Abstract
Three-week-old cesarean-derived colostrum-deprived (CD/CD) pigs were inoculated with porcine circovirus type 2 (PCV2, n = 19), porcine reproductive and respiratory syndrome virus (PRRSV, n = 13), concurrent PCV2 and PRRSV (PCV2/PRRSV, n = 17), or a sham inoculum (n = 12) to compare the independent and combined effects of these agents. Necropsies were performed at 7, 10, 14, 21, 35, and 49 days postinoculation (dpi) or when pigs became moribund. By 10 dpi, PCV2/PRRSV-inoculated pigs had severe dyspnea, lethargy, and occasional icterus; after 10 dpi, mortality in this group was 10/11 (91%), and all PCV2/ PRRSV-inoculated pigs were dead by 20 dpi. PCV2-inoculated pigs developed lethargy and sporadic icterus, and 8/19 (42%) developed exudative epidermitis; mortality was 5/19 (26%). PRRSV-inoculated pigs developed dyspnea and mild lethargy that resolved by 28 dpi. Microscopic lesions consistent with postweaning multisystemic wasting syndrome (PMWS) were present in both PCV2- and PCV2/PRRSV-inoculated pigs and included lymphoid depletion, necrotizing hepatitis, mild necrotizing bronchiolitis, and infiltrates of macrophages that occasionally contained basophilic intracytoplasmic inclusion bodies in lymphoid and other tissues. PCV2/ PRRSV-inoculated pigs also had severe proliferative interstitial pneumonia and more consistent hepatic lesions. The most severe lesions contained the greatest number of PCV2 antigen–containing cells. PRRSV-inoculated pigs had moderate proliferative interstitial pneumonia but did not develop bronchiolar or hepatic lesions or lymphoid depletion. All groups remained seronegative to porcine parvovirus. The results indicate that 1) PCV2 coinfection increases the severity of PRRSV-induced interstitial pneumonia in CD/CD pigs and 2) PCV2 but not PRRSV induces the lymphoid depletion, granulomatous inflammation, and necrotizing hepatitis characteristic of PMWS.
Porcine reproductive and respiratory syndrome (PRRS) is one of the most significant infectious diseases affecting the swine industry in the United States. 36 This syndrome and its causative agent, PRRS virus (PRRSV), an arterivirus, have been extensively reviewed. 36,44 Beginning in 1996, severe PRRSV-associated abortion storms described as acute PRRS, atypical PRRS, or sow abortion and mortality syndrome were reported in many Midwestern swine herds. 16 Outbreaks often occurred in sow herds that had received multiple doses of modified live PRRSV vaccines. Atypical PRRS outbreaks were characterized by a high incidence of abortions (10–60%), abortions at all stages of gestation, and sporadic sow mortality ranging from 5% to 10%. 19 Affected sows had lesions that had previously been associated with PRRS, including lymphohistiocytic interstitial pneumonia, encephalitis, endometritis, myometritis, and lymphoid hyperplasia. 18,36 PRRSV infection was confirmed by demonstration of intralesional viral antigen and/or virus isolation. Sows that died of atypical PRRS also frequently had hepatitis, a lesion that had not been associated with PRRS prior to 1996. Liver lesions were characterized by multifocal coagulative necrosis of individual hepatocytes and infiltrates of mononuclear inflammatory cells, suggestive of a viral etiology. 16,19,40 Preliminary experimental inoculation of cesarean-derived, colostrum-deprived (CD/CD) pigs with homogenates of hepatic tissue from affected sows indicated that the livers contained both PRRSV and porcine circovirus. 40
Porcine circovirus was first identified as a cell culture contaminant in 1974. 43 This porcine circovirus is now referred to as PCV type 1 (PCV1) and is nonpathogenic. 3 Porcine circovirus type 2 (PCV2) was first isolated in 1997, has less than 80% nucleotide homology to PCV1, and is associated with postweaning multisystemic wasting syndrome (PMWS). 5,27,29,30 PMWS is characterized by cachexia, dyspnea, and occasional jaundice or pallor in young pigs, typically between 8 and 16 weeks of age. 21 Predominant histologic lesions in affected pigs include depletion of lymphoid tissues and lymphohistiocytic to granulomatous inflammation in the lung, liver, kidney, and lymphoid tissues. 26,27,29,35 Diagnosis of PMWS is based on the demonstration of PCV2 within typical lesions. 1,39 Many of the clinical signs and lesions of PMWS have been reproduced via experimental inoculation of colostrum-deprived pigs with PCV2. 13 Coinfection with PCV2 and porcine parvovirus (PPV) induces more severe lesions and clinical disease. 2,24 PPV/PCV2 coinfection has been demonstrated in a significant portion of field cases of PMWS in western Canada. 11
PMWS has been reported in most swine-producing countries and has become a significant disease problem. 4,5,26,27,29,30,32,33,38 At the Iowa State University Veterinary Diagnostic Laboratory, both PRRSV and PCV2 antigen or genome have been detected in tissues from the majority of cases of PMWS. 39 Because PCV2/PRRSV coinfection has been associated with hepatitis in cases of atypical PRRS and PMWS, the objective of the present study was to compare clinical signs, gross and microscopic lesions, viral antigen distribution, PCV2 shedding, and serologic responses among CD/CD pigs experimentally inoculated with PCV2, PRRSV, or concurrent PCV2 and PRRSV.
Materials and Methods
Viral inocula
PRRSV strain NADC-20 was isolated in MARC-145 cells 25 inoculated with liver from a sow (No. 35358) that aborted in an atypical PRRS abortion storm and was euthanatized as part of a diagnostic investigation. Microscopic examination of the liver revealed hepatocellular swelling, multifocal hepatocellular necrosis, and periportal infiltrates of mixed inflammatory cells. The liver and several other tissues of this sow were positive for PRRSV antigen by immunohistochemical staining. The PRRSV inoculum used in this experiment consisted of cell culture medium harvested from the second passage. The inoculum contained 104. 5 TCID50/ml and was negative for PCV2 by polymerase chain reaction (PCR) methods. 29
In a preliminary study, 21-day-old CD/CD pigs inoculated with a homogenate of the liver from sow No. 35358 developed hepatic lesions similar to those present in the sow. A second passage of liver homogenate in CD/CD pigs again resulted in hepatitis. 40 PCV2 strain 35358 was isolated from pooled samples of liver and lymph node from a second passage study CD/CD pig with hepatitis and lymphoid lesions consistent with PMWS. Using hyperimmune serum collected from pigs and immunoperoxidase staining methods, PCV2 35358 was isolated in a PK-15 cell line (ATCC-CCL 33) determined to be free of PCV1, PCV2, and bovine viral diarrhea virus. 6 The PK-15 cells were determined to be free of porcine parvovirus by PCR. 28 The PCV2 35358 inoculum was prepared from the third cell passage from isolation and contained 103 TCID50/ml as determined by serial dilution on PK-15 cells and immunoperoxidase staining. 6 The PCV2 inoculum was negative for PPV by PCR as was the liver tissue from which PCV2 35358 was isolated. 28 A sham inoculum was prepared from uninfected MARC-145 cells.
Experimental design
CD/CD pigs obtained from 10 sows originating from a herd that was serologically negative for PRRSV were raised in individual incubators for 2 weeks and then placed into one of four treatment groups: control (n = 10), PCV2 (n = 19), PRRSV (n = 13), or PCV2/PRRSV (n = 17). More pigs were assigned to the PCV2 and PCV2/PRRSV groups to better assess the spectrum of lesions we anticipated would be induced by PCV2 and to accommodate the greater mortality expected in these groups. Additional group size variation was the result of the death of several pigs during the 1-week acclimation period prior to inoculation. Each group was housed in a separate room on raised decks. At 3 weeks of age, the pigs were inoculated intranasally as follows: control, 2 ml of sham inoculum; PCV2, 2 ml of PCV2 inoculum; PRRSV, 2 ml of PRRSV inoculum; and PCV2/PRRSV, 2 ml each of PCV2 inoculum and PRRSV inoculum. Clinical observations including respiratory disease scores 17 and rectal temperatures were recorded daily. Serum was collected weekly and stored at −70 C. At the end of the experiment, total serum bilirubin was determined using an automatic analyzer model 912 (Boehringer Mannheim, Indianapolis, IN) with 2,5-dichlorophenyldiazonium tetrafluorborate. Necropsies were scheduled for 7, 10, 14, 21, 35, and 49 days postinoculation (dpi). Moribund pigs were euthanatized, and necropsies were performed immediately. At necropsy, gross lesions and weights of liver, internal iliac lymph node, and superficial inguinal lymph node were recorded. Lung lesions were scored as previously described 17 to indicate the percentage of total lung that was consolidated. Rectal and nasal swabs were collected and stored at −80 C.
Histopathology
Samples of brain, lung, liver, thymus, mediastinal lymph node, tracheobronchial lymph node, mesenteric lymph node, medial iliac lymph node, superficial inguinal lymph node, ileocecal lymph node, tonsil, spleen, trachea, pancreas, duodenum, jejunum, ileum, colon, kidney, adrenal gland, lumbar vertebra, nasal turbinate, triceps muscle, quadriceps muscles, longissimus muscle, and skin were collected and fixed in 10% neutral buffered formalin for 2–4 days, routinely processed, and embedded in paraffin. Four-micrometer sections were stained with hematoxylin and eosin (HE), and light microscopic examination was performed by an observer (P. A. Harms) who was blind to the treatment groups. Lungs, livers, and lymphoid tissues were given a numerical score ranging from 0 to 6 (0 = normal; 6 = severe interstitial pneumonia, hepatic necrosis, and lymphoid depletion).
Immunohistochemistry
Immunohistochemistry for detection of PCV2 antigen was performed using a rabbit polyclonal antiserum as previously described 41 on paraffin-embedded tissues, including brain, lymph nodes, lung, liver, tonsil, spleen, trachea, thymus, skin, kidney, adrenal gland, duodenum, jejunum, and ileum, from pigs of control, PCV2, and PCV2/PRRSV groups. Immunohistochemistry for PRRSV was performed on paraffin-embedded lung and liver from pigs in the PRRSV and PCV2/PRRSV groups as described 15 using a cocktail of SR-30 and SDOW-17 monoclonal antibodies (Rural Technologies, Brookings, SD).
PCR for PCV2 DNA and PCV2 sequencing
DNA for the PCR assays was prepared using a DNeasy Tissue Kit® (Qiagen, Valencia, CA) according to the instructions provided by the manufacturer. DNA was extracted from 200 µl of serum or urine or from 10–15 µg of tissue. Nasal or rectal swabs were placed in 3–5 ml of physiological saline and mixed by vortexing, and 200 µl of the resulting suspension was used for DNA isolation. DNA was collected in 200 µl of elution buffer, and 3 µl of DNA was added to a PCR mixture with final concentrations of 1.5 mM MgCl2, 200 µM of each dNTP, 10 pM of each primer, 1 U of Taq DNA polymerase (Gibco BRL, Gaithersburg, MD), and 1× PCR buffer to make a final volume of 50 µl. The PCV2-specific primers PCV 1067 (5′-TTAGGGTTTAAGTGG-GGGGTC-3′) and PCV 1749 (5′-ATGACGTATCCAAGGAGGCG-3′) were designed to amplify a 702-base pair fragment containing open reading frame 2 (ORF 2) of PCV2 strains. Amplification of DNA was performed in 35 cycles with denaturing at 94 C for 20 seconds, annealing at 56 C for 20 seconds, and extension at 72 C for 40 seconds. Using DNA isolated from PCV1- or PCV2-infected PK-15 cells, these primers amplify PCV2 but not PCV1 (I. Morozov, unpublished data). The PCR products were analyzed with 1% agarose gel electrophoresis.
The entire genomic sequence of the PCV2 isolate used in this study was determined. The DNA was isolated from a sample of the initial inoculum and was amplified in four overlapping fragments. Priming pairs were as follows: PCV 1067 and PCV 1749, PCV 1U38 (5′-AGCTGAAAACGAAAGAAGTGCG-3′) and PCV 1045L (5′-ATGAATAATAAAACAATTACG-3′), PCV 75.1 (5′-CGGAAGGATTATTCAGCGTGAACACCC-3′) and PCV 1695 (5′-GGTAGCGGTGGCGGGGGTGGACGAG-3′), and PCV 985 (5′-ACAATCCACGGAGGAAGGGGGCCAG-3′) and PCV 1494 (5′-CAGAATAAGAAAGGTTAAGGTTG-3′). An additional primer, PCV 389 (5′-GCTGTGAGTACCTTGTTGGAGAGCGGG-3′), was used for sequence analysis of the fragment amplified with primers PCV1U38 and PCV 1045L. Sequence analysis was performed in the Nucleic Acid Facility of Iowa State University, and sequencing data were analyzed with the Geneworks program (IntelliGenetics, Mountain View, CA).
Serology
Blood was collected from each pig 6 days prior to and on the day of inoculation, at weekly intervals following inoculation, and at necropsy. Sera were stored at −70 C. Antibody reactivity was measured by immunofluorescence for PCV2, 34 by enzyme-linked immunosorbent assay (ELISA) (Herdcheck®, IDEXX, Westbrook, ME) for PRRSV, and by hemagglutination inhibition for PPV. 23
Data analysis
Statistical analysis of lymph node weights was performed using the analysis of variance (ANOVA) of SAS version 8.1 (SAS Institute, Cary, NC) and evaluated by Tukey's studentized range test. Nonparametric data (respiratory scores, lung lesion scores, and lymphoid depletion scores) were evaluated using the Wilcoxon test of the NPAR1WAY procedure of SAS 8.1 and further evaluated using the Kruskal–Wallis test. 8 The infectious nature of the treatments required that the groups be housed in separate, adjacent rooms. Rooms had identical equipment, including flooring, feeders, and waterers, and room temperatures were monitored daily to maintain a consistent environment in all rooms. All pigs had continuous access to water and were provided the same feed ad libitum. The same personnel provided daily animal care for all groups. Because the above efforts were made to minimize any differences among rooms that were unrelated to treatment, no pen effect was assumed in the statistical analysis. The control pigs that became infected with PCV2 were not included in the data analysis.
Results
Clinical signs
Control Pigs
Control pigs exhibited minimal respiratory disease (Fig. 1); all individual observations were of no disease or mild disease and were scored a 0, 1, or 2 on a scale of 0 to 6. No icterus was observed in any of the control pigs. During the first 2 weeks of the study, four pigs in the control group developed mild exudative epidermitis on the sides of the face and pinna that resolved with ceftiofur (Pharmacia–Upjohn, Kalamazoo, MI) therapy. One of two pigs remaining in the study through day 49 developed an intermittent fever (>40 C) beginning at 36 dpi and recurring through 49 dpi. This pig had a mild relapse of exudative epidermitis during the last 2 weeks of the study. No control pigs died or were euthanatized except for scheduled necropsies.
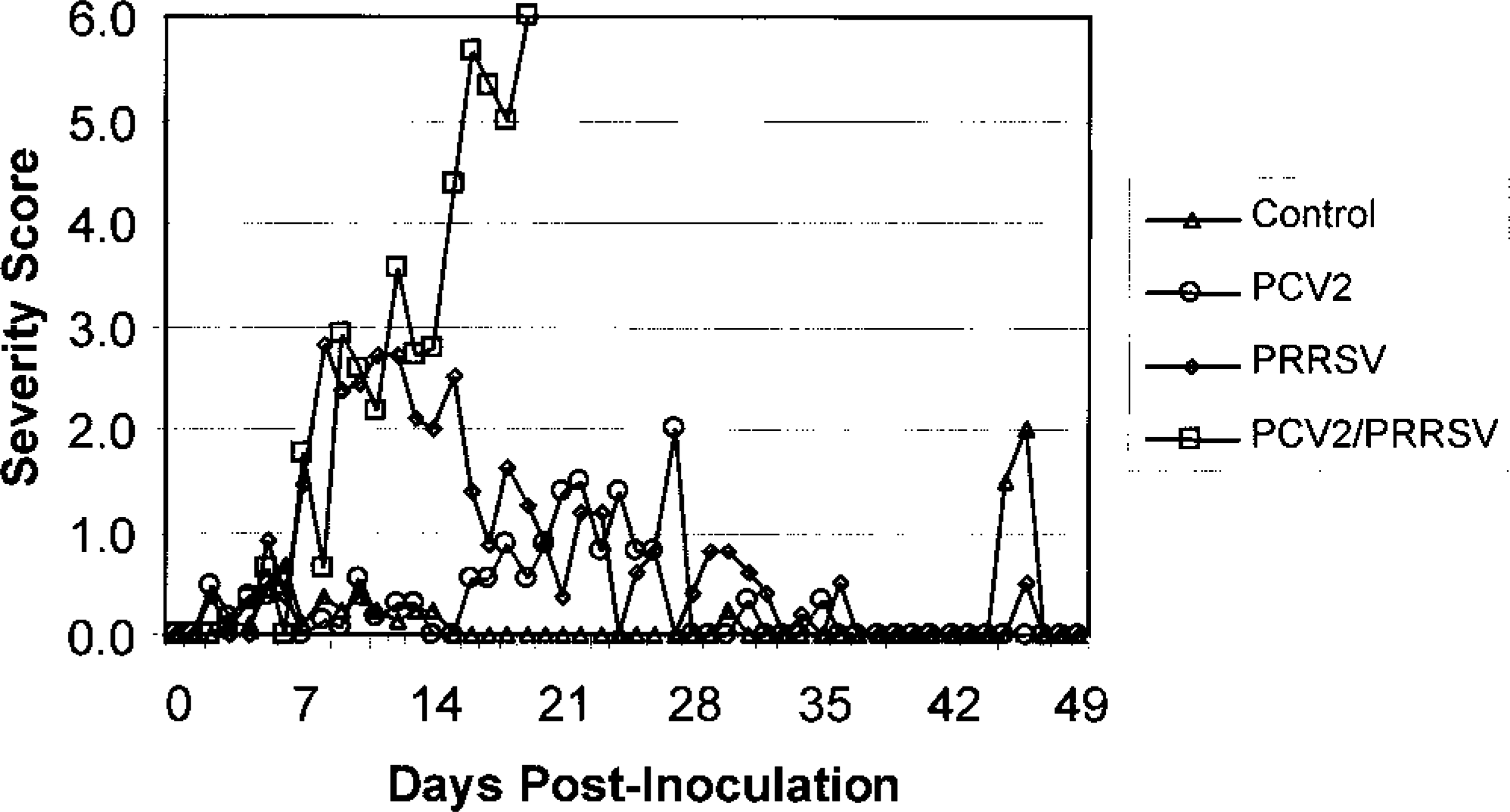
Mean respiratory scores for the four inoculated pig groups: sham (control), PCV2, PRRSV, and PCV2/PRRSV. Individual pigs were scored daily for respiratory distress based on the following scale: 0 = no respiratory distress, 1 = mild following activity, 2 = mild at rest, 3 = moderate following activity, 4 = moderate at rest, 5 = severe following activity, 6 = severe at rest.
PCV2-inoculated pigs
Mild clinical respiratory disease (tachypnea) was observed in the PCV2 pigs. Although respiratory disease was mild, it was significantly greater (P ≤ 0.05) than that of the control group during the 3rd week postinoculation. Mean rectal temperatures rose above 40 C beginning at 12 dpi and remained elevated through 28 dpi. Icterus was observed in 4/19 pigs and was most evident between 20 and 27 dpi. Exudative epidermitis first appeared at 3–7 dpi and was mild to moderate and multifocal in 14/19 pigs; it progressed to severe and diffuse in 8/19 pigs between 10 and 21 dpi. These pigs were treated with ceftiofur (Pharmacia–Upjohn), but not all pigs recovered. One pig died at 12 dpi with severe exudative epidermitis, and three pigs died with hemorrhagic gastric ulcers (at 21, 23, and 27 dpi). Two pigs were selected for euthanasia at 21 dpi because of severe depression, lethargy, and/or icterus.
PRRSV-inoculated pigs
PRRSV-inoculated pigs developed severe respiratory distress by 7 dpi that continued through 21 dpi, as demonstrated by a significant rise (P ≤ 0.05) in respiratory disease scores compared with those of the sham- or PCV2-inoculated pigs (Fig. 1). Tachypnea and pronounced abdominal breathing were first observed at 4 dpi. Mean respiratory scores began to decrease at 14 dpi, and by 28 dpi the surviving pigs were clinically normal. Mean rectal temperatures were increased during the period of maximal clinical disease until 14 dpi, after which temperatures remained normal. Neither icterus nor exudative epidermitis were observed. No pigs in this group died or were euthanatized except for scheduled necropsies.
PCV2/PRRSV-inoculated pigs
Respiratory disease scores in pigs receiving the combined inoculum were similar to those of the PRRSV group through 7 dpi but were significantly greater (P ≥ 0.05) than those of the control and PCV2 groups. However, mean respiratory scores increased steadily in the PCV2/PRRSV pigs from 4 to 20 dpi and were significantly higher (P ≤ 0.05) than those of the control, PCV2, and PRRSV groups between 14 and 21 dpi (Fig. 1). Mean rectal temperatures were similar to those of the PRRSV group through 7 dpi but remained elevated through 20 dpi. Icterus was observed in 7/11 pigs after 12 dpi. Scheduled necropsies of PCV2/PRRSV pigs occurred at 7 and 10 dpi. Between 10 and 20 dpi, 10 of the 11 remaining pigs developed severe dyspnea and depression and either died (4 pigs) or were euthanatized in extremis (6 pigs). The last pig in the PCV2/PRRSV group died at 20 dpi. One pig, not in extremis, was euthanatized at 14 dpi as scheduled.
Serum bilirubin
Total bilirubin in serum collected at necropsy was normal (>0.6 mg/dl) in 7/7 control pigs and 13/13 PRRSV pigs. Total serum bilirubin at necropsy was elevated (≥0.6 mg/dl) in 1/12 PCV2 pigs (21 dpi, 7.36 mg/dl) and 6/13 PCV2/PRRSV pigs (12–17 dpi, 0.75–9.23 mg/dl).
Macroscopic lesions
Control pigs
No macroscopic lesions were noted in any of the control pigs, other than the mild exudative epidermitis.
PCV2 pigs
Mild lung lesions consisting of patchy, tan to red mottled areas located primarily in the cranial and intermediate lobes were observed in the 9/13 PCV2 pigs studied between 7 and 21 dpi (Fig. 2). Lymph nodes were unremarkable, and the medial internal iliac lymph nodes were not significantly enlarged when compared temporally (8–21 dpi) with the controls by ANOVA and adjusted as a percentage of body weight (○ ± SE: 0.0191% ± 0.0017% compared with 0.0178% ± 0.0032% for the controls). Four pigs had swollen, mottled livers at necropsy (at 12, 14, 17, and 21 dpi), and icterus was evident in the sclera, skin, and connective tissues. Exudative epidermitis lesions consisted of thickened, reddened areas covered by greasy crusts. Staphylococcus hyicus was isolated from the lesions of 3/3 pigs. Ulcers of the gastric pars esophagea were present in three pigs found dead at 21, 23, and 27 dpi; in each pig, clotted blood filled the gastric lumen and extended into the small intestine.
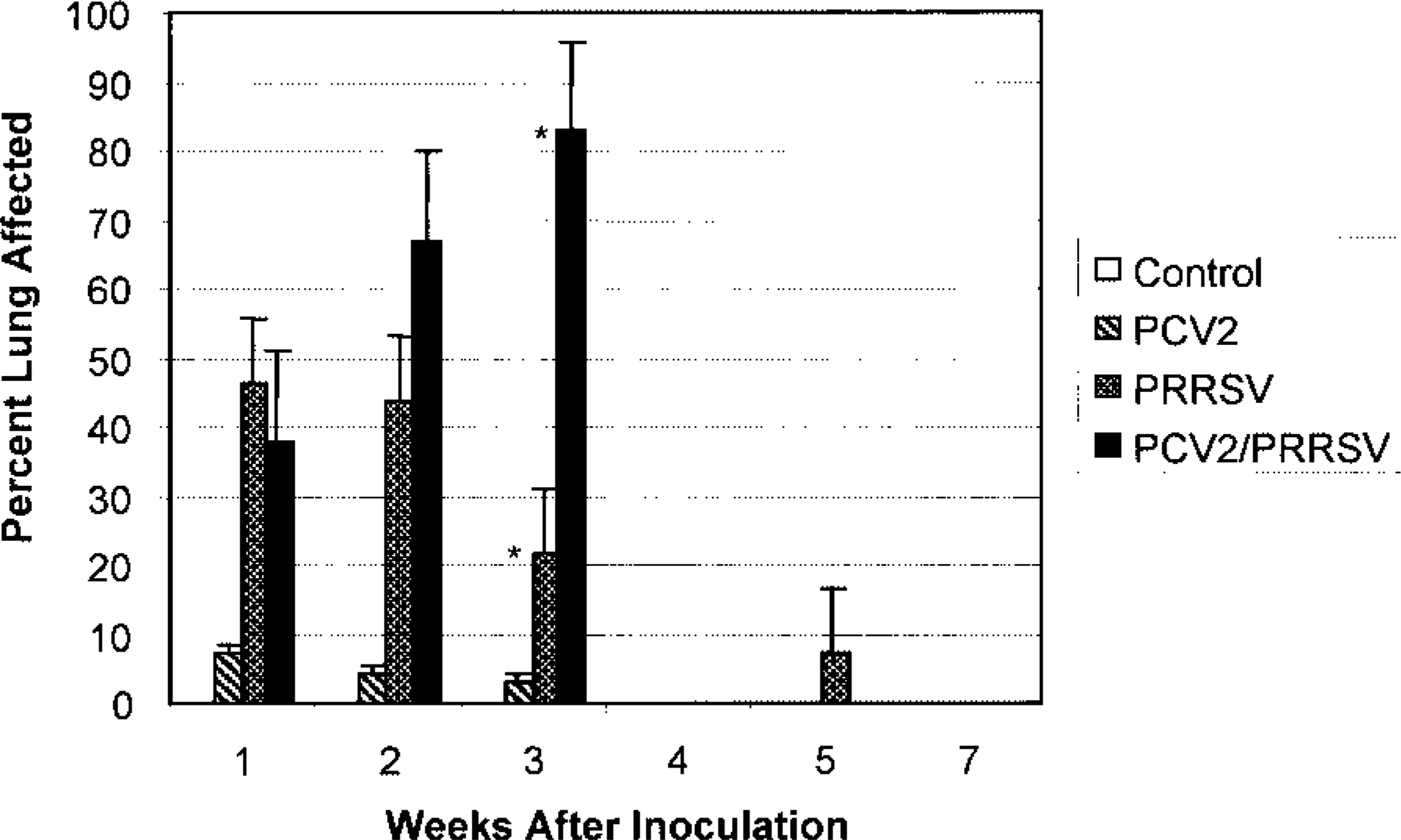
Mean grades for macroscopic lung lesions for the four inoculated pig groups: sham (control), PCV2, PRRSV, and PCV2/PRRSV. Lungs were graded based on a percentage of lung involvement. No pigs in the PCV2/PRRSV group survived past the 3rd week postinoculation, and no necropsies were performed in the sham- or PRRSV-inoculated groups at 4 weeks postinoculation. ∗Gross lung scores were significantly different (P ≤ 0.05) in the 3rd week postinoculation. Error bar = SE.
PRRSV pigs
At 7 and 14 dpi, lungs failed to collapse and had patchy to coalescing tan to purple areas of consolidation affecting all portions; less severe changes were present at 21 and 28 dpi. Lungs of pigs studied after 28 dpi were grossly unremarkable. Lymph node weights were compared for pigs studied during weeks 2 and 3. Lymph nodes were enlarged, and the medial internal iliac lymph nodes were significantly heavier (P ≤ 0.05) in the PRRSV group (0.0323% ± 0.0047% of body weight) than in the sham, PCV2, or PCV2/PRRSV groups (0.0178% ± 0.0032%, 0.0191% ± 0.0017%, and 0.0158% ± 0.0007% of body weight, respectively). Moderate to severe mesocolonic edema was present in four of the pigs (two pigs at 7 dpi, one pig each at 10 and 15 dpi). No icterus or macroscopic liver lesions were present in any of the pigs in this group.
PCV2/PRRSV pigs
Lung lesions were similar to, but more severe than, those observed in PRRSV pigs. The most severely affected lungs were diffusely tan to purple, noncollapsed, and rubbery with a meaty appearance on cut surface. At 7 dpi, mean gross lung lesion scores for PRRSV and PCV2/PRRSV pigs were significantly greater (P ≤ 0.05) than those of the sham or PCV2 pigs (Fig. 2). Among pigs studied between 15 and 21 dpi, mean lung lesion scores of the PCV2/PRRSV pigs (n = 8) were significantly greater (P ≤ 0.05) than those of the PRRSV pigs (n = 3) (Fig. 2). Lymph nodes were not visibly enlarged. Livers in 3/11 pigs studied after 10 dpi were swollen and mottled dark red-brown, and the serosa of the gallbladder and bile duct was edematous and icteric. Hemorrhagic ulcers of the gastric pars esophagea were present in two pigs that died at 17 dpi. Mesocolonic edema was present in six pigs (three pigs at 7 dpi, one pig each at 10, 13, and 15 dpi.). No significant differences in liver or superficial lymph node weights were present between the groups.
Histologic lesions
Control pigs
No lung lesions were identified in any of the tissues examined from control pigs at 7, 14, 28, and 36 dpi. At 21 dpi, one pig had multifocal necrotizing arteritis that was most severe in the lungs; bacterial culture, PCR for PCV2, immunohistochemistry for PRRSV and PCV2, and virus isolation for PCV2, PRRSV, classical swine fever virus, encephalomyocarditis virus, hemagglutinating encephalomyelitis virus, transmissible gastroenteritis virus, adenovirus, rotavirus, reovirus, Aujeszky's disease virus, PPV, enterovirus, and swine influenza virus (SIV) using tissues from this pig were all negative. Both pigs studied at 49 dpi had mild exudative epidermitis and small peribronchiolar infiltrates of mononuclear inflammatory cells. Mild lymphoid depletion in an isolated lymph node was present in 1/1 pig at 7 dpi, in 2/2 pigs at 14 dpi, and in 2/2 pigs at 36 dpi. Mild to moderate lymphoid depletion was present in several lymph nodes in one pig at 49 dpi; rare multinucleate giant cells were present within follicles of two of the lymph nodes.
PCV2 pigs
Between 10 and 49 dpi, there were small to occasionally medium-size peribronchiolar cuffs of mononuclear cells, primarily around terminal bronchioles. Coagulative necrosis of scattered individual bronchiolar epithelial cells and focal areas of mild transitional to squamous bronchiolar epithelial metaplasia (Fig. 3) were variably present between 14 and 21 dpi. Four of the seven pigs studied between 15 and 28 dpi had hepatitis and hepatic necrosis that was mild to severe. Mild lesions consisted of infiltrates of mononuclear inflammatory cells in the portal tracts and occasional small clusters of mononuclear cells within the acini. Moderate lesions also included hepatocellular swelling, individual hepatocellular necrosis, and increased numbers of mononuclear cell infiltrates. Severe hepatic lesions were characterized by periacinar or massive hepatocellular necrosis, disruption of the hepatic plates, expanded and congested sinusoids and large numbers of mononuclear cells within acini and portal tracts (Fig. 4).
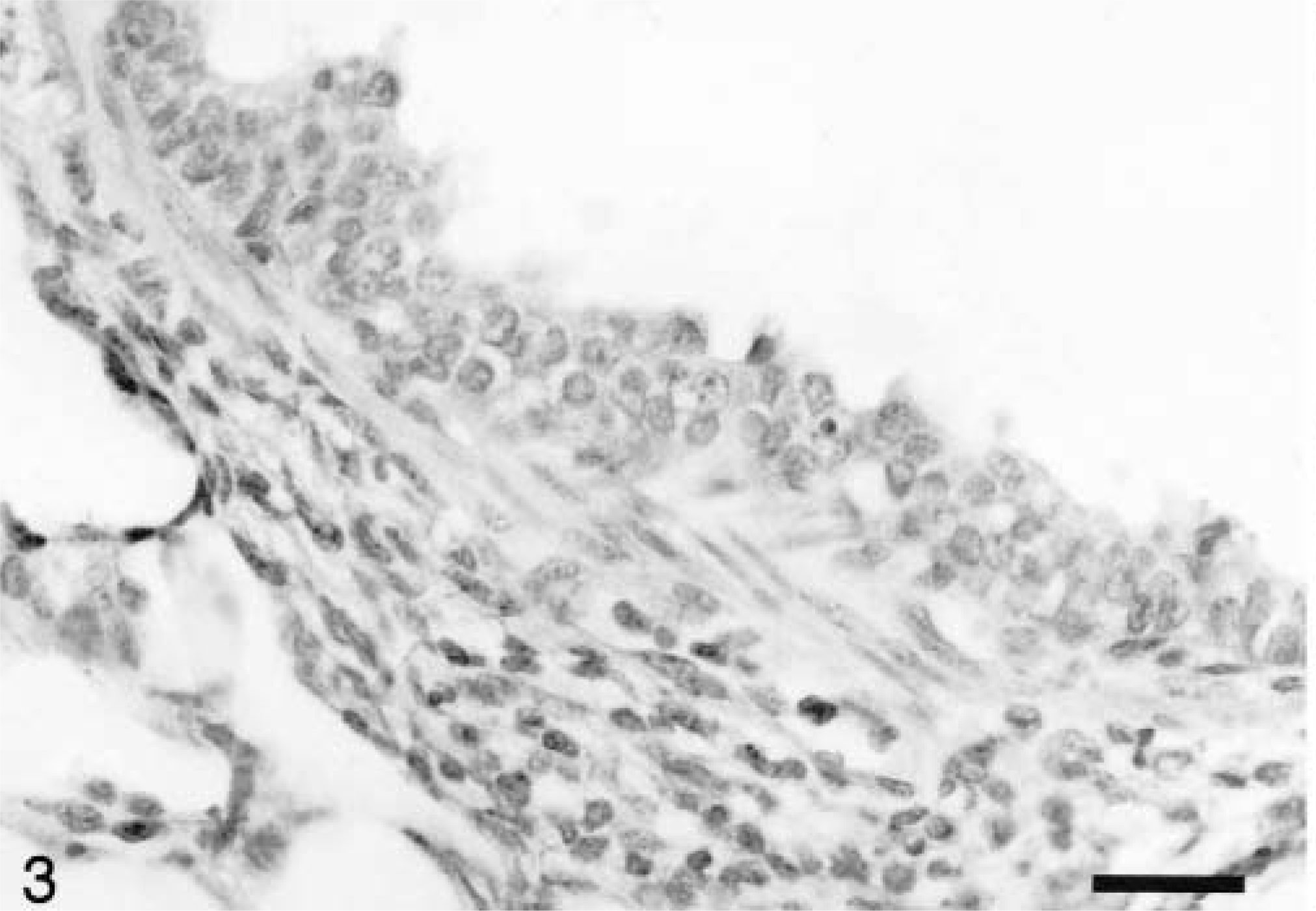
Lung; pig inoculated with PCV2 14 days previously. The bronchiolar epithelium exhibits transitional metaplasia, and there are peribronchiolar aggregates of mononuclear inflammatory cells. HE. Bar = 66 µm.
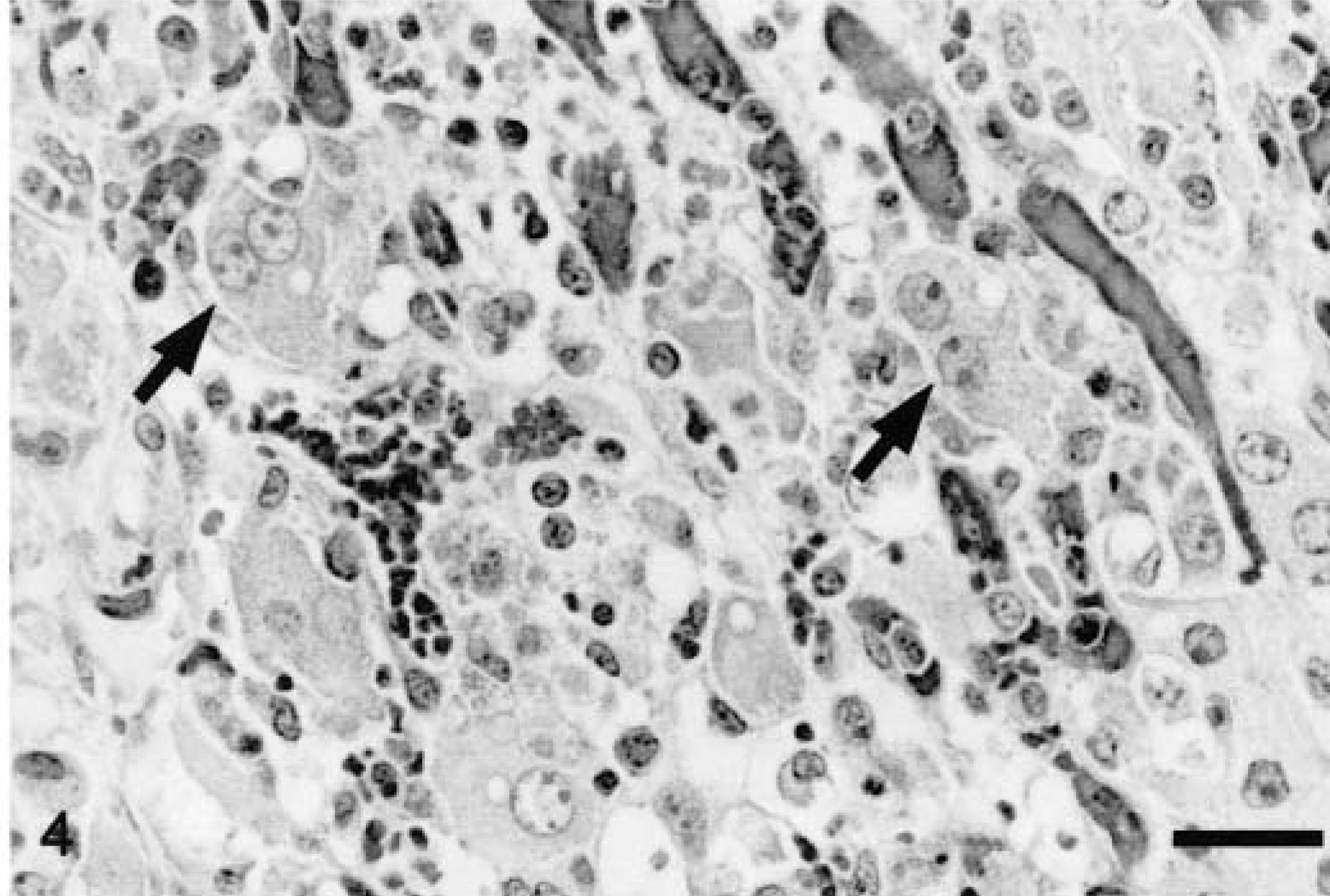
Liver; pig inoculated with PCV2/PRRSV 17 days previously. Hepatic plates are in disarray and hepatocytes are markedly enlarged and occasionally binucleate (arrows). Hepatocytes are separated by congested sinusoids and moderate numbers of plasma cells and macrophages. HE. Bar = 31 µm
Some degree of depletion of at least one lymphoid tissue (tonsil, spleen, thymus, Peyer's patch, and lymph nodes) was observed in all PCV2 pigs (Fig. 5) except for one pig at 7 dpi, one at 36 dpi, and one at 49 dpi. Lymphocyte numbers were reduced in both follicles (Fig. 6) and the paracortex. Macrophages in depleted follicles often contained clusters of round basophilic to amphophilic intracytoplasmic inclusions ranging from 2 to 10 µm in diameter (Fig. 7). These inclusions were most common at 21 and 28 dpi. Multinucleate giant cells were occasionally present but were not a consistent feature. Eosinophils were present in variable numbers in the lymph nodes. The lymphoid tissues affected and the severity of depletion were variable both within the same animal and between animals.
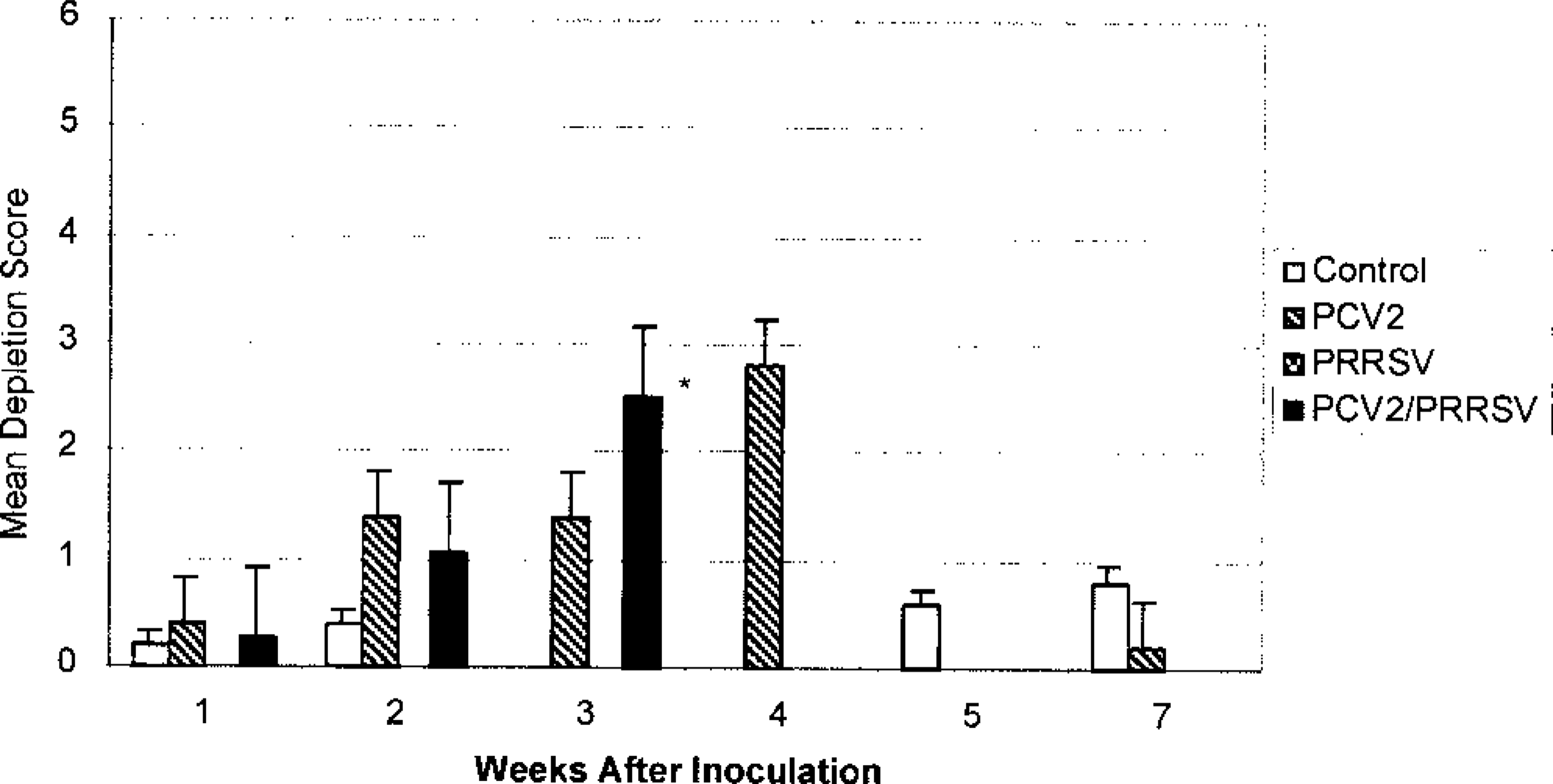
Mean composite score of lymphoid depletion in inoculated pigs. Individual composite scores were obtained as an average of the severity of lymphoid depletion in six selected lymph nodes, tonsil, spleen, Peyer's patches, and thymus. Severity score was based on a range of 0 to 6, where 0 = none and 6 = severe lymphoid depletion. None of the PCV2/PRRSV-inoculated pigs survived past 3 weeks postinoculation. ∗Lymphoid depletion for PCV2/PRRSV-inoculated pigs was significantly different (P ≤ 0.05) from that of the controls during the 3rd week postinoculation. Lymphoid depletion in the PCV2-inoculated pigs during the 4th week postinoculation was similar to that on the 3rd week postinoculation, but no control pig necropsies were performed during the 4th week postinoculation. Error bar = SE.
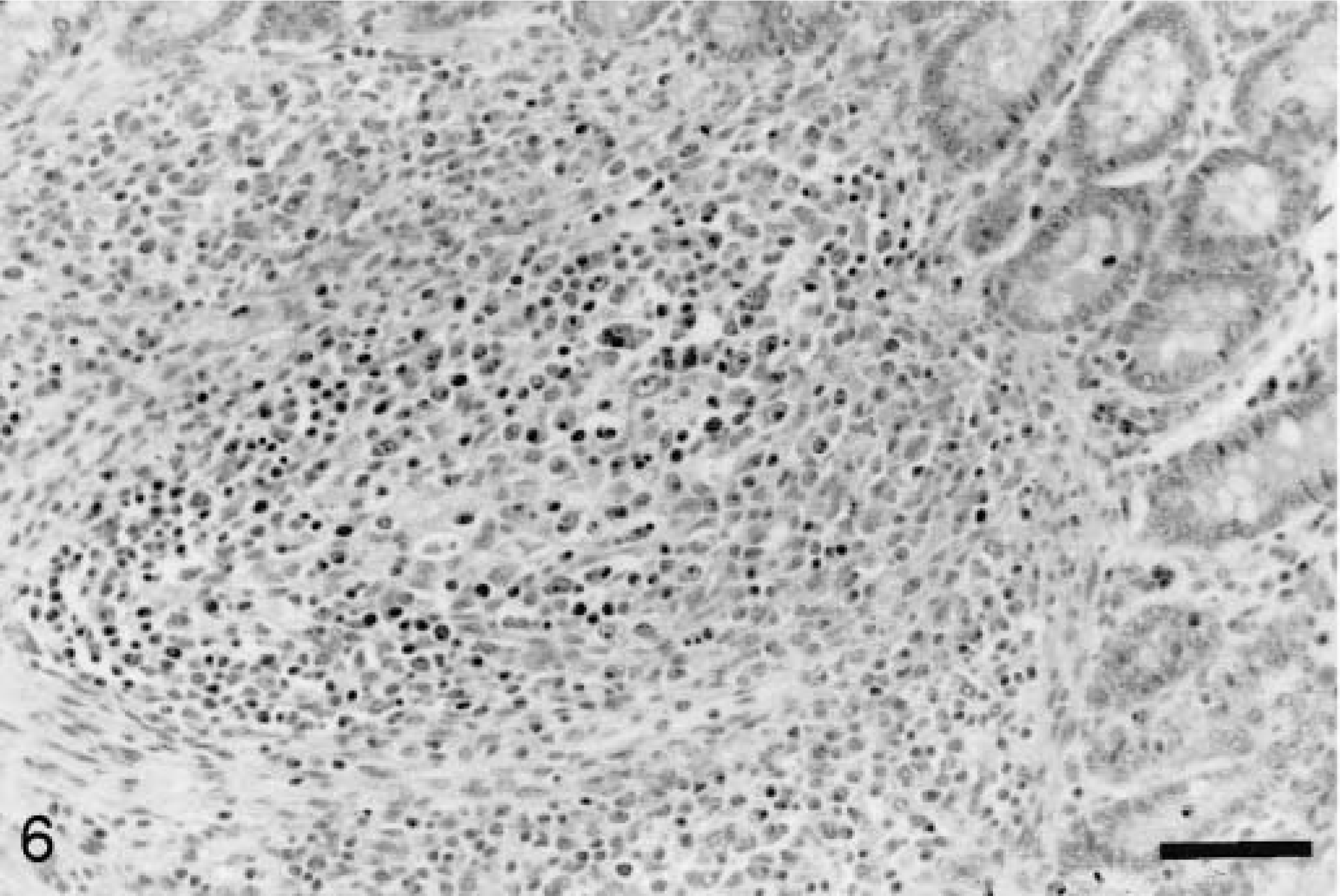
Ileum; pig inoculated with PCV2 21 days previously. Peyer's patch lymphoid tissue is predominantly replaced by large macrophages. HE. Bar = 83 µm.
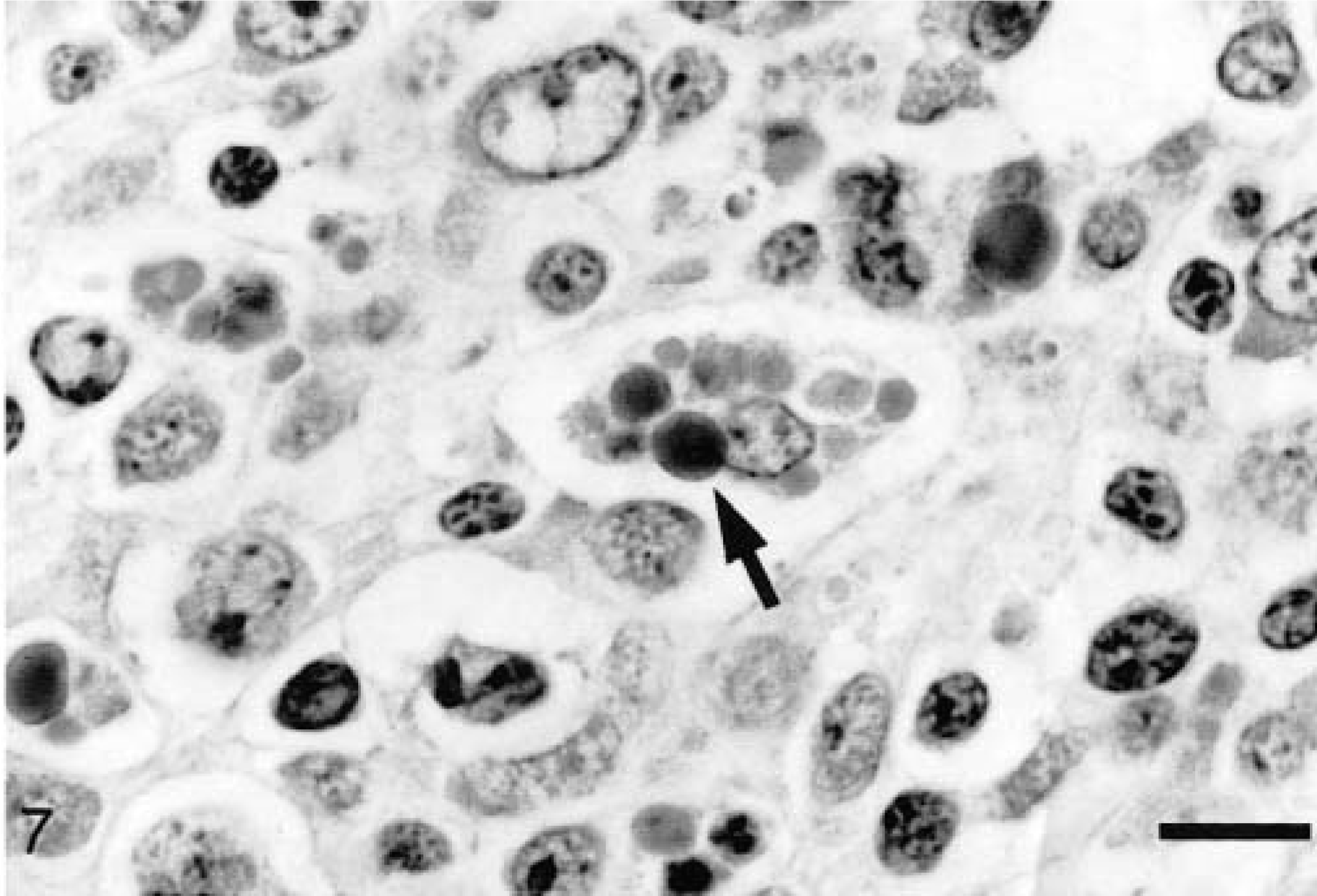
Ileum; pig inoculated with PCV2/PRRSV 21 days previously. Large macrophages in the Peyer's patch occasionally contain clusters of amphophilic to basophilic intracytoplasmic inclusions (arrow). HE. Bar = 12 µm.
In the small intestine, low to moderate numbers of lymphocytes and macrophages infiltrated the lamina propria in pigs studied after 14 dpi, predominantly in the ileum and duodenum; Brunner's glands were also infiltrated by lymphocytes and macrophages. Necrosis of Brunner's gland acini was present in 5/7 pigs between 21 and 35 dpi. Two pigs (at 21 and 23 dpi) had mild multifocal granulomatous colitis. Scattered individual pancreatic acinar epithelial cells were necrotic in three pigs at 14, 21, and 23 dpi. Kidneys of five pigs studied after 10 dpi had small perivascular infiltrates of lymphocytes and macrophages. Skin lesions were consistent with exudative epidermitis. No lesions were present in adrenal gland, brain, heart, or skeletal muscle.
PRRSV pigs
PRRSV pigs developed interstitial pneumonia consistent with that previously described for experimental PRRSV inoculation. 18,36 Alveolar septa were infiltrated by lymphocytes, plasma cells, and macrophages, type 2 pneumocytes were hypertrophied and moderately increased in number, and alveolar spaces contained small amounts of necrotic debris (Fig. 8). Interstitial pneumonia was first observed at 7 dpi, peaked in severity at 14 dpi, and was nearly resolved by 35 dpi. No unequivocal hepatic lesions or lymphoid depletion were observed in PRRSV-inoculated pigs. Lymphoid tissues were occasionally mildly hyperplastic. Between 7 and 21 dpi, 6/8 pigs had mesocolonic edema ranging from mild to severe. Necrotizing vasculitis was present in the mesocolon of three pigs at 14 and 21 dpi. Lymphocytic myocarditis was present in 7/13 pigs studied between 14 and 35 dpi. Brain tissue from 6/13 pigs had scattered small perivascular infiltrates of lymphocytes at 7, 14, and 21 dpi. No lesions were present in trachea, thymus, kidney, small intestine, skin, skeletal muscle, adrenal gland, stomach, or pancreas.
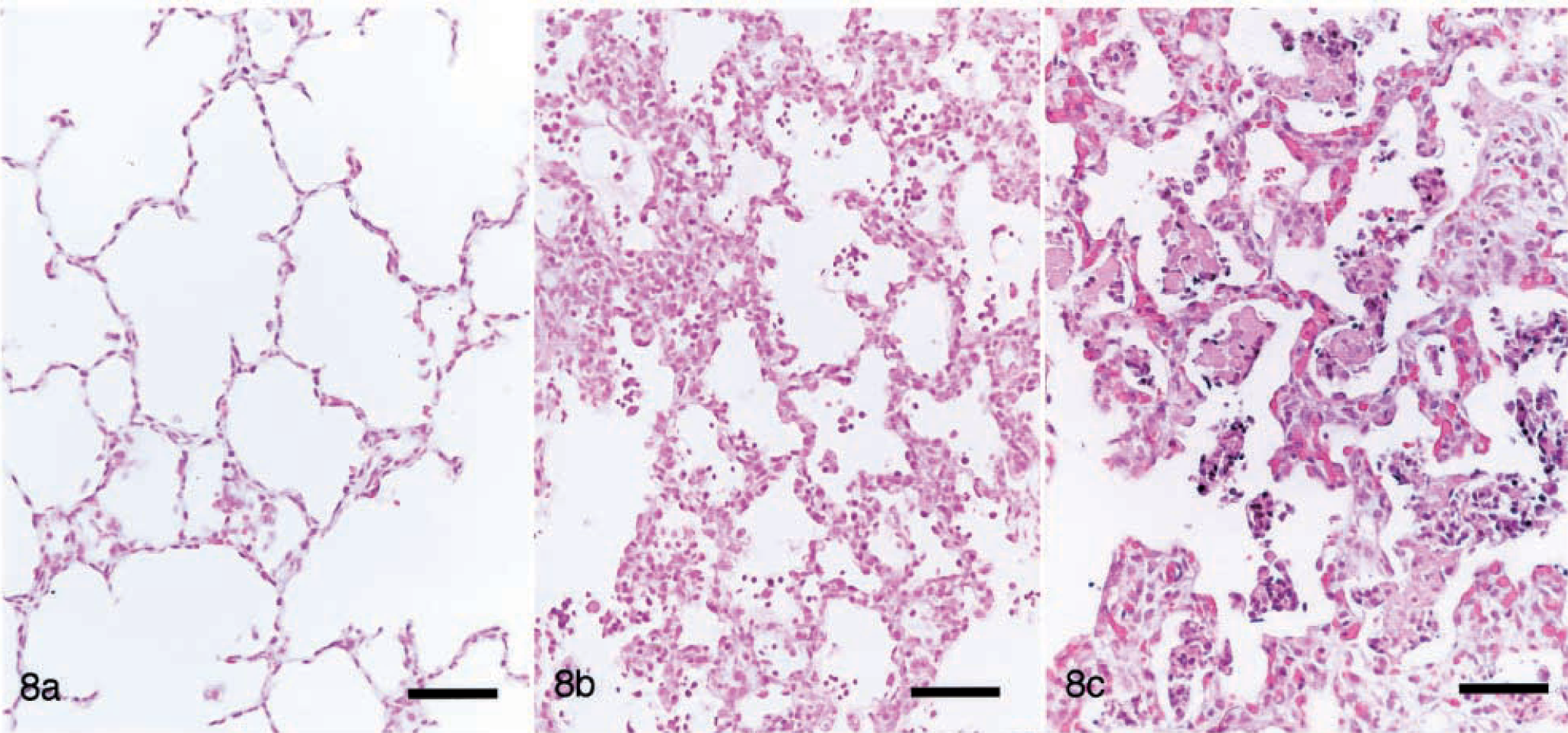
Lung. Fig. 8a Pig inoculated with PCV2 14 days previously. Unremarkable. Fig. 8b Pig inoculated with PRRSV 14 days previously. There is mild thickening of the alveolar septa, and low to moderate numbers of macrophages and cellular debris are within the alveolar spaces. Fig. 8c Pig inoculated with PCV2/PRRSV 17 days previously. Alveolar septa are markedly thickened by infiltrates of mononuclear cells and by type 2 pneumocyte hypertrophy and hyperplasia. Alveolar spaces contain large amounts of cellular debris and macrophages. HE. Bar = 75 µm.
PCV2/PRRSV pigs
Interstitial pneumonia in the PCV2/PRRSV pigs was similar to but more diffuse and severe than that in PRRSV pigs (Fig. 8). Alveolar septa were markedly thickened by infiltrates of macrophages and fewer lymphocytes and plasma cells, many alveolar septa were entirely lined by hypertrophied type 2 pneumocytes, and alveolar spaces contained abundant necrotic debris. Loose peribronchiolar accumulations of macrophages were often larger and more frequent than those in PCV2 pigs, and bronchiolar epithelial lesions were similar to but more severe than those in PCV2 pigs.
Hepatic lesions in the PCV2/PRRSV group were similar to those observed in the PCV2 pigs but were more severe and frequent. Necrotizing hepatitis was present in all 14 pigs studied between 10 and 20 dpi. Lymphoid tissues were depleted in 16/17 PCV2/PRRSV pigs, and depletion was more severe than in the PCV2 group (Fig. 5). Depleted lymphoid follicles often contained macrophages, and intracytoplasmic inclusions were common. Seven of 17 pigs (all between 10 and 20 dpi) had infiltrates of macrophages in the lamina propria of the small intestine, predominantly in the ileum. Mild mesocolonic edema was present in 10/16 pigs between 7 and 17 dpi. Kidneys in 5/10 pigs between 12 and 17 dpi contained scattered small interstitial infiltrates of lymphocytes and macrophages. Mild perivascular cuffing in the brain was present in one pig at 7 dpi and one pig at 10 dpi. There was coagulative necrosis of scattered individual pancreatic acinar epithelial cells in 7/11 pigs studied between 12 and 20 dpi. Skin lesions were limited to small multifocal perivascular accumulations of mononuclear inflammatory cells in one pig at 13 dpi. No lesions were present in the adrenal gland, heart, or skeletal muscle.
Immunohistochemistry for PCV2 antigen
Control pigs
PCV2 antigen was detected in 1/2 pigs at 36 dpi and in two pigs at 49 dpi. Labeling was confined to a few cells in lymph nodes and, in one of the pigs, a few cells in the lamina propria of the duodenum. No PCV2 antigen was detected in control pigs studied at earlier time points.
PCV2 pigs
Overall, PCV2 antigen was detected in at least one tissue of 17/19 PCV2 pigs. PCV2 antigen was most consistently detected in lymph nodes, tonsil, spleen, and Peyer's patches beginning with low numbers of positive cells in 2/3 pigs at 7 dpi. PCV2 antigen in lymphoid tissue was usually intracytoplasmic, in large macrophages within and adjacent to follicles. The number of PCV2-positive cells was greatest between 14 and 28 dpi, when the lymphoid depletion was most severe. The number of PCV2-positive cells was roughly proportional to the degree of lymphoid depletion. A few positive cells were detected in thymus of 7/19 pigs, predominantly at 14–23 dpi. Two of three pigs studied at 36 and 49 dpi had very low numbers of positive cells in the Peyer's patches and in 3/6 and 4/6 of the lymph nodes tested, respectively. PCV2 antigen was not detected in 1/2 pigs at 49 dpi.
PCV2 antigen was detected in 14/19 lungs. Antigen was first detected at 7 dpi within mononuclear cells in alveolar septa and around bronchioles. From 14 to 28 dpi, staining was also present within scattered bronchiolar epithelial cells (Fig. 9). No PCV2 antigen was detected in the lung at 36 or 49 dpi.
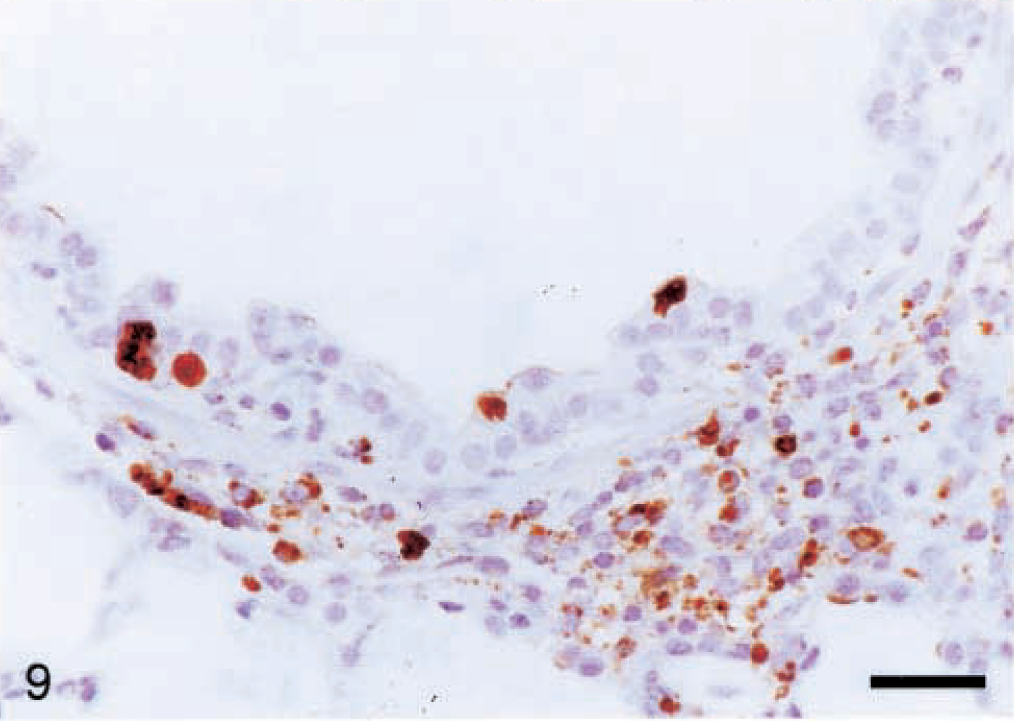
Lung; pig inoculated with PCV2 14 days previously. PCV2 antigen is within scattered bronchiolar epithelial cells and many macrophages in the peribronchiolar cuffs. Immunohistochemistry with anti-PCV polyclonal antibody, counterstained with hematoxylin. Bar = 66 µm.
PCV2 antigen was detected in 14/19 livers of PCV2 pigs. PCV2-positive cells were rare in the liver at 7 dpi. When present, PCV2 antigen was within the nuclei of hepatocytes and occasional Kupffer cells. After 14 dpi, the number of positive cells in the liver was variable but often high. PCV2 antigen was detected within nuclei of viable and necrotic hepatocytes and in the cytoplasm of numerous Kupffer cells and macrophages. PCV2 antigen distribution in severely affected PCV2 pigs was identical to that in severely affected PCV2/PRRSV pigs (Fig. 10). PCV2-positive cells were most numerous in livers with the most severe lesions; in this group, lesions were most severe and the density of PCV2-positive cells was greatest in pigs studied from 23 to 28 dpi. PCV2-positive cells were also present within inflammatory cell aggregates in the lamina propria of the gallbladder in 6/14 pigs studied between 10 and 20 dpi.
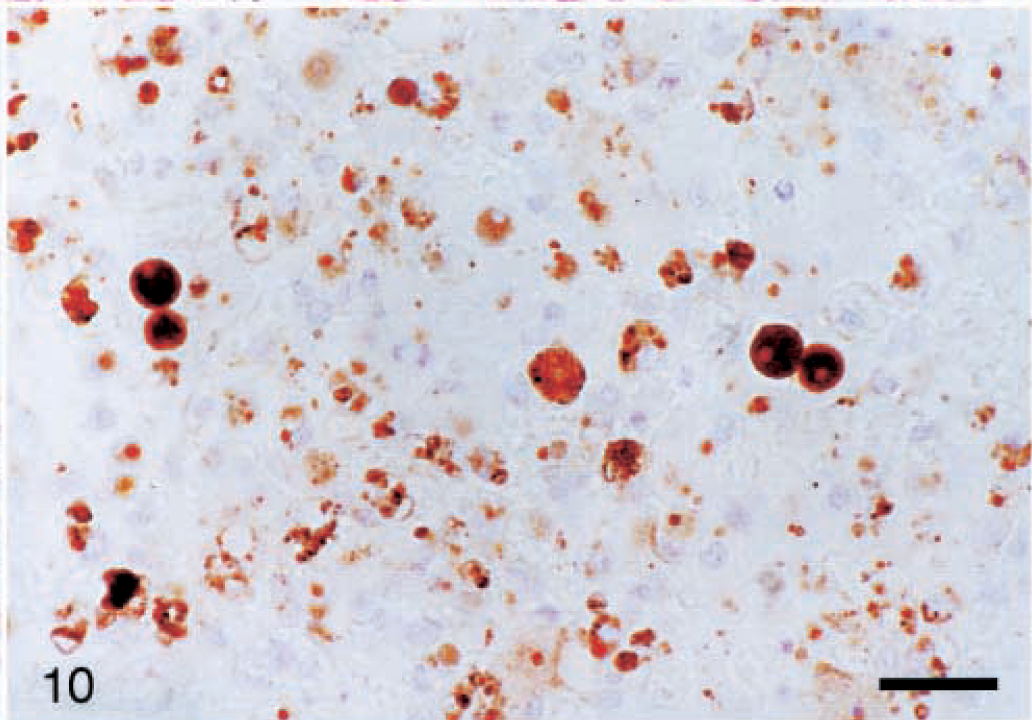
Liver; pig inoculated with PCV2/PRRSV 17 days previously. Large amounts of PCV2 antigen are present within hepatocytes and inflammatory cells. Immunohistochemistry with anti-PCV polyclonal antibody, counterstained with hematoxylin. Bar = 66 µm.
There were numerous PCV2-positive cells in the lamina propria of the small intestine of 15/19 pigs between 7 and 28 dpi, and positive cells were present in the Brunner's glands in 10/19 pigs between 10 and 28 dpi. PCV2 antigen was also detected in kidneys of 13/19 pigs. At 7 and 14 dpi, intracytoplasmic PCV2 antigen was present in a few mononuclear cells in the renal interstitium. Positive cells were most numerous in pigs studied between 21 and 27 dpi. Minimal staining was present in mononuclear cells in the superficial dermis of 2/19 pigs at 14 and 27 dpi. Low numbers of positive cells were present in the lamina propria of the trachea in 8/13 pigs between 10 and 28 dpi. No PCV2-positive cells were identified in brain or skeletal muscle.
PCV2/PRRSV pigs
The distribution and number of PCV2-positive cells were similar to those of the PCV2 group, except that there were higher numbers of PCV2-positive cells in lungs of the PCV2/PRRSV pigs. Positive cells were present in the alveolar septa, in bronchiolar epithelium, and within alveolar spaces. PCV2 antigen was abundant in severely affected livers (Fig. 10).
Immunohistochemistry for PRRSV antigen
PRRSV pigs
Between 7 and 35 dpi, lungs contained low to moderate numbers of PRRSV antigen–positive cells, predominantly within alveolar spaces. At 7 dpi, livers in two pigs contained a few PRRSV-positive Kupffer cells.
PCV2/PRRSV pigs
High numbers of PRRSV-positive cells were present in the lungs through 20 dpi, when the last pig died. Livers with the most severe hepatitis contained occasional PRRSV-positive cells (one or two cells/400× field).
PCR for PCV2
No PCV2 DNA was detected by PCR in serum from pigs sampled 6 days prior to or on the day of inoculation.
Control pigs
PCV2 DNA was detected in the serum of one pig at 21 dpi, at 28 dpi, and at 35 dpi, when it was euthanatized. Two other pigs were positive at 28 dpi and remained positive until they were euthanatized at 49 dpi.
PCV2 pigs
At 7 dpi, PCV2 DNA was detected in 15/19, 17/19, and 18/19 pigs in serum, nasal swabs, and rectal swabs, respectively (Table 1). PCV2 DNA was detected in all but one of the pigs at all subsequent time points. At 28 dpi, no PCV2 DNA was detected in one pig; samples from this pig were positive for PCV2 DNA at time points both prior to and after 28 dpi.
Detection of PCV2 DNA by PCR in PCV2- inoculated pigs.
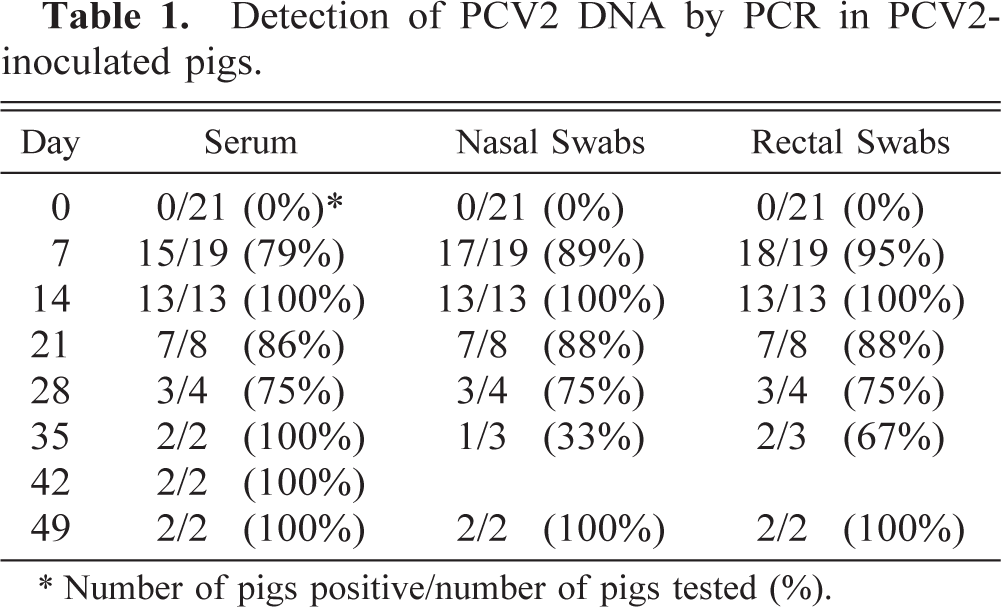
∗ Number of pigs positive/number of pigs tested (%).
Serology
Anti-PCV2 antibodies were first detected in PCV2 pigs at 14 dpi, and titers remained positive (≥1:20) in the pigs surviving to the end of the study at 49 dpi. PRRSV pigs remained negative for anti-PCV2 antibodies throughout the study, and PCV2/PRRSV pigs remained negative through the last necropsy at 20 dpi. Two pigs (also positive by PCR) in the control group became PCV2 seropositive at 49 dpi.
Pigs in the PRRSV and PCV2/PRRSV groups had PRRSV ELISA S/P values of >0.4 (seropositive) as early as 14 dpi. Control and PCV2 pigs remained PRRSV seronegative throughout the study. Hemagglutination inhibition titers to PPV remained <1:32 in all pigs throughout the study.
PCV 35358 genome characterization
The entire genome of PCV2 strain 35358 was amplified by PCR from the initial inoculum and then sequenced. The overall size of the genome was 1,768 nucleotides, similar to that of other published PCV2 strains. 14,20,27,29,30 Overall nucleotide homology between PCV2 35358 and the ISU31 strain 29 (from a field case of PMWS) was 99.5%, with only eight nucleotide differences: three nucleotide substitutions were in the ORF1 region, four in the ORF2 region, and one in the noncoding region between ORF1 and ORF2. On the amino acid level, there was 100% amino acid identity of the ORF1 predicted protein and only one amino acid difference in the sequence of the ORF2 predicted protein.
Because PCV2 DNA was detected in the sham-inoculated control group as early as 21 dpi, the PCV2 ORF2 amplicons from the serum of pig Nos. 1 and 11 (control group) and pig Nos. 19 and 33 (PCV2 group) were sequenced. Sequence comparison revealed 100% nucleotide identity of the ORF2 amplicons and 100% nucleotide identity with the ORF2 sequence amplified from the initial PCV2 35358 inoculum.
Discussion
Because two preliminary studies indicated that both PCV2 and PRRSV were present in the livers of sows with PRRS-associated hepatitis, the present study was conducted to evaluate the independent and combined effects of the PRRSV and PCV2 isolates. Hepatitis was only induced in the PCV2 or PCV2/PRRSV pigs and varied from mild to severe; severe cases were associated with hyperbilirubinemia. Livers in the control and PRRSV pigs were indistinguishable. We therefore conclude that the hepatitis previously observed in sows with “atypical” PRRS likely resulted from concurrent PCV2 infection. Several other researchers have associated hepatitis with PMWS and PCV2 infection, 2,24,35 and fulminant hepatitis and icterus are occasional features of clinical PMWS. 21,35
In addition to hepatitis and the associated icterus, PCV2-infected pigs developed lymphoid depletion and lymphohistiocytic to granulomatous inflammation in lung, liver, lymph nodes, spleen, kidney, ileum, jejunum, duodenum, and colon. These lesions are characteristic of PMWS. 26,27,29,35 The induction of PMWS with PCV2 alone confirms that PCV2 is pathogenic in CD/CD pigs and corroborates the findings of other investigators, 2,24 who have also demonstrated that pigs infected with both PCV2 and PPV develop more severe clinical disease and lesions than do pigs inoculated with PCV2. 2,24 Concurrent PCV2/PPV infection has been reported in field cases of PMWS in western Canada. 11 The original source inoculum and cell cultures for this experiment were negative for PPV by PCR, and none of the pigs in any group seroconverted to PPV. We are therefore confident that lesions identified in PCV2-inoculated pigs did not result from PPV coinfection.
Lymphoid depletion was a consistent finding in both the PCV2 and PCV2/PRRSV pigs and was often moderate to severe in nature. Lymphoid depletion is a subjective evaluation, and a mildly depleted lymph node may be indistinguishable from an inactive lymph node. Grossly enlarged lymph nodes were not present at necropsy in either group receiving PCV2 inoculum but were present in the PRRSV group. Enlarged lymph nodes observed in field cases of PMWS may be the result of a subsequent coinfection or a feature of a later stage of the disease.
PCV2/PRRSV induced severe clinical disease, high mortality, and lesions consistent with PMWS, including severe interstitial pneumonia and lymphoid depletion. PCV2 pigs developed lymphoid depletion, mild necrotizing bronchiolitis, and sporadic necrotizing hepatitis but did not develop the severe interstitial pneumonia often associated with field cases of PMWS 26,35 or induced by PCV2/PRRSV. Despite speculation that many of the lesions attributed to PRRSV may be caused by concurrent PCV2 infection, 12 the PRRSV pigs in this study developed moderate interstitial pneumonia, mild lymphocytic myocarditis, and other lesions consistent with previous experimental inoculation studies 17,37,42 and failed to seroconvert to PCV2. In the present study, the often severe interstitial pneumonia attributed to PCV2 required concurrent PRRSV infection.
PCV2/PRRSV induced an interstitial pneumonia with necrotizing bronchiolitis. This combination was not seen in the PRRSV or PCV2 groups, which had milder interstitial pneumonia or necrotizing bronchiolitis, respectively. Necrotizing bronchiolitis has not been previously reported in PCV2-inoculated pigs. PCV2 pigs exhibited only mild clinical respiratory disease that was significantly less severe than that in the PRRSV and PCV2/PRRSV groups. Chronic obliterative bronchitis and bronchitis have been reported in field cases 21 of PMWS and may represent a sequela of PCV2 infection. Necrotizing bronchitis and bronchiolitis are readily induced by SIV, 10 a very common pathogen of North American swine. Because previous SIV infection is difficult to rule out in clinical cases, a potential role for SIV in PMWS-associated obliterative airway lesions cannot be excluded.
The PCV2/PRRSV group also had more severe lymphoid depletion, hepatocellular necrosis, and granulomatous inflammation in lung, liver, lymph nodes, spleen, skin, kidney, stomach, ileum, jejunum, duodenum, and colon than did the PCV2 pigs. These findings are consistent with potentiation of PCV2-induced disease by PRRSV. Because both PRRSV and PPV may potentiate PCV2 infection, other infectious agents may have similar effects. In contrast to the PCV2/PPV model, the most striking and consistent lesion of the PCV2/PRRSV model was interstitial pneumonia. This interstitial pneumonia is compatible with the lesions seen in field cases of PMWS at our diagnostic laboratory.
Seroconversion of pigs to PCV2 was detected at 14 and 21 dpi in the PCV2 pigs, but the PCV2/PRRSV pigs did not develop antibodies to PCV2, probably because of the debilitated state of these pigs and the fact that all were dead by 20 dpi. The PCV2/PRRSV pigs did develop antibodies to PRRSV by 14 dpi. Production of antibodies to PRRSV may occur sooner postinoculation, allowing for seroconversion prior to the onset of debilitating disease. PCV2 DNA was detected in nasal and rectal swabs, urine, and serum in the PCV2 pigs beginning at 7 dpi and continuing through 49 dpi, suggesting that the virus is shed by several routes and for an extended period of time.
Regrettably, three pigs in the sham-inoculated control group were infected with PCV2 during the study. Although the exact time of infection is unknown, the controls probably did not become infected until after 14 dpi, and infection probably was due to cross-contamination with PCV2 from the PCV2 group. PCR did not detect PCV2 DNA in any pig in any experimental group at 0 or 6 dpi. At 7 dpi, PCV2 DNA could be detected consistently in the serum of PCV2 pigs but was not detected until 21 or 28 dpi in the three control pigs. Although the PCV2 pigs seroconverted at 14 and 21 dpi, the controls did not seroconvert until 49 dpi. Nucleotide sequences of the PCV2 ORF2 amplicons from the PCV2 and control pigs were identical, further indicating that the control pigs were exposed by lateral transmission of the same virus and were not infected by a different virus through the environment, vertical transmission, or other introduction. The pigs were housed on raised decks located in separate but adjacent isolation rooms with negative pressure ventilation. Workers were required to have no other pig contact for 12 hours and to shower and change clothes prior to entry into the isolation rooms. At entry, personnel were required to wear coveralls, hairnet, face mask, gloves, and disposable boots and to use a footbath. People flow was unidirectional from noninoculated rooms to inoculated rooms, and separate equipment was supplied to each room. Cross-infection, even with these biosecurity measures, suggests that PCV2 may be easily spread from pig to pig by fomites or airborne means.
Exudative epidermitis developed in both the PCV2 group and the control group. The disease was severe in several of the animals in the PCV2 group, resulting in the death of at least one animal (15 dpi). The control pigs responded to antibiotic therapy, whereas the PCV2 pigs did not. The two control pigs that seroconverted at 49 dpi required treatment for exudative epidermitis during the last week of the experiment, suggesting an immunosuppressive role for PCV2. 7,12,24
Lesions induced by PCV2/PRRSV coinfection in this study are consistent with those seen in field cases of PMWS. 26,27,29,35 CD/CD pigs are more susceptible to many infectious agents because of their lack of maternal antibodies. Limited studies have shown that maternal anti-PCV2 antibody drops to undetectable levels at approximately 8–10 weeks of age. 9 PCV2 infection at 8–10 weeks of age with the development of PMWS 2–3 weeks later is consistent with the age at which most field cases of PMWS are diagnosed in our diagnostic laboratory. Seroconversion to many other common potential pathogens, including PPV, PRRSV, SIV, and Mycoplasma hyopnemoniae, commonly occurs near this same time. 22,31,44 The timing of exposure and decay of maternal antibody to PCV2 and other pathogens may play a critical role in determining whether PCV2 infection induces PMWS or remains subclinical.
Footnotes
Acknowledgements
We thank D. Oedekoven, K. Harmon, J. Fosse, and the Iowa State University Veterinary Diagnostic Laboratory serology section for their technical assistance. This research was funded in part by grants from the National Pork Producers Council and the Iowa Livestock Health Advisory Council.
