Abstract
The prevalence of organisms known to be associated with bovine respiratory disease (BRD) was investigated in cattle prior to export. A quantitative reverse transcription polymerase chain reaction assay was used to detect nucleic acids from the following viruses and bacteria in nasal swab samples: Bovine coronavirus (BoCV;
Introduction
Respiratory disease associated with long distance transport of animals was first reported in the scientific literature in 1925, although the disease had been recognized by veterinarians for at least 20 years prior to that publication. 34 Bovine respiratory disease (BRD) is an important cause of death in cattle exported live from Australia.47,49 Morbidity and mortality due to BRD is the culmination of a combination of host, potential pathogen, and environmental risk factors. As the pathogens involved in fatal BRD on live export voyages from Australia are similar to those associated with BRD in feedlot cattle, 47 it is likely that the pathogenesis of BRD in these 2 cattle productions systems is similar.
Inconsistent access to feed and water, exposure to noise and vibration during transportation, exposure to a new environment, and comingling with unfamiliar animals activate the stress response in cattle (Sainsbury D, Sainsbury P: 1988, Livestock health and housing.
The Australian Government requires animals destined for export to be held in a registered facility (also known as an “assembly depot”) for a minimum period of time prior to being loaded onto a vessel (Australian Standards for the Export of Livestock, Standard 3, http://www.agriculture.gov.au/biosecurity/export/live-animals/advisory-notices/2011/export_advisory_notice_2011-01). Time in the assembly depot allows the animals to recover from road transport, acclimatize to shipboard rations, and receive any health checks or treatments required by the importing country. However, comingling, handling, and sorting of animals that have been recently transported from multiple farms also provides an ideal environment for transmission of viruses and bacteria between animals in the assembly depot.
Viruses implicated in the pathogenesis of BRD include
The current study reports the pre-export prevalence of viruses and bacteria associated with BRD in Australian live export cattle and identifies a number of factors that influence the prevalence of viruses and bacteria in nasal secretions. The seroprevalence of BoHV-1, BRSV, BVDV-1, and BPIV-3 were also determined. The baseline data generated by the present study will be used to develop pathogen management strategies, both on-farm and at the assembly depot, with a view to reducing the impact of important organisms on morbidity and mortality during live export voyages.
Materials and methods
Study population
Nasal swab samples were collected from 1,484 cattle on 4 voyages between December 2010 and May 2012. The number of cattle sampled represented 1–5% of the total cattle on the voyage (Table 1). The percentage of animals sampled from each cohort ranged from 1.3% to 12.5% (mean = 5.0, median = 4.9, standard error = 1.18).
Voyage data for voyages from which nasal swabs and/or serological samples were collected.
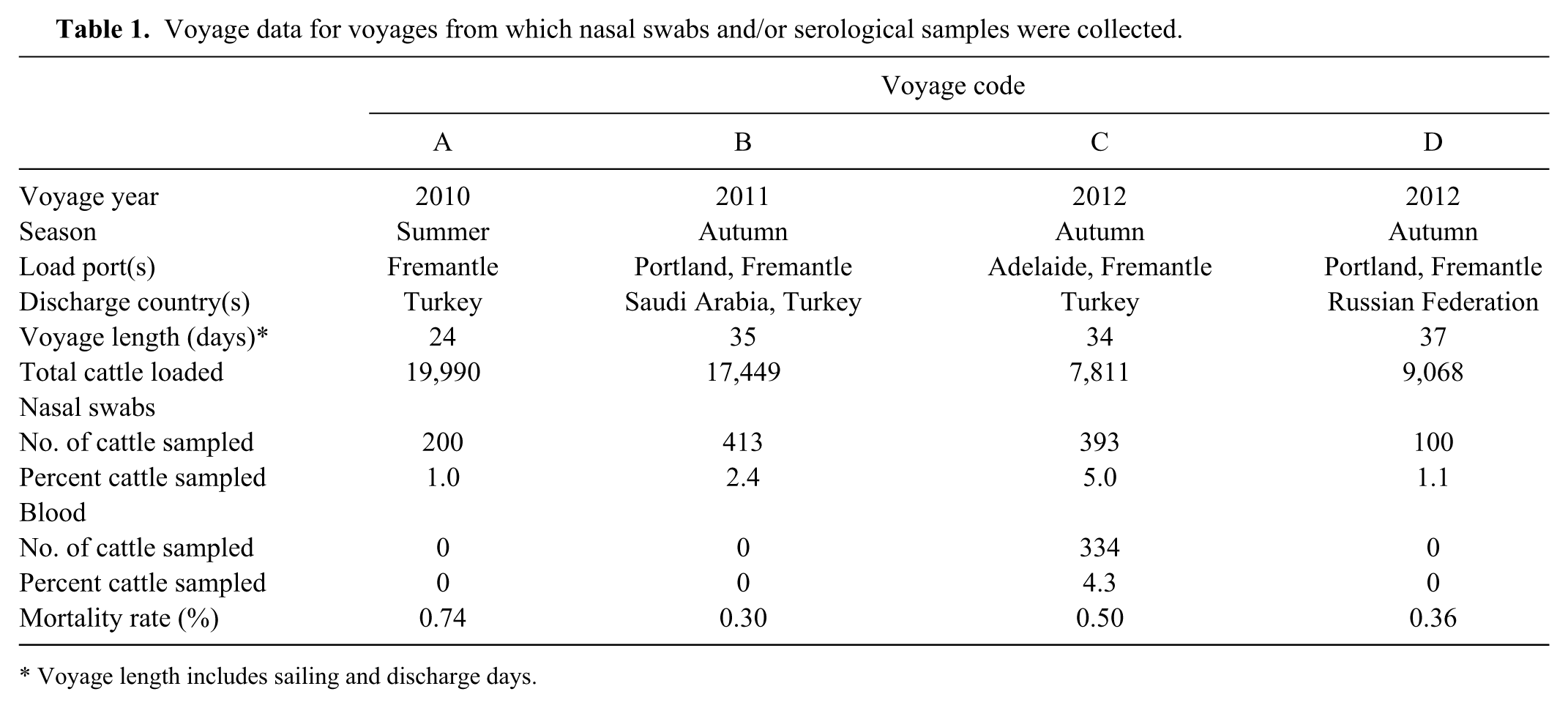
Voyage length includes sailing and discharge days.
Cattle were sampled at 8 assembly depots across 3 states (Table 2): 6 depots in Western Australia (depot codes 1–6) all loading out of Fremantle, 1 in Victoria (depot code 7, load port = Portland), and 1 in South Australia (depot code 8, load port = Adelaide). Animals were grouped into 14 cohorts based on class (feeder, slaughter, breeder) and load-out port (Fremantle, Portland, Adelaide; Table 2).
Voyage, assembly depot, and animal data for cohorts from which nasal swabs ± blood were collected.*
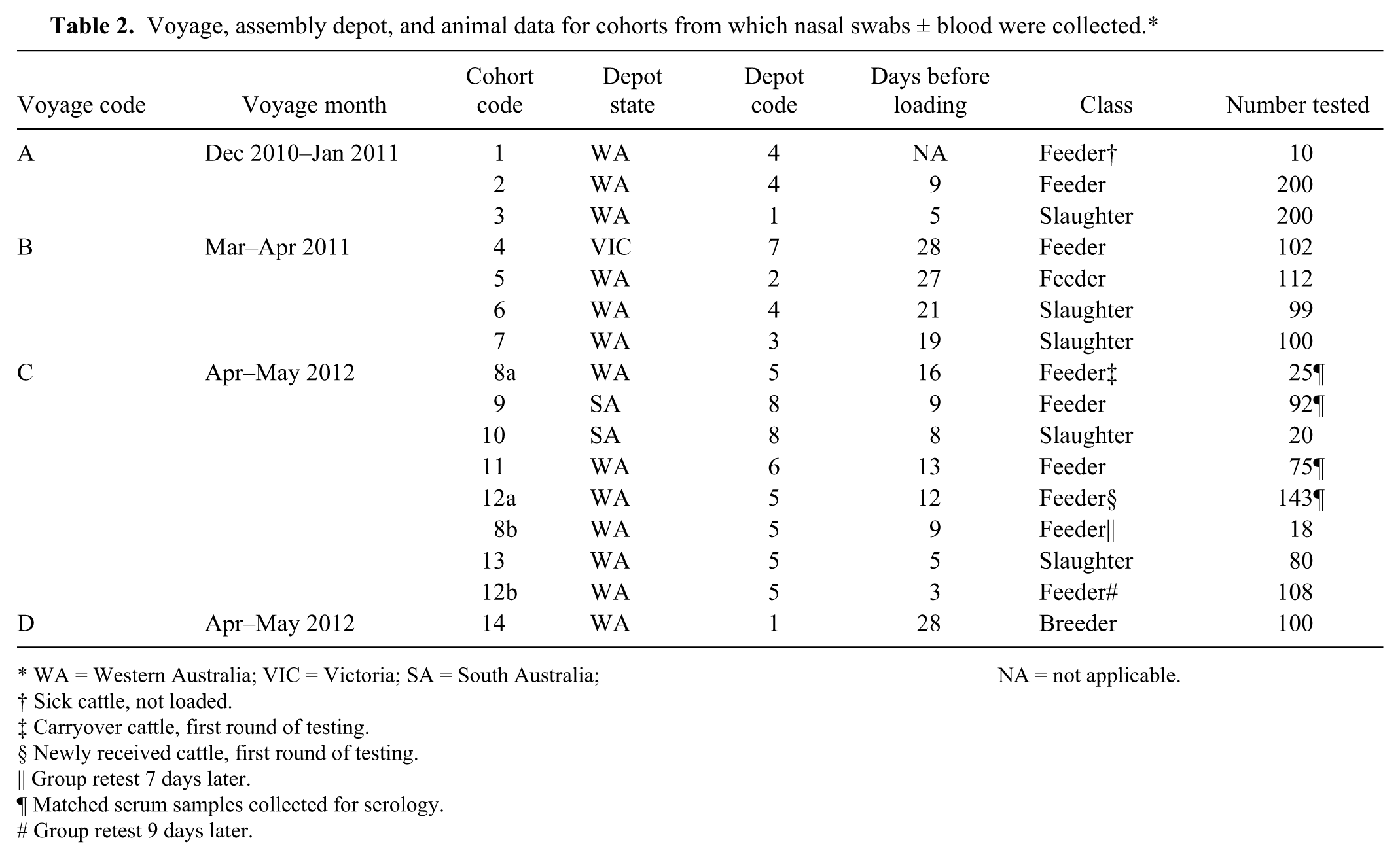
WA = Western Australia; VIC = Victoria; SA = South Australia; NA = not applicable.
Sick cattle, not loaded.
Carryover cattle, first round of testing.
Newly received cattle, first round of testing.
Group retest 7 days later.
Matched serum samples collected for serology.
Group retest 9 days later.
Selection of voyages for sampling was opportunistic and based on the depot induction timetable and location of the assembly depots. Cohorts of cattle were selected to provide a representative subset of the final composition of the shipment (with regard to load port, class, type, and sex) from as many depots and properties as possible, and to collect enough samples to be able to perform statistical comparisons between explanatory variables, while minimizing the impact of study sampling on processing of cattle.
Data collected from each animal at sampling included the radio frequency identification (RFID), property identification code (PIC), breed, sex, and weight. With the exception of RFID, not all data were available for all animals.
For voyages A–C, the consignments comprised a mixture of feeder and slaughter cattle. Voyage D was a shipment of breeder cattle to the Russian Federation. Overall, the class breakdown was 60% feeder, 34% slaughter, and 6% breeder. Eighty-four percent of cattle were
As per import protocol requirements, all feeder cattle were vaccinated against BoHV-1, and breeder cattle on voyage D were vaccinated against BoHV-1, BVDV-1, and BRSV. All vaccinations were administered as a single shot at depot entry.
Nasal swab samples
Nasal swabs were collected from live export cattle during induction processing up to 24 hr after the cattle entered the pre-export assembly depot. All nasal swab sampling was performed by a single member of the project team (SJ Moore) with the exception of sampling of slaughter cattle at depot 7 (cohort 10) that was undertaken by an Australian Quarantine and Inspection Service–accredited third-party veterinarian.
For nasal swab collection, the animal was restrained in a head bail and a 20-cm cotton swab was inserted approximately 10 cm into the nasal cavity. The swab was rotated across the nasal mucosa to collect a sample of the nasal secretions. During both insertion and removal, care was taken to prevent the swab being contaminated by dirt et cetera on the nostrils. The swab was immediately placed into a 5-ml plastic tube filled with 1–2-ml viral transport media (VTM; Hanks balanced salt solution, penicillin G [1,000 units/ml], streptomycin [25 mg/ml], and amphotericin B [0.1 mg/ml]; Department of Agriculture and Food, Western Australia) and kept chilled until transport back to the laboratory.
To investigate whether the prevalence of viral and bacterial shedding changed with time, 2 cohorts of animals were sampled 7 days (cohorts 8a and 8b) and 9 days (cohorts 12a and 12b) apart. Cohorts 8a and 8b were carryover animals from a previous shipment, and cohorts 12a and 12b were newly received animals (Table 2). It was not possible to select the same individuals for sampling at the first and second sampling sessions, but animals that were sampled at the second session were selected from pens containing animals that had been sampled at the first session. Nasal swab samples were processed, and nucleic acids from the organisms commonly associated with BRD
52
—BoCV, BoHV-1, BVDV-1, BRSV, BPIV-3,
Serological samples
Serological samples were collected from animals from a single voyage (voyage C; Table 1). Serum and nasal swab samples collected from the same animal at a single time-point (depot entry) were available for 334 feeder cattle: a single cohort of 91 animals from South Australia, and 3 cohorts from Western Australia, a total of 243 animals (Table 2). Cohort 8a comprised animals that had been carried over from a previous shipment. Cohorts 9, 11, and 12a were newly received animals.
Whole blood was collected at entry to the pre-export assembly depot as part of the pre-export health checks required by the importing country. While the animal was restrained in a crush, blood was collected into sterile plain Vacutainer tubes
a
by venipuncture of the coccygeal vein. Serum samples were sent to the Department of Agriculture and Food, Western Australia, for commercial
Aliquots of 200 μl of serum were tested for the presence of circulating antibodies to the viruses of interest using commercially available enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer’s instructions for the following viruses: BoHV-1, b BRSV, c BVDV-1, d and BPIV-3. e It was not possible to test for antibodies to BoCV because a validated commercial test is not available. Samples with optical density reading above the level set by the kit manufacturer were considered positive, below negative, and in-between were inconclusive. Inconclusive samples were not retested, and animals with inconclusive test results were not included in the analysis.
Statistical analysis
Sample size estimates were based on power analyses conducted using Win Episcope 2.0 (http://www.clive.ed.ac.uk), suggesting that 20–100 samples per property of origin would be sufficient to allow description of prevalence and preliminary assessment of association between risk factors and pathogen prevalence while adjusting for property level clustering. The final approach involved sampling 20–25 animals per property and up to 4–5 properties per cohort.
The statistical software package R was used for descriptive statistics 55 and commercial software f for regression analyses. Ninety-five percent confidence intervals for descriptive prevalence estimates were generated using the Wilson approach and expressed as percentages.7,48 Overall prevalence estimates were generated as an average of the individual cohort prevalence estimates, and as an overall individual animal prevalence.
Negative binomial regression with adjustment for clustering at the property of origin (PIC) level was used to investigate associations between the apparent prevalence of viruses and bacteria of interest and available explanatory variables: voyage, sampling month, sampling year, assembly depot, assembly depot state (Western Australia, Victoria, South Australia), sex, type (
Results
Nasal prevalence
One or more of the viruses or bacteria of interest were detected in the nasal swabs from 1,150 out of 1,484 (77%) cattle. The remaining 334 (23%) animals were negative for all organisms tested. Pathogen prevalences for all viruses and bacteria were found to vary significantly between cohorts (
Nasal pathogen prevalences for viruses and bacteria of interest can be found in Table 3. Bovine coronavirus was the most commonly detected virus (Table 3). The remaining viruses were present in cattle from 2 (BoHV-1), 3 (BVDV-1, BPIV-3), or 4 (BRSV) voyages but generally at low prevalences. Exceptions included the prevalence of BRSV for voyage D (10%) and the prevalence of BVDV-1 for 2 cohorts in voyage C (cohort 9 = 12%, cohort 11 = 10%; Table 3).
Cohort and individual level pathogen prevalences based on quantitative reverse transcription polymerase chain reaction testing of nasal swab samples. See Table 2 for sampling and animal data for each cohort.*
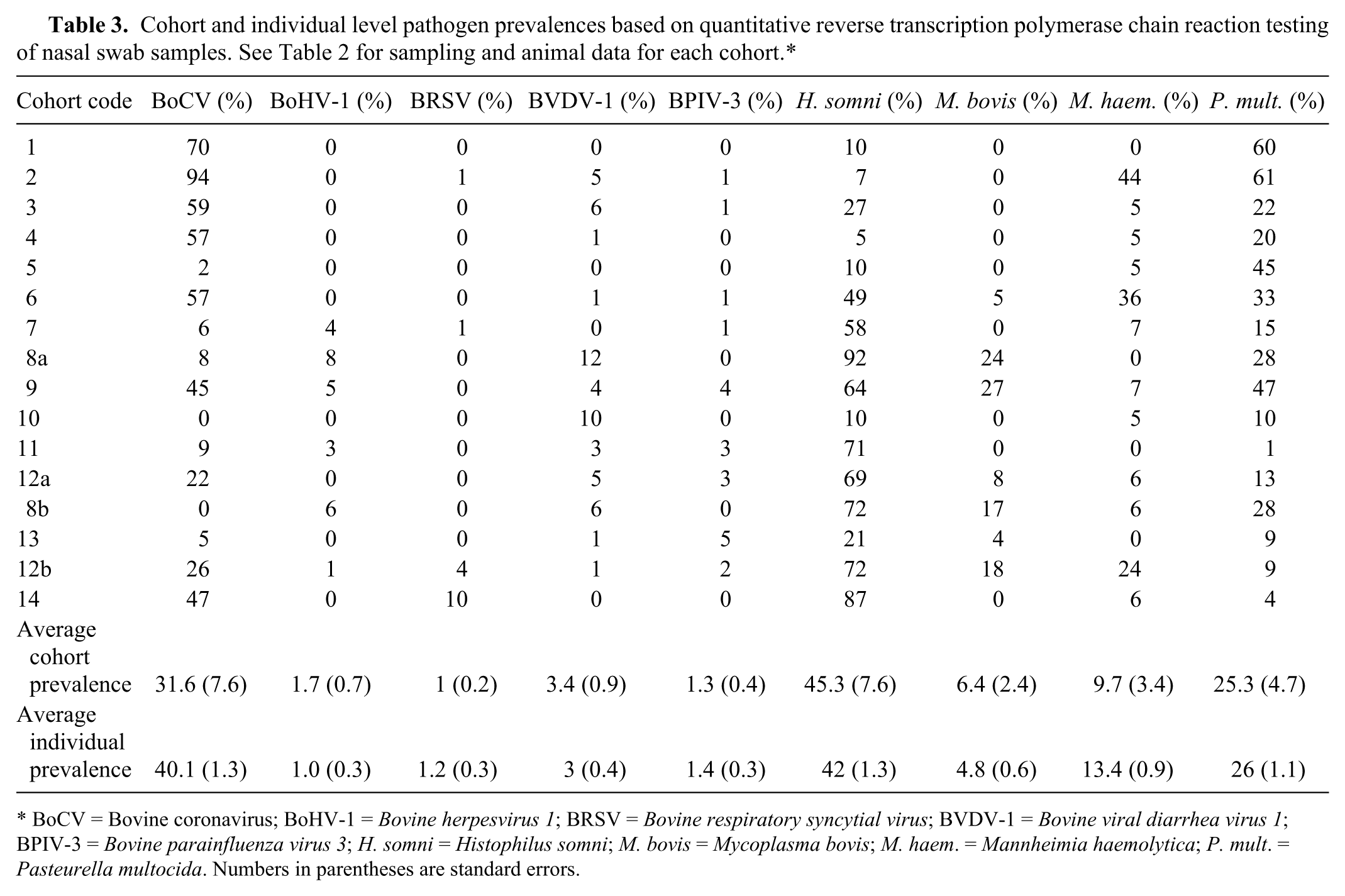
BoCV = Bovine coronavirus; BoHV-1 =
Multiple organisms were detected in nasal swab samples from many animals in the current study. Overall, a single bacterium only was detected in 503 out of 1,150 (43.7%) animals, while 1 or more viruses and bacteria were detected in 434 (37.7%) animals, and 1 or more viruses in 213 (18.5%) animals. Bovine coronavirus was present in 92% of single viral detections and 92% of mixed viral–bacterial detections. The presence of BoCV was significantly correlated with the presence of BRSV,
Of the resampled cohorts, significant decreases in pathogen prevalence were observed for BoCV and
Analysis of nasal prevalence versus explanatory variables
The total number of animals that tested positive for BoCV, BoHV-1, BRSV, BVDV-1, and BPIV-3 was 594, 15, 17, 44, and 21, respectively. For
Negative binomial regression with PIC as a fixed effect showed a number of associations between pathogen prevalence for BoCV,
There were significant differences between voyages, sampling month, and sampling season for all organisms (all
Seroprevalence
Overall, 88% of animals were seropositive for BPIV-3, 58% for BVDV-1, 50% for BRSV, and 43% for BoHV-1 (Table 4). There were significant differences in seroprevalence between PICs (all
Summary data showing the number of animals returning conclusive seroprevalence test results and the seroprevalence estimates (percentage positive [mean] and 95% confidence interval) for 4 sampled cohorts each tested for presence of antibodies to 4 bovine respiratory disease viruses.*
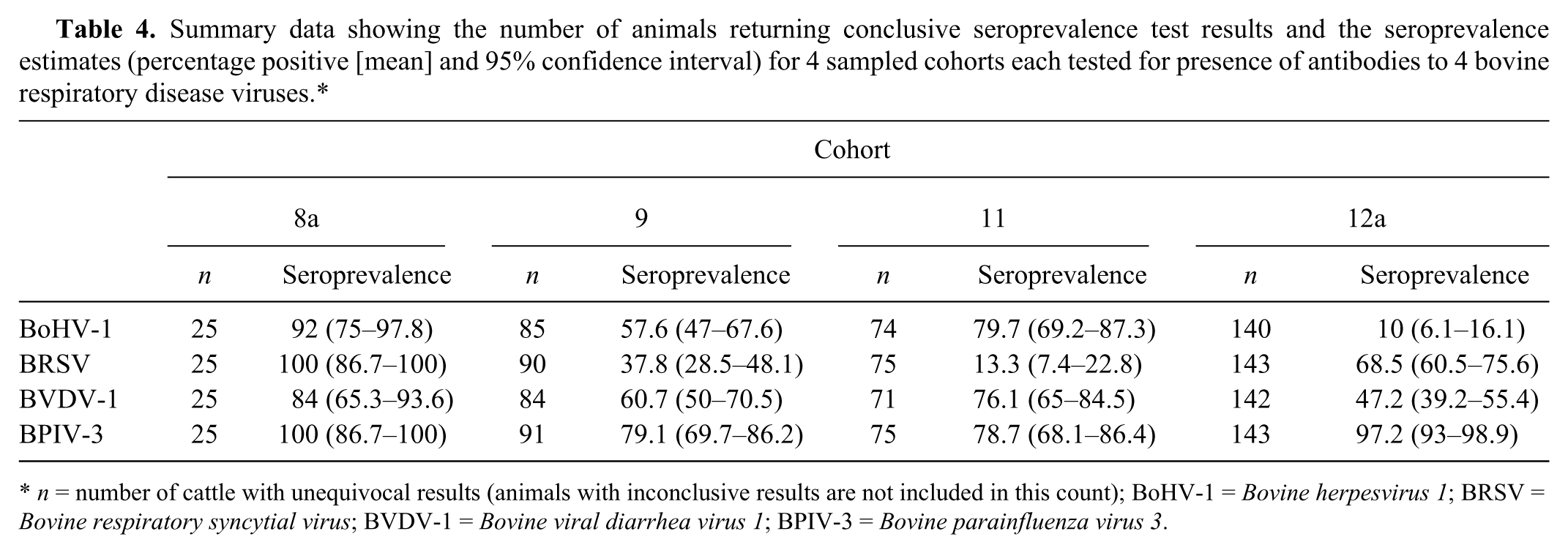
Combinations of seropositive results to single or multiple viruses. Animals with inconclusive results for 1 or more viruses are not included in this table.*
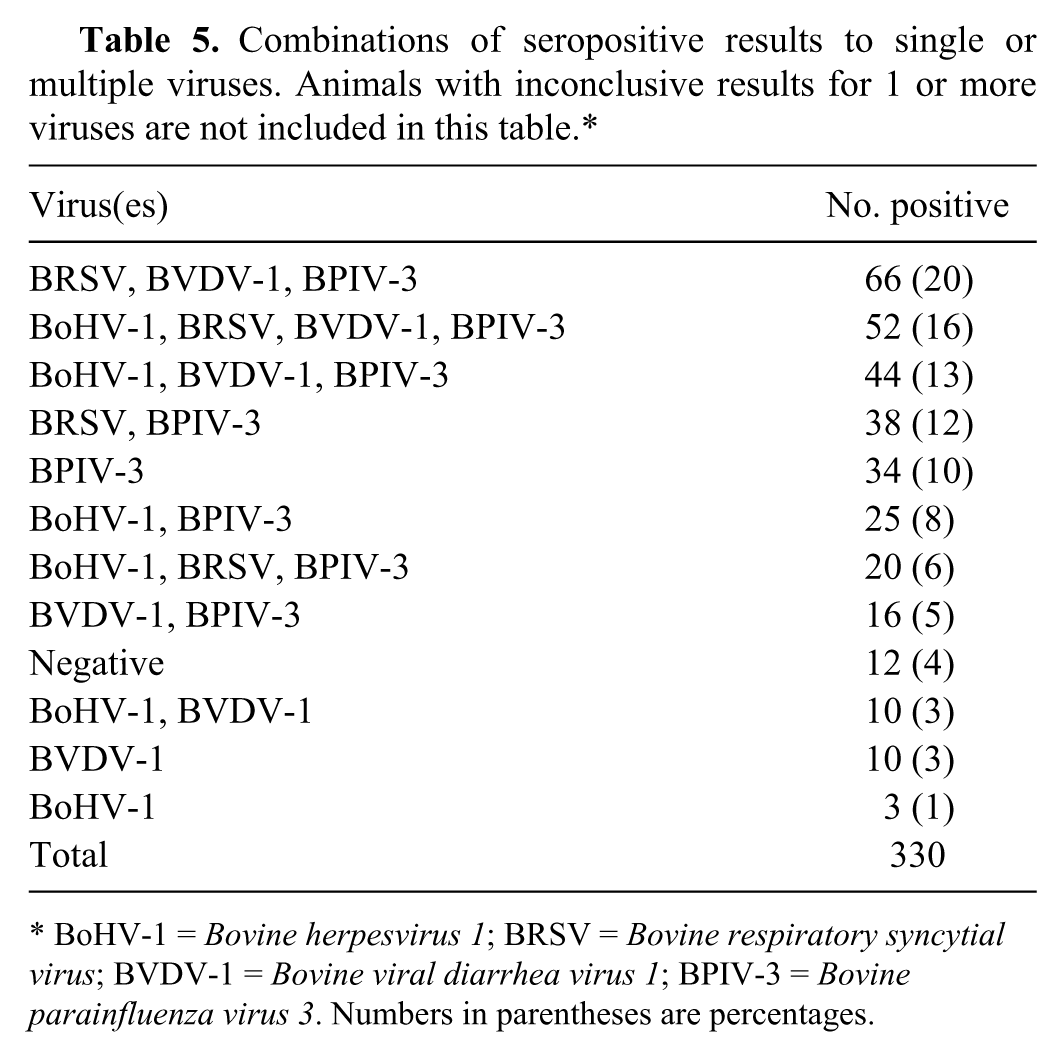
BoHV-1 =
The results from the matched nasal swab and serum samples for each animal were combined to determine that animal’s shedding and serological status for each virus at a single time-point. Animals were placed into 1 of 4 groups: seropositive and nasal swab negative, seronegative and nasal swab negative, seropositive and nasal swab positive, and seronegative and nasal swab positive (Table 6). For all viruses, the proportion of animals that were nasal swab negative and seropositive was significantly higher (all
Serological status and nasal swab results for each virus for newly received animals.*
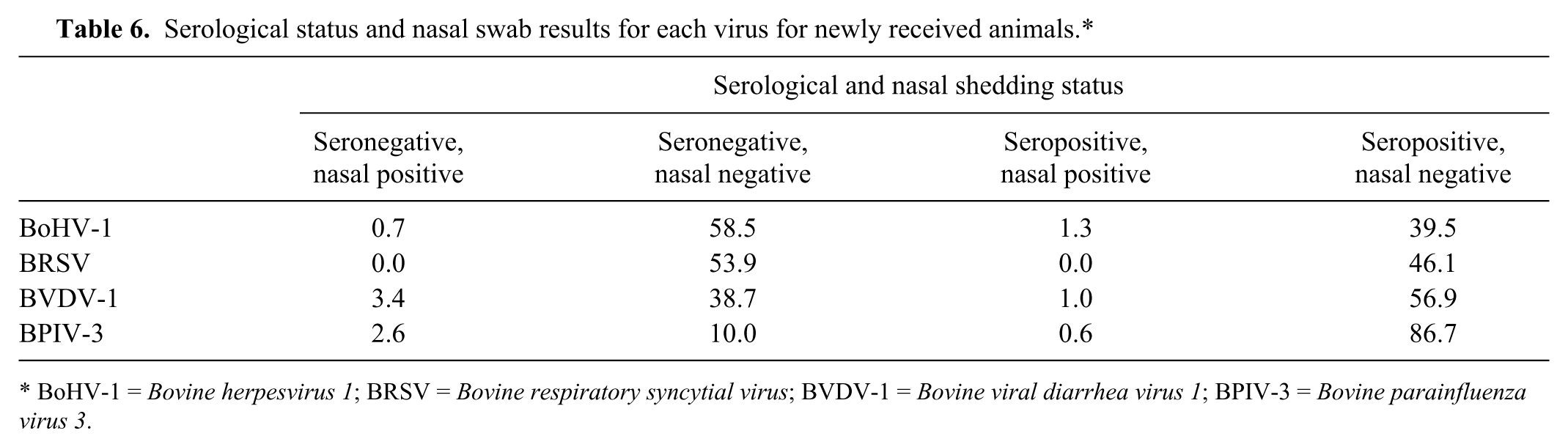
BoHV-1 =
The percentage of animals that were nasal swab positive and seronegative was higher in the carryover cohort (cohort 8a; 3/25 animals; 12%) than in the 3 cohorts of newly received cattle (cohorts 9, 11, and 12; 2.7–4.9%). Animals in cohort 8a had been carried over from a previous shipment for reasons including a positive serum BVDV-1 protein A antibody-capture ELISA (PACE) antigen test.
Discussion
The current study describes a systematic evaluation of the prevalence of organisms known to be associated with BRD, based on qPCR testing of nasal swab samples, for Australian cattle. In addition, to the authors’ knowledge, this is the first time that the seroprevalence of viruses known to be associated with BRD has been determined for Australian live export cattle, although there have been a number of previous serosurveys for these viruses in Australian cattle (Dunn SE, Godwin J, Hoare R, Kirkland PD: 2000, Diseases of feedlot cattle in eastern Australia: Meat Research Corporation project DAN.064 (1990–1994).
The major constraints on the number of samples and range of animals (type, class, sex) that could be collected were finances, time and/or labor, and industry tolerance. Because of the large numbers of animals that must be processed and the economic incentives to minimize the length of time that the animals spend in the assembly depot prior to load-out, animals are protocolled through multiple depots on the same day. Depots may be hundreds or thousands of kilometers apart so it was not possible to sample cattle at all depots on all days.
Nasal swab samples were collected from 1,484 out of 55,902 (2.6%) cattle and matched serum samples from 334 (0.6%) cattle across the 4 study voyages. This means that, particularly in the case of serological results, the prevalences reported for sampled animals may not necessarily represent the prevalence of these organisms across the entire shipment. The collection of paired sera and calculation of seroconversion rates would have increased the power of the current study. It was not possible to access the same animals for a second sampling occasion during the assembly period, and there are no facilities on board the ship to rebleed these animals.
The individual animal prevalence of BoCV was similar to that reported previously for cattle on the south coast of New South Wales with clinical BRD, 35 and within the range of previously reported values for clinically normal beef feedlot cattle (0–85%).10,27,32,39,40,54,71 An association between nasal shedding of BoCV and clinical signs of respiratory disease has been reported in some studies32,66,67,71 but not others.10,33 The presence of BoCV in nasal secretions has been linked to increased odds of death due to BRD during live export voyages, 47 and an increased risk of treatment for respiratory disease,27,38,54 reduced weight gain, 71 and increased incidence of pulmonary lesions at slaughter 38 in feedlot cattle.
Serological testing for BoCV antibodies was not performed as part of the study reported herein due to a lack of a commercially available test kit. However, animals with BoCV in their nasal swabs tend to have low serological titers for BoCV antibodies,32,54,71 and low antibody titers at feedlot entry are associated with an increased likelihood of developing clinical respiratory disease38,53,71 and requiring treatment for respiratory disease.27,54 It is therefore likely that live export cattle that are shedding BoCV on entry to the assembly depot will have low antibody titers and will thus be more likely to develop and require treatment for respiratory disease during voyages than animals without BoCV in their nasal secretions.
To the authors’ knowledge, BVDV has not been previously detected in individual animal nasal swabs from naturally exposed cattle including beef and dairy cattle with clinical BRD,13,66 and beef calves at feedlot entry.22,24 The lack of previous reports of BVDV in nasal swabs may be unexpected considering that BVDV is a well-known BRD pathogen. However, this is likely to be a methodological rather than an epidemiological effect; most previous BVDV studies have used serology or detection of virus in blood or tissue samples to demonstrate infection, rather than PCR testing of nasal swab samples.
The seroprevalence of BVDV-1 (56%) was within the range of previously reported values for Australian cattle (45–77%; Dunn SE, et al.: 1995, Diseases of feedlot cattle),15,41,64,69 and similar to the “approximately 60%” reported for Western Australian live export cattle in 1985. 41 Seroconversion during the feeding period is linked to an increased likelihood of requiring treatment for BRD. 50
In the present study, the 16 animals that were nasal swab positive and seropositive or seronegative for BVDV-1 were either transiently infected and captured immediately prior to, or soon after, mounting an immune response, or they were persistently infected (PI). Persistently infected animals have viral antigen in their nasal secretions and are mostly seronegative, although they may be seropositive if they have been exposed to a heterologous BVDV-1 strain.5,46
In feedlots, PI animals can infect 70–100% of susceptible contacts in a pen.22,25 Acute BVDV infection is associated with impaired function of cells of the innate and acquired immune systems, in particular lymphoid cells, which predisposes newly infected animals to secondary bacterial infections.21,57 The risk of initial treatment for BRD was reported to be 43% greater in cattle exposed to PI cattle than unexposed cattle, 42 although another study did not find an association between exposure to a PI animal and an increase in BRD prevalence. 51 Consideration should be given to the management of potential PI animals, particularly carryover animals, during the assembly period to minimize the risk of BVDV infection and subsequent development of BRD, bearing in mind that the application of a single PACE test to remove PI animals does not guarantee the removal of the virus from a live export consignment. 47
Thirty-nine percent (122/309) of newly received animals were seropositive for antibodies to BoHV-1. This is higher than that found in a 1967 study on Western Australian cattle (29%) but within the range of previously reported seroprevalences for Australian beef cattle (13–85%; Dunn SE, et al.: 2000, Diseases of feedlot cattle in eastern Australia; Dunn SE, et al.: 1995, Diseases of feedlot cattle).15,64,83 The 61% of animals that were seronegative in the study reported herein were susceptible to infection by pen-mates shedding BoHV-1. However, no significant increase in BoHV-1 nasal prevalence was recorded for newly received animals sampled at feedlot entry and again 9 days later. Therefore, it is likely that BoHV-1 does not play a primary role in the development of BRD during voyages.
The seroprevalence of BRSV in the study reported herein (46%) is higher than that reported from a serosurvey of Australian feedlot cattle (27%; Dunn SE, et al.: 1995, Diseases of feedlot cattle) and lower than that detected in cattle in an outbreak of respiratory disease in New South Wales (100%). 35 Seroconversion during the feeding period has been linked to an increased risk of treatment for BRD.8,71
The seroprevalence for BPIV-3 (87%) was the highest out of the 4 viruses. This, combined with a low nasal prevalence (1.4%) and lack of evidence for an association between BPIV-3 and respiratory disease in Australian live export cattle 47 or Australian (Dunn SE, et al.: 2000, Diseases of feedlot cattle in eastern Australia; Dunn SE, et al.: 1995, Diseases of feedlot cattle) 76 or North American 24 feedlot cattle, suggests that BPIV-3 plays a secondary role, if any, in the development of BRD in Australian live export cattle.
In all previous studies on the prevalence of bacteria in nasal swab samples, bacterial culture was used for isolation and identification of bacteria, while in the present study, culture was not performed prior to PCR detection of bacterial DNA. Bacterial culture has been found to be less sensitive than PCR for identifying bacteria of interest in lung swabs 70 and milk.23,44 Therefore, it is likely that some of the differences in prevalence observed between the current study and previous studies are due to diagnostic sensitivity and sample location.
The prevalence of
The prevalence of
Significant associations were detected between the presence of BoCV and BRSV,
Of the epidemiological factors for which data were available, property of origin had the most significant effect on both nasal and seroprevalence. When differences in nasal prevalence between properties was taken into account, no significant differences were detected between animals from different states, or of different sex, type (
Given that there are significant differences in nasal and seroprevalence between properties, strategies to boost immunity prior to loading (e.g., vaccination) would be more effective if they were applied at the property of origin, rather than once the animals have entered the pre-export assembly depot. However, on-farm vaccination is associated with logistic and planning implications that are not compatible with the existing live export cattle sourcing process. Therefore, while exporters could encourage producers preparing cattle for sale to the live export market to consider on-farm vaccination, the development of single shot, fast acting, efficacious vaccines that can be administered at arrival at the assembly depot may prove to be more cost effective and feasible from a management perspective.
Based on the results of the current study, BoCV would appear to be the viral pathogen of most significance for future studies into feedlot vaccination protocols. The other viruses of interest—BoHV-1, BPIV-3, BRSV, BVDV-1—were present at low prevalences in the assembly depot, and none of these viruses were significantly associated with BRD during voyages. 47 However, it is possible that these 4 viruses may be contributing to the development of BRD through their adverse effects on the respiratory tissues and immune system.52,68
The results of the present study show a mixed level of nasal prevalence and immunity to organisms associated with BRD in animals entering preexport feedlots. Therefore, the implementation of a vaccination strategy to raise antibody levels to the common viral BRD pathogens (e.g., BoCV) prior to export may mitigate the risk of development of BRD during voyages. In addition, informed selection of animals and the application of commingling strategies for animals from different properties have the potential to reduce exposure of naïve animals to BRD potential pathogens and thus reduce losses due to BRD morbidity and mortality during voyages.
Footnotes
Acknowledgements
This project would not have been possible without the cooperation of exporters, third-party Australian Quarantine and Inspection Service–accredited veterinarians, and stockmen. Thank you to Dr. Anne Barnes (Murdoch University) and Dr. John Creeper (Department of Agriculture and Food, Western Australia [DAFWA]) for editorial comments on this article. Special thanks to staff of the Bacteriology and Virology Departments at DAFWA for help with sample processing and testing.
a.
Greiner Bio-One VACUETTE TM, Greiner Bio-One, Kremsmünster, Austria.
b.
Infectious bovine rhinotracheitis (IBR) gB X2 Ab test, IDEXX Laboratories Pty. Ltd., Rydalmere, New South Wales, Australia.
c.
Bovine respiratory syncytial virus (BRSV) IgG antibody test kit, IDEXX Laboratories Pty. Ltd., Rydalmere, New South Wales, Australia.
d.
Bovine viral diarrhoea virus (BVDV) antibody test kit, IDEXX Laboratories Pty. Ltd., Rydalmere, New South Wales, Australia.
e.
Parainfluenza-3 virus (PI3) antibody test kit, IDEXX Montpellier SAS, Montpellier, France.
f.
Stata version 13, StataCorp LP, College Station, TX.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Funding for this project was provided by Meat & Livestock Australia and LiveCorp (project no. W.LIV.0252).
