR amplicon was obtained but DNA sequencing was not successful. Doxycycline (6 mg/kg q12h) was prescribed, and after 3 wk, enrofloxacin (6 mg/kg q12h) was added. The presence of anti-Bartonella antibodies was investigated at day 44 by indirect fluorescent antibody assays (IFAs), as described previously. 11 The dog was seroreactive to Bvb genotype II (reciprocal titer of 1:64), B. koehlerae (Bk; 1:128), and B. quintana (1:64). The dog was non-seroreactive to Bh and Bvb genotype I (<1:64). Testing for additional vector-borne pathogens by IFA (Rickettsia rickettsii, Ehrlichia canis, Babesia canis) and ELISA (Babesia gibsoni) was performed at the Vector Borne Disease Diagnostic Laboratory, North Carolina State University (Raleigh, NC, USA); the dog was non-seroreactive for all of these pathogens.
PCR results from specimens collected at various times from a dog with a histologic diagnosis of peliosis hepatis at day 38 and end-stage liver disease at day 292.

By the end of July (day 82), there was marked improvement in the anemia and there were no other hematologic abnormalities. Hepatic biochemical parameters remained elevated. Other biochemical abnormalities included hypoalbuminemia, hyperglobulinemia, and elevated SDMA. The dog was non-seroreactive to Bvb (genotype I and II) and Bh but continued to be seroreactive to Bk (1:256) and B. quintana (1:64). In September (day 129), after completion of the antibiotic course, the dog was more active, according to the owner, and had gained 1 kg (body weight of 17.9 kg). Mild anemia and hypoalbuminemia persisted. ALT, ALP, and AST activities remained elevated.
Despite remaining subclinical, similar hematologic and biochemical abnormalities were observed in December (day 213). At a scheduled re-evaluation in January 2023 (day 270), the owner reported hyporexia and mild lethargy in the dog. ALT and ALP activities had increased. Doxycycline (6.3 mg/kg q12h) and Denamarin (Nutramax) were prescribed. Three weeks later (day 292), the dog was examined at an emergency hospital due to anorexia of 1-wk duration, hindlimb weakness, and acute-onset vomiting. The dog was alert, responsive, and weighed 17.7 kg; mild dehydration was the only abnormal physical examination finding. Ultrasound documented abdominal and scant pericardial fluid. Hematologic abnormalities included normocytic, normochromic non-regenerative anemia; neutrophilia; monocytosis; and thrombocytopenia. Biochemical abnormalities included hypoalbuminemia, elevated ALT and ALP activities, and azotemia. Intra-abdominal bleeding was suspected; the owner elected euthanasia.
The main findings at autopsy were icteric subcutaneous and visceral adipose tissues, end-stage liver (Suppl. Fig. 4A), a small amount of abdominal effusion, and a pale heart. Multifocal hemorrhage was suspected in the right brain hemisphere. At the microscopic evaluation of the liver, there was marked chronic hepatitis with hepatocellular loss, nodular hepatocellular regeneration, ductular reaction, and porto-portal bridging fibrosis (Suppl. Fig. 1B). Necrosis (single cell or small foci) was infrequent. Hepatic infiltrates, portal and occasionally panlobular, were composed of neutrophils, lymphocytes, plasma cells, and macrophages with frequent intracytoplasmic hemosiderin. There was multifocal capillary proliferation, especially within inflammatory foci (Suppl. Fig. 4B). Using the same techniques mentioned previously, Bartonella spp. were not visualized. Extramedullary hematopoiesis and lymphatic dilation were still present. Hepatocytes frequently contained small lipid vacuoles. Copper accumulation was not observed with rhodanine stain. Acute, mild, multifocal hemorrhage was present in the meninges, and, to a lesser extent, in the perivascular spaces. Other lesions included myocardial fatty infiltration, chronic glomerulitis and tubulointerstitial nephritis, white pulp depletion and diffuse extramedullary hematopoiesis in the spleen, chronic sinus erythrocytosis in the mesenteric lymph node, mild multifocal myofiber necrosis with histiocytic infiltrates in the diaphragm, a thyroid adenoma, multifocal adrenocortical nodular hyperplasia, mild chronic lymphoplasmacytic cystitis with acute hemorrhage, mild plasmacytic enteritis, and rare foci of pulmonary mineralization. No histologic lesions were found in the esophagus, pancreas, and trachea.
Conventional PCR was performed retrospectively on blood specimens that had previously tested negative by qPCR and a FFPE liver sample from autopsy, using Bartonella genus and Bk-specific 16S-23S rRNA ITS primers (Table 1; Suppl. Table 4). B. rochalimae and Bk DNA were amplified and sequenced at days 82 and 230. In addition to Bk, B. alsatica–like bacterium DNA was found at day 270. Bk DNA was also sequenced from the end-stage liver sample.
Hepatic peliosis is characterized by randomly distributed, dilated, blood-filled spaces. It can be an incidental autopsy finding or associated with hepatic dysfunction or hemorrhage from liver rupture. In humans, the cause can be noninfectious (e.g., anabolic steroids, oral contraceptives) or infectious. Although Bh has been identified as the main causative agent of hepatic peliosis in people with acquired immunodeficiency syndrome (AIDS), 9 Bvb or B. elizabethae infection has been documented in single case reports,5,17 emphasizing the possibility that several Bartonella species can induce hepatic peliosis. Molecular detection of Bvb from blood and lesional tissue has also been reported in a 13-y-old immunocompromised patient with hepatic epithelioid hemangioendothelioma. 3
In dogs, molecular evidence of Bh has been described in one case of hepatic peliosis. 8 We retrieved no cases of Bvb-infected dog with peliosis hepatis in a search of PubMed, Web of Science, and CAB Direct, using search terms “Bartonella” and “dog”, suggesting that this condition has not been reported previously. Four genotypes of Bvb have been characterized by DNA sequence differences within the 16S-23S ITS region, and Pap31 gene. Consistent with our case, genotype II is, to date, the most frequent genotype in dogs and humans from the United States. Dogs and wild canids are considered the primary reservoir hosts; the mode of transmission among canids is unknown. Clinical manifestations of Bvb infections in dogs include endocarditis and/or myocarditis, 2 idiopathic thoracic effusions, 4 and granulomatous lymphadenitis. 14 Molecular evidence of Bvb genotype II have also been reported in vasoproliferative lesions, including bacillary angiomatosis in a therapeutically immunosuppressed dog (for presumptive idiopathic immune-mediated pancytopenia) 19 and perivascular wall tumors in 2 dogs. 1
Diagnosis of bartonellosis remains challenging due to the fastidious and slow-growing nature of Bartonella spp., and low and intermittent bacteremia. Despite a low sensitivity of IFA, the bacteremic dog in our study was seroreactive to at least one Bartonella antigen both times she was tested. Given the fact that cross-reactivity can occur across Bartonella spp. antigens, 13 molecular testing is required to define the species. DNA of different species (Bvb, Bk, B. rochalimae, and B. alsatica–like) was amplified and sequenced at various times from this dog’s blood and liver. Nevertheless, these results do not establish whether the presence of Bartonella spp. was a cause or contributor to the progression of liver disease documented in our case. Testing for other common vector-borne pathogens was negative.
Coinfection is technically difficult to document with PCR targeting the genus level. Amplification of all species may not be possible due to either differences in bacteremia level among species, or selective amplification of one species over the other(s) due to differences in sequence homologies with the oligonucleotides used as primers. 12 In addition, if both species are amplified at the same time, amplicon DNA sequence quality could be impaired due to overlap of sequences between amplified species. We could not determine whether undiagnosed coinfection or sequential infection could have happened, or the exact role of each species in the disease progression. We retrieved no cases of dog for which molecular evidence of 4 Bartonella species was reported in a search of PubMed, Web of Science, and CAB Direct, using search terms “Bartonella” and “dog”, suggesting that this condition has not been reported previously. In our opinion, the occurrence of infection with multiple Bartonella species at the same time or consecutively is likely underestimated in both animals and humans. Bk DNA was amplified at 2 times 6 mo apart, indicating potential long-term bacteremia. Bk has been reported in people with cardiovascular or neurologic symptoms. Both B. rochalimae and Bk have been associated with endocarditis in dogs. 16 These 2 species have not yet been described in dogs with liver disease, in contrast to Bh and B. clarridgeiae. 6
Despite amplification of Bartonella DNA from the FFPE liver biopsies and autopsy liver, bacteria were not visualized with Warthin–Starry stain, immunofluorescence, or chromogenic RNAscope ISH. Similar negative results have been documented in both dogs and humans, likely due to higher sensitivity of molecular techniques compared to visualization techniques. Microscopically, hepatic peliosis in our case was associated with perisinusoidal fibrosis, an increase of α-SMA–positive cells, and subsequent progression to an end-stage liver. These pathologic associations have been reported in humans.7,18,21 Liver sinusoidal endothelial cells have a key role in fibrosis initiation and progression, by directly secreting extracellular matrix and regulating the activation of hepatic stellate cells. 10
Most people with Bartonella-associated vasoproliferative lesions are immunocompromised because of AIDS, solid organ transplantation, cancer, or immunosuppressive treatment. Immunosenescence, immune dysregulation, and/or drug-induced immunodeficiency may have been predisposing factors in acquiring infection with multiple Bartonella species in our case, a senior dog being treated for atopy. There was depletion of the splenic white pulp, indicating inadequate immune defense. Dogs experimentally infected with Bvb had diminished antigen presentation, which might promote acquisition of additional bacterial infections. 15 Experimentally, cats infected with one Bh strain and subsequently treated became bacteremic when challenged with a different Bh strain or Bartonella species, suggesting that prior infection with Bvb would not have afforded protection against subsequent infection with other Bartonella species documented in our case. 20
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387241310868 – Supplemental material for Peliosis hepatis and hepatic fibrosis in a dog infected with multiple Bartonella species
Supplemental material, sj-pdf-1-vdi-10.1177_10406387241310868 for Peliosis hepatis and hepatic fibrosis in a dog infected with multiple Bartonella species by Cynthia Robveille, Cody Atkinson, Jerry Cowart, Ricardo G. Maggi, Neelesh Narurkar and Edward B. Breitschwerdt in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Emily Kingston and Chance Liedig for their assistance in generating the molecular results in the Intracellular Pathogens Research Laboratory, North Carolina State University (NCSU). We also thank the NCSU CVM histology laboratory for the histologic processing of the samples and the α-SMA immunohistochemistry.
Declaration of conflicting interests
In conjunction with Dr. S. Sontakke and North Carolina State University, Edward B. Breitschwerdt holds US Patent 7,115,385 Media and Methods for Cultivation of Microorganisms, which was issued on 2006 Oct 3. He is a co-founder, shareholder, and Chief Scientific Officer for Galaxy Diagnostics, a company that provides advanced diagnostic testing for the detection of Bartonella spp. infections. Ricardo G. Maggi is a co-founder and the Chief Technical Officer for Galaxy Diagnostics Inc. All other authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was supported in part by the State of North Carolina, and donations to the Bartonella/Vector Borne Diseases Research Fund.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
