Abstract
Porcine circovirus type 2 (PCV2)–associated disease (PCVAD) continues to be an important differential diagnosis on pig farms in the United States and worldwide. Case trend analyses indicate that the incidence of PCVAD is on the rise in the United States. Accurate diagnosis is important in order to implement appropriate intervention strategies. PCVAD can manifest as a systemic disease, as part of the respiratory disease complex, as an enteric disease, as porcine dermatitis and nephropathy syndrome, or as reproductive problems. PCVAD may be only a sporadic individual animal diagnosis; however, PCVAD may also manifest as a severe herd problem accelerated and enhanced by concurrent virus or bacterial infections. This article is intended to discuss the most common disease manifestations, pathogenesis, diagnostic approaches, and intervention strategies associated with PCVAD in North America.
Introduction
Porcine circovirus type 2 (PCV2) is now considered one of the most important viral pathogens in the U.S. pig population. Case trend analysis based on submissions to the Iowa State University Veterinary Diagnostic Laboratory indicates a marked increase in PCV2-associated disease (PCVAD) cases in 2006 (Fig. 1). This may be the combined result of increased awareness of the disease, increased case submissions, and true increased incidence of PCVAD in the Midwest.
Historical background
Porcine circovirus (PCV), a small, nonenveloped, single-stranded DNA virus with a circular genome, 133 was first recognized as a contaminant of the continuous porcine kidney cell line PK-15 (ATCCCCL31) in 1974. 136 Under experimental conditions, the PK-15-derived PCV isolate did not produce disease in pigs. 5,134 In the late 1990s, a variant strain of PCV was associated with a newly emerged disease syndrome in pigs, which became known as post-weaning multisystemic wasting syndrome (PMWS). 7 Sequence analyses of the PMWS-associated PCV revealed significant genetic differences compared with the PK-15 cell-derived PCV. 10,29,42,88,92 To distinguish the two, the pathogenic PMWS-associated PCV was designated as porcine circovirus type 2 (PCV2) and the nonpathogenic PCV as porcine circovirus type 1 (PCV1). 88
The presence of PCV2 can be traced back to 1969 in Belgium (Sanchez R, Nauwynck H, Pensaert M: 2001, Proc Conference of ssDNA Viruses, Plants, Birds, Pigs, and Primates, p. 122), 1970 in the United Kingdom, 40 1973 in Ireland, 139 1985 in Canada, 83 and 1985 in Spain. 115 Archived serum samples obtained from slaughterhouses in Belgium were tested for PCV2-specific antibodies by indirect immunoperoxidase monolayer assay (IPMA), and it was found that all samples (50 from 1969, 50 from 1975, and 50 from 2000) were positive for PCV2-specific antibodies (Sanchez R, Nauwynck H, Pensaert M: 2001, Proc Conference of ssDNA Viruses, Plants, Birds, Pigs, and Primates, p. 122).
Sporadic cases of PMWS were retrospectively identified in archived tissues, before the emergence of PMWS in the 1990s, in formalin-fixed tissues from 68 porcine cases that had been submitted to a laboratory in England between 1970 and 1997. 40 PCV2-specific nucleic acids were found in 41% (9/22) of the submissions from the 1990s, in 31% (4/13) of the submissions from the 1980s, and in 32% (8/25) of the submissions from the 1970s. Sequence analyses of PCV2 sequences from 5 archived tissues revealed a high sequence identity to PCV2 isolates obtained from a 2000 porcine dermatitis and nephropathy syndrome (PDNS) case, implying that a similar PCV2 isolate has been present in the UK pig population for more than 30 years. 40
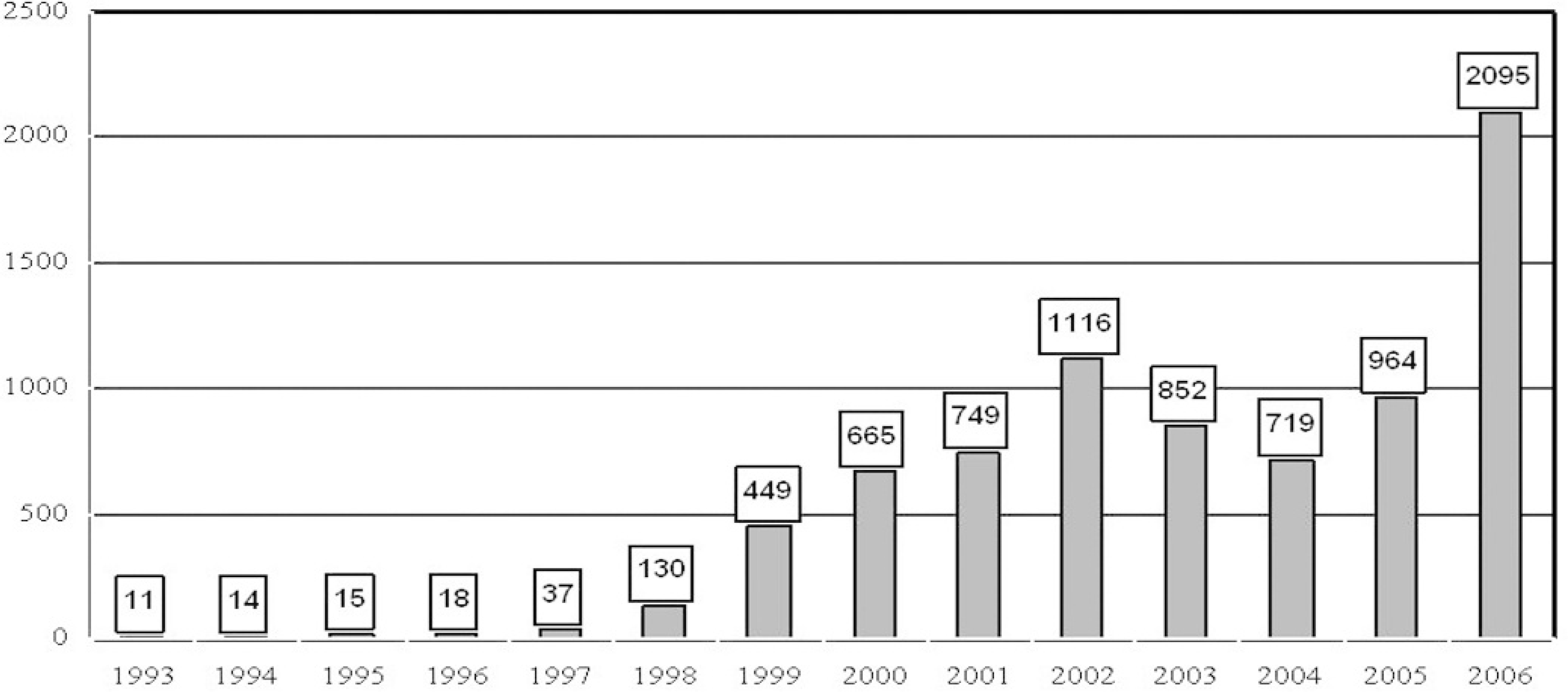
Overall trend of porcine circovirus type 2-associated disease (PCVAD) cases submitted to the Veterinary Diagnostic Laboratory at Iowa State University.
Archived tissues from 189 pigs and archived sera from 388 pigs collected from 1985 to 1997 in Spain were tested for the presence of PCV2 DNA by in situ hybridization (ISH) and for the presence of PCV2-specific antibodies by IPMA. 115 Approximately 41.3% (78/189) of the tissues were found to be ISH positive for PCV2, and 72.7% (282/388) of the sera were found to be IPMA positive, which is indicative of enzootic infection in Spain since 1985. 115
PCV2 antibodies were detected in most pig serum samples collected in Northern Ireland from 1973 to 1999. 139 The percentage of PCV2-seropositive sera showed an increased incidence in the samples collected in 1988 (100%; 80/80) and 1999 (92.1%; 129/140) when compared with 1973 (69.1%; 56/80), 1981 (61.3%; 49/80), and 1984 (55%; 44/80). 139
Archived serum samples obtained from Canadian slaughterhouses were tested by indirect fluorescent assay (IFA) for antibodies to PCV1 and PCV2. 83 In 1985, 8% (14/177) of the sera were positive for PCV1 and 13.6% (24/177) were positive for PCV2. In 1989, 41.4% (60/145) of the sera were positive for PCV1 and 72.4% (105/145) were positive for PCV2. In 1997, 38.1% (56/147) of the sera were positive for PCV1 and 66.7% (98/147) were positive for PCV2. 83
Taxonomy
Both PCV1 and PCV2 are members of the Circoviridae family. 137 The Circoviridae family is divided into the genera Circovirus (Circo indicates that the viral genome has a circular conformation) and Gyrovirus (Gyro is a derivation from the Greek work gyrus, meaning “ring” or “circuit”). The genus Circovirus contains the following species: beak and feather disease virus (BFDV), canary circovirus, goose circovirus, pigeon circovirus, PCV1, and PCV2, and tentatively duck circovirus, finch circovirus, and gull circovirus. The genus Gyrovirus contains only chicken anemia virus (CAV). 137
Viruses that belong to the Circoviridae family have characteristic virions that exhibit icosahedral symmetry and lack an envelope. The genomes are covalently closed, circular, single-strand DNA molecules, which range in size from 1.8 to 2.3 kb. The genome polarity of CAV is negative sense, whereas those of the other circoviruses are ambisense. 137 CAV, PCV2, and BFDV were found to have an icosahedral structure containing 60 capsid protein molecules arranged in 12 pentamer-clustered units. 25 Circoviruses are host specific or exhibit a narrow host range, and most of the known circoviruses infect avian species. 137 Subclinical infections are common; however, singular circovirus infections are associated with clinical disease in some cases, such as with infectious chicken anemia, psittacine beak and feather disease, and circovirus disease of pigeons. Circovirus infections in several species cause varying degrees of lymphoid depletion and are thought to be immunosuppressive. 137
Phylogenetic analyses of PCV1, avian circovirus, plant geminiviruses, and nanoviruses classified PCV1 as most closely related to BFDV and were intermediate between the 2 plant viral groups. 95 Furthermore, it has been proposed that a predecessor to PCV1 and BFDV may have originated from a plant nanovirus that infected a vertebrate host and recombined with a vertebrate-infecting RNA virus, most likely a calicivirus. 37
Recent research has shown that PCV2 isolates can be grouped into 2 major groups, PCV2 group 1 and PCV2 group 2, which can be further divided into clusters: PCV2 group 1 viruses can be divided into 3 clusters (1A–1C), and the PCV2 group 2 can be divided into 5 clusters (2A–2E). 97 Simultaneously with the introduction of the terms PCV2 group 1 and PCV2 group 2, North American laboratories started to group PCV2 field isolates into European-like isolates or PCV2b (falls into PCV2 group 1) and into North American-like isolates or PCV2a (falls into PCV2 group 2; Gagnon CA et al.: 2007, Proc Am Assoc Swine Practitioners 38:535–540). In addition, some North American laboratories report the predicted restriction fragment length polymorphism (RFLP) patterns rather than sequence information, and most isolates fall into 1 of 2 RFLP patterns designated 422 and 321. Isolates with the RFLP pattern 422 typically cluster into PCV2 group 2 (or PCV2a or North American-like isolates), whereas isolates with a 321 RFLP pattern can be either PCV2 group 2 (PCV2a or North American-like isolates) or PCV2 group 1 (PCV2b or European-like isolates). This has led to the use of the term old 321, which is associated with PCV2 group 2, and new 321, which is associated with PCV group 1 that can be verified only by sequencing. Proposed nomenclatures of PCV2 subgroupings are summarized in table 1.
Proposed nomenclature for subclassification of porcine circovirus type 2 (PCV2).

Based on restriction fragment length polymorphism; old versus new can be distinguished only by sequencing.
Biological and physical properties
The buoyant density of PCV1 in CsCl has been reported to be 1.37 g/cm 3 by Tischer et al. 136 and 1.36 to 1.37 g/ml by Allan et al. 12 The sedimentation coefficient (S) was determined to be 57S when compared with the sedimentation coefficient of a bovine enterovirus. 12 PCV1 was found to be stable at pH 3, at 56°C and at 70°C for 15 minutes, and was resistant to inactivation after exposure to chloroform. 12 The infectivity of PCV2 was reduced by 1.6 log by pasteurization for 10 hours at 60°C, by 0.75 log by dry heat treatment for 72 hours at 80°C, and by 1.25 log by extreme dry heat treatment for 30 minutes at 120°C. 140 PCV2 is readily isolated from tissues that have been stored at −70°C. 29
Transmission
Transmission of PCV2 is thought to occur through direct contact via oronasal, fecal, and urinary routes. 16,82 Direct contact with pigs inoculated with PCV2 42 days previously resulted in the transmission of virus to 3 of 3 control cesarean-derived, colostrum-deprived pigs. 16 PCV2 shedding in experimentally infected cesarean-derived, colostrum-deprived pigs was determined by polymerase chain reaction (PCR) on oropharyngeal swabs, nasal swabs, feces, whole blood, and serum. 125 All pigs tested were PCV2 DNA positive on nasal swabs, feces, and oropharyngeal swabs 1 day after inoculation, and PCV2 DNA was detected in all samples, with the exception of oropharyngeal swabs (1 of 2 pigs positive) up to 70 days post-PCV2 inoculation. Serum and whole-blood samples were first tested at 7 days postinoculation, at which time they were positive by PCR. 125 Tonsillar, nasal, tracheobronchial, urinary, and fecal swabs of pigs with and without severe systemic PCVAD were tested by quantitative real-time PCR, and PCV2 DNA was detected in a high percentage of the samples, leading to the conclusion that PCV2 is most likely excreted through respiratory secretions, oral secretions, urine, and feces of both PCVAD-affected and clinically healthy pigs, with higher viral loads in the PCVAD-affected pigs. 124 PCV2 nucleic acids were detected in the digestive tract of pigs with (14/54 intestines and 4/9 feces) and without (3/14 intestines and 16/20 feces) enteric disease by PCR, suggesting fecal-oral transmission of PCV2 via feces. 145
Vertical transmission has been demonstrated to occur in individual sows in the field 73,96 and experimentally. 52 There are reports of vertical intrauterine transfer of PCV2 resulting in viremic or persistently infected piglets at birth. 144 PCV2 can also be demonstrated in semen samples. Seven-month-old boars were inoculated intranasally with PCV2. 75 PCV2 DNA was detected at the first serum collection day at 4 days postinoculation in serum samples of 3 of the 4 boars, and serum samples were positive up to 35 days postinoculation but negative by 90 days postinoculation. PCV2 DNA was detected as soon as 5 days postinoculation in semen of 2 of the boars and intermittently through 47 days postinoculation in all 4 boars. 75 Semen from 98 one-year-old boars from 49 herds in Korea were tested by PCR, and 13 of 98 semen samples were found to be positive for PCV2 by conventional PCR, 26 of 98 semen samples were positive by seminested PCR, and 11 of 98 semen samples were positive by virus isolation. 66 The same study also investigated the prevalence of PCV2 in seminal fluid, nonsperm cells, and sperm heads, and it detected the greatest amount of PCV2 DNA in the seminal fluid and nonsperm fraction. 66 The frequency of PCV2 DNA in semen from naturally infected boars was found to be low and sporadic, and boars seropositive for PCV2 may have persistent shedding of the virus in semen. 84 PCV2 DNA in semen did not appear to affect the percentage of morphologically normal or live sperm cells in PCV2-shedding boars, and boars older than 17.5 months of age did not appear to shed PCV2 DNA in semen. 84 Although PCV2 DNA has been demonstrated in semen, experimental confirmation that PCV2 can be transmitted via artificial insemination is lacking to date.
Case trends in porcine circovirus type 2–associated disease based on submissions to the Veterinary Diagnostic Laboratory at Iowa State University.
Disease terminology
The first report of disease associated with PCV2 was described as postweaning multisystemic wasting syndrome, or PMWS. 44 Since PCV2 is ubiquitous in the pig population and infection does not necessarily equate to disease, a case definition for PMWS was proposed by Sorden. 128 Based on this definition, a diagnosis of PMWS required 1) the presence of clinical signs such as wasting, weight loss, and respiratory disease; 2) the presence of the hallmark PCV2-associated microscopic lesions (lymphoid depletion and/or histiocytic replacement of follicles in lymphoid tissues), and 3) PCV2 antigen or nucleic acids associated with the microscopic lesions as determined by immunohistochemistry or ISH. 128
Soon it became evident that PMWS described only a portion of the PCV2-associated diseases. For example, large amounts of PCV2 antigen can be found in older pigs or even fetuses and neonates that do not manifest wasting, or PCV2 antigen may be found solely in an organ system other than lymphoid tissues such as lungs or intestines. In 2002, it was proposed that existing abbreviations, including PMWS, should be replaced with porcine circovirus disease, or PCVD. l PCVD is now used in Europe to summarize diseases associated with PCV2. 123
In North America, it was felt that any new term used in connection with PCV2 should include the word associated, which led to the creation and introduction of the term porcine circovirus–associated disease (PCVAD) in March 2006 by the American Association of Swine Veterinarians (AASV). There are several reasons why the term wasting has been eliminated from the case definition and categorizations: 1) wasting is a subjective clinical term typically used in reference to weight loss, 2) weight loss or failure to gain weight is common in animals with a wide variety of different infectious and noninfectious diseases, and 3) the term wasting may incorrectly associate PCVAD with transmissible spongiform encephalopathies observed in cervids. The AASV PCVAD case definition was posted in October 2006 at http://www.aasp.org/aasv/position-PCVAD.htm (accessed February 4, 2007). Based on this case definition, PCVAD can be subclinical or include 1 or more clinical manifestations such as multisystemic disease with weight loss, high mortality (doubling of the mortality rate without introduction of a new known pathogen), respiratory signs, PDNS, enteric signs including diarrhea, and reproductive disorders individually or in combination in a herd or group of pigs.
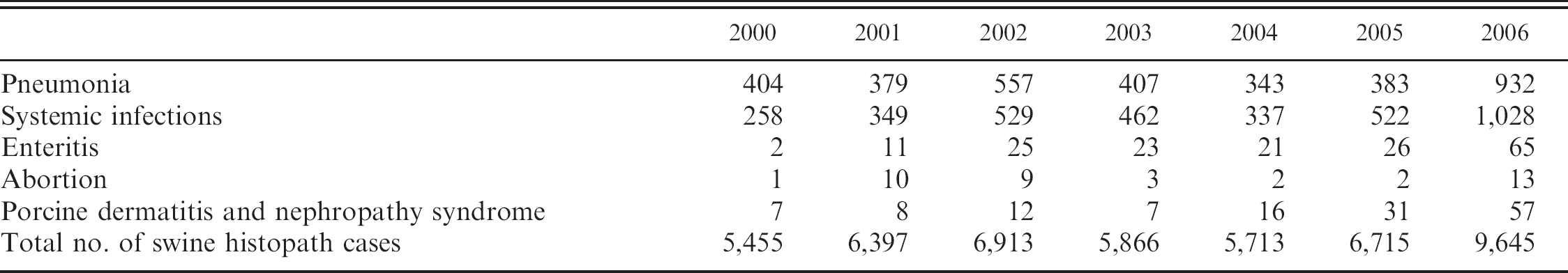
In the diagnostic database of the Veterinary Diagnostic Laboratory at Iowa State University (ISU VDL) in Ames, Iowa, PCVAD now includes systemic infection (the category in which PMWS now fits), PCV2-associated pneumonia, PCV2-associated enteritis, PCV2-associated reproductive failure, and PCV2-associated PDNS (Table 2). Cases that are categorized in this way may have PCV2-associated lesions that vary in severity from mild to severe, and such descriptions are added to the case reports. Diagnosticians at the ISU VDL generally feel that this classification scheme allows for better and more uniform classification of the different manifestations of PCVAD.
Pathogenesis of PCVAD
The pathogenesis of PCV2 infection and the major cell types that support PCV2 replication are still not fully understood. Lymphoid depletion and lymphopenia in peripheral blood is a consistent feature in pigs that develop clinical PCVAD. Immunohistochemistry (IHC) or ISH techniques demonstrate large amounts of PCV2 antigen or nucleic acids in the cytoplasm of macrophages and dendritic cells replacing lymphocytes in the depleted follicles in lymphoid tissues. 3,20,128 However, PCV2 antigen in lymphocytes was only sporadically detected, 20 and it is still unknown whether the reduction of lymphocytes in PCVAD-affected pigs is due to reduced production in the bone marrow, reduced proliferation in secondary lymphoid tissues, or increased loss of lymphocytes in the bone marrow, peripheral blood, or secondary lymphoid tissues via virus-induced necrosis or apoptosis.
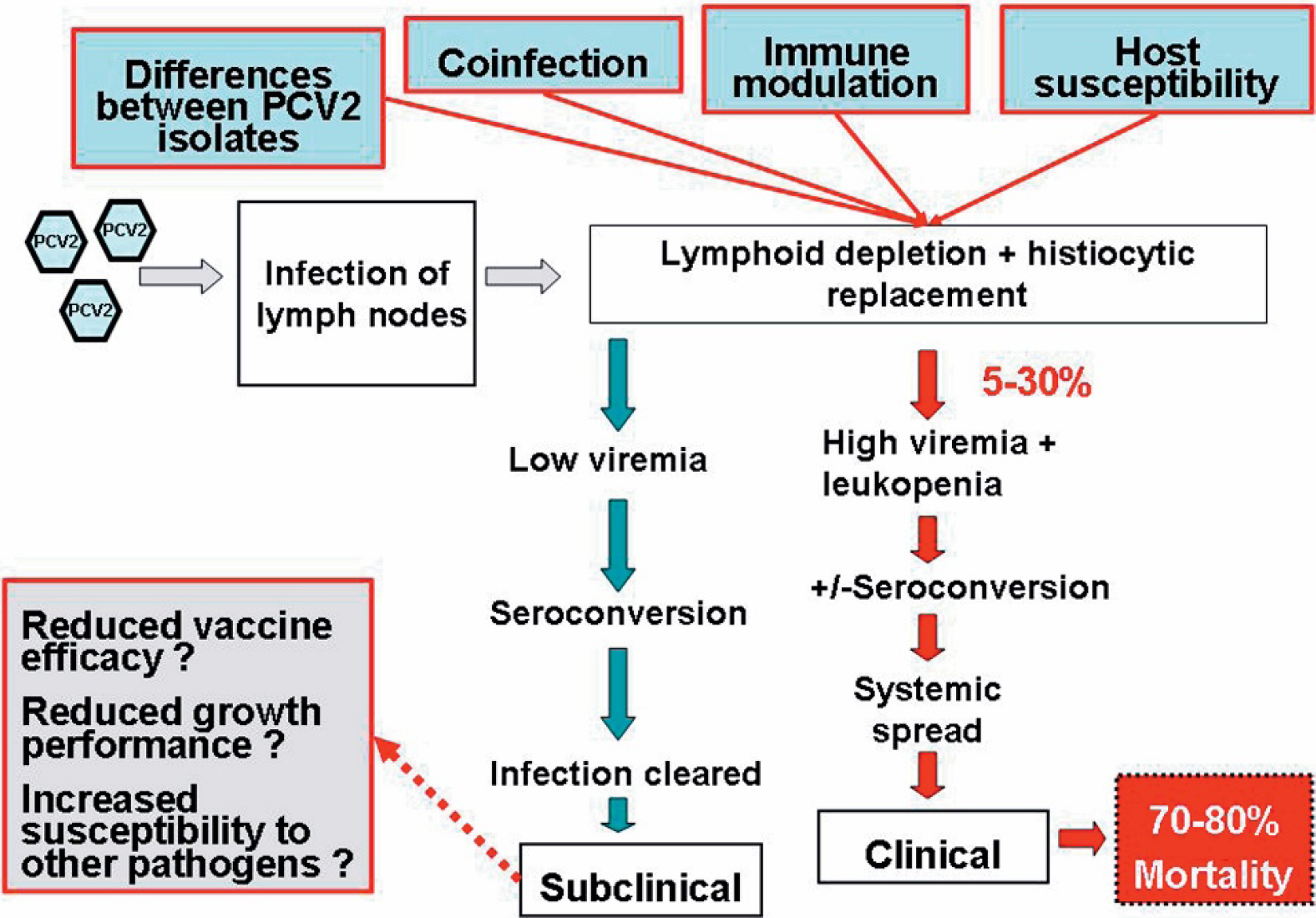
A current model of the understanding of the progression of porcine circovirus type 2 (PCV2) infection toward porcine circovirus-associated disease (PCVAD). Adapted from Dr M. Fenaux, personal communication, 2004.
Despite the presence of PCV2 in macrophages and dendritic cells, recent in vitro studies suggest that monocytic cells may not represent the primary target for PCV2 replication. 38 Monocytes and macrophages were tested for the ability to support PCV2 replication in vitro. PCV2 replication in these cell types was not observed; however, PCV2 was not degraded in the cytoplasm of the cells. 38 Similarly, no evidence of in vitro virus replication in dendritic cells was found by Vincent et al. 138 ; however, PCV2 did persist in dendritic cells without loss of infectivity or the induction of cell death. It has been speculated that because of their migratory capacity, dendritic cells can provide a vehicle for transport of the virus throughout the host. 138
To clarify the role of PCV2 in systemic PCVAD and other clinical manifestations solely attributed to PCV2 infection, an infectious DNA clone of PCV2 was developed. 32 Using an infectious DNA clone has ensured the purity and homogenicity of the inoculum in in vivo animal studies and has allowed for genetic manipulation of the virus at the molecular level to evaluate the biological effects of the genetic changes. 32,35,36,105 Specific pathogen-free (SPF) pigs infected with the cloned genomic PCV2 DNA developed PCV2-associated lymphoid lesions. 32 Evidence of wasting was not observed in the 35-day duration of the study; however, the PCV2 infectious DNA clone research clearly established PCV2 as the cause of the hallmark lymphoid lesions of PCVAD. 32 In 2005, a French group similarly demonstrated that SPF pigs inoculated with a cloned PCV2 DNA developed lesions consistent with systemic PCVAD. 39 Taken together, the data from the infectious DNA clone work support the hypothesis that PCV2 is essential for the development of PCVAD; however, most strains of PCV2 likely require other cofactors or building blocks to induce the full spectrum of clinical signs and lesions associated with advanced cases of PCVAD.
Evidence from the field supports the thought that PCVAD is multifactorial in causality and that not all pigs that are infected with PCV2 will develop clinical PCVAD (Fig. 2). Factors that are currently thought to influence the outcome of PCV2 infection can be broken down into 4 main areas of focus: virus, host, coinfections, and immune modulation (Fig. 2). Experimental work has further confirmed that at least these 4 components are the key building blocks, identified to date, in the PCVAD models.
Virus-dependent factors
A high percentage of clinically healthy pigs are known to be infected with PCV2, whereas others develop severe disease. Genetic analyses and sequence comparison of PCV2 isolates to date have failed to fully explain differences in clinical manifestations. The complete genomes of 10 Dutch PCV2 isolates from PCVAD-affected and -unaffected farms were sequenced and when compared were found to have 95.6% to 100% sequence identity. 41 No consistent pattern was evident between PCV2 isolates from affected and unaffected pigs, leading the authors to conclude that the differences in clinical manifestations were likely due to something other than the virus. 41 Thirty-four PCV2 isolates from eastern Canadian herds with varying clinical manifestations of disease were sequenced and found to be closely related to each other and to Canadian, U.S., European, and Asian strains. 76 The complete sequences of 38 PCV2 isolates from PCVAD-affected and -unaffected herds in France were determined, but molecular markers of virulence could not be identified. 26 It was concluded that all of the farms studied in Brittany are infected with closely related yet distinct PCV2 isolates and that recent PCVAD outbreaks are most likely not due to the emergence of a new genotype of PCV2. 26
It was also demonstrated that a virus strain that persisted for 10 years in an SPF herd in Sweden without causing clinical PCVAD was able to induce systemic disease under experimental conditions in pigs. 2,48 When compared with a recent Canadian reference isolate of PCV2 (PCV2–1010), it was found that both isolates were highly virulent in the PCV2-porcine parvovirus (PPV) coinfection model, which led to the overall conclusion that the virulence of the Swedish isolate was indistinguishable from that of the more recent reference Canadian strain. 48
Research on the construction and characterization of 2 chimeric infectious DNA clones of PCV1 and PCV2 has provided further insight into the virus-dependent factors. 33 The chimeric PCV1–2 DNA clone contained the PCV2 capsid gene cloned in the backbone of the nonpathogenic PCV1. The chimeric PCV1–2 virus induced a strong and specific antibody response to the pathogenic PCV2 capsid antigen and was attenuated (minimal to no lesions, low level and reduced length of viremia, low or nondetectable levels of viral antigen in lymphoid tissues) when inoculated into pigs. 33
Two amino acid mutations occurred in the capsid protein of PCV2 after 120 serial passages in cell culture and resulted in attenuation of the virus in vivo. 35 Significant differences were observed in the PCV2 genomic copy numbers in serum, and the gross and microscopic lesions in pigs inoculated with the wild-type PCV isolate were more severe than those inoculated with the passage 120 PCV2 isolate. 35 This study confirmed that minimal changes in the genome of a PCV2 isolate can markedly alter the virulence of PCV2 viruses.
Possible differences in virulence among Midwestern US field isolates of PCV2 with 98.9% nucleic acid and 96.7% amino acid sequence identities in ORF2 were investigated in an in vivo study reported in 2006. 105 PCV2 isolate ISU-40895 was isolated from a case with severe lymphoid depletion and inflammation associated with high amounts of PCV2 antigen consistent with PCVAD. In contrast, PCV2 isolate ISU-4838 was isolated from a case with no PCV2-associated lesions. The in vivo study using SPF pigs confirmed that PCV2 isolates with minimal genomic differences can differ significantly in virulence as measured by levels of virus load in serum and tissues and severity of PCV2-associated lesions. 105
Since late 2004, an increase in the incidence and severity and a change in the clinical manifestation of PCV2-associated diseases have been observed in Ontario and Quebec, Canada. Practitioners, diagnosticians, and researchers reported their observation of pathological lesions new to Ontario, including pulmonary edema, granulomatous enteritis, more severe lymphoid depletion with large numbers of circoviral inclusion bodies, and lymphoid necrosis associated with PCV2 antigen. Analysis of PCV2 isolates demonstrated a change in the type. 27 Similar observations have been reported in Quebec, Canada (L. Batista, personal communication, 2005; Gagnon CA et al.: 2007, Proc Am Assoc Swine Practitioners 38:535–540). Similar reports of increased incidence and severity of PCVAD associated with a newly emerging genotype were subsequently reported in the United States. 19 PCV2 type 1 isolates had not been reported in the United States prior to 2005 but appear to be associated with the 2006 outbreaks of PCVAD in Kansas, North Carolina, and Iowa. 19 It is of interest to note that, to date, the PCV2 isolates compared in experimental models or field studies have compared isolates only within but not across types 1 or 2 in the same model.
It is unknown if the increased finding of PCV2 type 1 (PCV2b) is due to a change (increase) in virulence, due to new introduction into the area (i.e., via semen, etc.), or if some other factor × might support increased replication of a previously insignificant PCV2 genotype. The factor × theory is supported by a retrospective cohort study done on 116 British farms between 2003 and 2004 (Green LE, Woodbine KA, Turner MJ: 2005, Proc Intern Conf Animal Circoviruses and Associated Disease, pp. 23–24). The main conclusions from this work were that PMWS behaved as an epidemic moving through a naïve population without obvious causal association between PMWS and PCV2 antigen and antibody and indicative of a novel infectious agent. Similarly, a New Zealand research group found that PMWS can be transmitted via direct and indirect contact from PMWS-affected pigs to healthy pigs, but exposure of naïve pigs to PCV2 alone did not cause disease indicative of a transmissible agent other than PCV2 as the necessary cause of PMWS (Jaros P, McIntyre LH, Morris RS, et al.: 2006, Proc Intern Pig Vet Soc, 19:168).
Host-dependent factors
Pigs of all breeds seem to be susceptible to PCV2 infection, and clinical PCVAD has been observed in a wide variety of purebred and crossbred pigs submitted to a U.S. diagnostic laboratory (PG Halbur, unpublished data). A cohort study was conducted to investigate a suspected decreased susceptibility to PCV2-associated disease in Pietrain pigs by manipulating the genetics via artificial insemination on 4 PMWS-affected farms. 116 Half of the sows were inseminated with Pietrain semen, whereas the remaining sows received the semen that was typically used on these farms. The PCV2-associated disease in the Pietrain offspring did not differ from that observed in other pigs on these farms in terms of PCV2 seroconversion, morbidity, and mortality. 116 In contrast, a field study using 2 identical 5000-sow herds with 3 different paternal genetic lines (100% Pietrain, 50% Large White/50% Pietrain, 25% Large White/75% Duroc) demonstrated that, under the circumstances of that field study, host genetics influenced the expression of PCVAD manifest as increased mortality in the offspring of the Large White/Duroc paternal line compared with the offspring of the 2 other lines. 79
Host susceptibility and its effect on the outcome of PCV2 infection were recently investigated in a controlled pilot project.” Three breeds were compared in this study: Duroc, Landrace, and Large White. The incidence of systemic PCVAD based on gross and microscopic lesions was 0% (0/23) in Durocs, 15.8% (3/19) in Landrace, and 0% (0/21) in Large White.” The purebred Landrace pigs used in this experiment were clearly more susceptible to PCV2-associated diseases as measured by severity of clinical signs and microscopic lesions associated with PCV2.” Singular experimental infection using the same PCV2 isolate at a similar cell culture passage and dose had failed to induce clinical PCVAD in crossbred SPF pigs. 32–36,49,100,101,104,106,107 Another research group experimentally induced systemic PCVAD in colostrum-deprived Landrace/Large White crossbred pigs inoculated with a PCV2 isolate recovered from a subclinically infected Yorkshire pig, implying host- and/or environment-dependant factors associated with the development of PMWS. 2
A correlation between the type of adaptive immune response against PCV2 and the level of virus replication provided evidence of host variations in the onset of the adaptive immune response, which may account for the differences in PCV2 replication and clinical manifestation and outcomes of PCV2-associated disease among pigs. 90 A recent in vitro study investigating the replication patterns of PCV2 in pulmonary alveolar macrophages found clear differences between macrophages derived from different conventional crossbred pigs suggestive of differences in susceptibility to PCVAD. 89 Although clinical and experimental evidence is minimal, further investigation of differences in host susceptibility to PCVAD is warranted.
Effect of coinfections
Experimental coinfection of pigs with PCV2 and other viruses such as ppv, 4,11,55,70,100 porcine reproductive and respiratory virus (PRRSV), 6,46,120 or bacteria such as Mycoplasma hyopneumoniae 106 has been shown to enhance the amount of PCV2 viral load and PCV2-associated lesions and to increase the incidence of PCVAD. In fact, the enhancing effect of coinfections on PCV2 replication and disease was accidentally detected when gnotobiotic pigs were inoculated with filtered cell culture material and filtered lymphoid tissues from pigs with naturally acquired PCVAD. 30 The experimentally inoculated pigs developed clinical PCVAD; however, both PCV2 and PPV were retrospectively detected in the inoculum and in the pigs.
Common coinfections that occurred in 484 US pigs with PCV2-associated systemic disease were summarized in 2002. 110 PRRSV was detected in 52% (251/484) of the cases, M. hyopneumoniae in 36% (172/484) of the cases, bacterial septicemia and/or pneumonia in 22% (105/484), swine influenza virus (SIV) in 5.4% (26/484), and singular PCV2 infection in only 2% (9/484) of the cases. 110 Aujeszky disease virus infection concurrent with systemic PCVAD was demonstrated in pigs associated with multifocal necrotizing tonsillitis and lymphadenitis. 114 Pogranichniy et al. 112 performed a case-control study on pigs with a clinical history of wasting and microscopic lesions characteristic of PCVAD and on control pigs without clinical signs or microscopic lesions typical of PCVAD. Among all viruses tested, PCV2, PRRSV, PPV, porcine enterovirus types 1 to 3, SIV, porcine respiratory coronavirus, transmissible gastroenteritis coronavirus (TGEV), porcine endogenous retrovirus, porcine lymphotropic herpesvirus type 1, and bovine virus diarrhea virus, PCV2 had the strongest association with PCVAD. The risk for clinical PCVAD was much higher if the pig was coinfected with PCV2/PRRSV. 112
In the authors' opinion, there is no single other copathogen (i.e., agent X) that can be solely attributed to enhancing the severity of PCV2-associated disease and incidence. At this time, it appears more likely that several known and unknown (agent X) pathogenic and nonpathogenic organisms that vary from region to region may be able to trigger progression of PCV2 infection to PCVAD.
Effect of immune modulation
Immunostimulation. Studies have demonstrated that immunostimulation may trigger progression of PCV2 infection to disease and lesions characteristic of PCVAD. Krakowka et al. 68 reproduced clinical PCVAD in gnotobiotic pigs stimulated with keyhole limpet hemocyanin in incomplete Freund adjuvant and inoculated with PCV2. Based on this initial work, there has been considerable interest and concern about the effect of adjuvanted vaccines on the enhancement of PCV2-induced disease, and there is a growing body of evidence in the literature to support the hypothesis that common vaccine regimens, under the right set of circumstances, may actually enhance PCV2-associated disease. 9,71,107 In contrast, others have demonstrated that PCVAD could be induced by PCV2 infection without coinfection or immunostimulation, implying that PCV2 may be a primary pathogen in some cases. 13,16,72
In addition to the type of vaccine used, other factors such as the timing of injection of adjuvanted vaccines and the age of the pig at the time of PCV2 infection may also influence the outcome of PCV2 infection. A recent study to determine if the timing of vaccination with a commercially available M. hyopneumoniae vaccine had an effect on PCV2 replication and PCV2-associated lesion severity confirmed differences in PCV2-associated lesions among the treatment groups and concluded that no or minimal PCV2-associated lesions were observed when pigs were vaccinated 2 to 4 weeks prior to expected PCV2 exposure. 101
A study was conducted to determine if the adjuvants (as opposed to the antigen) in commercial swine vaccines induce increased replication of PCV2 and increased incidence of PCV2-associated disease and lesions and if there is a difference among adjuvants in this effect. 49 The pigs were vaccinated at 4 and 6 weeks of age and were inoculated with PCV2 at 6 weeks of age. Under the conditions of the study, it was found that by the later stages of infection (35 days postinoculation), the pigs vaccinated with the oil-in-water adjuvants had an increased length of PCV2 viremia, increased amount of PCV2 in serum and tissue, and increased severity of lymphoid depletion compared to pigs vaccinated with the aqueous and aluminum hydroxide products. 49
Immunosuppression. Twelve gnotobiotic pigs were inoculated with PCV2 at 1 day of age. 69 In addition, 4 pigs received cyclosporine orally on a daily basis, and 4 pigs received corticosteroid (triamcinolone aceonide suspension) intramuscularly twice a week. The cyclosporine treatment, but not the corticosteroid treatment, resulted in increased PCV2 replication in tissues and promoted the spread of PCV2 to hepatocytes. Inflammatory reactions typical of PCVAD were absent, although tissues contained a high titer of virus, which led to the conclusion that granulomatous inflammatory lesions are immune mediated. 69 In another study, 7 cesarean-derived, colostrum-deprived pigs were inoculated with PCV2 intransally and intraperitoneally. 54 Three of the 7 pigs were treated with dexamethasone at 8 days of age and developed granulomatous lyamphadenitis, which was not observed in pigs inoculated with PCV2 alone. It was concluded that dexamethasone treatment influenced PCV2 infection of lymphoid tissues and that suppression of the cell-mediated immunity may play a role in the etiology of PCVAD. 54
Manifestations of PCVAD
PCVAD in an individual pig is diagnosed by the presence of characteristic microscopic lesions that are associated with a moderate-to-abundant amount of PCV2 antigen. To distinguish the different manifestations of PCVAD, it is important to evaluate intestines, lungs, and lymphoid tissues by immunohistochemistry for the presence of PCV2 antigen. Furthermore, on a herd basis, there is a distinction between sporadic PCVAD and PCVAD manifest at a level that it is considered a herd problem. It has been proposed that PCVAD is a herd-level problem if there is an increase in mortality of equal to or more than the mean of historic levels plus 1.66 times the standard deviation (Segalés J: 2006, Proc Am Assoc Swine Veterinarians: PCV2/PMWS seminar 12, 37:1–7). Alternatively, the chi-square test may be used to determine if the current mortality is higher than that of previous periods. If there is no historical herd mortality data available, an increase in mortality that exceeds the national or regional level by 50% is thought to be indicative of PCVAD at the herd level. In summary and for simplification, PCVAD diagnosis on a herd basis takes into account 1) the percentage of pigs diagnosed with PCVAD from all submitted pigs and 2) an increase of mortality on the particular farm investigated. In general, if PCVAD is diagnosed in 50% or more of the pigs from a representative sample of the farm and if there is a significant increase in mortality compared to previous herd parameters, PCVAD is considered to be a herd problem. If PCVAD is diagnosed in less than 50% of the pigs in a representative sample of the farm with a concurrent increase of mortality or if PCVAD is diagnosed in more than 50% of the pigs in a representative sample of the farm without concurrent increase in morality, then this is considered as sporadic PCVAD.
Subclinical PCV2 infection
A diagnosis of subclinical PCV2 infection implies that although PCV2 is present, it is not responsible for the disease observed in the pig (i.e., low amounts of PCV2 antigen associated with no to minimal lesions). Based on experimentally PCV2-inoculated pigs, it is known that PCV2 infection and lesions can be limited to 1 or 2 lymph nodes in a pig without causing any apparent clinical problems. 102,106 However, it also has been demonstrated experimentally that subclinical PCV2 infection may be associated with decreased vaccine efficacy. 104
PCV2-associated necrotizing lymphadenitis in individual lymph nodes in clinically healthy pigs has also been described. 61,102 The main lesion is follicular necrosis in the center of prominent lymphoid follicles and is usually restricted to 1 or 2 lymph nodes. The significance of this is unknown, other than in cases in which carcasses with large lymph nodes are condemned or deemed unfit for human consumption at the slaughterhouse and submission of samples to diagnostic laboratories confirms necrotizing lymphadenitis.

Eight-week-old pig experimentally coinfected with porcine circovirus type 2 (PCV2) and porcine parvovirus (PPV) showing icterus and a poor body condition typical of systemic porcine circovirus–associated disease (PCVAD).
PCV2-associated systemic infection
The primary clinical signs include weight loss or decreased rate of weight gain, paleness or icterus, and gauntness and ill thrift (Fig. 3). The infected pigs may also experience labored respiration with coughing and dark-colored diarrhea. Macroscopic lesions include but are not limited to enlarged lymph nodes. Lungs often fail to collapse and are mottled tan, and in chronic cases, the kidneys may have white streaks or spots. Less commonly, gastric ulceration can also be observed. As the name implies, systemic PCV2 infection is characterized by lymphohistiocytic to granulomatous inflammatory lesions in lymphoid tissues (Fig. 4) and/or lung, liver, kidney, heart, and intestines. Scoring systems for lesion severity and virus antigen estimation have been described (Table 3; Fig. 5). 106 One scoring system uses 7 lymphoid tissues and accounts for severity of lesions, amount of PCV2 antigen, and distribution of lesions. In brief, each lymphoid tissue (tracheobronchial lymph node, mesenteric lymph node, mediastinal lymph node, superficial inguinal lymph node, external iliac lymph node, tonsil, and spleen) is given a score ranging from 0 to 9 (lymphoid depletion score 0–3, granulomatous inflammation score 0–3, PCV2 IHC score 0–3). The individual scores of the 7 lymphoid tissues are added together and divided by 7. Depending on the final number, a pig is categorized as having no PCV2-associated lesions (score = 0), mild PCV2-associated lesions (score = 1–3), moderate PCV2-associated lesions (score = 4–6), or severe PCV2-associated lesions (score = 7–9). This scoring system has been useful for classification of the severity of experimentally induced PCVAD; however, it is unrealistic to expect this to be done on field cases, as an incomplete set of lymphoid tissues is typically submitted. The authors propose that to diagnose systemic PCV2 infection, it is necessary to at least demonstrate PCV2 antigen in more than 1 lymphoid tissue (lymph node, tonsil, spleen) or in 1 lymphoid tissue (lymph node, tonsil, spleen) and at least 1 other organ system (i.e., lung, liver, kidney, intestines) or in 2 two organ systems such as lung, liver, kidney, intestines. If abundant PCV2 antigen is associated with only 1 specific organ system, it should be referred to as PCV2-associated respiratory disease, PCV2-associated enteritis, or PCV2-associated reproductive failure rather than PCV2-associated systemic infection. If there is a limited amount of PCV2 antigen present but the lesions are severe, this is consistent with severe chronic PCVAD. With combined scoring of lesions and the amount of PCV2 antigen, it may also be possible to draw conclusions on the stage of infection (Table 4).
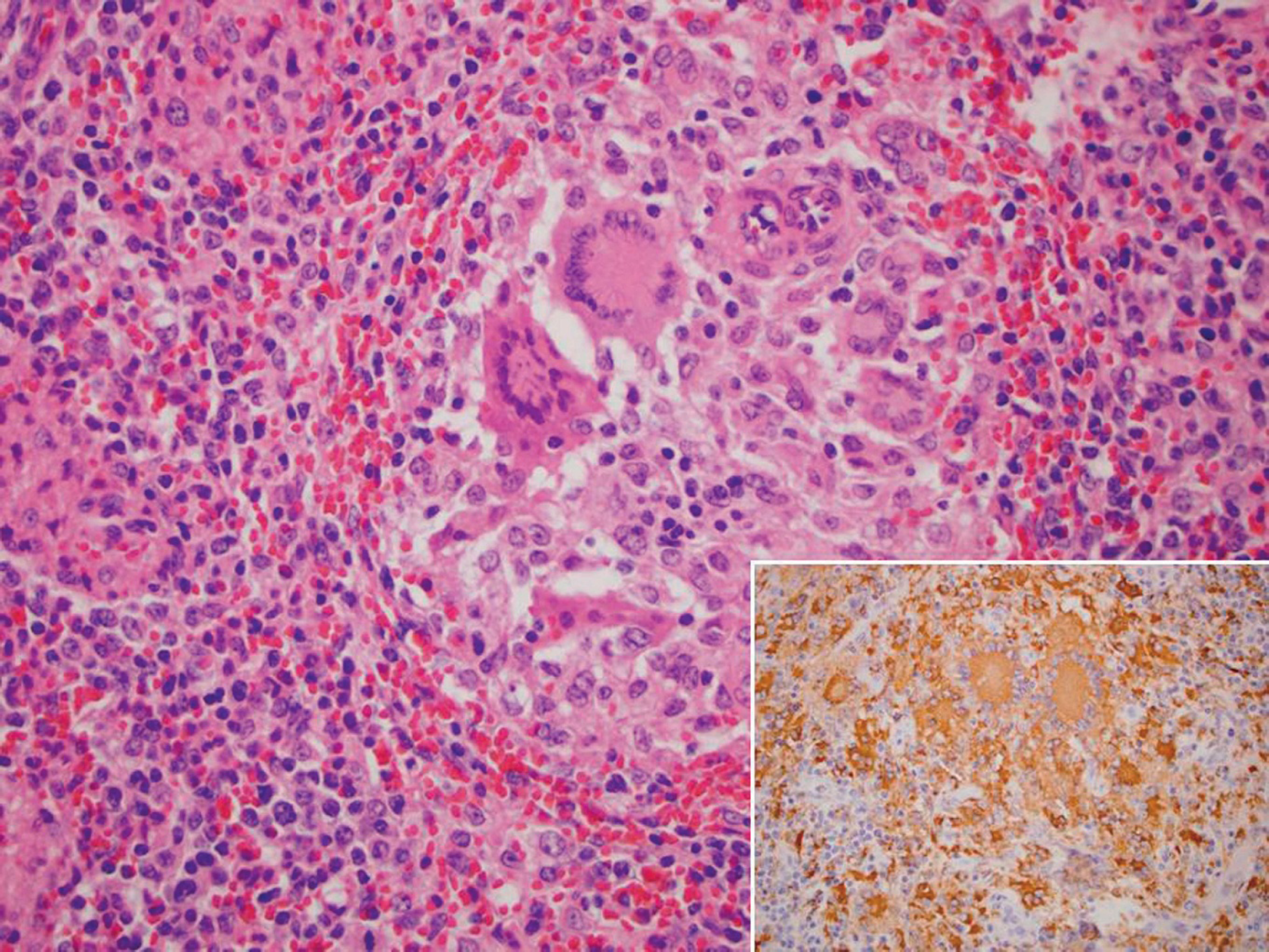
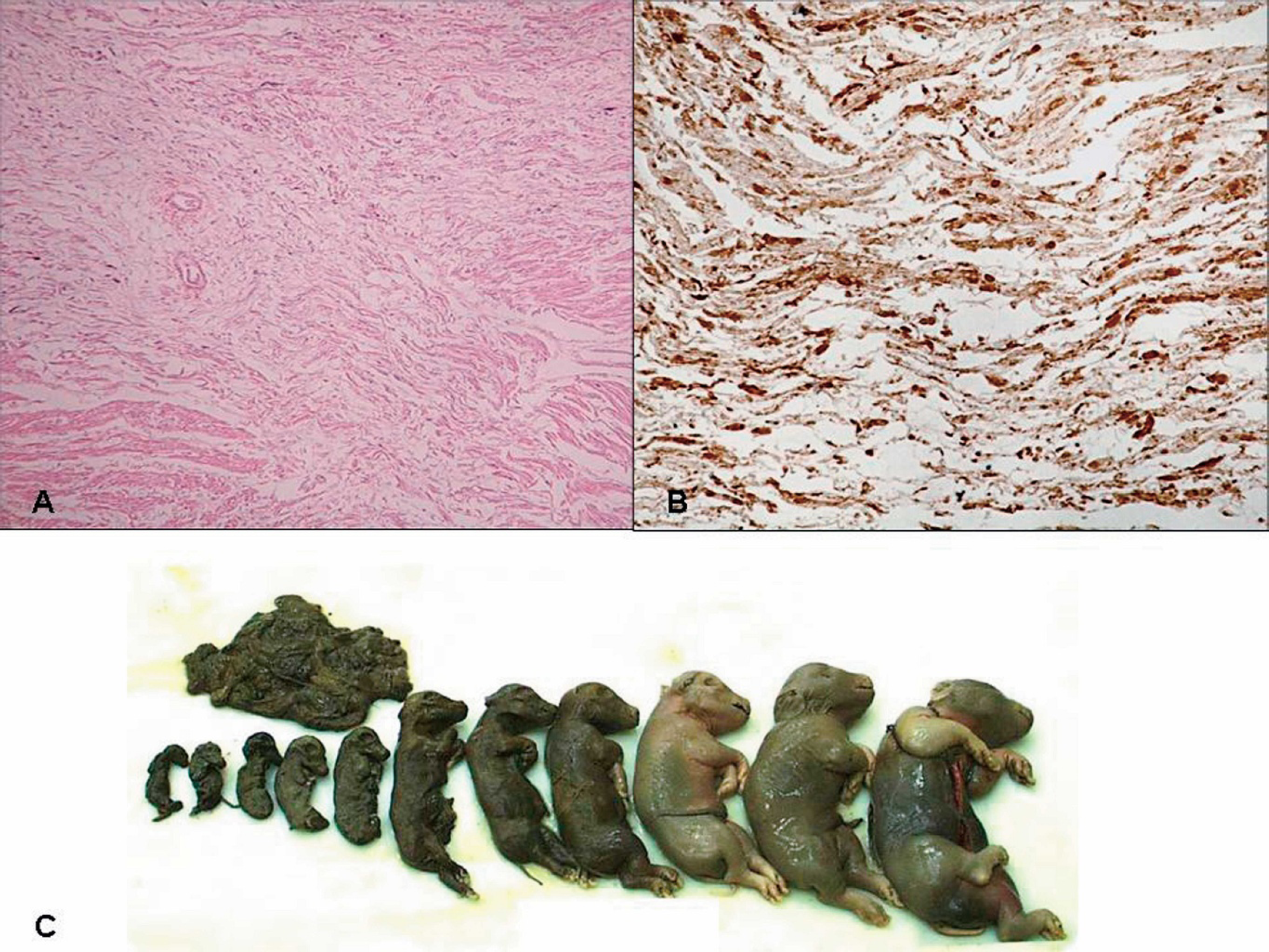
Lymph node, pig, field case. Porcine circovirus type 2 (PCV2)–associated lymphoid depletion and histiocytic-to-granulomatous replacement of the follicle with multinucleated giant cells in the center. Hematoxylin and eosin. Inset, abundant PCV2 antigen (brown staining), immunohistochemistry. Streptavidin-biotin peroxidase complex method, hematoxylin counterstain.
Scoring system for the severity of porcine circovirus type 2 (PCV2)–associated lymphoid lesions and amount of PCV2 antigen.

Percentage of lymphoid follicles that have cells with staining for PCV2 antigen demonstrated by immunohistochemistry. 106
PCV2-associated respiratory disease
Recent field investigations 45,63 and case trend analysis at U.S. diagnostic laboratories (Table 2) suggest that PCV2 may play an important role in the porcine respiratory disease complex (PRDC). PRDC is a condition observed mainly in 8- to 26-week-old pigs and is associated with multiple respiratory pathogens including PRRSV, SIV, and M. hyopneumoniae. PRDC is characterized by a decreased rate of growth, decreased feed efficiency, anorexia, fever, cough, and dyspnea. There may be diagnostic overlap between PCV2-associated systemic infection and PCV2-associated pneumonia. The presence of prolonged and unusually severe clinical respiratory disease, granulomatous bronchointerstitial pneumonia with bronchiolitis and bronchiolar fibrosis, and abundant PCV2 antigen associated with the lesions is suggestive that PCV2 may play a role in the PRDC problem.
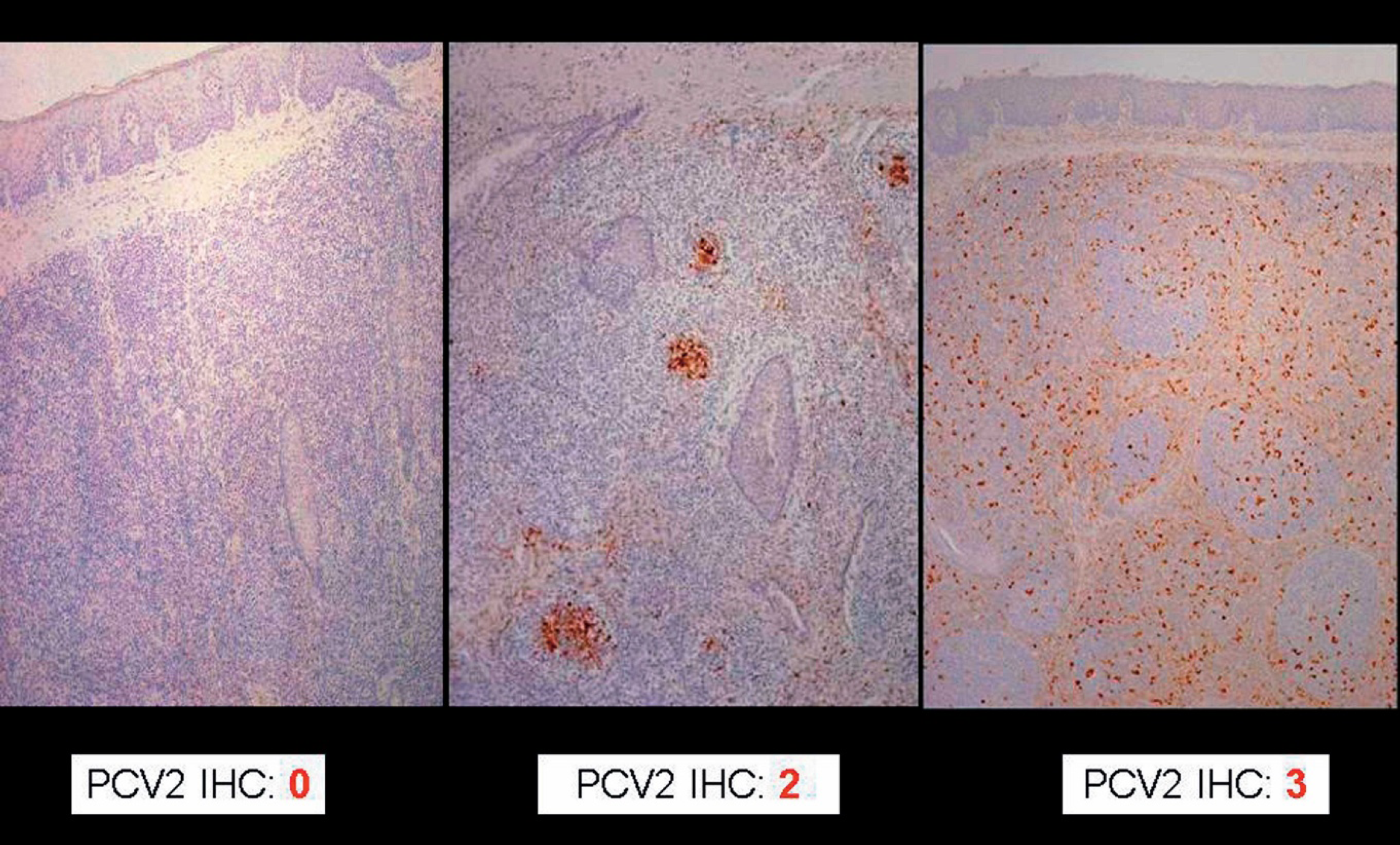
Grading of porcine circovirus type 2 (PCV2) antigen (brown staining) associated with PCV2-induced lymphoid depletion in the tonsils. Immunohistochemistry (IHC). Streptavidin-biotin peroxidase complex method, hematoxylin counterstain. The severity of lesions and amount of antigen ranges from none (IHC score of 0) to severe (IHC score of 3).
Interstitial pneumonia with bronchiolitis was reported in the first cases of PMWS (Clark EG: 1997, Proc Am Assoc Swine Practitioners 28:499–501). 29 PCV2-associated pneumonia is characterized by lymphohistiocytic to granulomatous interstitial pneumonia, peribronchiolar fibroplasia, and mild-to-severe necrotizing and ulcerative bronchiolitis (Fig. 6). The PCV2-associated bronchiolitis lesions can resemble those induced by swine influenza virus or porcine respiratory coronavirus.
Bronchointerstitial pneumonia was reproduced in addition to the hallmark lymphoid lesions of PMWS in deprived pigs inoculated with PCV2. Mild respiratory disease and multifocal interstitial pneumonia were induced in cesarean-derived, colostrum-deprived pigs inoculated with PCV2. 16 Mild interstitial pneumonia and lymphoplasmacytic rhinitis was induced in conventional pigs inoculated with PCV2. 120 Mild granulomatous bronchointerstitial pneumonia was reproduced in conventional pigs using an infectious DNA clone of PCV2. 32
Relative timing of porcine circovirus type 2 (PCV2)-associated lymphoid lesions (depletion, histiocytic-to-granulomatous inflammation, and amount of PCV2-antigen ranging from 0 to 3) based on observations in pigs experimentally infected with PCV2.
DPI 5 days postinfection with PCV2.
PCV2-associated enteritis
Cases of PCV2-associated enteritis are increasingly common. Most of the PCV2-associated enteritis field cases are from 8- to 16-week-old pigs. PCV2-associated enteritis cases often clinically and grossly resemble subacute or chronic ileitis associated with Lawsonia intracellularis. The intestinal mucosa is grossly thickened, and mesenteric lymph nodes are enlarged. 51 Microscopic examination confirms the presence of granulomatous enteritis, which is usually associated with abundant PCV2 antigen by IHC staining (Fig. 7).
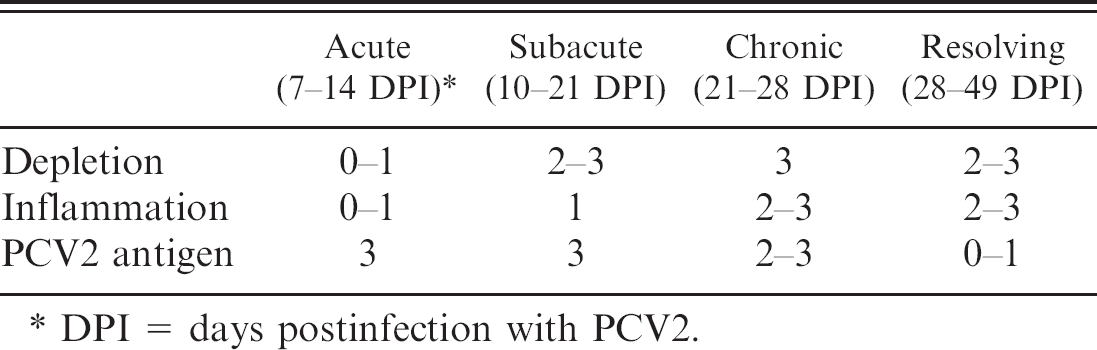
Lung, pig, experimentally infected with PCV2. A, moderate peribronchiolar lymphohistiocytic infiltration and mild necrotizing and ulcerative bronchiolitis. Hematoxylin and eosin. B, PCV2 antigen (brown staining) within the cytoplasm of macrophage-like cells, immunohistochemistry. Streptavidin-biotin peroxidase complex method, hematoxylin counterstain.
PCV2-associated enteritis was diagnosed in 6 weanling pigs by histopathology, PCV2 isolation, and PCV2 ISH 64 from a herd that was apparently free of PMWS and PDNS. The affected pigs had no lymphoid depletion or histiocytic replacement of follicles in lymphoid tissues. The authors proposed that diagnosis PCV2-associated enteritis occurs only 1) if diarrhea is present, 2) if characteristic lesions are present in Peyer patches but not in other lymph nodes, and 3) if PCV2 antigen or nucleic acids are present within the lesions. 64
PCV2-associated reproductive failure
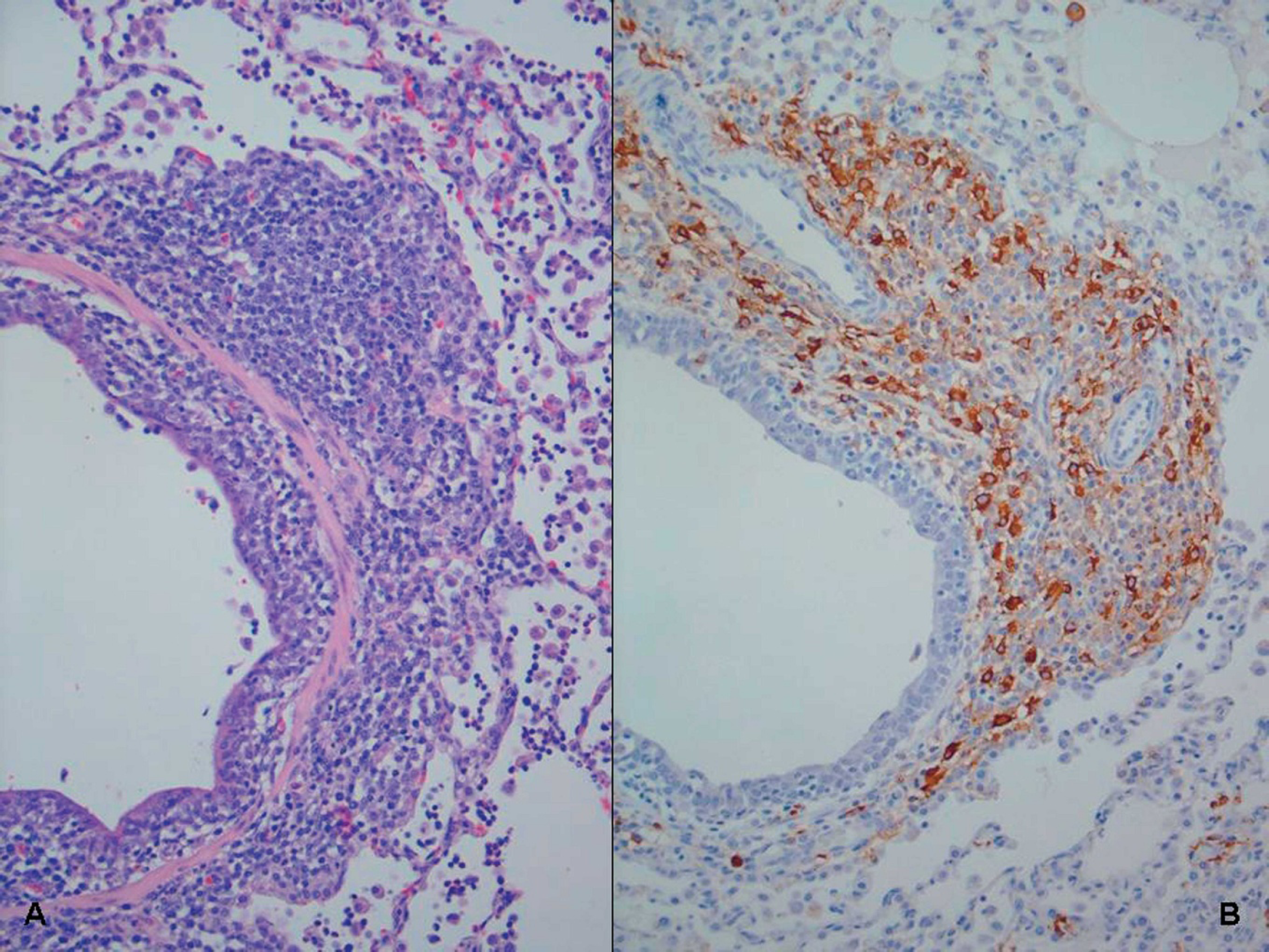
There have been several reports of PCV2-associated reproductive failure 73,96 since the original report of West et al. 144 in western Canada in 1999. Clinical manifestations on affected farms include more abortions, stillbirths, and fetal mummification (Fig. 8) and increased preweaning mortalities. Affected herds are typically gilt startups or new populations. Nonsuppurative to necrotizing or fibrosing myocarditis associated with abundant PCV2 antigen (Fig. 8) is the hallmark lesion in stillborn and neonatal pigs from field cases. 91
Experimental intrauterine infection of fetuses with PCV2 resulted in virus replication in the fetuses and supports the heart as the primary site of PCV2 replication in fetuses. 122 When fetuses were inoculated in utero on 57, 75, and 92 days of gestation, it was found that virus replication was significantly higher in fetuses inoculated at 57 days of gestation compared to those inoculated at 75 and 92 days of gestation. All fetuses were killed at 21 days postinoculation and, lesions (edema, enlarged livers, congestion) were observed only in the fetuses inoculated at 57 days of gestation. 122 In another study, 37 fetuses from 3 sows were inoculated at 86, 92, and 93 days of gestation intramuscularly, and at parturition, 24 normal pigs and 13 mummified, stillborn, or weak-born pigs were observed, confirming that PCV2 can infect late-term fetuses and cause reproductive abnormalities. 52 Although evidence is convincing that PCV2 is a reproductive pathogen, data from field cases suggest that most breeding herds are apparently immune and PCV2-associated reproductive failure is relatively rare.
PCV2-associated enteritis.
PCV2-associated PDNS
PDNS is characterized clinically by acute onset of skin lesions (raised purple skin lesions progressing to multifocal raised red-purple scabs with black centers most prominent on the rear legs; Fig. 9), fever, and lethargy. PDNS is often fatal. Other characteristic macroscopic lesions include enlarged tan, waxy-appearing kidneys with petechial hemorrhages. Microscopically, there is systemic vasculitis with dermal and epidermal necrosis and necrotizing and fibrinous glomerulonephritis. The hallmark microscopic lesions of PDNS, generalized vasculitis and glomerulonephritis, are suggestive of a type 3 hypersensitivity reaction, which is characterized by deposition of antigen-antibody aggregates or immune complexes at certain tissue sites. Several pathogens including viruses (PRRSV) 22,130 and bacteria (Pasteurella multocida, Streptococcus suis type 1 and 2, Escherichia coli, Proteus sp., Haemophilus parasuis, Actinobacillus pleuropneumoniae, Bordetella bronchiseptica, Arcanobacterium pyogenes, Staphyloccoccus aureus, or Salmonella sp.) 74,131 have been incriminated as possible etiologies for PDNS.
The association of PCV2 with PDNS was first reported in 2000. 119 Investigation of PDNS cases observed in Northern Ireland in 1990, which at the time was PRRSV free, demonstrated the presence of PCV2 antigen associated with granulomatous lymphadenitis. 8 A recent case-control study investigating PDNS in the Netherlands found that there was a significant association of high anti-PCV2 antibody titers to PCV2 and the development of PDNS. 141 The authors were not able to show PCV2 antigen by IHC in all of the PDNS cases, but they were able to confirm the presence of PCV2 DNA by PCR in all cases of PDNS. Importantly, the authors were able to show that porcine parvovirus or PRRSV nucleic acids were not present in many of the PDNS cases as determined by PCR. 141 A study comparing PCV2 serum viral load in PMWS and PDNS cases found that PDNS cases had significantly lower numbers of PCV2 DNA in serum compared to healthy, sub-clinically PCV2-infected pigs. 98 This study further confirms that PDNS pigs are infected with PCV2. However, to the authors' knowledge, no one has yet experimentally reproduced PDNS.
Porcine circovirus-associated reproductive failure.
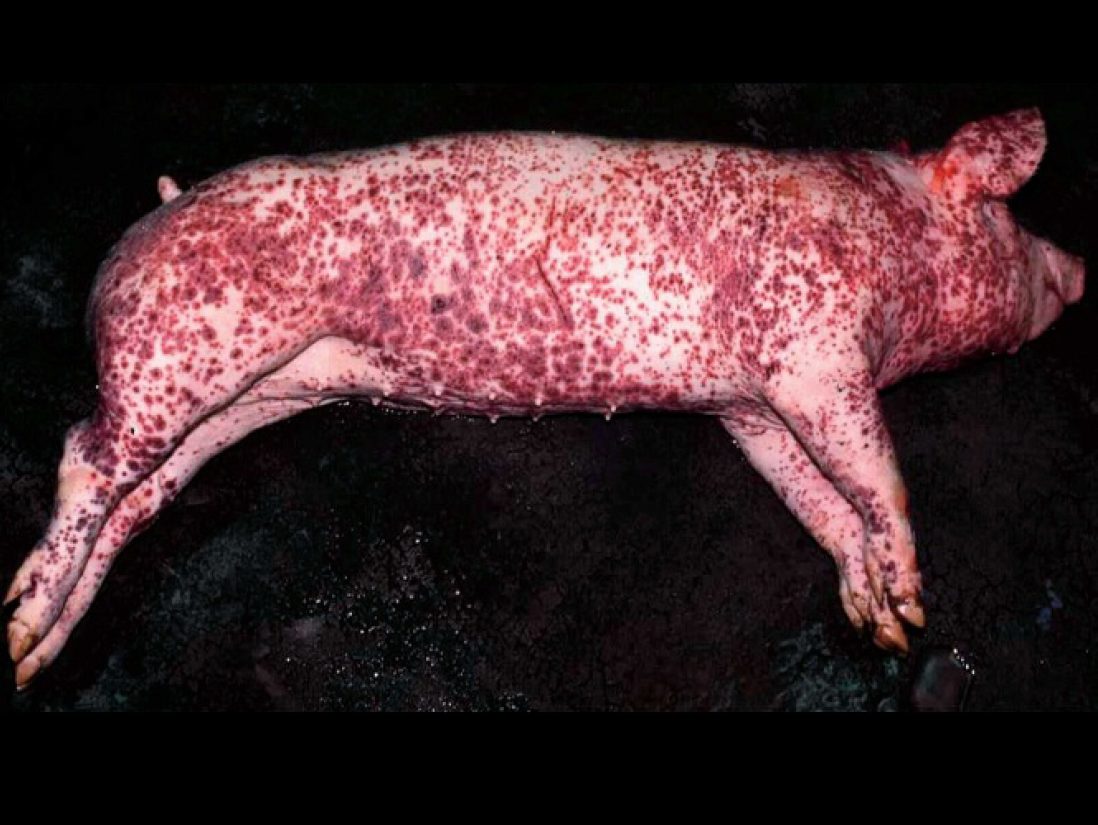
Twelve-week-old pig suffering from porcine dermatitis and nephropathy syndrome (PDNS). The perineal region, ventral abdomen, and legs are covered by raised coalescing red-purple lesions.
Diagnostic approaches and diagnostic tools available for PCVAD
PCV2 infection is ubiquitous in the global pig population. PCV2 can be found in healthy pigs as well as in diseased pigs. 3 This makes the choice and interpretation of diagnostic tests important for confirmation of PCVAD. PCVAD (systemic infection, enteritis, pneumonia, PDNS, abortion) is diagnosed by demonstration of characteristic lesions associated with PCV2 antigen or nucleic acids in the respective organs. Currently, IHC or ISH are considered the gold standard for detecting PCV2 as part of the diagnosis of PCVAD. 128
Detection of anti-PCV2 antibodies by indirect fluorescent antibody, IPMA, enzyme-linked immunosorbent assay, and serum virus neutralization assays
Serology is best used on a herd basis to determine the time of PCV2 infection by sequential or cross-sectional analyses of the population. Serological studies have found that PCV2 antibodies are present globally in almost all swine herds tested and in up to 100% of individual pigs within herds. 83,108,139 Most US breeding herds and most of the sows within those herds were found to be seropositive for PCV2. 108 The mean PCV2 antibody half-life in weanlings was estimated to be 19.0 days, and the window for PCV2-passive antibody decay within a population is relatively wide. Passively acquired anti-PCV2 antibodies present at 10 to 12 days of age were found to decay below enzyme-linked immunosorbent assay (ELISA) cutoff levels by approximately 4.9 ± 1.2 weeks of age in piglets with low levels of antibodies at weaning, by approximately 8.1 ± 1.9 weeks of age in piglets with moderate levels of antibodies at weaning, and by approximately 11.1 ± 2.5 weeks of age in piglets with high levels of passive antibodies at weaning. 108 Although PCV2 is widespread, PCV2-naïve populations and subpopulations such as young boars within boar studs exist and have been found to be susceptible to PCVAD. 103 Early identification of such naïve populations by serology may be useful to determine appropriate strategies to reduce the risk of subsequent PCV2 exposure.
Indirect fluorescent antibody assay. This assay is not automated and is subjective. Indirect fluorescent antibody (IFA) assays for PCV2 have been described in the literature. 7,111,132 Recently, an IFA assay based on an open reading frame (ORF) 2 protein has been described, 113 and it was found that the regular whole PCV2-based IFA assay had only a 57.1% relative sensitivity compared to the ORF2 protein-based IFA assay. 113 Immunofluorescence assays have also been described for antibodies against nonpathogenic PCV1 and appear to be specific. 36 Studies have shown that there is a low level of cross-reactivity between PCV1 and PCV2 on the IFA test. 7,111
IPMA. This assay is also not automated, and end points are determined subjectively. IPMA for PCV2 is widely used. 14,29 Interlaboratory testing comparing IFA and IPMA results on the same 20 serum samples performed in different laboratories in Europe and Canada found a wide variation in titers among laboratories. 85 In general, IPMA gave higher titers than IFA, and paraformaldehyde used as fixative gave higher titers than did acetone or ethyl alcohol. 85
ELISA. The ELISA is a sensitive technique for detecting and measuring serum antibodies. There are several publications describing PCV2 ELISAs. 14,77,93 Recently, commercially available IgG (Ingezim PCV IgG a ) and IgM PCV2 ELISAs (Ingezim PCV IgM a ) have been introduced in Europe. A comparison of IgG and IgM values might be useful for determining the timing of PCV2 infection: IgM value ≥IgG value: early active infection (within the first 21 days postinoculation); IgM value < IgG value: active infection (approximately between 20 and 50 days postinoculation); high IgG value and negative IgM value: late or resolving infection of convalescent (approximately 2 months after infection; Segalés J, Rodriguez J, Resendes A, et al.: 2005, Proc Intern Conf Animal Circoviruses and Associated Disease, p. 61). A variant of the regular ELISA is the competitive (blocking) ELISA. 139 A competitive ELISA specific for PCV2 antibodies is available in Europe (SERELISA PCV2 Ab Mono Blocking b ). This ELISA can also be used to detect PCV2-specific antibodies in feces (Lopez P, Guillossou S, Deshaies E, et al.: 2005, Proc Intern Conf Animal Circoviruses and Associated Disease, p. 91).
Serum-virus neutralization assay. Neutralizing antibody assays for PCV2 have been described in the literature. 90,111 Neutralizing antibodies were detected between 15 90 and 28 111 days after PCV2 infection and were correlated with protection or clearance of PCV2 infection in gnotobiotic pigs. 90 Since PCV2 does not induce a visible cytopathic effect in infected cells, the serum-virus neutralization assay requires either fluorescent antibody (FA) or immunoperoxidase staining at the end of testing to determine the presence or absence of virus replication. Requirements for pretreatment of cells to synchronize the cell cycles make serum virus neutralization tests hard to perform, and end-point titers may be less accurate. Typically, the percentage reduction is used to assess the neutralizing activity of a serum sample.
Detection of PCV2 nucleic acids by PCR and ISH
PCR. There are several PCR assays for the detection of PCV2-specific nucleic acids described in the literature. 40,43,92,125 Variants of the regular PCR assay include the following. 1) Multiplex PCR. More than 1 target sequence is detected in a single PCR step. The following multiplex PCR assays have been described in the literature: PCV2/PCV1, 109,111 PCV2/PPV, 62 and PCV2/pseudorabies virus/PPV. 50 2) Nested PCR. To increase the ability to detect very small amounts of the target sequence, nested PCR assays have been described. 57,60,75 3) Multiplex-nested PCR assays have been described for the seassays detection of PCV1/PCV2/PPV 58,66 and PCV1/PCV2. 65 4) Quantitative real-time PCR. Quantitative real-time PCR assays have been developed, and these assays allow for the determination of the amount of PCV2 genomic copy numbers in the serum or tissues. PCR reaction and detection are combined in 1 step, which decreases the turnaround time. 17,24,72,78,98,107,120 5) Reverse transcription PCR. This is used to detect PCV2 RNA, which is present only if PCV2 replicates. 146 Reverse transcription of RNA is required to make a complementary DNA for further atsomepointintheirlives.
Implications and applications for PCV2 PCR assays. Most pigs in the field (healthy or diseased pigs) are infected with PCV2 at some point in their lives. Therefore, the use of PCR, which theoretically can detect 1 genomic copy, is considered by many to be too sensitive for most applications. Demonstration of PCV2 nucleic acids by PCR cannot replace the clinical assessment of the pigs and microscopic evaluation of the tissues since PCV2 is ubiquitous, and many healthy pigs are therefore positive for PCV2-specific DNA without necessarily being affected by PCVAD. One notable exception may be the use of PCR for detection of PCV2 nucleic acids in semen to best ensure a negative status. PCR assays to detect PCV2 in semen have been described. 65,66,75 The amount of PCV2 nucleic acids in serum and tissues as determined by quantitative real-time PCR has been demonstrated to be predictive of the clinical outcome and thus may be of use to practitioners and researchers. 17,98 A threshold of 107 or greater PCV2 genomic copies per milliliter of serum was found to correlate well with severe PCV2-associated lesions and disease and poor prognosis. 17,98 Quantitative PCR may be used to accurately differentiate PCV2 infection from PCVAD by further defining PCR as positive with or without PCVAD based on the amount of PCV2 present. A PCR result would therefore be reported as negative; positive, no PCVAD (<106 PCV2 DNA copies); positive, PCVAD suspect (106 PCV2 DNA copies); or positive, PCVAD (107 PCV2 DNA copies or greater).
ISH. The ISH for PCV2 uses a labeled DNA probe that corresponds to a specific portion of the PCV2 genome. 59,86,118,126 Several ISH assays that detect multiple viruses within the same tissue section have been described: PCV1/PCV2, 56,94 PCV2/PRRSV, 22,127 and PCV2/PPV. 21
Detection of PCV2 virus or viral antigen by IHC, virus isolation, IF A, FA on tissue sections, and antigen-capture ELISA
IHC. IHC uses a monoclonal or polyclonal antibody to detect PCV2 antigen in formalin-fixed, paraffin-embedded tissue sections. 86,129 With this method, localization of the antigen within a tissue section is possible. It has been determined that an estimated viral load of a minimum of 108 PCV2 genomes per 500 ng DNA was required to give a visible staining in IHC. 17 A comparison of ISH and IHC on tissues from diseased pigs that were stored for up to 6 months in 10% neutral buffered formalin before being embedded in paraffin found that both techniques were able to detect antigen or nucleic acids in all tissues examined. 86 ISH was found to be more specific than IHC, especially when compared to IHC performed with polyclonal antibodies. 60
Virus isolation. PK-15 cells support PCV2 replication in vitro, and these cells can be inoculated with body fluids or homogenate from pigs suspected to be infected with PCV2. 111 Glucosamine treatment of the cells has shown to be effective in increasing PCV2 replication. 135 A PCV2-induced cytopathic effect is typically not observed, and to determine viral replication, immunofluorescent or immunoperoxidase staining has to be performed. Virus isolation (VI) is not routinely done for PCV2 because it is time-consuming and not always efficient, as viable virus is required and prolonged transit time and autolysis in mailed-in tissue submissions can further decrease the success of virus isolation. Applications for the use of VI may include determining if PCV2 shed in semen is infectious and the need to recover PCV2 for use in autogenous vaccine production.
Another version of virus isolation is the quantitative virus isolation. 86 For this assay, 10-fold dilutions of clinical specimens (serum, tissue homogenates) are inoculated on PK-15 cells. This test has been found to be useful in discriminating subclinical PCV2 infection from clinical PCV2 infection. 86
IFAIFA on tissue sections. IFA/FA uses a monoclonal antibody or polyclonal antiserum to detect antigen(s) in frozen tissue sections. 87 The assay is rapid but the antigen cannot be confidently associated with lesions, and the assay is relatively subjective. Studies using polyclonal antisera and monoclonal antibodies against PCV1 and PCV2 isolates on cells infected with either PCV1 or PCV2 have shown that there was no cross-reaction. 10
Antigen-capture ELISA. Anantigen-capture ELISA on tissue homogenates has been described, and the results were found to be comparable to quantitative virus isolation and IHC. 11 An antigen-capture ELISA (SERELISA PCV2 Ag Capture b ) optimized for fecal samples was also developed and tested on samples with and without a history of PMWS (Lopez P, Guillossou S, Deshaies E, et al.: 2005, Proc Intern Conf Animal Circoviruses and Associated Disease, p. 91). In the authors' experience, use of this test in the United States has produced variable results.
Electron microscopy. This method is used to demonstrate circovirus-like particles directly within a cell and to study the virus structure and size. Electron microscopy is not routinely done in diagnostic laboratories because it is time-consuming and expensive. The overall sensitivity is low, and there has to be an abundance of virus present in the tissue (at least 105 virus particles) to be detectable by electron microscopy.
Further characterization of PCV2 isolates by RFLP and sequencing
RFLP. In swine medicine, differentiation of isolates by RFLP is routinely done for PRRSV 143 and has been found to be a useful epidemiological tool to determine whether new PRRSV strains were introduced to a farm, although the stability of the RFLP pattern of a particular PRRSV is in question because of continuous genetic changes. 18 An ORF2-based PCR-RFLP assay described in 2000 using HinfI, HinP1I, KpnI, MseI, and RsaI enzymes was able to distinguish among PCV2 isolates (PCV2A, B, C, D, and E). 43 A PCR-RFLP assay using the NcoI enzyme that differentiates between PCV1 and PCV2 was also described in 2000. 31 An ORF2-based PCR-RFLP assay using Sau2AI, BanII, NspI, XbaI, and CfrI enzymes has been described recently and was able to distinguish 9 different PCV2 patterns. 142 In general, RFLP pattern analysis offers a quick means to broadly categorize PCV2 isolates; however, genomes often vary from each other outside the restriction sites, and care must be taken with interpretation.
Sequencing. With sequence analysis, it is possible to characterize the genetic information and compare isolates to each other. 23,26,31,41,67 PCV2 has 2 major genes that are oriented in opposite directions and that represent 93% of the entire genome: rep gene (replication associated; ORF1) and cap gene (capsid; ORF2). 26 The genetic difference among PCV2 isolates is mainly due to the variability in the ORF2 (90.1–100% amino acid sequence identity). 26,31 The ORF1 gene appears to be highly conserved among PCV2 isolates (99–100% amino acid sequence identity). To further investigate possible differences among PCV2 isolates, diagnostic and research laboratories may sequence only the ORF2 gene or sequence the entire PCV2 genome. 26,31 At present, sequencing results may be a useful epidemiological tool, but the knowledge to determine virulence based on sequence information is not yet available.
PCV2-associated microscopic lesions
A diagnosis of PCV2-associated disease cannot be confirmed without evaluation of microscopic lesions (depletion and histiocytic-to-granulomatous replacement of follicles in lymphoid tissues) and demonstration that PCV2 is associated with the characteristic lesions. 128 Syncytial cells are also characteristic of PCV2 infection, especially in lymph nodes, Peyer patches, and lamina propria of the intestinal villi. 118 Macrophages in affected lymphoid tissues may contain sharply demarcated, spherical, basophilic cytoplasmic inclusion bodies. The inclusions are either large and single or smaller and multiple, with groups of up to 12 inclusions. 118 The lesions typically vary from mild to severe, and a scoring system for evaluation of PCV2-associated lymphoid lesions has been described. 106
A selected group of practicing veterinarians in the Midwestern United States were encouraged to submit a complete and specific set of tissues from cases with a clinical history consistent with PCVAD (wasting/weight loss with or without respiratory disease, diarrhea, jaundice, and/or anemia) over a time period of 1 year as part of a PCV2 surveillance project in 2002 to 2003. A total of 100 cases were included in this study. All of the microscopic evaluations were performed by the same veterinary pathologist. Formalin-fixed and paraffin-embedded sections of lymphoid tissues (lymph nodes, spleen, and tonsil) were scored for the presence of PCV2 infection. After microscopic evaluation, the cases were classified as PCV2-associated systemic infection, as PCV2-associated lymphoid depletion, or as having no PCV2 association based on the presence, severity, and distribution of PCV2-associated lesions (Table 5). Only 54 of the 100 field cases suspected by experienced practitioners to be PCVAD had high amounts of PCV2 antigen associated with severe lymphoid depletion and inflammation and thus were confirmed to be PCV2-associated systemic disease. This means that 46 of the cases thought to have clinical disease consistent with PCVAD did not actually have PCV2-associated lesions and thus were clinically misdiagnosed. This further highlights the need for microscopic examination of tissues in cases suspected to be PCVAD.
Classification (porcine circovirus type 2–associated disease [PCVAD], mild porcine circovirus type 2 [PCV2]–associated lesions, or no PCV2-associated lesions) and coinfection status of 100 cases (1–2 pigs are included in each case) from farms thought to have PCVAD.

Isolation of 1 or more of the following: Streptococcus suis, Pasteurella multocida type A or D, Salmonella sp., Bordetella bronchiseptica, Haemophilus parasuis, Arcanobacterium pyogenes, Actinobacillus pleuropneumoniae, Actinobacillus suis, Escherichia coli, and Lawsonia intracellularis.
Intervention strategies
Good management practices
Prior to the availability of PCV2 vaccines in 2006 in North America, successful treatment and control of PCVAD had primarily focused on ensuring good production practices that minimize stress, eliminating coinfections or minimizing their effect, and eliminating potential triggering factors that induce immune stimulation and trigger progression of PCV2 infection to PCVAD. A 20-point plan to control PCVAD on severely affected farms was proposed. 81 The main points of this plan have been summarized as the 4 golden rules (www.thepigsite.com, accessed April 10, 2007) and include 1) limiting pig-to-pig contact, 2) reduction of stress, 3) good hygiene, and 4) good nutrition.
Risk factors for PCVAD in French farrow-to-finish herds include PPV or PRRSV coinfection of finishers, large pen size versus small pen size for weaners, and increased levels of cross-fostering, whereas long empty periods in the pig flow, regular treatment against external parasites, pen versus crated gestation, and internal versus external gilt replacement decreased the risk for PCVAD. 117 An exploratory study on risk factors for PCVAD involving 62 Spanish farms found that vaccination of gilts against PRRSV increased the odds of PCVAD expression and that vaccination of sows against atrophic rhinitis decreased the odds of the disease. 80 Investigation of the management factors associated with case herd status in farms in Manitoba, Canada, resulted in a strong association of increased piglet mortality with M. hyopneumoniae infection, PRRSV, diarrhea caused by E. coli K88, close proximity to other herds, multiple suppliers, large within-group range in age of the pigs, and not using spray-dried plasma in the first nursery ration. 28
Disinfection
Use of disinfectants in buildings and transport vehicles that have been demonstrated to be efficacious against PCV2 121 is recommended. Reduction of virus titers in vitro was observed with sodium hydroxide, Virkon S,c Roccal D Plus, d Clorox bleach, e 1-Stroke Environ, f Fulsan, g and Tek-Trol h in a controlled laboratory setting. The effectiveness of the disinfectants in commercial operations has not been tested and is unknown. In the research facility at Iowa State University, the following protocol is usually applied: after the animals are removed, the room and penning are covered with a degreaser detergent product, which is applied with a foamer at a dilution of 1:64. After 10 minutes, the degreaser detergent product is rinsed off with hot water through a pressure washer. Decontamination occurs by applying Virkon S c at a dilution of 1:30 for a contact time of 10 minutes, followed by rinsing with hot water. Prior to new occupancy, the room is fogged with Clidox-S i at a dilution of 1:5:1 and left to dry. To minimize corrosion, the room is rinsed with water 6 to 12 hours after fogging and then allowed to dry again before refilling with pigs. This procedure has been highly effective in disinfecting facilities that housed known infected pigs, as measured by the lack of transmission to subsequent groups placed in the facilities.
Control of coinfections
Coinfections play a major role in PCVAD, and evidence from the field and experimental trials have indicated that diagnosis and control of other infectious agents found in pigs with PCVAD decrease the severity of the coinfection and improve the outcome. The effect of PRRSV can be eliminated or minimized by breeding herd stabilization, pig flow changes, and/or vaccination. The effect of SIV can be eliminated or minimized by breeding herd and piglet vaccination. Specific bacterial infections (S. suis, H. parasuis, P. multocida, L. intracellularis, Salmonella sp.) need to be confirmed and can be minimized by using appropriate antimicrobials and bacterins. Use of chlortetracycline (Aureomycin j ) at an approximate dose of 22 mg/kg in the feed of pigs experimentally coinfected with M. hyopneumoniae and PCV2 provided evidence that the chlortetracycline treatment is highly efficacious in reducing lesions associated with PCV2 and M. hyopneumoniae coinfection (Opriessnig T, Thacker E, Halbur PG: 2006, Proc 19th International Pig Veterinary Society Congress 2:302).
A recent study evaluated the losses or gains associated with the use of 3 different commercially available M. hyopneumoniae bacterins in pigs experimentally coinfected with M. hyopneumoniae and PCV2 (Halbur PG, Rapp-Gabrielson V, Hoover T, et al.: 2006, Proc 19th International Pig Veterinary Society Congress, p 271). Two hundred ninety-six M. hyopneumoniae-negative pigs were randomly assigned to 1 of 4 treatment groups. Three commercial vaccines, administered as per label direction, were tested: 2 bacterins containing an oil-based adjuvant and 1 bacterin containing an aqueous-based adjuvant. The M. hyopneumoniae challenge resulted in severe macroscopic and microscopic lesions in the nonvaccinated pigs. Pigs in all vaccine-treatment groups had significantly higher mean body weights and an average daily gain on 100 and 131 days postinoculation compared with the unvaccinated controls (Halbur PG, Rapp-Gabrielson V, Hoover T, et al.: 2006, Proc 19th International Pig Veterinary Society Congress, p. 271). In contrast, another recent field study including 930 naturally infected, 53- to 54-day-old finisher pigs found no difference in the incidence of PMWS (weight loss or wasting, characteristic histopath lesions, intralesional PCV2 antigen) among vaccinated and sham-vaccinated pigs. 47 To minimize the enhancing effect of adjuvanted M. hyopneumoniae vaccine products on PCV2 replication and PCV2-assoicated lesions, it has been shown that pigs should be vaccinated 2 to 4 weeks prior to expected PCV2 exposure. 101
The use of anti-inflammatory drugs on pigs that are slow to respond may also be useful. It has been shown that the use of in-feed acetylsalicylic acid significantly (P = 0.008) reduced the incidence of antibiotic treatments in treated pigs compared with nontreated pigs on a Danish farm, with a postweaning mortality of 10% to 15% (Fruergaard M, Bækbo P, Enøe C, et al.: 2006, Proc 19th International Pig Veterinary Society Congress, p 98).
PCV2 vaccines
Experimental PCV2 vaccines have been described, tested, and generally found to be efficacious in several different in vivo models. A cestrum-derived, colostrum-deprived pig model was used to test chemically inactivated or ultraviolet-irradiated US-PCV2 isolate preparations (Pogranichniy RM, Yoon KJ, Yaeger M, et al.: 2004, Proc Am Assoc Swine Veterinarians 35:443–444). Conventional pig models were used to test a live chimeric PCV1–2 virus with the immunogenic capsid gene of PCV2 cloned into the backbone of PCV1 33,36 and to test the use of PCV2 DNA of baculovirus-expressed ORF2 protein injections as vaccine candidates. 15 A BALB/c mouse model has also been used to test a plasmid PCV2 DNA coated onto gold particles. 53
Commercial PCV2 vaccines for use in growing pigs and breeding-age animals became available in North America in 2006 (Table 6). Evidence to date indicates that the commercial vaccines are a remarkably effective tool to reduce losses in herds and production systems experiencing PCVAD in growing pigs. The inactivated, oil-adjuvanted PCV2-vaccine (CIRCOVAC®k) licensed for use in breeding-age animals was one of the first vaccines on the market and has been used most extensively in Europe. CIRCOVAC has been shown to be beneficial in reducing PCV2 circulation and shedding in the first weeks of life and improving pig health under experimental conditions (Charreyre C, Bésème S, Brun A, et al.: 2005, Proc Intern Conf Animal Circoviruses and Associated Disease, pp. 26–30). During field efficacy studies in Germany and France, the use of CIRCOVAC resulted in a rise in PCV2 antibody levels in the breeder herds and a concurrent decrease in PMWS rates in the growing pigs originating from the vaccinated breeding herds (Charreyre C, Bésème S, Brun A, et al.: 2005, Proc Intern Conf Animal Circoviruses and Associated Disease, pp 26–30). When used in Canada in 2006, field trials demonstrated that CIRCOVAC significantly (P < 0.05) decreased mortality (6.4–8.3% compared with prior to vaccination) on 67 of 77 farms after 6 months of use (Plourde N, Machell N: 2007, Proc Am Assoc Swine Vet 38:139–140).
A conditionally licensed product l for use in growing pigs became available to the North American market in April 2006. Investigations including 35,000 pigs on 21 Canadian farms showed that the mortality rate in vaccinated pigs was lowered by 77.5% when compared with nonvaccinated pigs (Grau AF, Jorgensen J, Thacker B, et al.: 2007, Proc Am Assoc Swine Vet 38:159–161).
Commercial porcine circovirus type 2 (PCV2) vaccines available in North America as of February 2007.
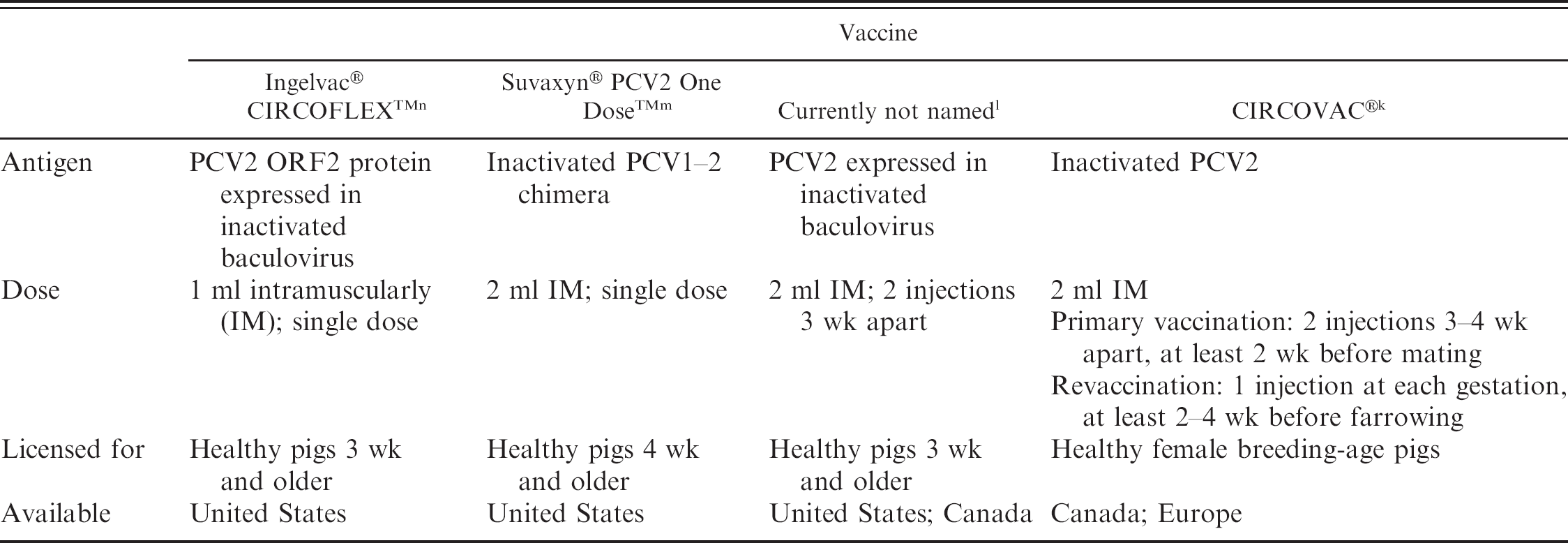
Suvaxyn® PCV2 One Dose™m is the first US Department of Agriculture-approved and fully licensed commercial PCV2 vaccine in the United States. This is the killed version of the live chimeric PCV1–2 virus. 33,36 Preliminary results from several large field studies in the United States using the killed chimeric vaccine™ demonstrated significantly (P < 0.001) decreased mortality and treatment costs in vaccinated pigs compared with nonvaccinated pigs (Connor J, Elsener J, 2007: Proc Am Assoc Swine Vet 38:151–152). The Suvaxyn PCV2 One Dose killed vaccine may have added safety features since it is based on an already attenuated chimeric PCV1–2 virus.
Preliminary results from field trials using the baculovirus expressed PCV2 vaccine Ingelvac® CIRCOFLEX™ 11 also demonstrated significant (P < 0.003) reduction in mortality in vaccinated (n = 1910) pigs compared with nonvaccinated (n = 1927) pigs on 4 different Canadian finishing sites (Desrosier R, Clark E, Tremblay D, et al.: 2007, Proc Am Assoc Swine Vet 38:143–145).
Autogenous vaccines prepared from lung or lymphoid tissue homogenates obtained from PCVAD pigs and inactivated with 2% formaldehyde have been used by some practitioners facing severe losses associated with PCVAD and the inability to attain a commercial vaccine because of limited supplies. Those practitioners generally reported marked reduction of mortality (i.e., from 20% to 3%) and limited to no adverse side effects with this regimen. The industry remains concerned about the safety and potential legal ramifications from using killed autogenous viral vaccines.
Summary
Much has been learned about the diagnosis, pathogenesis, and control of PCVAD since the disease was first recognized in the late 1990s in Canada. The scientific community now generally agrees that PCV2 is an essential component of PCVAD and that in most cases, it takes the right combination of other building blocks such as coinfections, immune stimulation, or host genetics to trigger PCV2 infection to become a herd-level problem. It has also become evident that PCVAD, when it manifests clinically, displays several disease forms such as severe systemic disease, respiratory disease, enteric disease, reproductive failure, or other as yet unrecognized manifestations. A broad selection of excellent diagnostic tools now exist for the confirmation of PCV2-associated disease and lesions, and the combination of those tools used should confirm that PCV2 is associated with the lesions. However, much knowledge is lacking about PCVAD, including understanding the molecular pathogenesis of the apparent differences in virulence among PCV2 isolates.
Indications are that the use of current vaccines in combination with good management practices are effective in reducing the impact of severe PCVAD; however, it remains unknown how pressures from vaccination and changes in production practices will affect the evolution of the virus in the pig population and, with that, the efficacy of the current vaccines. Recent findings suggest that evidence for differences in virulence of PCV2 viruses is building, and these differences in virulence may have important implications for understanding the differences in clinical manifestations of PCV2 infections and may influence future PCV2 vaccine development. However, the knowledge to predict virulence of PCV2 isolates does not yet exist, and it is unlikely that differences in virulence of PCV2 viruses fully explain the remarkably wide variations in clinical manifestations of PCV2-associated diseases from farm to farm in certain countries or across the world. Herd immunity also appears to play major role, as suggested by the declining incidence and severity of clinical PCVAD in the United Kingdom and Spain, where vaccines have not yet been widely used (Segalés J: 2006, Proc Am Assoc Swine Vet, seminar 12 [PCV2/PMWS], pp. 21–25).
Acknowledgements
The authors recognize and appreciate the financial support from National Pork Board Pork Check Off Dollars, Iowa Livestock Health Advisory Council, Pfizer Animal Health Inc, Schering Plough Animal Health Inc, US Department of Agriculture National Research Initiative Competitive Grants Program, and Fort Dodge Animal Health Inc toward PCV2 research at Iowa State University and Virginia Polytechnic Institute and State University. The authors recognize and appreciate the collaborations on PCV2 experiments by Dr. Martijn Fenaux and Dr. Nicole Juan from the Virginia Polytechnic Institute and State University and by Dr. Eileen Thacker's research group from Iowa State University. The authors also appreciate the help with animal care provided by Peter Thomas, Paul Thomas, Brian VanderLey, Matt Boogerd, Diane McDonald, and the staff from Laboratory Animal Resources at Iowa State University.
Footnotes
a.
Ingenasa, Madrid, Spain.
b.
Synbiotics Corporation, Lyon, France.
c.
Antec International, Sudbury, Suffolk, UK.
d.
Pharmacia and Upjohn, Peapack, NJ.
e.
Clorox Company, Oakland, CA.
f.
Steris Corporation, Road Mentor, OH.
g.
Fuller Brush Company, Great Bend, KS.
h.
Bio-Tek Industries Inc, Atlanta, GA.
i.
US Pharamacal Com LLC, Erie, CO.
j.
Alpharma Inc, Fort Lee, NJ.
k.
Merial Com, Lyon, France.
l.
Intervet Inc, Millsboro, DE.
m.
Fort Dodge Animal Health, Fort Dodge, IA.
n.
Boehringer Ingelheim Vetmedica Inc, St. Joseph, MO.
