Abstract
Mycobacterium fortuitum is a saprophytic, fast-growing, nontuberculous, and nonlepromatous mycobacterium that can cause infections in animals and humans. In dogs and cats, it is one of the most common agents of ulcerative dermatitides and panniculitides caused by atypical mycobacteria. In humans, it is frequently found in lipoid pneumonias or contaminated surgical sites. We report a cat with granulomatous pneumonia caused by M fortuitum resembling lipoid pneumonia in humans. The similarity between the histopathology of the lung and skin lesions caused by this organism in dogs and cats is emphasized. We discuss the role of lipids in the pathogenesis of mycobacterioses and suggest an association between atypical mycobacteria and lipid-rich environments. We conclude that M fortuitum should be included as a differential in cases of lipid-rich pneumonias that do not respond to common antibiotics.
Mycobacterium fortuitum is classified among the nontuberculous, nonlepromatous, fast-growing mycobacteria from the Runyon group IV and is one of the causative agents of “atypical mycobacterioses.” Other members of this group include Mycobacterium chelonae, Mycobacterium abscessus, and Mycobacterium smegmatis. M fortuitum is a saprophytic, ubiquitous organism found in soil, water (including potable water), and wet substrates. It can be cultured from houseflies, sawdust bedding in pig farms, and unpasteurized milk. M fortuitum is frequently isolated from reptiles, amphibians, invertebrates, and a variety of nondomestic species such as wild boars, swamp buffalos, seals, and armadillos. 3
Atypical mycobacteriosis in domestic animals usually presents in the form of an ulcerative, nonhealing, pyogranulomatous dermatitis and panniculitis in cats and rarely in dogs, following bite wounds. M fortuitum and M chelonae are the most commonly isolated organisms in such cases. 14 In this syndrome, macrophages and neutrophils often surround clear vacuoles that are thought to represent remaining lipid secondary to damage of the panniculus adiposus of the skin. The organisms are most easily identified inside these vacuoles and occasionally in the cytoplasm of macrophages using a modified Fite-Faraco acid-fast stain. 8 Classically, the presence of the vacuoles in the center of pyogranulomas in dermal lesions is one of the features suggestive of atypical mycobacteriosis, which prompts the pathologist to evaluate and acid-fast stain in order to identify the mycobacteria.
In humans, M fortuitum is found in contaminated surgical sites and rarely as the cause of disseminated disease in immunosuppressed individuals. 3 There are also numerous reports in the literature of M fortuitum as a complicating factor of lipoid pneumonia in children and adults. 1, 2, 10, 15, 22 Pneumonia or pulmonary granulomas caused by M fortuitum have been reported in dogs and in Florida manatees, but never in cats. Furthermore, descriptions of the histopathologic features and discussions of the pathogenesis of the pulmonary lesions in animals are scant in the currently available literature. 13, 20, 21
A 4-year-old, indoor-outdoor, male castrated domestic short-haired cat (Felis catus) presented to the Animal Medical Center with a 3-week history of increased respiratory effort and dyspnea. He had responded well to antibiotics (enrofloxacin, Baytril) when treated for a similar episode of dyspnea a few months previously. The cat also had a history of atresia anus, which had been surgically corrected when he was 1 year old. On clinical examination, the cat was emaciated and had severe periodontal disease. The owner revealed that the cat had been on 2.5 ml of oral lactulose (Belmalax) daily to treat chronic constipation due to atresia anus and due to a prolonged history of poor appetite and weight loss. Thoracic radiographs showed severe diffuse alveolar infiltrates that obscured the cardiac silhouette. Ancillary test results revealed a high white blood cell count (18,300 cells/ml) with mature neutrophilia (14,091 cells/ml) and monocytosis (2,196 cells/ml) and normal chemistry values, except for hypoglycemia (50 mg/dl). Serology tests for retroviruses (feline immunodeficiency and feline leukemia) were negative, and a high Toxoplasma gondii immunoglobulin M titer (1 : 256) was reported. Aerobic culture of a lung aspirate was negative, and cytologic evaluation showed pyogranulomatous inflammation without evidence of organisms. Tentative treatment with ampicillin, enrofloxacin (Baytril), and clindamycin was started without response. The cat was later euthanized due to poor prognosis.
On necropsy, the cat was emaciated and had severe periodontal disease with almost complete absence of all teeth. The lungs were diffusely consolidated, failed to collapse, were tan to light pink, and oozed tan, creamy material upon compression of sectioned parenchyma. The lumina of large bronchi and trachea appeared clean (Fig. 1). The tracheobronchial lymph nodes were soft and enlarged. Colonic dilation and fecal impaction were also noted, as well as a faint perianal scar and slightly abnormal anal conformation.
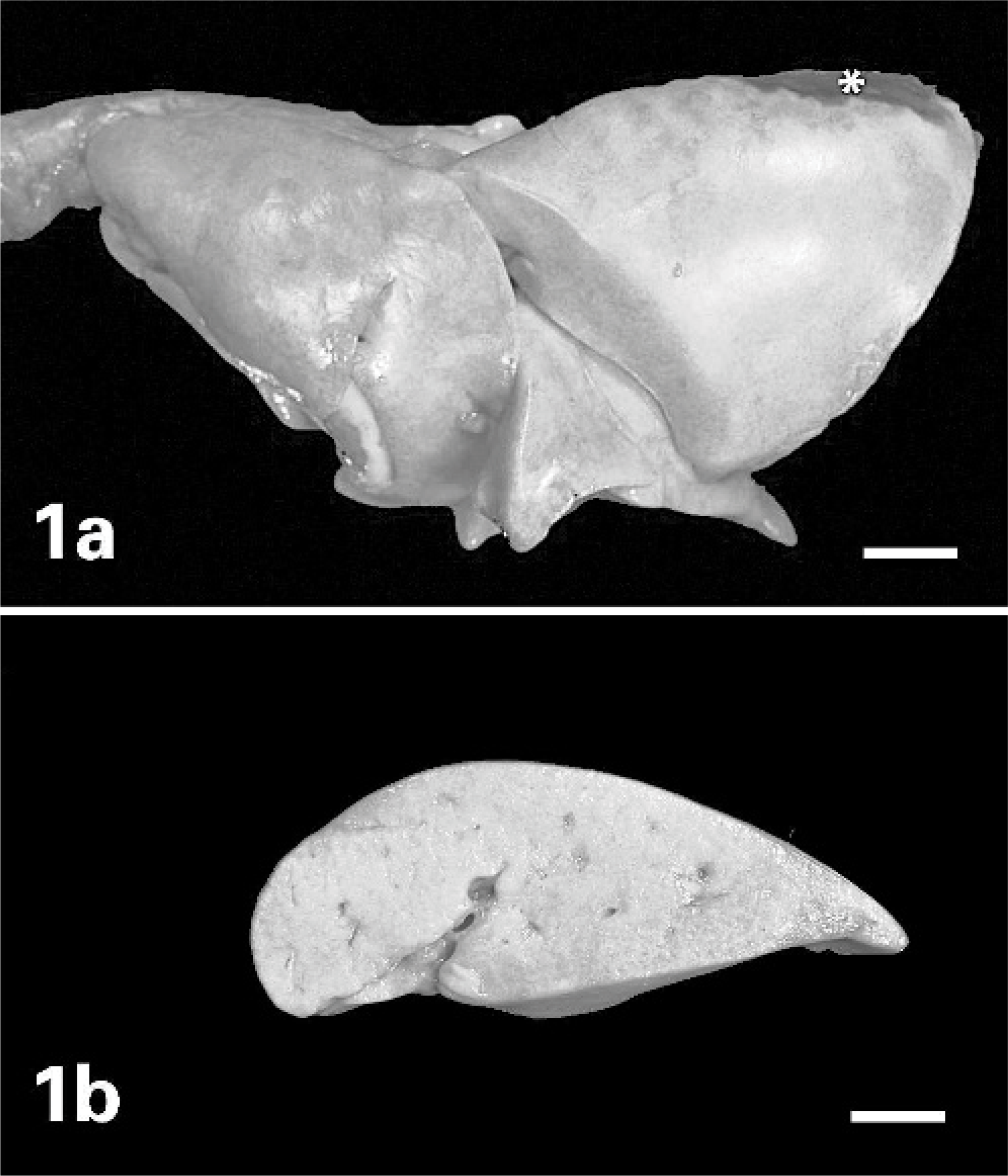
Lung; cat.
Histologically, there was a severe and diffuse pyogranulomatous bronchopneumonia, characterized by numerous macrophages and neutrophils, and rare lymphocytes and plasma cells filling the alveolar spaces. The macrophages often contained variably sized lipid droplets and a small amount of eosinophilic, granular-to-glassy, proteinaceous material in their cytoplasm (Fig. 2). The alveolar inflammatory infiltrate often surrounded a central, round vacuole with sharp borders. Wispy, delicate, filamentous eosinophilic material was noted within some of these vacuoles. The epithelium of many bronchioli was sloughed, degenerate, or necrotic. Pink amorphous-to-fibrillar material admixed with inflammatory cells and cellular debris was within the bronchiolar lumina. There was also mild type II pneumocyte hyperplasia throughout the lungs. Special stains (Brown-Brenn and Fite-Faraco) showed Gram-positive and acid-fast–positive bacilli in the center of the vacuoles and within some of the macrophages (Fig. 3). M fortuitum was cultured from frozen lung samples at the Mycobacterial Laboratory of the New York City Health Department.
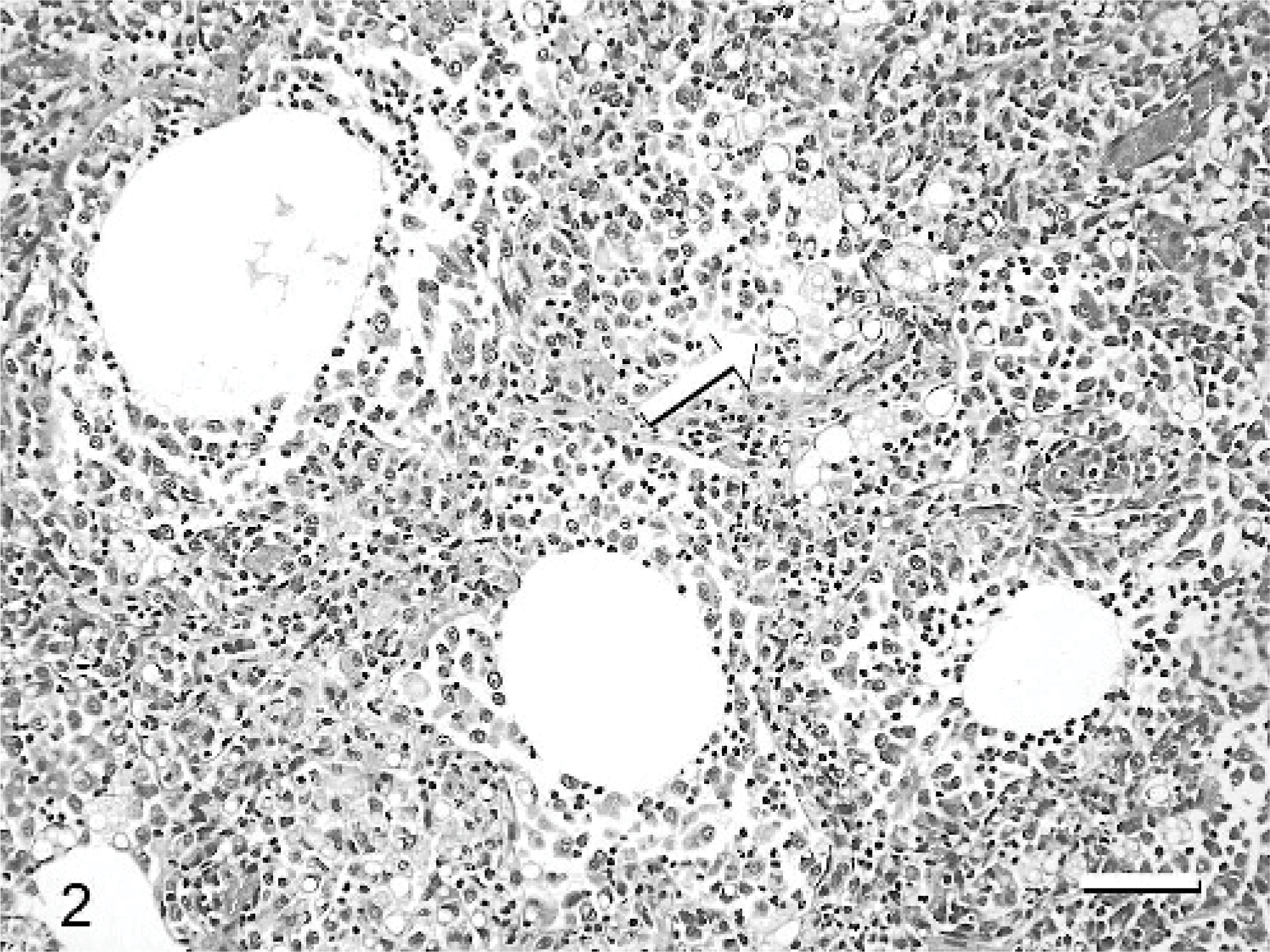
Lung; cat. Diffuse granulomatous inflammation effaces the pulmonary parenchyma. Note large round vacuoles with wispy material and sharply defined lipid droplets in the cytoplasm of macrophages (arrow). HE. Bar = 120 μm.
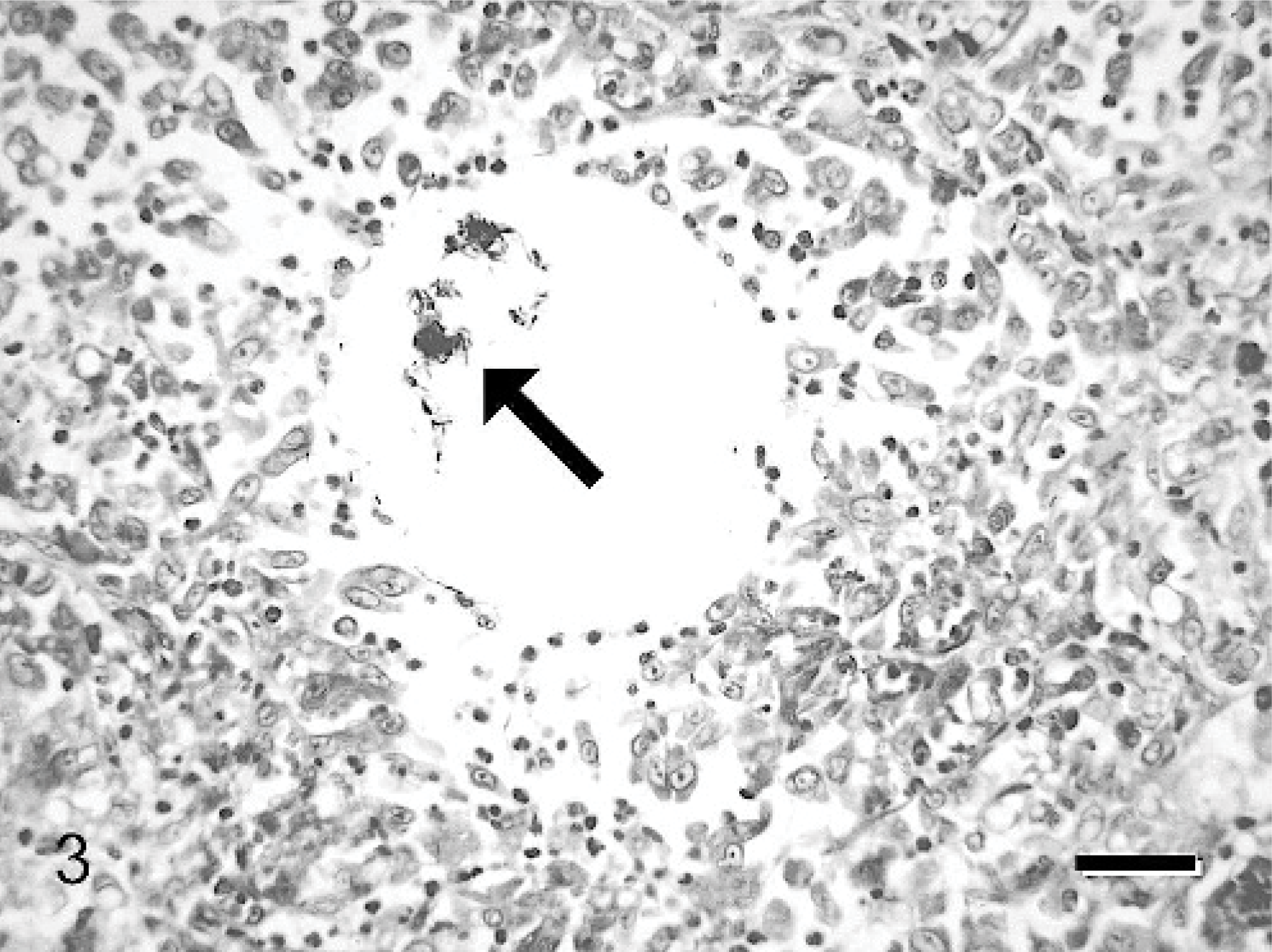
Lung; cat. Acid-fast–positive mycobacteria are noted in the lumen of a lipid vacuole within the pulmonary parenchyma. Fite-Faraco modified acid-fast. Bar = 80 μm.
There were many unusual features in this lesion, including the diffuse involvement of all lung lobes, the abundant macrophages containing lipid within alveoli and in bronchioli, and the presence of free, extracellular mycobacteria inside lipid vacuoles. The latter is a location identical to that described for granulomatous panniculitides caused by the M fortuitum-chelonae complex in cats. The striking difference is, however, the obvious lack of naturally occurring adipose tissue in the lung to explain the origin of the vacuoles. Aspiration was considered a likely source of lipid and was substantiated by the history of oral lactulose administration, which was probably mixed with milk in this case.
Interestingly, many of the human M fortuitum infections occur in children as a complication of pneumonia caused by aspiration of oily substances such as ghee (clarified butter) and in adults that aspirate lipid substances such as mineral oil (used in chronic cases of constipation). 1, 2, 15 In addition to cases of aspiration, M fortuitum has been identified as the cause of pneumonia, due to embolization of bone wax (a ceroid, lipid-based substance) from sternotomy incisions and of sternal osteomyelitis, where bone wax is used as a homeostatic sealant. The incidence of such infections was sufficiently high to grant the discontinuation of the use of this sealant in surgical procedures in humans around 1976. Embolism of this lipoid material was proved to be the vehicle of infection when radioactively labeled bone wax applied to sternotomy sites could be later found in the bone marrow and lungs of experimental animals. 18, 19 Furthermore, in the only detailed microscopic description of M fortuitum pneumonia in a dog, organisms were reported within lipid vacuoles similar to those described in this cat's lung lesions and in feline panniculitis caused by the same organism. 21
It is apparent from the previously described lesions that lipid plays an important role in the pathogenesis of atypical mycobacterioses. While lipids act as a mechanical protection for the mycobacteria and favor tissue necrosis facilitating secondary infection, the most important role of lipids in lesion development is probably related to the biochemistry of the mycobacterial cell wall. 11, 12, 15, 16 Mycolic acids that compose the mycobacterial cell wall are glycolipids thought to regulate the intracellular (intramacrophage) growth rate of mycobacteria, as well as the host immune response. 17 Among the many known mycolates is trehalose 6,6′-dimycolate (TDM), which is abundant in virulent strains of Mycobacterium tuberculosis, but also found in many saprophytic mycobacteria including M fortuitum. 7 It has been recently shown that TDM induces caseating granulomas in mice only when inoculated in the presence of lipid, which indicates that its cytotoxic effects are activated by oily substances. 11 In experimental infection of mice with M tuberculosis, abundant lipid accumulates in alveolar spaces and in foamy alveolar macrophages in early infection, similar to what was seen in this cat. Lipid accumulation is also seen in chronic human pulmonary tuberculosis, in the absence of aspiration. 11 In these cases, alveolar surfactant is likely the source of lipid since it consists mainly of disaturated phospholipids. 5 Aside from lipids, surfactant also contains proteins that can mediate the interaction between mycobacteria and alveolar macrophages. 6, 9, 11 Conversely, mycolic acids, and especially TDM, can modulate and inhibit the lipid fraction of surfactants, leading to low surface tension and alveolar collapse, which contribute to lesion development. 4 These data underscore the importance of both surfactant and mycolic acids in pulmonary mycobacterioses.
M fortuitum contains TDM in its wall, as well as other toxic glycolipids such as 2,3-diacyl trehalose. It is possible that lipid present at the site of infection activates these virulent mycolic acids, eliciting granulomatous inflammation in M fortuitum infection. The diffuse nature of the lesion may be explained by the proportion of virulent and nonvirulent mycolates within the cell wall of M fortuitum, since the relative composition of the cell wall greatly influences the host response to different species of mycobacteria. 17 In summary, lipid-rich environments seem to be ideal for all mycobacteria and essential in infections caused by less pathogenic, saprophytic organisms such as M fortuitum.
On the basis of the lesions seen in this cat and of previous reports of human and canine pulmonary cases, as well as feline mycobacterial panniculitides, we suggest that lipid-rich environments are required by M fortuitum. The source of lipids in the lung remains unclear, although aspirated oily substances, surfactant, and lipids released from the mycobacterial cell wall may play a key role in pathogenesis. We conclude that infection with mycobacteria of the M fortuitum-chelonae complex should, therefore, be considered in pneumonias associated with lipids that do not respond to the usual antibiotic therapies.
Footnotes
Acknowledgements
We thank Dr. Andrew Loar for cytologic evaluation of lung aspirates, as well as the staff of the Animal Medical Center for their help in multiple aspects of the management of this case. We also thank Sunny Yeung at Memorial Sloan-Kettering Cancer Center for assistance with digital images and the Mycobacterial Laboratory of the New York City Health Department for the culture of the organism.
