Abstract
Three kittens, ages 5, 9, and 17 weeks, were found dead by separate caregivers and were submitted for necropsy. At gross necropsy, each kitten had hemorrhagic or bloody fibrinoserous thoracic fluid and differing distributions of pulmonary consolidation. On histologic examination, the pulmonary lesion in each kitten was similar and was characterized by acute necrotizing and hemorrhagic pneumonia and pleuritis, with numerous intralesional small Gram-negative rods. A pure culture of a distinct serotype of Escherichia coli was identified in lung tissue from each kitten (O4:H5, O6:H7, O6:H5). Lung isolates, genotyped by polymerase chain reaction, carried genes that are characteristic of extraintestinal pathogenic E. coli (ExPEC), including cnf-1, papG allele I, papA, papC, sfa, fim, hlyD, malX, iroN, fyuA, kpsMII, and ompT. Escherichia coli isolates from the intestines of 2 of the kittens were 100% related to the respective lung isolate, as determined by pulsed-field gel electrophoresis. Cultures of fecal samples collected from a clinically healthy cohort population of kittens revealed 16 of 19 tested kittens (84%) to be shedding hemolytic E. coli. Ten different serotypes were identified from 43 hemolytic E. coli fecal isolates from the cohort population, each of which had a genetic profile consistent with that typical of ExPEC. To the authors’ knowledge, this is the first report to describe a cluster of isolated cases of pneumonia in kittens caused by distinct serotypes of ExPEC and to evaluate the prevalence of hemolytic E. coli carrying ExPEC-associated genes in the feces of a cohort population of kittens.
Introduction
Escherichia coli are common commensal bacteria that reside in the mammalian gut. Although many strains of E. coli reside as host flora, some strains are recognized pathogens capable of causing either enteric or extraintestinal infections and disease. Intestinal pathogenic E. coli have been subdivided into different groups based on clinical disease manifestations and the presence or expression of genes that encode for known virulence factors. These groups include enteropathogenic strains that carry bundle forming pili (bfp) and intimin (eae), enterotoxigenic strains that are capable of expressing heat stable (STa, STb) and heat labile (LT) enterotoxins, and shiga toxin producing E. coli strains that express one or more cytotoxins (Stx1 and Stx2). 1 Extraintestinal pathogenic E. coli (ExPEC) have virulence gene profiles that are typically distinct from commensal and intestinal pathogenic strains of E. coli. Reported ExPEC-associated genes include cytotoxic necrotizing factors (cnf-1, cnf-2), P fimbriae adhesins (papG allele I and papG allele III), major structural subunit of the P fimbrial shaft (papA), pilus assembly protein (papC), S fimbriae (sfa), F1C fimbriae (foc), type 1 fimbriae (fim), hemolysin D (hlyD), pathogenicity-associated island marker (malX-“PAI”), novel catecholate siderophore receptor (iroN), Yersinia siderophore receptor (fyuA), genes associated with capsular polysaccharide synthesis (kpsMTII), and outer membrane peptidase (ompT). 11–13,16,18 The roles that ExPEC virulence attributes play in the pathogenicity of ExPEC strains were previously summarized. 1,11–13,18 In brief, pili or fimbriae and fimbrial adhesin molecules promote adherence and colonization in tissue, Cnf-1 and HlyD are cytotoxins that evoke tissue necrosis, and siderophore receptors are important for iron acquisition. ExPEC isolates were associated with urinary tract infections, pneumonia, septicemia, and meningitis in humans, 17 urinary tract infections, genital infections, and septicemia in companion and food animals, 1,17 and necrohemorrhagic pneumonia in dogs, in a horse, and in a group of shelter cats. 3,6,9,18
The present report describes a cluster of 3 isolated cases of necrohemorrhagic pneumonia and pleuritis in kittens, induced by distinct serotypes of ExPEC. To evaluate the prevalence of E. coli carrying ExPEC-associated genes in the feces of kittens, a cohort population of clinically healthy kittens was screened.
Material and methods
Animals
Over a 2-month period in the early fall of 2007, 3 kittens were submitted for gross necropsy after being found dead by their caregivers. The kittens had no identified environmental overlap. The first kitten (kitten A), a 5-week-old female Domestic Shorthair, had a 1-week history of intermittent diarrhea. This kitten was reported to be febrile approximately 12 hr before being found dead. This kitten was housed with one reportedly healthy littermate. The second kitten (kitten B) was a 9-week-old, male Domestic Medium Hair that died 24 hr after an acute onset of lethargy and weakness. This kitten had mild upper respiratory disease that was reported to have clinically resolved 1 week before death with no antibiotic treatment. The 3 littermates and queen were healthy at the time of kitten B's death. The caregiver had supplemented all 4 kittens with bottle-feeding at 4–5 weeks of age because of a nonspecified illness that resulted in insufficient milk production by the queen. The third kitten submitted for gross necropsy (kitten C) was a 17-week-old male Domestic Shorthair that was found dead 1 week after clinical resolution of a 2-week history of antibiotic responsive lower airway disease. Treatment included 1 week of oral enrofloxacin administration followed by 1 week of amoxicillin-clavulanic acid administration. Antibiotics were discontinued 1 week before kitten C was found dead. This kitten was individually housed in an enclosure that housed multiple other adult cats and kittens, and there were no reports of illness among the other cats in the enclosure.
Fecal samples were collected from a cohort population. The sampled population was composed of 19 kittens, ages 6–12 weeks, which were living in foster homes. Caregivers of the kittens were asked to provide the signalment, medication and vaccination history, and prior and present health status of each kitten. Only 1 kitten per household was accepted into the test population. A single fecal sample from each kitten was collected from the litter box. Each kitten was required to be clinically healthy at the time of fecal collection and could not have received antibiotics for a minimum of 5 days before fecal collection.
Necropsy and histology
A complete gross necropsy examination was performed on the bodies of the 3 deceased kittens, and a standard set of tissues was collected and fixed in 10% neutral buffered formalin. Fixed tissues collected from kittens A and B were submitted to the histology laboratory at the William R. Pritchard Veterinary Medical Teaching Hospital (VMTH; University of California, Davis, CA) and tissues from kitten C were submitted to the Comparative Pathology Laboratory (CPL; University of California, Davis, CA) for standard processing and hematoxylin and eosin staining. Lung sections from each kitten were stained with Brown and Brenn Gram stain. Segments of intestine, including jejunum and colon from kitten A, and duodenum and jejunum from kitten B, were collected and stored at −70°C.
Bacterial isolation
Fresh samples of lung tissue from kittens A and B were submitted to the microbiology laboratory at the VMTH. Gram-stained impression smears were prepared from the lung tissue of each kitten. Lung samples were inoculated directly onto 5% defibrinated sheep blood agar and prereduced Brucella sheep blood agar and were incubated at 35°C under aerobic (with 5% CO2) and anaerobic (85% N2, 10% H2, and 5% CO2) conditions, respectively. Lung tissue from kitten C was cultured by using similar methods at the CPL. The pure lung culture isolate obtained from kitten C was submitted to the microbiology laboratory at the VMTH for identification confirmation.
The intestinal segments collected and frozen at the time of gross necropsy from kittens A and B were thawed, and the lumina were swabbed for bacterial culture. Samples from each of the intestinal segments of kittens A and B and from the fecal samples collected from the cohort population were inoculated separately for aerobic culture on 5% defibrinated sheep blood agar to demonstrate hemolysis and on MacConkey agar to enable the identification of E. coli in the event that the blood agar media was overgrown with other bacteria. Isolates were identified as E. coli based on the following biochemical reactions: spot oxidase negative, spot indole positive, acid slant over acid base, with gas production in triple sugar iron agar, urease negative on Christensen's urea agar, sulfur negative-indole positive-motility positive on sulfur-indole-motility deep agar, and no utilization of citrate on Simmons’ citrate agar. 7
Bacterial serotyping and genotyping
Isolated colonies of hemolytic E. coli cultured from the lungs (kittens A, B, and C), intestinal samples (kittens A and B), and feces (cohort population) were selected from the blood agar plates and serotyped and genotyped for virulence attributes. Each of the 3 pulmonary isolates, 5 enteric isolates from kitten A, 4 enteric isolates from kitten B, and up to 3 isolates from each of the hemolytic E. coli-positive cohort fecal samples were analyzed. Serotyping, bacterial DNA isolation, and virulence genotyping were performed by using previously described methods. 5,14,15,18 Initially, isolates were genotyped to assess the presence of genes encoding for the following virulence attributes: Cnf-1, Cnf-2, LT, STa, STb, Stx-1, Stx-2, and Eae. Isolates that were positive for cnf-1 and negative for all other genes were then screened for ExPEC-associated genes, including sfa, foc, fim, papG alleles I and III, papA, papC, hlyD, fyuA, iroN, malX, ompT, and kpsII. Relatedness of the lung and intestinal hemolytic E. coli isolates from kittens A and B was assessed by pulsed-field gel electrophoresis. 8
Antimicrobial susceptibility
Bacterial isolates from the lungs of kittens A, B, and C were tested for antimicrobial susceptibility at the microbiology laboratory at the VMTH by using a microdilution technique. a Tested antimicrobials included amikacin, amoxicillin-clavulanic acid, ampicillin, cefazolin, cefoxitin, cefpodoxime, cephalothin, chloramphenicol, enrofloxacin, gentamicin, imipenem, marbofloxacin, orbifloxacin, tetracycline, ticarcillin-clavulanic acid, ticarcillin, and trimethoprim-sulfamethoxazole.
Copathogen screening
Formalin-fixed, paraffin-embedded (FFPE) tissues from kittens A, B, and C were submitted to the Lucy Whittier Diagnostic Laboratory (University of California, Davis, CA) and to the Pathobiology Diagnostic Services, College of Veterinary Medicine (Auburn University, AL) for DNA isolation and polymerase chain reaction (PCR) analysis. Lung tissue from each of the deceased kittens was screened for Bordetella bronchiseptica, Chlamydophila felis, Feline calicivirus (FCV), and Felid herpesvirus 1 at the Lucy Whittier Diagnostic Laboratory. FFPE tissues, including thymus, lymph nodes (mediastinal and ileocecal), and distal intestinal sections from kittens A and B and thymus, mesenteric lymph nodes, bone marrow, and spleen from kitten C, were submitted to the Lucy Whittier Diagnostic Laboratory for Feline panleukopenia virus (FPV) and Feline leukemia virus (FeLV) screening and to the Pathobiology Diagnostic Services, College of Veterinary Medicine (Auburn University, AL) for Feline immunodeficiency virus (FIV) screening.
Results
Necropsy
At necropsy, kitten A was in moderately thin nutritional condition, kitten B was in mildly thin nutritional condition, and kitten C was in good nutritional condition. Kittens B and C had bloody fluid around and in the mouth and within the large airways. Each of the kittens had blood-tinged fibrinoserous (kitten A) or hemorrhagic (kittens B and C) pleural fluid that ranged in volume of approximately 3–5 ml (Fig. 1). Each kitten had differing severities and distributions of pulmonary parenchymal consolidation. The pulmonary lesion in kitten A was restricted to a diffusely dark red and tan mottled consolidated left caudal lung lobe. The lungs of kittens B and C were mottled red to dark red, with large coalescing regions of consolidation (Fig. 1). On cut section, hemorrhagic fluid seeped from the affected pulmonary parenchyma. Additional gross necropsy findings for kitten A included a deep focal ulcer in the proximal duodenum, mildly gas-distended distal small intestine, and enlarged cranial sternal and mesenteric lymph nodes. The ileocecal lymph node of kitten B was enlarged. All other tissues from the kittens were otherwise grossly unremarkable.
Histology
On histologic examination, the affected regions of the lungs in each kitten exhibited acute coagulative necrosis of terminal bronchiolar walls, alveolar septae, and pleural surfaces, with associated fibrinoid necrosis of vessel walls and hemorrhage. Proteinaceous fluid and fibrin frequently filled terminal bronchiolar lumens and alveolar spaces within the affected regions. Numerous Gram-negative rod-shaped bacteria were in the airways along bronchioloalveolar junctions, within alveolar spaces, lining necrotic alveolar septae, and within necrotic pleura and subpleural spaces (Figs. 2–4). There were inflammatory cell infiltrates within the affected regions of the lungs that ranged from a few to moderate (kittens B and C) to numerous (kitten A) variably degenerate neutrophils, intermixed with a few to moderate numbers of alveolar histiocytes that occasionally contained small rod-shaped bacteria. In kitten A, there was progression to lobar pneumonia.
Kittens A and B had histologic evidence of a mild enteritis and/or colitis, and the duodenal ulcer described grossly in kitten A was acute, with minimal associated inflammation. The cranial sternal, mesenteric, and ileocecal lymph nodes of kitten A exhibited lymphoid hyperplasia and sinusoidal plasmacytosis, and there was a sinus histiocytosis within the examined tracheobronchial lymph node. Lymphoid hyperplasia with follicular lympholysis was noted within Peyer's patches and lymph nodes (tracheobronchial, mesenteric, and ileocecal) and the thymus exhibited marked lympholysis in kitten B. Additional histologic lesions were minimal to mild and included acute random hepatic necrosis and perivascular lymphoplasmacytic meningitis.
Bacterial isolation
Gram-stained impression smears, performed on lung tissue submitted for culture from kittens A and B, revealed moderate to large numbers of Gram-negative rod-shaped bacteria. Moderate numbers of hemolytic E. coli were isolated in pure culture from the lung tissue of kittens A and B. No anaerobic bacteria were isolated from the lungs of either kitten after 5 days of incubation. A pure culture of hemolytic E. coli was isolated in large numbers from the lung tissue of kitten C.
Small to moderate numbers of hemolytic E. coli and moderate numbers of nonhemolytic E. coli were isolated from the intestine of kitten A. Small to large numbers of hemolytic E. coli were detected in the intestine of kitten B. Small numbers of mixed growth and no other Gram-negative rods were detected in the intestine of kitten B. Very small to moderate numbers of hemolytic E. coli were isolated from 16 of the 19 fecal samples (84%) collected from the cohort population of kittens.
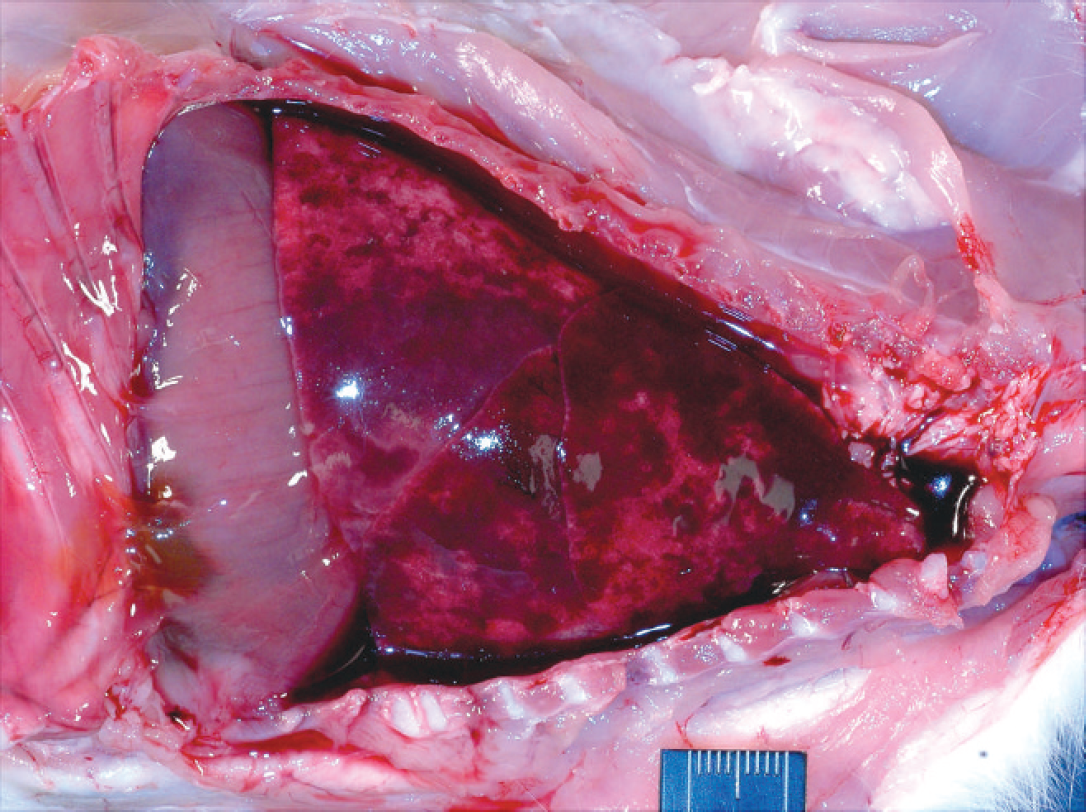
Thoracic cavity; kitten B. Hemorrhagic pleural fluid, consolidation of the lungs, and fibrin adhered to the visceral pleura.
Antimicrobial susceptibility and copathogen screening
Escherichia coli isolates from the lungs of the 3 deceased kittens were susceptible to all tested antimicrobials. FFPE tissues from kittens A, B, and C were PCR negative for Bordetella bronchiseptica, Chlamydophila felis, FCV, FHV-1, FeLV, and FIV. FFPE tissues from kittens B and C were positive for Mycoplasma felis, and FFPE tissues from kitten B were positive for FPV as detected by PCR.
Bacterial serotyping and genotyping
Deceased kittens. Hemolytic E. coli isolates from the lungs of kittens A, B, and C were serotyped as
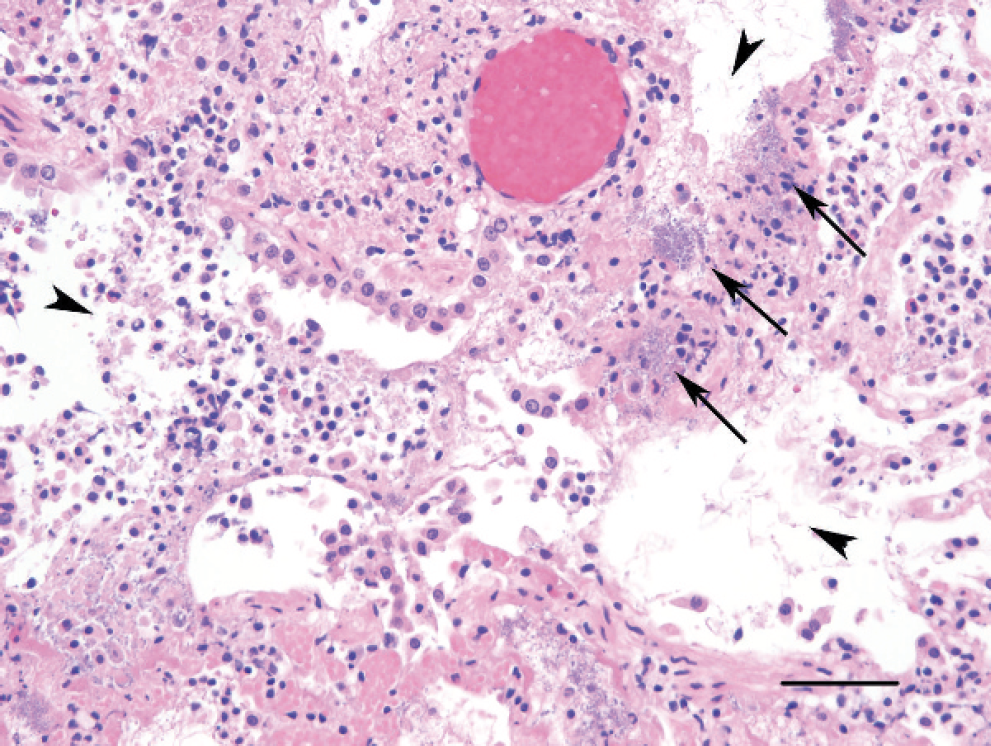
Lung; kitten C. Terminal bronchiolar walls and alveolar septae exhibit acute coagulative necrosis. Terminal bronchiolar lumens and alveolar spaces contain proteinaceous exudate, fibrin, moderate numbers of sloughed epithelial cells, and few neutrophils and histiocytes (arrowheads within the bronchiolar-alveolar airways). Numerous bacteria line the walls of the bronchiolar-alveolar junction and alveolar septae (arrows). Hematoxylin and eosin stain. Bar = 50 μm.
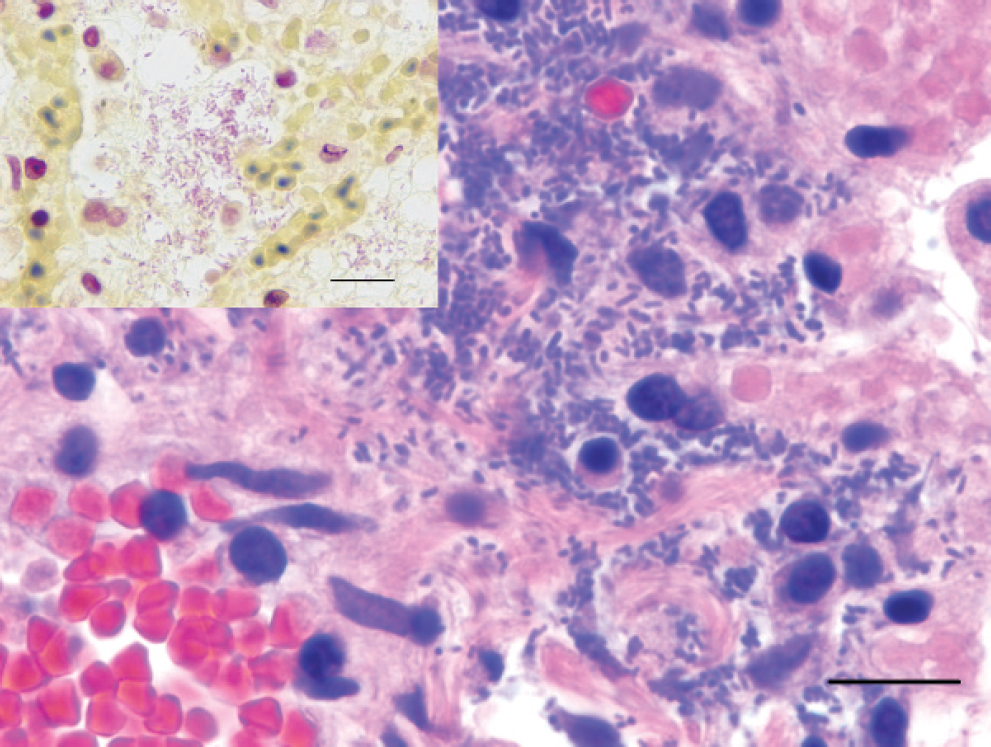
Lung; kitten C. Higher magnification shows the bacterial morphology to be small, approximately 1–3 μm long, rod-shaped, and Gram-negative. Hematoxylin and eosin stain. Bar = 10 μm. Inset: Brown and Brenn Gram stain. Bar = 10 μm.
O4:H5, O6:H7, and O6:H5, respectively. The only hemolytic E. coli isolate tested from the jejunum and 3 of 4 isolates tested from the colon of kitten A were of the same serotype (O4:H5) as the isolate from the lung of this kitten. Similarly, 1 of 2 hemolytic E. coli isolates tested from the duodenum, and 1 of 2 isolates from the jejunum of kitten B, were of the same serotype (O6:H7) as the isolate from the lung of this kitten.
Each of the O4:H5, O6:H7, and O6:H5 isolates from the lungs and intestines carried virulence genes characteristic of ExPEC, including cnf-1, papG allele III, papA, papC, sfa, foc, fim, hlyD, malX, iroN, fyuA, kpsMII, and ompT. In addition, the O6:H7 lung and intestinal isolates from kitten B also carried the papG allele I gene. Other genes, encoding for virulence
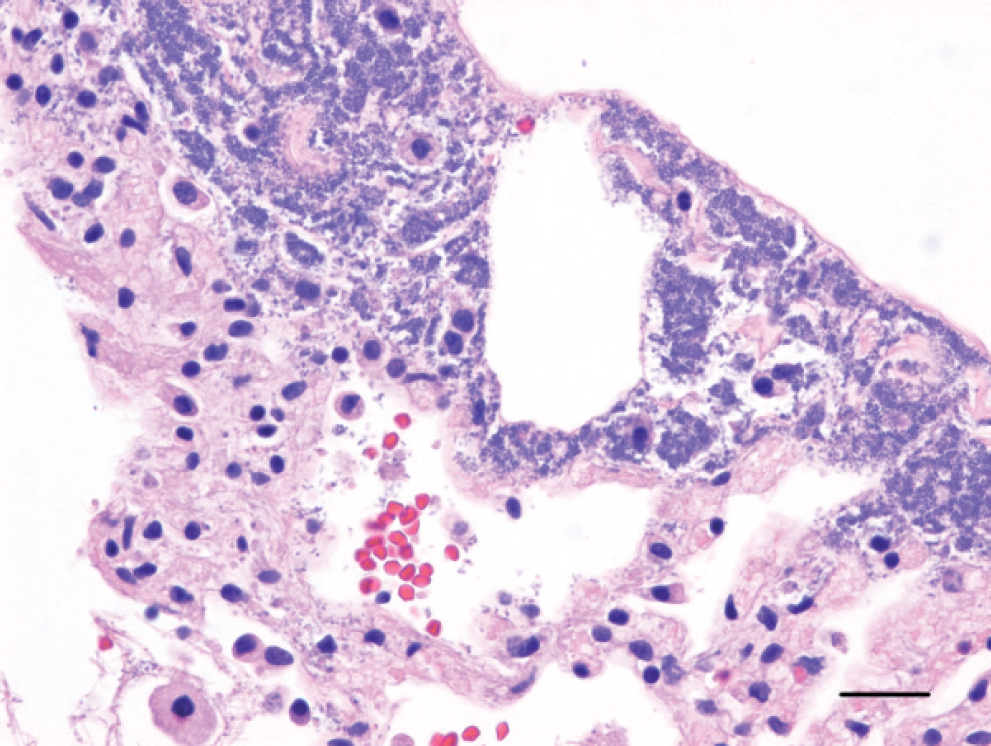
Lung; kitten B. The visceral pleura and alveolar septae exhibit acute coagulative necrosis. Numerous bacteria are in the necrotic pleura, fill the expanded subpleural space, and line the subpleural alveolar septae. Erythrocytes are seen within alveolar spaces. Hematoxylin and eosin stain. Bar = 50 μm.
Hemolytic Escherichia coli serotype and genotype profiles from the deceased kittens and fecal isolates from the cohort population. *
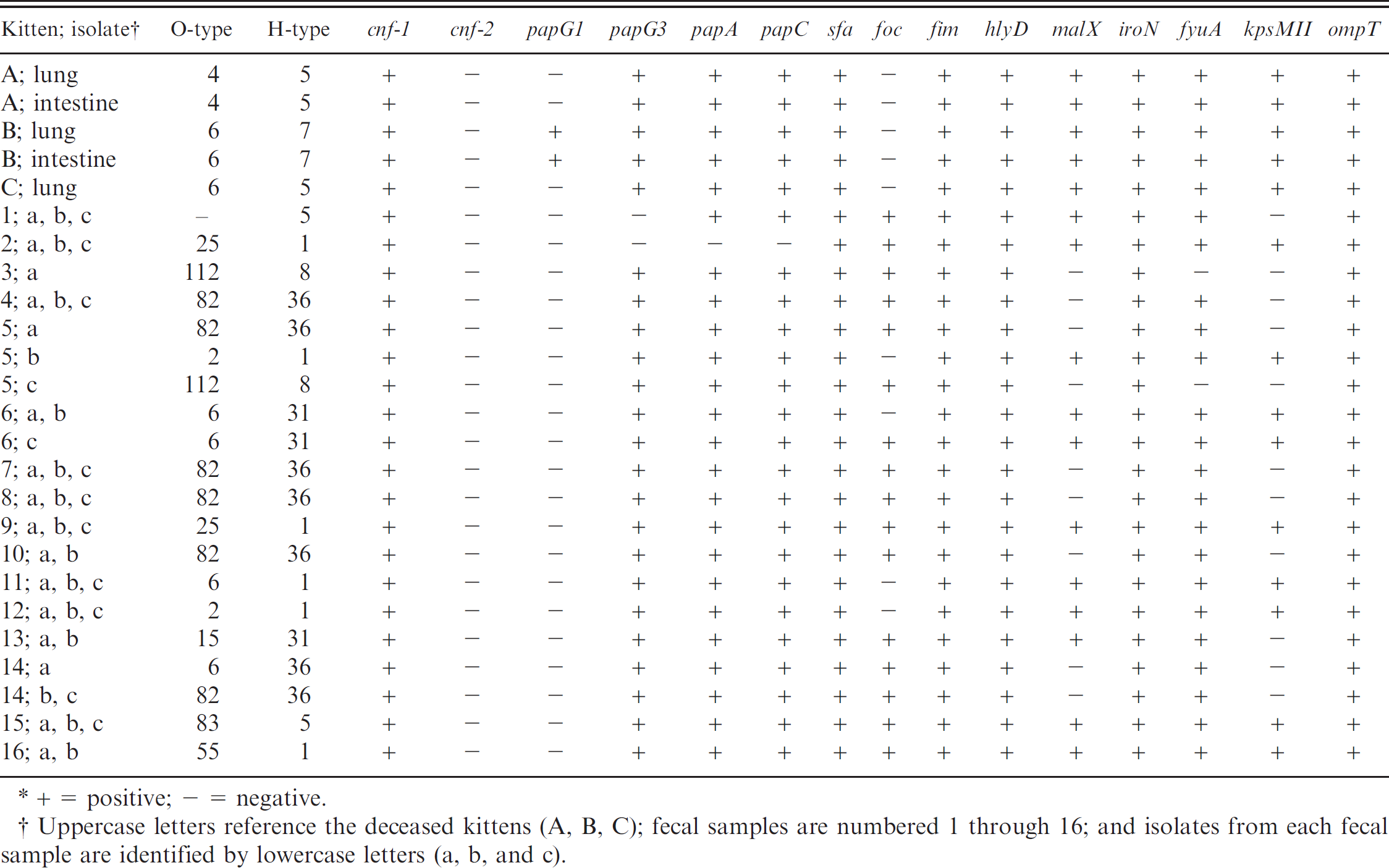
+ = positive; — = negative.
Uppercase letters reference the deceased kittens (A, B, C); fecal samples are numbered 1 through 16; and isolates from each fecal sample are identified by lowercase letters (a, b, and c).
attributes typically associated with enteropathogenic strains of E. coli, including LT, STa, STb, Stx-1, Stx-2, and Eae, were not detected in any of the isolates. Serotypes and virulence profiles of the hemolytic E. coli isolates from lungs and intestines of the 3 deceased kittens are summarized in Table 1. Pulsed-field gel electrophoresis indicated that the lung and intestinal E. coli isolates with the same serotype and similar genetic profiles were 100% related and confirmed that the isolates from each kitten were genetically distinct.
Cohort population. Hemolytic E. coli isolates from the 16 positive fecal samples were serotyped and genotyped for virulence attributes. The analysis included a total of 43 isolates, with 3 isolates submitted from each of 12 fecal samples, 2 isolates submitted from each of 3 fecal samples, and 1 isolate submitted from 1 of the fecal samples. Ten different serotypes were identified, and 3 of the isolates had an unidentified O antigen. Serotypes identified included 14 isolates of O82:H36, 6 isolates of O25:H1, 4 isolates of O2:H1, 3 isolates of O6:H1, 3 isolates of O6:H31, 3 isolates of O83:H5, 2 isolates of O112:H8, 2 isolates of O15:H31, 2 isolates of O55:H1, and 1 isolate of O6:H36. The 3 isolates having an unidentified lipopolysaccharide antigen (O) were each from the same fecal sample, and each had the same flagellar antigen type (H5). Each of the 43 tested hemolytic E. coli isolates carried at least 10 virulence attribute genes. All of the isolates (100%) carried cytotoxic necrotizing factor (cnf-1), fimbriae (sfa, fim), and a hemolysin toxin (hlyD). Thirty seven of the isolates (86%) carried a fimbrial adhesin gene (papG allele III), and 34 of the isolates (79%) carried the gene encoding for F1C fimbriae (foc). The genes encoding for the following virulence attributes were not detected in any of the analyzed fecal isolates: PapG allele I, Cnf-2, LT, STa, STb, Stx 1, Stx 2, and Eae. The serotypes and virulence factors identified in each of the fecal isolates are summarized in Table 1.
Discussion
The current article describes fulminant necrotizing and hemorrhagic pneumonia and pleuritis in 3 kittens. Pure hemolytic E. coli isolates were cultured from lung tissue of each kitten. The clinical history together with the histopathologic distribution of the lesions are most consistent with bronchopneumonia and subsequent pleuritis, which may indicate an aerogenous rather than a hematogenous source of infection. 4 On histologic examination, the inflammation and bacterial colonies were often centered on bronchioloalveolar junctions, a common site for inhaled pathogens to lodge and a feature of bronchopneumonia. Extending from the bronchioloalveolar junctions, the surrounding alveolar septae and blood vessels were frequently necrotic, and the possibility of an interstitial component to the pneumonia, likely from a septicemia, cannot be completely ruled out. At the time of death, these kittens were eating solid food and not being bottle-fed. This fact, in addition to the lack of identifiable foreign material in airways and a pure culture of E. coli from the affected lungs, makes aspiration pneumonia an unlikely underlying etiology. Although ExPEC commonly has been associated with urinary tract infections in cats, 1 these kittens had no gross or histologic evidence of cystitis or pyelonephritis.
Further characterization of the pulmonary hemolytic E. coli isolates demonstrated that each kitten was infected with a genetically distinct isolate of ExPEC. Hemolytic E. coli isolated from the intestines of 2 of the kittens were 100% related to the respective lung isolate, which implicated the intestine as the possible source for the pulmonary pathogen. To investigate fecal shedding as a potential source of ExPEC infections, fecal cultures were performed on a cohort population of clinically healthy kittens, revealing an 84% prevalence of hemolytic E. coli isolates that have genetic virulence profiles characteristic of ExPEC. This suggests that fecal shedding may indeed provide a source of ExPEC-associated infections, including respiratory infection as in the cases documented in the current report.
Two previous reports described a similar fulminant hemorrhagic pneumonia in which ExPEC were implicated as the etiology in 5 young dogs. 3,9 Similar to the 3 analyzed fecal isolates from kitten 6 (cohort population) in this report, the ExPECs that were isolated in pure culture from the lungs of one of these dogs was serotyped as 06:H31 and carried genes encoding for Cnf-1 and papG allele III. 3 A 2007 report described a suspected point source outbreak of fatal ExPEC-induced necrotizing and hemorrhagic pneumonia in 13 kittens and adult cats in a shelter. 18 The ExPEC identified in that outbreak was serotyped as O4:H5, the same serotype isolated from the lungs and intestine of kitten A in the current report. Similar to the kittens described in the present report, intestinal cultures from 3 of the dogs and 2 of the cats in these previous reports identified intestinal ExPEC isolates having similar genetic virulence profiles as the lung isolates in those animals. Serotypes and virulence genes identified in the hemolytic E. coli isolates from one of the deceased kittens (kitten B) and from the cohort population (kittens 6 and 11) in the current report are similar to ExPEC strains that are known to cause urinary tract infections in humans and dogs. 10 The similarities between serotype and genotype among human and animal isolates of ExPEC certainly raise concern for zoonotic potential.
A few reports demonstrated that strains of E. coli capable of producing Cnf-1 can be isolated from feces of healthy cats 2 and that E. coli having genetic profiles typically associated with ExPEC can be isolated from the feces of healthy cats and dogs. 19,20 The selection criteria of the screened E. coli isolates in those reports was either not described 19,20 or not specific for hemolytic isolates. 2 In addition, these reports screened fewer virulence genes in their isolates compared with the screening performed in this report.
The present report shows that potentially pathogenic E. coli are present within the feces of clinically healthy kittens, with an 84% prevalence within the test population in this report. It remains unclear why certain strains or isolates of E. coli with virulence profiles characteristic of ExPEC cause disease in some kittens and/or cats while not affecting others that are in close proximity. It is unknown if stressful conditions, such as environmental stressors or concurrent disease, increase the prevalence of intestinal E. coli that carry ExPEC-associated virulence genes with a subsequent increase in fecal shedding. Interestingly, the 3 kittens with pneumonia and the cohort population in this report, as well as the kittens, cats, and young dogs previously reported in the veterinary literature, were all in potentially stressful environments (group housing, foster homes, recent relocations or travel). In addition, each of the deceased kittens in the current report had either recent clinical respiratory disease (kitten B and C) or intermittent intestinal illness (kitten A). Mycoplasma felis was detected in lung tissue from 2 of the kittens (kitten B and C), and FPV was detected in lymphoid tissue from 1 of the kittens (kitten B) by PCR. These observations raise questions as to the effect that the host's immune competency, health status and/or previous antibiotic therapy may have on the prevalence of ExPEC strains in the intestine, shedding of the bacteria, and host susceptibility.
To the authors’ knowledge, this is only the second report that describes a fulminant hemorrhagic pneumonia in cats induced by ExPEC and the first report describing a cluster of isolated cases in kittens caused by genetically distinct strains of ExPEC. The current report provides further evidence that ExPEC should be recognized as an etiologic agent that can cause fulminant necrotizing and hemorrhagic pneumonia in kittens and implicates the host's intestine and feces as a potential source of infection.
Acknowledgements
The authors thank Dr. Dennis Wilson for his pulmonary pathology insight, Dr. Joaquin Ortega for his technical assistance, Anita Wong for assistance with isolation and identification of fecal E. coli, and the University of California, Davis, School of Veterinary Medicine, Orphaned Kitten Project for providing fecal samples for screening.
Footnotes
a.
Sensititrer, Trek Diagnostic Systems Inc., Westlake, OH.
