Abstract
Porcine circovirus type 2 (PCV2), an economically important pathogen of swine, is the necessary cause of post weaning multisystemic wasting disease (PMWS); PCV2 infection is associated with porcine dermatitis and nephritis syndrome (PDNS). Current immunohistochemical (IHC) methodologies identify PCV2 antigens but are not capable of differentiating replicating virus from nonreplicating virion particles in tissue sections. In this paper, a combination of IHC using commercial monoclonal antibodies specific for single stranded (ss) and double stranded (ds) DNA and PCV2 specific in situ hybridization (ISH) was used to show the specificity of the former for PCV2 DNA in tissue sections from PCV2-infected gnotobiotic pigs. Cold-ethanol-fixed tissue sections were superior to formalin-fixed tissues for detection of PCV2 DNA, presumably due to the lack of protein cross-linking in the latter. These data demonstrate that conventional IHC detects PCV2 DNA forms in experimentally infected PCV2-positive gnotobiotic porcine tissue sections that are minimally compromised by either formalin fixation or the hybridization conditions needed for ISH.
Introduction
Porcine circovirus type 2 (PCV2) is a single stranded circular DNA viral pathogen of swine. 1,7,12,19 It is the essential infectious component of post-weaning multisystemic wasting syndrome (PMWS). 2,11,18 PMWS is an economically important disease of piglets characterized by stunting of growth and multisystemic dysfunction including granulomatous lymphadenitis with lymphoid depletion, hepatitis, pneumonia, and myocarditis. 1,5 The circular single stranded DNA (ssDNA) genome of PCV2 is approximately 1,760 bases. A double-stranded (dsDNA) replicative isoform (RI) is produced during viral replication. 17 The replicase and nucleocapsid proteins are produced from the viral open reading frames (ORF) 1 and ORF2, respectively. 13,14 PCV2 has an affinity for cells of monocytic derivation and is thought to require replication of viral DNA within infected cell nuclei. 11,20 Viral antigens are produced within 18 hours of PCV2 entry into the cell. 11,20 Previous studies have reported both intranuclear and cytoplasmic localization of viral DNA and viral proteins, however it is not clear which stage(s) of the viral replicative cycle corresponds to intranuclear versus intracytoplasmic localization of viral proteins and DNA. The nucleocapsid consists of 60 monomeric subunits each measuring 28 kDa. 6 The assembled PCV2 nucleocapsid is 20.5 nm in diameter. 6 Monoclonal antibodies are capable of detecting viral proteins, 16 however they are not capable of distinguishing between replicating virus and encapsidated nonreplicating virus. In situ hybridization can identify viral DNA with great sensitivity and specificity, 3,15 but does not discriminate between ssDNA of the formed virion from the dsDNA forms of replicating viral DNA. In this study, the ability of commercially produced antibodies against double stranded (ds) and single stranded (ss) DNA to specifically identify PCV2 dsDNA and ssDNA, respectively is demonstrated.
Materials and methods
Tissue samples and handling of specimens. Both formalin- (10% v/v neutral buffered formalin) and ethanol-fixed paraffin-embedded 5-micron tissue section replicates from previously reported experimental disease studies of PCV2 infection in gnotobiotic piglets 11 were available for study. For the latter, fresh tissues from both PCV2-infected and PCV2-free gnotobiotic piglets were trimmed, placed into cassettes and fixed in 100% (v/v) cold (4°C) ethanol on ice for 18–24 hr. Fixed blocks were then immediately processed through an automated tissue processor (Fisher histomatic 266 a ) and embedded into paraffin.
In situ hybridization (ISH) procedure for PCV2 DNA. The PCV2 specific probes (NCBI Database 4 ) were created with a commercially available kit (PCR DIG Probe Synthesis Kit b ) using the following “PCV2-specific” primer set c : Forward: CGGATATTGTAA TCCTGGTCG Reverse: ACTGTCAAG CGTACCACAGTCA. The probes were applied as an in situ hybridization (ISH) mixture that contained 50% formamide, 10% dextran sulphate, 2× SSC buffer, 1× Denhardt's solution, 200 μg/ml herring sperm DNA, 11% autoclaved distilled water, 0.05% PCV probe. Section replicates from tissue blocks were deparaffinized through 3 changes in xylene and 2 changes in absolute ethanol and then rehydrated in graded ethanol (95-70%, v/v) into PBS. Between each subsequent step, the tissues were rinsed twice in 5 mM TBS for 10 min each. Endogenous peroxidase activity was blocked with a 20 min incubation in 0.5% H2O2in methanol. The sections were then incubated in 0.05% protease XIV for 15 min, 37°C, rinsed in distilled water, and washed twice in 5 mM TBS. Fifty microliters of hybridization mixture were added and the sections were coverslipped. The target DNA in the tissue and the PCV2 probe in the ISH mixture were denatured by incubating at 90°C for 10 min followed by immediate cooling on ice packs. Hybridization was performed for 18–22 hr at 37°C in a humid environment. After the hybridization, the slides were washed twice in double strength saline sodium citrate. The slides were washed again in TBS followed by an incubation in a maleic acid buffer (0.1 M maleic acid, 0.15 M NaCl, pH 7.5) for 20 min, 37°C. The sections were then transferred to the maleic acid buffer, which contained 1% blocking reagent (Roche, b 11096176001), followed by an incubation in anti-digoxygenin POD b (horseradish peroxidase -conjugated anti-digoxygenin antibody raised in sheep) diluted 1:200 in the 1% blocking reagent/maleic acid buffer. The sections were washed first in maleic acid buffer, then in 5 mM TBS. Finally a 3,3'diaminobenzidine e (DAB) solution was applied. The slides were lightly counterstained with Harris hematoxylin, dehydrated in graded alcohols to xylene, mounted, and examined by light microscopy.
Immunohistochemistry for viral DNA. Murine monoclonal antibodies to both single stranded (ss) and double stranded (ds) DNA were purchased from a commercial source d . The dsDNA-specific reagent (AE-2MAB1293 d ) is reported to stain nuclear dsDNA in both normal and neoplastic cells by IHC (www.chemicon.com). The ssDNA specific reagent (F7-26 MAB3299 d ) is highly ssDNA-specific but is also reported to weakly stain cell nuclei. 8,9 It is used to detect apoptotic cells (www.chemicon.com).
Section replicates from both formalin- and ethanol-fixed tissue blocks were deparaffinized through 3 changes in xylene and 2 changes in absolute ethanol, and then rehydrated in graded ethanols (95-70%, v/v) into PBS containing 0.1 mg/ml saponin. Antigen retrieval was accomplished through 20 min incubation in 20.0 μg/ml proteinase K in PBS, followed by 20.0 μg/ml pronase E for 20 min, 22°C. The sections were washed 3 times in distilled de-ionized water (ddH2O), incubated for 20 min in 50% v/v formamide f in ddH2O, 56°C, and then quickly transferred into cold (4°C) PBS for 5 min. Endogenous peroxide activity was quenched with 3.0% v/v H2O2 for 5 min followed by a 5 min rinse in ddH2O. Nonfat dry milk reconstituted to 3.0% (w/v) in ddH2O for 15 min was used as a blocking agent and was followed by a 5 min rinse in PBS. The primary antibody was applied and the sections were incubated for 25 min, 22°C. The anti-ssDNA antibody (F7-26) was used at a 1:10 dilution while the anti-dsDNA antibody (AE-2) was used at a 1:250 dilution. The slides were then rinsed 3 times for 5 min each in PBS. The secondary antibodies (biotinylated goat anti-mouse IgM f for F7-26 and biotinylated horse anti-mouse IgG e for AE-2) were then applied at a 1:100 dilution and incubated for 30 min, 22°C. The slides were rinsed twice again in PBS then the ABC reagent e was applied for 30 min. The sections were rinsed again in PBS then developed by incubating with DAB peroxidase substrate e at room temperature for 3–5 min. The slides were rinsed with distilled water, counter-stained with Harris hematoxylin, dehydrated through graded alcohols into xylene, mounted, coverslipped, and examined by conventional light microscopy.
Procedure for combined Fluorescent ISH (FISH) and fluorescent IHC (FIHC). The combined FISH/FIHC procedure used was identical to the IHC until the sections were removed from the ice cold PBS. After immersion in ice cold PBS for 5 min, the sections were incubated in 0.5% v/v H2O2 in methanol for 20 min, 22°C. The blocking reagent was 3% reconstituted milk protein. The dsDNA antibody was used at a dilution of 1:200 in PBS while the ssDNA antibody was used at a 1:10 dilution. After a 25 min, 22°C, incubation with the primary antibodies, the slides were rinsed 4 times in PBS, 5 min each. The secondary antibodies were applied and incubated as described previously. The remainder of the protocol was followed as per the ISH protocol, except all washes were repeated 4 times instead of twice, and probe detection was accomplished with fluorochromes instead of HRP-DAB. The ISH DNA probe was detected with a fluorescein isothiocyanate (FITC) conjugated antidigoxygenin antibody f . Once the fluorochrome was applied, all subsequent reactions/incubations were shielded from light. After a 60 min, 37°C incubation with the FITC conjugated antidigoxygenin (1:100 dilution), the sections were washed twice in maleic acid buffer, then 4 times in PBS. The ABC reagent was used as per the manufacturer's directions, followed by 3 rinses in PBS. The ssDNA and dsDNA antibodies were detected with a 1:100 dilution of avidin-conjugated tetramethylrhodamine c in PBS. The incubation was carried out for 45 min, 37°C, followed by 3 rinses in PBS. The slides were then coverslipped with an aqueous mounting solution and read on a Leica TCS SP2 AOBS confocal laser scanning microscope g .
Results
ISH. Cellular morphology was reduced in both formalin and ethanol-fixed tissues when compared to hematoxylin and eosin stained (HE) sections of the same tissues and fixation methods. The ethanol-fixed tissues had a more pronounced decrease in morphology when ISH was compared to HE or ISH on formalin-fixed tissues. Both ethanol and formalin fixation methods produced the same pattern of staining in PCV2-positive tissues, as described below. Ethanol-fixed, PCV2-negative lymph node and thymic sections did not contain ISH-positive cells. The PCV2-infected sections consisted of PMWS-affected ethanol-preserved and formalin-preserved swine tissues. The ethanol-fixed PMWS-affected thymi exhibited DAB reaction product deposition ranging from distinct inclusion bodies or cytoplasmic accumulations to diffuse cytoplasmic staining indicating the presence of viral DNA in the cytoplasm of histiocytic/macrophage cells. Occasional histiocyte nuclei were positive, while rare cells with pyknotic nuclei exhibited ISH nuclear staining. Ethanol-fixed PMWS-affected lymph nodes exhibited cytoplasmic staining for viral DNA in histiocytes and macrophages as distinct inclusion bodies or cytoplasmic aggregates of viral material. Some positive cells exhibited diffuse dense cytoplasmic staining. The positive cells were randomly distributed throughout the thymic medullary and cortical tissue. The formalin-fixed PMWS-affected lymph nodes exhibited strongly positive cytoplasmic material within the cytoplasm of histiocytes and activated phagocytic cells. There was no difference in the cell types stained or intracellular staining pattern between the two fixation methods tested. In the lymph nodes many of the medullary histiocytic cells were ISH positive, compared to only a few scattered cells in the cortical cell population.
ssDNA immunohistochemistry. Ethanol-fixed and formalin-fixed swine tissue sections were stained with F7-26 MAB3299 monoclonal antibody d . All of the tissues stained with this method retained morphological detail comparable to hematoxylin and eosin stained (HE) sections of the same tissues; the formalin-fixed tissues exhibited better tissue and cellular morphological preservation as compared to the ethanol-fixed tissues. The PCV2-positive sections were PMWS-affected PCV2-positive lymph node and thymic tissues. PCV2-free sections were processed in parallel with the PCV2-infected sections. PCV2-free sections had a few histiocytes and lymphocytes present with small strongly ssDNA-positive nuclei; however the tissues were otherwise negative. The ethanol-fixed PCV2-infected thymic sections had histiocytes containing heavy cytoplasmic DAB reaction product deposits in the form of round aggregates of material. Rare histiocytic cells had large lightly-staining nuclei or darkly staining positive pyknotic nuclei. Positive cells were randomly distributed throughout the thymic tissue. The PCV2-infected ethanol-fixed lymph node sections had similar staining characteristics and histiocytic cells exhibited cytoplasmic DAB reaction product aggregates (Fig. 1). For ssDNA, the only notable difference between the DAB staining pattern in PCV2-infected lymph node and thymus, was that in the thymus the aggregates were distributed randomly across the lobule (cortex and medulla), whereas those in the lymph node were concentrated in follicular dendritic cells.
Slides containing formalin-fixed PCV2-infected, PMWS-affected lymph nodes were also processed as previously described alongside the ethanol-fixed sections. A few cells in both the PCV2-infected formalin-fixed thymic and lymph node sections exhibited strong PCV2-positive nuclear staining and only rare scattered cells had positive cytoplasmic staining. Although ethanol-fixed tissues allowed for the identification of ssDNA and dsDNA using the appropriate antibodies, formalin-fixed tissues yielded little to no positive IHC reactions with these same reagents. For this reason ethanol-fixation was considered to be a better method for tissue preservation for use with these DNA-IHC procedures. Formalin-fixed tissues not evaluated further.
dsDNA immunohistochemistry. These studies used PCV2-infected PMWS-affected and PCV2-free ethanol-preserved control swine tissue sections that were stained with the AE-2 MAB1293 anti-dsDNA antibody d . PCV2-free control swine tissues were processed in parallel with the PCV2-infected tissues. In the PCV2-free control sections, a few cells in the thymus and lymph nodes had small nuclei with strong nuclear staining; none of them exhibited cytoplasmic staining. In contrast, PCV2-infected sections, a small number of cells in the thymi exhibited either light cytoplasmic DAB staining and morphologically normal nuclei, or dark densely-staining pyknotic nuclei. Some histiocytic cells contained aggregates of cytoplasmic material that stained strongly positive (Fig. 1). The cells with cytoplasmic staining appeared randomly distributed throughout the thymic tissue. A small number of the histiocytic cells within the lymph node follicle centers contained aggregates of positive staining material in the cytoplasm. Slides containing PMWS-affected formalin-fixed swine tissue as described above were also included; only rare cells had positive nuclear staining in both the thymic and lymph node sections. All of the tissues stained with this method retained excellent morphological detail compared to hematoxylin and eosin stained sections of the same tissues.
Dual FISH/FIHC. Fluorescent ISH (FISH) combined with the ssDNA fluorescent IHC (FIHC) staining techniques were applied to ethanol-fixed bronchial and peripheral lymph node sections and the tissues were examined by confocal microscopy. The image produced by each of the IHC techniques was merged with the ISH image to confirm colocalization and specificity of each DNA monoclonal with respect to PCV2 DNA as determined by ISH.
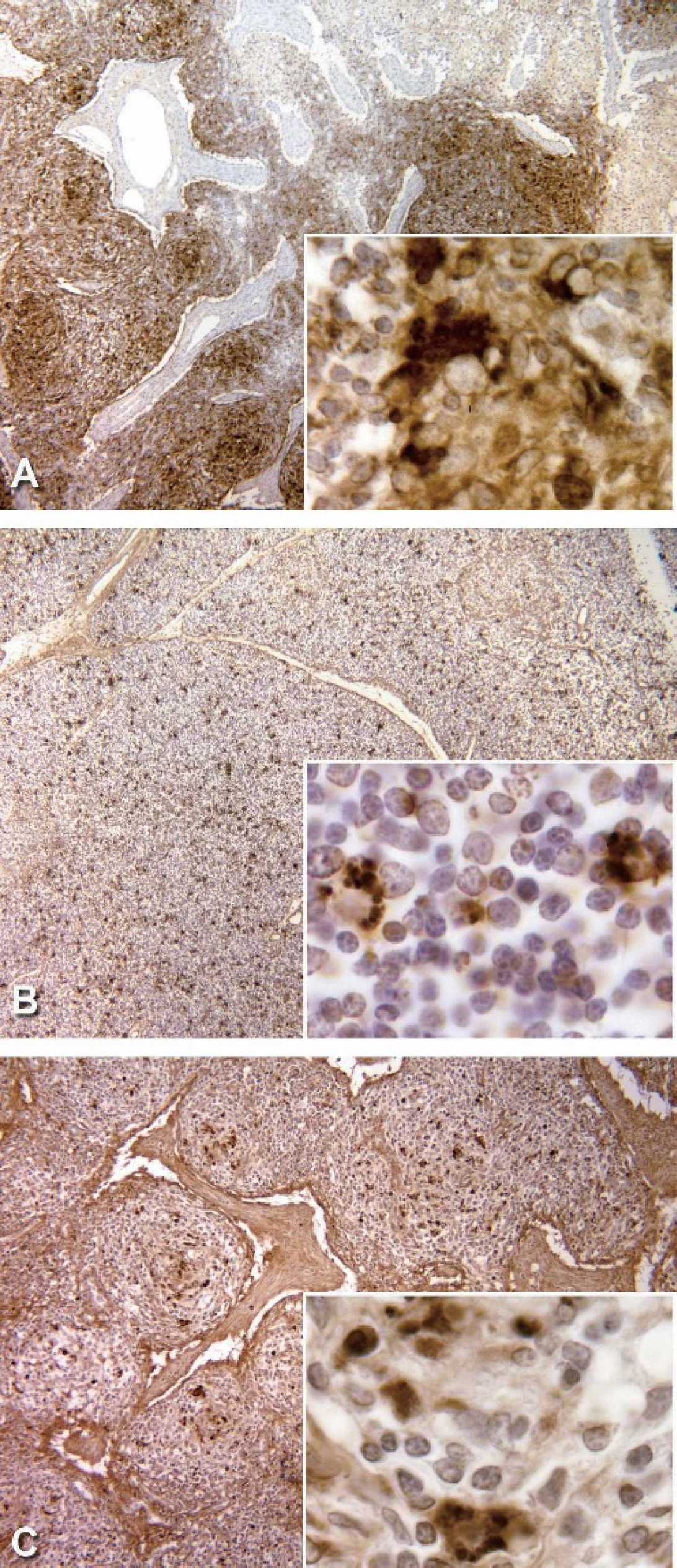
Lymph node: PCV2 specific ISH positive control slide, 40X. This photomicrograph illustrates the large portion of cells were positive for PCV2. The inset (400X) demonstrates the cytoplasmic accumulations of virus.
The PCV2-free tissues exhibited no significant staining with this method. In contrast, PCV2-infected sections exhibited a high degree of overlap (yellow) between the ssDNA IHC stained areas (red) and the ISH stained areas (green). A few scattered points throughout the combined photomicrograph were stained either with red or green but not both (Fig. 2). The dsDNA fluorescent IHC was also combined with FISH. The merged pictures were similar to those of the ssDNA FISH/FIHC sections. Tissues subjected to the dual FISH/IHC staining process exhibited a loss of tissue and cellular morphology, which prevented definitive identification of the infected cell types and cellular compartments.
The dual IHC/ISH staining protocol failed when it was attempted with the Roche blocking reagent. The milk protein blocking reagent was substituted for the Roche blocking reagent in later attempts and found to produce minimal background staining without inhibiting either the ISH or IHC procedures.
Discussion
Detection of viral DNA for PCV2 and other DNA viruses in histologic sections are routinely accomplished by ISH. This technique relies upon the specificity of the DNA probes used and the conditions for staining are adjusted to maximize the DNA-DNA hybridization opportunities. These methods have the strong advantage of high specificity (predicted by the probes), ease of use and adaptation to automated staining equipment. Off-setting these advantages are the harsh conditions necessary to denature the DNA for hybridization. This affects cellular morphology. In this study, ISH worked well on both formalin- and ethanol-fixed tissues. In order to detect viral PCV2 DNA in tissue sections without compromising the cellular morphology, we explored the feasibility of using monoclonals specific for ss and dsDNA as a substitute for ISH. Specificity of each monoclonal for PCV2 DNA was shown with the dual FISH/IHC technique. This demonstrated the specificity of the ss and dsDNA IHC techniques and hence identified putative nonreplicating and replicating PCV2 DNA, respectively, in PCV2-infected gnotobiotic swine tissues. Specificity of these monoclonals for PCV2 DNA isoforms was confirmed with confocal microscopy using concurrent FISH. Although monoclonal and polyclonal antibodies specific for PCV2 viral proteins are available, none have been shown to differentiate replicating PCV2 from nonreplicating PCV2 or possess specificity for PCV2 DNA forms.
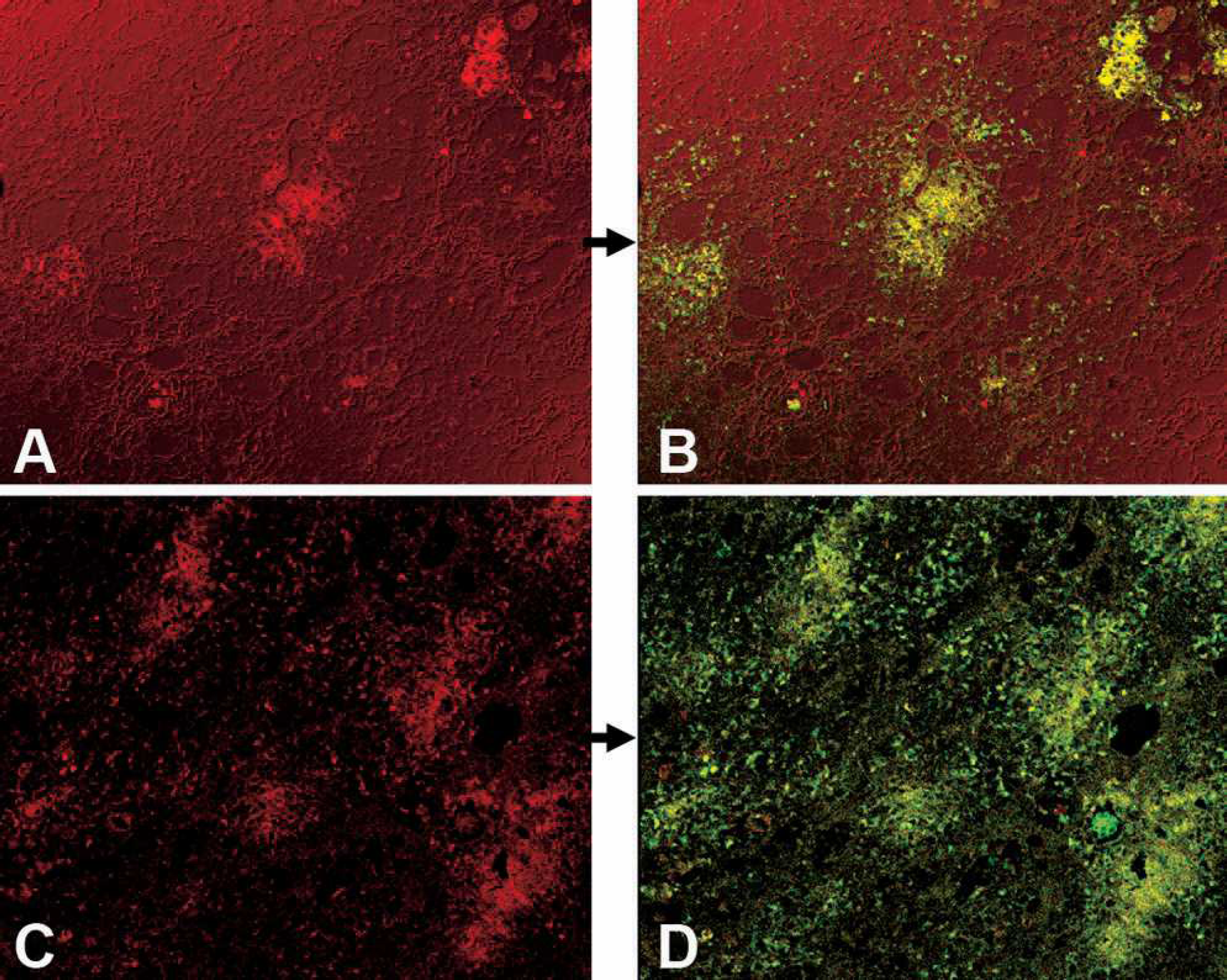
ssDNA FIHC (red), 20X
The distribution of PCV2 ssDNA identified by IHC demonstrated the reaction product accumulation in inclusion bodies in the cytoplasm of thymic histiocytic cells and/or macrophages and in the lymph node dendritic cells. This is likely viral ssDNA in the cytoplasm and suggests that the encapsidated ssDNA virions are both focally and diffusely distributed within the cytoplasm of these cells. This stain is specific for PCV2 DNA in gnotobiotic pigs infected with PCV2. The small amount of scattered anti-ssDNA staining, which did not co-localize with ISH, likely identified occasional apoptotic cells that were unrelated to PCV2 interactions, as these cells were ISH-negative. In support of this interpretation, pyknotic nuclei were strongly positive for ssDNA. One of the documented uses of the ssDNA monoclonal is as a cytomarker for apoptosis. 8 Importantly, these positive cells were detected in both the positive and negative control slides in similar numbers. Deeply basophilic nuclei were also present in the ISH negative controls but did not hybridize either. Thus, the apoptotic/necrotic cells are not a direct result of PCV2 infection. This is consistent with a previous study that demonstrated that PCV2 infection does not cause lymphoid depletion via apoptosis. 10 Apoptotic cells are expected because the largest concentration of PCV2 is in the germinal centers of follicles, which also are active in cell production and destruction and subsequently contain a large number of apoptotic cells.
The manufacturer's literature on the F7-26 “ssDNA-specific” monoclonal indicates that the antibody recognizes DNA that has been denatured at reduced temperatures due to enzymatic hydrolysis of DNA bound proteins (e.g. histones). Proteolysis by pronase E in addition to proteinase K was found to be necessary for the F7-26 to stain DNA. The areas marked by ISH but not the ssDNA IHC likely represent either viral DNA in a ds state, or that which was not bound by the anti-ssDNA antibodies due to stoichiometric constraints. Replicative forms of PCV2 DNA were represented by dsDNA-positive foci that were also positive for viral DNA-ISH. 17
The loss of tissue and cellular morphology due to the combined processes of IHC and ISH precluded definitive identification of the cell type(s) that contained dsDNA. This problem was avoided when IHC techniques were performed individually. The ssDNA and dsDNA IHC techniques, when used individually, indicated that the majority of cytoplasmic staining occurred in histiocytic and dendritic cells. Therefore, the material that was positive for viral DNA as per ISH, and either ssDNA or dsDNA as per IHC lies in the cytoplasm of mononuclear cells.
The decreased amount of positive staining material observed in IHC stained formalin-fixed tissues when compared to the ethanol-fixed tissues indicates a decrease in IHC sensitivity due to fixation of the former. This is supported by the similar amount and distribution of positive material observed in the formalin- and ethanol-fixed PCV2- positive ISH tissues. The decreased sensitivity of the stains in formalin-fixed sections with the IHC is likely associated with the cross-linking of proteins induced by formalin fixation. Since ethanol is a noncross-linking fixative, it is less likely to destroy or hide immunoreactive epitopes. Because ISH does not rely on protein recognition, protein-protein cross linking is not a consideration, and for this reason ISH performed well on both formalin and ethanol-fixed tissues.
In comparison to ISH, the IHC methods caused little damage to tissue and cellular architecture. This allowed for easy identification of cells and lesions by morphology. While the ISH is harsher on the tissue and more disruptive of morphological characteristics, it is successful when used in formalin-fixed tissues.
Since PCV2 DNA is in a single stranded form when the virus is not in the process of replication, the ssDNA IHC modality is appropriate for identifying the majority of virus within a tissue. It is also possible that these IHC methods may pick up other DNA viruses that may be present in porcine tissues, making these techniques most useful when staining tissues from gnotobiotic or other “clean pigs” (e.g., snatch farrow) in which it is known that the tissues are infected with PCV2 and the morphology must be preserved. Thus, the ds and ssDNA IHC can be used as another method for identifying PCV2 nucleic acid forms in experimentally-infected gnotobiotic porcine tissues. Additionally, the IHC techniques can be accomplished in a few hours without the need for 18-hour incubation, as is required by ISH.
Acknowledgements
The authors wish to acknowledge the excellent technical assistance of Ms. Amy Davis and Ms. Anne Saulsbery.
Footnotes
a.
Fisher Scientific, Hampton, NH.
b.
Roche Applied Science, Indianapolis, IN.
c.
Invitrogen Inc., Carlsbad, CA.
d.
Chemicon Inc., Thousand Oaks, CA.
e.
Vector labs, Burlingame, CA.
f.
Sigma-Aldrich, St. Louis, MO.
g.
Leica Microsystems GmbH, Wetzlar, Germany.
