Abstract
A 4-year-old male ferret (Mustela putorius furo) had a 6-month history of weight loss and gradual development of depression and coughing. Necropsy findings included pale gray tissue around the distal trachea, multiple nodules in the lungs, a single nodule in the stomach wall, gray foci in the liver, and enlarged lymph nodes. Histologic examination revealed multifocal to coalescing granulomatous inflammation in the trachea, lungs, stomach, liver, and lymph nodes, with acid-fast bacteria in epithelioid cells and macrophages. The acidfast bacteria were identified as Mycobacterium celatum (type 3) using DNA sequence analysis of the 16S ribosomal DNA gene. M. celatum is a recently described mycobacterium isolated mainly from immunocompromised humans. This is the first report of M. celatum infection in an animal.
Mycobacterium celatum, which was described for the first time in 1993, is a slowly growing, potentially pathogenic mycobacterium. The organism has been isolated from human patients. The new species is divided into three groups (types 1, 2, and 3) based on genomic sequencing. 2,4 M. celatum is mainly isolated from immunosuppressed patients, and the most common lesions associated with this novel mycobacterium are pneumonia and disseminated infection. 10,16 The bacterium has also been isolated from immunocompetent patients, where it caused cervical lymphadenitis in a child and fatal pneumonia in a 73-year-old woman. 5,6 Ferrets are susceptible to infection with various mycobacteria, and Mycobacterium bovis, Mycobacterium avium, and Mycobacterium microti have been isolated from diseased ferrets. 11,14 Domestic ferrets (Mustela putorius furo) kept as pets live close to their owners, and thus there is a possibility for transmission of infectious agents between humans and ferrets. We report here a case of M. celatum infection in a ferret in Norway. Infection with M. celatum in an animal has not previously been described.
A 4-year-old male ferret was presented to the Department of Small Animal Clinical Sciences, Norwegian School of Veterinary Science, in March 1999, with a history of weight loss and coughing. The animal had been depressed for at least 6 months, with a gradual decline in clinical condition; the cough had persisted for approximately 3 months.
Dyspnea, dehydration, depression, emaciation, and poor coat quality were noted during clinical examination. Auscultation revealed harsh, dry lung sounds. A radiograph of the thorax (Fig. 1) showed multiple nodular and peribronchial densities disseminated throughout both lungs. In addition, a cystic structure was visualized in the lung parenchyma. Because the prognosis was poor, the animal was euthanatized.
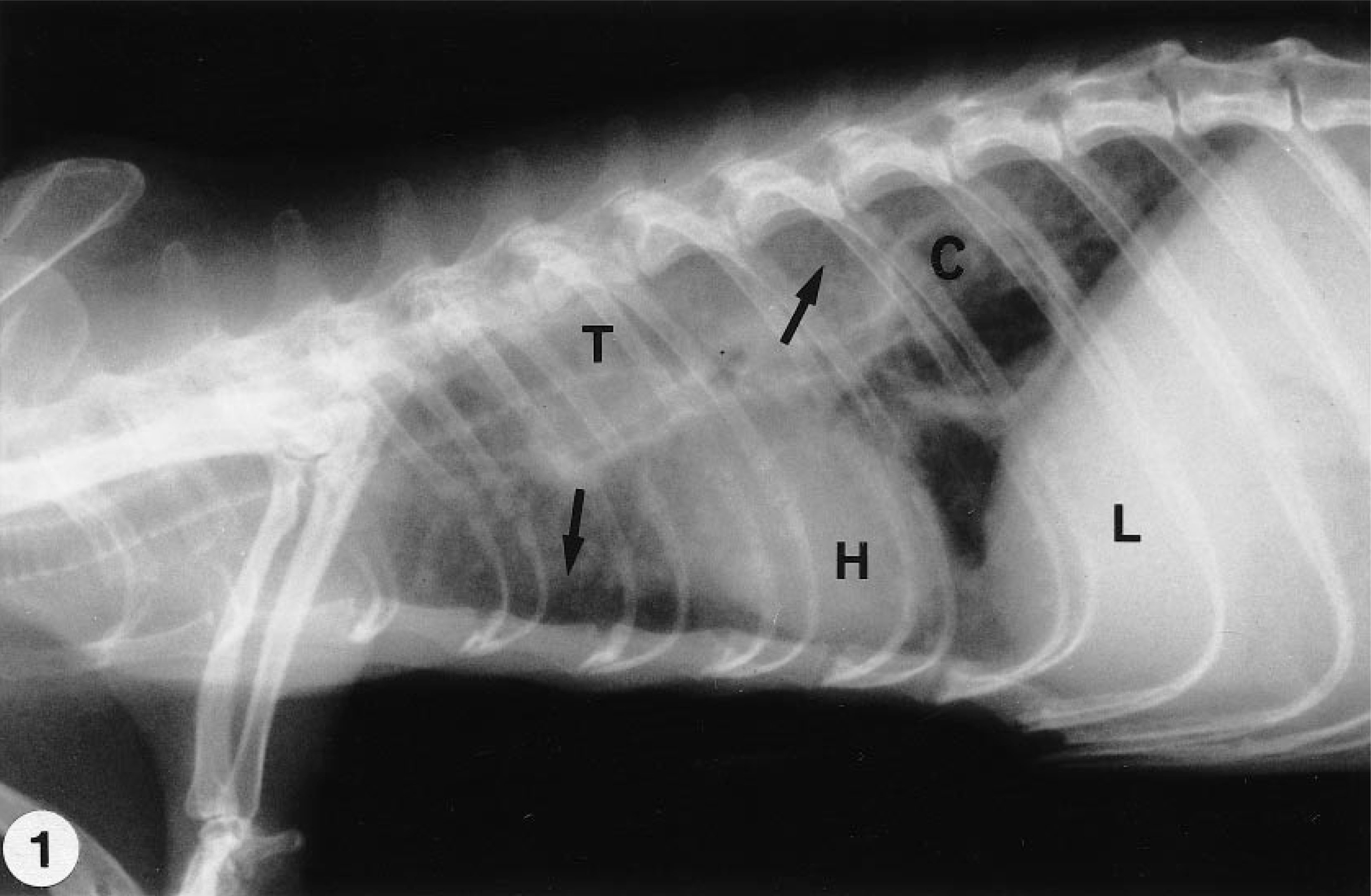
Thorax; ferret. Radiograph reveals multiple nodular and peribronchial densities (arrows) and a cystic structure (C) in the lungs. T = trachea; H = heart; L = liver.
At necropsy (Department of Morphology, Genetics, and Aquatic Biology, Norwegian School of Veterinary Science), the ferret appeared in poor body condition. The lungs had multiple firm, pale gray nodules, 2–10 mm in diameter, randomly distributed throughout the lung parenchyma. Nodular tissue was also found around the distal trachea. An area about 15 × 15 mm in the stomach wall was thickened, and the serosa was roughened. The cut surface showed pale gray tissue. There were multiple 1–2-mm gray foci in the liver parenchyma. All lymph nodes were moderately to severely enlarged. The spleen was pale but of normal size. Samples from the lung, trachea, heart, liver, kidney, stomach, and retropharyngeal and gastric lymph nodes were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Tissues were sectioned at 3–4 µm and stained with hematoxylin and eosin (HE), Gram's stain, periodic acid–Schiff (PAS) reaction, and Ziehl-Neelsen stain (ZN).
Histopathologic examination of the lungs showed multiple inflammatory foci (Fig. 2) containing epithelioid cells with abundant eosinophilic cytoplasm, macrophages, neutrophils, and lymphocytes (Fig. 3). Multinucleated giant cells were not observed. Necrotic areas were present in some of the granulomatous lesions. Similar granulomatous inflammation was identified in the mucosa, submucosa, and adventitia of the trachea. In the stomach wall, large areas of granulomatous inflammation were present in the lamina propria, submucosa, muscle wall, and serosa. The liver had small scattered foci of macrophages and neutrophils in the parenchyma, and a moderate lymphoplasmacytic infiltrate with some macrophages was present in periportal areas. Macroscopically visible gray foci were areas with vacuolated hepatocytes that most likely contained lipid. The architecture of the lymph nodes was distorted by multiple large foci of granulomatous inflammation in the cortex and medulla.
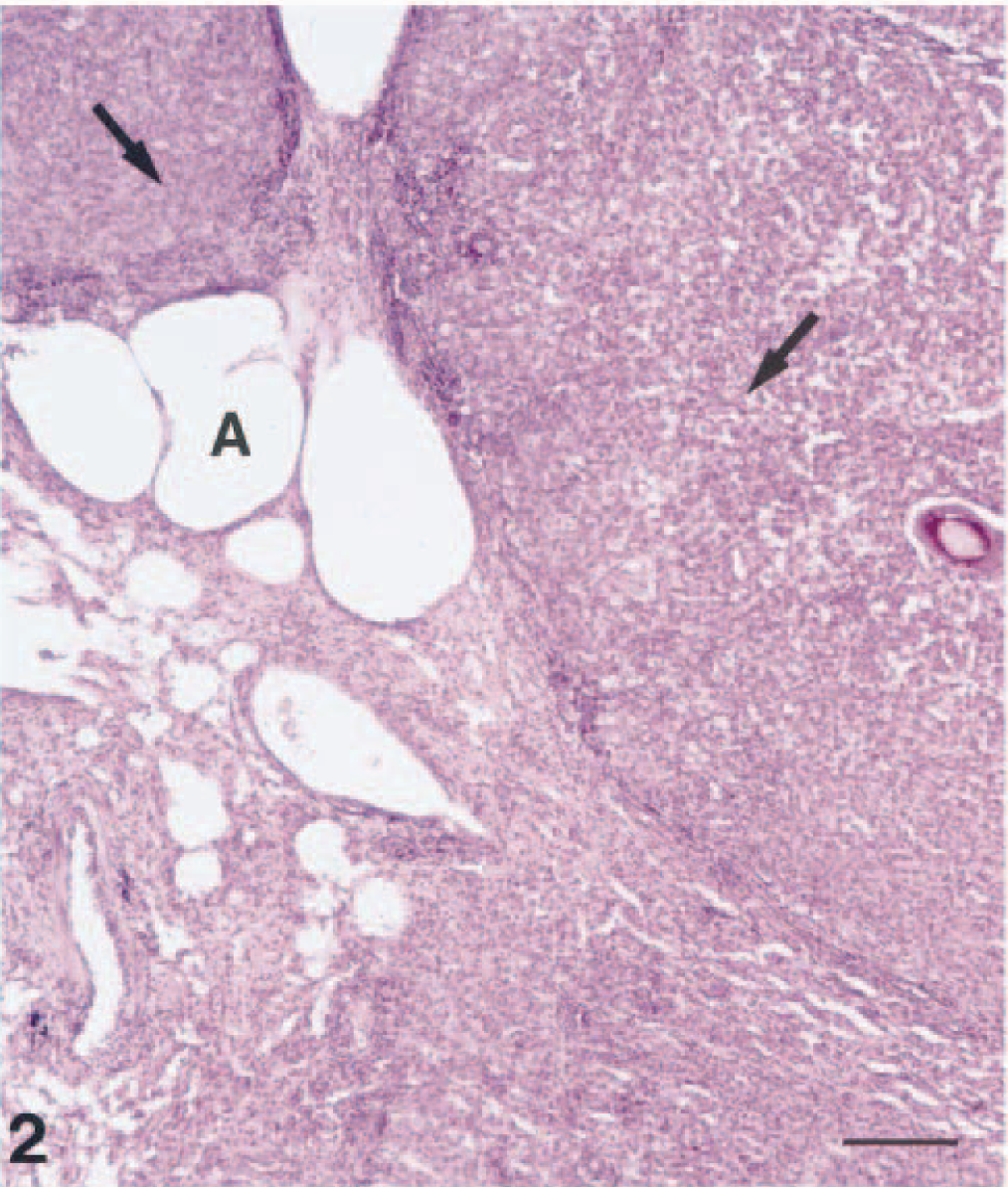
Lung; ferret. Multiple foci with granulomatous inflammation (arrows) were observed in the lung parenchyma. A = alveolar lumen. HE. Bar = 120 µm.
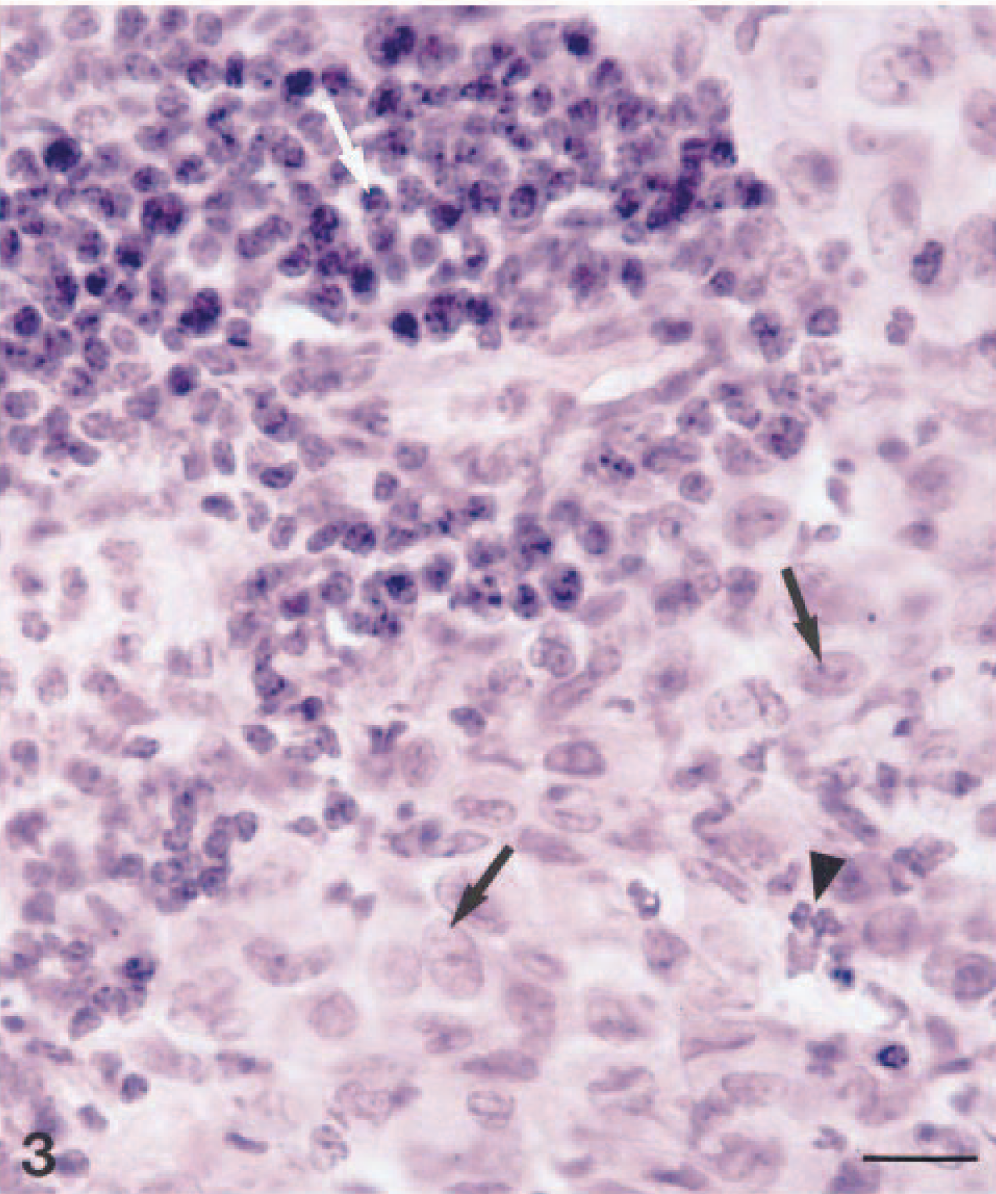
Lung; ferret. Granulomatous inflammation containing epitheliod cells with abundant cytoplasm (arrows), lymphocytes (white arrow), and neutrophils (arrowhead). HE. Bar = 15 µm.
Delicate, filamentous, sometimes branching acid-fast bacteria were observed in granulomatous lesions in the trachea, lung, stomach, liver, and retropharyngeal and gastric lymph nodes (Fig. 4). Bacteria were located in the cytoplasm of epithelioid cells and macrophages. Macrophages in the mucosa of the stomach and trachea contained large numbers of bacteria, but few to moderate numbers of bacteria were observed in lesions in other tissues. The bacteria did not stain with Gram's stain and PAS methods.
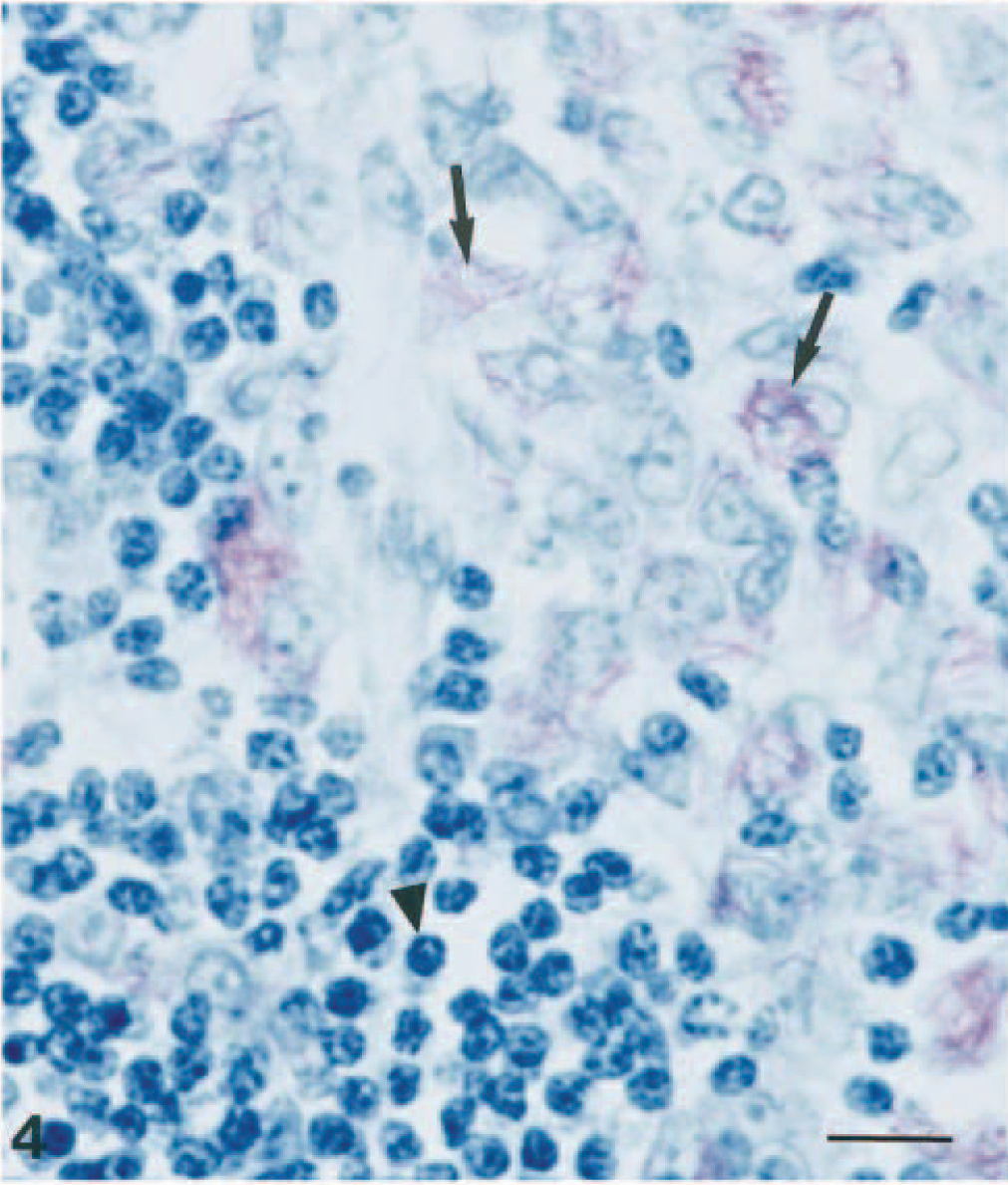
Stomach; ferret. A large number of acid-fast bacteria in macrophages (arrows) and aggregates of lymphocytes (arrowhead) were observed in the lamina propria. ZN. Bar = 10 µm.
Immunohistochemical analysis for the presence of mycobacteria was performed on paraffin-embedded sections of the stomach. Rabbit polyclonal antibodies against Mycobacterium avium subsp. paratuberculosis (DAKO, Glostrup, Denmark) and rabbit polyclonal antibodies against M. bovis (Bacille Calmette-Guérin) (DAKO) were used as primary antibodies in a streptavidin–biotin–alkaline phosphatase method. 15 There was staining with the polyclonal antibodies against M. avium subsp. paratuberculosis and M. bovis (Fig. 5).
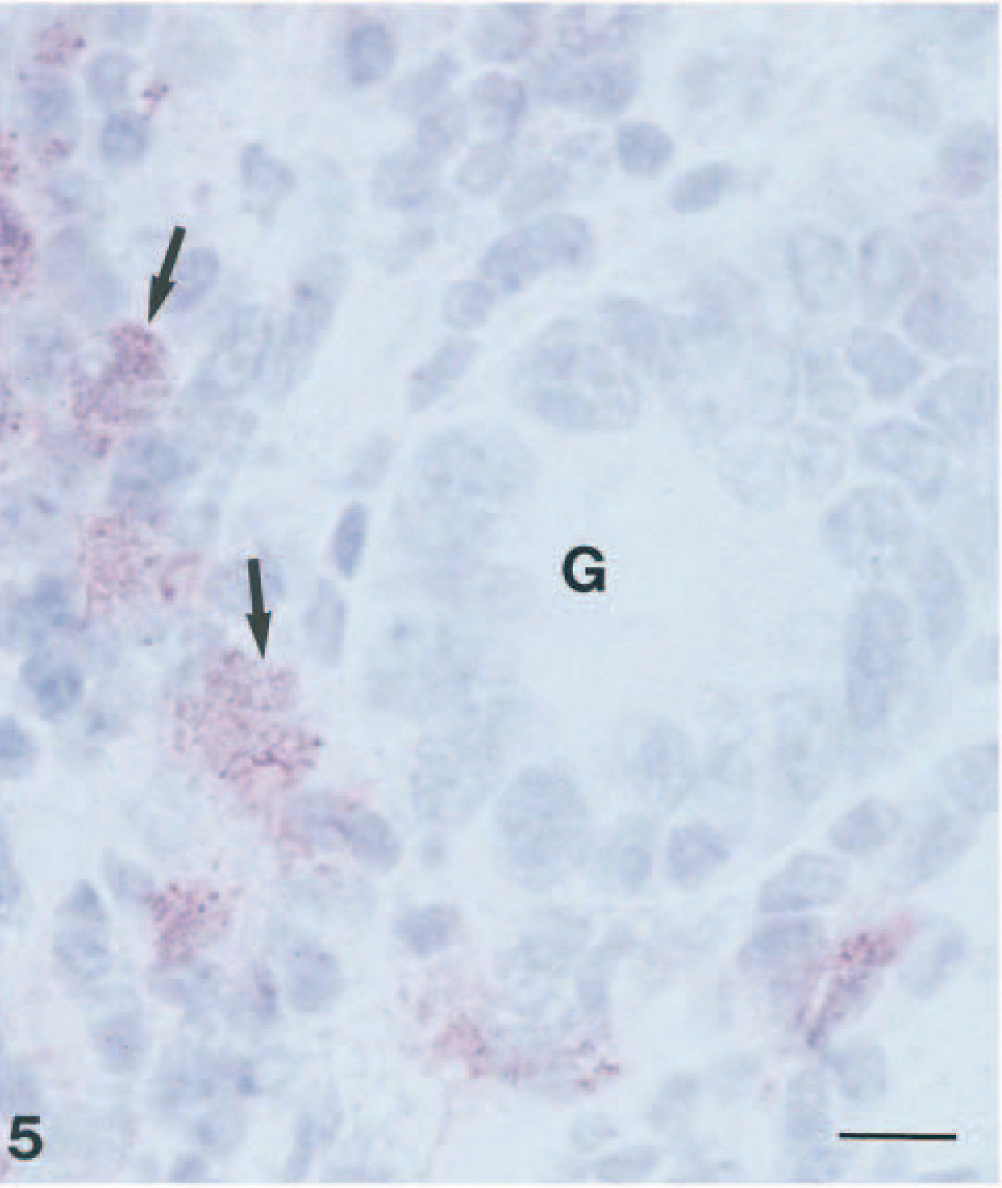
Stomach; ferret. Immunohistochemical staining for M. bovis in macrophages (arrows) was observed in the lamina propria. G = gastric pit. Streptavidin–biotin–alkaline phosphatase method, hematoxylin counterstain. Bar = 10 µm.
Liver and lung tissue was cultured for mycobacteria at the Department of Bacteriology (National Veterinary Institute, Oslo, Norway). The samples were decontaminated with 5% oxalic acid for 25 minutes and plated on slants of Petragani medium, Stonebrink medium, and Middlebrook 7H10 medium (Difco Laboratories, Detroit, MI). 8 The slants were incubated aerobically at 37 C for 2 months and checked for growth every week. Visible growth was observed on all media from both the liver and lungs after 10–14 days. The cultures were acid-fast bacilli, as determined by the ZN method, that did not form spores, capsules, or aerial hyphae. The colonies developed yellow pigmentation independent of light. The isolates exhibited reactions below the cutoff with the AccuProbe MAC (M. avium complex) and AccuProbe MTB (Mycobacterium tuberculosis) identification kits (Gen-Probe, San Diego, CA) when the unbound probe hydrolysis incubation time was 10 minutes. However, the isolates gave false-positive reactions with AccuProbe MTB when the unbound probe hydrolysis incubation time was 5 minutes. Species identification was performed at the Department of Bacteriology (National Institute for Public Health, Oslo, Norway) by sequencing a fragment of the 16S ribosomal DNA (rDNA) gene. For polymerase chain reaction (PCR) amplification, the previously described primers pA and pD were used. 13 After visualization on a 0.7% agarose gel using ethidium bromide, the PCR product was purified for sequencing using shrimp alkaline phosphatase and exonuclease I (Amersham Pharmacia Biotech, Buckinghamshire, England), according to the manufacturer's instructions. Sequencing was performed using the ABI Prism Big Dye terminator Cycle Sequencing Ready Reaction kit (PE Applied Biosystems, Foster City, CA), as recommended by the manufacturer, using the primers pB and pC described previously. 13 Sequencing reactions were run on the ABI prism 377 (PE Applied Biosystems) using 5% Long Ranger Gels (FMC Bioproducts, Rockland, ME). Sequences were assembled using Auto Assembler DNA Sequence Software, version 2.0. The consensus sequence was analyzed using a GenBank BLASTN search. The isolate had a 16S rDNA sequence identical to that of M. celatum type 3. 2
Based on the above findings, M. celatum type 3 was considered to be the causal agent of the disseminated granulomatous infection observed in the ferret. In the tissues of the ferret, M. celatum grew intracellularly as long, slender, sometimes branching bacteria. The organism induced diffuse granulomatous infiltrates without the formation of discrete granulomas. The type and distribution of lesions observed corresponded to observations in humans of poorly defined granulomas and pulmonary or disseminated infection. 1,9 However, descriptions of the pathology associated with the lesions in humans are scarce.
This was the first identification of M. celatum type 3 in Norway. During the next 2 months, two human isolates were received for identification at the National Institution of Public Health in Oslo. There was no known connection between the diseased ferret and the human patients; all originated from different regions of the country. During 1999, no further human or animal isolates were reported in Norway.
The epidemiology of nontuberculous mycobacteria is not well understood, but person-to-person transmission has not been demonstrated. This ferret was kept as a pet in close contact with humans. The animal most probably shed M. celatum in sputum and feces, resulting in mycobacterial contamination of the environment. Thus, transmission of mycobacteria to humans might have occurred. In humans, M. celatum is mainly isolated from immunocompromised patients. The diseased ferret had no history of previous recurrent disease, and no age-related immunodeficiency was likely in this 4-year-old animal; the average life expectancy of ferrets is about 10 years.
Ferrets might be more susceptible to mycobacterial infections than other species. An epidemiologic investigation in New Zealand showed that infection with M. bovis is common in feral ferrets (Mustela furo), and the prevalence of tuberculosis is higher in feral ferrets than in stoats and cats. In New Zealand, 17% of captured ferrets in an area endemic for bovine tuberculosis were infected. 11 In the M. bovis-infected ferrets, only 2.9% of the pathologic changes were localized to the respiratory tract, whereas 34.5% of the mesenteric lymph nodes had tuberculous lesions, suggesting the importance of oral infection. 12 The route of infection of M. celatum is not known, but the widespread lesions in the respiratory tract of the ferret in the present study may suggest transmission by inhalation.
By immunohistochemistry, M. celatum reacted positively with polyclonal antibodies against M. paratuberculosis and M. bovis. This finding was not surprising because these antibodies are reported to react with a multitude of antigens that are common to mycobacteria. 7
The isolated mycobacterium was identified as M. celatum type 3 using 16S rDNA sequence analysis. M. celatum cannot be identified by biochemical characteristics because these are identical to those of bacteria of the M. avium complex. 2,16 Furthermore, DNA probes cannot be used for identification because these probes may give misleading results. An M. tuberculosis DNA probe used for culture confirmation showed cross-reactivity with M. celatum type 1 and type 3 infections. 2,3 Determination of the signature sequence of the 16S rDNA is a quick and reliable method for identification of this rare mycobacterium, and the method differentiates among the three groups of M. celatum. 2,3
This case report shows that mycobacterial infection should be considered as a possible cause of chronic respiratory disease in ferrets.
Footnotes
Acknowledgements
We thank Anne-Marie Klem, Vivi Myrann, and Birgit R⊘e for their skillful technical assistance.
