Abstract
A 21-mo-old Japanese Black beef cow had swollen mandibular and superficial cervical lymph nodes. Fine-needle aspiration cytology of the superficial cervical lymph node revealed large lymphoblasts with mitoses present. The bovine leukemia virus (BLV) proviral load was relatively high, and phylogenetic analysis of the whole BLV genome classified the BLV strain as one with high viral replication activity. Genotyping of bovine leukocyte antigen genes indicated that the cow was susceptible to enzootic bovine leukosis (EBL). The bone morphogenetic protein 6 (
Bovine leukosis is divided into 2 types: enzootic bovine leukosis (EBL) caused by bovine leukemia virus (BLV; genus
Both viral and host factors must be considered when elucidating viral infectious diseases. Differences in pathogenicity of BLV strains,
14
sensitivity to EBL onset based on bovine leukocyte antigen (BoLA) DRB3 polymorphism,
8
and methylation status of bone morphogenetic protein 6 (
A 21-mo-old Japanese Black beef cow was presented with emaciation and mandibular swelling. Initial examination by a local veterinarian (day 1) revealed a body temperature of 40.6°C (RI: 38.0–39.2°C) and heart rate of 100 beats/min (bpm; RI: 60–84 bpm). 3 Emaciation, difficulty standing, and mandibular swelling were noted. The cow was treated for mandibular inflammation with 10 mg/kg oxytetracycline and 2.5 mg/kg NSAID medication. By day 6, superficial cervical and subiliac lymph nodes were enlarged. Bovine leukosis was suspected by a local veterinarian, and, on day 12, the cow was transferred to the Animal Teaching Hospital at the Obihiro University of Agriculture and Veterinary Medicine (Obihiro, Hokkaido, Japan) to confirm the diagnosis.
On initial physical examination at the hospital, high rectal temperature (40.2°C), normal heart rate (66 bpm), polypnea (42 breaths/min; RI: 18–28 breaths/min), 3 emaciation, and difficulty standing were noted. Swelling in lymph nodes, including parotid, mandibular, superficial cervical, and subiliac lymph nodes, was observed (Fig. 1). Rectal palpation revealed masses (1–4 cm) in the pelvic cavity. Fine-needle aspiration (FNA) cytology of superficial lymph nodes revealed medium-to-large lymphoblasts with a small amount of cytoplasm and occasional mitoses (Suppl. Fig. 1).
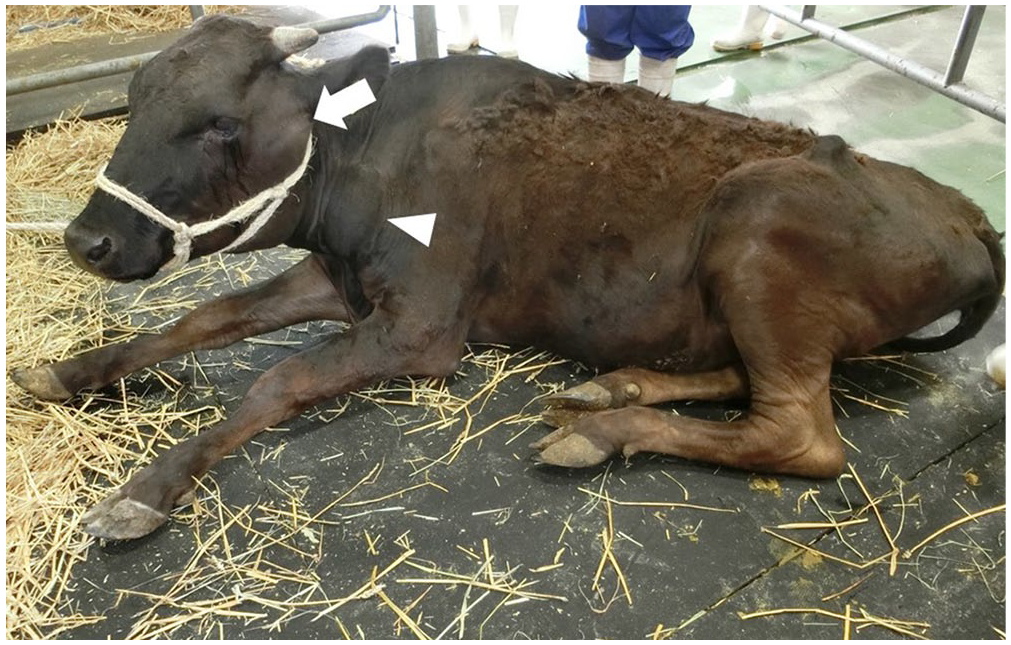
Swelling of parotid (arrow) and prescapular (arrowhead) lymph nodes on day 12 in a Japanese Black cow with enzootic bovine leukosis.
Hematologic examination showed mild anemia (RBC, 6.08 × 1012/L, RI: 5.10–7.60 × 1012; Hb, 70 g/L, RI: 85–122 g/L; PCV, 0.20 L/L, RI: 0.22–0.33 L/L). 3 WBC (11.2 × 109/L; RI: 4.9–12.0 × 109/L) and platelet count (606 × 109/L; RI: 193–637 × 109/L ) were within RIs. 3 In peripheral blood smears, 40% (4.48 × 109/L ) of WBCs were medium-to-large atypical lymphoid cells (Suppl. Fig. 2). Serum biochemical analysis revealed increased activities of lactate dehydrogenase (3,660 U/L; RI: 697–1,450 U/L) and thymidine kinase (818 U/L; RI: <5.4 U/L).6,18
The BLV proviral load of DNA extracted from the peripheral blood sample was quantified (CoCoMo-BLV primer/probe, Riken Genesis; TaqMan gene expression master mix, Thermo Fisher) according to the manufacturer’s instructions. The BLV copy number in peripheral blood was 2,720 copies/50 ng DNA. The DNA from peripheral blood was used as a template for the PCR assay, which was performed using PrimeSTAR GXL DNA polymerase (Takara Bio) and 2 primer pairs (BLV 1-17 F: 5′-TGTATGAAAGATCATGC-3′, BLV 4565-4586 R: 5′-AATCTGATTGTGAGTCCAGAGG-3′; BLV 4416-4436 F: 5′-CAGTTCGGAGTTTCCCTTTCT-3′, BLV 8703-8720 R: 5′-TGTTTGCCGGTCTCTCCT-3′). 14 The PCR reaction was as follows: amplification with 30 cycles of denaturation at 98°C for 10 s, annealing at 55°C for 15 s, extension at 68°C for 5 min, and final extension at 68°C for 2 min. PCR products were treated with ExoSAP-IT express (Thermo Fisher). DNA libraries were prepared (QIAseq FX DNA library kit; Qiagen) following the manufacturer’s protocol, and sequenced (iSeq system; Illumina) using 2 × 150 bp paired-end reads. Quality control procedures were performed (Trim Reads tool, Genomics Workbench v.20.0; CLC bio). Unless otherwise stated, all software was used with default values applied. Mapping of quality-filtered reads against a reference BLV genome (EF600696.1) was performed using the CLC mapping tool, and 568 BLV genome sequences reconstructed by the sequencing reads (568 coverage). The whole genome sequence of BLV in our case was identified. A phylogenetic tree was constructed by neighbor-joining methods (1,000 bootstrap replications) using the whole genome sequence of BLV in our case (LC659972) and complete genome sequences of BLV obtained from GenBank (LC164086, AP018006–AP018032). BLV in our case was classified as a strain with high viral replication activity (group A; Fig. 2).
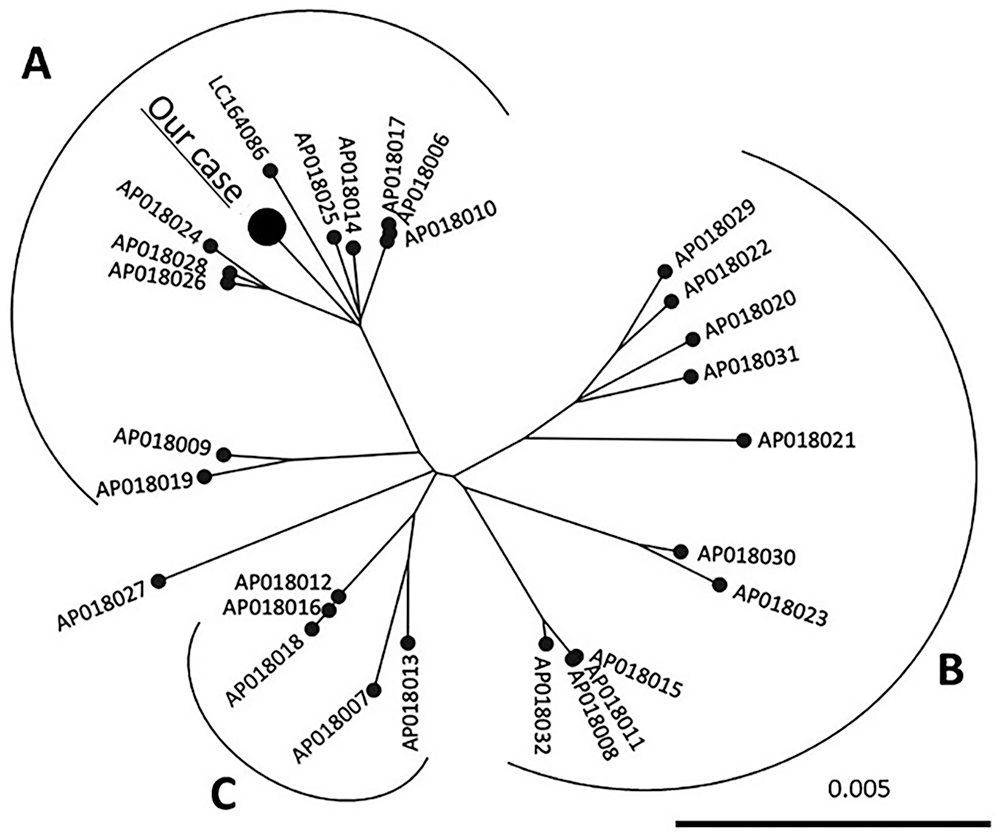
Phylogenetic analysis of the whole bovine leukemia virus (BLV) genome sequence in our case. A maximum-likelihood phylogenetic tree was constructed from whole BLV genome sequences of our case and 28 reference sequences. The BLV strains were divided into A–C based on a previous study. The bar denotes distance.
Methylation status of the
B-cell clonality of peripheral blood and swollen superficial lymph node tissue obtained by FNA was examined using PCR for immunoglobulin heavy-chain gene
BLV proviral integration clonality was analyzed using inverse PCR (iPCR). The procedure for iPCR was performed as reported previously.
11
DNA samples used for the B-cell clonality analysis were digested with
The cow was euthanized on day 14 for autopsy according to the ethical and animal welfare requirements under the guidelines of the Care and Use of Agriculture Animals of Obihiro University (approval 29-38). Gross examination revealed swelling of systemic lymph nodes. Spinal canal invasion of tumor tissue at vertebrae L5-6 was observed. Histologic examination revealed infiltration of neoplastic lymphoid cells in enlarged lymph nodes, liver, spleen, heart, lung, adrenal, uterus, and the adipose tissue around the spinal cord (Fig. 3A). Immunohistochemically, tumor cells within enlarged lymph nodes stained positive for BLA36 (BioGenex; Fig. 3B) and negative for CD3 (BioGenex; Fig. 3C).
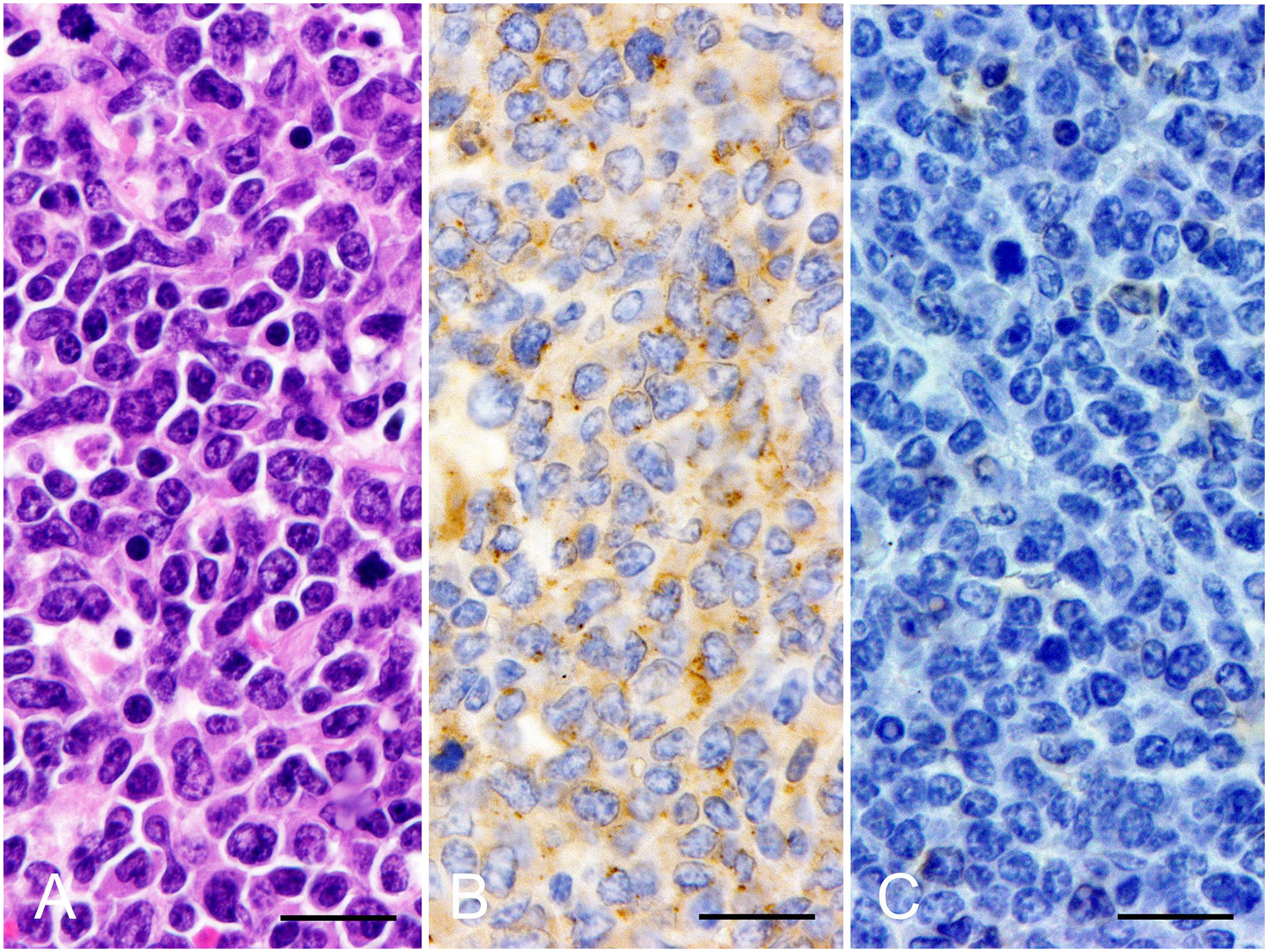
Histopathology and immunohistochemistry of superficial lymph node. Neoplastic cells had a round nucleus with nuclear atypia and granular patterned chromatin. These cells stained positive for BLA36 and negative for CD3.
EBL is typically observed in cattle >3-y-old,1,4,21 but cases of EBL in cattle <3-y-old have also been reported. 16 Moreover, onset of SBL in BLV-infected cattle has been reported. 5 Therefore, diagnosing EBL in young cattle is difficult. In our case, leukemia or lymphoma was suspected based on physical examination, FNA cytology, and hematologic examination. The BLV proviral load in our case was higher than the criterion for BLV infection associated with tumor development in a previous study (2,000 copies/50 ng DNA). 15 Monoclonal B-cell proliferation and monoclonal integration of the BLV provirus were detected by PCR for the Ig heavy-chain gene and iPCR, respectively. Histology demonstrated neoplastic proliferation of B cells. Based on these findings, we diagnosed EBL, despite the young age of the cow.
We also analyzed the BLV strain,
The MHC plays a crucial role in antigen presentation and immune responsiveness,17,20 and is also associated with infectious diseases including viral infection. The MHC system associated with disease susceptibility in cattle is known as BoLA.
13
The DNA sequence polymorphisms of
BMP6 reduces proliferation and induces apoptosis in B cells.
7
Epigenetic modifications, including DNA methylation, can alter the transcription activity in mammals.
2
A study found that the
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221102123 – Supplemental material for Enzootic bovine leukosis in a 21-month-old Japanese Black cow with high susceptibility
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221102123 for Enzootic bovine leukosis in a 21-month-old Japanese Black cow with high susceptibility by Masaki Maezawa, Kana Sakaguchi, Yuka Tagaino, Yuki Fujii, Masataka Akagami, Junko Kawakami, Ken-ichi Watanabe, Yoshiyasu Kobayashi, Haruko Ogawa and Hisashi Inokuma in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank all staff members of the Department of Veterinary Medicine at Obihiro University, those at Ibaraki Prefecture Kenpoku Livestock Hygiene Service Center for their technical assistance, and the Tokachi Agricultural Mutual Aid Association for bringing this clinical case to our attention.
Authors’ note
The data that support the findings of this study are available from the corresponding author upon request.
Declaration of conflicting interests
Laboratory of OSG Veterinary Science for Global Disease Management is an endowment laboratory, supported with a grant from OSG Corporation.
Funding
This work was supported by JSPS KAKENHI grants 20H03142 and 20J10567.
Supplemental material
Supplemental material for this article are available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
