Abstract
A 14-y-old pony mare was referred after 30-d duration of intermittent pyrexia, anorexia, weight loss, and change in manure consistency. Physical examination revealed a palpable but reducible ventral abdominal mass. Transabdominal ultrasonography revealed multiple distended, hypomotile, and thickened small intestinal loops in close approximation with numerous, well-defined, hyperechoic masses. There was a large amount of echogenic peritoneal fluid; abdominocentesis revealed a neutrophilic and macrophagic inflammatory exudate, and a mixed bacterial population was cultured. Given the poor prognosis, the mare was euthanized. The autopsy findings included a large abdominal abscess, serosanguineous peritoneal fluid with fibrin strands, and ~50 outpouches communicating with the lumen and extending from the anti-mesenteric aspect of the duodenum, jejunum, and ileum. These structures were classified as pseudodiverticula based on the histologic absence of the tunica muscularis layer of the intestinal wall. Pseudodiverticula should be included as a differential etiology in horses when clinical signs consistent with colic, diarrhea, or weight loss are recognized and, when on examination, one or more organized masses are palpated or visualized on transabdominal ultrasound, as well as visualization of small intestinal loops with thickened walls.
A 14-y-old, ~180-kg, Welsh pony mare, was admitted to the Virginia-Maryland College of Veterinary Medicine Teaching Hospital (VTH; Blacksburg, VA, USA) for evaluation of fever of unknown origin, intermittent anorexia, lethargy, and weight loss of ~30-d duration. Treatment on the farm prior to admittance included a variety of antimicrobials and nonsteroidal anti-inflammatories: oxytetracycline IV, flunixin meglumine (Banamine; Merck) IV and PO, minocycline hydrochloride PO, trimethoprim–sulfamethoxazole PO, and phenylbutazone PO (VetriBute; VetOne). Probiotics and feed supplements had also been administered on the farm; specific product information, as well as frequency and dosages of all medications administered on the farm, are unknown. Rectal temperature was checked on the farm twice per day by the owners; the highest temperature reported prior to admittance to the VTH was 39.9°C. The owners reported a mass on the ventral abdomen caudal to the last rib that progressively increased in size in the 2 wk prior to admission to the VTH. Manure consistency on the farm varied from formed fecal balls to diarrhea, and owner-reported hematology results included persistent hyperfibrinogenemia.
Upon admission to the VTH, the mare was quiet, alert, and responsive and had a body condition score of 3 of 9. Physical examination revealed a rectal temperature of 38.5°C, respiratory rate of 12 breaths per minute, tachycardia (60 beats/min), and distinct halitosis, which may have been the result of anorexia and overgrowth of bacterial organisms within the mouth. Gastrointestinal (GI) borborygmi were appreciable in all quadrants; there was bilateral hypermotility in the ventral quadrants. There was a firm mass over the right jugular vein that was ~3 cm in length; the left jugular vein was firm upon palpation; both of these findings were interpreted as likely thrombosis. A firm, reducible mass of ~12 cm diameter was palpable on the ventral midline.
A complete blood count (CBC; Suppl. Table 1) at the VTH revealed normochromic, normocytic anemia, hyperproteinemia, and normal peripheral lactate. The overall total white blood cell count was within normal limits with evidence of inflammation (significant hyperfibrinogenemia, mild band neutrophilia, panhyperproteinemia, elevated serum amyloid A) and glucocorticoid stress (mild lymphopenia, mild monocytosis). Review of a blood smear revealed toxic neutrophils; there was no evidence of an infectious agent. Serum biochemistry abnormalities (Suppl. Table 2) included low urea and creatinine, hyperproteinemia characterized by significant hypoalbuminemia and hyperglobulinemia, elevated gamma-glutamyl transferase (GGT) activity, low creatine kinase (CK) activity, and electrolyte derangements including mild hyponatremia, mild hypochloremia, and mild hypomagnesemia. Metabolic acidosis, characterized by a low tCO2 and an elevated anion gap, was also noted. Abdominocentesis yielded a septic exudate characterized by a mixed neutrophilic and macrophagic inflammatory population and rare bacterial rods. Culture of this exudate grew small numbers of Escherichia coli, alpha-hemolytic Streptococcus spp., Fusobacterium necrophorum, and Prevotella heparinolytica. No neoplastic cells were observed.
Transabdominal ultrasound was performed (2.5–5 MHz curvilinear transducer, Affinity 50G ultrasound; Philips Ultrasound). The abdomen contained a large amount of echogenic fluid, and multiple loops of small intestine were distended up to 5 cm in diameter (normal = 2–3 cm) with wall thickness of 3–6 mm (normal = 2–3 mm); many loops were hypomotile. The liver had rounded edges and subjectively appeared mildly enlarged. Multifocal, hyperechoic foci were appreciable bilaterally within the abdomen and appeared to extend from, or were attached to, small intestinal loops (Fig. 1); potential differentials for these structures included masses or abscesses.
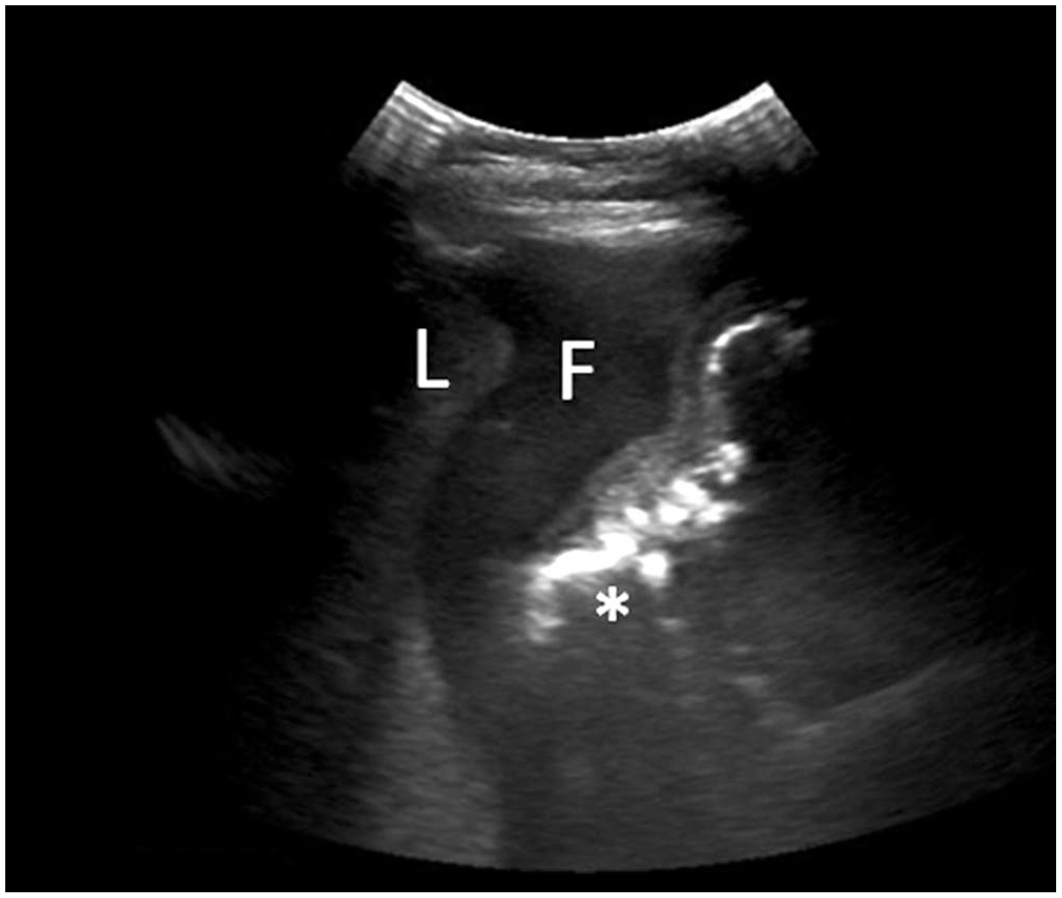
Transabdominal ultrasound images of the right abdomen. Rounded liver margins (L) surrounded by echogenic fluid (F) and an organized but poorly delineated hyperechoic structure (*), which probably was thickened small intestine.
Thoracic ultrasound revealed focal areas of pleural roughening in the left hemithorax and evidence of pleural fluid up to 3 cm deep in the right ventral hemithorax. Ultrasound of the right jugular vein had a large thrombus and was ~75% obstructed, with some blood flow evident.
Differential diagnoses based on the history, examination, and clinical findings included septic peritonitis of unknown etiology, abdominal abscess, and neoplasia, including lymphoma, GI stromal tumor, and/or leiomyoma or leiomyosarcoma. The prognosis for successful treatment was guarded-to-poor, and the course of therapy was likely to be prolonged. Ultimately, euthanasia was elected given the poor prognosis, and an autopsy was performed.
On postmortem examination of the pony, ~50 dilations, of 2–4 cm diameter, extended from the antimesenteric aspect of the serosal surface and communicated with the lumen of the duodenum, jejunum, and ileum (Fig. 2A, 2B). These outpouchings were up to 3.0 cm deep and variably contained impacted digesta or purulent material. Such abnormalities were not noted in the cecum or colon. The mass described on clinical examination corresponded with a 12-cm diameter abscess containing purulent-to-caseous material adhered to the left ventral body wall and adjacent jejunum. Although no communication was identified between this abscess and the jejunal lumen, abundant serosal fibrinous material may have obscured such a communication. There was ~5 L of serosanguineous fluid within the abdomen, and numerous fibrinous strands were firmly attached to the serosal surface of the liver, spleen, small intestines, cecum, and large colon; hence, adjacent viscera often adhered to one another. The right jugular vein contained a 3.0-cm thrombus cranial to an intravenous catheter; no thrombus was found in the left jugular vein. No significant gross findings were detected in other internal organs.
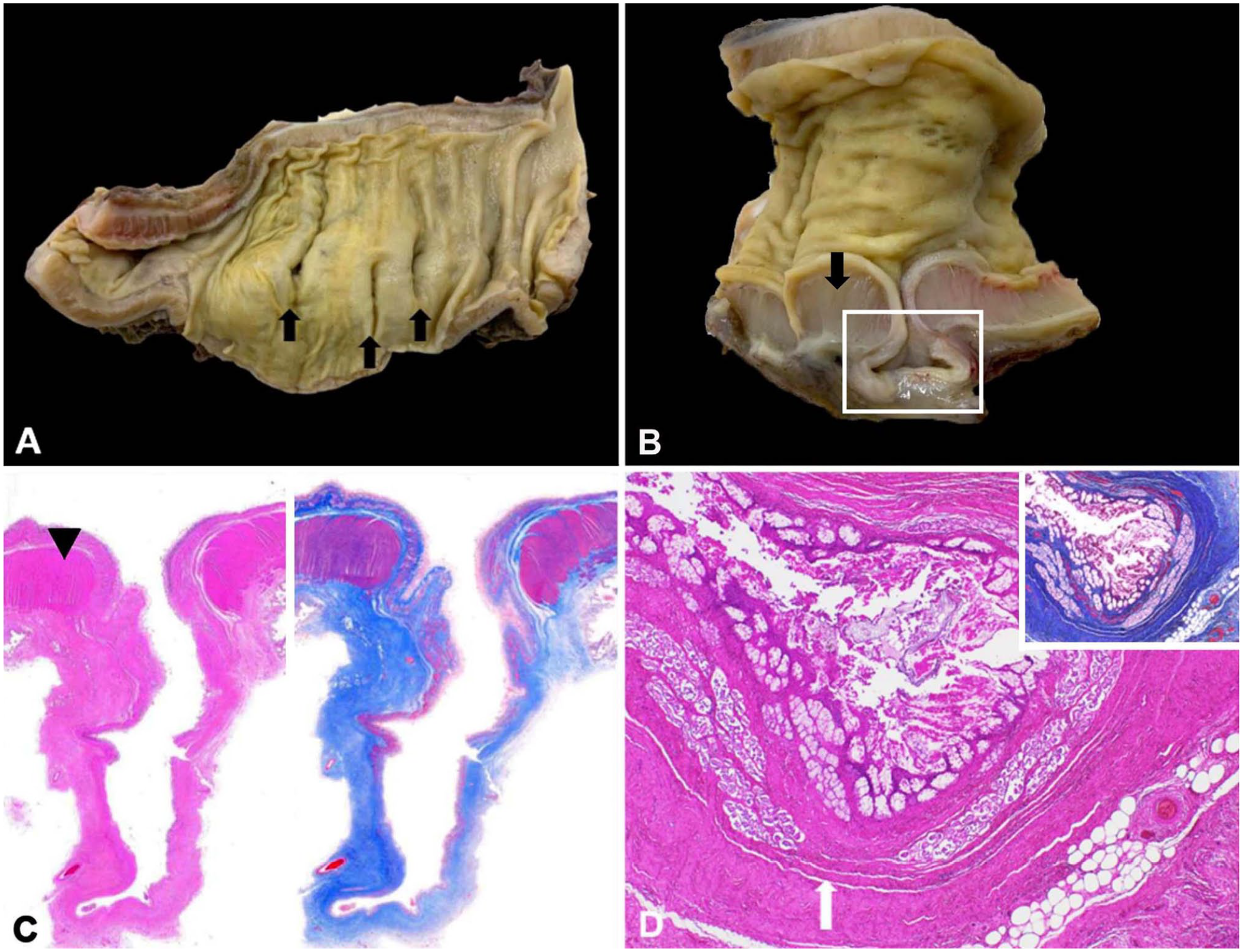
Small intestine of a pony mare with pseudodiverticulosis.
Multiple sections of the small intestine were examined with routine hematoxylin and eosin stain as well as trichrome stain (Fig. 2C, 2D). The inner, circular smooth muscle layer of the tunica muscularis immediately orad and aborad to the dilations was diffusely hypertrophic and hyperplastic. In all examined sections, the wall of the outpouchings lacked this layer of the tunica muscularis, but otherwise contained all normal structures between the mucosal and serosal surfaces. Based on the lack of the tunica muscularis, the outpouchings were diagnosed as pseudodiverticula. Additionally, there was significant peritonitis; serosal surfaces of the normal intestine, pseudodiverticula, and other abdominal organs were markedly thickened with fibrin, edema, congestion, and an inflammatory infiltrate composed of neutrophils, large foamy macrophages, lymphocytes, and plasma cells. Other histologic findings included mild periportal, lymphoplasmacytic hepatitis, mild hepatic lipidosis, and right jugular vein thrombosis.
Several reports identify diverticula of the GI tract in various animal species, including horses.3–9,12 These abnormalities have been classified as either true diverticula or acquired diverticula, which are also known as pseudodiverticula or false pulsion diverticula. 2 True diverticula consist histologically of all layers of the intestinal wall. In humans, true diverticula occur as a result of improper embryologic development, and clinical signs most frequently occur at a young age 1 ; likewise, in animal species, true diverticula often appear congenitally.8,10 In contrast, pseudodiverticula lack the tunica muscularis, and the walls of these dilations are composed of the mucosa, submucosa, and serosa.1,2,10 As seen in our case, pseudodiverticula are frequently seen concurrently with hypertrophy and hyperplasia of the tunica muscularis of the adjacent intestinal wall. Hypertrophy and hyperplasia of the smooth muscle can be idiopathic (primary) or secondary (compensatory) to increased contraction force as a result of partial aboral stenosis or obstruction.3,11 Hence, pseudodiverticula may occur because of a weakened tunica muscularis and/or increased intraluminal pressure, resulting in expulsion of the mucosal and submucosal layers through a defect in the tunica muscularis.2,8
Pseudodiverticulosis, the disease associated with the formation of pseudodiverticula as in our case, has been described in the small intestine of horses, pigs, sheep, cats, elk, a wallaby, and most recently, rabbits.1,5,7–9,12 Small intestinal pseudodiverticula are uncommon in horses, with only a handful of cases reported.3,4,7,12 Pseudodiverticula in horses have most commonly been reported along the mesenteric border of the intestine, and likely result from weakness associated with perforation of the intestinal wall near vasculature.3,7,12 However, one published case series reports pseudodiverticula along the antimesenteric border in 3 horses with concurrent intestinal lymphoma in close association with Peyer patches. 8
Given the history, presentation, evidence of inflammation, and presence of a septic exudate antemortem, the abdominal abscess discovered postmortem in this pony was not surprising; however, the finding of pseudodiverticula throughout the small intestine was unexpected. Abnormalities noted on abdominal ultrasound were identified on postmortem examination as a single large abscess with many small intestinal pseudodiverticula and small intestinal smooth muscle hypertrophy and hyperplasia; the latter two were confirmed by histologic evaluation. Smooth muscle hypertrophy and hyperplasia were most likely idiopathic, given that no obstruction or stenosis was identified. Perforation of pseudodiverticula was thought to be the cause of the septic peritonitis because many of these outpouchings grossly contained impacted feed and/or abscess material and the culture of abdominal exudate revealed a mixture of bacteria. However, translocation of septic material and bacteria through the thinned wall of the pseudodiverticula without perforation was also possible antemortem and may have caused or contributed to the septic peritonitis that was recognized on abdominocentesis. Definitive diagnosis could not be made because no perforation was identified on gross evaluation, and the mixture of bacteria on culture could have been attributed to contamination. The presence of pseudodiverticula along the antimesenteric border in the absence of concurrent neoplastic lesions is a unique presentation that has not been reported previously in horses, to our knowledge.
Many of the clinical signs in our case were consistent with previous reports of pseudodiverticulosis in horses, including thin body condition, chronic anorexia, weight loss, ventral abdominal edema, and lethargy.8,9 Although pyrexia is not consistently reported in cases of pseudodiverticulosis, it is not surprising in our case given the presence of the abdominal abscess, septic exudate, and associated severe peritonitis. Absence of pyrexia in previous reports may represent less complicated cases without a severe inflammatory component.
Diagnosis of pseudodiverticulosis of the equine GI tract, specifically the small intestine or the large colon, is a challenge given the complex anatomy of the equine GI tract and the inability to physically palpate the entirety of the abdomen during examination, as well as the inability to visualize all structures with imaging modalities, such as ultrasound imaging. Thus, diagnosis of pseudodiverticulosis in the horse requires histologic examination from samples obtained surgically or during postmortem examination. 3 Noninvasive antemortem diagnostic tests, as performed in our case, provide important but incomplete information. Although ultrasound findings in our case were similar to other reported equine cases, visualization and identification of the 5 intestinal wall layers are not always possible in all horses because of the depth of retroperitoneal fat. 5 Hence, it is possible that antemortem testing may misclassify pseudodiverticula as true diverticula. Additional antemortem tests that may have provided a more complete diagnostic picture include endoscopy of the stomach and proximal duodenum, and abdominal palpation per rectum. Endoscopic examination is limited by the length of the endoscope, although given the size of the patient, visualization of the proximal duodenum and the duodenal papillae would have been achievable. Thickened loops of small intestine likely would be palpable on rectal examination 3 ; however, it is unknown if the pseudodiverticula would have been palpable or recognized as an abnormality depending upon their size and anatomic location. The abdominal abscess would not have been palpable given its cranioventral location. The small size of this pony precluded rectal palpation.
The reported empiric treatment administered prior to presentation at the VTH raised concerns regarding possible resistant microorganisms. Additionally, antimicrobial therapy in horses has an associated risk of colitis and systemic complications. Furthermore, the health and integrity of the jugular veins were also concerns regarding possible medical treatment and sequelae of jugular vein thrombosis. There is one case report of successful surgical treatment of pseudodiverticulosis in a young Arabian filly. 12 However, the pony mare in our report had evidence of systemic disease, and the success of the published case may have been related to a clinically stable patient. Although uncommon, pseudodiverticula and pseudodiverticulosis have been reported in several veterinary species and should be considered when one or more organized, hyperechoic masses are visualized on transabdominal ultrasound or when an abdominal abscess or mass is suspected.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387211032001 – Supplemental material for Small intestinal pseudodiverticulosis in a pony mare
Supplemental material, sj-pdf-1-vdi-10.1177_10406387211032001 for Small intestinal pseudodiverticulosis in a pony mare by Hannah R. Leventhal, Anna M. Hassebroek, Francisco Carvallo and Harold C. McKenzie in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Valentina Stevenson for her assistance with obtaining gross and histopathology images.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
