Abstract
Bovine herpesvirus 1 (BoHV-1) is 1 of several viruses considered to be causal agents of bovine abortion. The current retrospective study examined data on bovine abortion submissions from 5 veterinary diagnostic laboratories from 2000 to 2011. The objective of the study was to describe prevalence of BoHV-1–associated abortion in submissions at veterinary diagnostic laboratories. There were 3 specific aims: 1) to examine the proportion of BoHV-1–related abortions with the introduction of new diagnostic assays such as polymerase chain reaction (PCR), 2) to evaluate the agreement of the histopathology report of the abortion submissions and the result of the assay used, and 3) to evaluate if there was an association between farm history of vaccination against BoHV-1 and BoHV-1–positive abortion submissions. An extended Mantel–Haenszel χ2 for linear trend was used to analyze the prevalence of BoHV-1 over the study period and showed that collectively there is evidence of an increase of positive BoHV-1 abortions (P < 0.001). The comparison of the proportion of BoHV-1–positive submissions pre- and postadoption of PCR was not significant (P = 0.25). Using Cohen kappa coefficient test of agreement, a kappa value of 0.81 (P < 0.001) was found, suggesting high agreement of lesions reported and assay result. It was found that using a χ2 test, a P value of 0.068 for nonmatched data (i.e., a history of vaccination against BoHV-1 in the herd) was associated with reduced detection of BoHV-1–positive abortion submissions.
Bovine herpesvirus 1 (BoHV-1; order Herpesvirales, family Herpesviridae, subfamily Alphaherpesvirinae, genus Varicellovirus) infection is 1 of several viruses considered to be a causal agent of bovine abortion. 1 Infection with BoHV-1 is also associated with infectious pustular vulvovaginitis and respiratory, ocular, neonatal, gastrointestinal, and neurologic disease. Vaccination against BoHV-1 is a means of protecting animals against the sequelae of infection including abortion. 4 Although vaccines are available for BoHV-1, vaccination may not provide complete protection or efficacy may be reduced by factors such as poor vaccination technique, handling, or misuse of modified live vaccines (i.e., administering to pregnant cows). 3 Furthermore, only 7 of 10 national operations (68.9%) vaccinated their beef cow–calf herd for BoHV-1 in 2007. 5 Consequently, BoHV-1 infection remains a potential differential diagnosis for abortion of beef cow–calf herds.
The diagnosis of BoHV-1–associated abortions is commonly made through the observation of gross and histopathologic lesions in fetal and placental tissues. Diagnoses are supported by the detection of viral antigen by fluorescent antibody test (FAT), immunohistochemistry (IHC), or viral DNA by polymerase chain reaction (PCR) methods. The choice of BoHV-1–specific detection methods varies among veterinary diagnostic laboratories (VDLs).
The overall objective of the current study was to describe the prevalence of BoHV-1–associated abortion in submissions at VDLs. To accomplish the objective, the study had 3 specific aims. The first aim was to evaluate changes in the proportion of BoHV-1–positive submissions over a decade and assess any association with assay type. Two null hypotheses were tested: 1) the proportion of BoHV-1 abortions had not increased over the past 10 years and 2) PCR-based BoHV-1 detection methods were not temporally associated with any observed increase in the proportion of BoHV-1–positive submissions. The working hypothesis for the second hypothesis was that as PCR-based detection methods for BoHV-1 become more frequently used, this could be associated with a concomitant increase in detection of BoHV-1 in abortion submissions because of increased sensitivity. The second aim was to evaluate the agreement of the histopathology report of the abortion submissions and the result of the assay used. The working hypothesis for this aim was due to increased use of PCR-based detection methods, detection of BoHV-1 associated with modified live infectious bovine rhinotracheitis (IBR) vaccines may lead to positive assay results without concomitant evidence of microscopic lesions in tissues consistent with BoHV-1. The third aim was to evaluate if there was an association between farm history of vaccination against BoHV-1 and BoHV-1–positive abortion submissions. The working hypothesis was that a history of vaccination against BoHV-1 in the herd should be associated with reduced detection of BoHV-1 in abortion submissions.
Data was received from 5 diagnostic laboratories across the United States for bovine abortion cases from January 2000 to January 2011. The participating laboratories were Iowa State University VDL (Ames, Iowa), California Animal Health and Food Safety Laboratory System at the University of California, Davis (Davis, California), Washington Animal Disease Diagnostic Laboratory at Washington State University, University of Minnesota VDL (Pullman, Washington), and South Dakota State University Animal Disease Research and Diagnostic Laboratory (Brookings, South Dakota). Not all of the laboratories had complete records available for the 11-year time period, as a change from paper to electronic record keeping took place and resulted in unavailability for complete records for the study period.
Animal-level data was requested from all bovine abortion submissions from 2000 to 2011, including the final diagnosis for each bovine abortion, the diagnostic assays used to test for BoHV-1 (not serology), and the test result. Assays of interest were FAT, IHC, and PCR. Virus isolation (VI) was not included in the study as it was not considered to be a predominately used assay. Furthermore, VI would usually only be conducted subsequent to other positive assays; therefore, the results would not contribute substantial information. Because the vernacular in veterinary science has been to refer to BoHV-1 as IBR virus, data was requested for IBR assays with the understanding that these tests were for BoHV-1. Data from all of the laboratories were harmonized to facilitate analysis, and all duplicate accessions were removed from the database. The statistical program R was used for data manipulation (http://cran.r-project.org/doc/manuals/R-intro.html), and all hypothesis tests were conducted in R using the EpiR package except where indicated (http://cran.r-project.org/web/packages/epiR/index.html).
For aim 1, the number of abortion submissions, the number of abortion submissions tested for BoHV-1, the number of BoHV-1–positive abortions, and the total number of BoHV-1 tests conducted were determined. The total number of BoHV-1 tests differs from the number of BoHV-1–positive abortions because some submissions were tested using more than 1 assay. When determining the total number of submissions tested for BoHV-1, only submissions with positive and negative results were used in the analyses. Results eliminated from the data included reports that were considered “inconclusive,” “suspect,” and “unable to evaluate.” The 3 BoHV-1 assays used were FAT, IHC, and PCR. The hypothesis put forth was that the proportion of BoHV-1–positive abortion accessions had not changed over time for each laboratory; all laboratories combined were tested using an extended Mantel–Haenszel chi-square test (http://www.openepi.com/Documentation/DoseResponsedoc.htm) for linear trend in the program Open Epi (http://www.openepi.com/Menu/OpenEpiMenu.htm). Also tested was the hypothesis that adoption of PCR-based BoHV-1 detection methods was not associated with the increased diagnosis of BoHV-1–positive abortions at laboratories that eventually adopted PCR. PCR-based BoHV-1 detection methods were first used to test submissions at the University of California, Davis, in 2008, at Iowa State University in 2009, and at the University of Minnesota in 2009. For this hypothesis, the data was categorized into diagnoses that occurred prior to and after PCR adoption, respectively, and the proportion of positive diagnoses in each period was compared using a chi-square test or Fishers exact test as appropriate.
The second aim was to evaluate the agreement between the histopathology report of the abortion submissions and the test result. For each unique BoHV-1–positive abortion accession, the set of BoHV-1–negative abortion accessions matched by month and year was identified. From this set, a single negative BoHV-1 accession was randomly chosen to create a pair of submissions matched by month and year. If no BoHV-1–negative submission occurred in the same month and year as a BoHV-1–positive submission, a match was chosen from the proceeding month. For each matched pair, the VDL laboratory report and VDL submission form were obtained. Depending on the laboratory, these forms may not have been available for the entire 11-year period due to deletion of records. For each matched pair, 1 of the authors (SG) redacted the information that described the final diagnosis and retained only the histopathology report, therefore making it unclear if the laboratory report was from a BoHV-1–positive or –negative submission. A diagnostic pathologist then reviewed the redacted laboratory reports and, based on the reported lesions, made the determination if the lesions reported were consistent with BoHV-1. The histopathology criteria used included multifocal necrosis in liver, lung, kidney, spleen, and brain or placenta if examined, with or without described nuclear inclusion bodies. The 2 possible outcomes were “yes” the reported histopathology lesions are consistent with BoHV-1 or “no” the lesions are not consistent with BoHV-1–associated abortion.
The Cohen kappa coefficient test of agreement was used to test the null hypothesis that kappa equals zero comparing the assay results with the histopathology result. When the diagnostician and the assay result did not agree, the reports were further broken down into 4 categories: 1) histopathology description available but vague or noninformative, 2) histopathology consistent with another etiology, 3) histopathology description of no lesions, and 4) histopathology description of tissue autolysis.
For the third aim, the purpose was to assess the association between vaccination history and BoHV-1–positive abortion submissions. For each matched pair in the second aim, VDL submission forms were used to extract the vaccination history of the submitting farm. Possible outcomes were 1) history of BoHV-1 vaccination, 2) no history of BoHV-1 vaccination, 3) no vaccination history reported on submission form, or 4) no record available. Many submission forms did not include the time vaccines were administered, therefore 2 analyses were conducted. First, only the matched pairs where both accessions contained information about vaccine use (yes/no) were used in the analysis. McNemars test was used to assess if vaccination history (yes/no) was associated with submission status (positive/negative). The analysis was then repeated using any record where part of the matched pair dataset reported farm history of vaccination (yes/no). As matching by month was ignored, a chi-square test was used to compare if vaccination history (yes/no) was associated with submissions status (positive/negative).
Data received contained 19,459 bovine abortions, of which 6,948 were tested for BoHV-1 and, of those 6,948 tested, 264 were positive (3.8%). The data relate only to samples with discernible results (i.e., positive and negative results; “inconclusive,” “suspect,” and “unable to evaluate” results were omitted). As a proportion of the total abortions, BoHV-1 diagnosis was rare (264/19,459; 1.4%). Combined analysis of the data from the 5 laboratories suggested evidence of an increase in BoHV-1–positive submissions (P value for linear trend < 0.01). Individually, only South Dakota showed evidence of a significant increase over the period (P value for linear trend < 0.01), as presented in Table 1.
Number of positive Bovine herpesvirus 1 (BoHV-1) submissions from 5 U.S.-based veterinary diagnostic laboratories per year that were tested for BoHV-1 ([no. of BoHV-1–positive submissions/no. of submissions tested for BoHV-1]%).*
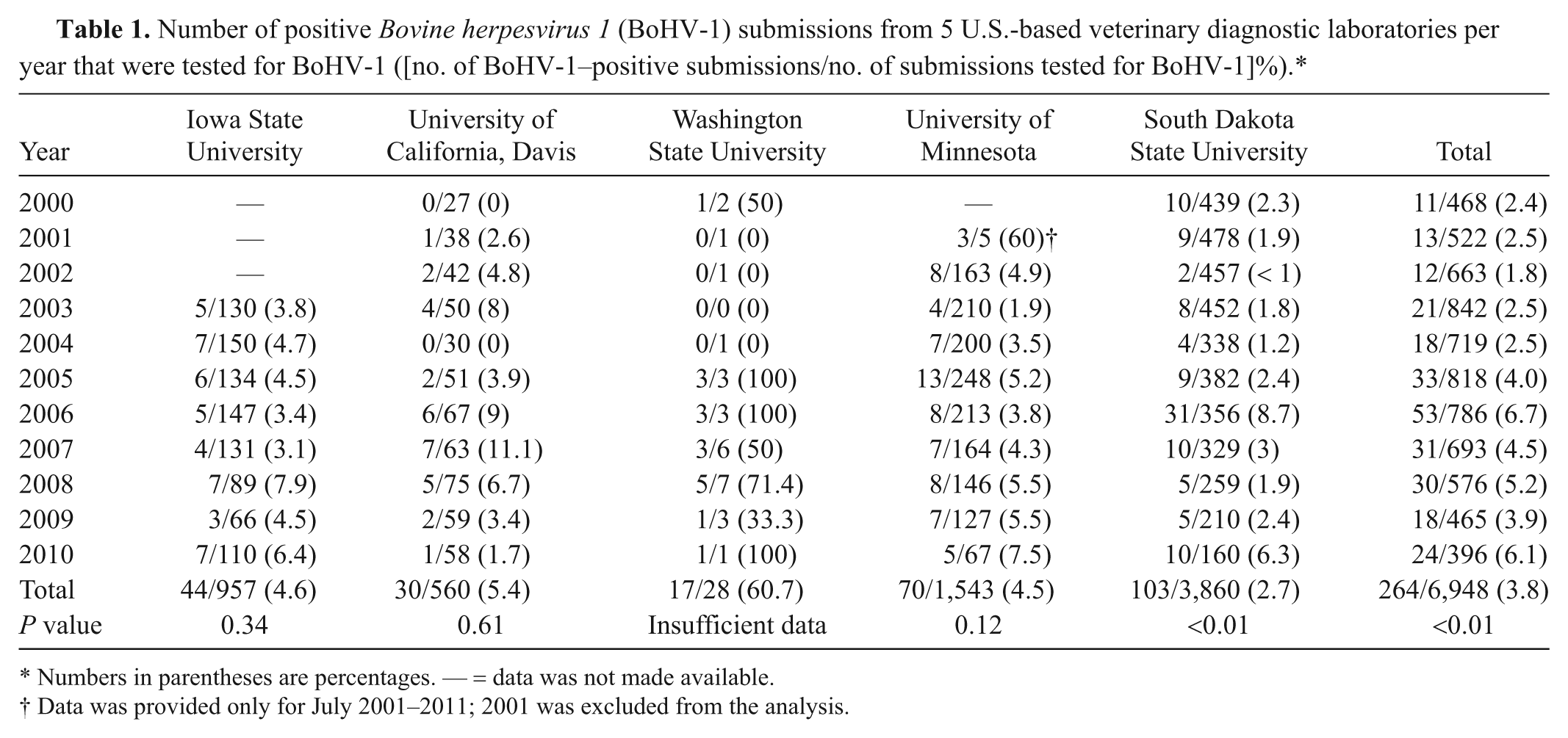
Numbers in parentheses are percentages. — = data was not made available.
Data was provided only for July 2001–2011; 2001 was excluded from the analysis.
For the comparison of the proportion of BoHV-1–positive tests prior to and after implementation of PCR, the results from Iowa State University and the University of California, Davis, were not used because use of PCR-based detection methods was rare, as presented in Table 2. Iowa State University VDL reported usage of PCR 14 times, with 2 positive results. The University of California, Davis, reported using PCR twice, and both results were negative. At the University of Minnesota VDL, 132 PCR tests were conducted after 2009. Prior to 2009, 1,344 submissions were tested for BoHV-1 using FAT and IHC; 55 (4.2%) were positive. After introduction of PCR testing, 194 animals were tested for BoHV-1 (132 using PCR), and 12 (6.2%) were positive. There was no evidence that these proportions were different (P = 0.25).
Frequency of use of Bovine herpesvirus 1 assays used at 5 U.S.-based veterinary diagnostic laboratories from 2000 to 2011.
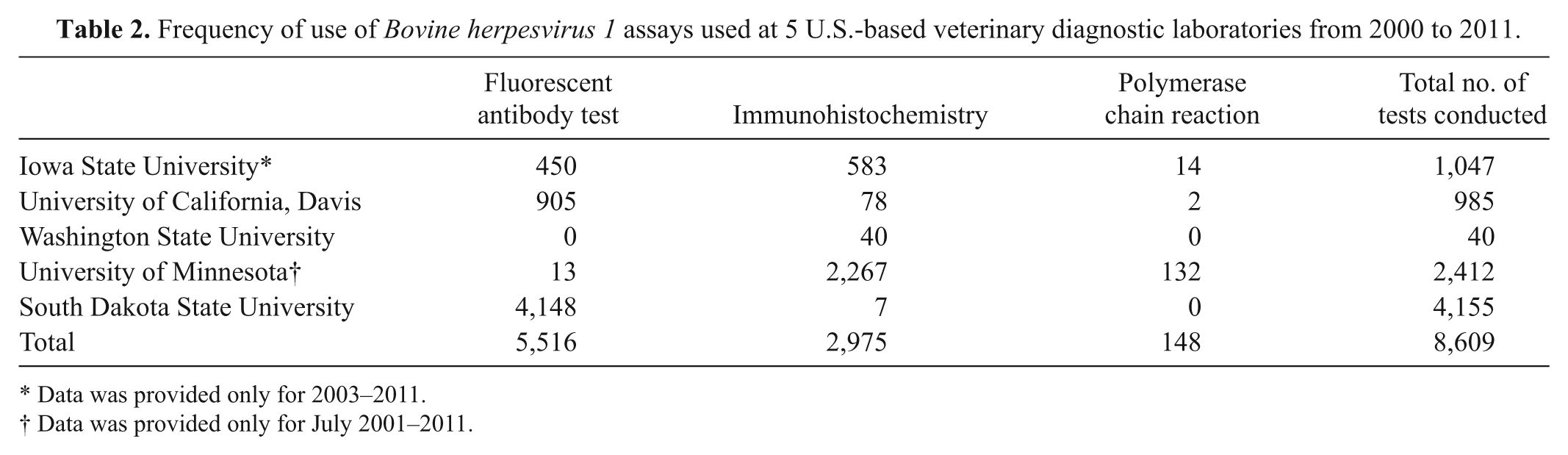
Data was provided only for 2003–2011.
Data was provided only for July 2001–2011.
For the second aim, due to unavailability of records, 345 of the 528 submissions were included in the analysis. The histopathology report and the assessment of the diagnostician agreed in 321 out of 345 (93%) submissions (kappa = 0.004). Of the 321 agreements, 153 were BoHV-1–positive submissions. Of the 24 nonagreements, 5 cases were BoHV-1 assay negative, but the diagnostician reported that the histopathology report appeared consistent with IBR infection. There were 19 BoHV-1 assay–positive cases where the diagnostician reported that the histopathology report did not appear to be consistent with IBR. There were 7 cases that had a histopathology description available but it was vague or noninformative, 2 cases had a histopathology description that was consistent with another etiology, 7 cases had a histopathology description of no diagnostic lesions, and 3 cases had a histopathology description of tissue autolysis.
For the third aim, farm history of vaccination of 245 positive submissions and 245 random negatives was identified, making 245 matched pairs representing 490 submissions; however, only 38 of the 245 matched pairs had complete data. Of the 38 pairs, 22 submissions had a history of vaccination for BoHV-1, 3 submissions had no history of vaccination for BoHV-1, and 13 had 1 vaccinated and 1 not vaccinated. There was no evidence of an association between farm history of vaccination and BoHV-1–positive abortion submissions (McNemars test statistics = 1.23, degrees of freedom = 1, P = 0.27). The number of pairs with complete data was very low because 324 of the randomly selected 490 accessions did not have any farm history data. Given the incomplete records, an analysis was conducted ignoring the matches (i.e., a cross-tabulation of data from any of the BoHV-1–positive and –negative submissions that included vaccine history). Using this data arrangement, of the 86 BoHV-1–positive submissions, 62 reported using a BoHV-1 vaccine and 24 did not (24/86; 27.9%). Of the 80 BoHV-1–negative submissions, 68 reported using a BoHV-1 vaccine and 12 did not (12/80; 15%). The data indicates that the failure to report a history of vaccination against BoHV-1 was almost twice as common in BoHV-1–positive submissions as BoHV-1–negative submissions (27.9% vs. 15%; chi-square P value of 0.068 for nonmatched data).
As found in other studies, the data shows that the diagnosis of BoHV-1–associated abortion is rare (264/6,948, 3.8% tested for BoHV-1; 264/19,459, 1.4% of total bovine abortions). This period prevalence estimate is consistent with prior published reports.2,6 A retrospective study 2 based on 10 years of data conducted at South Dakota Animal Disease Research and Diagnostic Laboratory reported data on viral diagnoses associated with bovine abortions and stillbirths. The study reported that of 8,962 abortions tested for BoHV-1, 485 (5.4%) were positive for BoHV-1. 2 The study also reported a 50% decrease of BoHV-1 abortions from 1971 to 1991. Another 18-year study 6 conducted at the Diagnostic Center for Population and Animal Health, which received aborted bovine fetuses from Wisconsin, Indiana, Ohio, and Minnesota, reported that out of 1,618 bovine abortions submissions, 3.2% were attributed to IBR. The study focused on the prevalence of BoHV-1 and the specific diagnosis of the cause of the abortions versus looking for an increase of BoHV-1 over time and therefore did not discuss trends in BoHV-1 diagnosis. Despite the consistency with prior reports, the results from the current study might not be predictive of results at other laboratories as the original request for participation from laboratories was not the result of a random selection process.
For the first aim, analyses were not able to detect consistent changes in prevalence during the study periods (i.e., only at 1 laboratory). Therefore, the hypothesis that PCR was associated with increased prevalence could be assessed at only 1 laboratory. No evidence was found to support an association between the use of PCR and an increase in BoHV-1–associated abortion diagnoses. This may be partially due to the infrequent usage of PCR for diagnosing BoHV-1 abortions. PCR-based detections methods are more expensive than IHC and FAT, and without compelling evidence that increased sensitivity is needed, the motivation to use a more expensive test may not exist.
For the second aim, strong agreement was found, as expected, between the diagnostician and the histopathology reports. One concern is that although the diagnostician of the present study was blinded to the assay results, the original author of the report was certainly not; therefore, it might be argued that the agreement was not unexpected and perhaps should have been higher. Given this potential bias (i.e., 2 observations are not necessarily independently made), it would not be proposed that assay results be considered as a substitute for histopathology reports.
For the third aim, it was found that, of the complete data provided, fewer BoHV-1–positive submissions had a reported history of vaccination compared to BoHV-1–negative submissions. A major issue identified herein was the inadequacy of the original submission forms. This is a consistent concern with retrospective analyses of diagnostic laboratory data that is passively collected. Data submitted to diagnostic laboratories may represent a biased population (i.e., producers that submit specimens may be fundamentally different from those that do not submit). To then have further selection bias based on the large amount of missing data leaves even greater potential for selection bias. It is suspected that the direction of the bias for these data is toward the null. It is proposed that those who submit and those who complete forms may tend to be more conscientious producers; therefore, the observed association between vaccination history and BoHV-1–positive abortions may be even stronger than that observed.
Footnotes
Acknowledgements
The authors thank Larry Holler, Randy Berghefer, Daniel Able, Timothy Baszler, James Evermann, Lucy Gomes, Mark Anderson, Mary Thurn, and James Collins.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Support for this project was provided by a grant from Pfizer Animal Health. As a courtesy, Pfizer Animal Health was provided with the publication after preparation, and prior to submission for publication, but did not alter or edit the results or interpretation.
