Abstract
A 12-year-old Simmental cow was presented with a moderately firm irregular whitish mass of approximately 5 cm in diameter, occupying the right orbit. Microscopically, a poorly differentiated neoplasm was observed. The immunohistochemical panel included cytokeratins, vimentin, epithelial membrane antigen, Factor VIII, CD34, Mart-1, Melan A, smooth muscle actin, desmin, chromogranin, neuron-specific enolase, S-100 protein, and MIB-1. The neoplasm was negative for all of them, with the exception of vimentin and S-100 protein. Transmission electron microscopy revealed abundant desmosomes. These findings support the diagnosis of orbital (retrobulbar) meningioma.
Keywords
Meningiomas are derived from meningothelial cells, particularly of the arachnoid and pia mater; therefore, they are far more common in the central nervous system than in any other location in the body. Meningioma is the most common tumor of the central nervous system in dogs and cats, but it is considered rare in sheep, horses, and cattle. 7 In the dog, which has the higher incidence among the domestic species, 82% of the meningiomas are intracranial, 15% are intraspinal, and the remaining 3% are retrobulbar or orbital. 7
Approximately 5–10% of all human orbital tumors are meningiomas of the optic nerve. 10, 11 Several cases of orbital meningioma have been reported in the dog. Clinically, they are frequently associated with unilateral protrusion of the ocular globe and blindness. Apparently there is no predilection for either side. 2– 4, 8, 9 Although most of the reported cases do not metastasize, a few of these cases were associated with pulmonary metastasis. 3, 4 Extension of a retrobulbar meningioma to the chiasmal area with extension to the thalamus and to the optic disc and sclera has been reported, 2 as well as the presence of osseous or cartilaginous tissue within the neoplasm. 2– 4 In cattle, there have been a few cases of meningioma in the central nervous system. 5 During the course of preparation of this article, a case of extracranial meningioma with features of orbital meningioma was described in a Holstein cow in Japan. 12
A 12-year-old Simmental cow, weighing 700 kg, was presented with exophthalmia and a moderately firm irregular whitish mass of approximately 5 cm in diameter, occupying the right orbit (Fig. 1). The right orbital mass was exenterated after a clinical presumptive diagnosis of neoplasia. During the period of 5 months that followed the surgery, the cow did not have any sign of recurrence or metastasis as well as no clinical signs of neurologic disease.
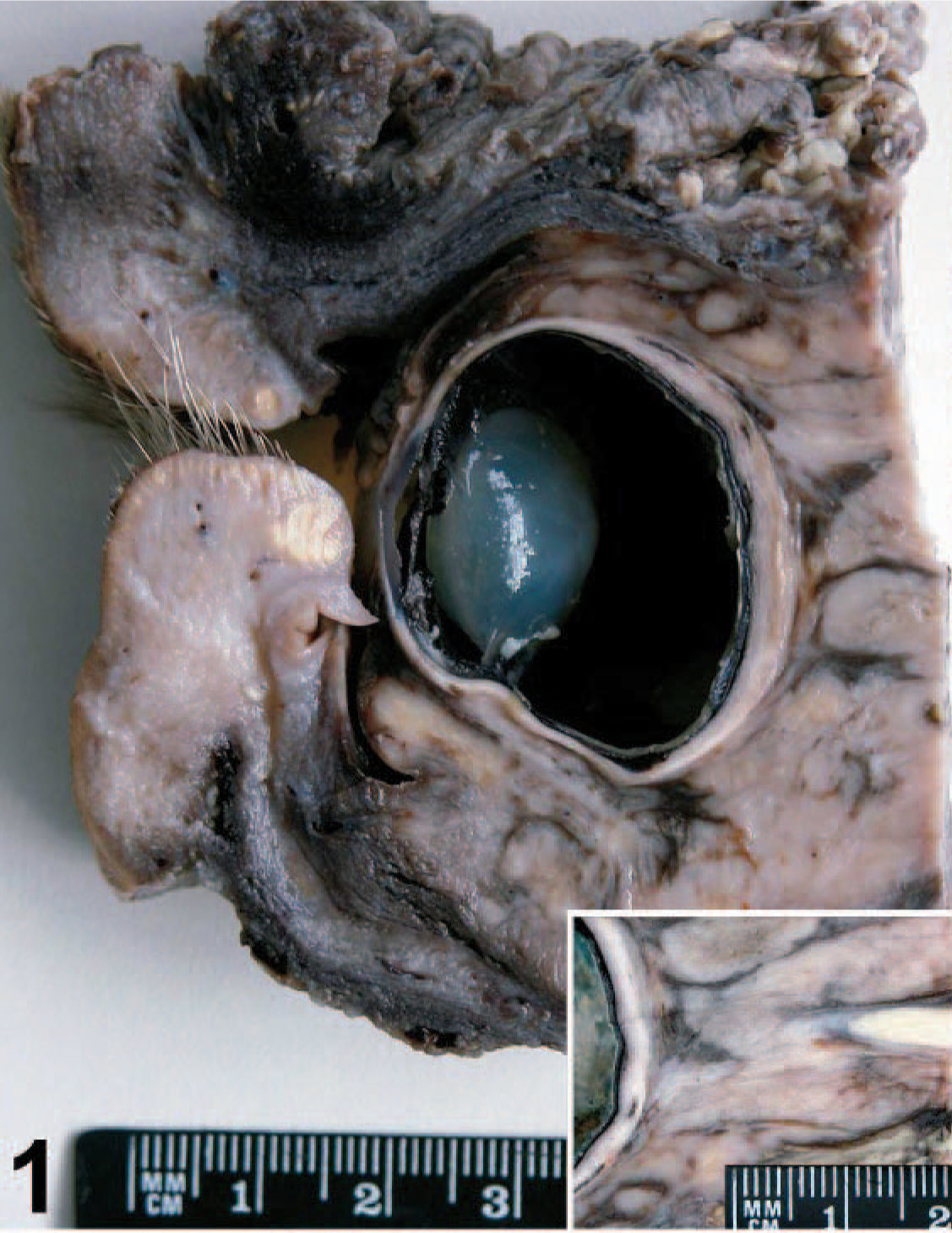
Orbital meningioma; cow. Tan retrobulbar neoplastic mass occupies the orbital cavity. Inset: retrobulbar neoplastic tissue surrounding the optic nerve without invasion of the nerve.
Fragments of the neoplastic tissue and retrobulbar structures were fixed by immersion in 10% buffered formalin, embedded in paraffin, cut at 5 μm, and stained with HE, periodic acid–Schiff stain (PAS), Masson trichrome, and picrosirius. Three-micrometer sections were mounted onto silane-coated slides for immunohistochemical staining with the streptavidin-peroxidase complex (LSAB+ Kit, Dako, Carpinteria, CA, USA) and diaminobenzidin substrate. Antibodies and details of the immunohistochemistry protocol are summarized in Table 1. Formalin-fixed fragments of the tumor were transferred to 2.0% paraformaldehyde and 2.5% glutaraldehyde in 0.08-M Sorensen's phosphate buffer at pH 7.2 and further processed for transmission electron microscopy.
Antibodies and protocol used for immunohistochemistry.∗
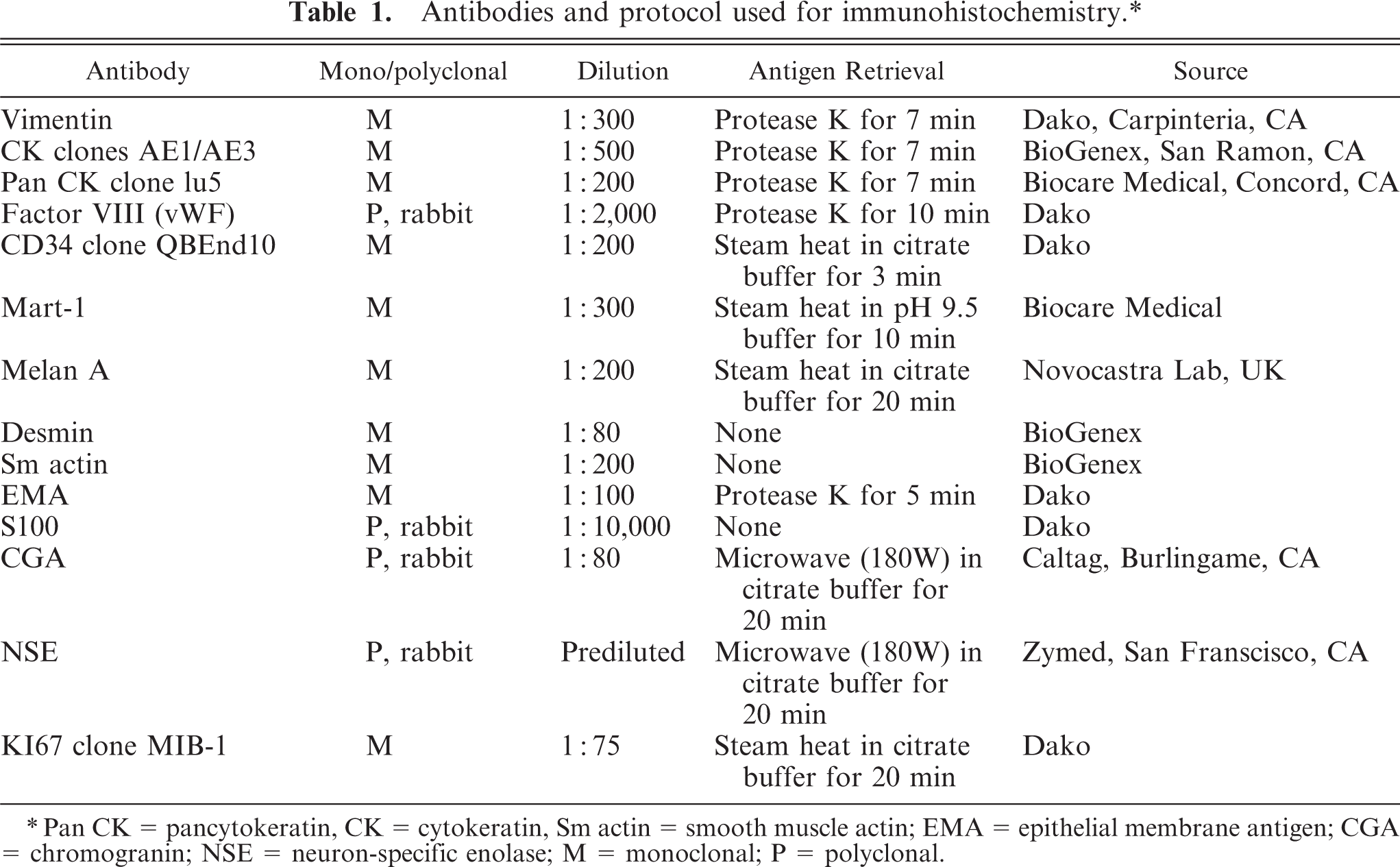
∗Pan CK = pancytokeratin, CK = cytokeratin, Sm actin = smooth muscle actin; EMA = epithelial membrane antigen; CGA = chromogranin; NSE = neuron-specific enolase; M = monoclonal; P = polyclonal.
Microscopically, the orbital mass was composed of an invasive, nonencapsulated, and poorly delimitated neoplastic tissue. The neoplastic cells were poorly differentiated, mostly polygonal with some spindle-shaped cells and arranged in sheets (Fig. 2); some areas had a tubule-papillar pattern, forming structures similar to vascular channels containing no cells, supported by moderately abundant fibrovascular stroma. The neoplastic tissue was located around the optic nerve without invading the nerve fibers (Fig. 3) but with extensive invasion of the adjacent retrobulbar muscular tissue. The cells were highly anaplastic and pleomorphic, with a high nucleus/cytoplasm ratio. The nuclei were predominantly oval with vesicular chromatin and mostly 1 mildly prominent nucleolus, with a few multinucleated cells. Neoplastic cells had a high mitotic rate characterized by multiple mitotic figures per high-magnification microscopic field. MIB-1-positive cells corresponded to approximately 14.2% of the neoplastic cell population (a total of 7,731 cells were counted), which is similar to proliferation indexes reported for anaplastic human meningiomas. 1 There was a diffuse lymphocytic infiltrate among the neoplastic cells, multifocal small areas of necrosis, and mild multifocal hemorrhage. On the basis of the morphologic features, a presumptive diagnosis of orbital meningioma was established, but because of the poor differentiation of this neoplasm, an undifferentiated carcinoma and an endothelial sarcoma, possibly of lymphatic origin, were considered in the differential diagnosis. Some neoplastic cells had PAS-positive intracytoplasmic granules, and Masson trichrome evidenced a small amount of collagen fibers among the cells, with almost exclusively type III collagen as demonstrated in sections stained with picrosirius under polarized light.
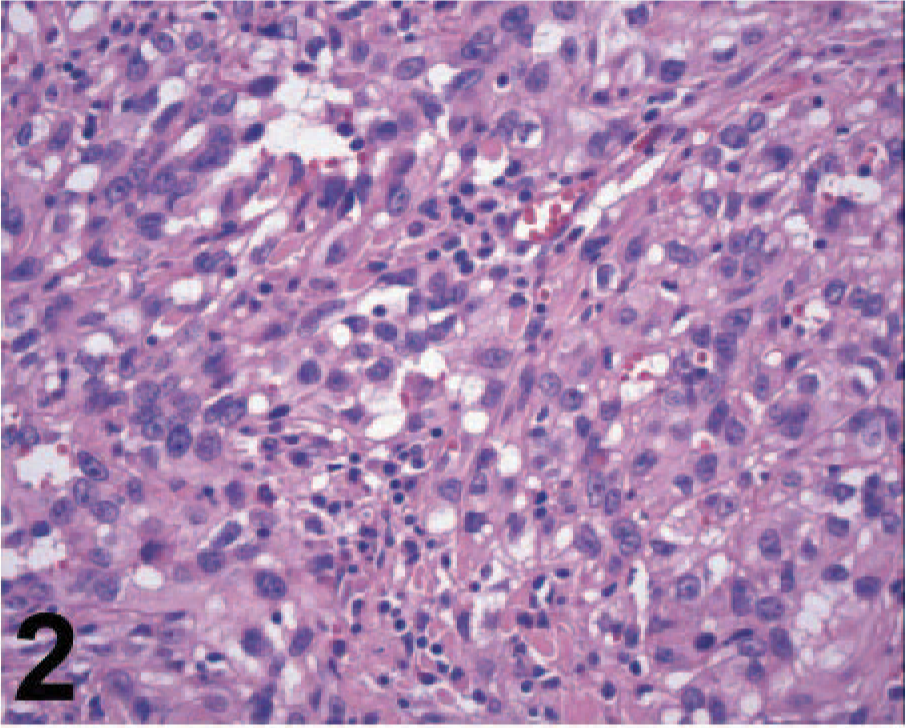
Orbital meningioma; cow. Anaplastic meningothelial cells have diffuse lymphocytic infiltrate. HE.
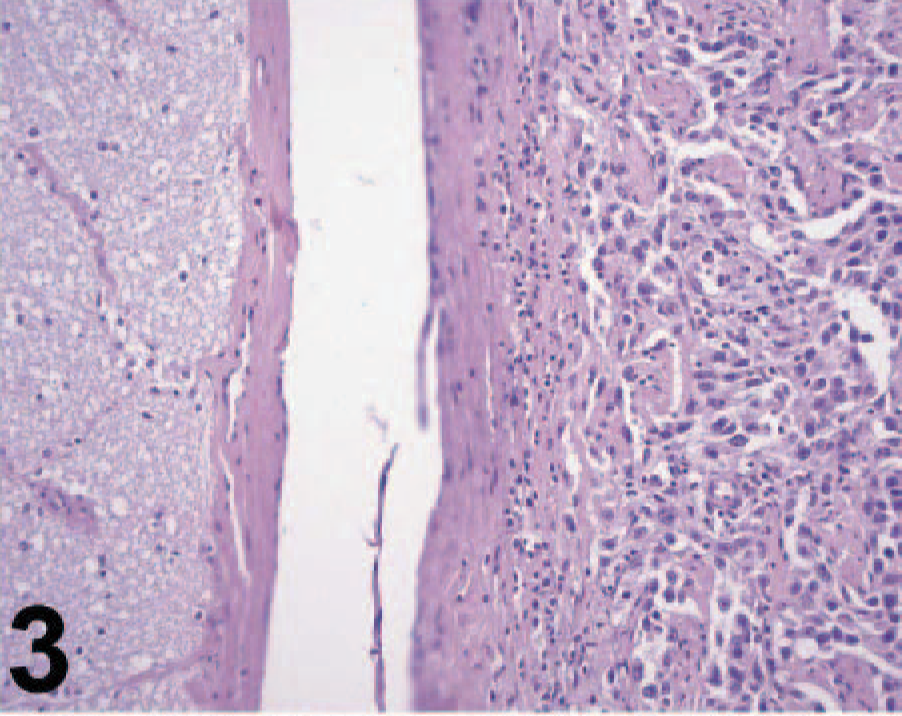
Orbital meningioma; cow. Neoplastic tissue surrounds the optic nerve. HE.
The immunohistochemical panel included cytokeratins, vimentin, epithelial membrane antigen (EMA), CD34, Factor VIII, Mart-1, Melan A, smooth muscle actin, desmin, chromogranin, neuron-specific enolase, and S-100 protein. This tumor was negative for all these markers, with the exception of vimentin (Fig. 4) and S-100 protein. Transmission electron microscopy revealed abundant desmosomes (Fig. 5). Although human meningiomas are positive for EMA, the antibody does not detect this antigen in canine tissues 7 and possibly not in cattle since sections of bovine kidney did not stain with this antibody, while human renal epithelial cells stained in the positive control section. Because of the lack of a specific marker for meningiomas in cattle and the poorly differentiated morphology of this tumor, our diagnostic strategy was largely based on the elimination of other possible poorly differentiated neoplasms such as carcinomas (negative for cytokeratins), endothelial sarcomas (negative for factor VIII and CD34), malignant melanomas (negative for Mart-1 and Melan A), sarcomas of muscular origin (negative for smooth muscle actin and desmin), neuroendocrine tumors (negative for chromogranin), and neuroectodermal tumors (negative for neuron-specific enolase). Taken together, these findings support the diagnosis of orbital meningioma. Several morphologic subtypes of meningioma have been recognized in domestic animals. The current classification includes the following subtypes: meningothelial, fibrous, transitional, psammomatous, angiomatous, papillary, granular cell, myxoid, and anaplastic. 6, 7 Because of the morphologic features, the present case can be classified as an orbital (retrobulbar) anaplastic meningioma. In spite of its malignant histologic appearance, neither recurrence nor metastasis was noticed up to 5 months after surgical removal of the tumor.
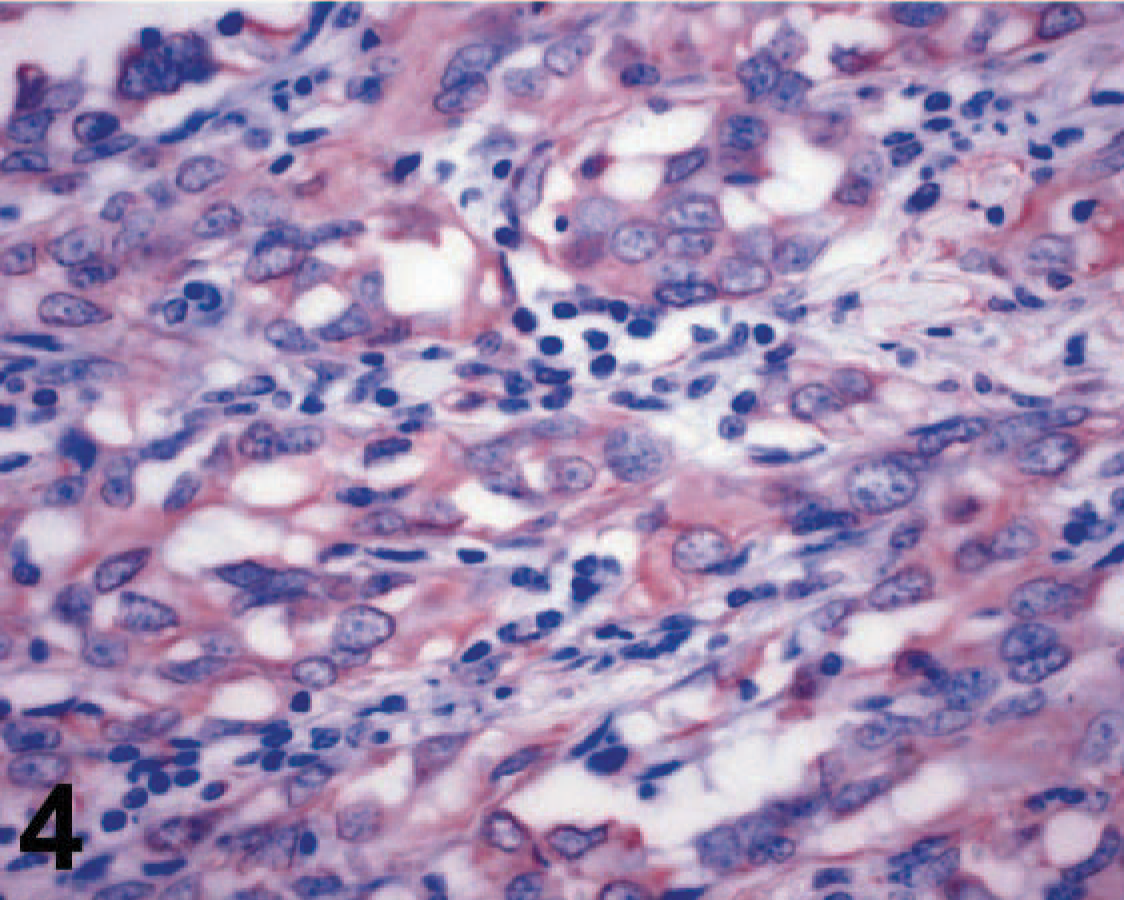
Orbital meningioma; cow. Diffusely positive immune staining for vimentin. Streptavidin-peroxidase complex.
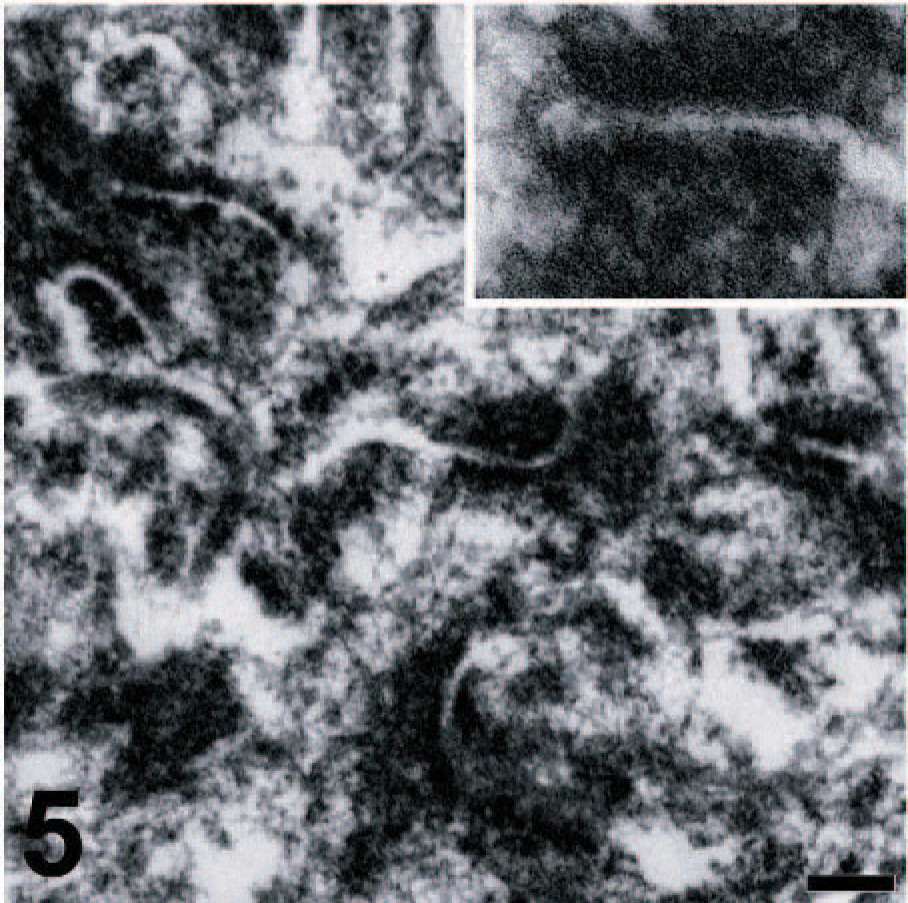
Orbital meningioma; cow. Abundant desmosomes are seen between adjacent neoplastic cells. Inset: detail of desmosome cell junction. Transmission electron microscopy. Bar = 0.5 μm.
Footnotes
Acknowledgements
We thank G. Adamson (Electron Microscopy Laboratory, Department of Medical Pathology and Laboratory Medicine-UC Davis) and D. K. Naydan (Department of Pathology, Microbiology and Immunology, School of Veterinary Medicine, UC-Davis) for technical assistance and Dr. A. J. Baumler for supporting the microscopic analysis performed by RLS during his sabbatical at UC Davis.
