Abstract
Assessment of the clinical severity, pathogenesis, and prognosis of canine chronic liver disease poses significant challenges to clinicians and pathologists, relating in part to a lack of standardized terminology and assessment methods and also to a lack of understanding of the pathogenesis of chronic liver disease in the dog. This study graded the severity of necroinflammatory activity in chronic liver disease in dogs using a modification of Ishak’s grading scheme for human chronic liver disease and examined the association of grade score with hepatocellular apoptosis, regeneration, nitric oxide synthase isoform expression, copper and iron accumulation, and indicators of oxidative stress. Formalin-fixed, paraffin-embedded hematoxylin and eosin (HE)–stained liver biopsies from 45 dogs with chronic liver disease and 55 healthy control dogs were graded for various morphologic components of liver injury and response. The cumulative score for grade of necroinflammatory activity was strongly and significantly correlated with immunoreactive labels for hepatocellular proliferation (Ki-67); apoptosis (cleaved caspase-3); inducible nitric oxide synthase (iNOS) in lobular, portal, and septal stromal cells; endothelial nitric oxide synthase (eNOS) in hepatocytes and lobular, portal, and septal stromal cells; and total stainable hepatic iron. A weaker significant correlation was found between grade and accumulation of hepatocellular copper. No significant correlation was found between grade and immunoreactivity for malondialdehyde-protein adducts. These results document a method for grading of the severity of necroinflammatory disease in canine liver biopsies and show an association with increased iNOS and eNOS expression.
Chronic liver disease involves ongoing injury to hepatocytes and prolonged regenerative, inflammatory, and profibrotic responses that affect liver function and recovery. In chronic liver disease of human patients and associated rodent models of human disease, much is known regarding the various contributors to chronic liver injury and repair, and the various etiologies share some common mechanistic factors. Parenchymal cell loss can occur as a result of both hepatocellular necrosis and apoptosis; hepatocellular apoptosis contributes to the loss of hepatocytes in alcoholic hepatitis, nonalcoholic steatohepatitis, chronic viral hepatitis B and C, severe cholestatic liver disease, copper accumulation disorders, graft rejection, and autoimmune hepatitis. 3,18,19,22,36,45 Apoptosis has also been demonstrated to contribute to the progression of COMMD1 mutation-associated hepatitis in dogs. 17 Replication of differentiated hepatocytes increases after parenchymal loss or resection, but replication can be impaired in some conditions. 16,61 Proliferation of nonparenchymal cells, including cholangiocytes and less differentiated oval cells (especially in rodents), can occur when hepatocellular loss is severe or when replication of mature hepatocytes is delayed or blocked. 16
A common contributor to hepatocellular injury is oxidative stress, an imbalance between levels of antioxidant molecules and reactive oxygen species (ROS) (including singlet oxygen molecules, superoxide radicals, hydrogen peroxide, and hydroxyl radicals) that are highly reactive with various organic molecules 7 and can direct hepatocytes toward necrosis or apoptosis, 9,47 as demonstrated in studies of alcoholic and nonalcoholic steatohepatitis, 40,57 various other toxic insults to the liver, 1 chronic hepatitis C virus infection, 29 and type I autoimmune hepatitis. 48 Oxidative stress has been implicated as a contributor to canine chronic copper toxicosis of Bedlington Terriers, chronic extrahepatic cholestasis, and chronic hepatitis in dogs. 60 Copper is well recognized as a contributor to oxidative stress in the liver. A transitional metal ion, copper accumulates as the primary mediator of chronic liver disease in human Wilson disease, 41 in the Long-Evans Cinnamon rat, 20 and in Bedlington Terriers with COMMD1 mutations and likely other canine breeds, 17,23,24,26,54,62 but also accumulates in humans with alcoholic liver disease 51 and viral hepatitis with cirrhosis, 39 as well as in advanced stages of primary biliary cirrhosis. 32 Similarly, the transitional metal ion iron is associated with chronic liver disease as a result of primary iron overload in hereditary hemochromatosis in humans 46 but also accumulates in chronic liver disease associated with hepatitis C virus 59 and alcohol-related liver disease. 50
The effects of nitric oxide (NO) on hepatocytes are complex and dependent on physiologic context; by itself, NO has been demonstrated to be antiapoptotic for hepatocytes, 11,25,33 –35,38,63 but with concurrent oxidative stress, NO potentiates hepatocellular necrosis through the evolution of peroxynitrite, a highly reactive oxygen species that can damage a broad variety of organic molecules producing nitrotyrosine-protein adducts. 11 Nitric oxide is produced by 2 constitutive isoforms of the enzyme nitric oxide synthase (endothelial or type 3 NOS/eNOS and neuronal or type 1 NOS/nNOS) and 1 inducible isoform (iNOS or type 2 NOS). There is general agreement in the literature that both iNOS and eNOS are upregulated in various cell populations of the liver in chronic hepatopathies of human patients, although there is disagreement as to the microanatomic tissue compartments affected. 2,21,42,44
Most cases of chronic necroinflammatory liver disease in the dog are idiopathic, whereas most chronic liver diseases in humans have identifiable causes. In humans, the severity of liver disease can be graded in liver biopsies using scoring systems that rate necroinflammatory and reparative responses. 4,5,15,28,55,56 Such scoring systems have been used extensively for correlative studies investigating factors involved in the pathogenesis of chronic liver disease and biomarkers for chronic liver disease. Detailed descriptive classifications of the more common chronic liver diseases of dogs have been refined in recent years, 53 but there remains a need for standard ways to assess severity of the disease entities using criteria that do not require a known cause. The objective of this study was to examine the contribution of apoptosis, hepatocellular proliferation, nitric oxide synthase, and oxidative stress to the pathogenesis of canine chronic liver disease, in which inflammation and necrosis were assessed by a histopathological scoring system adapted from one originally developed by Ishak et al. 28
Materials and Methods
Case Selection
Pathology reports of canine liver biopsies submitted to the diagnostic pathology services of the Department of Pathobiology and the Animal Health Laboratory, University of Guelph, Guelph, Ontario, Canada (1998–2006), and Histovet Surgical Pathology, Guelph, Ontario, Canada (2001–2006), were reviewed. A total of 45 cases were selected based on the following inclusion criteria: (1) a compatible morphologic diagnosis of chronic liver disease (chronic hepatitis, chronic-active hepatitis, hepatic fibrosis/nodular regeneration/cirrhosis, hepatic postnecrotic collapse and nodular regeneration, lobular dissecting hepatitis) and description including the presence of 1 or more compatible morphologic findings (hepatic fibrosis, parenchymal collapse, single-cell necrosis/apoptosis, piecemeal necrosis, mononuclear cell infiltrates, nodular regeneration, biliary proliferation), (2) adequate biopsy size (for the purposes of this study, only surgical incisional biopsies were included, with a minimum of 10 portal tracts), and (3) adequate tissue fixation. Criteria for exclusion from the study included the presence of other forms of liver disease (including acute hepatic zonal or massive necrosis typical of primary acute toxic or hypoxic liver injury, acute cholangiohepatitis, extrahepatic cholestasis, hepatic abscessation, chronic passive congestion and periacinar fibrosis secondary to cardiac failure, congenital vascular anomalies, and hepatic neoplasia); however, lesions with extensive neutrophil involvement were not excluded if they met inclusion criteria for chronicity without any indication of abscessation. Needle biopsy specimens were excluded due to size limitations (many were less than 2 cm in length, fragmented, and included fewer than 10 portal tracts below the minimum guidelines suggested in human biopsy studies to avoid inaccuracies of assessment 49,52,58 ). Case dogs had an average age of 7.6 years and a median age of 7.7 years. Purebred dogs of 22 different breeds made up 88.9% of case dogs; the remaining 11.1% were of mixed breed. Intact males (46.7%) and intact females (44.4%) were nearly equally represented, as were spayed females (4.4%) and neutered males (4.5%) (Table 1).
Signalment Statistics for Case (n = 45) and Control (n = 55) Study Animals.

Control surgical biopsies were obtained from 55 clinically normal preconditioned dogs of varying ages and breeds with normal hematology and serum biochemistry profiles (including serum alanine aminotransferase [ALT] and γ-glutamyltransferase [GGT]) used for surgical teaching exercises approved by the University of Guelph Animal Care Committee and in accordance with the guidelines of the Canadian Council on Animal Care, at the Ontario Veterinary College, University of Guelph (Table 1). Intraoperative guillotine biopsies of the liver were collected, immediately fixed for 24 hours in 10% neutral buffered formalin, and routinely processed for histopathology. Control specimens were selected based on (1) absence of histologic evidence of significant primary or secondary liver disease in biopsy specimens, (2) adequate biopsy size (for the purposes of this study, only surgical incisional biopsies were included, with a minimum of 10 portal tracts), and (3) adequate tissue fixation. Control dogs had an average age of 3.5 years and a median age of 3.0 years. Purebred dogs of 3 different breeds made up 12.7% of control dogs, with the remaining 87.3% being of mixed breed. Intact males (45.5%) and intact females (38.2%) again made up the majority, with fewer spayed females (5.5%) and neutered males (11.0%).
Case and control tissue samples were fixed in 10% neutral buffered formalin for 24 hours prior to routine processing and embedding in paraffin. HE-stained sections were prepared for the case (n = 45) and control (n = 55) biopsies using standard methods by the histotechnology laboratory of the Animal Health Laboratory, University of Guelph, Guelph, Ontario, Canada. Additional 6-μ serial tissue sections were mounted on charged glass slides for immunohistochemical staining.
Histologic Scoring
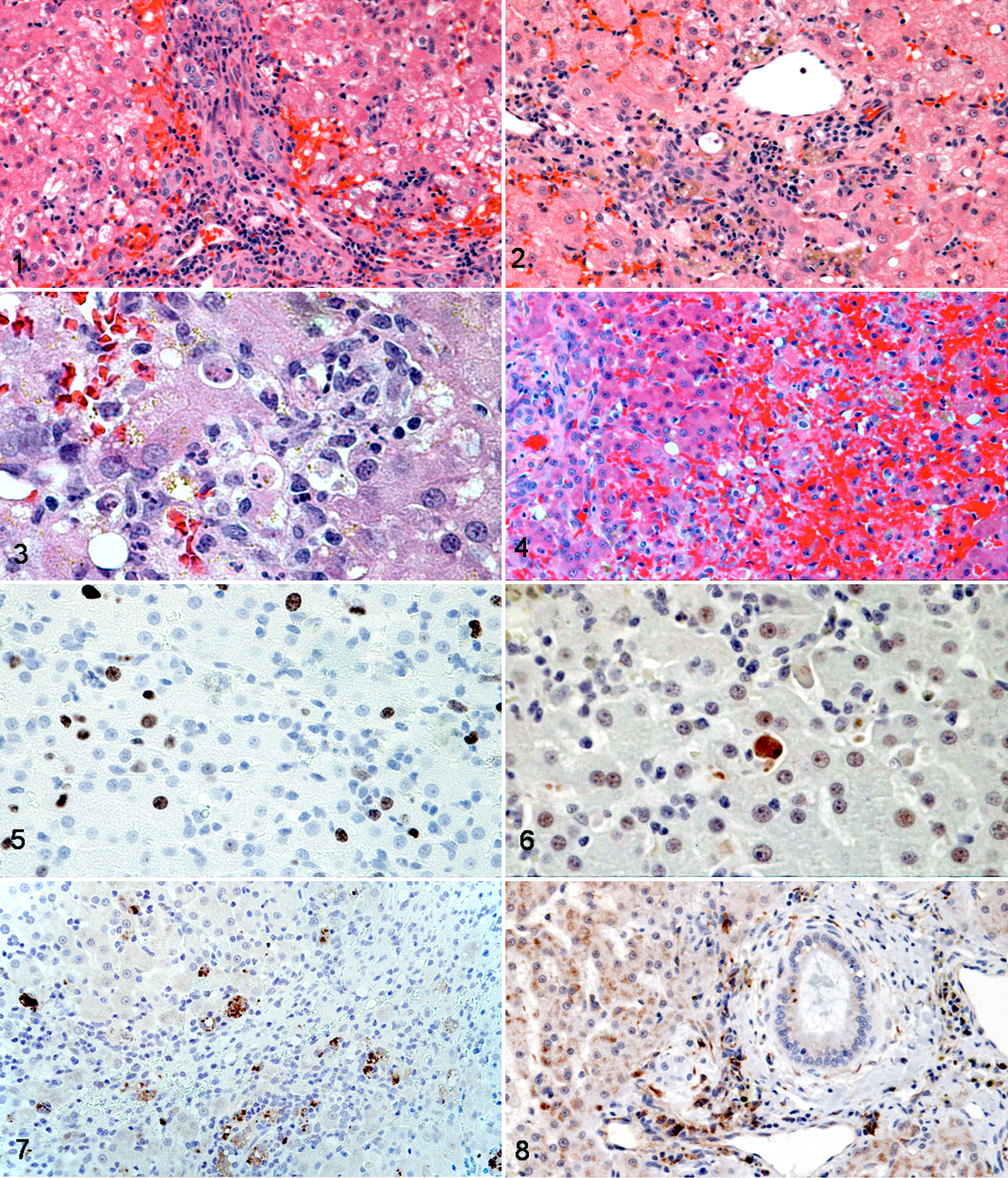
For the purpose of scoring the specimens, a modification of the scheme developed by Ishak et al 28 was used (Table 2). Liver biopsy specimens were graded according to the degree of necroinflammatory activity present and staged according to the extent and pattern of fibrosis. HE-stained histology slides from cases and controls were examined blindly and assigned 4 individual semiquantitative scores for the presence and severity of (1) portal inflammation: defined as predominantly mononuclear cell inflammation (lymphocytes, plasma cells, and macrophages) within the fibrous stroma of the portal tract 30 (Fig. 1); (2) periportal or periseptal interface hepatitis (“piecemeal necrosis”): defined as predominantly mononuclear cell inflammation and cell death of individual or small groups of hepatocytes at the interface of parenchyma and connective tissue of the intact or scarred portal tract 30 (Fig. 2); (3) focal lytic necrosis, apoptosis, and focal inflammation: defined as cell death of random individual or small focal groups of hepatocytes throughout the lobule, with associated inflammation 30 (Fig. 3); and (4) confluent necrosis: defined as larger confluent areas of adjacent dead hepatocytes or a site where a group of hepatocytes has previously undergone liquefactive necrosis and removal, forming areas of parenchymal collapse. These areas are often centrilobular, affecting zone 3. Confluent necrosis linking vascular structures and portal zones is termed bridging necrosis and may link terminal hepatic venules (central-central bridging) or terminal hepatic venules to portal tracts (central-portal bridging). Panacinar or panlobular necrosis is the result of more extensive confluent necrosis, involving zones 2 and 1 as well as 3, and destroying entire acini 30 (Fig. 4).
Grade and Stage Scoring Criteria for Chronic Hepatitis (Modified From Ishak et al 28 ).

These 4 scores are summed to produce a final cumulative score for the grade of necroinflammatory activity present. HE-stained slides from all specimens were examined and scored no fewer than 3 times, twice by 1 pathologist (A.R.V.) (for purposes of determining repeatability; repeat scores were performed within 2 weeks of initial scoring) and once by a second pathologist (M.J.S.) (for purposes of determining agreement). Prior to examination, both pathologists were given written definitions of the specific criteria evaluated and the thresholds for each score. In those circumstances in which scores for each criterion were identical between pathologists, these scores were used as a final score for purposes of comparison with other criteria. In those circumstances in which scores differed between pathologists, the specimens were reexamined and assigned scores by consensus.
The distribution and extent of hepatic fibrosis was also scored (Table 2) to determine the stage of chronic liver disease as part of a larger study; correlations between scored stage and immunoreactivity for Ki-67, cleaved caspase-3 (CC-3), malondialdehyde adducts, and NOS isoforms, as well as accumulation of transitional metals, were absent, lacked statistical significance, or were of lesser significance than those seen with scored grade and are not discussed here.
Mitotic Index
The hepatocellular mitotic index was determined as the cumulative number of mitotic figures counted in hepatocytes in 10 random high-power (400×) fields. Random fields were identified using randomly generated coordinates and were excluded if less than 75% of the visible area was composed of hepatic parenchyma.
Immunohistochemistry
Six-micron unstained serial sections from all formalin-fixed, paraffin-embedded case and control livers were mounted on charged glass slides for immunohistochemical evaluation. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide. The details of each primary antibody used, antigen retrieval and protein block procedures, secondary antibodies, chromogens, and positive and negative controls are listed in Table 3. Slides were counterstained with Meyer’s hematoxylin.
Details of Immunohistochemical Reagents.
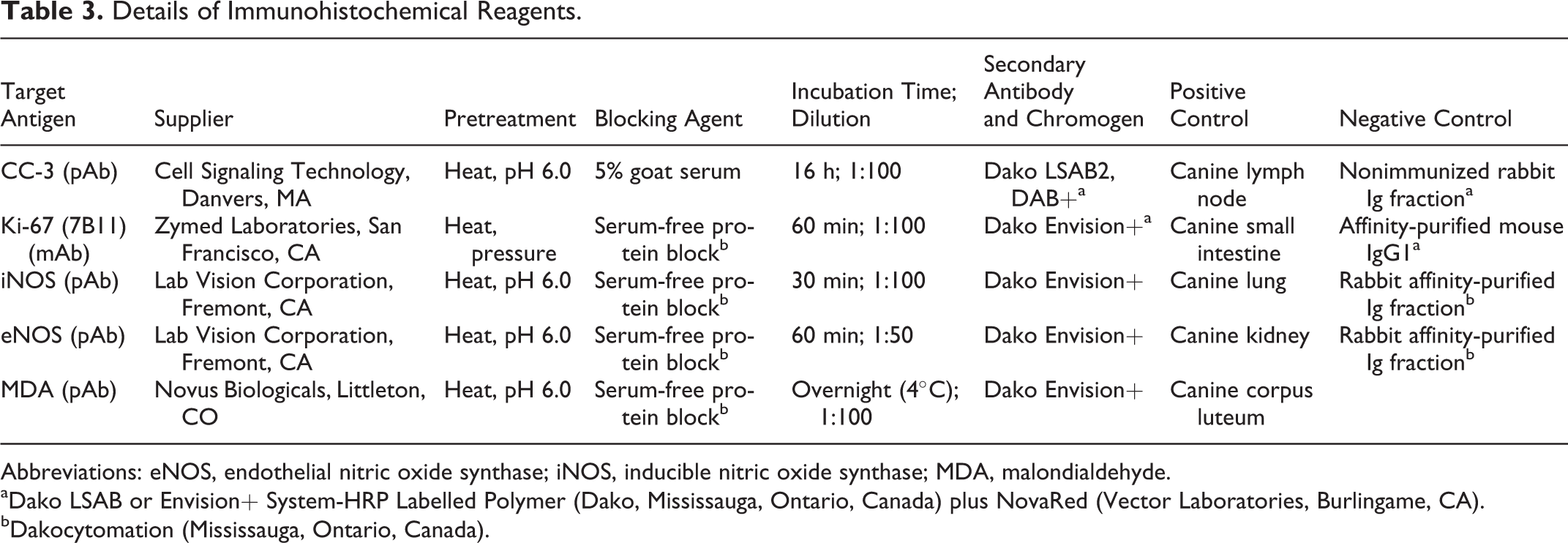
Abbreviations: eNOS, endothelial nitric oxide synthase; iNOS, inducible nitric oxide synthase; MDA, malondialdehyde.
aDako LSAB or Envision+ System-HRP Labelled Polymer (Dako, Mississauga, Ontario, Canada) plus NovaRed (Vector Laboratories, Burlingame, CA).
bDakocytomation (Mississauga, Ontario, Canada).
CC-3 and Ki-67 immunoreactivity were used to assess apoptotic and proliferative indices, respectively. Stained hepatocytes were identified and counted in 10 randomly chosen high-power fields (random fields were identified using randomly generated coordinates and were excluded if less than 75% of the visible area was composed of hepatic parenchyma).
The percentage of hepatocytes with cytoplasmic staining of each NOS isoform was scored on a 0 to 3 scale as described in Table 4. Degrees of lobular and portal/septal stromal cell NOS isoform immunoreactivity were separately scored on a 0 to 2 scale (Table 5). Scores for parenchymal (0–3), lobular stromal cell (0–2), and portal/septal stromal cell (0–2) immunoreactivity were then summed to produce a total NOS isoform staining score (0–7).
Scoring of Parenchymal Immunoreactivity for Nitric Oxide Synthase (NOS) Isoforms.

Scoring of Stromal Immunoreactivity for NOS Isoforms.

Abbreviations: eNOS, endothelial nitric oxide synthase; iNOS, inducible nitric oxide synthase.
A marker of oxidative stress, parenchymal malondialdehyde (MDA)–protein adduct immunoreactivity was reported as the percentage of hepatocytes staining for MDA adducts (Table 6). The distribution of MDA staining was also evaluated, as zonal (including which zones affected), random focally extensive (affecting large areas of parenchymal cells but without zonal distribution), or diffuse.
Scoring Immunoreactive Malondialdehyde (MDA) Adducts in the Parenchymal Cell Compartment.

Histochemistry: Copper and Iron
Six-micron histologic sections were stained for iron content and distribution using Perl’s Prussian blue stain 31 and for copper content and distribution by rhodanine stain, 8 following standard protocols. The Perl’s stain for iron only demonstrates ferric forms of iron but has been found to correlate well with tissue iron content using semiquantitative methods of evaluation; iron content and distribution were evaluated semiquantitatively as for evaluation of human liver biopsies with chronic liver disease. 6 Perl’s-stained sections were examined and assigned 3 semiquantitative scores based on the presence of positive staining in hepatocytes, Kupffer cells, and portal tracts; these were then modified by specific coefficients to produce weighted scores, 6 then summed to produce a final score (Table 7). The rhodanine stain for copper detects copper at tissue levels above 400 ppm 43 and has been shown to correlate strongly with actual tissue levels within the liver. 27 Copper staining was limited to hepatocytes; the proportion of positively stained cells throughout the biopsy was estimated and scored (Table 8), and the zonal distribution of staining was noted.
Semiquantitative Scoring of Hepatic Iron Content (Adapted From Brissot et al 6 ).

Parenchymal Hepatocellular Copper Scoring System.

Statistical Analysis
All data collected from examination of slides were analyzed using the SAS 9.1 software package (SAS Institute, Inc, Cary, NC). Numerical scores for grading and staging were considered categorical data, requiring nonparametric analysis. Repeatability was calculated as a κ value by standard methods 37 ; agreement was determined using a weighted κ statistic.
Correlations were made between grading scores and immunohistochemical and histochemical scores by calculating a Spearman correlation coefficient (SCC). In those tests where exact probability (P) values could not be determined, exact P values were estimated using Monte Carlo modeling, extrapolating the data set to 1 million samples to achieve the closest approximation; Monte Carlo P values were calculated with the StatDistrib software package (William Sears, University of Guelph, 2001), using sums of squares, mean squares, and degrees of freedom as determined using SAS. P values less than .05 were considered significant. For each criterion examined, the distribution of data points relative to case or control status was examined using the Mann-Whitney-Wilcoxon test (MWW). P values less than .05 were considered to indicate a significant divergence from the null hypothesis (that the distribution of data points is random with respect to case/control specimen origin) with respect to a specific criterion.
Relationships involving nominal variables (Ki-67 and CC-3 index) were examined by generating χ2 tables, with likelihood χ2 values greater than 3.8416 considered to have a significant deviation from the null hypothesis. Coefficients of correlation were established by calculating a size-adjusted contingency coefficient. In those tests where exact P values could not be determined, the exact P values were estimated using Monte Carlo modeling, extrapolating the data set to 1 million samples to achieve the closest approximation. P values less than .05 were considered significant.
Multiple linear regression using a negative binomial model was performed on a subset of the data (NOS isoform immuno-reactivity, CC-3 index, and grade of disease) to examine the relative component each criterion plays in the detected correlations; χ2 values greater than 3.8416 were considered to represent significant relationships between the criteria in question. P values less than .05 were considered significant.
Graphical representations of data were generated using Stata 9.1 (StataCorp, College Station, TX).
Results
Most control biopsies (47/55) had summative scores of 0 of 18 for the grade of necrosis and inflammation present (Fig. 9). Five control specimens scored 1 of 18, 2 specimens scored 2 of 18, and 1 specimen scored 3 of 18, due to minimal quantities of portal inflammation (7/55 specimens scored as 1), focal necrosis (4/55 specimens scored as 1), and/or confluent necrosis (1/55 specimens scored as 1). The summative scores of necroinflammatory activity for case biopsies ranged from 0 of 18 to 14 of 18 (Fig. 9). The cumulative grade score had high interobserver agreement (κ = 0.83; 95% confidence interval [CI], 0.75–0.92) and intraobserver repeatability (κ = 0.82; 95% CI, 0.74–0.87).
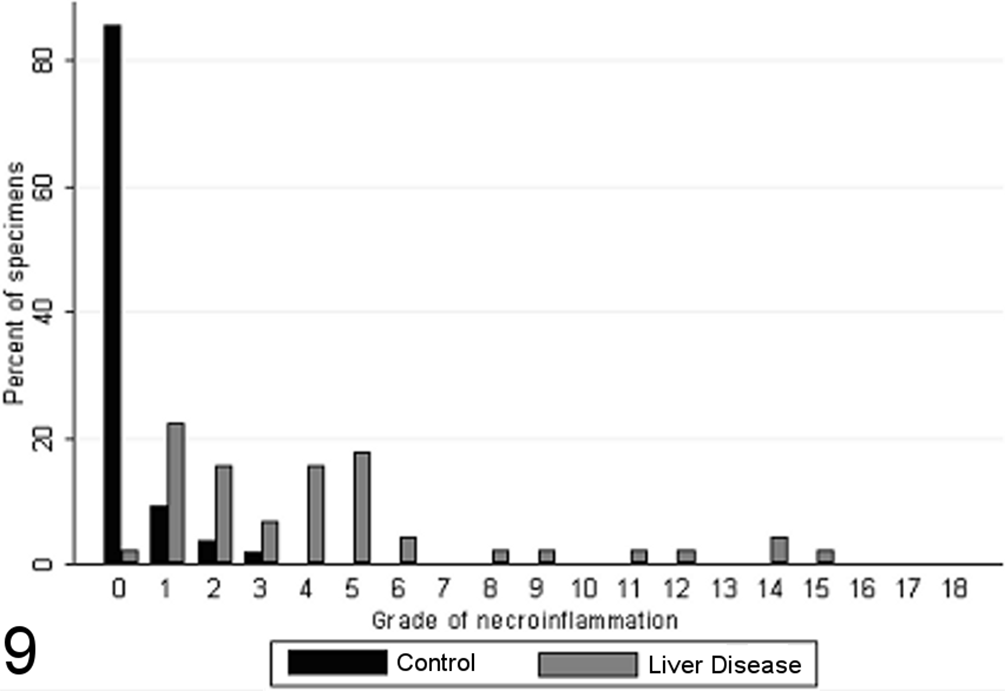
Grade of necroinflammatory activity present in case (n = 45) and control (n = 55) canine liver biopsy specimens.
All specimens from healthy control animals contained between 0 and 1 mitotic figures per ten 400× fields, while specimens from case animals exhibited wide variation in mitotic index, with a trend toward higher mitotic indices; 57% of liver specimens from case dogs had 0 to 1 mitotic figures per ten 400× fields, while the remaining 43% of case dogs had 3 to 10 mitotic figures per ten 400× fields (Fig. 10). There was a significant difference in the distribution of mitotic indices between the case and control specimens (MWW p<0.0001), and a strong significant positive relationship between mitotic index and cumulative grade score (SCC=0.57, p<0.0001).
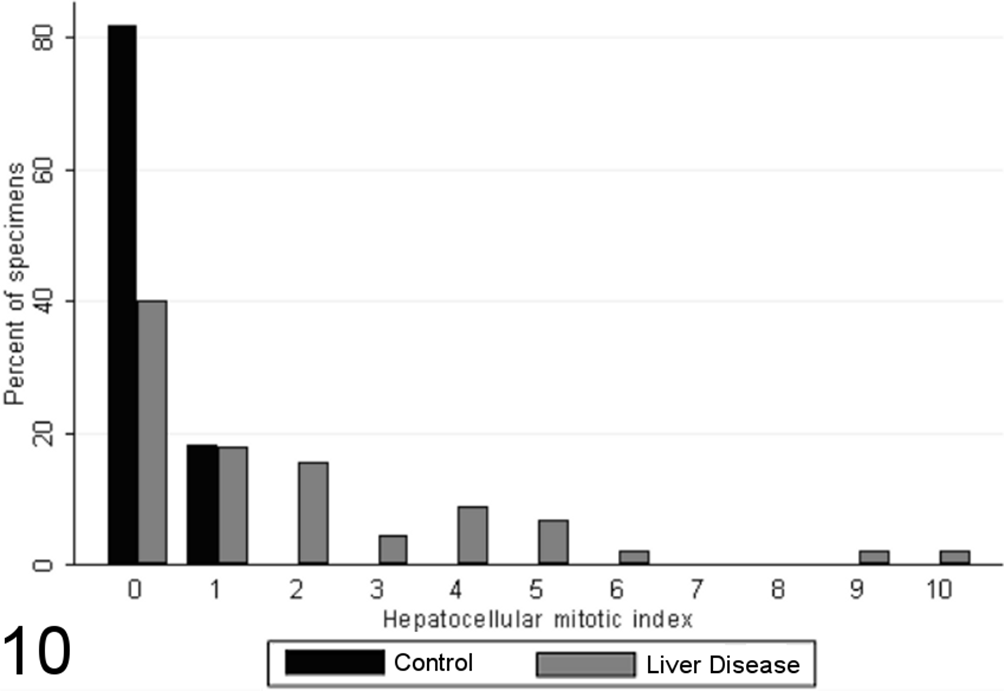
Hepatocellular mitotic index (mitotic figures per 10 random high-powered fields) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
Similarly, there was a significant difference in the distribution of Ki-67 indices between the case and control specimens (P < .0001, MWW), and the Ki-67 index was found to correlate strongly with cumulative grade score (SCC = 0.57; P < .0001), although a proportion of high-grade lesions had relatively low proliferative indices. Of control dogs, 61.8% had no Ki-67–positive hepatocytes per ten 400× fields, and all had no more than 5, while 53% of liver specimens from case dogs had more than 5 Ki-67–positive hepatocytes per ten 400× fields (Fig. 5), up to a maximum counted of 321 positive cells per ten 400× fields (Fig. 11).
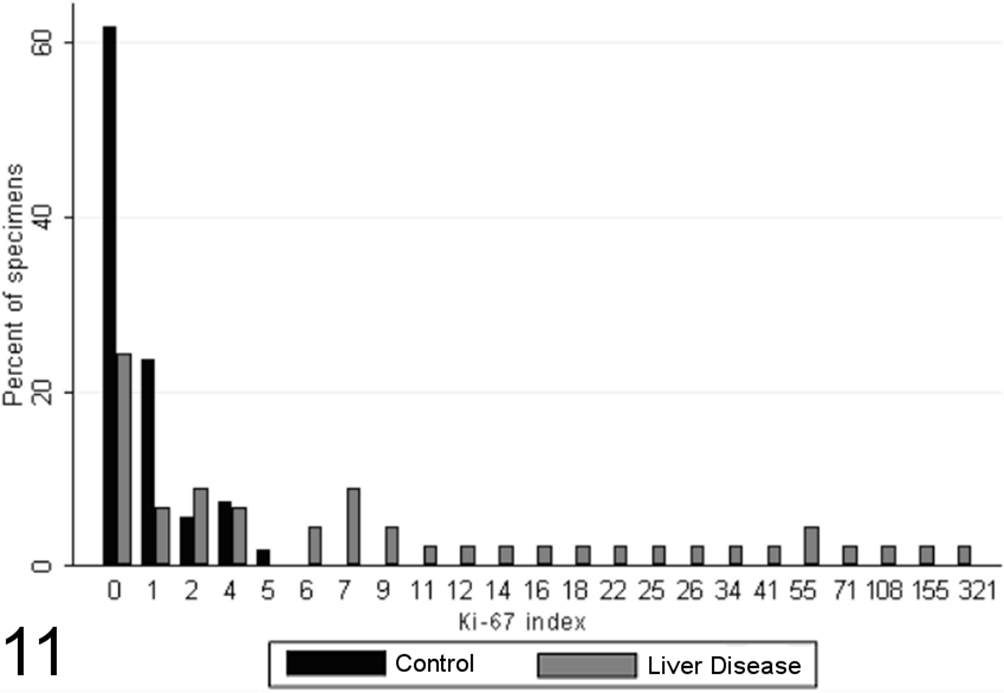
Ki-67 indices (immunoreactive cells per 10 random high-powered fields) for case (n = 45) and control (n = 55) canine liver biopsy specimens.
No hepatocytes with detectable CC-3 were found in 78% of biopsies from healthy control animals, with the remaining 22% having fewer than 6 positive cells per 10 high-power fields (Fig. 6). There was significantly increased staining in animals with chronic liver disease (P = .0009, MWW) and a significant positive relationship between apoptotic index and cumulative grade score (SCC = 0.51; P < .0001). Liver specimens from case dogs exhibited a broad variation in apoptotic indices, varying from no detectable stained cells up to a maximum index of 17 stained hepatocytes per 10 high-power fields (Fig. 12).
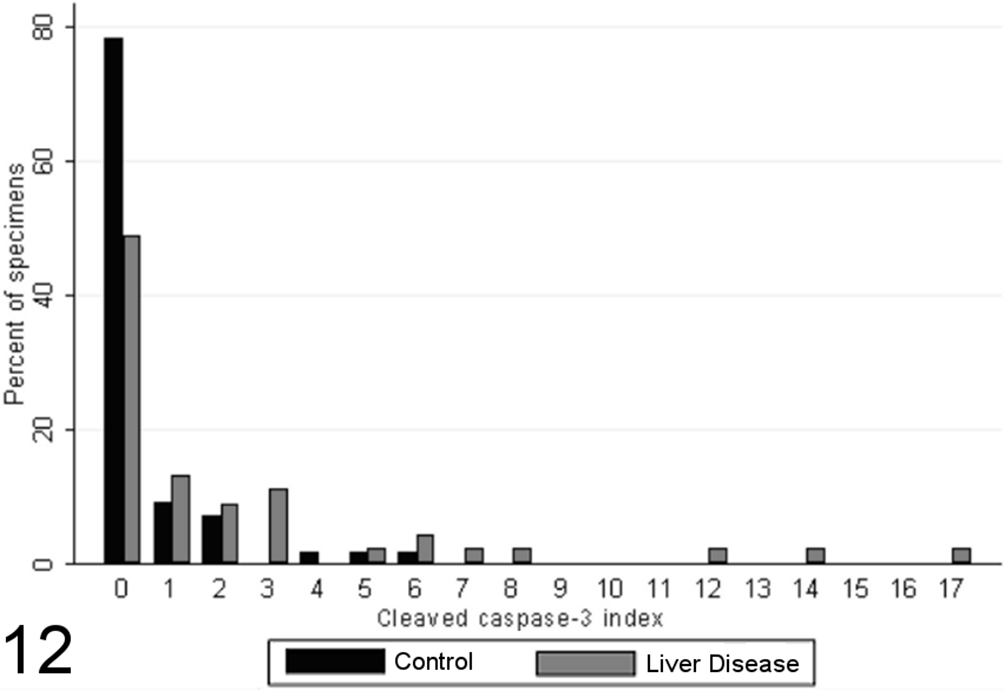
Cleaved caspase-3 index (immunoreactive cells/10 random high-powered fields) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
In most liver biopsies from both control and case dogs, MDA adduct immunoreactivity was largely limited to fewer than 10% of hepatocytes, as individual or small clusters in lobules or adjacent to portal tracts or central veins. Both groups had similar distributions of hepatocellular MDA adduct scores, with few specimens exhibiting an absence of staining (Fig. 13). There was no significant difference in the proportion of MDA adduct–positive hepatocytes between specimens from case and control animals (P = .1005, MWW) or any significant correlations noted between grade score and the score for MDA adduct immunoreactivity (P > .05). However, a strong, significant correlation was noted between the presence of a diffuse pattern of hepatocellular MDA adduct immunoreactivity and the grade of necrosis and inflammation present (SCC = 0.57; P = .03).
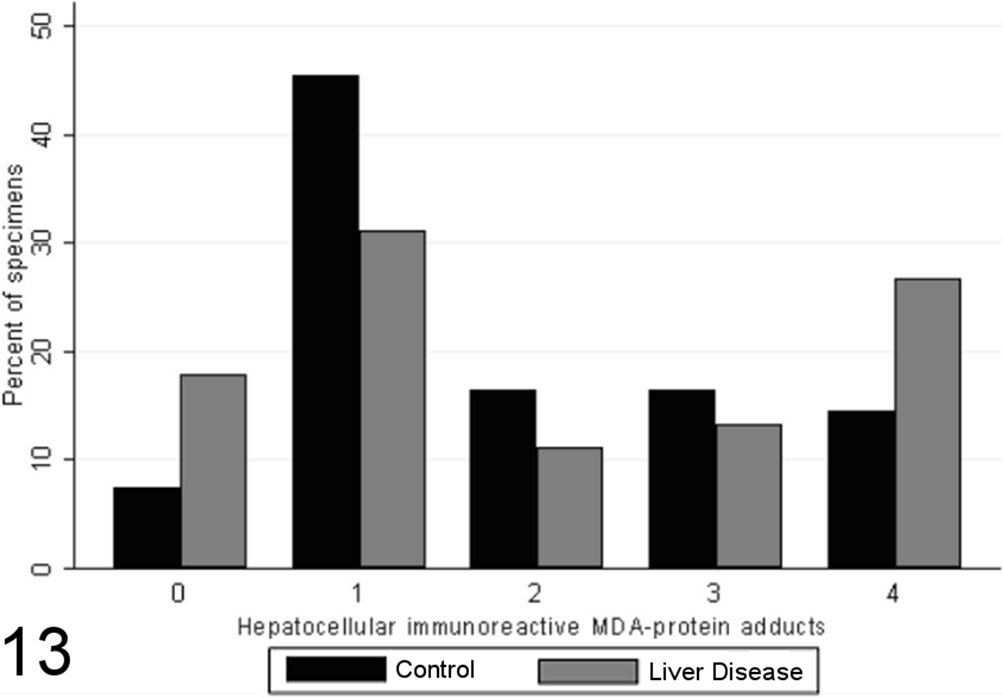
Scores for hepatocellular immunoreactive malondialdehyde (MDA)–protein adducts (0–4) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
In both case and control specimens, copper accumulation was limited to the cytoplasm of hepatocytes; 72% of liver specimens from control dogs and 42% of liver specimens from control dogs had no detectable copper staining (Fig. 14). A significant difference was noted in the distribution of hepatocellular copper scores between biopsies from case and control animals (P = .0011, MWW), while a significant weak positive correlation (SCC = 0.39; P < .05) was identified between semiquantitative copper scores and the grade of inflammation and necrosis present within liver biopsies. No significant correlations were noted between the score of immunoreactive MDA and semiquantitative measurement of copper.
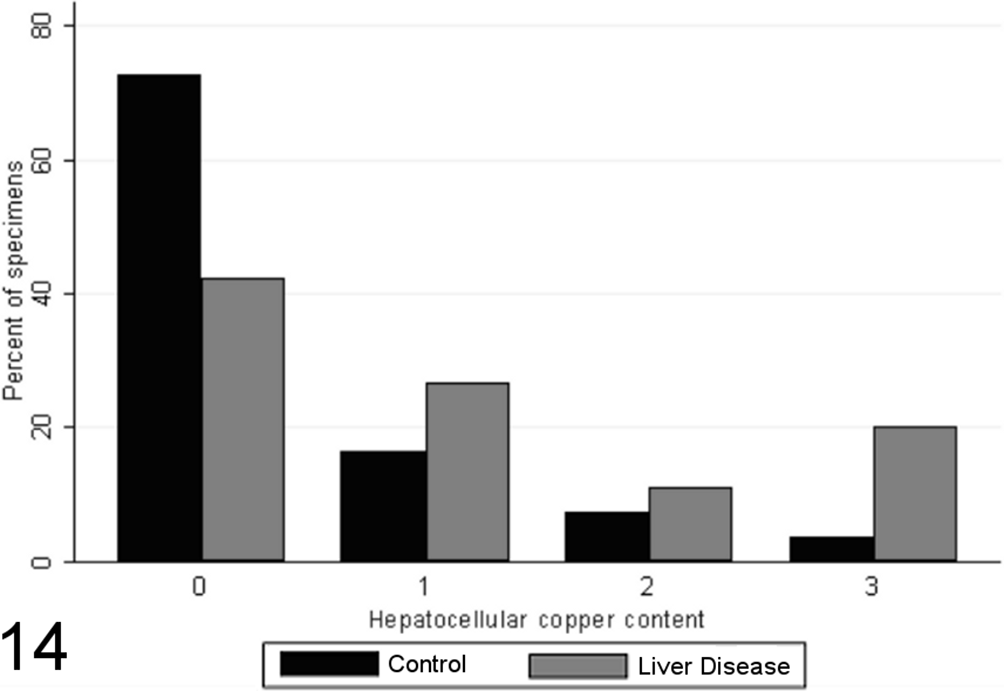
Scores for hepatocellular copper content (0–3) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
Total scores of hepatic iron staining varied significantly between specimens from case and control animals (P < .00001, MWW), with little iron in control liver specimens, increased amounts with chronic disease (Fig. 15), and a positive correlation with the grade of necrosis and inflammation (SCC = 0.61; P < .0001). Significantly more stainable iron was observed within hepatocytes, Kupffer cells, and portal/septal stromal connective tissue (P < .00001, MWW) in chronic liver disease, with significant correlations with the grade of necrosis and inflammation present (Spearman correlation coefficients of 0.57 for hepatocellular iron, 0.55 for iron in Kupffer cells, and 0.63 for iron in portal/septal stromal tissue; P < .0001). However, no significant correlations were noted between semiquantitative measurement of total hepatic iron and the score of immunoreactive MDA-protein adducts.
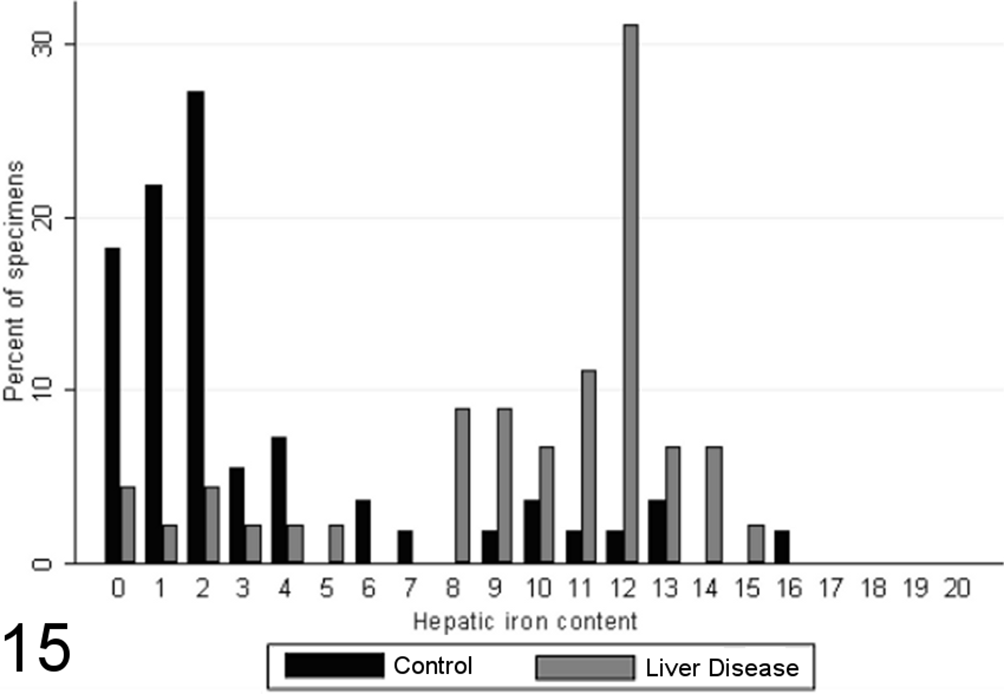
Cumulative scores for hepatic iron content (0–20) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
Most specimens from healthy control animals contained little or no iNOS immunoreactivity within any of the tissue compartments examined, whereas there was increased total iNOS staining in dogs with chronic liver disease in comparison to healthy controls (P < .00001, MWW; Figs. 16 –19). While hepatocellular staining for iNOS did not vary significantly between case and control specimens (with little immunoreactivity in either group; P = .8847, MWW), iNOS immunoreactivity was elevated in case specimens relative to controls within the stromal cells of the lobules, portal tracts, and fibrous septae (P < .00001, MWW; Fig. 7). Total, lobular stromal, and portal/septal stromal iNOS scores correlated strongly and significantly with the grade of inflammation and necrosis present (SCCs of 0.64, 0.64, and 0.62, respectively; P < .0001). A weak positive correlation (SCC = 0.38; P < .0001) was present between immunoreactivity for CC-3 and total iNOS. Multiple linear modeling was performed to further examine the possible interrelationships between CC-3 immunostaining, the grade of necroinflammation present, and iNOS immunostaining in each tissue compartment (Table 9), and χ2 values indicated that the correlations noted between iNOS immunostaining and CC-3 immunostaining are actually a reflection of the relationship noted previously between the CC-3 index and the grade of disease present.
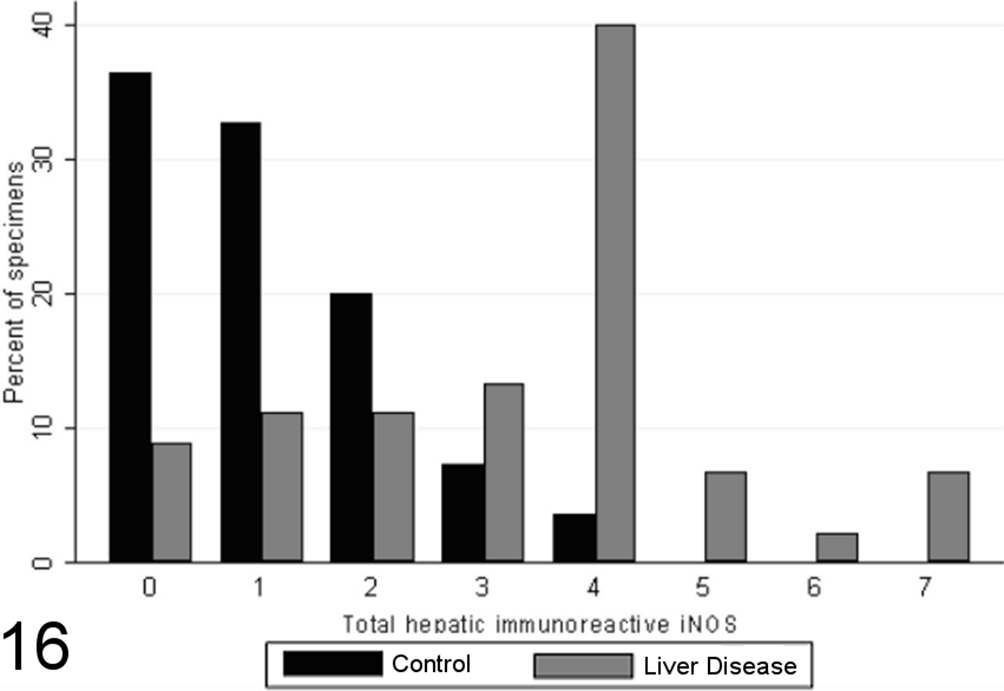
Scores for total hepatic inducible nitric oxide synthase (iNOS) immunoreactivity (0–7) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
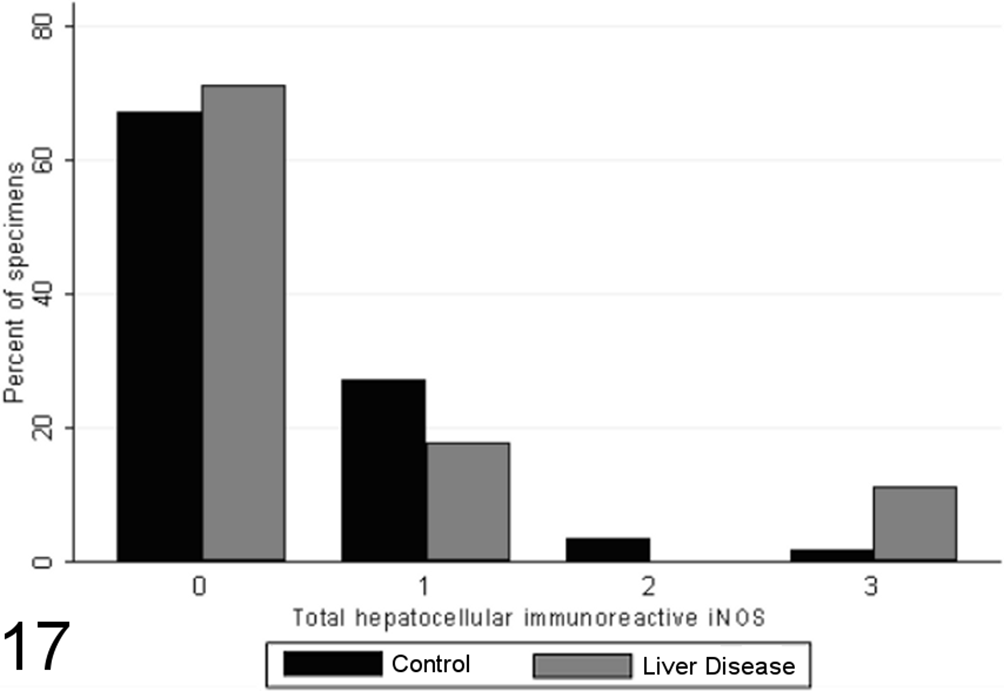
Scores for hepatocellular inducible nitric oxide synthase (iNOS) immunoreactivity (0–3) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
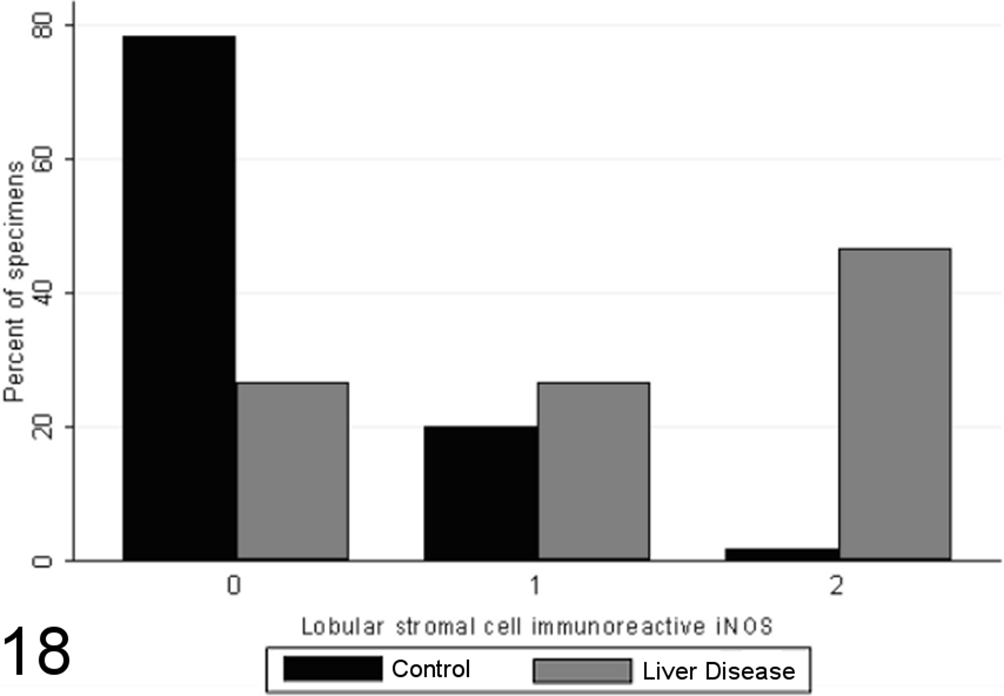
Scores for lobular stromal cell inducible nitric oxide synthase (iNOS) immunoreactivity (0–2) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
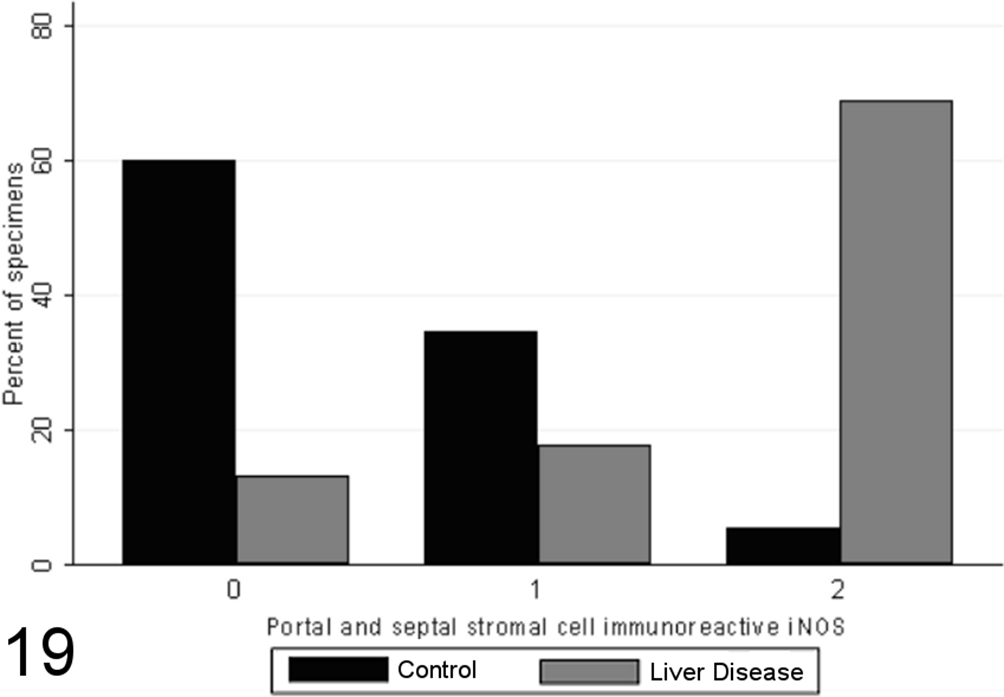
Scores for portal and septal stromal cell inducible nitric oxide synthase (iNOS) immunoreactivity (0–2) in case (n = 45) and control (n = 55) canine liver biopsy specimens.
Relationship χ2 Values for Multiple Linear Regressions Between Summed Grade of Necroinflammation and iNOS and eNOS Immunoreactivity in Different Tissue Compartments With Respect to Cleaved Caspase-3 Index (Number of Immunoreactive Cells per 10 Random High-Power Fields) in Case (n = 45) and Control (n = 55) Canine Liver Biopsy Specimens.
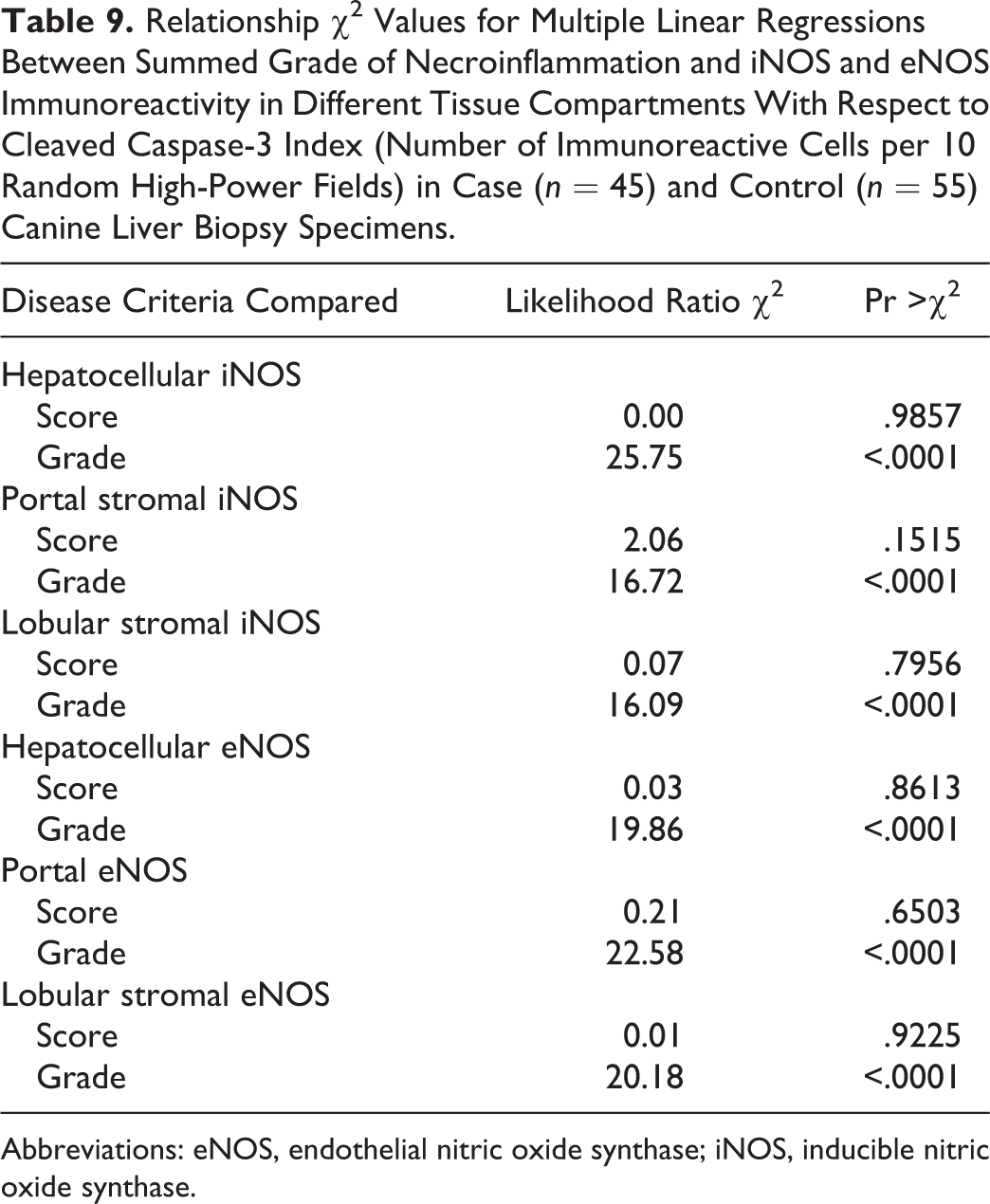
Abbreviations: eNOS, endothelial nitric oxide synthase; iNOS, inducible nitric oxide synthase.
Total scores of eNOS immunoreactivity varied significantly between specimens from case and control animals (P < .0001, MWW), with increased expression with chronic disease and correlation with the grade of necrosis and inflammation (SCC = 0.64; P < .0001). Specimens from healthy control animals contained eNOS immunoreactivity within a limited proportion of hepatocytes (usually few than 30%, typically Rappaport zone 1 hepatocytes), while in control specimens, eNOS immunoreactivity tended toward a panlobular immunoreactivity, involving a greater proportion of hepatocytes (Figs. 20 –23), the extent of which significantly correlated with the grade of necrosis and inflammation (SCC = 0.57; P < .0001). eNOS immunoreactivity was absent or low within the stromal cells of the lobules, portal tracts, and fibrous septae of control specimens and was significantly elevated in case specimens (P < .00001, MWW) (Fig. 8). Lobular stromal and portal/septal stromal eNOS scores correlated strongly and significantly with the grade of inflammation and necrosis present (SCCs of 0.60 and 0.55, respectively; P < .0001). Only a weak positive correlation (SCC = 0.27) was present between immunoreactivity for CC-3 and total eNOS; as with iNOS, this correlation is likely a reflection of the relationship noted previously between the CC-3 index and the grade of disease present, rather than a direct relationship. Multiple linear modeling was performed to further examine the possible interrelationships between CC-3 immunostaining, the grade of necroinflammation present, and eNOS immunostaining in each tissue compartment (Table 9), and χ2 values indicated that the correlations noted between iNOS immunostaining and CC-3 immunostaining are actually a reflection of the relationship noted previously between the CC-3 index and the grade of disease present.
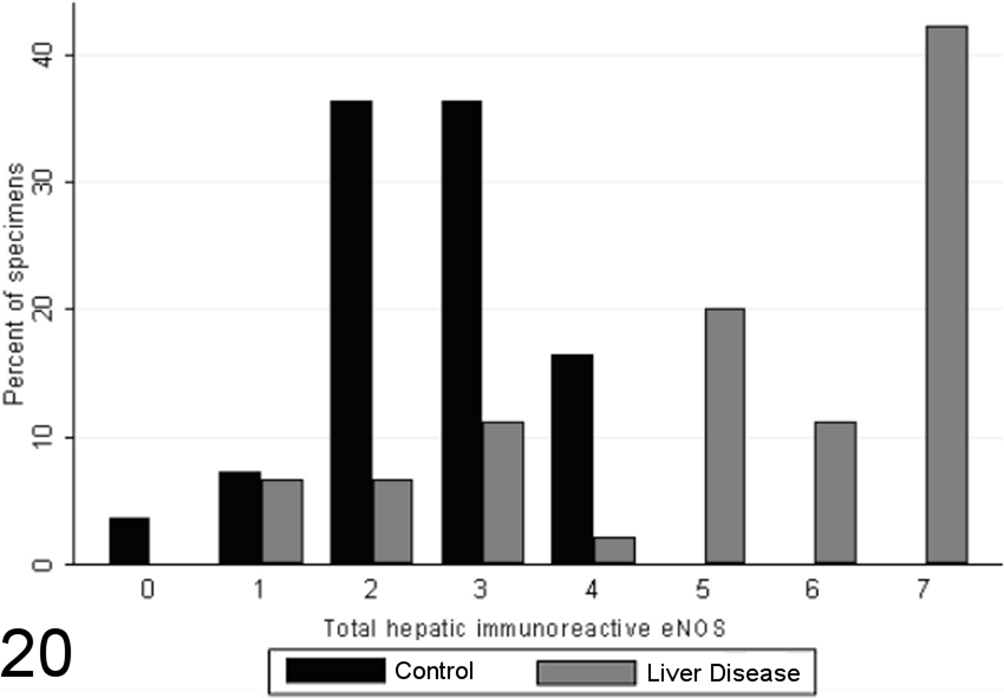
Scores for total hepatic endothelial nitric oxide synthase (eNOS) expression in case (n = 45) and control (n = 55) canine liver biopsy specimens.
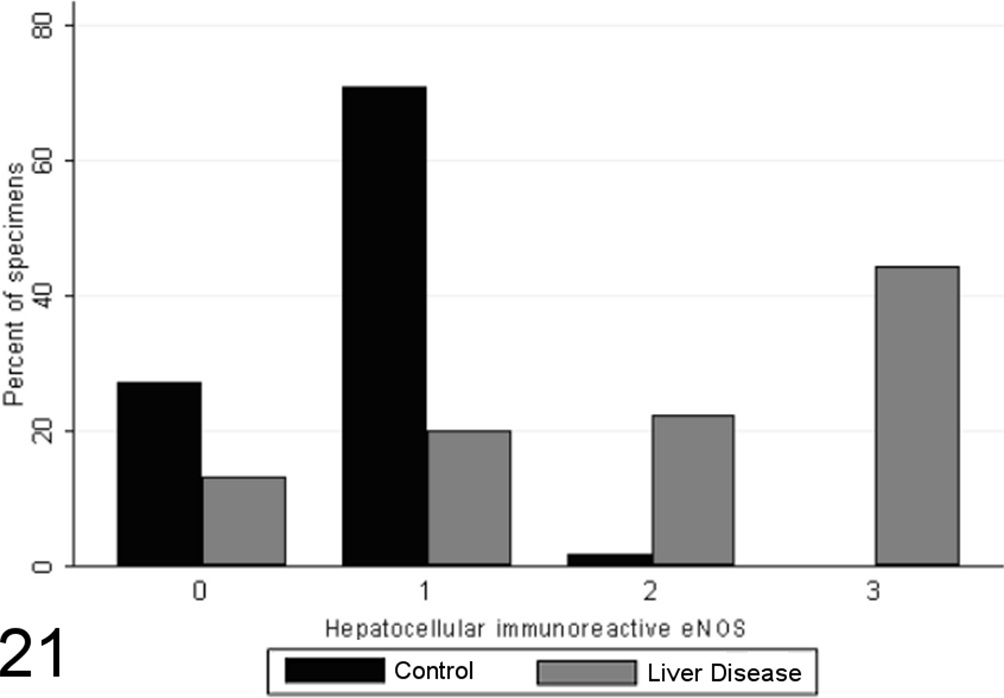
Scores for hepatocellular endothelial nitric oxide synthase (eNOS) expression in case (n = 45) and control (n = 55) canine liver biopsy specimens.
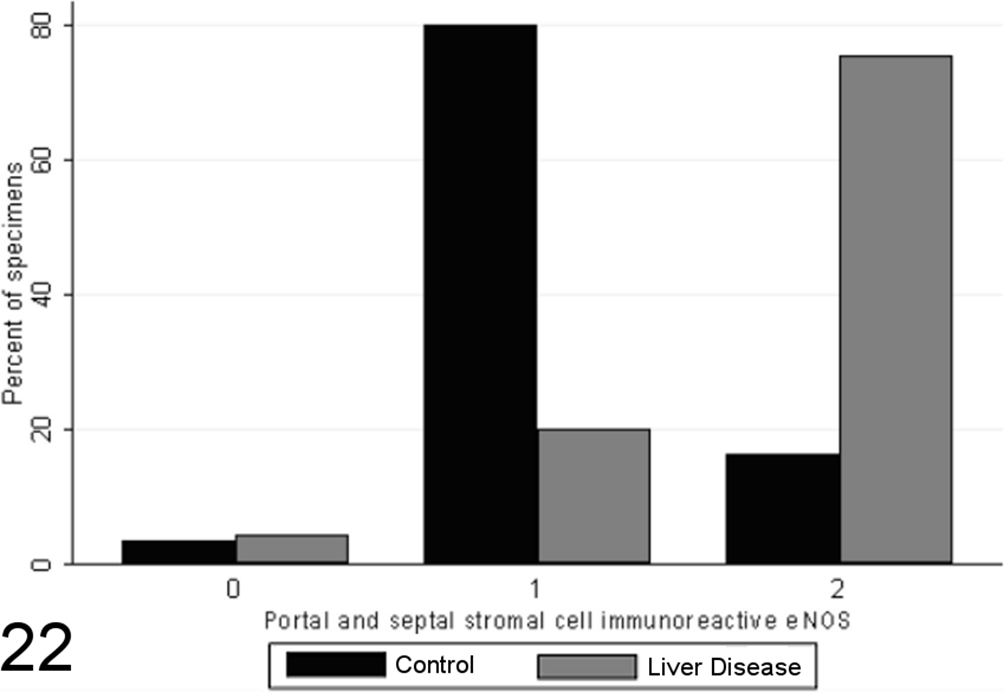
Scores for portal and septal stromal cell endothelial nitric oxide synthase (eNOS) immunoreactivity in case (n = 45) and control (n = 55) canine liver biopsy specimens.
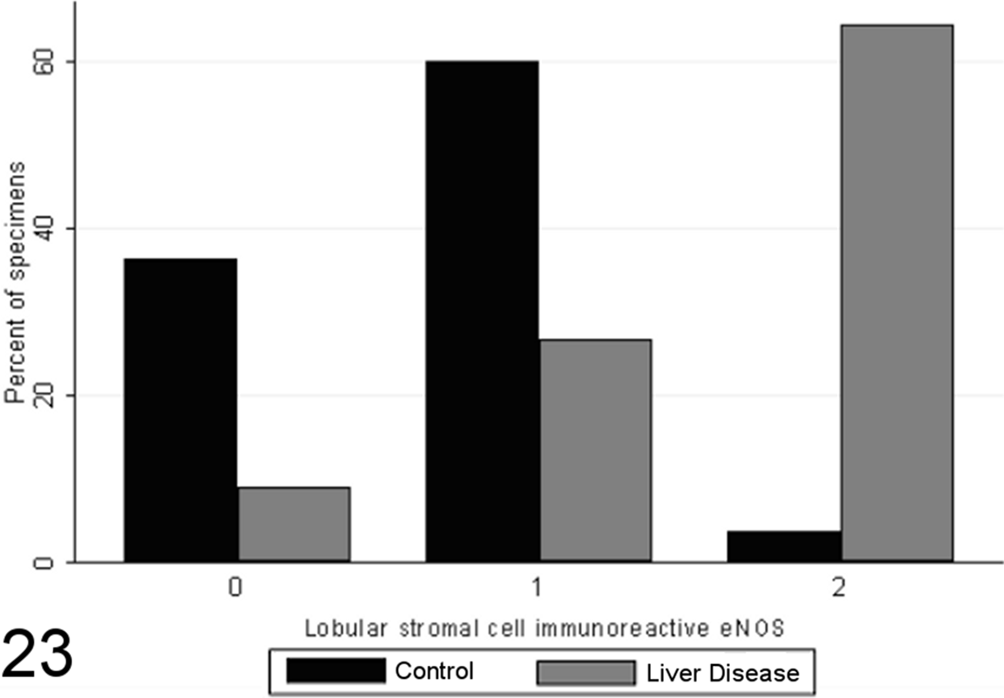
Scores for lobular stromal cell endothelial nitric oxide synthase (eNOS) immunoreactivity in case (n = 45) and control (n = 55) canine liver biopsy specimens.
Discussion
Histologic scoring systems for evaluating the presence and severity of necrosis, inflammation, and fibrosis in biopsies from patients with chronic liver disease are used extensively in human hepatopathology. 4 Such systems facilitate the comparison of biopsy specimens by allowing standardized assessment of various morphologic alterations, including those associated with clinical improvement or decompensation over time. These systems provide a valuable scaffold upon which one can examine prognostic data, risk factors in disease, mechanisms of pathogenesis, and the relative efficacies of different therapeutic modalities. 30 In this study, we adapted a histologic scoring system to evaluate grade of necroinflammatory tissue change, then queried the association of grade score with several mechanistic contributors to liver injury and repair, including markers of hepatocellular apoptosis, regeneration, nitric oxide synthase isoform expression, copper and iron accumulation, and MDA adducts as an indicator of oxidative stress. This particular scheme was selected for this study since it has relatively well-defined criteria and associated thresholds, evaluates stage of fibrosis separately from grade of necrosis and inflammation, and produces a summative score based on various criteria determined to be clinically relevant in human hepatopathology. The scoring system was adopted for our investigative objectives; its potential utility in routine diagnostic grading and staging was not assessed.
In canine chronic liver disease, increasing severity of hepatocellular injury and tissue inflammation is associated with both increased hepatocellular replication (assessed by Ki-67 and mitotic index) and apoptosis (assessed by CC-3), predictable in a milieu of progressive tissue injury and attempted repair. There is also increased iron accumulation in all compartments of the liver, similar to what has been reported in various human chronic liver diseases 50,59 and suggested to be a result of the phagocytosis of dead cells, particularly hepatocytes, with local concentration of iron storage products for which there is no excretory mechanism. 12 There was no significant correlation between hepatocyte immunoreactive MDA-protein adduct score and presence and severity of liver injury and inflammation, an unexpected finding that is in contrast to the findings of Spee et al, 60 who found that dogs with chronic inflammatory and cholestatic liver diseases tended to have reduced GSH:GSSG ratios (an alternate indirect indicator of oxidative stress). This contrast may relate to differences between these indirect measures of oxidative stress. It may be that progression to the formation of MDA adducts is not an early or important outcome in canine liver disease, although when MDA-protein adduct expression was analyzed according to the pattern of expression (rather than the % hepatocyte expression), a generalized diffuse pattern of MDA adduct formation was found to correlate strongly with grade of disease, suggesting that oxidative stress may progress to the formation of MDA adducts in those cases with profound inflammation and necrosis. Similarly, there is only a weak relationship between histochemically detectable hepatocellular copper and disease grade in our study. Copper accumulation in chronic liver disease has been attributed to impaired copper excretion secondary to impaired biliary flow 13 in addition to primary copper storage abnormalities. The absence of significant correlations between stainable copper and iron and scores of MDA adduct immunoreactivity suggests that stored amounts of these redox active metals are not directly associated with the degree of oxidative stress, at least as measured by formation of MDA-protein adducts.
There is a strong association between the severity of hepatocellular injury and tissue inflammation and the expression of NOS isoforms. iNOS expression is upregulated in stromal compartments in proportion to the severity of inflammation and cell death, a pattern of response in accordance with the findings of Mohammed et al, 44 who reported induction of iNOS within stromal, endothelial, and inflammatory cells without a significant hepatocellular contribution. Various inflammatory mediators, particularly interleukin-1β, tumor necrosis factor, interferon-γ, and lipopolysaccharide upregulate iNOS in other mammals, 14 suggesting that the observed increase of iNOS in canine liver disease is a possible indicator of inflammatory activity in the canine liver. eNOS expression, normally present in a small proportion of hepatocytes in health, is upregulated in both parenchymal and stromal cell compartments in relation to the severity of tissue injury and inflammation. Often regarded as a constitutive form of NOS, 10 eNOS expression is increased in chronic liver disease of humans. 2,42,44 McNaughton et al 42 similarly reported eNOS at a basal level within hepatocytes (although also in vascular and sinusoidal endothelium, as well as biliary epithelium) in control human liver with intensification of hepatocellular nuclear staining with disease. The pathophysiological consequences of these NOS alterations in chronic liver disease are unknown. Nitric oxide alone appears to be associated with protective effects against hepatocellular apoptosis, 11,25,33 –35,38,63 but a direct relationship was not found between NOS isoform and CC-3 immunoreactivity. Further studies are needed to determine if the expression of eNOS and iNOS or their products might have an alternate mechanistic role in the pathogenesis of canine chronic liver disease, such as the direct or indirect propagation of cell death.
As a retrospective investigation, several limitations must be considered in the interpretation of these data. The case and control populations are, by necessity, heterogeneous, with differing spectrums of animal breeds, ages, and sexes, which could bias or confound the results. Chronic liver disease is more common in the older dog but is not restricted to this age group (as evidenced by the 2-year-old dog in the case group), and liver biopsies are rarely available from age-matched clinical cases without liver disease. While we assessed case and control animals together in the correlations of various grade and stage end points, it is possible that some associations partly reflect age-related rather than disease-related change. The independent comparison of these cytochemical and immunohistochemical stains to the severity of disease in addition to case/control status attempts to mitigate the effects of this interference. Further prospective investigation would permit age/breed/sex matching, as well as alternative methodologic assessment of the factors identified as potentially important.
In summary, the severity of inflammation and necrosis in canine chronic liver disease strongly correlates with the expression of markers for hepatocellular apoptosis and regeneration, with immunoreactivity for the endothelial and inducible isoforms of nitric oxide synthase in several hepatic tissue compartments, with a diffuse pattern of MDA-protein adduct immunoreactivity and with accumulation of stainable iron, and only weakly with the accumulation of stainable hepatocellular copper. The significance of oxidative stress and interaction with accumulation of transitional metals is uncertain and warrants further study using alternate methods. Further prospective studies are required to determine how the grade of tissue injury in canine liver biopsies might relate to clinical outcomes.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by the Pet Trust Foundation of the Ontario Veterinary College, University of Guelph.
