Abstract
Porcine circovirus type 2 (PCV2) is associated with several syndromes in growing pigs, including postweaning multisystemic wasting syndrome and porcine dermatitis and nephropathy syndrome. In the present study, a previously undescribed neurovascular disorder associated with a PCV2 infection is described. Sixteen pigs showed clinical signs of wasting and neurologic deficits. Acute hemorrhages and edema of cerebellar meninges and parenchyma due to a necrotizing vasculitis resulted in degeneration and necrosis of the gray and white matter. Few to numerous PCV2 DNA and antigen-bearing endothelial cells were detected in affected areas of the brain using in situ hybridization and immunohistochemistry. Conventional histochemical stains, as well as the detection of caspase 3 activity and DNA strand breaks by the terminal transferase dUTP nick end labeling assay, showed numerous apoptotic endothelial cells in the vascular lesions observed. Sequencing of various brain-derived PCV2-specific amplicons revealed a strong identity between different isolates and an 89 to 100% identity to previous isolates. The phylogenetic tree showed that there was no clustering of isolates correlating to clinical signs or geographic distribution. This previously undescribed PCV2-associated neurologic disease has features of both postweaning multisystemic wasting syndrome and, to a lesser extent, porcine dermatitis and nephropathy syndrome. The available evidence suggests that direct virus-induced apoptosis of endothelial cells plays a role in the pathogenesis of this unusual PCV2-associated cerebellar vasculitis.
Introduction
The genus Circovirus belongs to the Circoviridae family and includes small, single-stranded, nonenveloped DNA viruses with a circular genome. They have a wide host range, including pigeons, psittacidae, finches, geese, ducks, and pigs. 5, 53 Porcine circoviruses (PCVs) are the only ones of this family that infect mammals. 51 Two PCVs, types 1 and 2, have been identified so far. 18 Porcine circovirus type 1 is considered nonpathogenic for pigs, whereas PCV2 is commonly accepted as a major pathogen for swine. 43, 44, 51, 52
Porcine circovirus type 2 is highly prevalent in swine herds worldwide, and infection is often subclinical; however, PCV2 has also been associated with multiple clinical disease syndromes, including postweaning multisystemic wasting syndrome (PMWS), porcine dermatitis and nephropathy syndrome (PDNS), reproductive failure, respiratory disease complex, proliferative and necrotizing pneumonia, and possibly congenital tremor. 11, 19, 43, 45, 48 Investigations of various PCV2 strains obtained from swine herds with a high incidence of PMWS and animals with PCV2 infection without clinical signs revealed several base exchanges, but virulence-associated genomic clusters were not demonstrated. 10, 18, 31, 32
Postweaning multisystemic wasting syndrome is characterized clinically by wasting, dyspnea, and lymphadenopathy. 19, 40 The most prominent histologic lesions in PMWS occur in lymphoid organs and consist of extensive lymphoid depletion, infiltrates of macrophages and fewer multinucleated giant cells, and intracytoplasmic inclusion bodies. Protein or DNA of PCV2 has been demonstrated by immunohistochemistry and/or in situ hybridization in affected and unaffected organs in the cytoplasm of macrophages; multinucleated giant cells; dendritic cells in lymphoid tissues; alveolar macrophages; Kupffer cells; and, to a lesser extent, epithelial cells in lung, liver, pancreas, and kidneys. Endothelial cells in several organs, including lymph nodes and spleen, are rarely positive for PCV2 DNA. 2, 37, 40 Lesions in the brain are detected only rarely in PMWS. 40, 43 A direct involvement of the brain or spinal cord during PCV2 infection has only been suggested in congenital tremor type A II in neonatal piglets but has not been confirmed by others, and therefore its existence is still controversial. 16, 20, 48 Although vascular lesions are not prominent in PMWS, vasculitis is the hallmark of PDNS. 43 Lesions in PDNS are characterized primarily by a cutaneous and subcutaneous necrotizing vasculitis and glomerulonephritis and are likely mediated by immune complex deposition. 46 Porcine circovirus type 2 and porcine respiratory and reproductive syndrome virus (PRRSV) are considered the most likely causative agents. 42 However, to date PCV2 antigen has not been detected within PDNS vascular lesions. 24, 43
This report describes 16 cases of a neurodegenerative disorder in wasting pigs due to a PCV2-associated cerebellar vasculitis, which exhibits some characteristics of PMWS and fewer changes consistent with PDNS.
Materials and Methods
Animals and clinical signs
Between 1985 and 2004, 16 Landrace pigs from different farms with a body weight of between 5.4 and 19.1 kg and an approximate age of 6 weeks to 2 months showed clinical signs of wasting, coughing, and neurologic deficits with opisthotonus, nystagmus, and convulsions. Death occurred spontaneously within a few hours after the onset of neurologic signs, or the animals were killed due to poor prognosis; they were submitted for necropsy.
Tissue samples, histology, and histochemistry
Specimens from the central nervous system (CNS), lymphoid organs, and a variety of other tissues and organs, including lung, heart, small and large intestine, bone marrow, liver, skin, and kidney, were collected at necropsy, fixed by immersion in 4% neutral-buffered formaldehyde, embedded in paraffin wax, cut at 3-μm thickness, and routinely stained with hematoxylin and eosin. Selected tissue sections of cerebrum, cerebellum, and brain stem were stained with Luxol-Fast-Blue cresyl echt violet and periodic acid-Schiff. In addition, samples from cerebellum, cerebrum, and lymph nodes from pig Nos. 6, 8, 9, 12, 13, and 15 were immediately snap frozen in liquid nitrogen. For additional etiologic examinations, tissues obtained immediately at necropsy from jejunum, lung, and mesenteric and bronchial lymph nodes of all animals were screened for PRRSV, porcine respiratory corona virus, porcine herpes virus type 1, porcine influenza virus, classical swine fever, and Escherichia coli bacteria in commercial and state laboratories.
Immunohistochemistry and in situ detection of DNA strand breaks
Immunohistochemistry was applied to formalin-fixed and paraffin-embedded tissue as previously described. 9, 33, 34 Briefly, deparaffinized and rehydrated sections were incubated with 1.5% hydrogen peroxide in methanol to block endogenous peroxidase activity. To reduce background staining, sections were mounted on Coverplates (Fa. Shandon, Germany) and incubated with 10% normal goat serum for 1 hour at room temperature. Subsequently, slides were incubated with antibodies against PCV2 (Ab 36A9, Ingenasa, Spain), glial fibrillary acidic protein (Z0334, Dako, Germany), neurofilament (clone 2F11, Dako, Germany), vimentin (clone V9, Dako, Germany), MAC387 (MAC387, Dako, Germany), lysozyme (Z0334, Dako, Germany), CD3 (clone 6F2, Dako, Germany), CD79α (clone HM57, Dako, Germany), factor VIII–related antigen (A0099, Dako, Germany), immunoglobulin (Ig) G (A100-104F, Bethyl Laboratories, USA), IgM (A100-100F, Bethyl Laboratories, USA), IgA (A100-102F, Bethyl Laboratories, USA), complement factor C3 (GAD/C3c, Nordic Immunological Laboratories, Denmark), and cleaved Caspase-3 (Asp175, Cell Signaling Technology, USA) to detect PCV2 antigen, astrocytes, neurons, mesenchymal cells, macrophages, T- and B-cells, endothelial cells, immunoglobulines, complement factor, and apoptotic cells. All primary antibodies were incubated overnight at 4°C in a humidified chamber. Antibody dilutions and tissue pretreatments are summarized inTable 1. After several washings with phosphate-buffered saline (PBS), sections were incubated with the secondary antibody for 30 minutes at room temperature in a humidified chamber. Finally, visualization of positive reactions was performed using the avidin-biotin-peroxidase complex method (Vectastain ABC kit and peroxidase substrate kit DAB, Vector Laboratories, USA) according to the manufacturer's instructions. Specificity of the primary antibody against PCV2 was confirmed by using formalin-fixed, paraffin-embedded cell pellets from PK15 cells that were either infected or noninfected with PCV1 and 2.
Antibodies and reaction conditions used for immunohistochemistry to determine cell types, mechanisms of cell death, pathogenesis of lesions and virus distribution.
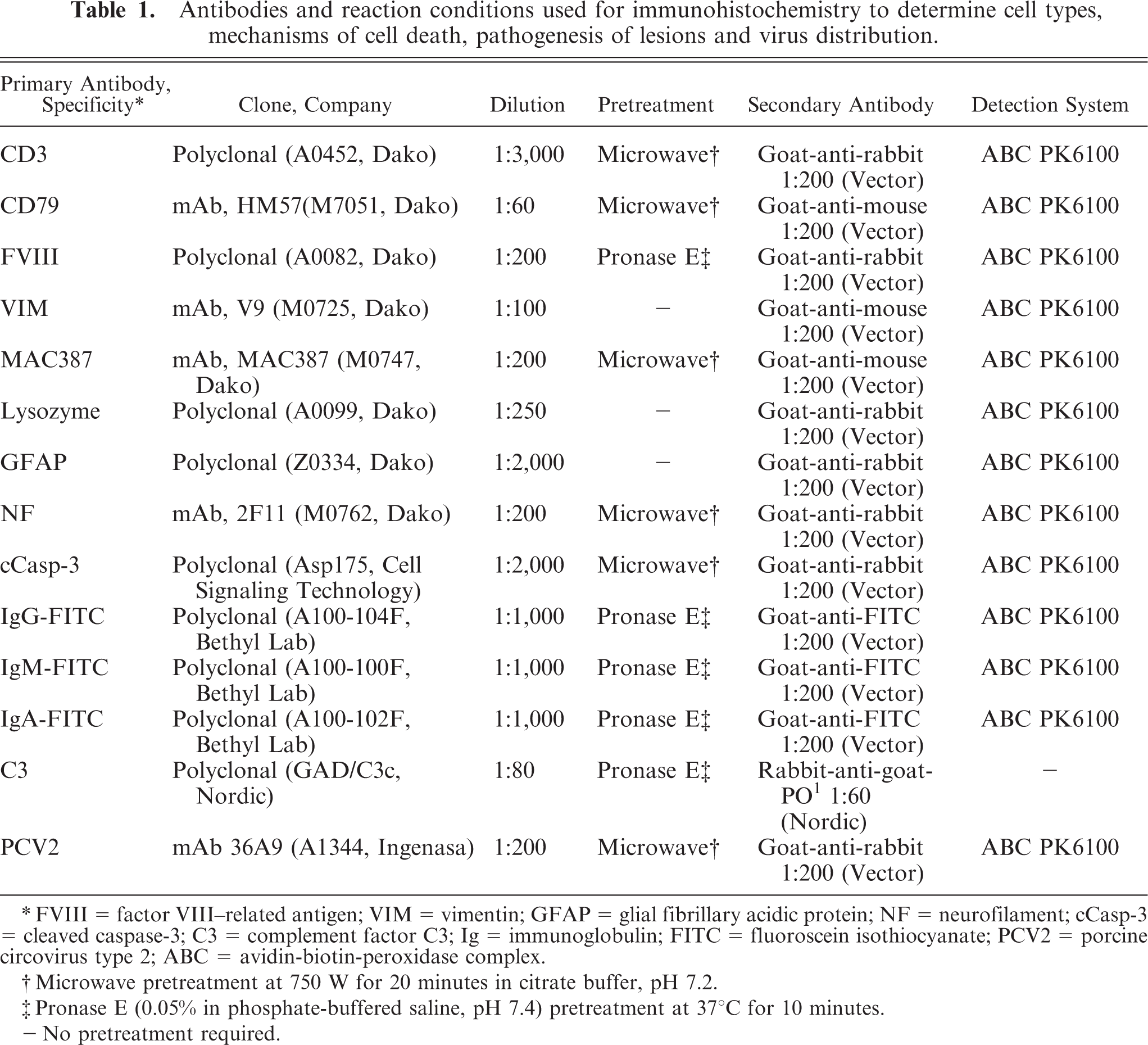
∗ FVIII = factor VIII–related antigen; VIM = vimentin; GFAP = glial fibrillary acidic protein; NF = neurofilament; cCasp-3 = cleaved caspase-3; C3 = complement factor C3; Ig = immunoglobulin; FITC = fluoroscein isothiocyanate; PCV2 = porcine circovirus type 2; ABC = avidin-biotin-peroxidase complex.
† Microwave pretreatment at 750 W for 20 minutes in citrate buffer, pH 7.2.
‡ Pronase E (0.05% in phosphate-buffered saline, pH 7.4) pretreatment at 37°C for 10 minutes.
— No pretreatment required.
Detection of DNA strand breaks was performed by terminal deoxynucleotidyl transferase-mediated dUTP-biotin nick end labeling (TUNEL) on serial sections. The ApopTaq Plus peroxidase in situ apoptosis detection kit (S7101, Chemicon, USA) was used according to the manufacturer's instructions.
In situ hybridization for detection of porcine circovirus type 2
For in situ hybridization, a PCV2-specific, digoxigenin-labeled oligonucleotide probe (DIG-5′-CCTTCCTCATTACCCTCCTCGCCAACAATAAAATAATCAAA-3′) was designed based on the sequence of PCV2 ORF 1 (nt position 168–208, accession number AF027217) and was used as previously described. 5 A PCV1-specific, digoxigenin-labeled oligonucleotide probe (DIG-5′-CCCTCTTCCAAACCTTCCTCTCCGCAAACAAAAT AATCAAA-3′) was designed based on the sequence of PCV1 ORF 1 (nt position 155–195, accession number NC 001792) and was used as a control. As a second control, a digoxigenin-labeled oligonucleotide probe with the reverse sequence of the PCV2-specific probe (DIG 3′-AAACTAATAAAATAACAACCGCTCCTCCCATTACTCCTTCC-5′) was applied. Formalin-fixed, paraffin-embedded cell pellets from noninfected and PCV1- and -2 infected PK15 cells served as positive and negative controls.
DNA in situ hybridization was performed as previously described. 14, 15, 33 Briefly, 5-μm thick sections of paraffin-embedded tissues were placed on Superfrost plus slides (Menzel-Gläser, Germany). All steps were performed in water treated with DNase-free glassware and diethyl pyrocarbonate. After deparaffinization in a descending alcohol series, sections were denatured with 0,2 HCl for 20 minutes. After 2 washing steps in 2 × standard saline citrate buffer, digestion was performed with proteinase K (Roche, Austria) at a concentration of 2 μg/ml for 15 minutes at 37°C. Digestion was stopped by incubation in 2% glycin PBS for 5 minutes and postfixation in 4% paraformaldehyde for 4 minutes. After 2 washing steps in 2 × standard saline citrate buffer, acetylation occurred in 0.25% acetanhydride in 0,1 M triethanolamine (pH 7) for 10 minutes. For prehybridization, sections were covered with a mixture of 450 ml 20 × standard saline citrate buffer, 675 ml 100 % deionized formalin, and 150 ml 50 × Denhardt's (Sigma, Austria) solution solved in 210 ml DNAse-free glassware and diethyl pyrocarbonate water and incubated at 55°C for 1 hour in a humidified chamber. For hybridization, the DIG-labeled oligonucleotide probe was adjusted to a concentration at 100 ng/μl in the hybridization mixture (16 ml 100% formamid, 8 ml 20 × hybridization salts, 3.2 ml 50 × Denhard's solution, 320 μl heparin, 320 μl 10% triton X, 18 μl RNS, 80 μl dextran sulfate). Hybridization was performed for 16 hours at 56°C in a humid chamber. A posthybridization hot wash was performed in 4 × saline sodium citrate + 30% formamid for 15 minutes at 48°C, 2 × saline sodium citrate for 5 minutes at room temperature, and 2 times in 0.2 × saline sodium citrate at 50°C for 15 minutes. After several washing steps, slides were incubated with a blocking reagent (1.2 ml sheep serum, 1.8 ml Triton-X-100, 60 ml 1M Tris-HCl, pH 7.5) for 30 minutes. For visualization of the reaction, sections were incubated with an antidigoxigenin, alkaline phosphatase-conjugated antibody (Fa. Boehringer-Mannheim, Germany, diluted 1:200 in blocking reagent) and subsequently stained with X-phophate-nitroblue-tetrazolium. After stopping the color reaction (1M Tris HCl, pH 8, with 0.5 M EDTA in 1,000 ml Aqua bidest), slides were counterstained with hematoxylin and covered with warm Glycergel (Fa. Dako).
In situ hybridization and immunohistochemistry double labeling
Double labeling to detect PCV2 genome and factor VIII–related antigen in formalin-fixed, paraffin-embedded sections of selected animals was performed as previously described. 15 Briefly, DNA in situ hybridization to visualize PCV2 genome was performed as described above. After stopping the final color reaction of the nitro blue tetrazolium solution and several washing steps using PBS, slides remained in PBS for 15 minutes. Thereafter, pronase E (0.05% in PBS, pH 7.4) pretreatment at 37°C for 10 minutes was used for demasking. Subsequently, slides were incubated with blocking reagent (10% normal goat serum) for 1 hour and incubated with the primary antibody directed against factor VIII–related antigen (A0082, Dako) and visualized with the Substrate-Chromogene Kit (K3469, Dako, Germany). The positive signal was red or blue for immunohistochemistry or in situ hybridization, respectively. Double staining of viral DNA and cell decoration resulted in a cytoplasmic pinkish-blue mixed color or the presence of both colors in the same cell.
Porcine circovirus type 2 isolation, polymerase chain reaction, and sequencing
For detection of the PCV2 genome, DNA was isolated from fresh brain tissue of pig Nos. 6, 8, 9, 12, 13, and 15, and polymerase chain reaction (PCR) was performed as previously described. 11, 12, 34 Briefly, DNA was isolated with an E.Z.N.A. Tissue DNA Mini Kit (PeqLab Biotechnology, Germany) according to the manufacturer's instructions. Subsequently, a 481-bp fragment within the region of the ORF2 was amplified with 2 primers specific for PCV2 (forward primer: p196 5′-CGGATATTGTAGTCCTGGTCG-3′ position 1095–1115; reverse primer: p197 5′-ACTGTCAAGGCTACCACAGTCA-3′ position 1570–1549; GenBank accession number AY484414). Initial denaturation was performed for 4 minutes at 94°C, followed by 35 cycles denaturation at 94°C for 1 minute, annealing at 65°C for 1 minute, and elongation at 72°C for 1 minute. The reaction ended with a final extension step at 72°C for 10 minutes. The amplicon was visualized by standard gel electrophoresis on a 2% agarose gel. Positive DNA fragments of specific sizes were cut out from the gels, purified by using a commercial kit (Nucleobond, Macherey & Nagel, Germany) and submitted for sequencing. The latter was performed according to the method of Sanger with the fluorescent-labeled BigDye Terminator sequencing kit using automatic sequencers (Applied Biosystems, USA) by an extern laboratory (SEQLAB, Germany). 6 Primers p196 and p197 served as forward and reverse primers, respectively, for sequencing. Sequences were analyzed with LALIGN online version (http://www.ch.embnet.org) to confirm identity and the CLUSTAL W online version (http://www.ebi.ac.uk/clustalw/index.html) for comparative alignments and were submitted to DDBJ/EMBL/GenBank database (PCR products from pig Nos. 6, 8, 9, 12, 13, and 15 accession numbers DQ174680 to DQ174685). PK15 cells that were not infected or infected with PCV1 and 2 served as positive or negative controls for DNA isolation and PCR. For constructing the phylogenetic tree, sequences from PCV2 isolates from different geographic locations and from animals displaying different clinical signs were downloaded from the EMBL/GenBank. They were aligned using CLUSTAL X version 1.83. 48 Alignments were optimized using BioEdit version 4.0.7.1. 17 The phylogenetic tree was calculated with the neighbor-joining algorithm as implemented in CLUTAL X; for calculating the bootstrap values, 1,000 repeats were performed. 49
Results
Necropsy findings
All animals displayed a poor body condition characterized by wasting and a rough hair coat. Similar gross lesions, consisting of a moderate to severe edema in the subcutis, mesentery, meninges, and gall bladder wall, were found in all pigs. Furthermore, a generalized, mild to moderate enlargement of visceral and peritoneal lymph nodes, most prominent in mesenteric lymph nodes, was detected. The cut surfaces of the mesenteric lymph nodes showed well-demarcated yellow and dry areas. Acute congestion and a diffuse alveolar and interlobular edema with mild to moderate areas of consolidation were found in the lungs of most animals. In the CNS, similar lesions consisting of moderate to severe acute hemorrhages randomly distributed in the meninges and parenchyma predominantly in the cerebellar gray and white matter were found in all animals (Fig. 1). The cerebral hemispheres were rarely and only mildly affected. The medulla oblongata and brain stem, as well as cerebral nuclei, were unremarkable.
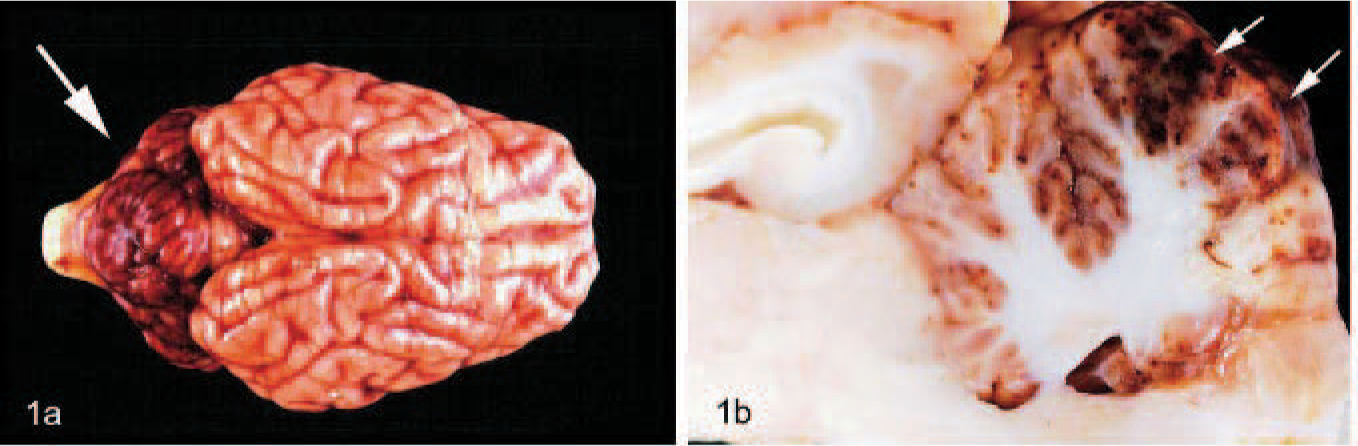
Pig No. 6, brain.
Histopathologic findings
The light microscopic examination of the brain revealed multifocal to disseminated, acute, severe leptomeningeal and parenchymatous hemorrhages and a moderate to severe lymphohistiocytic, necrotizing vasculitis of small to medium arteries and veins mainly in the cerebellum (Fig. 2). The alterations in the walls of small vessels ranged from diffuse, homogeneous, eosinophilic changes to dense infiltrates of macrophages, lymphocytes, and fewer neutrophils, thrombi, and perivascular hemorrhages. The granular, molecular, and Purkinje cell layer and the adjacent white matter of the folia were the most severely affected areas. Neuronal degeneration and necrosis, as well as a laminar spongiosis, were evident in the Purkinje and molecular cell layer of the cerebellum of most animals. The deeper white matter areas and neuronal nuclei, including the vestibular nuclei, were not affected. Some nuclei of endothelial cells showed typical signs of apoptosis, including chromatin condensation, karyorrhexis, and accumulation of 3 to 5 round, basophilic, nuclear fragments. The surrounding neuropil exhibited extensive edema with perivascular accumulation of protein-rich fluid (Fig. 3). In areas of extensive edema, mainly adjacent to the folia, a moderate to severe loss of luxol fast blue staining and erythrocyte-laden macrophages were noticed. Furthermore, a mild to moderate increase of gemistocytic astrocytes was detected in areas of severe edema, mainly in the white matter of the folia subjacent to the gray matter.
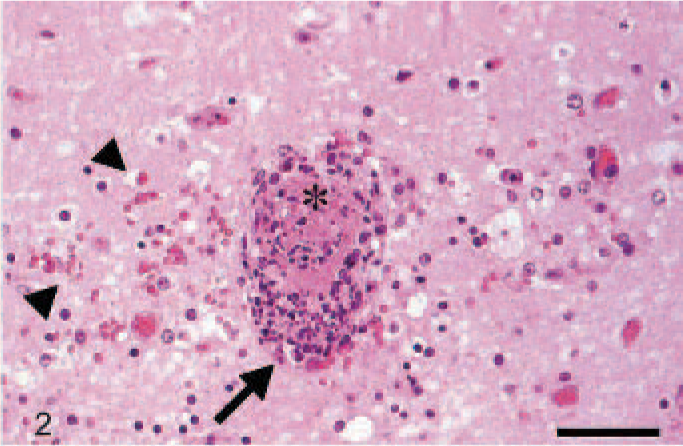
Pig No. 6, cerebellum. Necrotizing vasculitis (arrows), hemorrhages (arrow heads), and thrombosis (asterisk). HE stain. Bar, 50 μm.
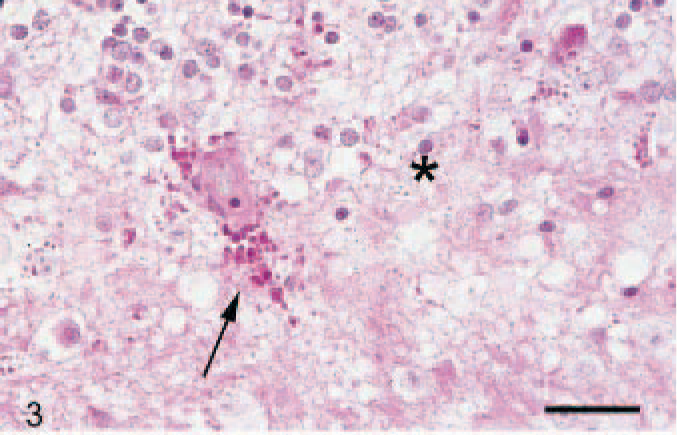
Pig No. 16, cerebellum. There is mild perivascular hemorrhage and proteinaceous fluid deposition (arrow) and moderate perivascular edema (asterisks). The vessel wall is mildly thickened and hypereosinophilic. Periodic acid-Schiff reaction. Bar, 25 μm.
Histologic findings of the remaining CNS and in non-nervous tissues are summarized inTable 2. Findings, most prominent in the mesenteric and pulmonary lymph nodes, were characterized by moderate to severe lymphoid depletion; histiocytosis; multinucleated cells; and numerous, cytoplasmic, basophilic, botryoid inclusion bodies in macrophages. Some mesenteric lymph nodes exhibited severe, diffuse coagulation necrosis and thrombi formation and, occasionally, necrotizing vasculitis. Lymphoid depletion was also commonly found in ileal Peyer's patches, the tonsils, and the spleen. Additionally, pig No. 12 showed a severe necrotizing hepatitis. A mild to moderate lymphohistiocytic, interstitial pneumonia was a common finding in most animals. Furthermore, 2 animals (Nos. 7 and 10) showed lung lesions consistent with proliferative and necrotizing pneumonia. Three animals, (Nos. 2, 6, and 14), showed moderate renal necroses and hemorrhages associated with vasculitis affecting the arcuate arteries and cortical arterioles. In addition, some animals showed mild interstitial histiocytic infiltrates in various organs, including the heart, kidney, liver, and pancreas.
Summary of histologic lesions in non-nervous organs and the spinal cord of pigs suffering from Circovirus type 2-associated encephalopathy.
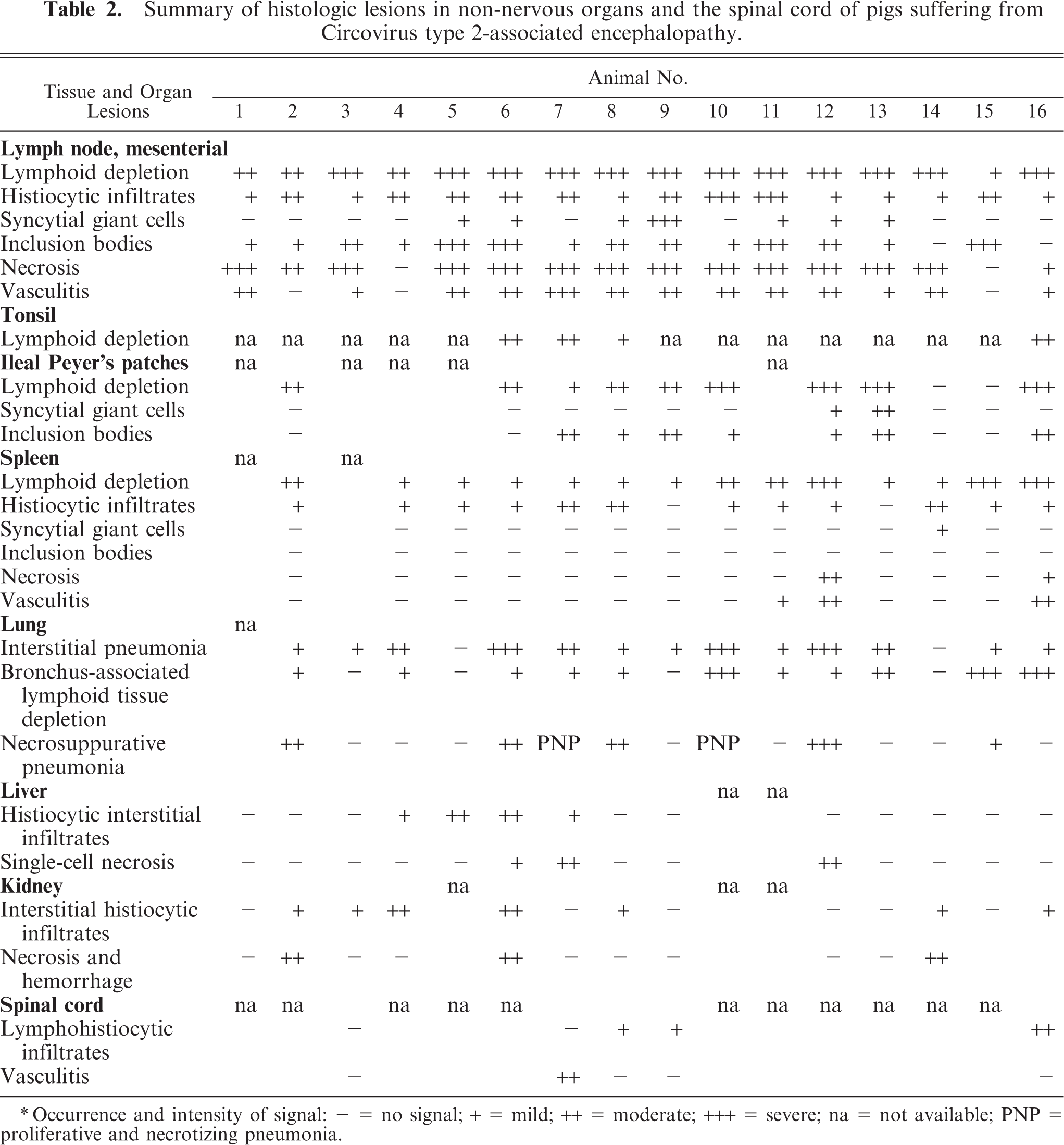
∗ Occurrence and intensity of signal: − = no signal; + = mild; ++ = moderate; +++ = severe; na = not available; PNP = proliferative and necrotizing pneumonia.
Phenotypical characterization of cells and detection of DNA strand breaks
Glial fibrillary acidic protein immunoreactivity of gemistocytic cells in areas of severe edema confirmed their identity as astrocytes. Neurofilament staining of neuronal cell bodies and axons showed a widespread disappearance of axons in the white matter in severely edematous areas. A few cells, expressing vimentin, MAC387, lysozyme, CD3, or CD79α, were detected perivascularly in meninges and the CNS tissue, especially in areas of hemorrhages. Furthermore, CD3-positive T lymphocytes and MAC387- and lysozyme-positive macrophages were found in vessels with severe leukocytic infiltrates. Antibodies against IgG, IgA, IgM, and C3 revealed positive signals in plasma accumulations in the lumen of all vessels. Furthermore, weak staining for these antigens was found in the thickened walls of small arteries and arterioles and in perivascular accumulations of proteinaceous fluid in areas of severe edema but not in unaffected vessels. Positive immunohistochemical signals of cleaved caspase-3 were found in cerebellar endothelial cells and in a few intra- and perivascular leukocytes of all animals (Fig. 4). This reaction was associated with moderate to severe vascular lesions and occasionally associated with apoptotic figures. There were also single positive cells in the surrounding neuropil or intravascular in areas of severe vascular leakage. Additionally, a similar distribution pattern was detected using the TUNEL assay; however, the number of positive endothelial cells was reduced to one third, compared with caspase-3 immunohistochemistry.
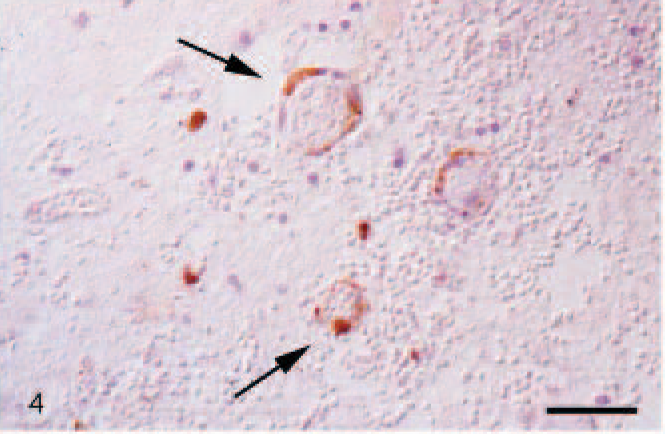
Pig No. 7, cerebellum. Immunohistochemical detection of cleaved caspase-3 (arrows) endothelial cells slightly counterstained with hemalaun. Avidin-biotin-peroxidase complex method. Bar, 50 μm.
Detection of porcine circovirus type 2–specific viral proteins and genome
In situ hybridization showed PCV2-specific signals for genomic DNA in the brains of all animals (Table 3). The specific dark blue signal was found in the cytoplasm of endothelial cells of small vessels affected by vasculitis and less frequently in unaffected vessels, mainly in the cerebellum and to a lesser extent in the cerebrum (Figs. 5, 6). Signal was also detected in occasional individual perivascular leukocytes. In animal Nos. 3, 7, and 10, positive signals were also found in endothelial cells of meninges and in the white matter of the spinal cord. Strong signals were also visible in the cytoplasm of histiocytic and syncytial giant cells in lymph nodes. A mild to severe reaction was also detected in leukocytes and resident histiocytic cells in tonsils, Peyer's patches of small intestine, spleen, bone marrow, lung, liver, heart, and kidney (Table 3). There were no detectable signals using the PCV1-specific oligonucleotide probe or the PCV2 reverse probe in the investigated tissues or controls.
Detection of the porcine circovirus type 2 genome by in situ hybridization in selected organs.
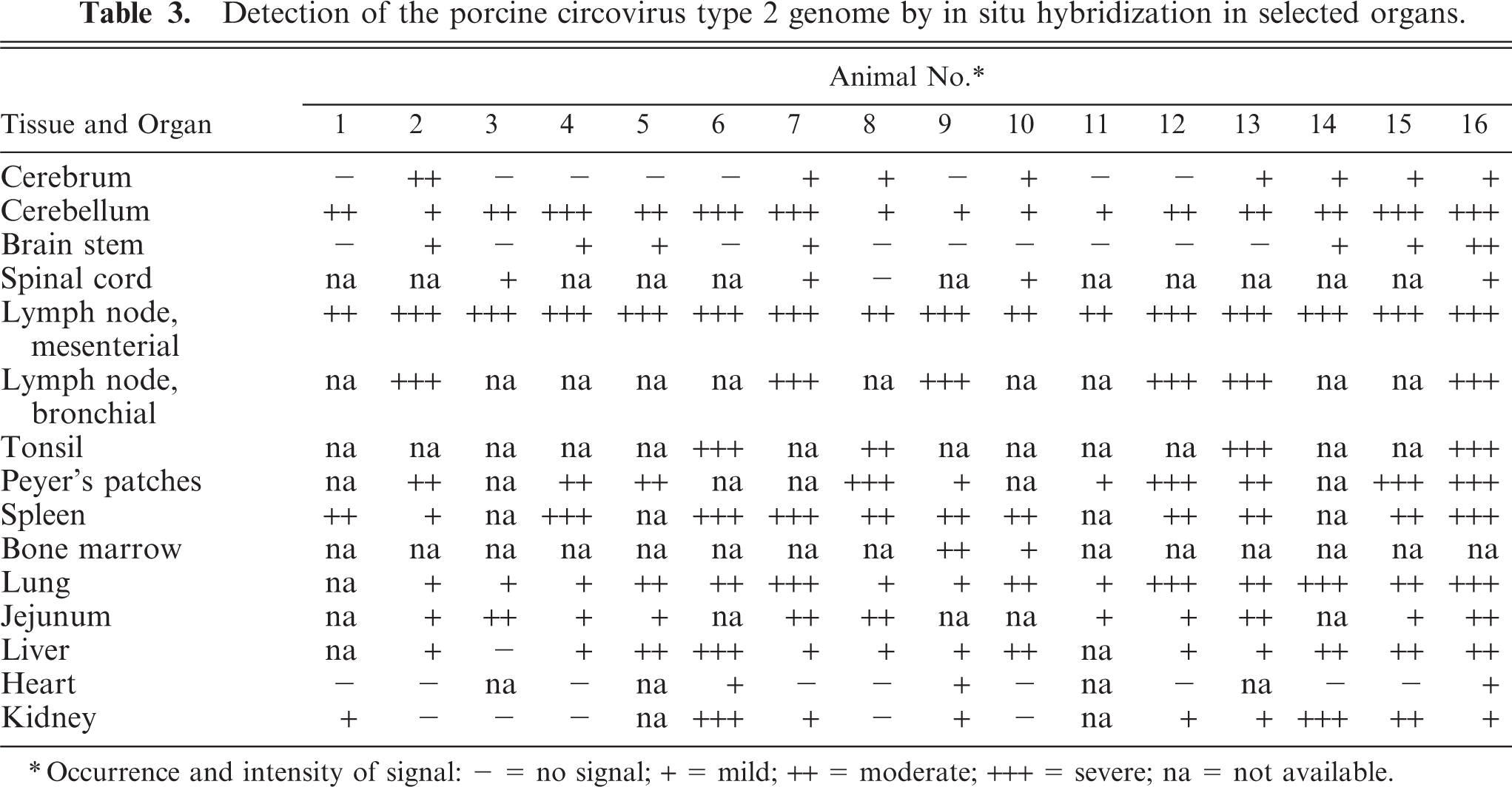
∗ Occurrence and intensity of signal: − = no signal; + = mild; ++ = moderate; +++ = severe; na = not available.
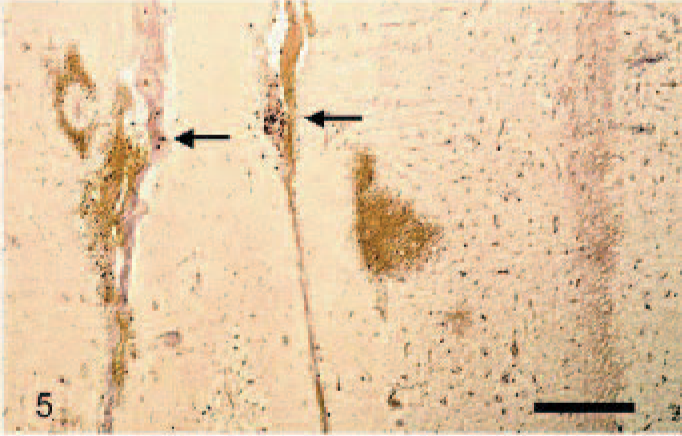
Pig No. 6, cerebellum. Porcine circovirus type 2–positive cells (arrows) in meninges; molecular Purkinje cell and granular cell layers; and areas of hemorrhage. In situ hybridzation. Bar, 200 μm.
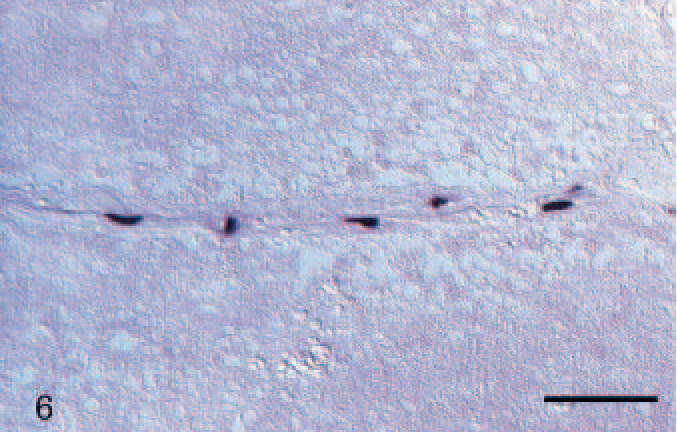
Pig No. 16, cerebellum. Porcine circovirus type 2–positive cells, presumably endothelial cells in a vessel. In situ hybridzation. Bar, 50 μm.
In situ hybridization results were confirmed by immunohistochemistry on selected tissues and organs. Immunohistochemical signals for PCV2 protein were detected in the brains of all animals, mainly in the cytoplasm of endothelial cells of small and medium vessels in affected and unaffected areas of the cerebellum (Fig. 7). Occasionally, intravascular and perivascular leukocytes showed a weak, cytoplasmic signal.
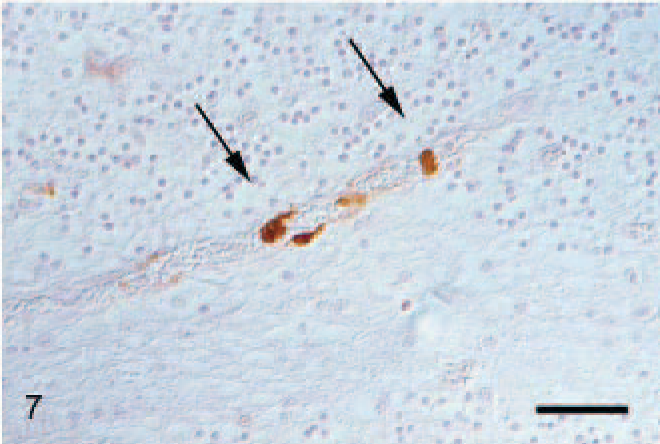
Pig No. 16, cerebellum. Immunohistochemical detection of PCV2 viral protein in endothelial cells (arrows) slightly counterstained with hemalaun. Bar, 50 μm.
The double-labeling technique using in situ hybridization specific for PCV2-DNA and immunohistochemistry against factor VIII–related antigen revealed coexpression of both signals in the cytoplasm of endothelial cells, predominantly in affected vessels (Fig. 8).
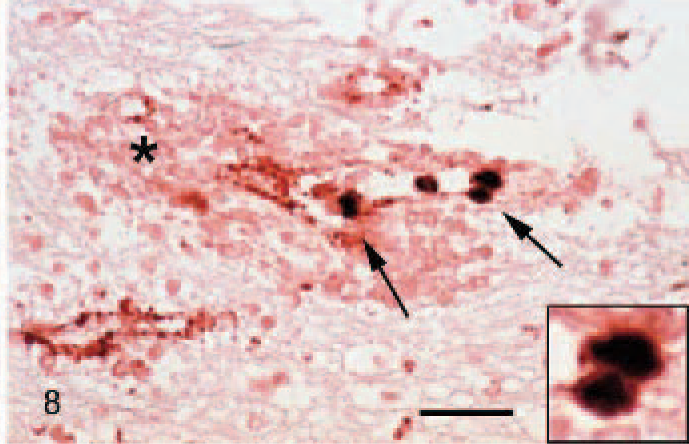
Pig No. 16, cerebellum. Detection of PCV2 DNA by in situ hybridization (bluish, arrows) and factor VIII–related antigen by immunohistochemistry (red, arrowhead) and extravascular erythrocytes (asterisks). Some endothelial cells show double labeling (arrows and inset). Bar, 50 μm.
Polymerase chain reaction and sequence alignments
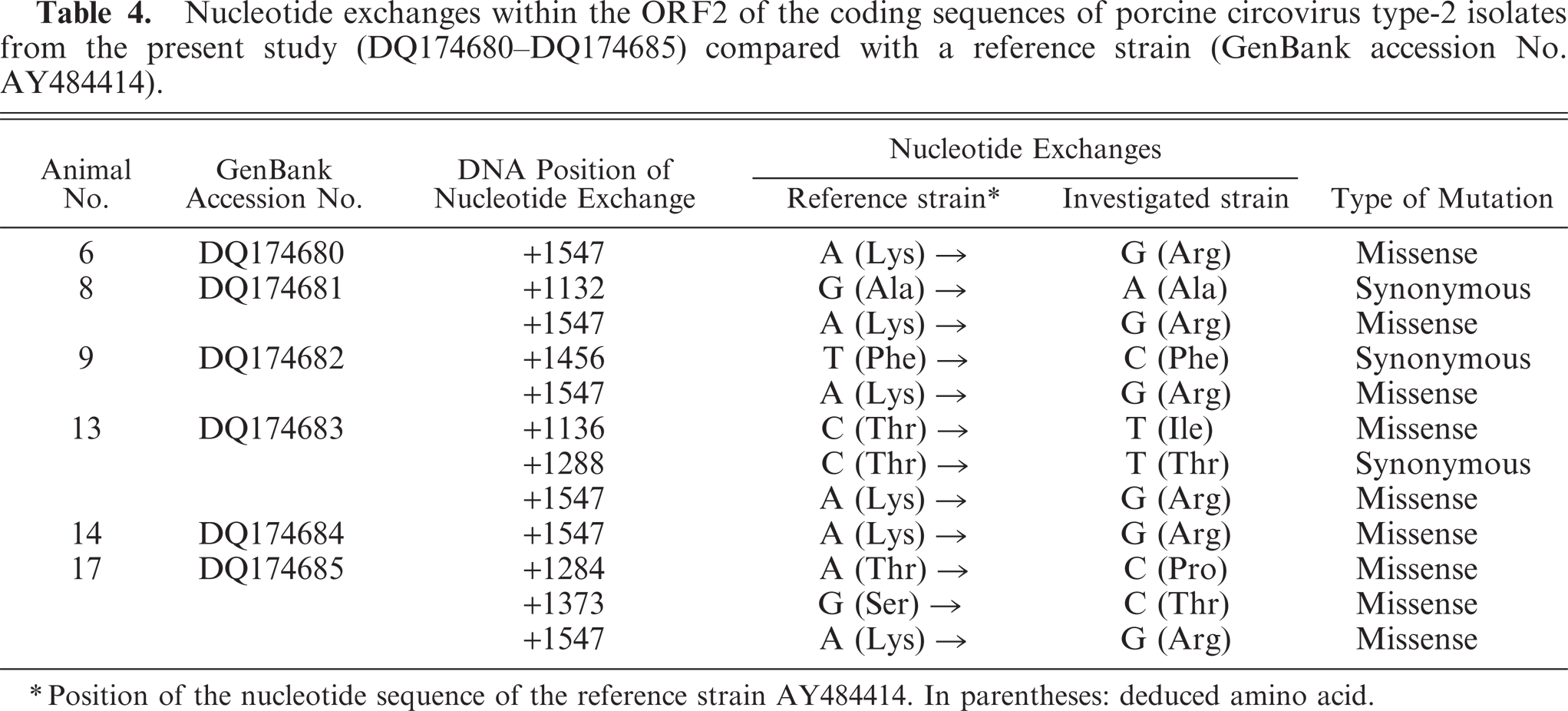
Polymerase chain reaction revealed PCV2-specific amplification products in the brains of 6 randomly selected pigs (Nos. 6, 8, 9, 12, 13, and 15;Fig. 9). Sequencing confirmed the identity of the PCR products as PCV2 specific. Several nucleotide exchanges were found in the 439-bp region of the ORF2 (Table 4). A strong identity of 99.3 to 99.8% between the different CNS isolates was found by CLUSTAL W analysis. BLAST analyses revealed a high identity between the different brain isolates and previously published PCV2 genome sequences ranging from 89.5 to 100%. The phylogenetic tree (Fig. 10) showed that there is no clustering of isolates correlating to clinical signs or geographic distribution. The 6 isolates analyzed in this study clustered together with isolates from different parts of Europe and China. Within this cluster, identities ranged between 99 and 100%. Identities with isolates in the other clusters, some of them consisting of only a single isolate, ranged between 89 and 92%.
Nucleotide exchanges within the ORF2 of the coding sequences of porcine circovirus type-2 isolates from the present study (DQ174680–DQ174685) compared with a reference strain (GenBank accession No. AY484414).
∗ Position of the nucleotide sequence of the reference strain AY484414. In parentheses: deduced amino acid.
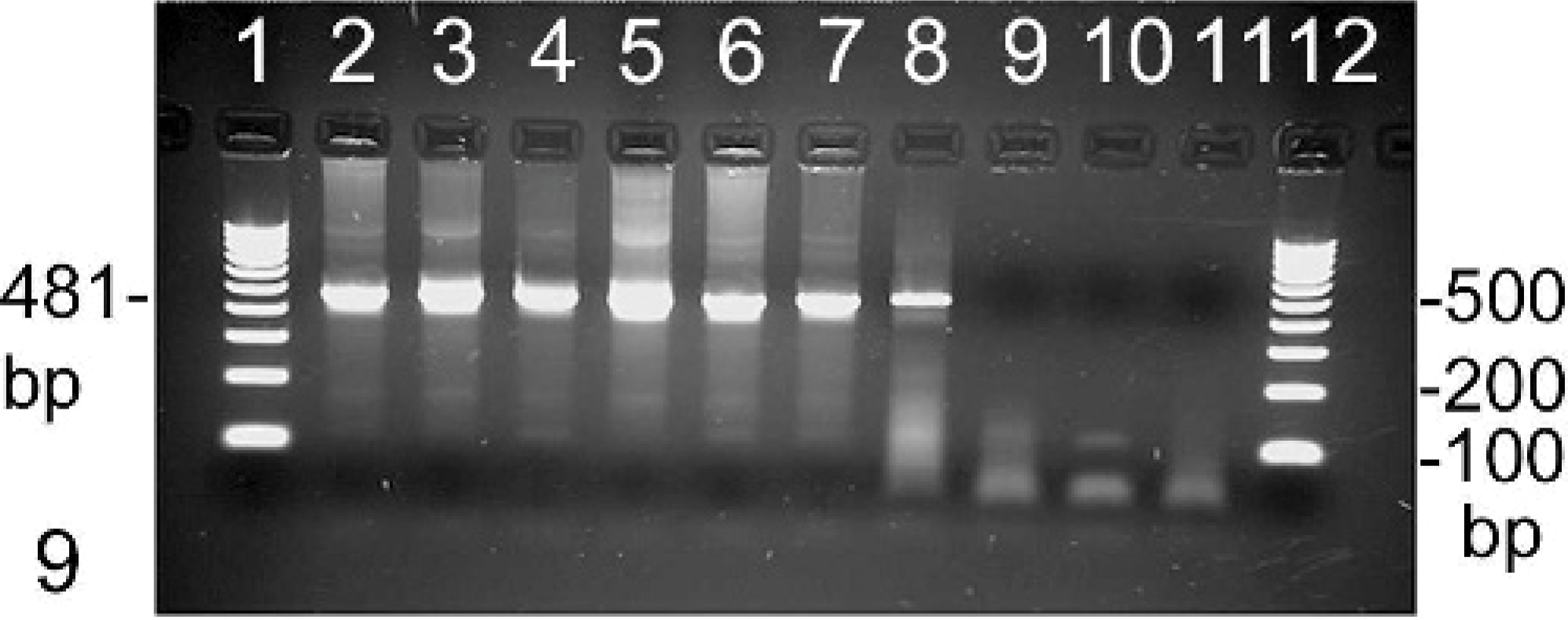
Identification of PCV2-specific amplicons in the cerebellum of affected pigs by PCR. Lanes 2 to 7, pig Nos. 6, 8, 9, 12, 13, and 15, respectively; lane 8, PK15 cells infected with PCV2; lane 9, PK15 cells infected with PCV1; lane 10, PK15 cells not infected; lane 11, nontemplate control; lanes 1 and 12, molecular size marker (100 bp).
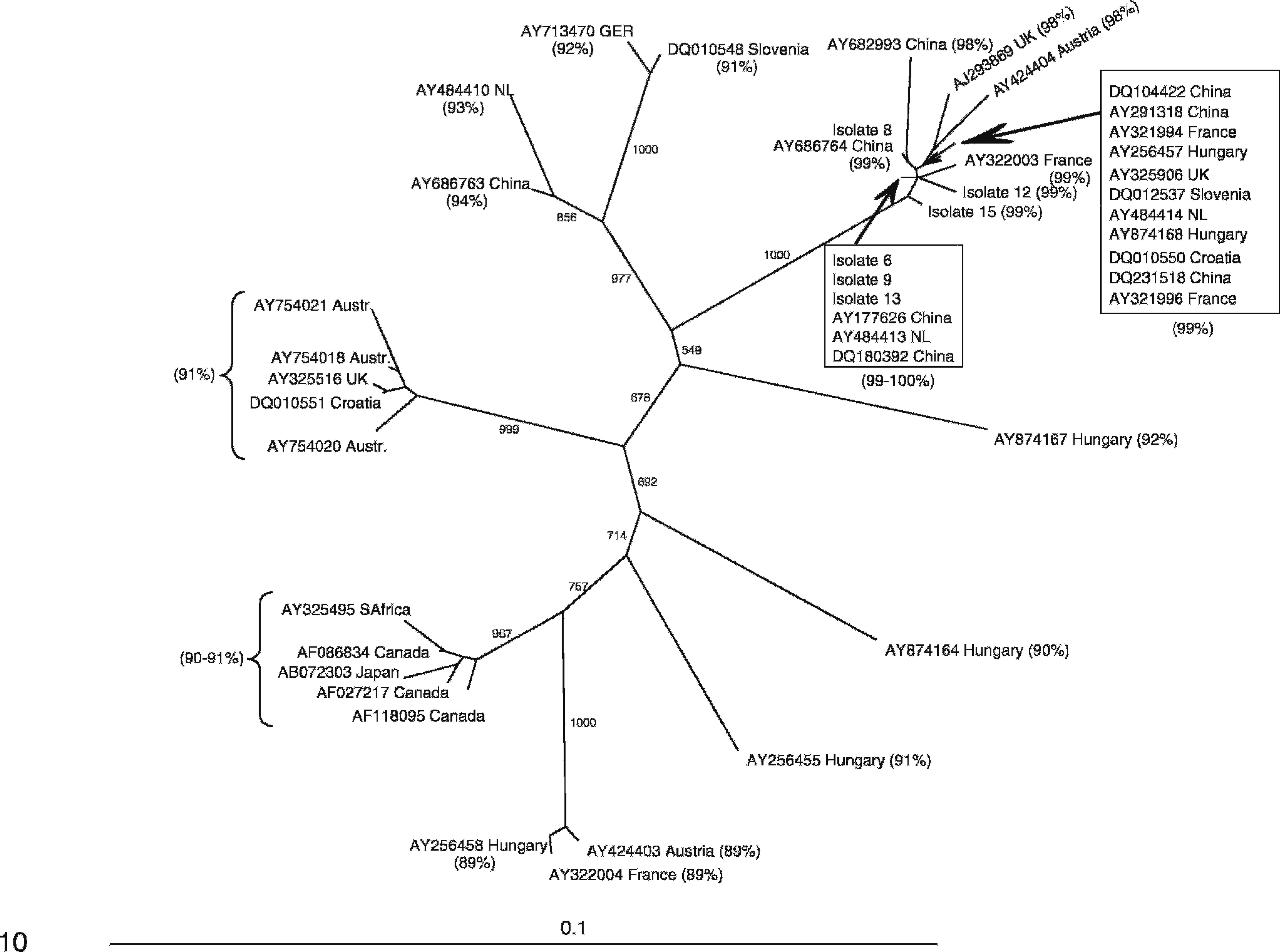
Unrooted neighbor-joining phylogenetic tree constructed from 437 nucleotides of the coding region for the capsid protein from 39 PCV2 strains and the 6 isolates from this study. The sequences obtained from the EMBL/GenBank are identified by their accession number and country of origin. Numbers on the branches: bootstrap values (1,000 repeats). Brackets: percent identity to the sequence of isolate 1. Bar: number of substitutions per site.
Additional etiological findings
The results of additional etiologic screenings are shown inTable 5. In summary, porcine respiratory coronavirus could be demonstrated in 8 of 8 cases, 5 of 12 animals were positive for PRRSV (pig Nos. 7, 10, 11, 12, and 13), and 1 of 15 (pig No. 1) was positive for classical swine fever.
Results of additional etiologic investigations for detection of viral and bacterial pathogens.
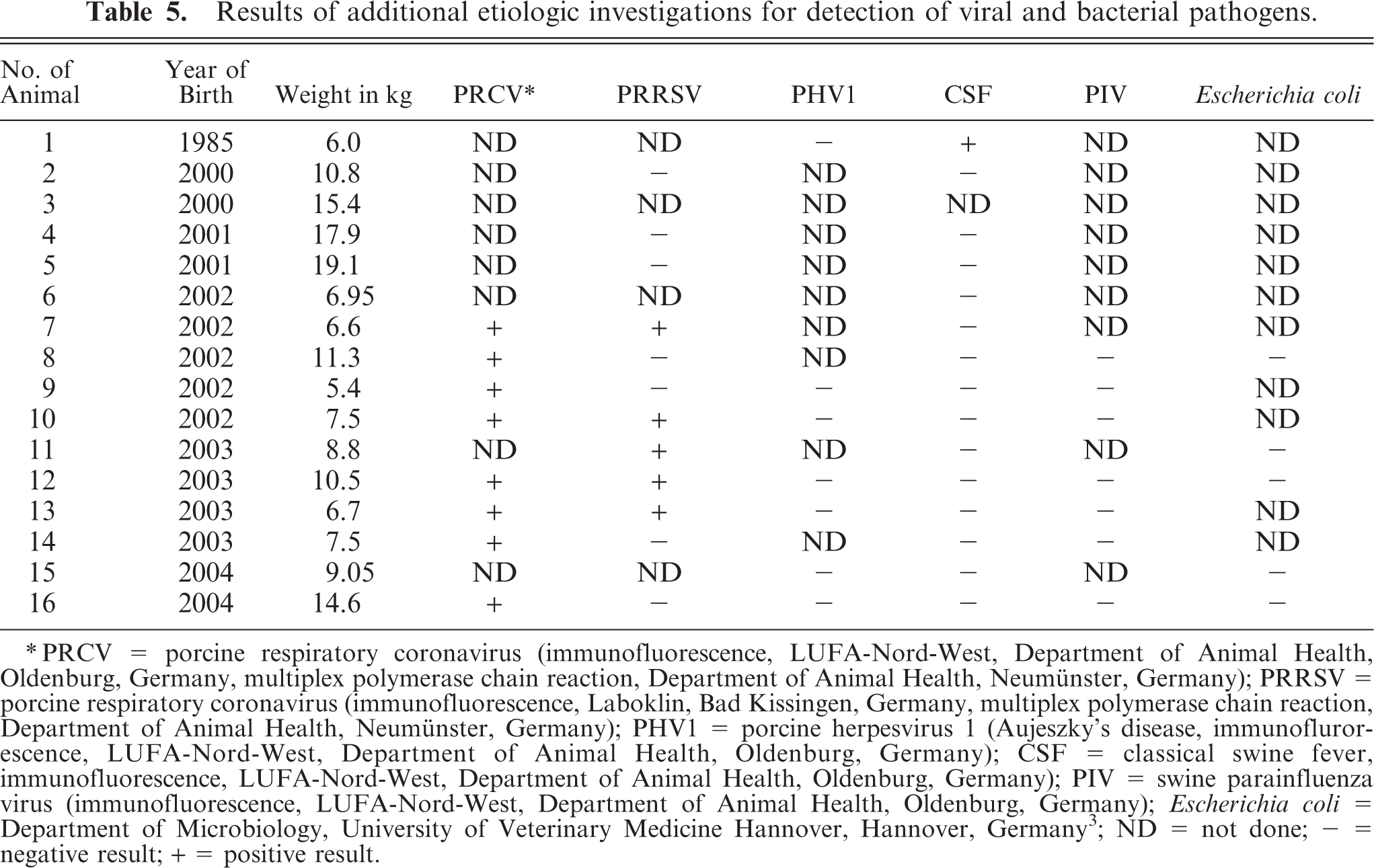
PRCV = porcine respiratory coronavirus (immunofluorescence, LUFA-Nord-West, Department of Animal Health, Oldenburg, Germany, multiplex polymerase chain reaction, Department of Animal Health, Neumünster, Germany); PRRSV = porcine respiratory coronavirus (immunofluorescence, Laboklin, Bad Kissingen, Germany, multiplex polymerase chain reaction, Department of Animal Health, Neumünster, Germany); PHV1 = porcine herpesvirus 1 (Aujeszky's disease, immunoflurorescence, LUFA-Nord-West, Department of Animal Health, Oldenburg, Germany); CSF = classical swine fever, immunofluorescence, LUFA-Nord-West, Department of Animal Health, Oldenburg, Germany); PIV = swine parainfluenza virus (immunofluorescence, LUFA-Nord-West, Department of Animal Health, Oldenburg, Germany); Escherichia coli = Department of Microbiology, University of Veterinary Medicine Hannover, Hannover, Germany3; ND = not done; − = negative result; + = positive result.
Discussion
The present study extends previous knowledge on PCV2-associated lesions and describes a new target tissue and associated pathology in PMWS. These 16 spontaneously occurring cases from different herds displayed a severe, acute, clinical neurologic disorder associated with acute hemorrhage and edema in the cerebellum resulting from necrotizing vasculitis. The presence of PCV2 within endothelial cells in the affected vessels suggests that PCV2 plays a causative role in this disease. Apparent targeting of the cerebellar vasculature by PCV2 in the cases described here and the acute nature of the clinical disease represent newly described and unexpected features of PMWS, which is usually characterized by a chronic clinical course with relatively low morbidity and a moderate though variable mortality. 44
Clinically, the investigated pigs displayed signs reminiscent of several common swine diseases, such as edema disease, streptococcal meningoencephalitis, classical swine fever, Aujeszky's disease, and salt intoxication. In contrast to most other infectious and noninfectious diseases, the occurrence of PCV2-associated cerebellar vasculitis was restricted to single young individuals at the age of 6 weeks to 2 months. Nevertheless, this unique condition has a remarkably low incidence, confirmed by a recent retrospective study, in which 18,550 necropsy results of pigs over 30 years (1962–1998) were screened. No lesions reminiscent of PCV2-associated cerebellar vasculitis as described in the present cases were found (L. Krüger and W. Baumgärtner, personal communication), except for one case in 1985, which has been included in this study.
Necrotizing vasculitis has been identified as the key underlying change resulting in severe leakage of vessels in the present cases. Vasculitis was an unexpected finding and does not represent a common lesion in PMWS. 43 Cardiovascular lesions due to a PCV2-associated vasculitis have been described recently in naturally and experimentally infected piglets and are characterized by myocardial necrosis and systemic vasculitis, but cardiac features could not be detected here. 37 To date, vasculitis and PCV2 infection have been described mainly as characteristic features of PDNS but not PMWS. 40, 43 Porcine dermatitis and nephropathy syndrome and PMWS share certain histologic similarities. In PDNS, as described for PMWS, a severe lymphocytic depletion, an increase of histiocytic infiltrates and multinucleated giant cells in lymph nodes, and an interstitial pneumonia were found in approximately 50% of all cases. 45 In PMWS, the main features found in nonlymphoid tissues included widespread interstitial infiltration of lymphocytes and macrophages in liver, kidney, heart, pancreas, adrenal glands, and rarely meninges. 40, 43 In PDNS, changes in nonlymphoid tissues were mainly found in skin and glomeruli and occasionally in other organs. 45, 50 Death is presumed to result from necrotizing glomerulonephritis and renal failure. Skin lesions consisted of confluent areas of dermal necrosis on hind legs, abdomen, and head due to a necrotizing vasculitis in the subcutis. 45 These characteristic lesions in kidneys and skin are probably caused by an immuncomplex-mediated type-III hypersensitivity reaction in small arterioles but could not be detected in the piglets described here. 46, 54 Only Nos. 2, 6, and 13 had renal necroses and hemorrhages due to a necrotizing vasculitis of arcuate arteries and cortical arterioles, but they lack glomerulonephritis and necrotizing dermatitis typical for PDNS. Furthermore, cases of PDNS without evidence of PCV2 infection but with a high incidence of infection with PRRSV or other infectious agents have also been described. 3, 8, 39 Detection of PCV2 and/or PRRSV in vascular lesions in PDNS has failed in most cases, and the precise role of PCV2 infection in PDNS still remains unclear.
Vasculitis in the brain may be primarily restricted to the CNS or occurs secondarily as part of a systemic infection or disorder. 47 In pigs, vessel-associated lesions in the brain, such as vasculitis and perivascular cuffs, have been described in numerous diseases, including classical and African swine fever, enterovirus and rubula virus infection, rabies, pseudorabies, vitamin E deficiency, and fungal and bacterial infections. The blood-brain barrier is a unique structure, and any disturbance of its integrity results in an uncontrolled fluid movement from the vascular space into the neuropil. 1, 7, 30 Most commonly, increased permeability is triggered by vasoactive mediators during acute inflammation, direct injury of the endothelial cells, or leukocyte-dependent injury. 26, 30 Immunohistochemistry showed a lack of immune cells and immune complex depositions in the vascular wall in early lesions. A weak vascular immunoglobulin reaction was observed in areas of severe vascular leakage most likely due to the transvascular movement of immunglobulin-rich plasma proteins. The endotheliotropism of PCV2, detected by in situ hybridization and immunohistochemistry, and the lack of detectable immune complexes in affected vessels pointed towards a direct PCV2-mediated vascular injury. In cases of PCV2-associated cardiovascular lesions in piglets, a similar pathogenic endotheliotropism of the virus has been described. 37
Additionally, several infected endothelial cells showed increased activity of cleaved caspase-3 and occasionally apoptotic figures. Programmed cell death or apoptosis is morphologically characterized by chromosomal condensation and fragmentation, the formation of apoptotic bodies, and an increase of apoptosis-specific proteins and lipids and plays an important role in physiologic and pathophysiologic processes. 32, 35, 41 Caspase-3 is a major effector caspase that is obligatorily activated by intrinsic, mitochondrial, and extrinsic, death receptor–initiated pathways. 13 The immunohistochemical detection of cleaved caspase-3 allows the specific identification of the activated death pathways, whereas the TUNEL assay only detects the more unspecific DNA double-strand breaks. The morphologic and immunohistochemical data indicated a role of apoptosis during PCV2 infection of cerebellar endothelial cells in the cases presented here. Previously, several studies failed to show a direct involvement of apoptosis in PCV2-associated lesions. 22, 29, 38 However, recently a novel PCV2-associated protein encoded by ORF 3, which is involved in virus-associated apoptosis by activating caspase-8 and caspase-3 pathways in vitro, was described. 28 Nevertheless, endothelial cell apoptosis secondary to local hypoxia resulting from the vascular damage cannot be ruled out in these cases.
Recent studies have shown that additional factors are required to induce classical PMWS lesions in PCV2-infected pigs. Several infectious agents, including PRRSV, PPV, and Mycoplasma hyopneumoniae, as well as noninfectious agents, such as vaccines against Actinobacillus pleuropneumoniae and keyhole limpet hemocyanin incomplete Freund's adjuvant, have been identified as costimulatory factors to induce the disease. 21, 23, 42 A high virus load has been shown to increase the severity of clinical signs in PMWS, whereas PCV2 antibody titers did not appear to influence the outbreak of the disease. 4, 25, 27 In contrast, in PDNS an excessively high PCV2 antibody titer, possibly triggering the formation of immune complexes, was commonly present. 31, 36, 54 Consequently, it has been assumed that the pathogenesis differs markedly between both syndromes. In the present study, a coinfection with porcine respiratory corona virus was identified in 8 animals and with PRRSV in 5 cases. The significance of these findings remains unclear, but a role of these agents in the pathogenesis of the described PCV2-associated vasculitis must be considered.
Edema disease may be associated with clinical signs and pathomorphologic lesions similar to those found here, but microbiologic examination of intestine failed to detect shigatoxigenic E. coli. Furthermore, the brain stem and medulla oblongata, which were commonly histopathologically affected in edema disease, were unaffected in all animals described here. Generally, cerebellar bacterial infection seems unlikely due to the lack of a primarily neutrophilic reaction in the brain or meninges. The causes of the edema in subcutis, gall bladder, and mesentery are also still unclear, but because in some animals PCV2 genome but no vasculitis was detected in the jejunum, a causative role of the virus could not be excluded.
The primer pair used amplified a genome fragment within the PCV2 ORF2. 31, 32 The 6 isolates analyzed here were almost identical and clustered together with isolates from Europe and Asia. A cluster of a specific genotype was not found. Alignments of the nucleotide sequences coding for the capsid protein of PCV2 isolates from different parts of the world and originating from pigs displaying different clinical signs did not reveal any type of correlation. Although distinct clusters can be distinguished, identities between isolates within a cluster ranged between 99 and 100% and within other clusters between 89 and 92%.
In summary, the association between PCV2-specific signals and occurrence of lesions, especially in the cerebellum, suggested a direct role of the virus in the pathogenesis of the observed vasculitis. Although PCV2 was detected in several organs and tissues, it was not possible to demonstrate viral protein or nucleic acids in cells of neural origin. Cerebellar vasculitis represents a new spontaneously occurring type of PCV2-associated disease. Although the principle lesion of this syndrome could be identified, virus-cell interaction and the role of viral coinfections for the pathogenesis of this unique cerebellar lesion need to be investigated in future studies.
Footnotes
Acknowledgements
We thank Danuta Waschke, Kerstin Rohn, and Petra Grüning for excellent technical support.
