Abstract
The cause of death in 215 cattle on 20 long-haul live export voyages from Australia to the Middle East, Russia, and China was investigated between 2010 and 2012 using gross, histologic, and/or molecular pathology techniques. A quantitative reverse transcription polymerase chain reaction (qRT-PCR) assay was used to detect nucleic acids from viruses and bacteria known to be associated with respiratory disease in cattle: Bovine coronavirus (Betacoronavirus 1), Bovine herpesvirus 1, Bovine viral diarrhea virus 1 and 2, Bovine respiratory syncytial virus, Bovine parainfluenza virus 3, Histophilus somni, Mycoplasma bovis, Mannheimia haemolytica, and Pasteurella multocida. The most commonly diagnosed cause of death was respiratory disease (107/180, 59.4%), followed by lameness (n = 22, 12.2%), ketosis (n = 12, 6.7%), septicemia (n = 11, 6.1%), and enteric disease (n = 10, 5.6%). Two thirds (130/195) of animals from which lung samples were collected had histologic changes and/or positive qRT-PCR results indicative of infectious lung disease: 93 out of 130 (72%) had evidence of bacterial infection, 4 (3%) had viral infection, and 29 (22%) had mixed bacterial and viral infections, and for 4 (3%) the causative organism could not be identified. Bovine coronavirus was detected in up to 13% of cattle tested, and this finding is likely to have important implications for the management and treatment of respiratory disease in live export cattle. Results from the current study indicate that although overall mortality during live export voyages is low, further research into risk factors for developing respiratory disease is required.
Introduction
Since the late 1990s, Australia has exported 600,000–985,000 live cattle each year to countries around the globe. Between 2010 and 2012, approximately 65% of cattle have been exported to countries in Southeast Asia and the Pacific with approximately 90% of these destined for Indonesia. Approximately 15% of cattle are exported to countries in the Middle East and North Africa, 10% to Asia (including China, Japan, Pakistan, and India), and 10% to countries near the Black Sea (Turkey and the Russian Federation; LiveCorp: 2012, Cattle live export: facts & stats. Available at: https://www.livecorp.com.au/sites/default/files/content_files/cattle_statistics_2010-12.pdf).
Between 2000 and 2011, the average mortality rate per voyage in cattle exported live from Australia was 0.14% (Department of Agriculture Fisheries and Forestry [DAFF], Australian Government: 2013, Livestock mortalities for exports by sea. Available at: http://www.daff.gov.au/animal-plant-health/welfare/export-trade/mortalities). However, because of the large numbers of animals exported each year and the relatively high value of each animal (approximately AUD $1,000 per head in 2012; Meat & Livestock Australia: 2012, Australian live export statistical review 2012. Available at: http://www.mla.com.au/files/f1d10d44-8433-4955-bedd-a18900f09cfc/Australian-livestock-export-industry-statistical-review_2012.pdf), even low mortality rates can have a significant economic impact.
Mortality rates and animal welfare are closely linked; increased mortality is often interpreted as an indicator of poor welfare. 26 Increased public awareness and concern for animal welfare issues creates ongoing pressure on the live export industry to demonstrate that it is actively committed to minimizing mortality rates and improving animal welfare. Previous studies on cattle deaths during sea transport from Australia to the Middle East 31 and Australia to Japan 20 identified heat stroke, trauma and/or lameness, and respiratory disease as the 3 most common causes of mortality. Heat stress is a recognized problem on export vessels for a number of reasons, including stocking densities on board the ship, high ambient temperature and humidity particularly in the Equatorial region and in waters around the Arabian peninsula (Gulf of Aden, Red Sea, Gulf of Oman, Persian Sea), and reliance on artificial ventilation especially on closed decks. 31
Bovine respiratory disease (BRD) has a multifactorial, polymicrobial etiology. Organisms known to be associated with BRD include viruses and bacteria: Bovine coronavirus (BoCV; Betacoronavirus 1), Bovine parainfluenza 3 virus (BPIV-3), Bovine herpesvirus 1 (BoHV-1), Bovine respiratory syncytial virus (BRSV), Bovine viral diarrhea virus 1 and 2 (BVDV), Arcanobacterium pyogenes, and Bibersteinia (formerly Pasteurella) trehalose, Histophilus somni, Mycoplasma bovis, Mannheimia haemolytica, and Pasteurella multocida. 33 In an effort to reduce the risk of cattle developing BRD during a voyage, cattle are often vaccinated against specific respiratory disease pathogens and/or treated with broad spectrum antibiotics prior to being loaded onto the vessel, and may be treated again before they reach their destination. These treatments may be required by the importing country or, with regard to antibiotics, applied at the exporter’s discretion to animals that they believe are at greater risk of developing BRD. During a voyage, treatments may be administered to animals showing clinical signs of disease or to animals considered at risk of developing disease (e.g., pen mates of sick animals). There is little data to document unequivocally whether any of these practices are effective at reducing morbidity or mortality.
The current study was initiated to determine the causes of mortality in live export cattle on long-haul voyages from Australia, based on necropsy reports and samples collected from study voyages. In addition, the study aimed to identify infectious agents involved in BRD in Australian live export cattle to inform future preventative treatment and management strategies.
Materials and methods
Twenty eligible voyages were enrolled during the period March 2010 to September 2012. Eligible voyages were defined as those loading cattle in Australia for a long-haul voyage (≥10 days) to destinations including the Middle East (Israel, Saudi Arabia, Bahrain, Turkey), North Africa (Egypt, Libya), and the Russian Federation, and carrying at least 1,000 head of cattle.
Data and sample collection
For 19 out of 20 voyages, data and sample collection was performed by the Australian Quarantine and Inspection Service (AQIS; now DAFF) Accredited Veterinarian (AA Vet) accompanying the voyage. A standardized protocol was provided, and all participating AA Vets received training in the data and sample collection protocols. Necropsy equipment, containers for collecting samples, and paper or electronic templates for recording observations were all provided on board each ship. The AA Vets accompanying the voyages were encouraged to collect a defined set of data and samples from each animal that died, but this was not always possible because of the demands of other tasks and duties that they are routinely expected to perform during voyages. For 1 out of the 20 voyages, data and sample collection was performed by a member of the research team (SJ Moore).
Standardized data recording forms were used to collect animal and epidemiologic data for each animal that died. Data included the animal’s location on the ship, animal characteristics (including visual ear tag, electronic identification tag, breed, and weight), clinical signs displayed before death, risk factors or events that may have contributed to death, gross necropsy findings if available, and a preliminary cause of death.
Necropsy samples included fresh tissue samples collected into 10% buffered formalin at a maximum ratio of 1 part tissue to 10 parts formalin, and tissue samples (approximately 5 mm2) and/or swabs collected into a 5-ml plastic screw top container filled with 2 ml of viral transport media (VTM; Hanks balanced salt solution, penicillin G [1,000 units/ml], streptomycin [25 mg/ml], and amphotericin B [0.1 mg/ml]; Department of Agriculture and Food, Western Australia) and frozen.
The number and type of samples collected at necropsy depended on the animal’s clinical signs prior to death and gross necropsy findings. Core samples collected from all animals were: lung (grossly normal and abnormal), trachea, heart, ileocecal junction, kidney, liver, and rumen into 10% buffered formalin, and nasal and lung swabs. When the animal showed clinical signs prior to death that were suggestive of a specific disease, which was confirmed at necropsy, then a range of additional samples were collected according to the suspected disease. When there was no obvious cause of morbidity/mortality and the cause of death could not be determined from gross necropsy findings then the core samples plus fixed skeletal muscle, reticulum, abomasum, small intestine, large intestine, pancreas, mesenteric lymph node, gall bladder, spleen, adrenal gland, and the brainstem and cervical spinal cord were collected into 10% buffered formalin.
Samples and data collection forms remained on the ship until the next time it berthed in Fremantle (Western Australia). Forms and samples were removed from the ship and taken directly to the quarantine approved premises at the Department of Agriculture and Food, Western Australia, for processing.
Processing of fixed tissues
Representative samples were taken from each fixed tissue and processed routinely for embedding in paraffin wax. Histologic sections were cut at 5 μm, stained with hematoxylin and eosin, and examined for pathologic changes using light microscopy.
Processing of samples in VTM
Tubes containing samples in VTM were thawed, vortexed, and allowed to settle for approximately 15 min. A 200-μl aliquot of VTM was taken and placed into a 96-well plate. a Both the original sample tubes and 96-well plates were stored at −80°C until required. Nucleic acid extraction from VTM aliquots was performed using a commercial kit b on a magnetic particle processor c according to the manufacturer’s instructions. Plates containing the eluate were stored at −80°C until required.
Detection of microbial nucleic acids
Quantitative reverse transcription polymerase chain reaction (qRT-PCR) was used to detect nucleic acids from organisms commonly associated with BRD. 33 The qRT-PCR was performed using a commercial kit d according to the manufacturer’s instructions.
Primer and probe sequences were sourced from available sequences for BoCV (data not shown), BoHV-1, 29 BPIV-3, 23 BRSV (Mahony TJ, Horwood PF: 2006, Rapid detection of bovine respiratory disease pathogens. Final report for Meat & Livestock Australia project FLOT.219. Available at: http://www.mla.com.au/Research-and-development/Final-report-details?projectid=1063), BVDV, 23 and the following 3 bacteria (Mahony TJ, Horwood PF: 2006): M. haemolytica, P. multocida, and H. somni. Mycoplasma bovis primer sequences were sourced from a previous study, 6 and the probe sequence was designed using commercially available software. e Primer and probe sequences can be found in Supplementary Table 1. To reduce the number of tests, required reactions were multiplexed as follows: BoCV and BPIV-3, BRSV and BVDV, M. haemolytica and P. multocida, H. somni and M. bovis. Bovine herpesvirus 1 was run as a single assay.
The final optimized multiplex qRT-PCR assays were as follows. All assays consisted of a 20-μl reaction mix containing 2 μl of the nucleic acid sample, 4 μl of proprietary master mix, 0.4 μl of ROX (5-carboxy-X-rhodamine) dye solution, and 0.2 μl of RT mix. d Primer and probe concentrations varied according to the assay. For BoHV-1, BoCV and BPIV-3, BRSV and BVDV, and M. haemolytica and P. multocida, 400 nM of each primer and 200 nM of probe were used. For H. somni and M. bovis, 100 nM of each H. somni primer and 50 nM of probe were used, and 400 nM of each M. bovis primer and 200 nM of probe. The cycling parameters were as follows: 50°C for 20 min, 95°C for 5 min, followed by 45 cycles of 95°C for 15 sec and 60°C for 45 sec.
Each run contained duplicate samples of a positive control (either a virus isolate or a clinical extract that had previously been characterized), a negative control (an extract of cell culture–grade fetal bovine serum), and a blank (PCR-grade water). Runs were only considered valid if the positive control was amplified at the expected threshold cycle, and the negative and blank controls showed no amplification.
Interpretation of qRT-PCR results
Positive samples were those with a characteristic sigmoidal curve similar to the positive control, crossing the threshold before 40 cycles. Negative samples were those with no characteristic sigmoidal curve. Samples crossing the threshold after 40 cycles were regarded as suspicious for containing the nucleic acid of interest and were retested before classifying as negative or positive.
Determining the cause of death
The final diagnosis for each animal was based on the combination of all available data: description of premortem clinical signs, gross necropsy findings, histology results, and qRT-PCR results. Not all data was available for all animals. Respiratory disease was considered the cause of death if there was cranioventral consolidation of more than 30% of the lung 14 and/or the AA Vet had assigned a preliminary diagnosis of pneumonia/BRD and there were histologic findings of moderate to severe pneumonia in lung tissue.
Heat stroke was considered the cause of death if the animal was noted to be panting heavily prior to death or was found dead with no previous clinical signs, the core body temperature (CBT) was ≥43°C (measured using a deep temperature probe), and necropsy findings were consistent with those described previously for heat stroke in live export cattle 31 : eyes sunken, muscles glowing pink and dry rather than red-brown and moist, heart tightly contracted with epicardial ecchymoses, severe acute diffuse pulmonary congestion, and edema causing the lungs and mucosa of the trachea and bronchi to be dark red. Ambient environmental temperatures were not recorded.
Statistical analysis
Statistical software package R was used for data analyses. 35 Unadjusted mortality rates were calculated as the cumulative incidence of deaths during the voyage with a denominator representing the total count of cattle loaded onto the ship at the port(s) of loading, and a numerator representing the count of deaths observed for the entire voyage (from first day of loading to last day of discharge). Adjusted mortality rates were estimated using Poisson regression with models incorporating an offset based on total cattle-days at risk during the voyage to adjust for variation in numbers of cattle and voyage duration.
Poisson modeling was used to screen for associations between mortality rate and relevant explanatory variables (Supplementary Table 2). Because of sparseness of the data and the presence of confounding between many explanatory variables, Poisson models were run as univariable screening models only to assess for associations between explanatory variables and voyage mortality rate. Additional analyses were conducted using linear regression for continuous outcomes (CBT, necropsy sampling rate, severity of myocardial degeneration) and logistic regression for binary outcomes (pathogen prevalence in nasal or lung samples) to explore associations with various explanatory variables, including cause of death, that were present in the dataset. Spearman rank correlation test was used to test for correlations between the presence of various organisms, as detected by qPCR, in the nasal secretions and lung. A threshold of P = 0.05 was used for all statistical analyses.
Results
Voyage data
During the study period of March 2010 to June 2012, there were 85 voyages to the Middle East and Russian Federation of which 59 were carrying more than 1,000 head of cattle. Most voyages took place in the autumn (17/59, 29%) with approximately equal numbers during the spring (n = 14), summer (n = 13), and winter (n = 15). The average total voyage length for the 85 voyages during the study period was 25.3 ± 6.8 days (sailing plus discharge, expressed as mean ± standard error).
Necropsy samples were collected from 20 out of 59 (34%) eligible voyages during the study period (Table 1). The majority of study voyages took place in the southern hemisphere in autumn (n = 8 voyages) and winter (n = 5) with fewer voyages in summer (n = 4) and spring (n = 3). Seven ships were used to carry livestock with each ship making 1–5 voyages over the study period. The average total voyage length for the 20 eligible voyages was 27.4 ± 7.0 days.
Voyage data. Voyage number 18 is not included in this table because it was not relevant to this phase of the study.
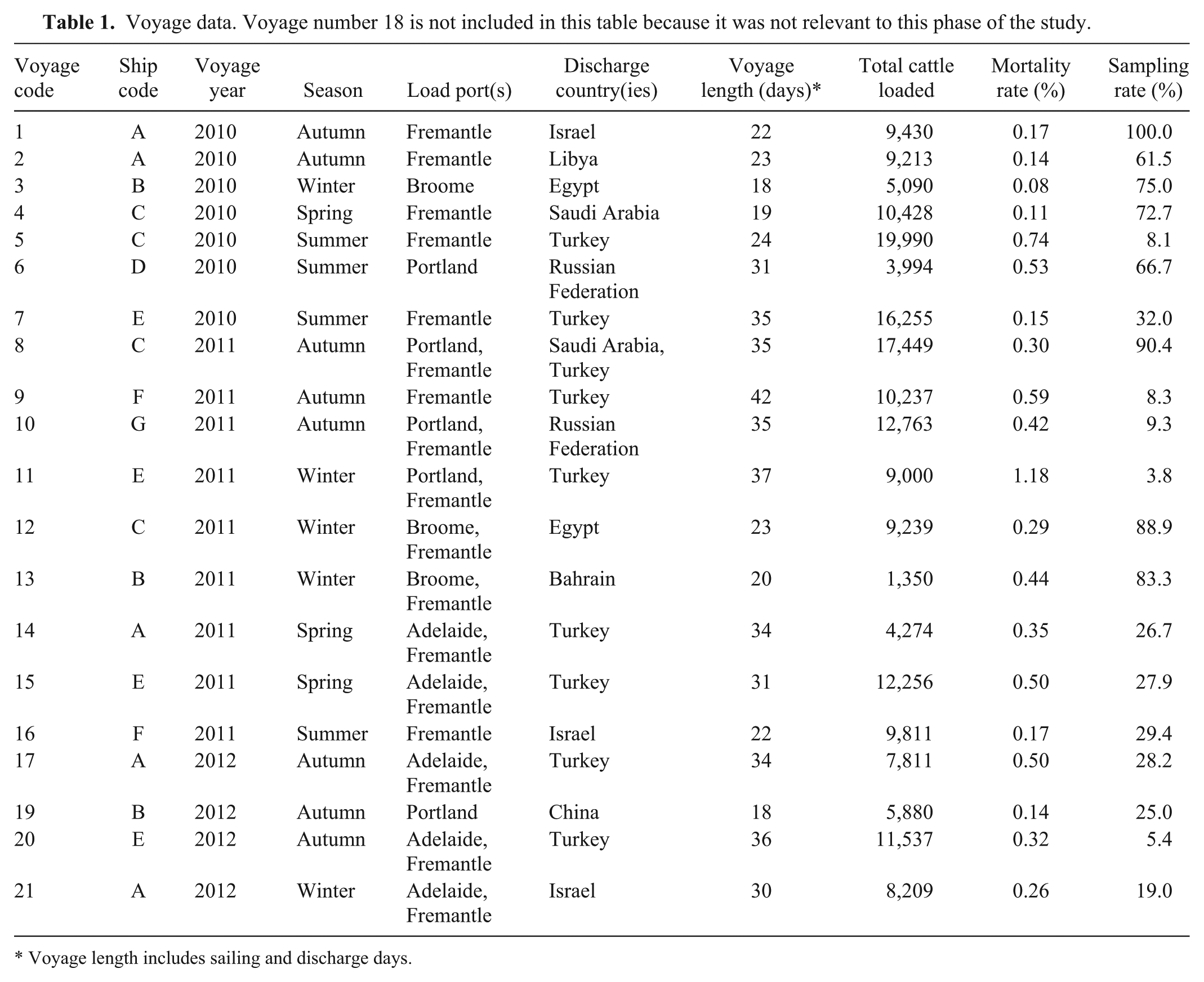
Voyage length includes sailing and discharge days.
Cattle were loaded out of 4 ports across 3 states: Fremantle and Broome in Western Australia, Portland in Victoria, and Adelaide in South Australia. The majority of cattle were loaded out of the southern port of Fremantle (Table 2). Cattle from southern ports (Portland, Adelaide, Fremantle) tend to be primarily of the Bos taurus subspecies while those from the northern port (Broome) are primarily Bos indicus. 9
Loading and discharge data, including the type of loading (single vs. split) and the number of cattle loaded and discharged at each port.
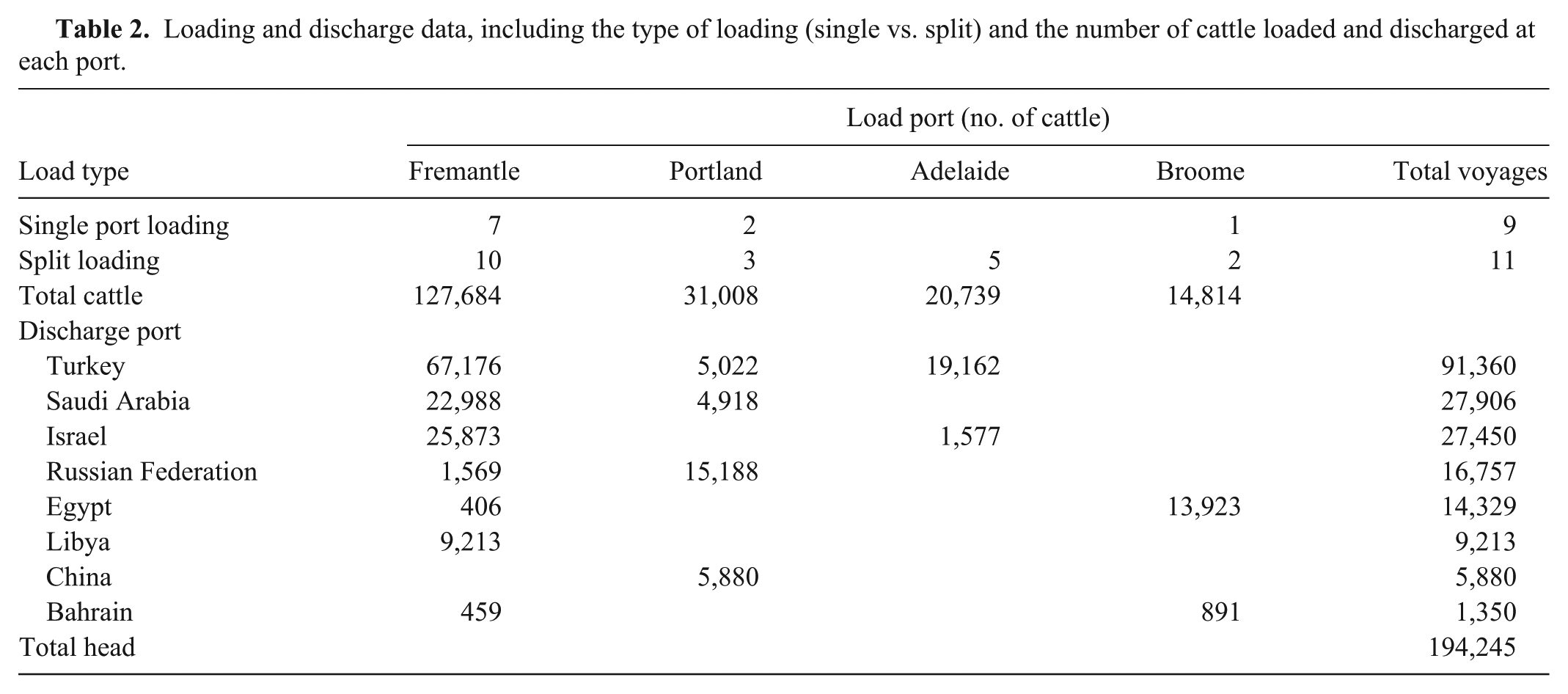
Cattle were loaded out of a single port for 10 voyages while split loadings were used on the remaining 10 voyages. For split loadings, cattle were loaded at Broome, Portland, or Adelaide and then the ship sailed 4–5 days to Fremantle where additional cattle were loaded. All voyages out of Adelaide were split loaded while the remaining ports had a mixture of single and split loadings. The total number of cattle on a fully loaded ship varied from 3,994 to 19,990 head (Table 1).
With the exception of a single voyage, cattle were discharged at ports in 2 broad geographic zones: the Middle East and North Africa including Israel, Saudi Arabia, Bahrain, Libya, and Egypt, and the Black Sea zone including Turkey and the western Russian Federation (Table 2). Turkey was the primary discharge country taking 47% of animals. Saudi Arabia and Israel accounted for approximately 14% each.
Mortality
The unadjusted average of voyage-specific cumulative incidence estimates was 0.37% (range: 0.08–1.18%). With the exception of voyage 11, the mortality rates for all study voyages were below the reportable level of 1% 9 (Table 1). The average adjusted mortality rate derived from Poisson regression modeling was 0.125 deaths per thousand cattle-days (95% confidence interval [CI]: 0.121–0.139). Adjusted mortality rates were significantly associated with voyage (P < 0.001) and the risk of mortality was higher in summer and winter than in spring and autumn (P < 0.001).
No significant differences were detected between mortality rate for single versus split loadings (P = 0.175) or loadings out of southern (Fremantle, Adelaide, Portland) versus northern (Broome) ports (P = 0.651). The mortality rate for voyages loading out of eastern ports (Adelaide, Portland) was significantly higher than western ports (Fremantle, Broome; P < 0.01) and this was because the mortality rate for voyages loading out of Portland was significantly higher than all other ports of loading (P < 0.01).
The mortality rate for voyages discharging in Turkey and the Russian Federation was significantly higher than for countries in the Middle East and North Africa (Bahrain, Egypt, Israel, Libya, Saudi Arabia) or China (P < 0.001). The mortality rate for split discharges was lower than for single port discharges (relative risk = 0.69, 95% CI: 0.58–0.82, P < 0.001).
Clinical presentation
Necropsy reports and/or fixed tissue samples were received from 215 of the 741 deaths (29%) that occurred on the study voyages. Animal data, a description of clinical signs, a description of necropsy findings, and/or fixed or frozen tissue were not received from the remaining 526 animals so these animals were not included in the study. Of the 215 animals, 135 (63%) were recorded as found dead and 57 (26%) as euthanized. The circumstances of death were not recorded for the remaining 23 (11%; Table 3).
Clinical category and circumstance of death for animals from which epidemiologic data and/or necropsy samples were received.

Clinical signs recorded by the AA Vets were grouped into 8 clinical categories: no previous clinical signs, lameness, weakness and/or recumbency, ill-thrift and/or shy feeders, respiratory signs, neurologic signs, misadventure, and miscellaneous. Of the 134 animals that were found dead and for which clinical data was available, 79 (59%) were recorded as having shown no previous clinical signs. Of the remaining 55 animals, 50 animals had been hospitalized, and the most common reasons for this were ill-thrift and/or shy feeders (32%), respiratory signs (22%), lameness (20%), and weakness and/or recumbency (16%; Table 3). Five were found dead with their head stuck under a pen rail (misadventure) and were assumed to have died from circulatory collapse and asphyxiation as a result of being cast.
Fifty-seven animals were euthanized, 20 (35.1%) of these for weakness and/or recumbency and 19 (33.3%) for lameness that was not responsive to treatment (Table 3). Seven animals were recorded as having neurologic signs prior to euthanasia. Two animals presented with respiratory signs that were not responsive to treatment. Of the 2 animals euthanized due to misadventure, 1 went down in the race during discharge and would not stand up again, and another was found trapped in a gate. Four animals that had been sedated for treatment of leg wounds failed to recover from the anesthetic.
Gross pathology
Gross necropsies were performed on 197 of the 741 deaths (27%) that occurred on the study voyages, representing 92% of the 215 animals that formed the study population.
Core body temperature was measured using a deep probe thermometer in 97 animals across 14 voyages. Twelve animals were found to be hyperthermic (CBT ≥43°C) but only 2 animals were assigned a final diagnosis of primary hyperthermia (i.e., heat stroke). Hyperthermia secondary to moderate-to-severe respiratory disease was identified in 9 animals. One case was found dead with no previous clinical signs, and no evidence of respiratory or other disease, including heat stroke, could be found. Overall, there was a significant association between CBT and time since death (P < 0.001), and this was found to be due to a drop in CBT at between 14 and 16 hr after death.
Gross pathology indicative of respiratory disease was confirmed in 12 (80%) of the 15 animals that were recorded as having clinical signs of respiratory disease prior to death. Two animals (13.3%) were found to have cardiovascular disease—1 with traumatic reticuloperitonitis (hardware disease) and 1 with severe restrictive pericarditis. One animal had an impacted abomasum.
Respiratory disease was the most common necropsy diagnosis for animals presenting with no previous clinical signs (54/79, 68%), ill-thrift and/or shy feeders (13/23, 57%), or weakness and/or recumbency (13/31, 42%). Approximately 10% (13/31) of animals presenting with lameness and 12.5% (1/8) of those that died from misadventure were found to have concurrent respiratory disease at necropsy.
Data on the presence and gross distribution of lung lesions was available for 157 out of 197 cases. No significant lesions were seen in 35 out of 157 (22.3%) cases. Of the animals with grossly visible lung pathology, diffuse discoloration and/or consolidation was seen in the cranioventral lobes only in 85 out of 122 (69.7%) cases and all visible lung in 25 (20.5%) cases. Multifocal lesions were present in 8 (6.6%) animals, focal lesions in 3 (2.5%) animals, and a single animal showed both cranioventral and multifocal lesions.
A gross necropsy diagnosis of inappetence was recorded for 7 out of 22 (31.8%) animals in the ill-thrift and/or shy feeders category and 5 out of 31 (16.1%) of animals presenting with weakness or recumbency. Inflammation of joints and muscle was found in 17 out of 30 (56.7%) animals presenting with lameness, and 3 of these also had histologic lesions consistent with septicemia.
Histology
On average, necropsy samples were collected from 43.1% (range: 3.8–100%) of the animals that died on each study voyage. The sampling rate was found to vary with time since the start of the project (P < 0.05) and the total number of deaths that occurred during the voyage (P < 0.05). Sampling rates for voyages at the beginning of the project were generally higher than at the end. The AA Vet accompanying the voyage, project month, total voyage length, number of deaths, and mortality rate had a significant effect on sampling rate in a univariate model (all P < 0.05), but not in a multivariate model.
Between 1 and 16 tissues were collected from each animal, with an average of 6 ± 3 tissues per animal. The most commonly collected tissues were lung (collected from 99% of necropsies), heart (80%), kidney (74%), liver (73%), trachea (67.5%), and rumen (46.2%). Approximately 50% of tissues were histologically normal, while in approximately 40% there were pathologic changes. The remaining tissues were considered unsuitable for examination because the tissue structure(s) of interest was not present or the tissue was too autolyzed.
The heart was collected from 158 out of 197 (80.2%) necropsies. Of the 16 cases that showed some pathology in the heart, mild to moderate, multifocal myocarditis was seen in 10 (62.5%). Acute myocarditis was observed in half of the cases, and 3 of these cases had lesions in other tissues consistent with a diagnosis of systemic infection with H. somni. The remaining 5 cases showed lymphoplasmacytic foci indicative of chronic myocarditis; sarcocysts (the asexual replication stage of Sarcocystis spp.) were observed in 3 of these cases.
The lung was collected for histologic examination from 195 out of 197 (99%) necropsies. Of the 127 animals with histologic evidence of respiratory disease, bronchopneumonia was seen in 104 (81.9%) and interstitial pneumonia in 19 (15%), with or without pleuritis. Histologic lesions considered typical of specific organisms, and, where present, the morphology of intralesional bacteria, were used to determine the viruses and/or bacteria that were most likely to have caused the observed lung pathology (Table 4).
Histologic type of pneumonia versus putative causative organisms detected using a quantitative reverse transcription polymerase chain reaction assay on lung swab and/or tissue for animals from which matched fixed lung and lung swab and/or tissue was available.* The number in each cell is the number of animals positive.
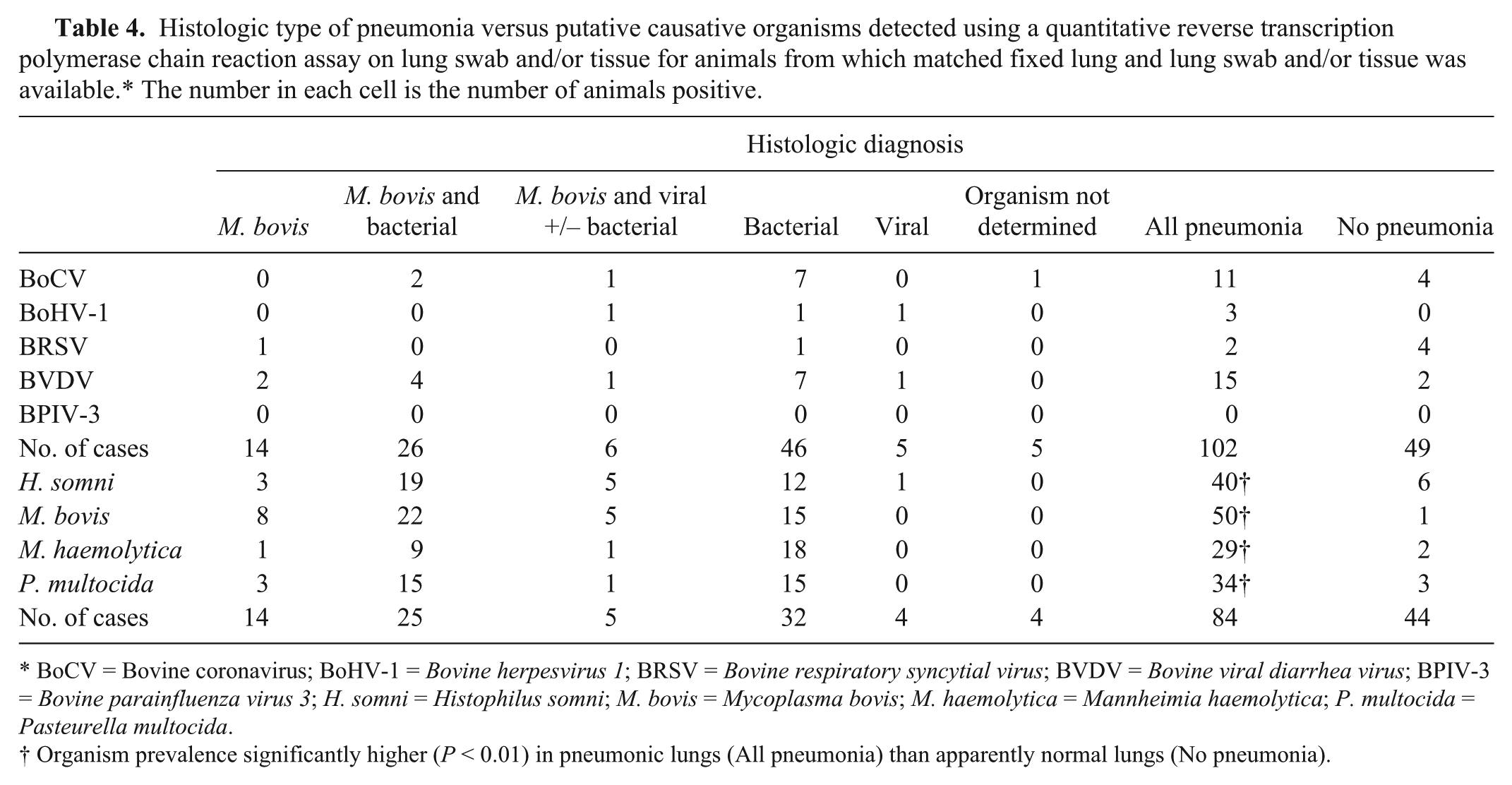
BoCV = Bovine coronavirus; BoHV-1 = Bovine herpesvirus 1; BRSV = Bovine respiratory syncytial virus; BVDV = Bovine viral diarrhea virus; BPIV-3 = Bovine parainfluenza virus 3; H. somni = Histophilus somni; M. bovis = Mycoplasma bovis; M. haemolytica = Mannheimia haemolytica; P. multocida = Pasteurella multocida.
Organism prevalence significantly higher (P < 0.01) in pneumonic lungs (All pneumonia) than apparently normal lungs (No pneumonia).
Molecular results
Lung tissue or swabs or both were collected for molecular pathology from 159 animals across 15 study voyages. Matched fixed and frozen lung samples were available for 151 cases.
The overall prevalence of the viruses of interest in lung samples was low; BoCV and BVDV were detected in approximately 10% of animals, BoHV-1 and BRSV were detected in less than 5% of cases, and BPIV-3 was not detected. Bacteria of interest were detected in 24% (M. haemolytica) to 60% (M. bovis) of animals tested (Table 4).
It was found that the prevalence of viruses was up to 4 times higher (all P > 0.07), and the prevalence of bacteria up to 26 times higher (all P < 0.01) in animals with histologic evidence of respiratory disease than animals with histologically normal lungs. The only exception was BRSV, for which the prevalence was 4 times higher in animals with normal lungs (Table 4), although the sample size was very small (n = 6).
Of animals with histologic features considered to be characteristic of infection with a particular organism, nucleic acid from the organism of interest was also detected by qRT-PCR in 45% of cases for viruses (no significant correlation, P = 0.076), and approximately 73% of cases for bacteria (statistically significant correlation, P < 0.001).
The presence of H. somni in lung tissue was significantly correlated with the presence of M. bovis, P. multocida, and viral pathogens (all P < 0.001), in particular BoCV and BVDV (both P < 0.01). The presence of M. bovis and M. haemolytica were significantly correlated with the presence of P. multocida (both P < 0.001).
Nasal swabs were collected at necropsy from 84 animals across 11 study voyages. Sterile swabs and VTM were included in the necropsy kit of all enrolled voyages but sampling of each case was at the discretion of the person performing the necropsy.
The viruses of interest were detected in the nasal swabs from up to 13% of animals, and bacteria from up to 49% of animals (Table 5). Matched nasal swab samples and lung histology results were available for 82 animals. Animals with M. bovis, M. haemolytica, or P. multocida in their nasal swabs had significantly higher odds of having histologic evidence of respiratory disease and a final diagnosis of respiratory disease. Bovine coronavirus was also significantly associated with higher odds of a final diagnosis of respiratory disease (Table 5).
Nasal swab results versus histologic diagnosis and final diagnosis.*
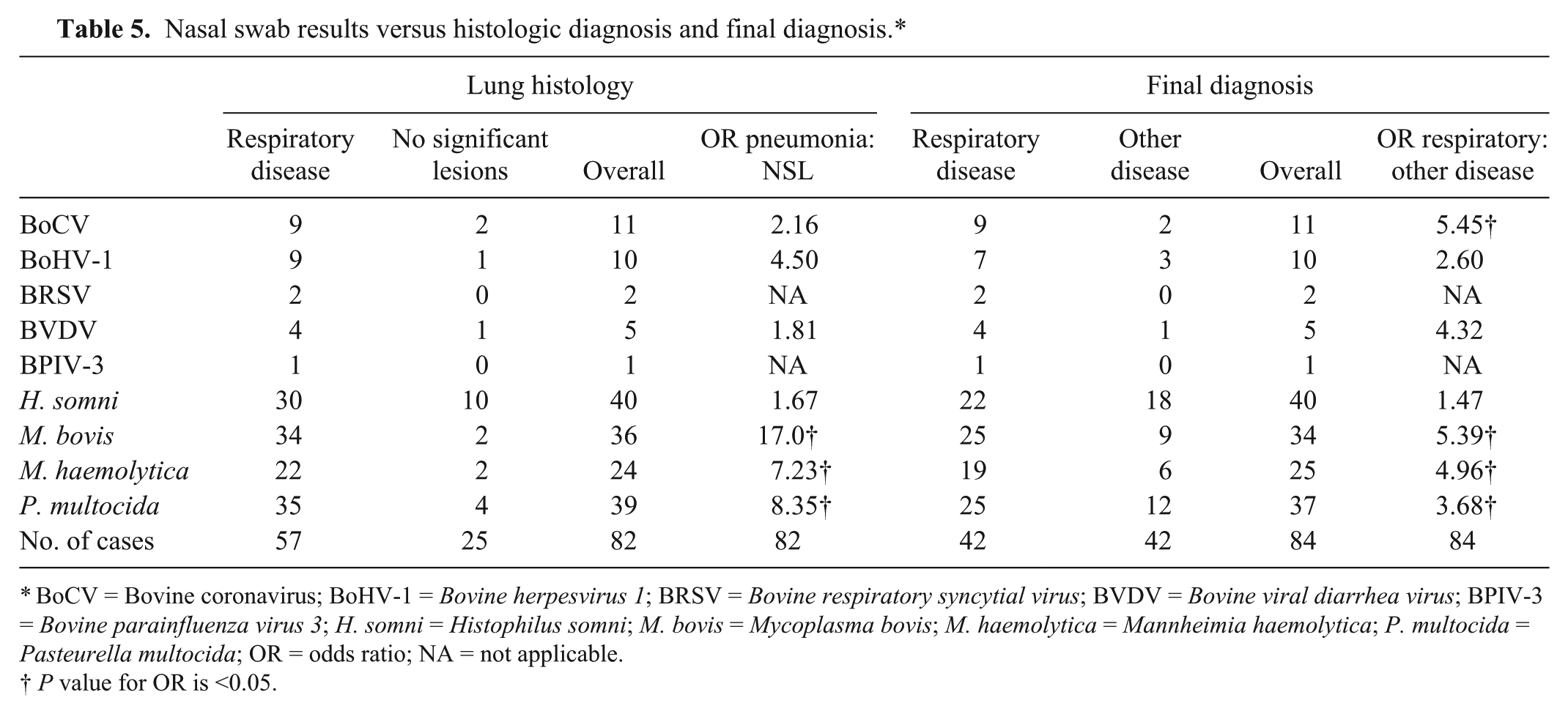
BoCV = Bovine coronavirus; BoHV-1 = Bovine herpesvirus 1; BRSV = Bovine respiratory syncytial virus; BVDV = Bovine viral diarrhea virus; BPIV-3 = Bovine parainfluenza virus 3; H. somni = Histophilus somni; M. bovis = Mycoplasma bovis; M. haemolytica = Mannheimia haemolytica; P. multocida = Pasteurella multocida; OR = odds ratio; NA = not applicable.
P value for OR is <0.05.
Matched frozen nasal and lung samples were available for 79 cases. Of the 34 animals that were found to have M. bovis in their nasal swabs, M. bovis was detected in the lungs of 67.6% (Table 6). Coexistence of nasal shedding and presence of the organism of interest in the lung was also detected in at least half of the animals tested for BVDV (n = 5; 60%), H. somni (n = 39; 53.8%), and BRSV (n = 2; 50%).
Detection of viruses and bacteria in nasal swab and/or lung tissue samples.*

BoCV = Bovine coronavirus; BoHV-1 = Bovine herpesvirus 1; BRSV = Bovine respiratory syncytial virus; BVDV = Bovine viral diarrhea virus; BPIV-3 = Bovine parainfluenza virus 3; H. somni = Histophilus somni; M. bovis = Mycoplasma bovis; M. haemolytica = Mannheimia haemolytica; P. multocida = Pasteurella multocida.
Of the 9 animals from which swabs of grossly abnormal joints or cellulitic tissue were taken, 2 (22%) were positive for M. bovis. An additional 5 animals were positive for M. bovis on nasal or lung samples and had clinical and/or necropsy findings of lameness, but swabs of affected tissues were not taken. No significant association was found between an animal having at least 1 M. bovis–positive sample (i.e., nasal and/or lung and/or other tissue) and clinical signs or a final diagnosis of lameness.
Final diagnosis
Overall, causes of death were grouped into 9 categories: respiratory disease, lameness, misadventure, ketosis, septicemia, enteric disease, anesthetic complication, heat stroke, and other (Table 7). Respiratory disease was the most common cause of death, accounting for nearly 50% of deaths overall (Table 7) and 59.1% of deaths for which a final diagnosis could be made. Of the 100 animals with a necropsy diagnosis of respiratory disease, respiratory disease was confirmed by histology in 86 cases, no significant lesions were seen in 8 cases, and the lung was not sampled in 6 cases. Histologic evidence of concurrent respiratory disease was present in an average of 33% of animals for which respiratory disease was not considered the primary cause of death. Two thirds (130/195) of animals from which lung samples were collected had histologic and/or molecular results indicative of infectious lung disease: 93 out of 130 (72%) had evidence of bacterial infection, 4 (3%) had viral infection, 29 (22%) had mixed bacterial and viral infections, and, for 4 (3%), the causative organism could not be identified.
Final diagnosis versus voyage. The final diagnosis for each animal was based on the combination of all available data: description of premortem clinical signs, gross necropsy findings, histology results, and quantitative reverse transcription polymerase chain reaction results. Not all data was available for all animals.*
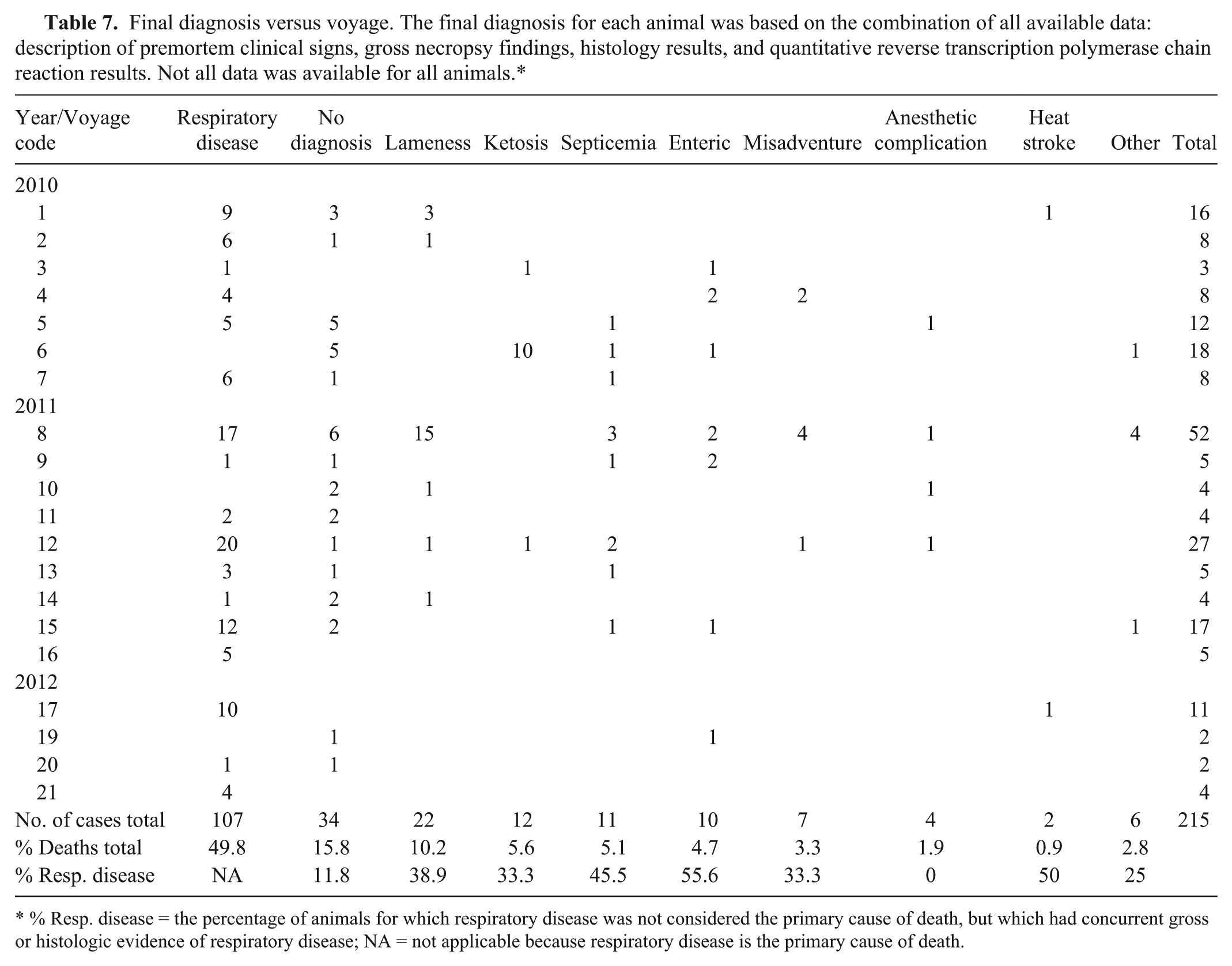
% Resp. disease = the percentage of animals for which respiratory disease was not considered the primary cause of death, but which had concurrent gross or histologic evidence of respiratory disease; NA = not applicable because respiratory disease is the primary cause of death.
Seventeen of the 22 animals diagnosed with lameness were euthanized because their injuries were significantly compromising their welfare. Animals that were sedated for treatment of leg wounds and failed to recover from anesthetic accounted for an additional 4 lameness-related deaths. The proportion of deaths due to lameness and misadventure was significantly higher on vessel C when compared with other vessels (P < 0.01).
Most of the cases of ketosis occurred on a single shipment of pregnant dairy and beef breeder cattle sailing from Portland to the Russian Federation. Suspected predisposing risk factors for death identified by the AA Vet accompanying the voyage included a 2-week delay from the scheduled sail date, voyage length (31 days), suboptimal access to feed and water, and hot and humid conditions in the Gulf of Aden and Red Sea followed by cold conditions in the Mediterranean Sea.
Miscellaneous causes of death included severe urinary tract infection and/or obstruction requiring euthanasia (n = 1, voyage 6), H. somni myocarditis (n = 2) with encephalitis (n = 1, voyage 8), severe pink eye requiring euthanasia (n = 1, voyage 8), and death due to pericarditis of unknown origin (n = 1, voyage 15). A final diagnosis could not be determined for 35 out of 215 (16.3%) cases. No significant association was found between “no diagnosis” cases and whether or not gross necropsy findings were recorded, whether or not the lung was sampled, or the number of tissues collected at necropsy (all P > 0.1).
Of the 79 animals that were found dead with no previous clinical signs, a gross necropsy diagnosis and histology samples were available for 68. The gross diagnosis was confirmed by histology in 56 out of 68 (82.4%) cases, no significant lesions were found on histology in 8 (11.8%) cases, and a different disease was diagnosed in 4 (5.9%) cases.
Of the 17 animals for which no significant lesions were found at necropsy, a final diagnosis based on histologic samples alone could be made for 5 (29.4%). No significant lesions were found on histology for 8 (47.1%) cases, and the remaining 4 cases had histologic lesions that were not severe enough to have been the cause of death.
Discussion
The current study involved the development of research protocols that were then applied by AA Vets on routine commercial voyages. This approach was aimed at providing a standardized collection of samples and data for the research project as well as longer term development of sustainable protocols for investigating morbidity and mortality under routine operating conditions.
The project enrolled approximately one-third of the eligible voyages that occurred during the time period and 20 voyages represents a substantive sample of export voyages. There were potential biases in the enrollment of voyages and in the collection of postmortem material, for example, the decision to participate in the project was made by the exporter and decisions about whether to perform a postmortem and collection of samples were made by the AA Vet.
Adjusted mortality rates were significantly associated with voyage but the implications of this finding are difficult to explain because of the sparse data and confounders at multiple levels, including exporter, ship, AA Vet, and other unmeasured voyage-related factors (e.g., sea condition, climate.). Adjusted mortality rates were higher for voyages in summer and winter than in spring and autumn. Increased mortality rates on long-haul shipments loaded out of southern ports in the Australian winter and arriving in the Middle East summer have been reported previously for both cattle 31 and sheep.22,37 A previous study 37 postulated that winter grazing on abundant pasture resulted in a metabolic state where sheep were adjusted to laying down body fat and so were less able to utilize adipose reserves for energy when required (e.g., during periods of inappetence prior to and during export), compared with animals grazing on poor pasture at other times of the year. Climatic factors are also likely to play a role; cattle on voyages in summer and winter are going from one climatic extreme to another (e.g., summer heat in Australia to winter cold in the Russian Federation), with little time for physiologic adaptation, which may predispose them to becoming sick or dying during the voyage.
Respiratory disease was the most common cause of mortality identified in the current study, accounting for 59.1% of the deaths for which a final diagnosis could be made. This prevalence is nearly 3 times that reported previously 31 (18.9%) but broadly similar to that reported in beef feedlots in Canada (46–65%)5,10,14,24,30 and the United States (55–75%).25,28 Histologic evidence of concurrent respiratory disease was also present in an average of 33% of animals for which respiratory disease was not considered the primary cause of death.
One possible reason for the discrepancy between the previous study 31 and the current study could be sampling bias. In the previous study, 31 necropsies were performed by designated research staff while in the current study necropsies were performed by the AA Vet accompanying the voyage, in addition to the duties that they are routinely expected to perform during voyages. Time pressures and varying levels of interest in the project, both between AA Vets and across time, meant that sampling was not consistent across all voyages. However, when only voyages on which at least 80% of deaths were necropsied were considered, deaths due to respiratory disease accounted for 48% of deaths, still more than twice that reported previously. 31
Quantitative RT-PCR assay was used in the present study to detect potential respiratory pathogens in samples collected from the nose and lungs of cattle on board the vessels. The prevalence of viruses of interest (BoCV, BoHV-1, BRSV, BVDV, BPIV-3) was less than 15% in both nasal and lung samples. The prevalence of these viruses was higher in animals with clinical or histologic evidence of respiratory disease than those without, with the exception of BRSV in lung samples. However, the only statistically significant association was between the prevalence of BoCV in nasal swabs and increased odds of a final diagnosis of respiratory disease (Table 5). In the pathogenesis of BRD, viruses play a primary role, causing damage to the respiratory tract and facilitating secondary invasion by bacteria that may go on to cause a fatal bacterial pneumonia. 33 Therefore the absence of a significant association between one particular virus and terminal BRD does not rule out the possibility that that virus may have contributed to the development of disease.
The current study reports a molecular diagnosis of BoCV in association with respiratory disease in Western Australian and South Australian cattle. In 2012, BoCV in association with BRD was reported on a property on the south coast of New South Wales, Australia, 21 and BoCV has been detected in fecal samples from scouring calves in Western Australia (MA O’Dea, personal observation).
Bovine coronavirus was detected in 11 out of 102 (10.8%) lungs with histologic evidence of respiratory disease. This is similar to that previously reported for cattle in a U.S. feedyard, where a conventional RT-PCR assay was applied to lung homogenates (21/194, 10.8%). 11 Other studies using non–PCR-based techniques for BoCV detection in lung samples have reported detection rates of 1.4% in Danish cattle, 44 2% in Canadian cattle, 14 and 69.2% in American cattle. 41 It is difficult to make direct comparisons between these studies because of differences in analytical methods, timing of data and sample collection, sample sizes, and environmental and animal factors. However, it would appear that, overall, BoCV detection rates in diseased lung tissue collected at necropsy are generally low. This is likely to reflect the role that BoCV is thought to play in the development of BRD (i.e., as a predisposing factor that allows commensal bacteria to infect the lungs, rather than a cause of terminal respiratory disease 38 ).
An association between BoCV in nasal swabs and clinical signs of respiratory disease has been found by some studies17,42,43,45 but not others.4,18 Nasal shedding of BoCV has also been linked to an increased risk of treatment for respiratory disease, 34 increased incidence of pulmonary lesions at slaughter, 27 and reduced weight gain. 45 In the study reported herein, animals with BoCV in their nasal swabs had higher odds of having histologic evidence of respiratory disease (odds ratio [OR] = 2.16, P > 0.05) and a final diagnosis of respiratory disease (OR = 5.45, P < 0.05) compared with animals that were BoCV negative. Therefore, although the detection rates of BoCV in lung samples and nasal swabs were relatively low (10.8% and 13%, respectively), it is likely that BoCV is important in the development of BRD in Australian live export cattle.
Bovine viral diarrhea virus was detected in the lungs of 15 out of 102 (15%) animals with histologic evidence of pneumonia, and this was the highest detection rate of any of the viruses of interest. Previous studies on the presence of BVDV in lung tissues have reported detection rates of 6–21% in diseased lungs.11,12,14,44 In feedlot cattle, the presence of BVDV in the lung has been found to be associated with earlier treatment for BRD, shorter treatment intervals when multiple treatments were required, and earlier death due to BRD than animals without BVDV in their lungs.11,14
In the current study, 7 out of 20 (35%) animals that tested positive on 1 or more samples were from consignments in which acutely or persistently infected animals had been identified by serum protein A antibody-capture ELISA (PACE) and removed from the consignment prior to loading. The detection of BVDV in animals on board the ship may be due to infection of naive animals by persistently infected animals during the assembly period or acutely infected animals that had insufficient viral loads to be detected by PACE. The present study suggests that a single test to remove persistently infected animals does not guarantee the removal of the virus from the consignment.
Mycoplasma bovis was detected in 51 out of 128 (40%) lung samples and in 34 out of 84 (40%) nasal swab samples. The presence of M. bovis in the lung and nasal secretions was significantly associated with higher odds of having histologic evidence of respiratory disease and a final diagnosis of respiratory disease. The prevalence of M. bovis in lungs with histologic evidence of respiratory disease was 60% (50/84). This is similar to that previously reported for beef feedlot cattle (51–88%).1,11,13 Mycoplasma bovis was most commonly found as a component of mixed bacterial infections, although all 6 animals from which M. bovis was the only organism detected in the lung had histologic evidence of respiratory disease.
No significant relationship was found between M. bovis and arthritis or lameness, although pneumonia and arthritis and/or tenosynovitis is a common presentation in animals with M. bovis. 3 This could be due to inadequate data collection, inadequate data on the presence of arthritis, difficulty in seeing lesions on board vessels because of darkness and manure, or the small number of samples that was taken from infected joints or adjacent tissues.
Mannheimia haemolytica, P. multocida, and H. somni have all been identified as important organisms in the development of BRD.7,15 Mannheimia haemolytica and P. multocida were detected in 24% and 29% of lung samples and 30% and 44% of nasal swab samples, respectively. Histophilus somni was detected in 36% of lung and 48% of nasal swab samples (Tables 4, 5) and was the only bacterial organism that was not significantly associated with histologic evidence of or a final diagnosis of respiratory disease (Table 5), although it was associated with increased severity of pneumonia.
Of particular interest was the identification of 3 cases with histologic findings consistent with a diagnosis of myocarditis (n = 3), and pneumonia (n = 1) or thrombotic meningoencephalitis (TEM; n = 1) associated with H. somni infection. Myocardial abscesses were smaller and less severe than described previously16,19 although it is possible that additional lesions were present but not seen, or seen but not recorded and/or sampled by the AA Vet performing the necropsy. Although TEM was the first manifestation of disease associated with H. somni to be described, 2 a number of subsequent reports suggests that case presentations of pneumonia, myocarditis, and/or arthritis in the absence of TEM are becoming more common.16,32,39,46
Heat stroke was the most common cause of death in previous studies20,31 accounting for 50 out of 92 (54.3%) and 58 out of 180 (32.2%) of deaths for which a diagnosis could be made, respectively. Heat stroke was identified as a primary cause of death in only 2 animals in the present study (<1% of deaths) although hyperthermia secondary to other disease processes (e.g., severe bronchopneumonia and septicemia) was recorded in an additional 9 animals. The reduced incidence of deaths due to heat stroke in the study reported herein compared with previous studies could be due to a number of factors, including misclassification, changes to the preexport and on-board management of cattle, and differences in the type of cattle being exported.
The present study depended on collection of samples by AA Vets. AA Vets collecting the samples received training on identifying gross necropsy changes typical of heat stroke in cattle to reduce the risk of misdiagnosis. If none of these signs were evident at necropsy then the animal was not recorded as having died of heat stroke. AA Vets were encouraged to take CBTs and record the number of hours between death and necropsy to assist in making an informed and evidence-based diagnosis. However, CBT was measured in less than half of the animals in this study so it is possible that animals that died and whose CBT was not measured may have had primary or secondary heat stress.
Since the late 1990s, a combination of industry initiatives (Australian Live Export Standards (ALES), 1997 40 ) and mandatory standards for animal health and welfare (Australian Standards for the Export of Livestock (ASEL), 2004 8 ) have led to changes in the management of cattle prior to and during export. Changes include seasonal restrictions on geographic regions from which cattle can be sourced and maximum stocking densities based on season, voyage length, and type of animal (Bos taurus vs. Bos indicus). In 2003, the Livestock Export Heat Stress Risk Assessment Model (HotStuff) was implemented to help exporters to estimate and minimize the risk of heat stress mortalities occurring during a voyage (Maunsell Australia: 2003, Development of a heat stress risk management model. Final report for Meat & Livestock Australia and LiveCorp Project LIVE.116. Available at: http://www.mla.com.au/Research-and-development/Final-report-details?projectid=1192). Ongoing public pressure ensures that exporters, AA Vets, and stockpersons are very aware that they are operating under and accountable to Commonwealth law. The results reported herein suggest that an increased awareness and consideration of animal welfare has achieved better conditions for live export cattle, including a reduction in deaths due to heat stroke.
The type of cattle sent for live export has changed over the past 25 years, mainly in response to import trade tariffs and consumer education. There is a trend toward shipping younger “feeder” cattle that are fattened at their destination, rather than older “slaughter” cattle that are killed soon after their arrival. Smaller animals have a larger surface area to volume ratio so they are better able to respond to changes in ambient temperature and humidity than larger animals. At the time of the previous studies (1985–1987 20 and 1998–2001 31 ), it is likely that the proportion of >600-kg slaughter animals in a shipment was higher than for the voyages in the current study. This difference in shipment composition may have contributed to the lower incidence of heat stress observed on the voyages reported herein.
Trauma, including lameness and misadventure, was the third most common cause of death. Deaths in the lameness category were generally the result of the animal being euthanized because its injuries were severely compromising its welfare. The incidence of trauma (13.5%) was lower than that reported previously20,31 (22.8% and 24.6%, respectively). This is likely due to a combination of improved vessel design and a change in the type of cattle being exported.
Lameness may begin prior to loading or during the voyage. During the assembly period, poorly designed or maintained yards or trucks combined with rough handling can lead to foot and leg injuries. During the voyage, lameness develops secondary to injuries and abrasive floor surfaces. Injuries are more common during rough weather, with reduced stocking densities, and in heavy animals (>380 kg; Banney S, Henderson A, Caston K: 2009, Management of bedding during the livestock export process. Final report for Meat & Livestock Australia project W.LIV.0254. Available at: http://www.mla.com.au/Research-and-development/Final-report-details?projectid=14966). 20 Ongoing maceration and bacterial contamination through contact with manure means that these lesions seldom heal without treatment.
Risk factors for the development of lameness include both animal and management factors (Banney S, et al.: 2009, Management of bedding during the livestock export process). 31 Heavy animals and those with soft feet, particularly southern and dairy cattle, are more likely to become lame. Management factors include the type of pen flooring, type and amount of bedding, frequency of deck washing (to remove soiled bedding), and stocking densities. Recommendations for best practice for cattle selection and bedding management to reduce the incidence of lameness have been made (Banney S, et al.: 2009), but morbidity and mortalities due to lameness are still relatively high, indicating that there is room for improvement.
Sedation for wound cleaning and bandaging carries its own mortality risk, as the 4 postanesthetic deaths show. Hyperthermia is known to occur following administration of tranquilizing drugs to sheep in hot weather. 36 Xylazine is commonly used for sedation of cattle on board vessels, and hyperthermia secondary to a reduced ability to thermoregulate is a recognized side-effect of deep sedation with xylazine, particularly when animals are in an environment where overheating could occur.
Enteric disease was uncommon. Conditions observed included bloat (n = 2), enteritis (n = 3), and gastrointestinal rupture (n = 2) or obstruction (n = 1). The most common disease involving an organ of the digestive system was fatty liver change secondary to inappetence (n = 3) associated with advanced pregnancy (n = 9). Animals are monitored daily during the pre-export period and voyage to identify and treat animals showing signs of inappetence (“shy feeders”). However, despite the best efforts of the AA Vets and on-board stockmen, a small proportion of animals fail to adapt to on-board conditions and rations.
The study reported herein builds on the previous study of cattle deaths during sea transport from Australia 31 by including a greater number of study voyages (20 vs. 4) and the application of qPCR assays to detect bacteria and viruses of interest in necropsy samples. Heat stress appears to be less of a problem than previously reported, and this is likely to be due to heat stress risk management strategies implemented by the export industry since the late 1990s and a change in the type of animals being exported. Lameness remains an important cause of morbidity and mortality, and further research is needed into ways to manage this problem.
Although overall mortality rates are low, respiratory disease is still an important cause of death. The current investigation provides baseline data on the bacteria and viruses involved in BRD in Australian live export cattle, which can be used to inform on future BRD prevention and management strategies. Further studies are already underway to investigate preloading risk factors, including the prevalence of potential pathogens during the assembly period.
Footnotes
Acknowledgements
This project would not have been possible without the cooperation of exporters, AA Vets, and stockmen. Special thanks to Greg Norman for managing all things “boat.” Thanks also to the staff of the Histology, Bacteriology, and Virology Departments at the Department of Agriculture and Food, Western Australia (DAFWA) and Virology Laboratory at Elizabeth Macarthur Agricultural Institute for help with sample processing and testing, and to John Creeper for assistance with sample processing, slide interpretation, and review of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this project was provided by Meat & Livestock Australia and LiveCorp (project W.LIV.0252).
a.
Greiner Bio-One GmbH, Hannover, Germany.
b.
Ambion MagMax Viral RNA Extraction Kit, Life Technologies, Grand Island, NY.
c.
MagMax Express 96 magnetic particle processor, Life Technologies, Grand Island, NY.
d.
QuantiTect virus +ROX vial kit, Qiagen Inc., Valencia, CA.
e.
Primer Express software v2.0, Applied Biosystems, Foster City, CA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
