Abstract
In cases of equine acute abdominal disease, where pancreatic damage is suspected, pancreatic damage can be assessed by measuring increased trypsin activity in the plasma of horses suffering intestinal obstruction and severe shock. The pancreas is particularly vulnerable to splanchnic hypoperfusion because it is a highly active tissue. In this study, 10 horses undergoing abdominal surgery for intestinal obstruction were assayed for trypsin activity on admission and, because of extensive intestinal lesions that were not amenable to surgery, euthanasia was selected; the pancreas was removed before euthanasia. Trypsin activity in the plasma of these horses was significantly higher than in healthy horses (196 ng/ml ± 128.2 versus 28.5 ng/ml ± 19.2; P = 0.0026). Light and transmission electron microscopy revealed slight to severe lesions of vacuolar degeneration, a few zymogen granules, dilation of the endoplasmic reticulum, and swelling of mitochondria in the exocrine pancreas. The activation of an inflammatory cascade occurring during strangulating intestinal obstruction could increase pancreatic anoxic lesions caused by severe shock and hypoperfusion in the horse. Further studies will show the significance of pancreatic lesions and the ensuing damage in equine acute intestinal obstruction and shock.
Introduction
In horses, acute pancreatic disease is a rarely studied entity, and pancreatic involvement in shock states after acute abdominal disorders has not yet been extensively described. But pancreatic injury after hypovolemic or septic shock has been widely demonstrated in animal models by histologic observations (acinar cell damage and necrosis) in pigs, 6 monkeys, 8,25,26 rabbits, 17 and rats 15 and by biochemical assays (increased pancreatic enzyme concentrations in blood). 7 This was attributed to splanchnic hypoperfusion on account of the particular susceptibility of the pancreas to decreased blood flow. The perturbation of the pancreatic microvascular perfusion is an important pathogenic factor in cases of pancreatitis. 17
In humans, shock, especially septic and traumatic shock, leads to hypoperfusion of the splanchnic area, often resulting in multiple organ failure. Among the splanchnic organs, the pancreas is particularly vulnerable to hypovolemia, a so-called shock organ. 31 Histologic studies have demonstrated profound ultrastructural alterations of this organ (particularly visible in the mitochondria and endoplasmic reticulum) after severe trauma with major blood loss not affecting the splanchnic area directly. 14,16 Biochemical assays of increased blood concentrations of amylase, lipase, and trypsin show pancreatic damage in various clinical situations such as cardiovascular surgery with extracorporeal circulation, severe sepsis, and septic shock as well as major trauma, generally without clinical signs of pancreatitis. 2,5,29,31 This unexpected pancreatic failure was related to hypoperfusion or neutrophil activation, and the particular vulnerability of the pancreas was attributed to the high metabolic activity of the acinar cells and to the specific structure of the blood distribution in this organ. 3
In an earlier study, we demonstrated a high level of trypsin activity in the plasma of horses submitted to surgery for intestinal obstruction, especially in those with severe signs of endotoxic shock. We also observed an increase of plasma trypsin activity in the postoperative period. 12 Because abnormal trypsin activity was also found in the plasma of horses with nonstrangulating large intestine obstruction, the increased plasma concentrations of trypsin could not solely be ascribed to increased reabsorption from the ischemic intestinal walls. Therefore, pancreatic lesions may be present. The aim of this study was the biochemical analysis of plasma trypsin activity and light and transmission electron microscopic study of pancreatic tissues from horses presenting severe and irreversible intestinal lesions not amenable to surgery and consequently euthanatized during anesthesia after extraction of the pancreas.
Materials and Methods
Case description
Ten horses (six warmbloods, two Lusitanians, one Welsh pony and one Arab, mean age 10 years, six geldings, three mares and one stallion) admitted for acute abdominal disease and diagnosed with a pathology needing surgery were used in this study. Clinical, hematologic, and biochemical parameters permitted classification of the horses regarding the degree of cardiovascular shock status (absent, mild, or severe shock). Blood was drawn on admission for the assay of enzymatically active trypsin (EAT) as described previously. 12
Three horses (two ponies and one warmblood, aged from 2 to 9 years) euthanatized for long bone fractures without acute abdominal disease or endotoxic shock were used as control.
EAT assay
Plasma samples drawn on admission were immediately stored at −20 C until assay. In brief, the assay is based on the proteolytic cleavage of a specific low molecular weight substrate by active trypsin.
18,27
This chromogenic substrate, carbobenzyloxyl-
The reproducibility was tested in our laboratory and the interassay variation coefficient (eight assays of the same sample) for normal plasma samples (n = 10) was of 2.1 ± 1.5%. For plasma samples with very high values (n = 12) the interassay coefficient was of 4.4 ± 1.5%. The accuracy of the test was adequate because added amounts of 100 ng/ml of trypsin were recovered at ≥90%. The physiologic range of EAT in healthy horses (n = 38) was previously determined to be 28.5 ng/ml ± 19.2 ranging from 3 to 78 ng/ml. 12 An unpaired t-test with Welch correction was used to compare the mean levels of EAT between healthy horses and the pathologic ones.
Histologic examination of the pancreas
Before euthanasia, the pancreas was extracted under general anesthesia for histopathologic evaluation in the cases described above. For comparison the pancreas of three horses being euthanatized for localized limb injuries (long bone fracture) was extracted in the same way. Small fragments (1 mm in diameter) were immediately immersed for fixation in 2.5% glutaraldehyde in phosphate buffer (with postfixation in 1% osmium tetroxide) for transmission electron microscopy and embedded in Epon. The ultrathin sections were mounted on copper grids and stained with uranyl and lead 9 and electron micrographs were taken by a transmission electron microscope Zeiss 910 80 KV (Zeiss, Esslinger, Germany). Larger fragments (2–3 cm in diameter) were fixed in 10% buffered formalin for at least 24 hours. The tissue fragments were then cut and embedded in paraffin wax for light microscopy. Sections of 3 mm were stained with hematoxylin and eosin.
Results
Case description
Small intestine strangulation obstruction was found in five cases, with absent (n = 1), mild (n = 2), or severe signs of shock (n = 2). In all the cases, the small intestine was judged nonviable (20 cm to 12 m). Euthanasia was selected because enterectomy was not permitted. The other five horses had large intestinal obstruction without shock symptoms (n = 1) or with mild shock (n = 4). Euthanasia was performed because the vascular lesions were too extensive because of multiple obstructing adhesions or a nonreducible intussusception. Table 1 shows a more detailed description of the cases, their pathology, and the corresponding intestinal lesion.
Horse Nos. (HN) 1 to 10, classified according to their shock score (SS1 no shock signs, SS2 mild shock signs, SS3 severe shock signs), with their corresponding duration of colic before presentation to the clinic, the intestinal disorder observed in surgery, and the type and degree of vascular compromise as well as the plasma trypsin activity (EAT in ng/mL) on admission. EAT values of control were also mentioned. ∗
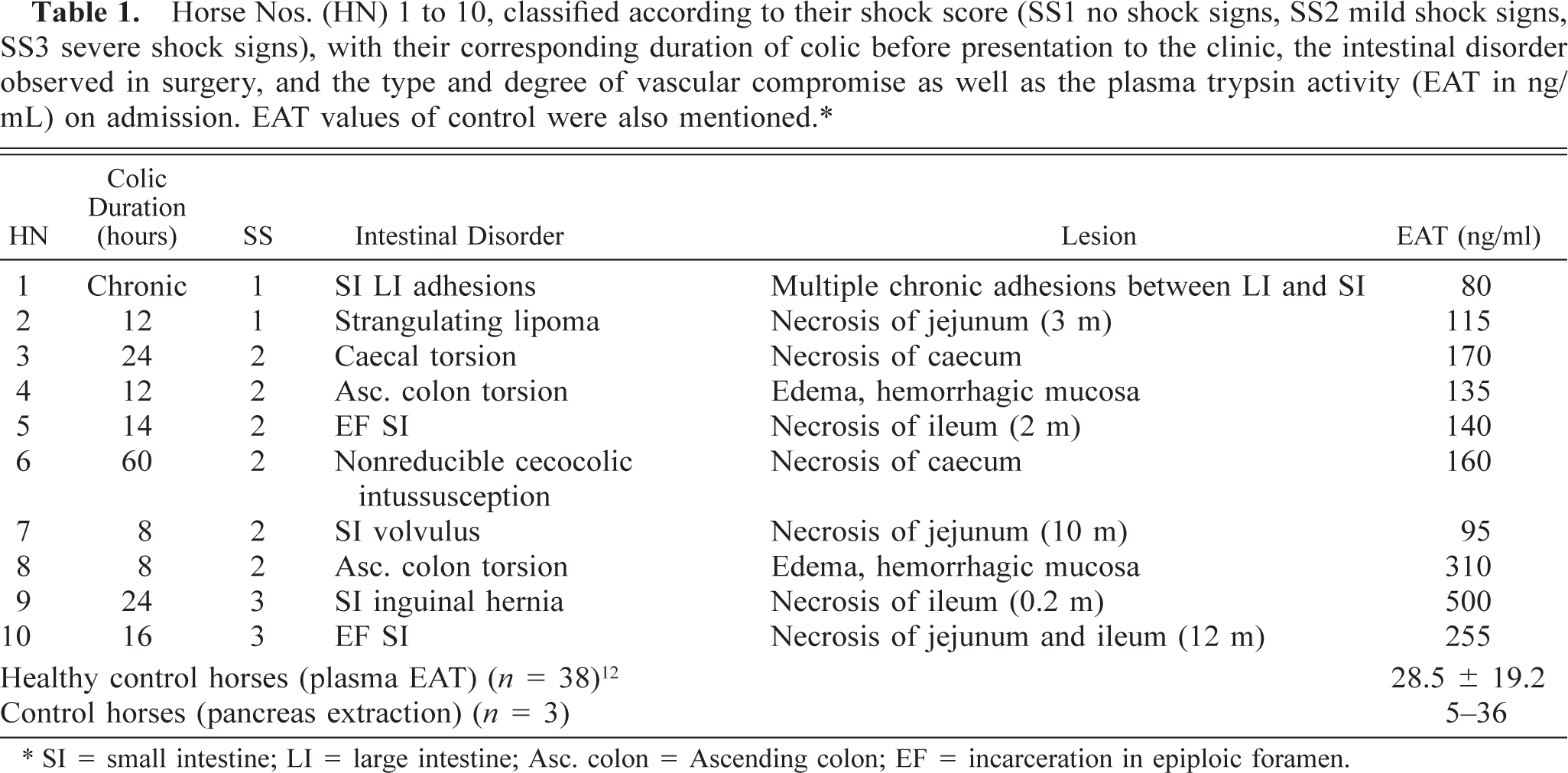
∗SI = small intestine; LI = large intestine; Asc. colon = Ascending colon; EF = incarceration in epiploic foramen.
Plasma EAT activity
The control horses had normal trypsin activity and horse No. 1 had plasma EAT approaching the maximal reference value, whereas all the other horses had high trypsin activity. Statistical analysis showed a mean value of 196 ng/ml ± 128.2 ranging from 80 to 500 ng/ml, differing significantly from the mean control (28.5 ng/ml ± 19.2) of healthy horses (P = 0.0026). Table 1 shows the corresponding EAT value of each case as well as the EAT mean value of the healthy control.
Histopathologic and ultrastructural evaluation
Histopathologic evaluation showed that the general structure of the pancreas was visible in all the preparations (three control horses and 10 horses suffering from intestinal obstruction). The exocrine pancreas showed disseminated endocrine islets. The exocrine pancreatic tissue was composed of glandular epithelial cells of pyramidal form. The acinus organization was not readily visible. The nucleus was large and hyperchromatic in parabasal position. Electron transmission micrographs of pancreatic cells of the control horses had well-conserved ultrastructural features (rough endoplasmic reticulum, mitochondria, zymogen granules, nucleus).
Horse No. 1 also had a well-conserved exocrine pancreatic tissue. Ultrastructural features of pancreatic cells from horse No. 1 were only slightly vacuolated. The rough endoplasmic reticulum (RER) was distributed throughout the cytoplasm with visible ribosomes well attached to the membrane. Mitochondria were intact. Few granules were seen at the apical pole of the acinar cells (Fig. 1). Horse No. 2 presented similar pancreatic aspect. The other horses and especially Nos. 9 and 10 had white vacuoles of variable size in the cytoplasm (degeneration). Other cells were distended and presented more eosin-staining cytoplasm (intracellular edema). Cell contours were less defined, with a disorganized aspect. Horse No. 10 also showed very pale nuclei. The RER from pancreatic cells of horses Nos. 3–10, with more severe shock, was vacuolated. The RER of acinar cells was markedly dilated and filled with amorphous material. The ribosomes were still attached to the RER membrane and some mitochondria appeared intact. Few granules were still present (Fig. 2). Several horses (especially horses Nos. 9 and 10) also showed mitochondrial swelling and disruption of the inner mitochondrial membrane, but the mitochondrial damage was not observed in all the mitochondria of the cell. Dilated cisterns of RER were observed with detached free ribosomes and homogenous material in the cisterns. Autophagic vacuoles were also visible (Fig. 3).
Discussion
In horses, pancreatic disease is not well documented. One study established normal values for amylase and lipase in blood and peritoneal fluid, 23 to develop diagnostic tools for pancreatic damage. The biochemical markers of pancreatic damage are routinely measured in pancreatic disease in humans, but in shock patients, plasma concentrations of pancreatic enzymes are not always assayed. Nevertheless, as early as in the 1970s, hyperamylasemia was observed after extra-abdominal surgery in humans. 20,29 Since these early observations, plasma increases of the pancreatic enzymes have been widely reported after invasive surgery and in critically ill patients, particularly when shock or sepsis were present. 2,5,21,31 These pancreatic enzyme increases occurred most often without clinical signs of pancreatitis, and histologic evaluation did not always show lesions especially where light microscopy was used. 30
Pancreatic involvement in equine acute abdominal disease was previously demonstrated by biochemical assay. 12 As in humans and other species, amylase can originate from salivary glands and lipase can be released from the liver, so that increased concentrations in blood were not specific signs of pancreatic injury. 33 The plasma trypsin activity was measured to evaluate pancreatic damage. Trypsin is specific to the pancreas and has a key function because of the activation of the pancreatic enzyme cascade. 24 It activates its own zymogen and those of all the other pancreatic enzymes. In this study, increased blood concentrations of trypsin were found, and this biochemical marker of pancreatic damage was accompanied by tissue alterations of the pancreas as observed by light and electron transmission microscopy.
The histopathologic and ultrastructural changes observed in the pancreatic tissue were vacuolar degeneration, swollen or partially destroyed mitochondria, RER enlargement, and detachment of ribosomes. Several studies on the human pancreas showed similar lesions in patients dying of severe trauma or septic shock. 16,32 The horse (No. 10) presenting the most severe pancreatic damage had incarceration of jejunum and ileum (10 m) in the epiploic foramen and had severe signs of shock, so that hypoperfusion as well as direct compression of the pancreas could have been the initial cause of the damage. The pancreatic lesions we observed can thus be linked to ischemic damage after hypoperfusion or mechanical compression. They cannot, however, be attributed to autodigestion of the gland because the pancreas was removed before euthanasia and the tissue samples were excised and treated directly in the surgery. Moreover, the control pancreatic samples were obtained under the same conditions from horses without shock and they showed normal structure.
A previous study of Tribl and collaborators 30 found decreased pancreatic function in patients with septic shock, but not in nonseptic shock patients. Thus, hypoperfusion was not the only mechanism involved in the observed pancreatic pathology, but the activation of neutrophils may also play a role. We previously demonstrated that neutrophils were stimulated in acute abdominal disease and measured increased plasma concentrations of myeloperoxidase, a marker of neutrophil degranulation. 11 Myeloperoxidase and other inflammatory mediators may be released by aggregation and trapping of neutrophils in splanchnic capillaries, exacerbating the ischemia-reperfusion injury to the pancreas and the local inflammatory reaction. But no infiltration of the pancreatic tissue by leucocytes was observed in our study.
Whatever the mechanisms that caused pancreatic damage, our observations demonstrated that the pancreas was involved in the pathophysiology and evolution of equine acute abdomen. Pancreatic failure cannot be regarded unequivocally as the cause of the acute abdomen, but it may be an aggravating factor in the disease. When acinar cells release active trypsin, the consequences need not be limited to intrapancreatic activation of the other zymogens and damage only the pancreatic tissue. 24 Active trypsin in blood plays a role in the activation of the complement and coagulation cascade, on neutrophil stimulation, and on protein degradation and proenzyme activation, despite the presence of plasma anti-proteases as demonstrated in pancreatitis. 10,22 The activation of the blood cascades and the release of active enzymes may overload the anti-protease capacity of the blood and trypsin bound to α2-macroglobulin (its main plasmatic inhibitor) remains active against low molecular weight substrates. 18 These systemic effects contribute to morbidity and the onset of multiple organ failure. 13 But in horses, little is known about the effect of trypsin in blood. More cases should be investigated to determine whether pancreatic damage could explain various mechanisms of continuing shock even after the resection of devitalized intestine.
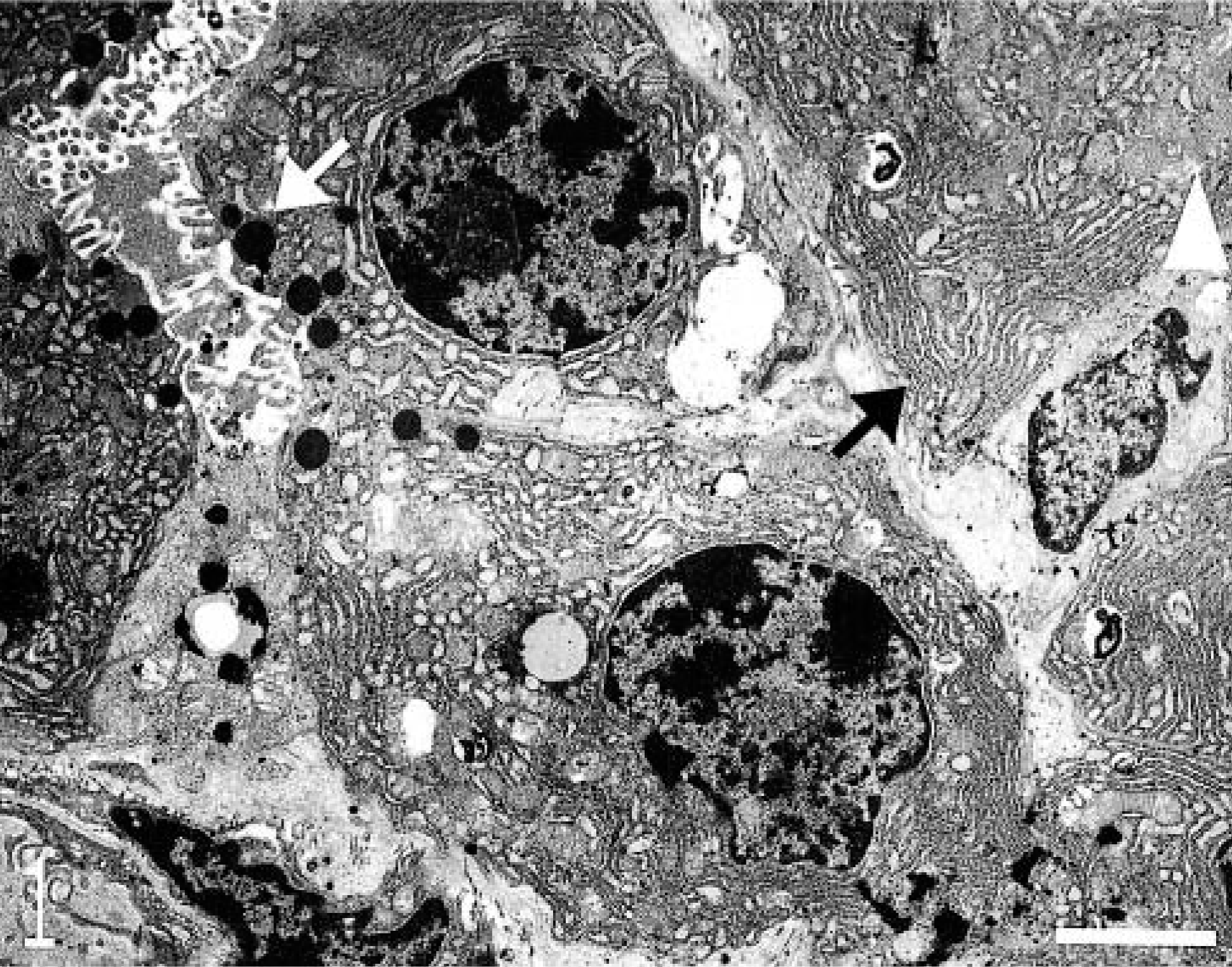
Transmission electron micrograph. Pancreas; horse No. 1. Acinar cell of exocrine pancreas with normal rough endoplasmic reticulum (RER) distributed throughout the cytoplasm (black arrow) and normal mitochondria (white arrowhead). Few zymogen granules (white arrow). Bar = 2 μm.

Transmission electron micrograph. Pancreas; horse No. 10. Acinar cells of exocrine pancreas presenting vacuolated rough endoplasmic reticulum (black arrows) with still attached ribosomes but filled with amorphous material. Very few zymogen granules (white arrow). Bar = 2 μm.

Transmission electron micrograph. Pancreas; horse No. 9. Acinar cells and capillary. Extensively dilated cisterns of RER, detachment of ribosomes (black arrowhead). Swelling of some mitochondria with disruption of the inner membrane (black arrow). Autophagic vacuole (white arrow). Bar = 2 μm.
Footnotes
Acknowledgements
This work was supported by a FRSM (National Fund for Scientific Medical Research—Belgium) grant no 3.4615.98. We thank J.F. Bradfer of the Electron Microscopy Unit of the Faculty of Veterinary Medicine (University of Liège) for technical assistance, as well as Prof. Dessy of the Department of Histology and Dr. E. Jacquinet of the Department of Pathology for scientific advice.
