Abstract
Osteochondrosis is a common and clinically important joint disorder that occurs in human beings and in multiple animal species, most commonly pigs, horses, and dogs. This disorder is defined as a
Osteochondrosis is a common disorder of growth cartilage in domestic animals and humans. 5 In animals, the disease has been described in pigs, 18 dogs, 77 horses, 76 cattle, 53 cats, 94 and rats. 57 It is regarded as the most important cause of leg weakness in swine 55 and is a frequent cause of lameness in young athletic horses 76 and dogs. 45 It also is a clinically important disorder in humans. 90, 100
The etiology of osteochondrosis has been intensively researched; however, these efforts have been hampered by confusion regarding the definition of the disease and a lack of understanding of the pathogenesis, particularly regarding the formation of primary lesions. This appears to be due to the fact that most researchers have focused their studies on lesions that cause clinical signs or are associated with radiographic lesions. At these chronic stages of the disease, however, the information obtained actually reflects secondary degenerative and regenerative processes, rather than the primary process(es). 3
The purpose of this review is to provide an overview of the disease, focusing on the most commonly cited theories, recent research findings, and our own views regarding the etiology and pathogenesis of osteochondrosis in order to provide a better understanding of this apparently complex disease.
Osteochondrosis—An Ambiguous Term That Has Stood the Test of Time
Osteochondrosis or, rather,
In human medicine, osteochondrosis has been defined (in the plural form) as “idiopathic conditions characterized by disorderliness of enchondral ossification, including both chondrogenesis and osteogenesis, that comes upon a formerly normal growth mechanism.”
107p.10
According to Siffert,
107
this definition includes a wide range of different diseases affecting growth cartilage in joints, physeal lines, and tendon and ligament attachments, including Legg-Calve-Perthes disease (epiphyseal osteonecrosis of the proximal femur), Köhler disease (osteonecrosis of the tarsal navicular bone or patella), Osgood-Schlatter disease (apophysitis of the tibial tuberosity), Scheuermann disease (epiphysial osteonecrosis of adjacent vertebral bodies in the thoracic spine), Freiberg infraction (epiphyseal osteonecrosis of the second metatarsal), Sever's disease (apophysitis of calcaneus), and
In veterinary medicine, the terms osteochondritis and osteochondrosis were not used until the 1960s. 71 The first studies of this condition mainly focused on lesions in the canine shoulder, but the term was soon introduced as an appropriate name for the prevalent lesions referred to as “chronic deforming arthrosis” associated with “leg weakness” of pigs. 73 After performing extensive studies on “leg weakness” in pigs, Grøndalen 32p.37 stated that a diagnosis of osteochondrosis should be applied to “local, primarily non-inflammatory disturbances in the endochondral ossification process in the epiphyseal plates and in joint cartilage with no superficial cartilage lesions.” However, at the same time, Reiland 96p106 concluded his extensive studies and stated, “Osteochondrosis was found to be a generalized disease in pigs, characterized by a disturbance of endochondral ossification.” This latter notion became widely accepted and has been used in many textbooks, 18, 88 referring to lesions both of the articular–epiphyseal cartilage complex and the physeal growth cartilage in many species.
We concur that osteochondrosis is a better general term for this condition than osteochondritis, with modifiers (
Enchondral Ossification
There are two regions of specialized growth cartilage that are present at the ends of long bones during the period of skeletal growth and development. The physis, or growth plate, is present on either side of the primary center of ossification and is responsible for longitudinal growth. The epiphyseal cartilage that is present between the secondary centers of ossification and the overlying articular cartilage (articular–epiphyseal cartilage complex) is responsible for producing the shape of the ends of the long bones. In both sites, the growth cartilage is replaced by bone through a sequential process of cell proliferation, extracellular matrix synthesis, cellular hypertrophy, matrix mineralization, and vascular invasion that is termed
The chondrocytes in both the epiphyseal and the physeal growth cartilages are organized into four different zones (Fig. 1). The resting zone contains infrequently dividing chondrocytes that function as precursors to the proliferative zone, in which the cells divide rapidly. In the physeal growth cartilage, the proliferating chondrocytes form easily distinguishable columns, whereas in the epiphyseal growth cartilage the columnar organization is less obvious, with the cells forming clusters rather than columns (see Byers and Brown 8 for a review). Terminally differentiated hypertrophic chondrocytes secrete and maintain a highly specialized matrix, the function of which is to promote calcification of cartilage that serves as a template for bone formation by osteoblasts. In the calcifying zone, clasts remove the transverse septa, 69 which allows invasion of vascular loops and osteoprogenitor cells into the lacunae vacated by the terminal hypertrophic chondrocytes. Vascular endothelial growth factor (VEGF), which is produced by hypertrophic chondrocytes, appears to be the factor responsible and necessary for vascular ingrowth into the growth plate. 27 The invasion of cartilage by blood vessels is required for enchondral ossification, as illustrated by the fact that genetic, biochemical, or mechanical disruption of angiogenesis results in thickening of the epiphyseal growth plate due to expansion of the hypertrophic zone (see Colnot 15 and Hall et al. 43 for reviews).
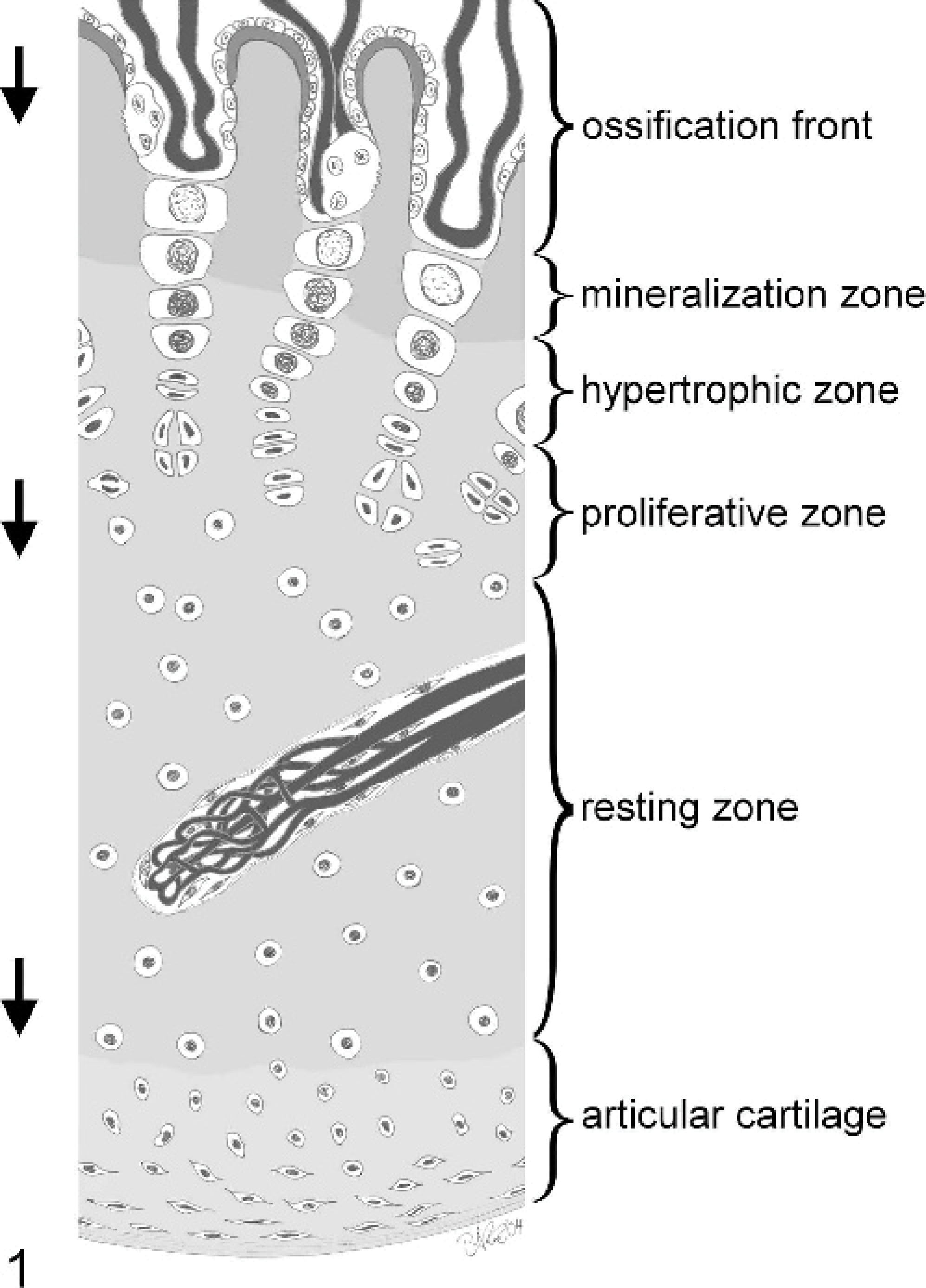
Schematic cross-section of an articular–epiphyseal cartilage complex including the terminal end of a cartilage canal. The cartilage canal contains an arteriole branching into numerous capillaries that rejoin into a venule. In the immature animal, the cells of the proliferative zone will continuously divide, and the cells of the hypertrophic zone will increase in size, causing the cartilage complex to grow in the direction of the arrows. On the ossification front, vessels and bone cells invade the cartilage in the same direction. Because the growth of the cartilage is slightly slower than the progression of the ossification front, the growth cartilage will become successively thinner.
On the ossification front, the osteoblasts envelop the surfaces of the remaining longitudinal septa, which appear in histologic sections as calcified cartilage spiculae, and deposit woven bone on them, thereby forming the primary spongiosa consisting of calcified cartilage, woven bone, and lamellar bone. The latter replaces both calcified cartilage and woven bone after chondroclastic removal and osteoclastic remodeling, respectively. 84 The rate at which differentiation and growth progress, the direction of the process in three-dimensional space, and the time at which the process terminates are thought to be affected by genetic, nutritional, metabolic (including hormonal), and mechanical factors. Regarding the latter, computer models of cartilage mechanobiology have been described, 13, 14 demonstrating that cartilage growth and ossification is accelerated by shear stress (tensile load) and inhibited by hydrostatic stress (compressive load).
Several studies (see Kronenbery 63 , Provot and Schipani 93 , and Goldring et al. 29 for reviews) provide evidence that the proliferation of chondrocytes in the growth plate is under the control of a local feedback loop that is dependent upon temporal and spatial location and primarily involves three signaling molecules synthesized by chondrocytes: parathyroid hormone–related peptide (PTHrP), Indian hedgehog (IHH), and transforming growth factor-beta (TGF-beta). This feedback loop acts to regulate the rate at which chondrocytes leave the proliferative zone and irreversibly commit to terminally differentiated hypertrophic cells. 1 PTHrP, a peptide hormone with homology to parathyroid hormone, is synthesized and secreted by periarticular perichondral cells and by chondrocytes later in development. It facilitates the continuous proliferation of chondrocytes in the growth plate and inhibits chondrocyte hypertrophy. 56 Thus, a pool of proliferating cells is maintained, and premature differentiation into prehypertrophic and hypertrophic chondrocytes is prevented (reviewed in Goldring et al. 29 ). IHH, a member of the hedgehog family of cell surface–associated ligands, is a secreted molecule produced by prehypertrophic and hypertrophic chondrocytes that promotes chondrocyte proliferation and provides the signal to maintain PTHrP expression at the ends of the developing bone. 106 By inhibiting chondrocyte maturation, PTHrP downregulates IHH in the cells near the ends of the bone. 106 Runx2, a transcription factor expressed in chondrocytes as they initiate chondrocyte hypertrophy, is believed to coordinate a proper rate of maturation and proliferation of chondrocytes by activating IHH and, consequently, PTHrP (reviewed by Provot and Schipani 93 ). TGF-beta is produced by perichondrial cells in response to IHH and acts on the perichondrial and periarticular cells to increase PTHrP syntheses (reviewed in Ballock and O'Keefe 1 ). TGF-beta also can act directly on chondrocytes to inhibit hypertrophy Although this PTHrP–IHH–TGF-beta feedback loop currently appears to be the primary regulator of cell proliferation in the growth plate, it is also likely that this regulatory network is modulated by other systemic and local signaling molecules. 1, 63, 93
Cartilage Canals
Although the epiphyseal growth cartilage matrix is macroscopically indistinguishable from the matrix of the overlying articular cartilage, it is distinguished histologically or through perfusion/clearing studies by the presence of vessels that invade the cartilage from the surrounding perichondral plexus
75
and course within channels termed
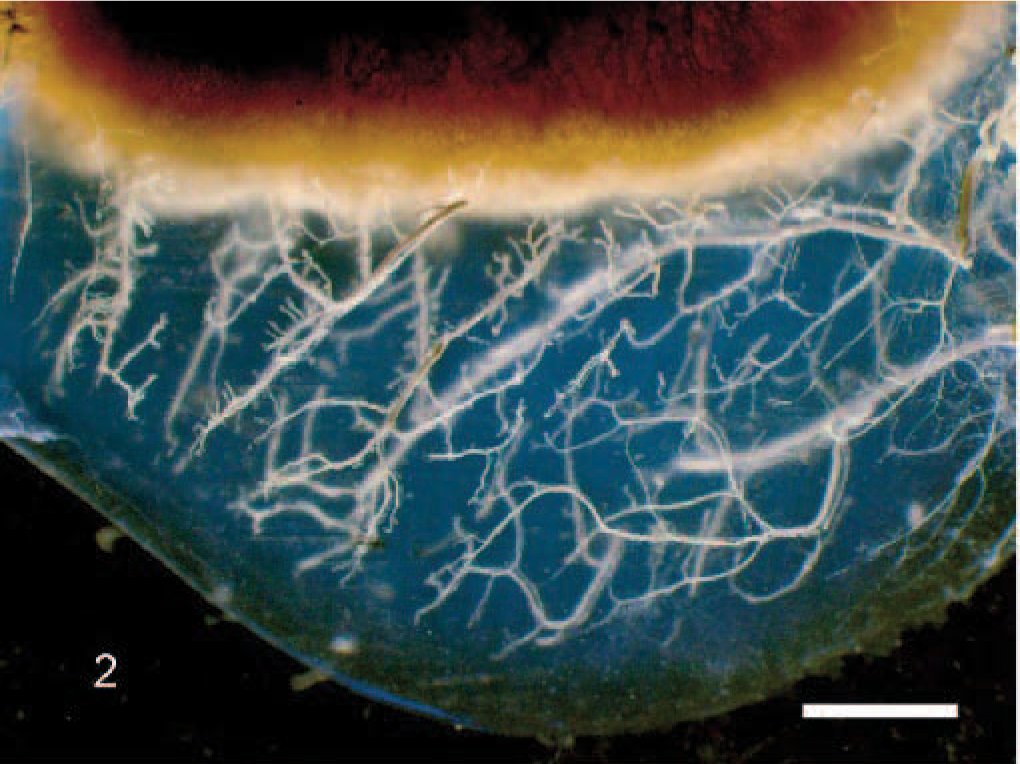
Cartilage canals in the articular–epiphyseal cartilage complex of the medial femoral condyle of a 7-week-old piglet viewed with transillumination. The image is from a 5-mm-thick cut frontal slab from an animal perfused with barium sulphate in which the tissues were cleared with methyl-salicylate (see Ytrehus 134 for full description). The epiphyseal growth cartilage is well vascularized with cartilage canals branching from the abaxial (to the right) and the axial (to the left) perichondrium, whereas the articular cartilage (lowest part) is avascular. Bar = 2 mm.
As the individual grows, the growth rate of the cartilage diminishes compared with the rate of progression of the ossification front, causing the ossification front to advance toward the articular cartilage and resulting in the layer of growth cartilage becoming progressively thinner (Fig. 1). Concurrently, the cartilage canals undergo a physiologic regression process known as chondrification. 42 This term implies a transformation of the cartilage canal in which the vessels regress and the mesenchymal cells are converted into proliferating and matrix-producing chondrocytes that obliterate the lumen of the preexisting canal. 42, 113 This transformation does not adversely affect the adjacent cartilage. By the time the individual reaches adulthood, the epiphyseal growth cartilage is replaced by bone, and no cartilage canals remain.
Manifestations of Osteochondrosis
The principal lesion of both articular and physeal osteochondrosis is a focal failure of enchondral ossification; that is, an area of growth cartilage fails to undergo matrix calcification or vascular invasion, and therefore does not become converted to bone.
19
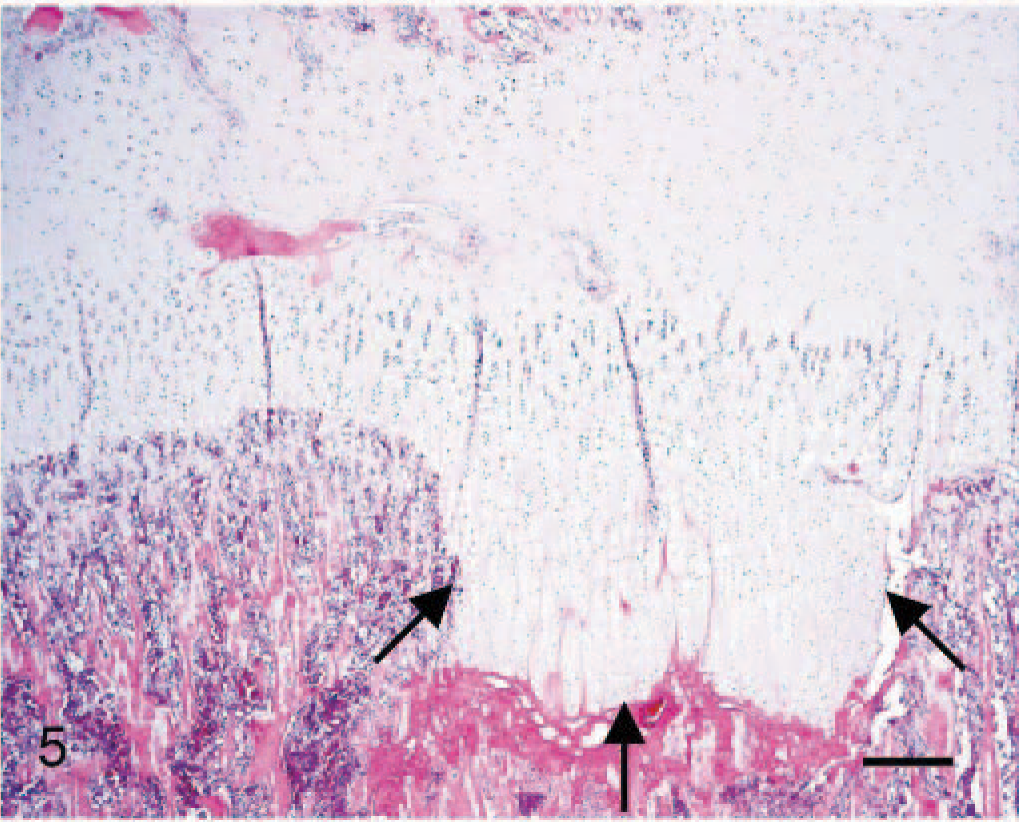
This is grossly visible in cut sections as a locally extensive zone of thickened cartilage accompanied by an irregular/uneven chondro-osseous junction. In more advanced cases, the persisting cartilage is soft and reddish, and the subjacent bone marrow is edematous and hyperemic. However, prior to the point of the disease at which a focal failure of enchondral ossification occurs and is grossly visible, the lesion in articular osteochondrosis is characterized histologically by a focal area of necrosis that is confined to the growth cartilage and involves neither the overlying articular cartilage nor the underlying subchondral bone (Fig. 3). In our previous studies we have designated the presence of a focal area of cartilage necrosis that is confined to the epiphyseal cartilage as
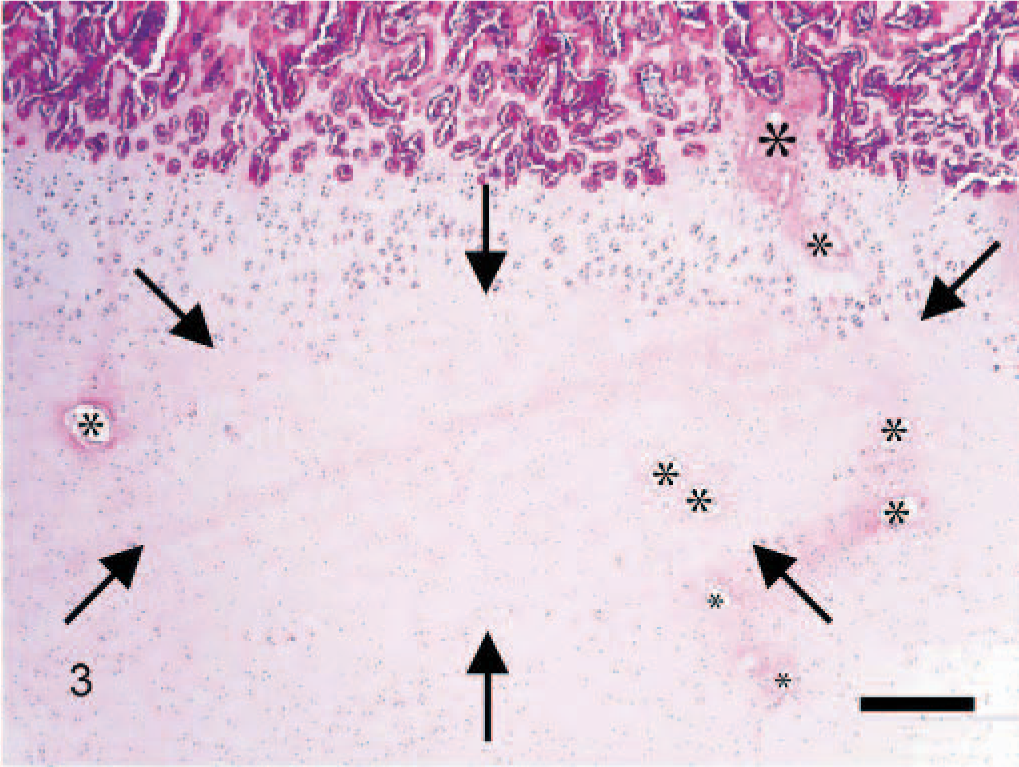
Microscopic section of the medial femur condyle of a 16-week-old pig containing an
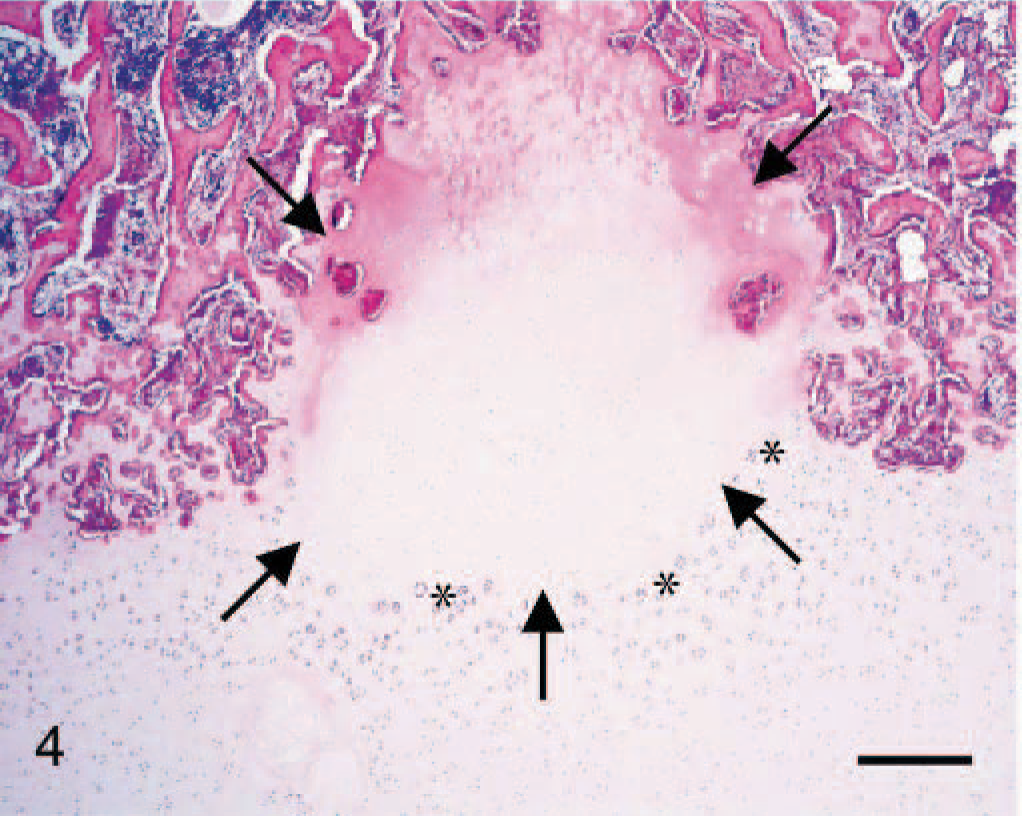
Microscopic section from the medial femoral condyle of a 16-week-old pig containing an
In pigs, typical lesions of articular osteochondrosis are found in the medial aspect and the sagittal ridge of the distal condyle of the humerus and the medial condyle of the femur. In addition, lesions often classified as osteochondrosis are commonly identified in the semilunar notch of ulna and in the distal ulnar physis. Lesions are found less often in the shoulder, hip, and hock joints, in the ischium-tuber ischium apophysis, in the vertebral joints, and in the costochondral junctions of the ribs (for review, see Nakano et al. 82 ).
Lesions may develop in several locations in a single animal and often occur in bilaterally symmetrical sites. However, a generalized distribution of lesions with involvement of hyaline cartilage in locations other than the predilection sites noted above is regarded as rare. Jørgensen et al. 55 and Jørgensen and Andersen 54 concluded that the correlations among the occurrences of lesions of osteochondrosis in different body locations (other than in bilaterally symmetrical sites) were low or insignificant, indicating that osteochondrosis, at least in pigs, is not a generalized disease. The primary changes of osteochondrosis in pigs are commonly observed by the time animals reach an age of 2 months and have been described as early as 2 weeks after birth. 48 Clinical signs of lameness are normally not evident at this age and are most prevalent in young adolescent animals (4–18 months of age). 18
In dogs, articular osteochondrosis is commonly seen in the caudal central aspect of the humeral head, the medial aspect of the distal humeral condyle, the medial and the lateral distal femoral condyles, and the medial and lateral trochlear ridges of the talus. In addition, lesions in the distal ulnar growth plate and the sacrum are often designated as osteochondrosis. 77 Medium- or larger-sized breeds are more commonly affected than smaller ones, and males are affected more often than females. Furthermore, osteochondrosis in specific sites occurs at high frequencies in certain breeds. 71
In horses, osteochondrosis is regarded as a frequent cause of lameness in young athletic individuals.
76
For example, Grøndahl and Dolvik
31
found a prevalence of OCD in the tibiotarsal joint of 14.3% in a radiographic survey of young Standardbred trotters. Unfortunately, the terms osteochondrosis and
In humans, over 50 syndromes (most of them bearing eponymic names) with lesions in a number of anatomic sites have been classified as osteochondroses. 107, 109 The articular osteochondroses with primary involvement of articular and epiphyseal cartilage appear to be similar to articular osteochondrosis in the pig. However, as stated by Bohndorf, 5 definitive knowledge of the course of OCD in vivo and its pathologic–anatomic correlations does not exist in humans. The reason for this is that the only tissues available for study are fragments and loose bodies that are identified at surgery in individuals exhibiting clinical signs of disease. These essentially represent end-stage disease; histologic data from early (subclinical) lesions of OCD are not available.
Due to this lack of consistency in classification, combined with the lack of knowledge about the early manifestations of the lesions in different sites, data on the disease in humans are not easily transferred to animals, or vice versa. However, the age distribution of the patients and the location and radiographic and macroscopic appearance of the lesions of osteochondritis dissecans of the distal, medial, and lateral femoral condyles; the medial aspect of the trochlea of talus; and the distal condyle of the humerus in humans appear to have many similarities to osteochondrosis in the corresponding locations in animals. 5, 99, 100 In fact, as early as 1978 Sten-Erik Olsson 86 suggested that the pathophysiology of osteochondrosis is essentially the same in all species, including humans.
Neither Grøndalen 37 nor Reiland 96 made any definitive nomenclature distinctions between lesions in the articular–epiphyseal cartilage complex and lesions in the physeal growth cartilage. However, while the primary lesions in the articular–epiphyseal cartilage complex are characterized by necrosis of cartilage, 9, 12, 22 the dominating morphologic feature of the lesions of the physeal plate is persistence of chondrocytes with a hypertrophic morphology (Fig. 5). 49 Failure of enchondral ossification in articular osteochondrosis occurs as a consequence of cartilage necrosis, whereas the failure of enchondral ossification seen in the physeal plate appears to be due to an altered development of the hypertrophic chondrocytes.
Microscopic section of the distal ulna of a 20-week-old pig. Physeal osteochondrosis is characterized by retention of hypertrophic chondrocytes (arrows) in the metaphyseal bone. Hematoxylin and eosin. Bar = 100 μm.
While this review focuses on articular osteochondrosis, some comments regarding the pathogenesis of the physeal lesions are warranted. There are two hypothetical vascular models that could explain how lesions in this location may be formed. The first is supported by the results of a recent study in birds that demonstrated that experimentally induced apoptosis of the endothelium lining cartilage canal vessels and, subsequently, the surrounding chondrocytes, initiates lesions of tibial dyschondroplasia, 95 a disease that has many morphologic similarities to physeal osteochondrosis in mammals. 19, 116 Hypothetically, a focal interruption of physeal cartilage canal blood supply followed by death of the chondrocytes closest to the metaphysis (where the ischemia presumably is more severe) would create a plate of abnormal matrix that would resist penetration by metaphyseal vessels when it was reached by the ossification front (Fig. 6). This plate would act as a barrier to the vascular invasion of normal matrix that is subsequently produced by chondrocytes closer to the epiphysis, resulting in persistence of hypertrophic chondrocytes and creating a mass of thickened physeal cartilage in the affected area. Further support for this theory comes from perfusion and histology studies of naturally occurring cases of tibial dyschondroplasia by Thorp and Duff, 115 who proposed that occlusion of epiphyseal cartilage canals caused formation of a band of abnormal cartilage, preventing invasion by metaphyseal vessels.
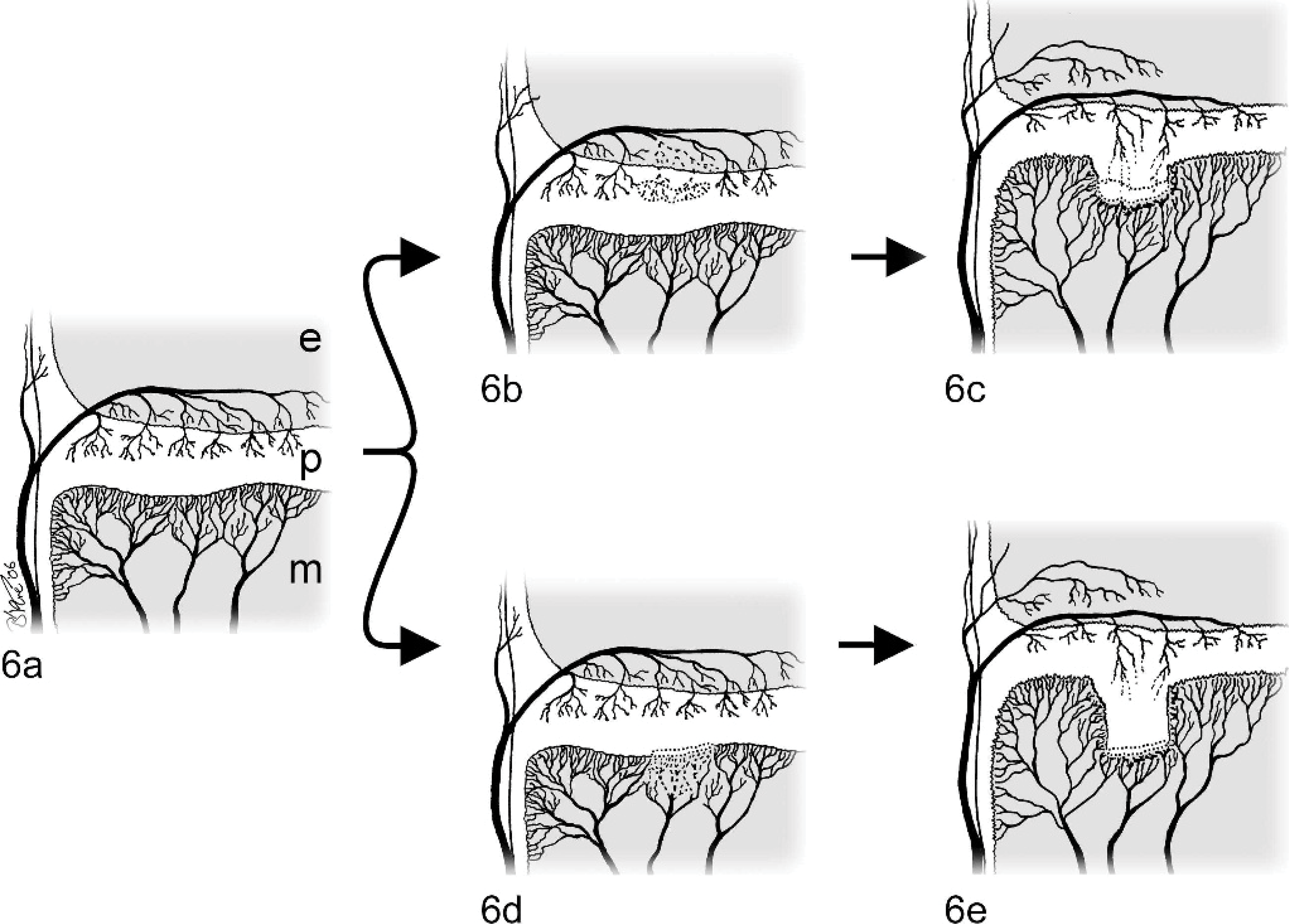
Hypothetical models of the pathogenesis of physeal osteochondrosis. Schematic drawings of sections of a physeal plate in different stages. The region of the physeal plate (p) that is closest to the epiphysis receives its blood supply from cartilage canals crossing into it from the epiphyseal bone marrow (e). The region of the physeal plate that is closest to the metaphysis (m) is largely avascular, but it is invaded by the vessels of the advancing ossification front (
Alternatively, microtrauma to the vessels of the metaphysis could induce an interruption of the blood supply to the ossification front, causing a failure of terminal differentiation of the hypertrophic cartilage and, thereby, persistence of a thickened physeal cartilage (Fig. 6). This theory is supported by studies demonstrating that 1) surgical interruption of the metaphyseal vessels at the ossification front induces lesions of tibial dyschondroplasia in birds, 98 2) interruption of metaphyseal vessels by compression induces persistence of cones of hypertrophic cartilage in the physis of rabbits, 118 and 3) overloading induces formation of lesions of osteochondrosis in the distal epiphyseal plate of pigs. 39 Whether failure of cartilage canal blood supply to the deep physeal resting zone cartilage or failure of metaphyseal blood supply to the ossification front is the initial step in the pathogenesis of physeal osteochondrosis is a subject for further research. In the remainder of this review, we will focus on lesions of the articular–epiphyseal cartilage complex, since the majority of the recent research work has focused on lesions in this site.
Proposed Etiologies of Osteochondrosis
Osteochondrosis is regarded as having a multifactorial etiology, with no single factor accounting for all aspects of the disease. 19, 100 Suggested factors that play a role in this disease include rapid growth, heredity, anatomic characteristics, trauma, dietary factors, and a defect in vascular supply to epiphyseal cartilage (discussed in detail below).
Rapid growth
The increasing frequency of leg weakness in pigs in the middle of the 20th century coincided with a dramatic increase in growth rate in this species, which is attributed largely to increasing economic pressures promoting a reduction in time from birth to slaughter. Hence, it was logical to consider rapid weight gain, either through increased feeding or through heritable traits, as an important predisposing factor for the development of osteochondrosis. Through the years, however, only a limited number of reports have supported this view (see Nakano 82 and Ekman and Carlson 19 for review). Data indicating that rapid growth is not central to the development of osteochondrosis include those demonstrating that the prevalence of lesions is not significantly altered by reducing growth rate through restricted feeding. 11, 34, 81, 128 In addition, animals with a genetically lower growth rate do not necessarily have a lower frequency of osteochondrosis. 121 However, some large population surveys have revealed a positive genetic correlation between rapid growth and prevalence of osteochondrosis. 54, 74
In our recent study of 1,690 offspring of 15 sires, all raised in the same herd under similar conditions, we were unable to demonstrate any correlation between slaughter weight or growth rate from birth to slaughter and prevalence of lesions of
Lundeheim, 74 however, observed that pigs with clinical signs of leg weakness at slaughter had grown more rapidly early in life than pigs without these signs, but they had grown more slowly closer to the time of slaughter. He proposed that an unfavorable relationship between constitution and growth rate was counteracted by discomfort, resulting in reduced feed consumption when the clinical signs of leg weakness began to appear.
It has been reported in dogs that rapid growth increases the incidence of skeletal diseases, including osteochondrosis. 97 Authors supporting this contention often cite the studies of Hedhammar et al. 47 In these studies 24 Great Dane dogs were fed either ad libitum or restricted amounts of a diet rich in protein, energy, calcium, and phosphorus. Two animals from each feeding regime were followed for 9, 12, 24, 36, 48, and 60 weeks before they were euthanatized and necropsied. The main pathologic finding of this study was a relative failure of cartilage maturation and bone resorption in the ad libitum group compared with the restricted group, which was attributed to a relative persistent hypercalcitoninism and hyperparathyroidism. Articular osteochondrosis was found in only 5 dogs; 3 of them (all euthanatized at 24 weeks: 2 on restricted feeding and 1 on ad libitum) had bilateral lesions of the humeral head, 131 whereas 2 others (euthanatized at 48 weeks: 1 on restricted feeding and 1 on ad libitum) had lesions on the articular processes of the cervical vertebra. 17 The fact that the numbers of affected animals in the ad libitum and restricted groups were nearly equal fails to support the hypothesis that rapid growth increases the incidence of osteochondrosis in dogs.
In horses it has been proposed that an increase in circulating insulin and a decrease in circulating thyroxine, which are caused by high-energy diets, may lead to a failure of maturation and hypertrophy of the chondrocytes to which the circulating hormones have greatest access; namely, the chondrocytes surrounding the cartilage canals. 52, 105 According to this hypothesis, chondrocytes under the influence of increased insulin and decreased thyroxine levels persist as cartilage cores. The cores subsequently become necrotic when subjected to biomechanical factors in the transition zone between cartilage and bone, 52 producing lesions similar to osteochondrosis in pigs. However, these theories are contradicted by the morphology of the initial lesions of articular osteochondrosis, which are characterized by sharply demarcated areas of chondrocyte necrosis within the epiphyseal cartilage. 9 The formation of cartilage cores is a subsequent event caused by the resistance of the necrotic cartilage to the processes of mineralization and vascularization. This theory also fails to account for the fact that lesions occur focally at specific predilection sites and are not generalized.
Thus, the majority of the published literature in all species fails to support a direct role of increased growth rate/overnutrition in the etiology of osteochondrosis.
Heredity
Differences in the prevalence of osteochondrosis between different breeds and different breeding lines of pigs, 33, 40, 54, 122 dogs, 66 and horses 31 strongly indicate that there is a heritable component to osteochondrosis. However, estimates of heritability in this disease vary greatly, ranging, for example, from 0.09 to 0.46 for the humeral condyle and from 0.20 to 0.49 for the femoral condyles of the pig. 54, 112, 133 Many authors have suggested that the inherited factor is closely associated with a predisposition toward increased growth rate; however, as mentioned previously, existing data on the association between osteochondrosis and rapid growth are contradictory. 11, 34, 80, 128 In our recent study of the offspring of 15 boars with high breeding values for osteochondrosis, it was found that inherited factors had considerable effects on the prevalence, severity, and location of osteochondrosis, 135 but there was no correlation between rapid growth and lesions of osteochondrosis. Instead, it has been suggested that an important heritable factor in pigs may be anatomic conformation (see below). 136 This also has been suggested in avulsion of the tibial tuberosity in certain dog breeds, a condition that may be the result of physeal osteochondrosis. 108
All of the equine inheritance studies indicate that osteochondrosis is inherited as a polygenetic trait, 91 and similar results have been found in studies in dogs. 87 Exactly which traits are predisposing for the disease and the nature of the interaction between environmental and genetic factors remains to be resolved. Evidence for a genetic contribution to diseases designated as osteochondrosis in humans is provided by case reports of bilateral Köhler disease, 120 Freiberg infraction, 4 and Scheuermann disease, 30, 41 sometimes with a simultaneous onset and parallel clinical course, in identical twins.
Thus, in all species that have been examined, there is compelling evidence that inheritance is important in the etiology of osteochondrosis. Notably, all of these studies have focused on the late stages of the disease (primarily
Anatomic characteristics
According to Schenck,
100
in 1933 the medical surgeon Fairbank had already proposed that repetitive impingement of the tibial spine on the lateral aspect of the medial femoral condyle during internal rotation of the tibia is the causative factor of
Grøndalen
37
measured 29 characteristics of skeletal shape and evaluated macroscopic lesions and exterior conformation in 912 slaughter and breeding pigs. He suggested a connection between exterior features, joint shape, and joint lesions and claimed that an unfavorable joint shape creating local overload was an important cause of
Based on necropsy findings, Olsson 85 hypothesized that local traumatic factors, which may vary among joints, are important in the etiology of osteochondrosis in dogs. He concluded that repeated microtrauma due, for example, to impingement of the dorso-caudal area of the humeral head in the shoulder joint and forced contact between the medial aspect of the lateral femoral condyle and the lateral part of the intercondylar eminence in the stifle joint, was an important factor in lesion prevalence.
Thus, anatomic characteristics appear to be important in the etiology of osteochondrosis; however, these are undoubtedly related, at least to some extent, to genetic factors. Since anatomic characteristics also are difficult to measure with consistency and are altered with disease progression, it is difficult to determine their precise contribution.
Trauma
Trauma has been one of the most widely proposed causes of osteochondrosis in all species (see Bohndorf
5
and Ekman and Carlson
19
for review). In support of a traumatic etiology, predilection sites in all affected species tend to be located in areas of local biomechanical stress. In addition, increased stress (e.g., increased athletic activity in humans)
5
appears to increase the prevalence and severity of macroscopic osteochondral lesions. Experimental studies also provide evidence supporting the role of trauma. Nakano and Aherne
79
found that pigs dropped from varying heights (<1 m) at a young age (average weight: 29 kg), had more lesions of
The role of trauma in the pathogenesis of osteochondrosis may depend on the stage of the disease that is considered. Although trauma may be involved in converting a subclinical
Thus, although major trauma has been documented as causing osteochondral fractures, particularly in humans, its role in the initial development of osteochondrosis appears to be limited, as evidenced by the lack of a history that includes a single impact traumatic event in most animal and human cases. 4, 19, 90, 99 Major trauma alone also fails to explain the fact that lesions occur in specific predilection sites and often are bilaterally symmetric.
Dietary factors
An imbalance in calcium-phosphorus supplementation, copper deficiency, zinc oversupplementation, vitamin C deficiency, vitamin A deficiency, vitamin D deficiency, biotin deficiency, and chronic metabolic acidosis all have been suggested and investigated as etiologic factors of osteochondrosis (see Nakano et al. 82 for a review of work done in pigs). However, experiments with dietary supplementation of the suspected deficient minerals and vitamins or supplementation of dietary sodium bicarbonate to counteract suspected metabolic acidosis have not produced significant effects on the prevalence and/or severity of osteochondrosis in pigs. 82
In a study of dietary effects on skeletal development in dogs, a high-calcium diet caused lesions typical of rickets, which resolved after the calcium intake was normalized. Dogs in all three groups (controls, high-calcium, and high-calcium/high-phosphorus diets) had focal delays in endochondral ossification involving the costochondral junctions of the ribs, the severity of which did not appear to be influenced by treatment group. 101 In another study in which dogs were fed a high–vitamin D3 diet versus a control diet, irregularities in the costal growth plate were observed in both groups and were described as being more severe in the high–vitamin D3 group; however, a quantitative assessment of these lesions was not done. 119
A lesion termed
Thus, the studies that have explored the relationship between dietary factors and osteochondrosis have failed to provide strong evidence of an association. 82 Overall, the literature to date regarding etiologic factors in osteochondrosis provides relatively strong evidence for the roles of genetic and anatomic factors in the etiology of this disease and little or no evidence for rapid growth, major trauma, or dietary factors. 19, 134– 136
Models of Pathogenesis of Osteochondrosis
Fragility of cartilage and/or bone
Grøndalen 37 suggested that “weak cartilage/bone tissue” could be one of the main causes of leg weakness in pigs and that genetic conditions and/or dietary factors could be underlying causes of the weakness. In a review based on studies in Great Dane dogs, Dämmrich 16 suggested that due to overnutrition and accelerated growth, a relative osteopenia of the subchondral bone causes a weakening of the biomechanical bone support for the overlying cartilage, thereby causing a disturbance in chondrocyte metabolism leading to osteochondrosis.
In horses on a diet having very low levels of copper, Hurtig et al.
51
found that in foals with articular lesions, cross-links between hydroxylysine groups on adjacent collagen fibers in cartilage and bone were missing, possibly because of a lack of the copper-dependent enzyme lysyl oxidase. Among other findings, the lesions of copper-deficient horses and bison have been characterized by microfractures in subchondral bone spiculae, diminished formation of primary spongiosa, and thinning/erosion of articular cartilage.
51,
130
Hence, Hurtig et al.
51
suggested a relationship between low copper intake in fast-growing horses, inferior collagen quality, biomechanically weak cartilage and bone, and lesions of
As mentioned previously, the focal nature of the lesions occurring in osteochondrosis make it unlikely that this disease is caused by a generalized weakness in either cartilage or bone per se. It is, on the other hand, theoretically conceivable that biomechanically weak tissues (due to a number of causes) may fail to give support to blood vessels in cartilage and/or bone, thereby lowering the threshold for vascular damage, inducing ischemic necrosis of growth cartilage (see below). However, taking into the account the morphology of the lesions, their high prevalence in otherwise healthy animals, the fact that these lesions occur in well-defined predilection sites, and the relative paucity of conditions that are associated with weakening of cartilage and bone, it is unlikely that formation of fragile supportive tissue plays a main role in the pathogenesis of osteochondrosis.
Primary dyschondroplasia
Olsson and Reiland 86 proposed that osteochondrosis may begin as a generalized abnormality of chondrocyte development and maturation (i.e., dyschondroplasia), leading to multifocally altered enchondral ossification. Ekman and Heinegård 20 suggested that the earliest lesion of osteochondrosis may be a change in the phenotype of chondrocytes, resulting in subsequent alterations in matrix synthesis and assembly compared with normal cartilage. The cause of the phenotypic change was not defined, but one suggestion was that it occurred secondary to ischemia induced by necrosis of vascular channels. 20, 21 These primary lesions would resist the ossification process and ultimately become retained cartilage, susceptible to further damage within the joint. These authors further hypothesized that secondary lesions of cartilage necrosis (osteochondrosis) may occur as a result of biomechanical stress. 52 This model, however, fails to account for several important features of osteochondrosis, including the fact that lesions occur in specific predilection sites that differ among species and that the lesions tend to be bilaterally symmetrical. In our opinion, there is no morphologic evidence for the theory that osteochondrosis occurs due to a generalized abnormality of cartilage development/maturation.
Necrosis of subchondral bone
The primary lesion of osteochondrosis in the human literature often has been interpreted to be necrosis of subchondral bone, 99, 123 with involvement of the overlying cartilage occurring secondarily. 5 According to Nagura, 78 Ludoff in 1908 hypothesized that the necrosis was caused by traumatic damage to the vascular supply of the bone. Furthermore, Smillie 110 concluded that the basic pathology of osteochondritis dissecans, whether of the Juvenile or Adult variety, is local ischemia, relative or absolute. Evidence that is widely used to support the theory that subchondral bone necrosis is the primary lesion of osteochondrosis is the presence of necrotic and viable bone in surgically removed osteocartilaginous flaps or loose bodies. However, as shown by Bravo et al., 6 surgical formation of a cartilaginous flap may induce ingrowth of cartilage canals and the formation of an accessory center of ossification, resulting in the presence of a trabecular bone component in a previously purely cartilaginous flap. Inadequate blood supply to the newly formed center of ossification, possibly caused by motion of the flap, may subsequently lead to necrosis of the bone. The finding of necrotic trabecular bone in osteocartilaginous flaps or loose bodies removed by surgery, therefore, does not prove that they originated from a focus of primary bone necrosis. In addition, the theory that ischemic necrosis of subchondral bone is the primary lesion of osteochondrosis is not supported by the veterinary literature, in which early, subclinical lesions are available for study. 19
Ischemic necrosis of growth cartilage
Studies of spontaneously occurring osteochondrosis in pigs
10,
59,
129
have indicated that the early (subclinical) lesions of osteochondrosis (
One of our recent studies, 134 however, failed to prove an association between a failure of chondrification of cartilage canals and formation of lesions of osteochondrosis, nor could it support an association between rapid growth and chondrification of cartilage canals or between rapid growth and lesions of osteochondrosis. Consequently, the study did not support the assumption that osteochondrosis is caused by a general failure of cartilage canal blood supply. Experimental studies, however, show that interruption of cartilage canal blood supply rapidly causes necrosis of cartilage canal vessels followed by necrosis of the surrounding resting zone growth cartilage, and so creates lesions typical of naturally occurring osteochondrosis. 12, 137 These findings suggest that naturally occurring osteochondrosis is a consequence of a local event that causes failure of a limited number of cartilage canals. In support of this theory, our recent studies of perfused and cleared slabs of articular–epiphyseal cartilage from growing swine reveal that cartilage canal necrosis usually occurs focally and immediately distal to a point where the vessels of the canal cross through the chondro-osseous junction. 136 The necrosis affects the entire canal from this point distally, implying that the primary lesion occurs at the interface between cartilage and bone. These results provide evidence that areas of the articular–epiphyseal cartilage of the distal femur in pigs sustain a shift in blood supply in animals in a certain age window, from cartilage canal vessels originating in the perichondrium to vessels originating in the bone marrow (Fig. 7). As the animal grows and the cartilage becomes thinner, some cartilage canal vessels having a convex course with a nadir in the median part of the cartilage become surrounded by the advancing ossification front and form new anastomoses with bone marrow vessels. It is likely that these new anastomoses, crossing through a zone of intense remodeling activity and surrounded by presumably biomechanically weak cartilage spiculae and primary spongiosa, are subjected to considerable mechanical stress and are prone to distortion and damage.
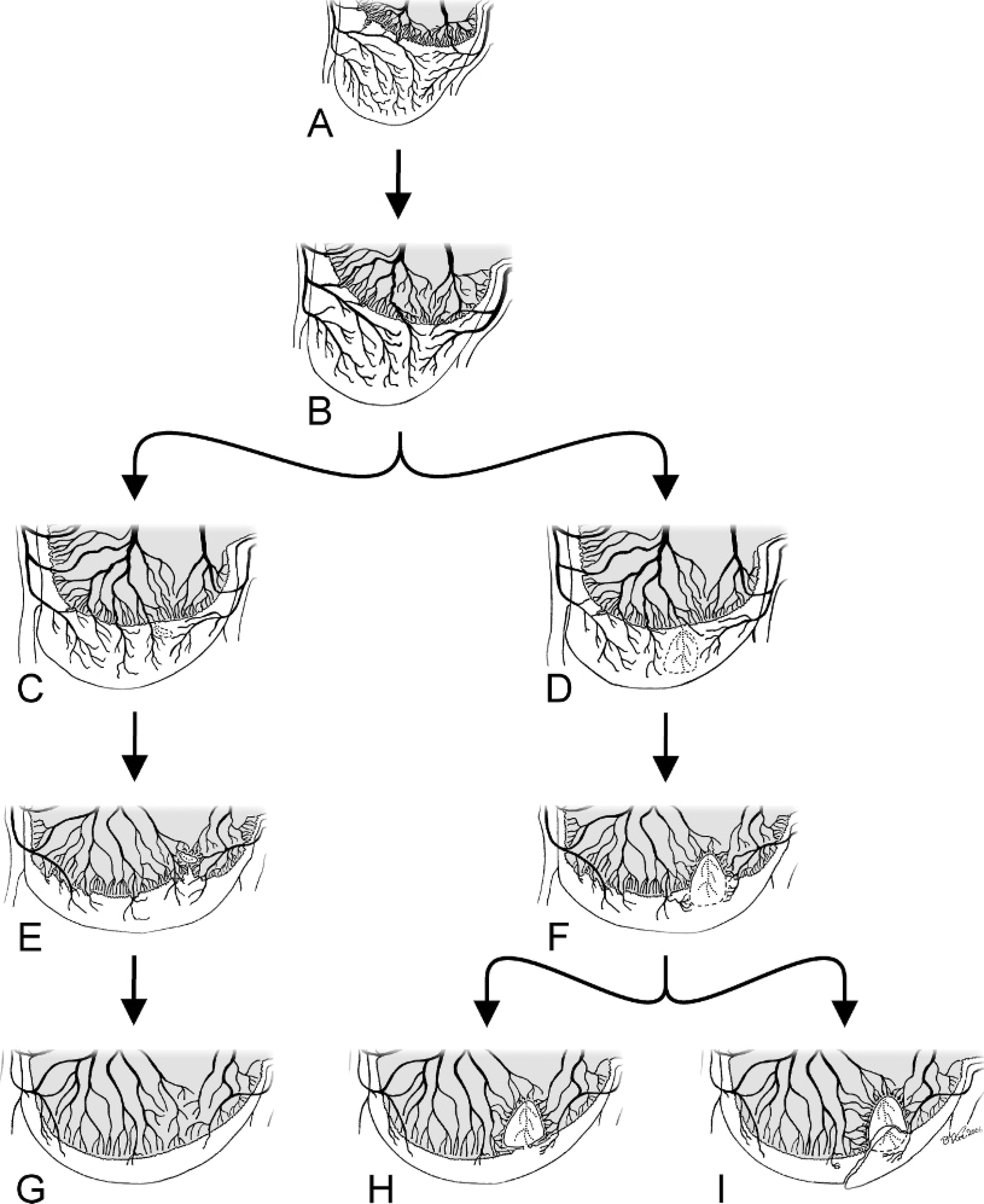
Pathogenesis of articular osteochondrosis. Schematic drawings of sections of an epiphysis at different stages of development. At an early age (
Therefore, we believe that the primary event in osteochondrosis is cessation of cartilage canal blood supply that occurs as a result of damage to temporal and newly formed vessels. In the medial femoral condyle of pigs, these vessels exist only in animals that are between approximately 8 and 13 weeks of age. 136 This theory, based on findings in pigs, explains the rather specific location of lesions of osteochondrosis and the existence of a defined time window of susceptibility for the disease, and it also provides an explanation as to why lesions often occur with bilateral symmetry.
Sequelae of Osteochondrosis May Overshadow the Disease Itself
After ischemic necrosis of growth cartilage occurs, two types of reaction may be predicted, and both have been demonstrated experimentally. 137 First, the cartilage matrix of the necrotic area degenerates, as it is no longer maintained by the chondrocytes. Second, a response is observed in the surrounding tissues (overlying articular cartilage, synovial membrane, adjacent growth cartilage and the tissues of the advancing ossification front) that is presumably initiated by the release of structural matrix components and matrix-bound enzymes and signaling molecules.
The overlying articular cartilage, being avascular, has limited ability to respond to injury; however, the growth cartilage along the margins of the necrotic area proliferates and exhibits increased matrix production, although no effective repair can occur prior to removal of the necrotic tissue. Chondrocytes of the growth cartilage also increase their production of the angiogenic factor VEGF
136,
137
and, presumably, a wide spectrum of proinflammatory markers.
24,
28,
83
Additionally, the cartilage canal vessels of the surrounding, viable cartilage proliferate toward the necrotic area,
136,
137
a process likely to be accompanied by proteolysis of cartilage matrix. In the subchondral bone subjacent to the necrotic cartilage, an inflammatory reaction occurs in which the bone marrow becomes populated with a mixture of active osteoblasts, active osteoclasts and chondroclasts, inflammatory cells, and spindle cells.
136
In cases of
A number of investigations have focused on biochemical and molecular characterization of the tissue composing lesions of osteochondrosis. 65, 67, 68, 102, 103, 117 The affected tissue is collected based on its gross appearance and is compared to site-matched normal-appearing tissue. The purpose of these studies is to provide a better understanding of the pathogenesis of the disease; however, these grossly visible lesions are, by definition, chronic, and the tissue collected, therefore, represents the late events occurring in the disease process. The results should be interpreted with care, as they are likely to produce evidence of an upregulation of a wide range of proinflammatory, catabolic, and anabolic factors, mirroring the ongoing process of degradation of necrotic cartilage and formation of reparative tissue. 3
Most previous studies have focused on the chronic stages of the disease; therefore, all factors affecting the diseased individual in the time span between the formation of the initial lesion and the chronic stages of the disease, often occurring over many months or even years, are considered to be involved in the initiation and progression of osteochondrosis, when many of these are more likely to represent repair processes. Moreover, molecular studies on tissue representing these chronic lesions are unlikely to yield information that is relevant to the initial development of the disease.
Conclusions
Based on all available evidence, we believe that the primary lesion of articular osteochondrosis should be defined as a
Given the long history and widespread use of the term
The majority of the evidence supports hereditary and anatomic factors as being the most important etiologic contributors to the pathogenesis of osteochondrosis (Fig. 8). The focal nature of the lesions suggests that future studies should focus on site-specific traits such as joint conformation and joint motion. It is likely that microtrauma affecting the vessels at the chondro-osseous junction may be the initiating event in the formation of lesions. An anatomic predisposition, such as faulty joint conformation, can increase the mechanical stress to this area in the limited period during which these vessels exist. It also is conceivable that the environmental factors may augment the mechanical stress to the structures at risk (Fig. 8).
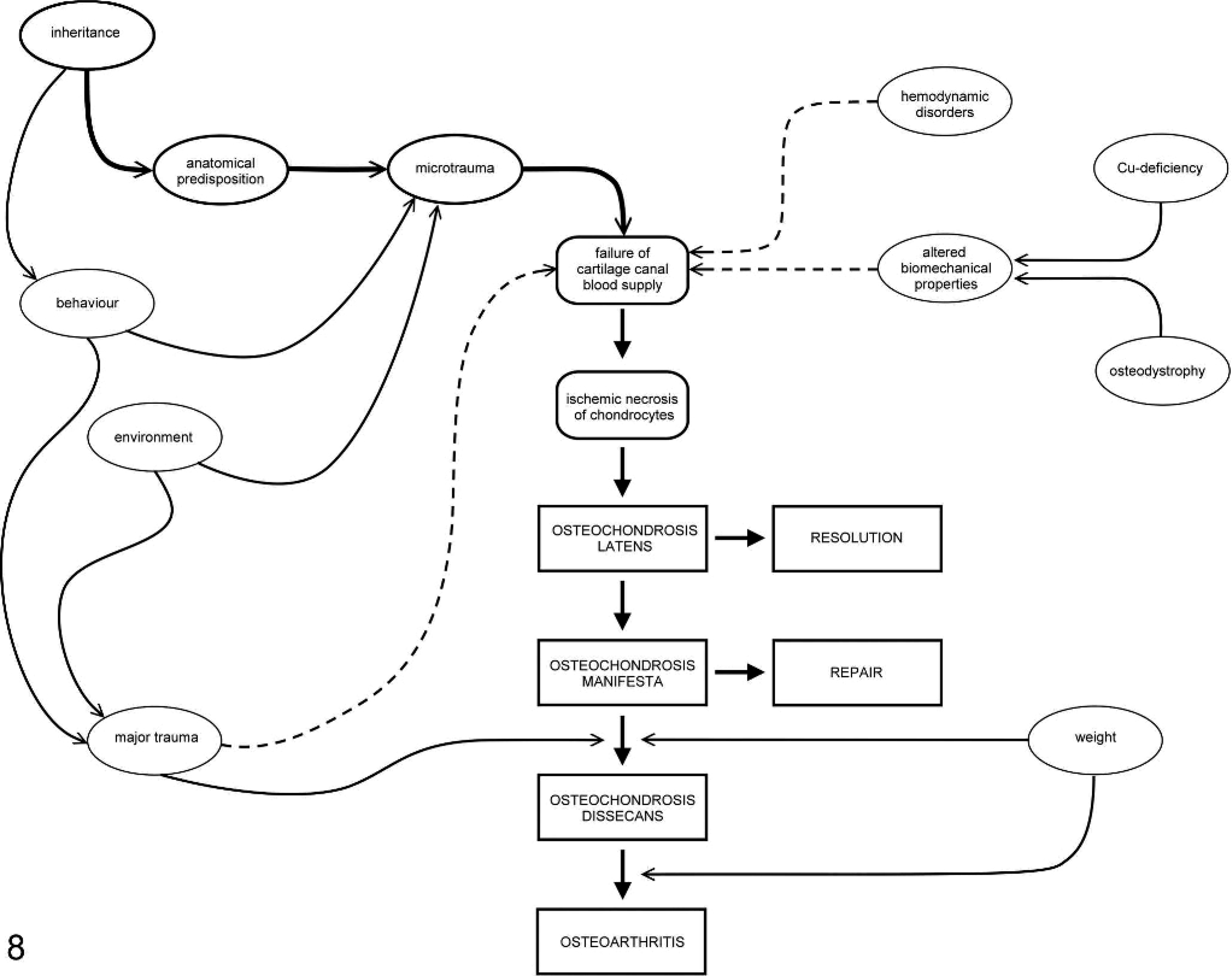
Causal diagram showing likely etiologic factors of osteochondrosis and how they may influence the development of the disease. The factors playing the most important role in primary osteochondrosis are emphasised with bold text boxes and solid arrow lines, whereas factors that may play a role in secondary osteochondrosis are depicted with dotted arrow lines.
Major trauma does not appear to play an important role in the majority of cases of osteochondrosis. Nevertheless, major trauma to or local infections in a growing epiphysis may alter blood supply in the affected location. These events may, consequently, cause rare cases of focal ischemic necrosis of growth cartilage (i.e., a secondary osteochondrosis) in any site, including locations other than the typical predilection sites (Fig. 8).
92
While trauma is not thought to be a major etiologic factor in osteochondrosis, it is quite likely that trauma plays an important role in the progression of lesions of
Generalized factors, such as hemodynamic disorders or an imbalance of dietary components, may be implicated in the pathogenesis of osteochondrosis only so long as they affect cartilage canal function, either through direct effects on the vessels or by resulting in altered biomechanical properties of the surrounding structures (Fig. 8). In these latter cases, it may be justified to regard the resulting lesions of osteochondrosis as being secondary to the generalized condition. 92
There is little evidence that the body weight of the animal affects the prevalence of early lesions of osteochondrosis. It is plausible, however, that increased body weight promotes the progression from osteochondrosis to osteoarthritis. Evidence for this statement includes the observations that obesity is an important factor in the progression of lesions of osteoarthritis in humans 23 and that the prevalence/severity of osteoarthritis is correlated with body weight in dogs (Fig. 8). 58, 111
A pathogenesis in which necrosis of cartilage canals is a crucial step accounts for many of the aspects of osteochondrosis that other theories do not. 19 For instance, because epiphyseal cartilage becomes avascular prior to adulthood, this theory explains why osteochondrosis only develops during skeletal growth. It also explains why the typical lesions occur in distinct predilection sites, as well as why they tend to occur in bilaterally symmetrical locations, since vascular supply to a site tends to be quite consistent within a species and has bilateral symmetry. An important clinical implication arising from this theory is that lesions of osteochondrosis are formed in a very limited window of time, namely the period when the epiphyseal cartilage is supplied by vulnerable blood vessels. Prophylactic measures, therefore, should be focused on this period of time, which varies somewhat depending on the species and site.
The choice of scientific approach used to study osteochondrosis relies on an understanding of the primary pathologic processes, the different stages of the disease (
Footnotes
Acknowledgements
We are very grateful to Professor Jon Teige at the Division for Anatomy and Pathology at the Norwegian School of Veterinary Science and Professor Finn P. Reinholt at the Institute of Pathology at University of Oslo and the Department of Pathology at Rikshospitalet University Hospital for valuable and inspiring discussions.
