Abstract
Two Vietnamese potbellied pigs (Sus scrofa) had respiratory disease and, on autopsy, both pigs had large masses in the lungs and thoracic cavity. Microscopically, pulmonary and pleural masses contained large areas with hyphae surrounded by hypereosinophilic cellular debris rimmed by abundant eosinophils, lymphocytes, plasma cells, and histiocytes with occasional multinucleate giant cells. The hypereosinophilic debris usually formed tight cuffs, or “sleeves” around the hyphae, compatible with Splendore-Hoeppli–like material. The fungal organisms were determined by PCR to be Conidiobolus incongruus in one pig and Mucor circinelloides in the other. Entomophthoromycosis and mucormycosis should be included in the differential diagnoses for swine pneumonia, particularly when there is evidence of granulomatous pulmonary masses and pleural effusion with eosinophilic inflammation.
Vietnamese potbellied pigs (Sus scrofa) were introduced into the United States in 1985 and are considered in many circles to be domesticated pets. Commercial swine and potbellied pigs have many similar disease conditions; however, differences occur primarily as the result of dissimilar housing, lack of intensive management, and usually longer lifespan in potbellied pigs. 17 Fungal organisms are considered to be sporadic causes of pneumonia in swine, with Pneumocystis carinii listed most frequently. 1 Reports of fungal respiratory infection in swine caused by organisms previously classified as “zygomycetes” (i.e., mucormycosis, entomophthoromycosis) are rare. 2 Mucormycosis or “zygomycotic” infection has been reported in the stomach 18 and lymph node2,3 in commercial swine and in the bone of the distal hindlimb of a potbellied pig. 11 We report fungal pneumonia in 2 Vietnamese potbellied pigs caused by orders Entomophthorales and Mucorales.
A 9-mo-old, 35.5-kg, neutered male Vietnamese potbellied pig was presented in August 2014 to the Louisiana State University, Veterinary Teaching Hospital and Clinic (LSU-VTH&C; Baton Rouge, LA) Food Animal Medicine service with a history of lethargy, acute hyporexia, and pain. The patient was up to date on vaccinations and deworming. Upon physical examination, the pig was pyrexic (T 39.8°C), tachypneic (RR 76), and tachycardic (HR 128). Additionally, the pig was dyspneic with a marked abdominal component to respiration (“thumps”) and increased lung sounds. No other physical abnormalities were noted. Given the primary differential diagnosis of bacterial pneumonia, empirical treatment was initiated with enrofloxacin (7.5 mg/kg intramuscularly; Baytril, Bayer, Osaka, Japan). Because no improvement was noted within 3 d of presentation, further tests were performed. Thoracic radiographs revealed severe bilateral pleural effusion and a mass in the right lung. Ultrasonographic findings of the thorax were similar with fibrinous or fibrous material in the effusion and marked atelectasis of the right lung lobes. Under general anesthesia, a thoracic drain was placed in the right hemithorax, where ~640 mL of pleural fluid were drained. Cytologic evaluation of the pleural fluid revealed increased nucleated cells (7.8 × 109/L, reference interval [RI]: <1.0 × 109/L) and protein (56 g/L, RI: <25 g/L) concentrations, with increased eosinophils (63% of nucleated cells). The cytologic diagnosis was eosinophilic inflammation. Despite all treatments, the patient’s health continued to decline and it was euthanized. A postmortem examination followed.
At autopsy, there was ~250 mL of serosanguineous fluid in the pleural cavity. Extensive fibrous adhesions connected lungs to right and left parietal pleura, and the diaphragm. Both caudal lung lobes and the left pleura had firm nodules; the largest was 12 × 9 × 6 cm. Cut surfaces of the masses were solid and pale tan with multifocal-to-coalescing dark red areas. Tracheobronchial lymph nodes were enlarged approximately twice normal size and diffusely pale on cut surface. Microscopically, hematoxylin and eosin–stained slides of lung and pleural masses contained large areas with hyphae surrounded by eosinophilic cellular debris or sleeves (Splendore–Hoeppli [SH]-like), and further surrounded by abundant eosinophils, lymphocytes, plasma cells, and macrophages. There were occasional multinucleate giant cells. Hyphae were ~7–10 μm diameter with occasional non-dichotomous branching and were poorly septate (Fig. 1). Other findings in the adjacent parenchyma included massive fibrin exudation and edema, marked fibroplasia and angioplasia, and numerous inflammatory cells, including a high percentage of eosinophils. The tracheobronchial lymph node was hyperplastic. Sinuses and sinusoids contained numerous eosinophils admixed with macrophages.
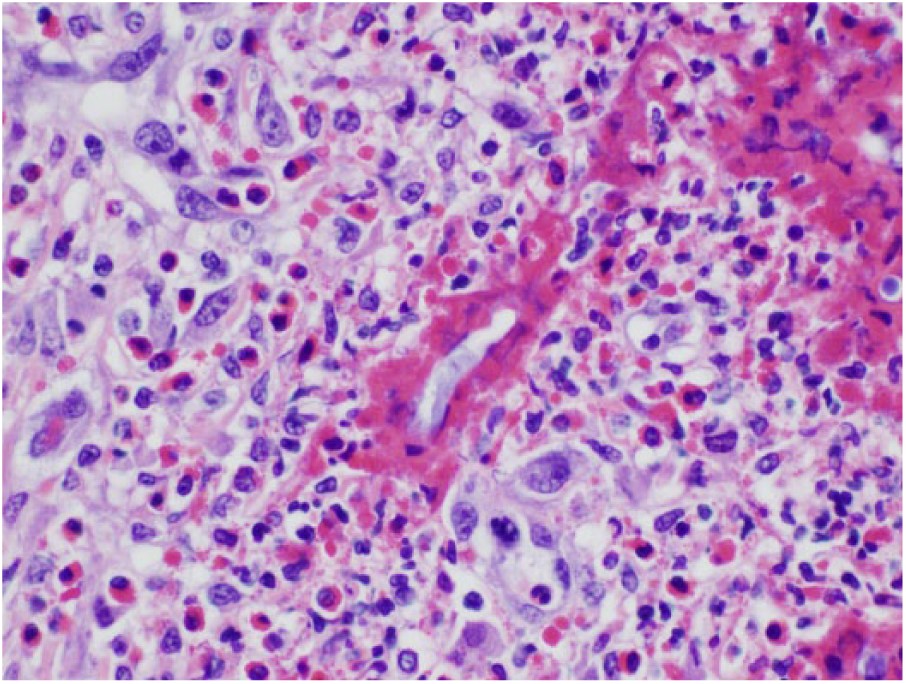
Microscopic evaluation of the granulomatous mass in case 1 has numerous Conidiobolus incongruus hyphae surrounded by Splendore-Hoeppli–like reaction.
Microbiologic culture of the mass did not yield any organisms or growth. A paraffin block was submitted to the Washington Animal Disease Diagnostic Laboratory (WADDL; Pullman, WA) for molecular sequencing, and extracted DNA was used as a template for PCR using pan-fungal primers, followed by direct sequencing and sequence analysis. 4 After DNA extraction, a portion of the 18S ribosomal RNA gene was amplified by PCR using published universal fungal primers. 23 When this PCR amplicon was directly sequenced, the sequence most closely matched that of Conidiobolus incongruus (>99% sequence identity [668/669-bp match] with GenBank accession AF114319).
A 20-mo-old male neutered pot-bellied pig from a different owner was presented in May 2015 to LSU-VTH&C Food Animal Medicine service with a history of coughing; the pig exhibited dyspnea and wheezing upon expiration. Temperature, pulse, and heart rate were within normal limits. A thoracic radiograph revealed a soft tissue mass in the left hemithorax and an alveolar pattern in the left caudal lung lobe. The only abnormalities on a complete blood count were mild neutrophilia (18.8 × 109/L, RI: 2–15 × 109/L) and marked eosinophilia (16.9 × 109/L, RI: 0–1.5 × 109/L). A blood chemistry panel indicated hyperglobulinemia (68 g/L, RI: 53–64 g/L), hypoglycemia (2.42 g/L, RI: 3.58–5.23 g/L), and titrational metabolic acidosis (bicarbonate 10.4 mmol/L, RI: 18–27 mmol/L; anion gap 41 mmol/L, RI: 10–25 mmol/L). Ultrasound revealed a moderate amount of free echogenic fluid in the left pleural space and a 5 × 8 cm heterogeneous mass in the left mid- to caudal thorax. Thoracentesis resulted in removal of 100 mL of yellow-green, clear fluid. Analysis of the thoracic fluid indicated increased nucleated cells (9.5 × 109/L, RI: <1.0 × 109/L) and total protein (67 g/L, RI: <25 g/L), with increased non-degenerate neutrophils (49% of nucleated cells) and eosinophils (20% of nucleated cells), consistent with neutrophilic and eosinophilic inflammation. The pig had a history of recent deworming with ivermectin, so a parasitic etiology for the eosinophilic inflammation was considered less likely. The owner elected euthanasia given the grave prognosis.
At autopsy, the left thoracic cavity contained 150 mL of serosanguineous fluid. The left lung was markedly and diffusely expanded by a large, firm mass of 14.5 × 11.5 × 11.5 cm; the mass was thickly coated with fibrin and adhered firmly to the left thoracic wall with extensive, mature fibrous strands (Fig. 2). The tracheobronchial lymph nodes were enlarged up to 5 times normal size and firm, with slightly irregular, nodular surfaces. Microscopically, in hematoxylin and eosin–stained slides, normal pulmonary architecture was completely effaced and replaced by coalescing eosinophilic granulomas centered on hyphae 7–8 μm wide, and occasionally septate with nonparallel walls and non-dichotomous branching (Fig. 3). Hyphae were surrounded by “eosinophilic sleeves” or SH-like material, and large numbers of degranulated eosinophils. Granulomas contained epithelioid macrophages, multinucleate giant cells, and peripheral dense fibrous tissue infiltrated by epithelioid macrophages, eosinophils, lymphocytes, and plasma cells. Other findings included mature granulation tissue infiltrated by similar inflammatory cells, pockets of fibrin, and markedly increased numbers of pulmonary alveolar macrophages. In the tracheobronchial lymph node, 75–85% of the parenchyma was replaced by dense fibrous tissue infiltrated by moderate numbers of eosinophils, macrophages, and fewer lymphocytes and plasma cells. Parasitologic examination of the lungs by soaking in heated saline did not reveal any worms.
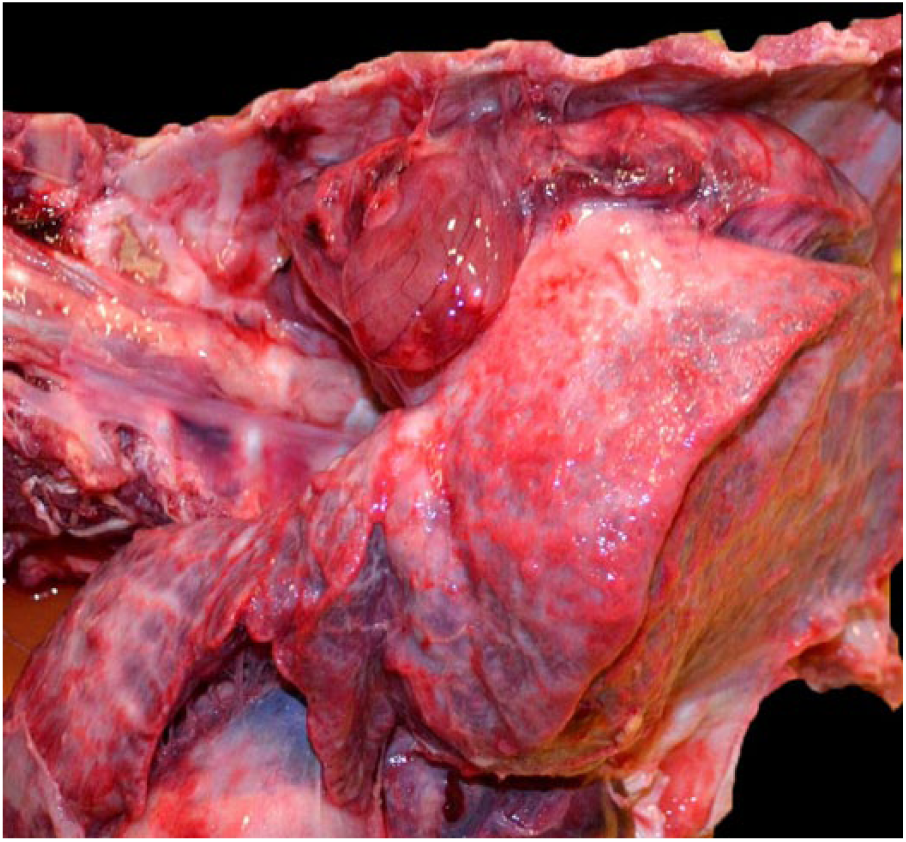
A large granulomatous mass in the lung is attached to the ribs of a 20-mo-old Vietnamese potbellied pig, case 2.
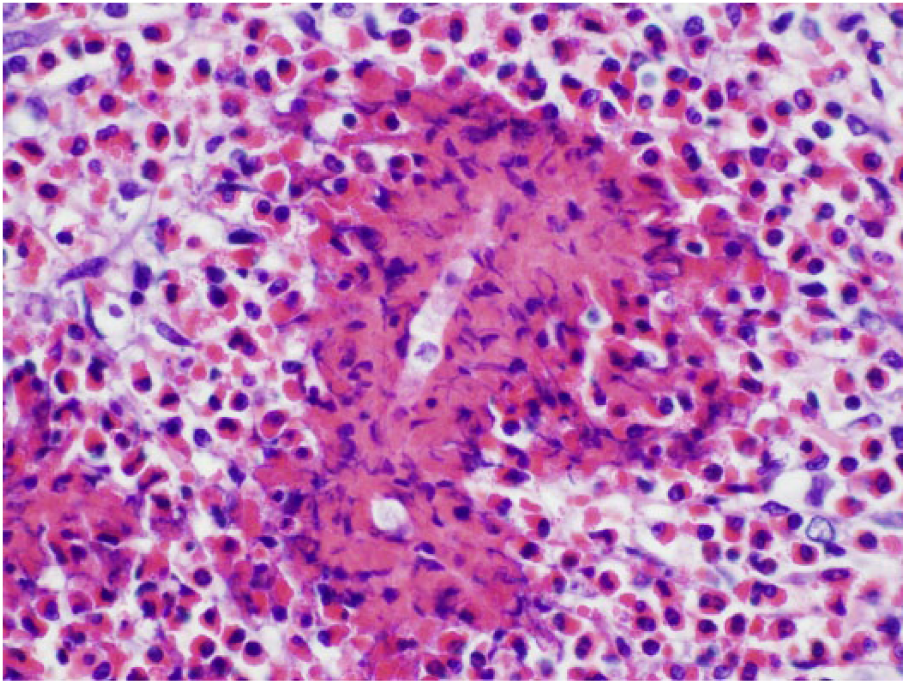
Microscopic evaluation of the granulomatous mass has numerous Mucor circinelloides hyphae surrounded by Splendore-Hoeppli–like reaction.
Sections of lung and tracheobronchial lymph node were plated on fungal agars, Sabouraud dextrose agar with chloramphenicol (Thermo Fisher Scientific, Remel Products, Lenexa, KS), and mycobiotic agar with cycloheximide and chloramphenicol (Thermo Fisher) at an incubation temperature of 25°C for 14 d. Identification was by direct exam wet mount using lactophenol cotton blue stain (Thermo Fisher). Subculture was to potato dextrose agar for sporulation (Hardy Diagnostics, Santa Maria, CA). Microbiologic culture yielded Rhizopus sp. from lung and lymph node. Sections of frozen lung were sent to WADDL. After DNA extraction, a portion of the large subunit RNA gene (D1-D2 region) of the 28S ribosomal RNA gene was amplified by PCR using published universal fungal primers. 7 When this PCR amplicon was directly sequenced, the sequence most closely matched Mucor circinelloides (100% sequence identity [678/678] with GenBank accessions FM246460 and AM933557). Given some basic similarity in the microbiologic culture appearance of Rhizopus and Mucor, it is most likely that the agent cultured was actually Mucor. However, this could not be confirmed because the culture isolate was not sequenced.
The causative agents of the granulomatous and eosinophilic pneumonia in these 2 Vietnamese potbellied pigs were C. incongruus (order Entomophthorales) and M. circinelloides (order Mucorales). Organisms within these orders were previously classified under the phylum Zygomycota and thus collectively termed “zygomycetes.” However, phylum Zygomycota is now obsolete because phylogenetic studies have revealed that the phylum is polyphyletic. 10 Because the terms “zygomycete” and “zygomycosis” are entrenched in the literature, the terms, when used in our report, appear in quotation marks.
In the order Entomophthorales, principle genus Conidiobolus, reports of disease are most common in dogs, horses, and sheep, which develop rhinofacial and nasopharyngeal conidiobolomycosis. 16 Disease progression to fungal pneumonia is usually as a manifestation of disseminated infection. In one report with disseminated disease caused by C. incongruus in a deer, the lung was considered the primary focus of infection. 21 Conidiobolomycotic pneumonia as a stand-alone entity in the literature, however, is rare. In one case report, a dog was treated for conidiobolomycotic pneumonia and recovered. Diagnosis was based on culture from a transtracheal wash. Pulmonary involvement was assessed as primary based on a clinical work-up that ruled out fungal rhinitis. Although the animal recovered, it was not determined if there was systemic or disseminated infection. 6 In case 1 of our report, conidiobolomycosis was diagnosed only in the lung, and to our knowledge, entomophthoromycosis has not been previously reported in porcine lungs.
Order Mucorales includes genera Lichtheimia, Rhizopus, Mucor, and Rhizomucor. 10 Rhizopus spp. have been cultured from liver and lymph nodes in swine,2,3 and mycotic inflammation was also identified in lungs, liver, and kidneys by light microscopy. 2 Mucorales respiratory infections in animals are uncommon. Pulmonary mucormycosis has been documented in a horse (caused by Absidia corymbifera, now Lichtheimia corymbifera) 5 and a harp seal. 9 Rhinocerebral mucormycosis is a commonly reported form in humans 6 ; however, in our mucormycosis case (case 2), there was no clinical evidence of upper respiratory disease or gross evidence of pharyngeal or nasal involvement. Fungus was recovered on culture of the lymph node, indicating local dissemination of disease.
Our 2 cases had several similarities. Both pigs were young and clinically the patients had dyspnea, pleural effusion characterized by eosinophilic inflammation, and pulmonary masses. Unfortunately, there was minimal information regarding the environment of the piglets or the level of inbreeding, although the 2 pigs were not related. Grossly, both patients had pulmonary masses and tracheobronchial lymphadenomegaly, and the pulmonary masses were found to represent mycotic eosinophilic granulomas with abundant hyphae and SH-like material. The SH reaction is described as a perihyphal eosinophilic cuff 8 and is associated with various fungal organisms, bacteria, oomycetes, parasites, and some non-infectious hazards. 8 The light microscopic appearance has been reported as both amorphous 13 and granular. 13 The composition in most cases includes immunoglobulins (antigen–antibody complexes), fibrin, and tissue debris 15 ; however, some cuffs have a major component of eosinophil major basic protein. 12 The dense aggregation and coating of hyphae by eosinophil granules has been reported with Pythium insidiosum and called SH-like material.12,14 The thick eosinophilic sleeves in Conidiobolus and Mucor infections in these 2 potbellied pigs is similar. Some standard veterinary textbooks state that the eosinophilic cuff that surrounds individual hyphae is a characteristic histologic change observed in entomophthoraceous infections,20,22 but light microscopic features for Mucorales and Entomophthorales hyphae in our reports are virtually identical.
P. carinii incites an interstitial-to-bronchointerstitial mycotic pneumonia in immunocompromised patients. The fungal organisms in our 2 cases have no resemblance to P. carinii, nor is host reaction typical of that infection. 1 There is no confirmed or suspected immunosuppression as a result of coinfection or prior viral infection in the 2 pigs in our report. Lesions caused by porcine circovirus 2 were absent from the lymph nodes of these 2 pigs. Additional testing for viral pathogens was not performed. Consideration, however, must also be given to secondary stress factors such as dietary changes, inadequate diet, etc. Aspergillus spp. have been reported in porcine lungs 19 ; however, the hyphae have uniform width, are regularly septate, and have dichotomous branching at acute angles, whereas “zygomycetes” have nonparallel walls, are infrequently septate, and have non-dichotomous, irregular branching.
Organisms in the order Entomophthorales and Mucorales should be considered in the differential list for primary fungal pneumonia, particularly in young Vietnamese pot-bellied pigs with eosinophilic inflammation in thoracic effusions. Our report adds to the literature base with gross and microscopic descriptions and definitive identification via DNA extraction, amplification of fungal DNA, sequencing, and molecular identification. We also documented the SH-like reaction in a clinical case caused by Mucorales fungi, indicating that this material is not a reliable way to differentiate between Mucorales and Entomophthorales on light microscopy.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
