Abstract
A well-circumscribed mass 70 X 35 X 28 cm in size and 41 kg in weight was detected at necropsy in a male adult horse within the omentum major without any association to the gastrointestinal tract. The tumor consisted of multiple white-to-yellow lobules and displayed a firm consistency. In addition, multiple cysts filled with blood-like fluid, and multifocal areas of necrosis were observed. Histologically, the mass consisted of slightly pleomorphic spindloid-shaped cells arranged in interlacing bundles containing elongated nuclei with blunt ends. The majority of tumor cells revealed a positive immunoreaction for α-smooth muscle actin, vimentin, and neuron-specific enolase and were negative for S-100, factor VIII-related antigen, and glial fibrillary acidic protein. Few tumor cells showed expression of desmin and c-kit. On the basis of macroscopy, histology, and immunohistochemistry, an omental leiomyoma was diagnosed.
Leiomyomas are benign mesenchymal tumors arising from smooth muscle cells. They occur in many domestic animals but most commonly in dogs. In the latter, leiomyomas were most frequently observed in the uterus and gastrointestinal tract. 4 Intestinal leiomyomas have also been found in horses. 3, 11, 13, 19 Apart from the intestine, leiomyomas of the female genital tract, testis, and urinary bladder have been described in horses. 4 So far, omental tumors have been reported in 2 horses. One was diagnosed as an omental fibrosarcoma, 12 and the other, as disseminated peritoneal leiomyomatosis. 17 A solitary leiomyoma originating in the omentum has been described in a dog. 4 Similarly, omental leiomyomas have been reported in humans occasionally. 16, 23, 25 This is the first report of a primary leiomyoma of a male horse arising from the omentum major.
A 16-year-old Hanoverian gelding was presented to the Equine Clinic of the University of Veterinary Medicine Hannover with a history of lethargy and a stilted gait for the previous 2–3 weeks. At the initial examination, the horse was depressed and reluctant to move and showed a poor body condition with a pot belly appearance. On rectal palpation, a ventral intra-abdominal solid-to-fluctuating mass was found. The cranial border could not be defined through transrectal palpation. Ultrasonographically, an inhomogeneous mass with hyperechoic texture and multiple variously sized hypoechoic caverns extending from the ventral inguinal region to the ventral cranial abdomen, approximately 10 cm caudal to the sternum, was seen. Next to the mass, several loops of small intestine could be identified, and their involvement into the mass could not be excluded ultrasonographically. The owner did not approve treatment, and the horse was euthanized.
At necropsy, in the left cranial abdomen in close proximity to the diaphragm, a well-circumscribed mass 70 × 35 × 28 cm in size and 41 kg in weight was detected within the omentum major without any association to the gastrointestinal tract and 20 cm apart from the greater curvature of the stomach (Fig. 1). The neoplasm was characterized by multiple white-to-yellow lobules with a firm consistency. Some areas contained multiple cysts filled with serosanguineous watery fluid, and multifocal areas of necrosis were found (Fig. 2). Additionally, the abdominal cavity revealed a moderate diffuse fibrinous inflammation. Findings in other organs included a moderate hypertrophy of the tunica muscularis of the ileum and the caudal region of the esophagus. A white dense area measuring 0.5 cm in diameter was detected within the epicardium in the right ventricle of the heart. Lung, spleen, liver, and kidney showed a moderate-to-severe congestion.
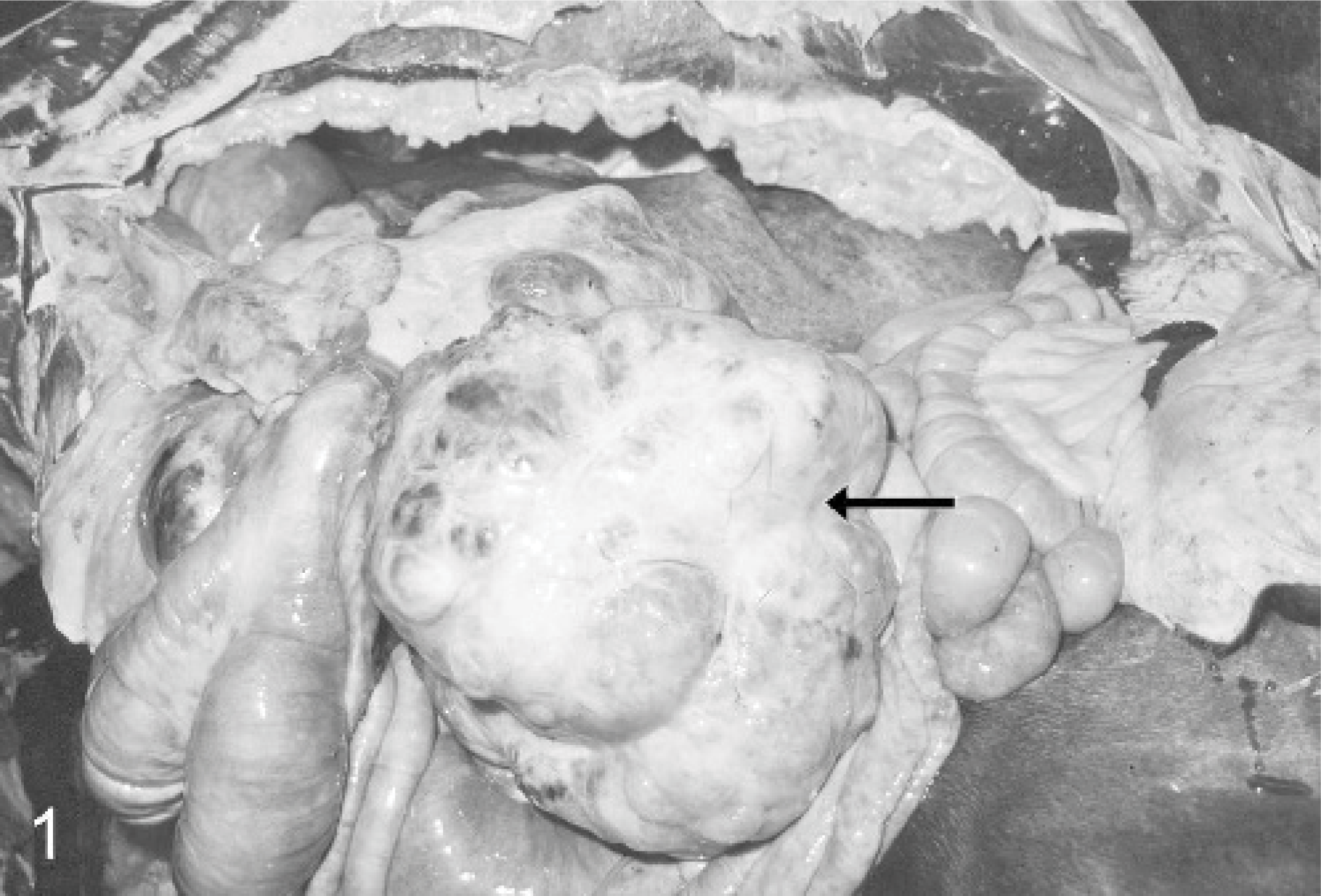
Abdominal cavity; horse. Well-circumscribed leiomyoma originates from the omentum major (arrow).
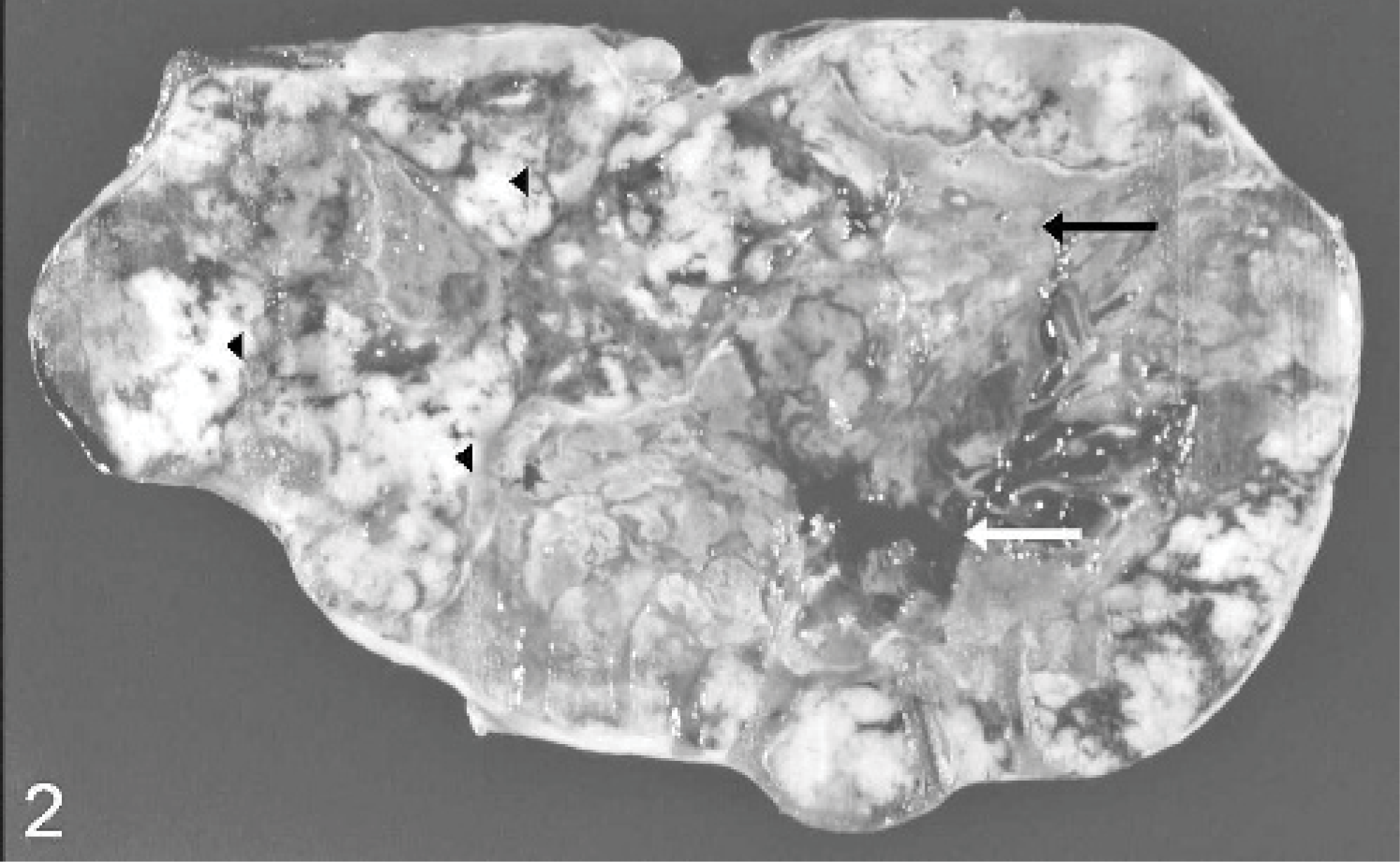
Omental leiomyoma; horse. Cut surface of the multilobular tumor has white-to-yellow firm areas (arrowheads), multifocal necrosis (black arrow), and multiple cysts filled with blood-like fluid (white arrow).
For histologic investigation, tissue samples from various organs, including the omental mass, heart, lung, kidney, liver, muscles, spleen, gastrointestinal tract, and abdominal lymph nodes, were fixed in 10% nonbuffered formalin, embedded in paraffin, and stained with HE.
Immunohistologic investigation on selected sections of the mass was performed with murine monoclonal antibodies specific for α-smooth muscle actin (diluted 1 : 200; Dako, Glostrup, Denmark), vimentin (diluted 1 : 100, Dako), desmin (clone D33, diluted 1 : 100, Dako) and neuron-specific enolase (NSE; diluted 1 : 200, Dako) as well as polyclonal rabbit antibodies specific for S-100 (diluted 1 : 800; Sigma, Taufkirchen, Germany), factor VIII (diluted 1 : 200, Dako), glial fibrillary acidic protein (GFAP; diluted 1 : 2000, Dako) and c-kit (diluted 1 : 60; Calbiochem, Darmstadt, Germany) as described. 20, 21 Briefly, after incubation with the primary antibody, a biotin-conjugated goat anti-mouse or goat anti-rabbit antibody was applied. The antigen-antibody complex was visualized using the avidin-biotin-complex (ABC) method (Vector, Peterborough, England). Tissue sections were counterstained with Mayeŕs hematoxylin. Negative controls consisted of sections incubated with mouse ascites fluid or inactivated rabbit serum instead of the primary antibody.
Histologic examination of the omental mass revealed an encapsulated, in most areas moderately dense cellular, in some areas acellular, mass and a fibro-vascular stroma. The numerous neoplastic mesenchymal cells were arranged in interlacing bundles (Fig. 3). These spindloid-shaped cells, varying from 10 × 20 μm to 15 × 40 μm in size, with poorly defined cell borders, were slightly pleomorphic. The nuclei were mostly elongated with blunt ends (Fig. 4). Tumor cells exhibited a moderate-to-abundant eosinophilic cytoplasm and occasionally small vacuoles. The frequency of mitotic figures was 0–1 per high power field. Mild multifocal infiltrations of neutrophilic granulocytes, macrophages, and lymphocytes were observed. The acellular areas were characterized by circumscribed necroses with extravasation of fibrin. In addition, mild multifocal hemorrhages were present.
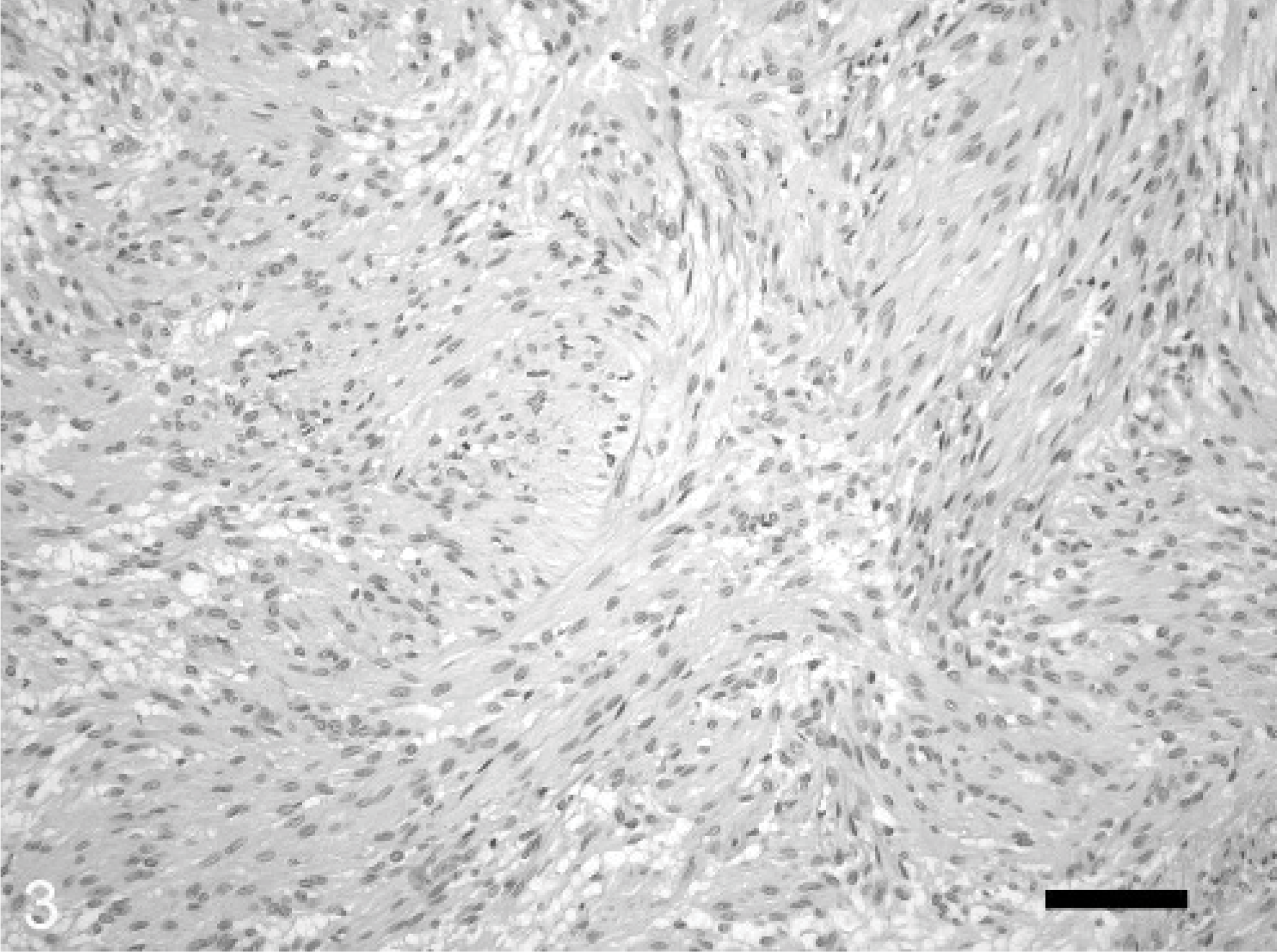
Omental leiomyoma; horse. Spindloid-shaped tumor cells are arranged in interlacing bundles. HE. Bar = 25 μm.
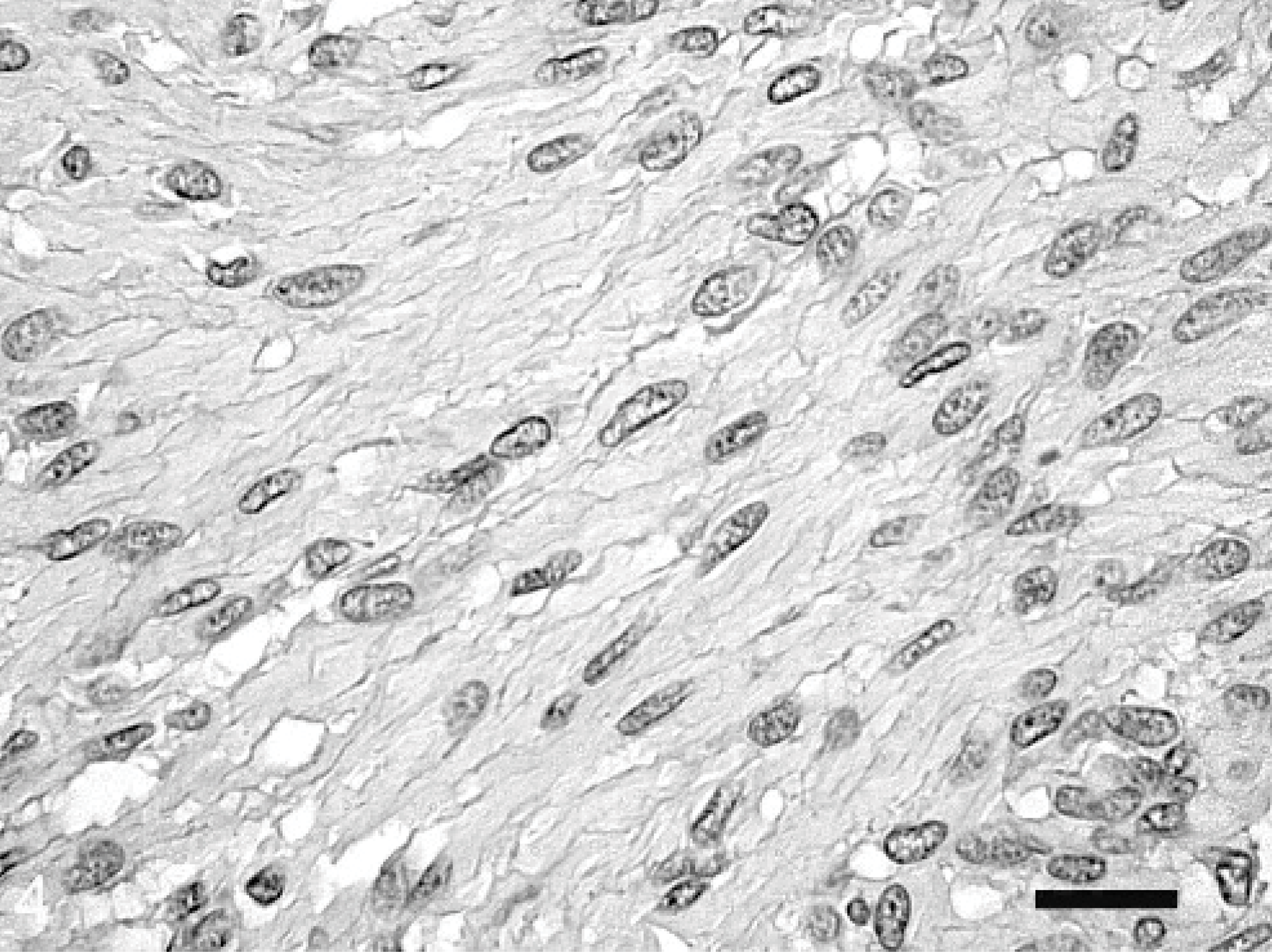
Omental leiomyoma; horse. Slightly pleomorphic tumor cells contain cigar-shaped, elongated nuclei with blunt ends. HE. Bar = 100 μm.
Adjacent areas of the omentum showed a mild multifocal lymphohistiocytic and neutrophilic inflammation and fibrosis. The mesenterial, aortic, and the portal lymph nodes were characterized by a moderate lymphocytic hyperplasia. The white dense area in the epicardium of the right ventricle consisted of fibrosis with squamous epithelial metaplasia. All other organs were without significant microscopic lesions.
Ninety-five percent of the tumor cells revealed a positive immunoreaction for α-smooth muscle actin; 80% of the tumor cells, for vimentin; and 60% of the tumor cells, for NSE. Five percent of the tumor cells were positive for desmin, and a focal area of the tumor (approximately 5%) showed a slight expression of c-kit. The tumor cells lacked immunoreactivity for S-100, factor VIII–related antigen, and GFAP.
Histologic appearance, especially the presence of well-differentiated spindled cells with cigar-shaped nuclei, led to the diagnosis of a leiomyoma in the omentum major. This was confirmed by immunoreactivity for α-smooth muscle actin and desmin in tumor cells (Fig. 5). In contrast to leiomyosarcomas, this tumor was encapsulated and showed no local invasiveness. The cells were only slightly pleomorphic and exhibited a low mitotic rate. Furthermore, no metastases were found in the regional lymph nodes or other organs, including the lung and heart.
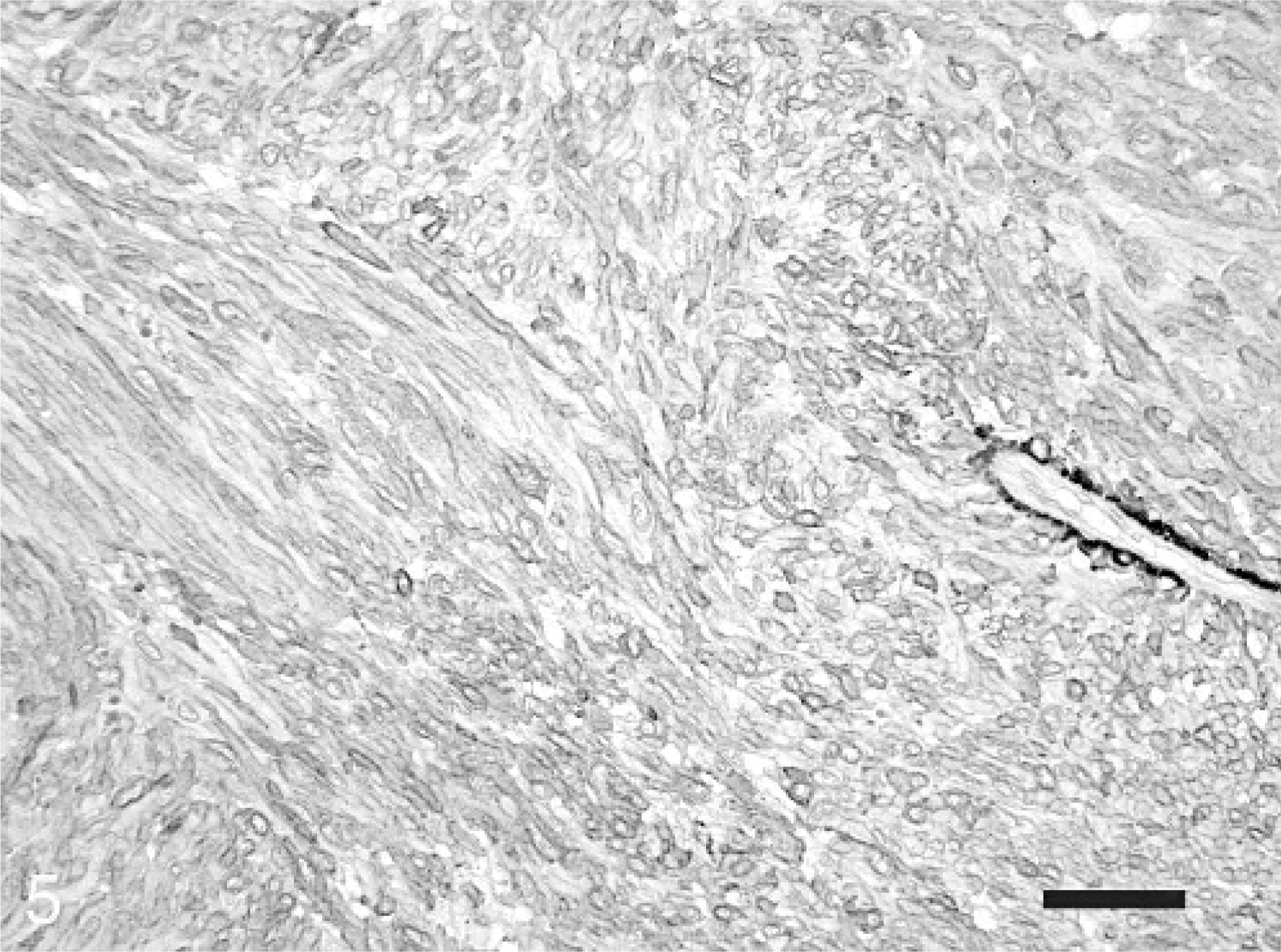
Omental leiomyoma; horse. Strong immunoreactivity for α-smooth muscle actin of the tumor cells is seen. Immunohistochemistry for α-smooth muscle actin and slightly counterstained with hematoxylin. Bar = 50 μm.
Smooth muscle cells are widely distributed throughout the body and are especially prominent in the gastrointestinal, genitourinary, and respiratory tract. They can also be detected in the walls of blood and lymph vessels. In humans, leiomyomas are divided according to their topographic distribution into cutaneous and vascular leiomyomas and leiomyomas of deep soft tissue. Vascular leiomyoma, a solitary form of leiomyoma that usually occurs in the subcutis, is composed of numerous thick-walled vessels and occurs more frequently in women. 7 Leiomyomas of deep soft tissue are predominantly located in the deep muscle of the extremities, within the abdominal cavity or retroperitoneally. 7
In horses, leiomyomas have been reported in the intestine, female genital tract, testis, and urinary bladder. 3, 4, 11, 13, 19 More recent studies have shown that some intestinal leiomyomas should be classified as gastrointestinal stromal tumors (GISTs). 6, 10 GISTs are, as defined by the Histologic Classification of Tumors of Domestic Animals of the World Health Organization, a spindle-cell neoplasm presumed to originate from primitive mesenchymal cells. 14 Human GISTs, like interstitial cells of Cajal (ICC), express a receptor tyrosine kinase (KIT) encoded by a c-kit proto-oncogene and CD34. Therefore, it is assumed that GISTs originate from ICC. 15 However, this conclusion is still discussed controversially. 27 The activation of KIT in GIST may represent part of an oncogenic process rather than indicating that these tumors derived from ICC. 27 Similarly, KIT-positive tumors, diagnosed as GISTs, have also been reported in humans at locations apart from the gastrointestinal tract, such as the pharynx and pancreas. 5, 26, 28 Furthermore, GISTs deriving from the omentum have been detected in humans. 8, 9, 16, 18
However, the cellular origin of these so-called extragastrointestinal stromal tumors (EGISTs) is not known, and the occurrence of EGISTs is still discussed controversially. 1 One study could find KIT-positive mesenchymal cells in the normal human omentum; 24 in another study, most cases of EGISTs were re-diagnosed as metastases of normal GISTs or as GISTs that have arisen from the outer muscle coat of the gastrointestinal tract and lost their contact due to an extensive extramural growth pattern. 1 So far, GISTs in horses have been reported in 2 studies, which were all associated to the gastrointestinal tract. 6, 10 Although GISTs, described by Del Piero et al. (2001), 6 showed expression of vimentin, S-100, and α-smooth muscle actin, all tumors lacked expression of NSE. In contrast, in the other study, GISTs were positive for NSE and negative for S-100. 10 In both studies, tumors lacked expression of desmin. 6, 10 However, this marker was detected in the tumor investigated in the present case.
Similar to equine GISTs, expression of α-smooth muscle actin, vimentin, and NSE was found in the present neoplasm. Although the tumor in this report showed also a mild focal expression of c-kit, no association of the tumor with the wall of the gastrointestinal tract could be demonstrated. GISTs in horses, such as omental GISTs in humans, lacked expression of desmin, 6, 10, 22 which was a dominant finding in smooth muscle tumors. 2 Therefore, macroscopy and the immunohistochemical findings support the diagnosis of a leiomyoma in the omentum. To our knowledge, in domestic animals, an omental leiomyoma has only been diagnosed once in a dog. 4 Whereas in humans, a leiomyoma in the omentum has been reported in a child and 67- and 41-year-old women. 16, 23, 25 In one case, the omental leiomyoma was associated with myomas in the uterus and a relationship between the uterine and the omental tumor was suspected. 26 In the other 2 reports, no uterine myomas were observed.
In horses, only 2 mesenchymal tumors originating in the omentum were reported so far. In 1987, Harvey et al. 12 described a 13-year-old Thoroughbred mare suffering from an omental fibrosarcoma of low-grade malignancy, 30 × 30 × 60 cm in size. Tumor cells were described as fusiform, with oval nuclei and moderate amounts of eosinophilic cytoplasm. Special staining techniques or electron microscopy to differentiate fibroblast from smooth muscle cells was not mentioned in that report, and no figures displaying the light microscopic appearance of the tumor cells were provided. Therefore, that presented neoplasm might possibly also have originated from smooth muscle cells. The second reported omental tumor in a horse was described as a single large omental mass weighting 34 kg in a 6-year-old female Quarter Horse. 17 This tumor was compared with disseminated peritoneal leiomyomatosis in humans and accordingly diagnosed.
Leiomyomatosis peritonealis disseminata is a rare condition in which multiple smooth muscle or smooth muscle-like nodules develop in subperitoneal locations throughout the abdominal cavity. 7 The lesion occurs exclusively in women, usually during the child-bearing years, and the involvement of hormonal factors in the pathogenesis of this condition is suspected. 7 In the reported equine case, 17 only 1 huge mass was detected in the omentum, indicating that the neoplasm might also represent a leiomyoma.
Although it cannot be excluded completely that the leiomyoma described in this report originated from blood or lymph vessels, the tumor should, on the basis of the nomenclature used for leiomyomas in humans and the location of the mass in the omentum, be classified as a leiomyoma of the deep soft tissue.
Footnotes
Acknowledgements
We are grateful to Bettina Buck and Petra Grünig for excellent technical assistance.
