Abstract
Influenza A virus infections may spread rapidly in human populations and cause variable mortality. Two of these influenza viruses have been designated as select agents: 1918 H1N1 virus and highly pathogenic avian influenza (HPAI) virus. Knowledge of the pathology of these virus infections in humans, other naturally infected species, and experimental animals is important to understand the pathogenesis of influenza, to design appropriate models for evaluation of medical countermeasures, and to make correct diagnoses. The most important complication of influenza in humans is viral pneumonia, which often occurs with or is followed by bacterial pneumonia. Viremia and extrarespiratory disease are uncommon. HPAI viruses, including HPAI H5N1 virus, cause severe systemic disease in galliform species as well as in anseriform species and bird species of other orders. HPAI H5N1 virus infection also causes severe disease in humans and several species of carnivores. Experimental animals are used to model different aspects of influenza in humans, including uncomplicated influenza, pneumonia, and virus transmission. The most commonly used experimental animal species are laboratory mouse, domestic ferret, and cynomolgus macaque. Experimental influenza virus infections are performed in various other species, including domestic pig, guinea pig, and domestic cat. Each of these species has advantages and disadvantages that need to be assessed before choosing the most appropriate model to reach a particular goal. Such animal models may be applied for the development of more effective antiviral drugs and vaccines to protect humans from the threat of these virus infections.
Influenza was first described as epidemics of acute, rapidly spreading catarrhal fevers in humans, and the first epidemic was probably described by Hippocrates in 412
Since 1900, there have been 4 pandemics of human influenza: 1918 H1N1 Spanish influenza, 1957 H2N2 Asian influenza, 1968 H3N2 Hong Kong influenza, and the 2009 H1N1 Mexican influenza. 31 Of these, the 1918 Spanish influenza was the worst, causing acute illness in 25 to 30% of the world’s population and the death of nearly 50 million persons. 57,141,145 Most fatalities occurred in the 15- to 34-year-old group. Most patients succumbed to bacterial pneumonia, but many had primary acute interstitial pneumonia, pulmonary hemorrhage, and pulmonary edema. 141,145 So severe was the impact on the young in the United States that life expectancy declined by more than 10 years during the pandemic. 145 The overall mortality was 2.5% among the infected, whereas in some remote populations, mortality rates as high as 70% were reported. In other influenza epidemics, mortality rates were typically under 0.1%. The pandemics of 1957, 1968, and 2009 were less severe than that of 1918 on the basis of lower mortality rates and a lesser severity of disease. Historically, pandemic influenza viruses arise through reassortment and specific adaptations. New viruses have novel surface proteins—principally, the hemagglutinin—that allow the virus to spread through the immunologically naive human population. 145
High pathogenicity avian influenza (HPAI) has caused 28 severe disease outbreaks in poultry since 1959, with mortality rates approaching 100%. Unlike human pandemic influenza, which has lesions mostly in the respiratory tract, HPAI viruses in poultry cause severe systemic disease with necrosis and inflammation in many visceral organs, the vascular system, skin, and brain. 102,103,138,140 The H5N1 HPAI virus lineage, which emerged in China in 1996, has become the largest HPAI outbreak in poultry during the past 50 years, and it has been the first to produce significant numbers of human infections and deaths—specifically, 471 infections with 282 fatalities since January 2004. 138,162 The production of H5N1 HPAI virus infections in humans, with an accompanying case fatality rate of 60%, raised concerns for HPAI viruses becoming the next human pandemic virus, whether through mutation, reassortment, or both. 160
Etiology
The etiology of influenza was established for fowl plague (ie, HPAI) as a filterable agent, or virus, by Centanni and Savonuzzi in 1901; for swine influenza by Shope in 1928; and for human influenza by Smith in 1933. 13,132,134 Influenza is caused by negative-sense single-stranded segmented RNA viruses in the Orthomyxoviridae family, which has 5 genera: Influenzavirus A, Influenzavirus B, Influenzavirus C, Isavirus, and Thogotovirus. 31,64 Influenza A virus infections occur in mammals and birds, whereas influenza B and C viruses are reported in mammals only. Influenza A viruses are further classified on the basis of surface glycoproteins into 16 hemagglutinin subtypes (HA subtypes; H1–H16) and 9 neuraminidase subtypes (NA subtypes; N1–N9). 140 All HA and NA subtypes have been reported in avian species, but the subtypes reported in mammals are more restricted and include endemic influenzas of H1N1 (humans and pigs), H3N2 (humans and pigs), and H3N8 (horses and dogs) and rare occurrence of other avian influenza (AI) subtypes that spill over from birds to mammals. Most AI viruses cause asymptomatic infection or mild disease in poultry (ie, low-pathogenicity avian influenza), and can be any combination of the 16 HA and 9 NA subtypes. However, some of the H5 and H7 AI viruses can be highly pathogenic. HPAI viruses produce 75% or greater mortality in intravenously inoculated chickens (Gallus gallus domesticus); they have a chicken intravenous pathogenicity index of 1.2 or greater; or they are H5 or H7 AI viruses, having an hemagglutinin proteolytic cleavage site with a polybasic amino acid sequence similar to that of other HPAI viruses. 101
Select Agent and Non–Select Agent High-Risk Influenza A Viruses
Only a few strains or lineages of influenza A virus are classified as select agents: first, all HPAI viruses, as regulated by US Department of Agriculture; second, reverse genetic constructs containing all 8 gene segments of the 1918 H1N1 Spanish influenza virus, as regulated by US Department of Health and Human Services. 142
No naturally occurring viable 1918 influenza virus exists. 142 However, with development of reverse genetic systems, influenza A viruses have been constructed from sequenced genetic code using plasmids that transcribe negative-sense RNA of individual gene segments in cell culture systems. 34,99, 141 Jeffrey Taubenberger and colleagues at the Armed Forces Institute of Pathology originally extracted and sequenced the viral genome from paraffin-embedded and frozen lung tissues of 1918 civilian and military victims. 9,115 –118,146 The reconstruction of the 1918 virus and the initial study of its pathology were accomplished over a 6-year period through collaborations between the Armed Forces Institute of Pathology, the Mount Sinai School of Medicine, the US Department of Agriculture, the Centers for Disease Control and Prevention, and the University of Washington. This culminated in 2005 with reconstruction of all 8 gene segments of 1918 into a complete viable virus. 142,149
Although not classified as select agents, the H2N2 Asian influenza virus of the 1957 pandemic and the H1N1 virus of the 2009 pandemic (pH1N1 virus) are of concern because they are considered to have higher risk to public health than that of seasonal influenza A viruses. The 1957 H2N2 pandemic caused 4 million excess deaths worldwide when compared to seasonal influenza virus. 57,141,143,145 Individuals born since 1968 have no protective immunity against the H2N2 virus because it stopped circulating in the human population during 1968, when the H3N2 pandemic virus appeared. 143 However, the currently circulating seasonal human H3N2 influenza virus contains the same N2 lineage as the H2N2 virus and may confer some partial protection. 115 The presence of the H2N2 virus in many laboratory freezers and the temporary inclusion of live H2N2 virus in some proficiency tests to laboratory and health care facilities in 19 countries during 2005 (http://www.cdc.gov/flu/h2n2backgroundqa.htm) created a risk for accidental or intentional release that could initiate another pandemic. 97,130
Beginning in April 2009, a novel H1N1 influenza A virus was identified as the cause of acute respiratory disease of humans in Mexico. 38 This virus was a complex reassortant influenza A virus, which had not been reported in animals but had gene segments related to North American classic H1N1 swine viruses (hemagglutinin, nucleoprotein, and nonstructural gene segments), North American avian viruses (polymerase A and B2 genes), human influenza A virus (polymerase B1), and Eurasian H1N1 swine viruses (neuraminidase and matrix). 38 As of February 14, 2010, more than 212 countries have reported laboratory-confirmed cases of this pandemic influenza H1N1 2009, including at least 15,921 deaths (http://www.who.int/csr/don/2010_02_19/en/index.html). In humans, pH1N1 virus infection usually caused uncomplicated influenza. 94 In contrast to seasonal influenza, severe disease from pH1N1 virus infection seemed concentrated in older children and young adults, with almost no cases reported in patients older than 60 years. Most complications occurred among previously healthy individuals, with obesity and respiratory disease as the strongest risk factors. Fatal outcome was seen in 0.5% of cases, usually associated with severe pneumonia and respiratory failure. Neuromuscular and cardiac complications were unusual. 123 In fatal cases, patients had diffuse alveolar damage, often with necrotizing bronchiolitis and alveolar hemorrhage. 89
Influenza in Humans
This section is situated in the context of the pathology of animal models for influenza in humans. As such, it reviews the pathology of influenza in humans to show (1) gaps in our knowledge of the disease in humans so that we know which areas to investigate in animal models, (2) whether the disease in animal models accurately mirrors the disease in humans or at least the aspect of disease under investigation, and (3) which countermeasures against the disease in humans need to be developed and how to read out their efficacy in animal models.
The pathology of influenza in humans has mainly been studied following influenza pandemics because they focus attention on influenza and because series of fatal influenza cases are available. The bird-to-human transmission of HPAI H5N1 virus also generated interest in the subject because of concern that it would cause a new influenza pandemic.
Human Influenza Viruses
The pathology of select agent H1N1 1918 virus infection is similar, at least superficially, to that of non–select agents H2N2, H3N2, and pH1N1 virus infections, even though the case fatality rate in the 1918 pandemic was considerably higher than it was in the other pandemics. Therefore, we describe the pathology of these human influenza virus infections together. For more detailed reviews, see Kuiken and Taubenberger 73 and Taubenberger and Morens. 144
Transmission of human influenza virus occurs by inhalation of infectious droplets or airborne droplet nuclei or by indirect contact followed by self-inoculation of the upper respiratory tract or conjunctival mucosa. The relative importance of these routes is still debated. 47
Attachment of the viral hemagglutinin to its host cell receptor, a sialic acid, is the first step in influenza virus replication following transmission. Because different influenza viruses use different sialic acids as their receptor and because the expression of sialic acids differs across the respiratory tract and across species, this step influences pattern of disease in the respiratory tract and host range of virus infection. The pattern of viral attachment can be measured by virus histochemistry, which is a direct measure of attachment of an influenza virus to its host cell. 155 Human influenza viruses in the human respiratory tract predominantly attach to ciliated epithelial cells, which are present throughout the respiratory tract. Attachment is most abundant to the ciliated epithelium of upper respiratory tract, trachea, and bronchi (Fig. 1). 153,154
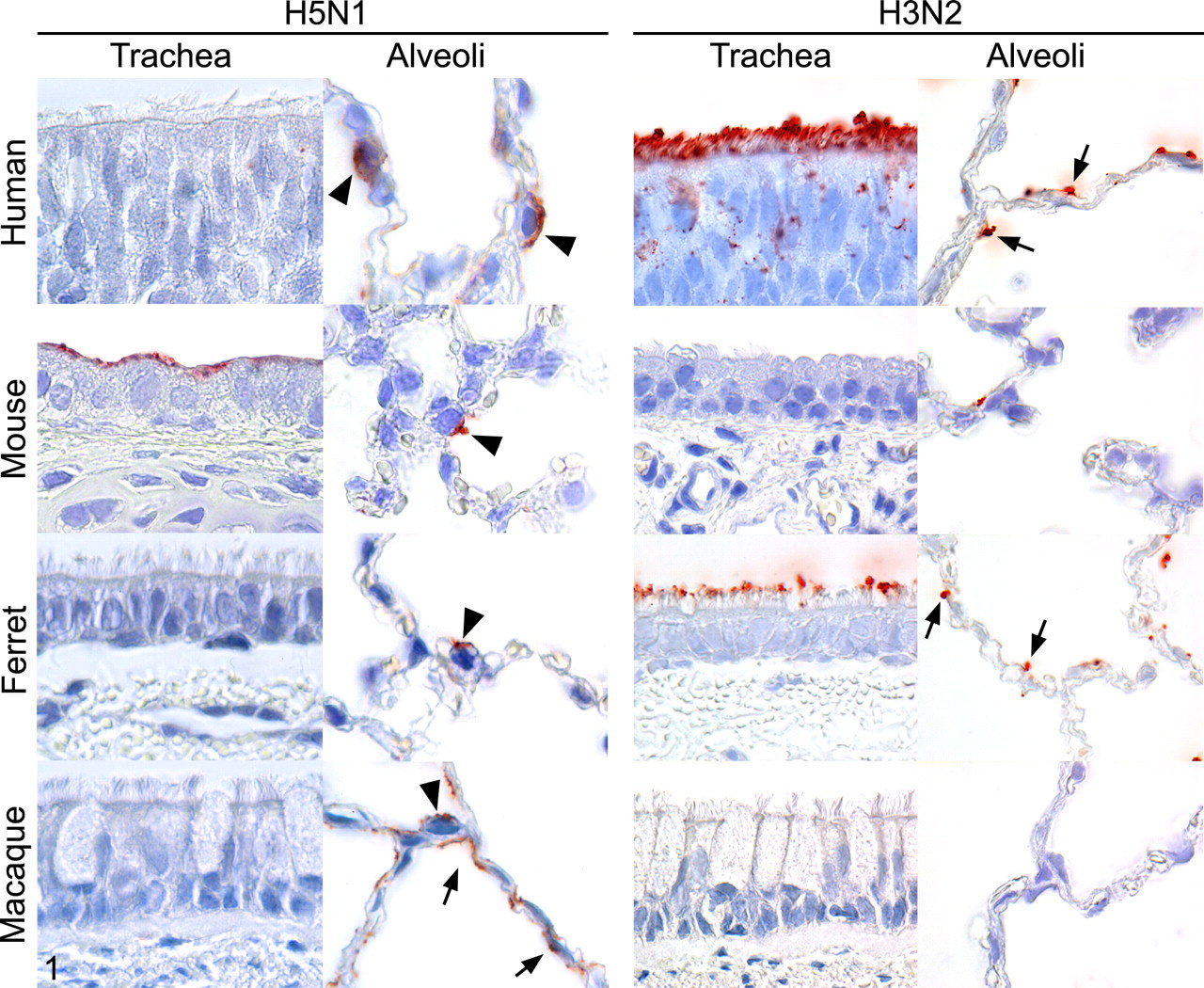
Pattern of attachment of highly pathogenic avian influenza H5N1 virus and seasonal human influenza H3N2 virus to trachea and alveoli of humans and commonly used experimental animal species. Influenza virus is visible as granular to diffuse red staining on the apical surface of epithelial cells. In the alveoli, influenza virus attaches to type I pneumocytes (arrows), type II pneumocytes (arrowheads), or both. Reprinted from Reference 153 with permission from the American Journal of Pathology and from Reference 155 with permission from Science.
The pathology of uncomplicated influenza corresponds to this pattern of attachment. Uncomplicated influenza is a mild inflammation of the upper respiratory tract—specifically, rhinitis, paranasal sinusitis, pharyngitis, and laryngitis 153 —and diffuse superficial necrotizing tracheobronchitis. There is desquamation of epithelial cells, edema and hyperemia in the lamina propria, and infiltration with lymphocytes and histiocytes. The inflammation is short-lasting: Epithelial regeneration is visible by 2 days after onset of symptoms. 156
The most common complication of influenza is extension of the virus infection to the lung, resulting in pneumonia. Rather confusingly, the type of pneumonia caused by influenza virus infection is classified as diffuse alveolar damage in human pathology 30 and as bronchointerstitial pneumonia in veterinary pathology. 26 Based on attachment studies, the primary targets of human influenza virus in the alveoli are type I pneumocytes and, less frequently, type II pneumocytes and alveolar macrophages (Fig. 1). 154 This corresponds with the few in vivo descriptions of the target cells of human influenza virus in fatal pneumonia: Virus antigen expression is seen in alveolar epithelial cells, alveolar macrophages, and interstitial macrophages. 41,50,87,93
Damage to the alveolar epithelium is due to a combination of the direct cytolytic effect of virus infection and the indirect effect of host response. Type I pneumocytes prevent leakage of fluid across the alveolar–capillary border, and type II pneumocytes resorb fluid from the alveolar lumen. Therefore, damage to type I and type II pneumocytes allows fluid to flood into and accumulate in the alveolar lumina, which has severe consequences for the gas exchange function of the respiratory tract, resulting in severe—and, in some cases, fatal—respiratory dysfunction. 157
The pathologic result of influenza virus infection of the alveoli is diffuse alveolar damage (Fig. 2). 144 In the early stage, it is characterized by necrosis of alveolar epithelium and flooding of the alveolar lumina by edema fluid, mixed with variable proportions of fibrin, erythrocytes, alveolar macrophages, and neutrophils. Some alveoli are lined by hyaline membranes. The alveolar septa are widened because of hyperemia, edema, and infiltration by mainly neutrophils. Alveolar capillaries and small pulmonary blood vessels may contain fibrin thrombi. At the later stage of diffuse alveolar damage, there is type II pneumocyte hyperplasia, interstitial fibrosis of alveolar septa, and infiltration by predominantly lymphocytes and plasma cells (Fig. 3).
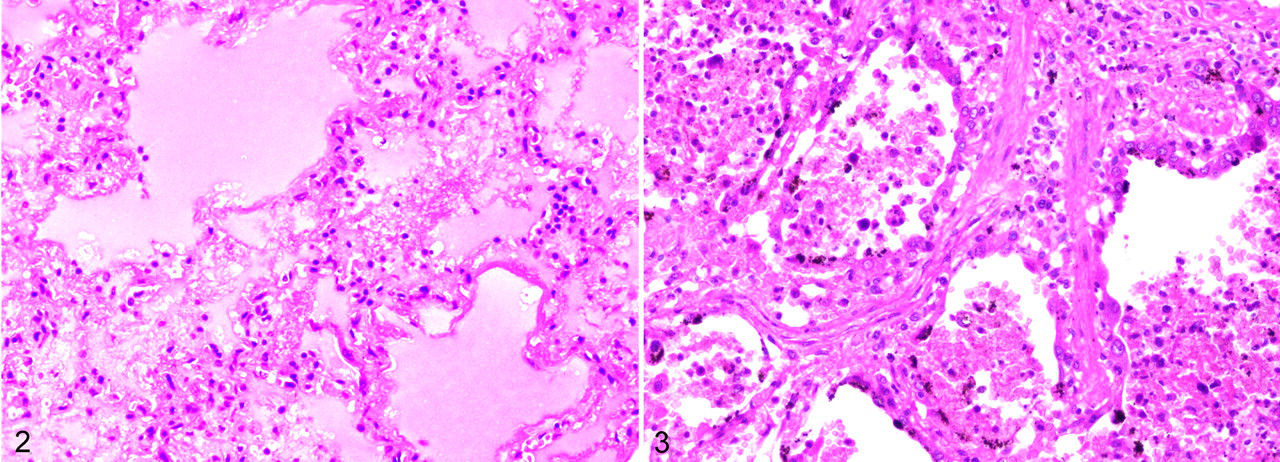
Influenza viral pneumonia often occurs with, or is followed by, bacterial pneumonia. Commonly involved bacteria are Staphylococcus aureus and Haemophilus influenzae. Pathologically, this leads to more extensive necrosis and more prominent infiltration of neutrophils: suppurative bronchopneumonia. In the 1918 H1N1 pandemic, 96% of deaths likely resulted from secondary bacterial pneumonia. 92 In later pandemics, this percentage declined in favor of deaths from primary influenza viral pneumonia, possibly because of the widespread administration of antibiotics. 49
Although human influenza virus primarily infects and causes disease in the respiratory tract, it is associated with disease in extrarespiratory organs, albeit to a lesser extent, including influenza-associated acute encephalopathy, 135 myocarditis, 114 and myopathy. 1 In general, there two explanations for the pathogenesis of these extrarespiratory complications. 73 The first is that influenza virus spreads via blood to these tissues and replicates there. However, viremia from human influenza virus infection appears to be rare after onset of symptoms and, if it occurs, is not sustained for long periods. 78 Proof of replication of human influenza virus in extrarespiratory tissues comes from direct immunofluorescence, immunohistochemistry, or in situ hybridization in the tissue concerned. However, such reports are rare, and further confirmation of the ability of human influenza virus to replicate in extrarespiratory human tissues in vivo is badly needed.
The second explanation for the pathogenesis of extrarespiratory complications is suggested by the link between acute respiratory distress syndrome and multiple organ dysfunction syndrome. 10 Acute respiratory distress syndrome, which has multiple causes (including influenza virus infection), commonly progresses to multiple organ dysfunction syndrome, most commonly involving the hepatic, renal, central nervous, gastrointestinal, hematologic, and cardiac systems. The pathogenesis of multiple organ dysfunction syndrome has not been elucidated, but it may involve the microcirculation and mitochondrial metabolism, as well as the release of cytokines into the circulation.
Highly Pathogenic Avian Influenza Viruses
Direct human infection with avian influenza viruses was considered to be rare and of little consequence to human health. The most common clinical presentation of these rare cases was a conjunctivitis that resolved within 2 weeks. 73 This changed in 1997, when HPAI H5N1 virus infection was diagnosed in people in Hong Kong, resulting in 6 deaths out of 18 confirmed infections. HPAI H5N1 virus continues to circulate among poultry in many countries in Asia, Africa, and Europe, occasionally spreading to humans, often with fatal consequences. Besides the severe disease consequences for the infected individual, HPAI H5N1 virus has the potential to cause widespread disease in the human population if it adapts to efficient human-to-human transmission. Our understanding of the pathology of HPAI H5N1 in humans is hampered by the few autopsies performed on people who have died of the infection. A recent review 69 identified only 9 full autopsies out of 216 laboratory-confirmed fatal cases. The presumed routes of infection in humans include exposure to infected poultry, swimming in contaminated water, and preparing or eating contaminated poultry products. 11
HPAI H5N1 virus in the human respiratory tract predominantly attaches to nonciliated epithelial cells. Attachment is rare in the upper respiratory tract 153 and trachea, and it increases progressively toward the bronchioles (Fig. 1). 155 Within the terminal bronchioles, HPAI H5N1 virus preferentially attaches to nonciliated cuboidal epithelial cells and, within the alveoli, to type II pneumocytes and alveolar macrophages. This pattern of attachment corresponds to the clinical evaluation of infected people, where primary disease centers on the lungs, 11 and to the rare autopsy reports, where lesions center on the alveoli and bronchioles, without reported lesions in trachea and bronchi. 69 This pattern of attachment could partly explain the severe pneumonia caused by H5N1 HPAI virus infection. In the alveoli, type II pneumocytes are the predominant epithelial cells and are metabolically active. Furthermore, type II pneumocytes are responsible for ion transport, fluid resorption, surfactant production, and re-epithelization after alveolar damage. 25 Alveolar macrophages protect the lung from influenza virus–induced damage by phagocytosis of virus particles and by control of the immune response. Therefore, targeting of these cells by H5N1 HPAI virus may hamper their protective function and contribute to a productive infection. 44,51
The pulmonary lesions of HPAI H5N1 virus infection are consistent with diffuse alveolar damage, both acute and chronic. They are characterized by type II pneumocyte hyperplasia, interstitial infiltration by lymphocytes (and, in some cases, neutrophils), and the predominance of alveolar macrophages (some with hemophagocytosis) in alveolar lumina. Other features include desquamation of epithelium into alveolar lumina, hemorrhage, and bronchiolitis. Virus antigen and viral RNA expression are seen in type II pneumocytes and in ciliated and nonciliated tracheal epithelial cells by immunohistochemistry and in situ hybridization, respectively. 69
HPAI H5N1 virus infection has also been associated with extrarespiratory disease. There is evidence for viral presence by immunohistochemistry, in situ hybridization, or both (often in association with lesions) in brain, intestine, liver, lymph nodes, placenta, and fetal lung. 69 Virus spread from the lungs to extrarespiratory tissues most likely occurs by viremia. The isolation of HPAI H5N1 virus from the blood of 2 patients and the detection of viral RNA by reverse transcriptase polymerase chain reaction in the blood of 9 of 16 patients suggest that viremia can occur at reasonably high levels and for prolonged periods in people with symptoms of HPAI H5N1 virus infection. 78
Another HPAI H5N1 virus that has been reported to cause fatal disease in humans is HPAI H7N7 virus. In 2003, infection with this virus caused an outbreak in poultry in the Netherlands, and the virus was detected in 86 people who had handled affected poultry, as well as 3 of their family members. Out of 89 people in whom HPAI H7N7 virus infection was confirmed, the majority (n, 78; 88%) presented with conjunctivitis alone, whereas a smaller proportion had conjunctivitis and influenza-like illness (n, 5; 6%) or influenza-like illness alone (n, 2; 2%). However, one patient developed severe pneumonia and died from acute respiratory distress syndrome and related complications. The virus was isolated from postmortem lung samples. Histologically, there was severe diffuse alveolar damage, with flooding of the alveolar lumina by serosanguineous fluid mixed with fibrin and neutrophils. There was no evidence for extrarespiratory spread of virus. 35,68
HPAI H 5 N 1 in Carnivores
In general, terrestrial carnivores are considered relatively resistant to disease from influenza virus infection. If disease does occur, it is mostly limited to the respiratory tract. 6 HPAI viruses of the H5N1 subtype are remarkable because of their ability to cause severe or fatal disease in several species of terrestrial carnivores and to spread beyond the respiratory tract. Information about the pathology of HPAI H5N1 virus infection in these species is not only useful to understand the pathogenesis and lesions in the affected species but to appreciate the potential portals of entry, mechanisms of spread, and range of affected tissues in humans. See Reperant et al for a detailed review. 119
Natural cross-species transmission resulting in fatal disease has been reported in tigers (Panthera tigris), leopards (Panthera pardus), domestic cats, a domestic dog, Owston’s palm civets (Chrotogale owstoni), a stone marten (Mustela foina), and an American mink (Mustela vison). 119 Ingestion of infected wild or domestic bird carcasses appeared to be the primary route of exposure. Experimental feeding of infected bird carcasses to red foxes (Vulpes vulpes) 120 and ferrets 79 also resulted in disease.
There are 2 potential portals of entry following ingestion of infected material: first, the respiratory tract, owing to inhalation of virus particles during ingestion; second, the digestive tract, owing to ingestion of infected material. These 2 routes are based on the difference in pathogenesis between intratracheally inoculated cats and those that fed on infected bird carcasses. 121 Proof that both the respiratory tract and the digestive tract can act as a portal of entry for HPAI H5N1 virus was first provided by experimental infections of ferrets and mice, some of which were inoculated directly into the stomach. 79
Virus spreading from these 2 initial sites of replication can take place via blood, lymph, or nerves. 119 Experimental infection of mice suggests that HPAI H5N1 virus may infect the brain from the nasal cavity, through neuronal transmission along the olfactory nerves. 106 The pattern of virus infection in kidney, liver, and adrenal gland of experimentally infected cats strongly suggests virus spreading via blood. 121 Despite a presumed viremic phase of HPAI H5N1 virus infection, viral replication in the endothelial cells of blood vessels, a characteristic finding in poultry, has rarely been documented in mammals. Occasional endothelial cells in the heart and pulmonary veins of experimentally infected cats expressed virus antigen. 121
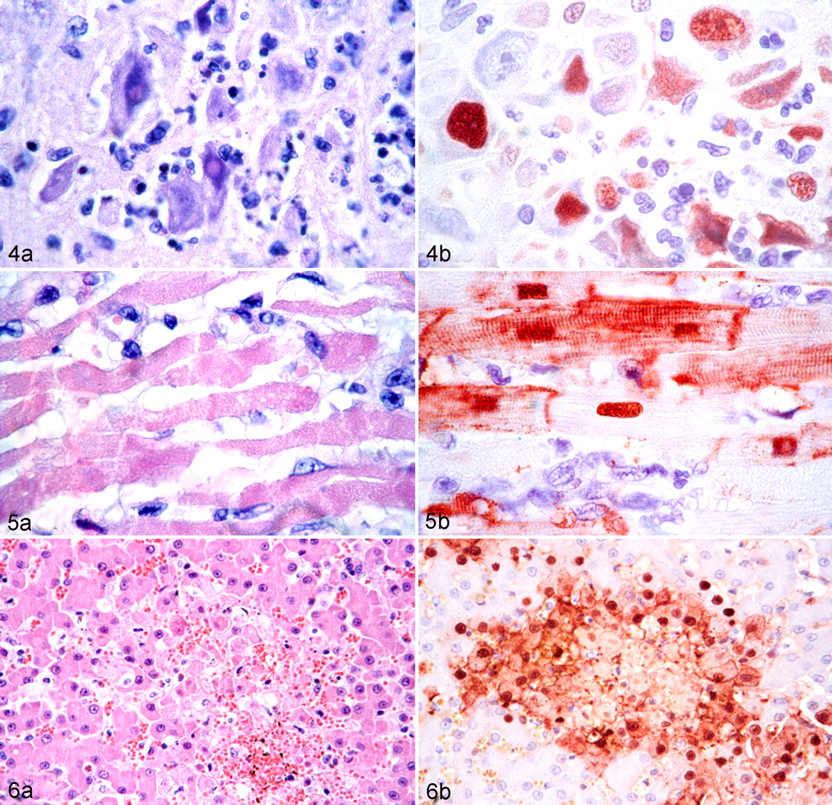
In the above-listed experimentally and naturally infected carnivore species, virus infection and associated disease were not limited to the respiratory tract but detected in multiple extrarespiratory organs: brain, liver, heart, kidney, spleen, intestine, pancreas, adrenal gland, or a combination of these tissues, with expression of virus antigen (by immunohistochemistry) or viral RNA (by in situ hybridization) mostly in the parenchymal cells of those organs (Figs. 4–6). Virus replication in most of these organs was associated with severe necrosis and inflammation, assumed to result from the cytolytic effect of viral replication and the indirect effects of the induced host immune response. 119
Extrarespiratory lesions and associated viral antigen expression in domestic cats infected with highly pathogenic avian influenza H5N1 virus infection and euthanized at 6 or 7 days after infection.
HPAI H 5 N 1 Virus Infection and Other HPAI Virus Infections in Birds
Infection with HPAI viruses, including HPAI H5N1 virus, cause severe systemic disease and mortality in chickens and other species of the order Galliformes. An unusual aspect of the ongoing outbreak of HPAI virus infection is that it has caused substantial mortality in several species of wild birds, some of which were previously considered resistant to disease from HPAI virus infection. This review provides summary information of the pathogenesis and lesions in birds of HPAI virus infection in general and HPAI H5N1 virus infection in particular to (1) understand the pathogenesis and pathology in the affected avian species, (2) compare the pathogenesis and pathology between birds and mammals (including humans), and (3) help determine the risk for direct bird-to-human transmission of HPAI H5N1 viruses. This summary is based on experimental infections. Detailed reviews are provided elsewhere. 104,105,139
Galliformes
HPAI virus infection has been most extensively studied in chickens and turkeys (Meleagris gallopavo), which are the most commonly affected species and which suffer the most severe disease. HPAI virus infections in other captive galliform species—chukar (Alectoris chukar), helmeted guinea fowl (Numida meleagris), Japanese quail (Coturnix japonica), and common pheasant (Phasianus colchicus) 108 —have a similar overall mortality as in chickens, although survival often is longer.
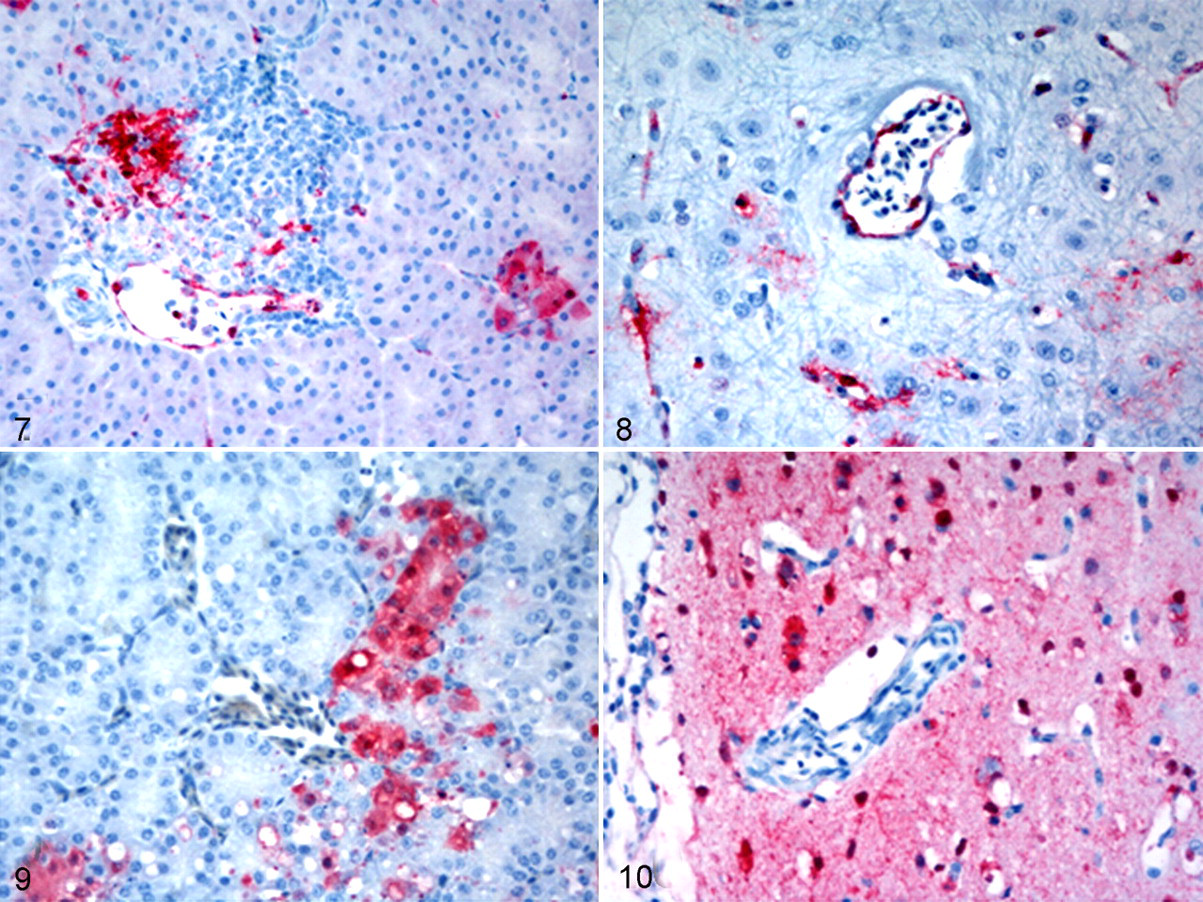
The basic pathogenesis of HPAI virus infection in chickens and other galliforms is as follows. After respiratory tract infection, the virus initially replicates in epithelial cells of the respiratory tract. From there, virus spreads through the vascular and lymphatic systems, where it replicates in macrophages and heterophils. This viremia allows the virus to disseminate to multiple organs, where it replicates in parenchymal cells. This basic scheme has 2 variations, depending on the HPAI virus involved. In the first variant, which occurs in HPAI H5N1 virus infection, viremia is associated with virus replication in endothelial cells (Figs. 7, 8). The damage to the blood vessels causes increased permeability resulting in edema, hemorrhage, microthrombosis, consumptive coagulopathy, multiple organ failure, and peracute death. In the second variant, viremia is not associated (or is rarely associated) with virus replication in endothelial cells. The birds survive longer, which allows more extensive virus replication in multiple critical organs, resulting in severe necrosis and inflammation. In this form, birds die at a later stage from single- or multiorgan failure, with involvement of the brain, autonomic nervous system, heart, endocrine glands (eg, adrenal gland), pancreas, or a combination of these organs. 139
Gross lesions are not consistently present in all infected birds. 139,140 In peracute disease, gross lesions may be absent. In acute disease, birds may have ruffled feathers and edema of the comb, wattles, periorbital and intermandibular areas, upper neck, leg shanks, and feet, with accompanying subcutaneous hemorrhages, especially of the nonfeathered skin. Some virus strains produce edema and hyperemia of the conjunctiva, eyelids, and trachea. The wattles, combs, and snoods may contain necrotic foci and hemorrhages, or they may be cyanotic. Internally, hemorrhages may be present on serosal and mucosal surfaces and accompanied by necrotic foci within multiple visceral organs. 105 With many HPAI viruses, foci of necrosis may be present in the pancreas, heart, and, occasionally, the liver and kidneys. Urate deposits may accompany renal necrosis. Occasionally, the lungs may be firm from edema and pneumonia and have congestion and hemorrhages. In young birds, the cloacal bursa and thymus may be atrophic, with or without hemorrhage.
Microscopic lesions are more frequent than gross lesions in most HPAI virus infections of chickens. Microscopic changes consist of necrosis, inflammation, or both in multiple organs. Such lesions are most common and most severe within the skin (including feather follicles), brain, heart, pancreas, lungs, adrenal glands, and primary and secondary lymphoid organs. 105 Expression of avian influenza viral antigen is associated with these areas of necrosis and inflammation. Lesions are similar in other galliform species, but because these species survive longer than chickens, inflammation in parenchymal organs is more common and prominent than necrosis. 140,140
Anseriformes
In domestic ducks (Anas platyrhynchos domesticus), most HPAI virus infections cause no or only limited virus replication and few clinical signs. 2,3,109 The early H5N1 HPAI viruses (1997–2002) replicated in only the respiratory tract of domestic ducks, resulting in mild respiratory lesions and no mortality. 109 However, some HPAI H5N1 viruses isolated after 2002 have caused clinical disease and high mortality in domestic ducks. This increased virulence is due to expanded tissue tropism with high virus titers in affected tissues. 14,52,76,82,104,126,136,137 An important difference in pathogenesis compared to that in chickens is the lack of virus replication in endothelial cells and the absence of associated vascular damage. Gross lesions in severely affected domestic ducks include dehydration, empty intestines, splenomegaly, thymus atrophy, dilated and flaccid heart with increased pericardial fluid, airsacculitis, and congested malacic brain. 82,104,112 Microscopic lesions and virus antigen expression are similar to those in chickens, except that there is no replication or associated lesions in blood vessel endothelium (Figs. 9, 10).
In wild duck species, the morbidity and mortality of experimental infection with HPAI H5N1 virus from Europe or Asia after 2002 highly depend on host species. Intranasal inoculation into mallards, northern pintails (Anas acuta), blue-winged teals (Anas discors), redheads (Aythya americana), and wood ducks (Aix sponsa) caused mortality only in wood ducks. 19 Intratracheal and intraesophageal inoculation into mallards, gadwalls (Anas strepera), Eurasian wigeons (Anas penelope), common teals (Anas crecca), tufted ducks (Aythya fuligula), and Eurasian pochards (Aythya ferina) caused mortality only in tufted ducks and Eurasian pochards. 63 The main gross lesions in species showing mortality were multifocal pancreatic necrosis, diffuse airsacculitis, and pulmonary consolidation. Microscopic lesions included severe diffuse neuronal necrosis in cerebrum and cerebellum, necrotizing pancreatitis, necrotizing adrenalitis, and multifocal myocardial necrosis. Virus antigen expression occurred mainly in the parenchymal cells of affected tissues; as in domestic ducks, it was absent in endothelial cells. 19,63
Wild swans, both whooper (Cygnus cygnus) and mute (Cygnus olor), were often found dead from HPAI H5N1 virus infection in Europe in 2006. Because of their high susceptibility and striking size and color, they acted as an indicator for the presence of the virus in the environment. 147,148 Experimental HPAI H5N1 virus infection in whooper, black (Cygnus atratus), trumpeter (Cygnus buccinator), and mute swans caused 100% mortality in all 4 species. 14 The main gross lesions in mute and whooper swans were widespread hemorrhages. Microscopic lesions were necrosis and inflammation in mainly brain, pancreas, and liver. Virus antigen expression occurred in the parenchymal cells of affected tissues and—uniquely among anseriformes species—endothelial cells. 59
All tested goose species suffered mortality from HPAI H5N1 virus infection. Domestic geese (Anser anser domesticus) showed neurologic signs and microscopic lesions consisting of nonsuppurative encephalitis with neuronal necrosis and necrotizing or lymphoplasmacytic pancreatitis. 109 Canada geese (Branta canadensis) most consistently had lesions in brain, spinal cord, parasympathetic ganglia of the gastrointestinal tract, heart, and pancreas. 98,107 Bar-headed geese (Anser indicus) and cackling geese (Branta hutchinsii) commonly had necrosis and inflammation in brain, pancreas, liver, and adrenal glands associated with virus antigen expression in the parenchymal cells of affected tissues. 21
Other Orders
Experimental HPAI H5N1 virus infections have been performed in bird species of other orders: Charadriiformes: laughing gulls (Larus atricilla)
19
and herring gulls (Larus argentatus)
22
Columbiformes: domestic pigeons (Columba livia domestica)
17,20,55,81
Struthioformes: emus (Dromaius novaehollandiae)
109
Passeriformes: American crows (Corvus brachyrhynchos),
139
zebra finches (Taeniopygia guttata),
110
house finches (Carpodacus mexicanus),
110
house sparrows (Passer domesticus),
110
and European starlings (Sturnus vulgaris)
110
Psittaciformes: budgerigars (Melopsittacus undulatus)
53,110
Mortality rates in these species ranged from 0 to 100%, and the range of tissues affected varied per species, which illustrates the great variability in the susceptibility to HPAI H5N1 virus infection among bird species. However, microscopic lesions in affected species were mostly in the brain and consisted of nonsuppurative encephalitis associated with virus antigen expression in neurons.
Animal Models for Influenza in Humans
Animal models help to better understand influenza in humans and develop medical countermeasures against this disease. These models differ according to the aspect of human influenza that they are designed to mimic and the goal for which they have been developed. Forms of human influenza for which animal models have been developed include uncomplicated influenza, influenza pneumonia, influenza-associated bacterial sepsis, influenza-associated neurologic disease, influenza in immunocompromised hosts, and virus transmission. 5 Goals for which animal models are designed include determining the transmissibility of different viruses, the virulence of different viruses, the pathogenesis of viral infection, and the efficacy of vaccines or antiviral drugs.
Important variables in animal models for influenza are route of inoculation and experimental animal species. Routes of inoculation include exposure to infected animals, exposure to aerosolized viruses, endotracheal or endobronchial instillation, and intranasal instillation. 91 Experimental animal species include laboratory mouse (Mus musculus domesticus), 12 domestic ferret (Mustela putorius furo), 12 Syrian hamster (Mesocricetus auratus), 36 chinchilla (Chinchilla lanigera), 39 domestic horse (Equus ferus), 159 laboratory rat (Rattus norvegicus), 125 domestic dog (Canis lupus familiaris), 24,40 domestic cat (Felis catus), 121 cotton rat (Sigmodon hispidus), 18 domestic pig (Sus scrofa), 80 guinea pig (Cavia porcellus), 74 and nonhuman primates (eg, squirrel monkey, Saimiri sciureus; cynomolgus macaque, Macaca fascicularis; rhesus macaque, Macaca mulatta). 27,72,95
We here review the pathology and pathogenesis of viral pneumonia in 3 commonly used experimental animal species: laboratory mouse, domestic ferret, and cynomolgus macaque. The use of each species has its advantages and disadvantages (Table 1 ).
Advantages and Disadvantages of Experimental Animal Species Commonly Used to Model Influenza in Humans
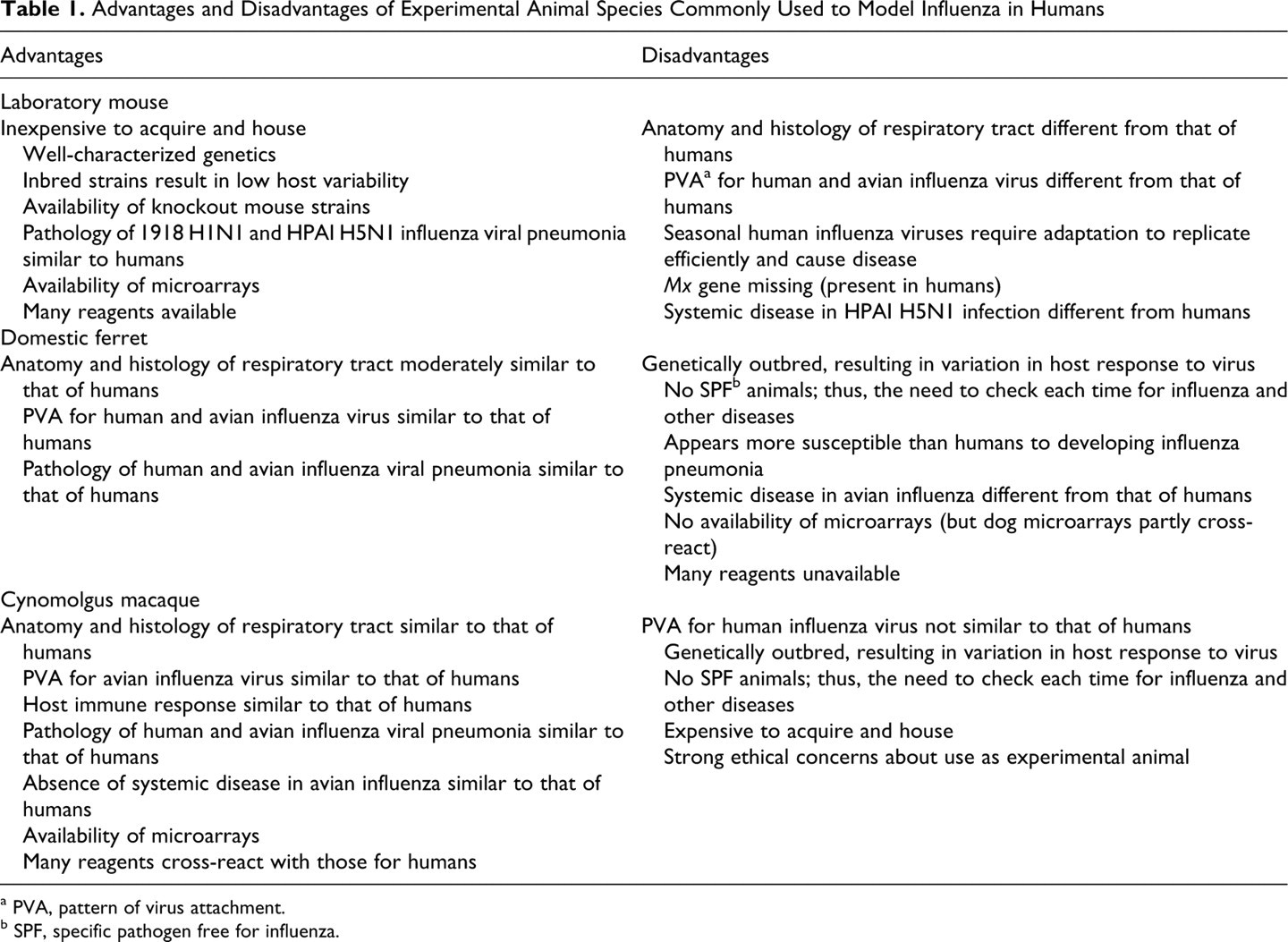
a PVA, pattern of virus attachment.
b SPF, specific pathogen free for influenza.
Influenza in Laboratory Mice
Susceptibility of laboratory mouse strains to influenza virus infection and associated disease differs greatly, 16 and so the strain chosen greatly influences the outcome of infection. Seasonal human influenza viruses require adaptation to mice to efficiently replicate and cause disease; in contrast, mice are generally more susceptible to 1918 H1N1, HPAI H5N1, and pH1N1 viruses. These phenomena are likely due to the differences in receptors for influenza virus in the respiratory tract of mice compared to those in humans (see pattern of virus attachment below). Mice are usually used to model influenza viral pneumonia. To this end, they are inoculated intranasally. For this technique, anesthetized mice are held in a vertical position; the viral suspension is deposited in the nares; and part of the inoculum is aspirated into the lungs. 91 Therefore, intranasal inoculation in anesthetized mice may be comparable to intratracheal inoculation in ferrets and macaques.
Human Influenza Viruses
By virus histochemistry, seasonal human influenza viruses of the H1N1 and H3N2 subtypes show no attachment to trachea, bronchi, or bronchioles of C57/BL6 mice (Fig. 1). There was occasional attachment to alveolar epithelial cells of indeterminate type. 154 The pattern of attachment of the 1918 H1N1 and pH1N1 viruses in the mouse respiratory tract has not been determined.
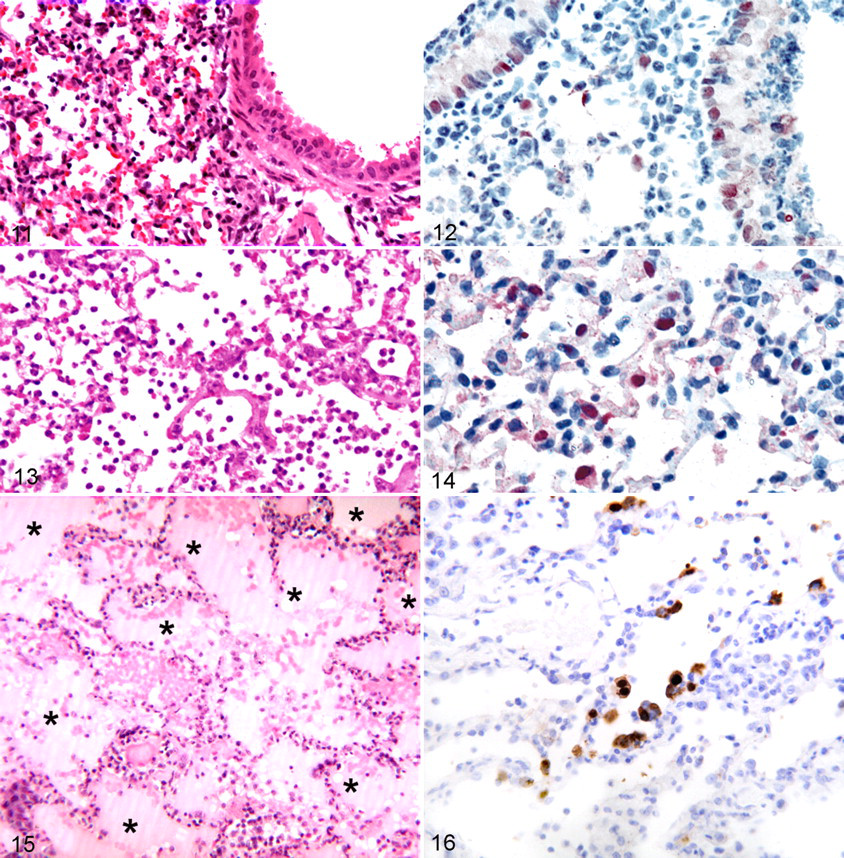
In general, 1918 H1N1 influenza virus infections have frequently been studied in mice. 60,61,90,111,113,161 Experimental infection with the 1918 H1N1 virus caused high mortality in mice, with lesions restricted to the respiratory tract. The most consistent histologic lesions included necrotizing bronchitis, bronchiolitis, and alveolitis with alveolar edema and alveolar hemorrhage (Fig. 11 ), with viral antigen in ciliated bronchiolar epithelial cells, type II pneumocytes, and alveolar macrophages (Fig. 12). 149 The alveolitis was peribronchiolar to diffuse in distribution and primarily neutrophilic in character, although histiocytes also were prominent. Lungs of mice infected with the 1918 H1N1 virus had about 3 times more inflammatory cells than those of mice infected with a seasonal H1N1 virus but similar numbers as those infected with HPAI H5N1 virus. 111
Not all gene segments had equal impact on the virulence of the 1918 H1N1 virus. An influenza virus with 5 gene segments from the 1918 H1N1 virus and with the remaining three from a seasonal human influenza virus (1918 HA/NA/M/NP/NS:Tx/91) produced slightly less severe lung lesions than those of the complete 1918 H1N1 virus. 149 Other recombinants containing the hemagglutinin of the 1918 H1N1 virus (1918 HA:M88 and 1918 HA/NA:WSN) also caused severe pulmonary lesions. However, a virus with the hemagglutinin of a seasonal human influenza virus and the remainder from the 1918 H1N1 virus (Tx/91 HA:1918) produced mild lesions. Together, these results indicate that the 1918 H1N1 HA gene segment is essential to produce severe pulmonary lesions. 60,66,149
Experimental 1918 H1N1 virus infection of mice markedly activated proinflammatory and cell death pathways. Infection with this virus increased expression of genes associated with T helper 1, natural killer, macrophage, and neutrophil populations in lungs of acutely infected mice and was associated with increased expression patterns for proinflammatory cytokines and chemokines, nitrous oxide metabolism, and expression of apoptosis-related genes. 61 Such gene expressions included significant activation of death receptor, interleukin-6 (IL-6), type I interferon, and Toll-like receptor responses, which produced further increases in proinflammatory and cell death responses. 61 Fas, caspase-8, and caspase-9, related to induction of apoptosis, also increased in expression. 61 Macrophage inflammatory protein 1α (MIP-1α), MIP-2, MIP 3α, monocyte chemotactic protein 1 (MCP-1), IL-6, IL-12, IL-1β, IL-18, and granulocyte colony-stimulating factor increased in lungs of mice infected with complete 1918 H1N1 or 1918 H1N1 HA:M88 viruses as compared to those infected with seasonal human H1N1 virus. These results indicate that the 1918 H1N1 HA gene induces macrophage activation and mixed leukocyte recruitment (including neutrophils). 60,111,131 By contrast, G2/M checkpoint control and glutathione metabolism, which are important for successful clearance of influenza virus infection and tissue remodeling, decreased in mice infected with 1918 H1N1 virus, compared to those infected with seasonal human H1N1 virus. 61
Experimental pH1N1 virus infection in mice resulted in prominent bronchiolitis and alveolitis with viral antigen in ciliated bronchiolar epithelial cells, type II pneumocytes, and alveolar macrophages. 54 The median lethal dose for mice (MLD50) for pH1N1 was 105.8 plaque-forming units (pfu), which is in the range of the MLD50 of some other H1N1 viruses: 104.5 to greater than 106.6 pfu. However, compared to currently circulating seasonal human H1N1 viruses, pH1N1 virus replicated more efficiently in the mouse lungs and caused more severe lesions. Virus antigen expression was restricted to the respiratory tract and was present in airway epithelium and in alveoli. 54
Avian Influenza Viruses
By virus histochemistry, avian influenza viruses in mice (including HPAI H5N1 virus) attached abundantly in the trachea to both ciliated and nonciliated epithelial cells (Fig. 1). Attachment was moderate in bronchi and bronchioles, predominantly to nonciliated cuboidal epithelial cells. Attachment was also moderate in alveoli, predominantly to type II pneumocytes. 154 The pattern of attachment of avian influenza viruses in the upper respiratory tract of mice has not been determined.
Experimental HPAI H5N1 virus infection in mice generally caused severe disease and high mortality. Grossly, pulmonary consolidation was observed, whereas heart, liver, and kidney had no significant changes. 100 The main histologic lesions occurred in the respiratory tract. There was epithelial necrosis in nasal cavity, trachea, bronchi, and bronchioles, with fibrin and neutrophils in the airway lumina. There was alveolitis characterized by the presence of serous fluid mixed with fibrin, erythrocytes, and neutrophils in alveolar lumina, as well as increased numbers of alveolar macrophages. 33,100 Virus antigen expression in the respiratory tract occurred in respiratory epithelium of nasal cavity, bronchi, and bronchioles. It also occurred in the olfactory epithelium of nasal cavity, alveolar macrophages, alveolar epithelial cells, neurons in the brain, and pulmonary ganglia. 33,84,100 The more pathogenic HPAI H5N1 isolates resulted in systemic disease, characterized by nonsuppurative encephalitis, myocardial necrosis, and (in one study with ddY mice) fat necrosis. 37,84,100 Virus antigen expression colocalized with these lesions in neurons, glial cells, and ependymal cells in the brain; cardiomyocytes in the heart; and adipocytes in fat tissue. 37,84,100 Virus antigen expression was also seen in hepatocytes in absence of associated lesions. 100
The viral genetic factors affecting the virulence of HPAI H5N1 viruses and other emerging avian influenza viruses were reviewed by de Wit and Fouchier. 32 Comparison of two 1997 HPAI H5N1 isolates, one from a patient with fatal infection and the other from a patient with mild disease, showed a similar difference in pathogenicity for mice. The main determinant for this difference was an E627K substitution in the gene for polymerase B2 (PB2). 45 HPAI H5N1 viruses isolated from healthy domestic ducks after 1999 differed remarkably in their pathogenicity in mice, as mainly determined by a D701N substitution in PB2. 77 Finally, the pathogenicity of 2 HPAI H5N1 viruses isolated in Vietnam in 2004, one from a chicken and the other from a human, were compared in mice. The human isolate was lethal whereas the chicken isolate was not. The pathogenicity was determined in part by the polymerase complex: Viruses with a PB2 or PB1 of the chicken virus in the background of the human isolate were attenuated. The polymerase activity of the human isolate was significantly higher than that of the chicken polymerase complex in human cells, indicating that increased virus replication correlated with pathogenicity. 127 Together, these results from experimental HPAI H5N1 viruses in mice indicate that the acquisition of virulence of these avian viruses for mammals is a polygenic trait. 32
Influenza in Ferrets
The domestic ferret is descended from either the European polecat or the steppe polecat, and it has been domesticated for thousands of years. 4 Ferrets develop a productive infection after inoculation with human and avian influenza viruses without prior adaptation of the virus. Experimental influenza virus infections of ferrets have been used to model different aspects of influenza in humans. First, they have been used to model uncomplicated upper respiratory tract infection—the most common use. For these models, ferrets are inoculated intranasally. 54,94 Second, they have been used to model the primary complication of influenza in humans: viral pneumonia. For highly virulent influenza viruses, such as HPAI H5N1 virus and 1918 H1N1 virus, intranasal inoculation is sufficient to consistently induce pneumonia. 150 Infection with these viruses also leads to systemic disease, which illustrates the high susceptibility of ferrets to influenza virus infection. For less virulent influenza viruses, such as pH1N1 virus, intratracheal inoculation is used to consistently generate pneumonia. 151 Third, ferrets have been used to model influenza virus transmission among humans. To this end, they are inoculated intranasally and placed in special cages with controlled air flow that is connected to cages with uninfected ferrets. 94
Human Influenza Viruses
By virus histochemistry, seasonal human influenza viruses of the H1N1 and H3N2 subtypes show abundant attachment in the upper respiratory tract 94 and trachea and progressively less attachment in bronchi and bronchioles (Fig. 1). In the upper respiratory tract, as well as in the trachea, bronchi, and bronchioles, virus attaches predominantly to ciliated epithelial cells. In alveoli, virus attaches more to type I than type II pneumocytes. 154 The pattern of attachment of the 1918 H1N1 and pH1N1 viruses in the ferret respiratory tract has not been determined, except that pH1N1 virus attaches abundantly to ciliated epithelial cells of the upper respiratory tract. 94
Experimental 1918 H1N1 virus infection in ferrets caused severe disease and high mortality. 150 Gross lesions were not reported, but histologic lesions consisted of moderate or marked necrotizing or purulent bronchiolitis and alveolitis with focal areas of acute alveolar edema and hemorrhage (Fig. 13 ). In addition, there was peribronchiolar accumulation of mixed inflammatory cells, alveolar epithelial hyperplasia, and epithelial necrosis in submucosal serous glands. 150 Virus antigen expression occurred in superficial epithelium of bronchi and bronchioles, in epithelium of submucosal serous glands, in alveolar macrophages, and in hyperplastic alveolar epithelium (Fig. 14 ). 54,94 The transmissibility of the 1918 H1N1 virus from ferret to ferret depended on specific gene segments being present. Direct contact transmission required the 1918 H1N1 HA, but efficient respiratory droplet transmission required the 1918 H1N1 PB2 gene segment. 152 The NP and 3 polymerase complex genes were necessary for high-replication efficiency in the respiratory tract of ferrets. 158
Experimental pH1N1 virus infection causes morbidity and mortality in ferrets. Different experimental setups have been used to model different aspects of the disease in humans. Experimental pH1N1 virus infection of ferrets to model viral pneumonia in humans has been performed by intratracheal inoculation. 151 Gross lesions were multifocal or coalescing pulmonary consolidation, involving 20 to 70% of the lung surface. Histologic lesions consisted of diffuse alveolar damage characterized by flooding of alveolar lumina with edema fluid containing alveolar macrophages, neutrophils, erythrocytes, fibrin, and cellular debris. There was a multifocal necrotizing bronchiolitis and a mild bronchitis. Virus antigen expression occurred in type I and type II pneumocytes, alveolar macrophages, bronchiolar epithelial cells, and bronchial epithelial cells. Quantitative scoring showed that pH1N1 virus caused diffuse alveolar damage intermediate in severity between that caused by seasonal H1N1 virus and HPAI H5N1 virus. 151
Experimental pH1N1 virus infection of ferrets to model human-to-human transmission has been performed by intranasal inoculation. 94 The resultant lesions occurred higher in the respiratory tract. There was a mild-to-moderate necrotizing rhinitis, tracheitis, bronchitis, and bronchiolitis, and virus was demonstrated in respiratory epithelium associated with necrosis or inflammation in nasal cavity and airways but rarely in alveoli. 94 The differences between the 2 studies 94,151 in pattern of disease and virus antigen expression are a good illustration of the effect that route of inoculation has on the pathogenesis of experimental influenza virus infection. With regard to transmission, pH1N1 was efficiently transmitted through respiratory droplets between ferrets. 94
Avian Influenza Viruses
In the upper respiratory tract of ferrets, HPAI H5N1 virus shows rare attachment by virus histochemistry. 28 In the lower respiratory tract, avian influenza viruses in ferrets, including HPAI H5N1 virus, show no attachment to tracheal or bronchial epithelial cells and rare attachment to bronchiolar epithelial cells (Fig. 1). They do show attachment in the alveoli, where attachment to type II pneumocytes is more prominent than that to type I pneumocytes. 154
Many experimental HPAI H5N1 virus infections in ferrets have been reported. 15,42,56,79,86,124,151,163,164 The virulence of HPAI H5N1 viruses is variable, with isolates from humans being more virulent than those from birds. 86 HPAI H5N1 viruses isolated from humans caused high morbidity and mortality in ferrets, often accompanied by neurologic signs. 23,42,124,151,164 The main gross lesion was multifocal consolidation of the lung parenchyma, which was dark red, firm, and raised. 151,164 Extrapulmonary lesions were less frequent and included hemorrhages on liver 164 and adipose tissue in peritoneal cavity. 86
Histologic lesions were observed in lung, brain, and liver. Lung lesions affected alveoli, bronchioles, and bronchi and were given different morphologic diagnoses, including diffuse alveolar damage, 151 interstitial pneumonia, 164 and bronchopneumonia. 163,164 Epithelial necrosis, as well as epithelial hypertrophy and hyperplasia, occurred in alveoli, bronchioles, and bronchi. Alveolar lumina contained edema fluid, fibrin, erythrocytes, cellular debris, and variable proportions of neutrophils and mononuclear cells. A mixed inflammatory exudate was also present in alveolar septa. Bronchiolar and bronchial lumina contained variable proportions of neutrophils and mononuclear cells, as well as cellular debris. 15,42,86,151,163,164 Brain lesions were a nonsuppurative necrotizing encephalitis, with changes observed in the cerebrum, cerebellum, brain stem, olfactory bulb, choroid plexus, and meninges. Lesions were characterized by neuronal degeneration, neuronophagia, glial nodules, and perivascular accumulation of mononuclear cells, sometimes with neutrophils. 15 Liver lesions included multifocal hepatitis characterized by hepatocellular necrosis, hemorrhage, and accumulation of mononuclear cells or mixed inflammatory cells. There was also necrosis and hyperplasia of bile duct epithelium. 15,42,163
Virus antigen expression occurred in lung, brain, and liver. In the lung, it occurred in type I and type II pneumocytes, bronchiolar epithelial cells, and bronchial epithelial cells. 86,151,164 Virus antigen expression was also seen in neurons 86,163 and hepatocytes. 163 In all three organs, virus antigen expression colocalized with characteristic histologic lesions.
Studies on the transmission of HPAI H5N1 viruses in ferrets have shown negligible transmissibility via respiratory droplets. Ferrets inoculated with HPAI H5N1 viruses rarely show sneezing, which is an efficient way to produce and spread infected respiratory droplets. 85 In contrast, there is repeatable, albeit inefficient, transmission of HPAI H5N viruses via direct contact. 163 Transmissibility of influenza viruses in ferrets is likely associated with their virus infectivity and level of virus shedding from the respiratory tract. 85 Reassortment of HPAI H5N1 virus genes with human H3N2 influenza virus genes was shown to be insufficient for ferret-to-ferret transmission of HPAI H5N1 virus, suggesting that additional unknown mutations are required for HPAI H5N1 virus to become efficiently transmissible and cause a human influenza pandemic. 85 It is possible that the currently circulating HPAI H5N1 viruses may require a receptor-binding change to a predominant α-2,6 sialic acid–binding preference to become efficiently transmissible among humans. 150 Some of the mutations in the hemagglutinin that might cause such a change have recently been tested. 28
Influenza in Cynomolgus Macaques
Macaque species used to model influenza virus infection in humans are cynomolgus (M fascicularis), 72 rhesus (M mulatta), 27 and pig-tailed macaques (Macaca nemestrina). 8 There is serologic evidence of influenza A and B virus infections in captive and free-living macaque species, 58,62 and no prior adaptation of human or avian isolates of influenza A virus is required to cause a productive infection in experimentally inoculated macaques. In nearly all studies, macaques have been used to model the pulmonary complication of influenza virus infection—namely, viral pneumonia. To this end, the preferred route of inoculation is intratracheal, either by itself 71 or with inoculation of tonsil, conjunctiva, and/or nose. 7,29,72,122 This macaque model of influenza viral pneumonia is used to compare virulence of different influenza viruses, 65 to study the pathogenesis, 7,29 and to test antiviral agents 133 or vaccines. 71
Human Influenza Viruses
By virus histochemistry, seasonal human influenza viruses of the H1N1 and H3N2 subtypes show no attachment to epithelial cells of the lower respiratory tract (Fig. 1). 154 The pattern of attachment of the 1918 H1N1 and pH1N1 viruses in the macaque respiratory tract has not been determined.
Experimental 1918 H1N1 virus infection into cynomolgus macaques caused acute respiratory distress, culminating in the death of some animals. Gross lesions included pulmonary consolidation involving 60 to 90% of the lung surface by 6 to 8 days postinoculation (dpi). Microscopic lesions included diffuse alveolar damage with extensive edema and hemorrhage (Fig. 15). Virus antigen expression occurred in both flat and plump alveolar cells, interpreted as type I and type II pneumocytes, respectively (Fig. 16). Compared to infection with a seasonal human influenza H1N1 virus, virus infection of the lung was protracted and associated with progressive pulmonary damage. There was more upregulation of IL-6, CCL11 (eotaxin 1), CXCL6 (granulocyte chemotactic protein 2), and CXCL1 (neutrophil-activating protein 3); note that CXCL6 and CXCL1 are involved in the activation and recruitment of neutrophils. There was less upregulation of IFN-α genes, DDX58 (retinoid acid–inducible protein I) and IFIH1 (melanoma differentiation–associated gene 5), which are involved in antiviral response. Together, these pathology and cytokine results suggest that the innate response to this 1918 H1N1 virus was dysregulated, resulting in insufficient protection against infection. 65
Pulmonary lesions and associated viral antigen expression in commonly used experimental animal species infected with 1918 H1N1 virus.
Experimental pH1N1 virus infection in cynomolgus macaques caused morbidity but no mortality. 48,54 Gross lesions included focal or multifocal pulmonary consolidation and enlargement of the tracheobronchial lymph nodes. Microscopically, there was diffuse alveolar damage characterized by flooding of alveolar lumina by highly proteinaceous fluid, rare hyaline membranes positive for periodic acid–Schiff, necrosis of alveolar epithelial cells, and accumulation of many neutrophils and alveolar macrophages. In addition, there was moderate bronchiolitis and mild bronchitis, tracheitis, and rhinitis. 48 Virus antigen expression occurred mostly in type I and type II pneumocytes and rarely in epithelial cells of larynx and nasal cavity. 48,54 The abundance of neutrophils and macrophages in pulmonary lesions corresponded with upregulation of CCL2, CCL3, CCL8, CXC10, IL-8, and CXCL1, which are known chemoattractants for neutrophils and monocytes. 48 Concentrations of MCP-1, MIP-1α, IL-6, and IL-18 were higher as compared to those of the lungs of cynomolgus macaques infected with seasonal human H1N1 virus. 54 In sum, the severity of pH1N1-virus-induced pulmonary lesions in cynomolgus macaques was higher than that induced by seasonal human influenza H1N1 virus but lower than that induced by HPAI H5N1 virus or 1918 H1N1 virus. 48,54
Avian Influenza Viruses
By virus histochemistry, avian influenza viruses in cynomolgus macaques, including HPAI H5N1 virus, show no attachment to tracheal epithelial cells and no or rare attachment to bronchial epithelial cells (Fig. 1). They do show attachment in the bronchioles, predominantly to nonciliated cuboidal epithelial cells. They also show attachment in the alveoli, where attachment to type I pneumocytes is more prominent than to type II pneumocytes. 154 The pattern of attachment of avian influenza viruses in the upper respiratory tract of macaques has not been determined.
Experimental influenza virus infections in macaques have been most extensively studied for HPAI H5N1 viruses. Infections with these viruses in cynomolgus macaques caused morbidity and mortality in part of the inoculated animals. The main gross lesion included focal, multifocal, or coalescing consolidation of the lung parenchyma, which was red-purple, firm, slightly depressed, and less buoyant than normal. 7,27,71,72,122 There was pulmonary edema characterized by a variable volume of clear or red foamy fluid in the trachea and bronchi 7,72 Tracheobronchial lymph nodes and tonsils were enlarged. 7,72 No gross lesions were observed elsewhere, despite careful examination of extrapulmonary organs.
The most severe histologic lesions at 4 dpi centered on the alveoli and bronchioles and consisted of diffuse alveolar damage. 72,122 The lumina of alveoli and bronchioles were variably filled with protein-rich edema fluid, fibrin, erythrocytes, and cell debris, admixed with many neutrophils and mononuclear cells and rare eosinophils and lymphocytes. The alveolar and bronchiolar walls showed extensive loss of epithelium and were thickened by the presence of edema fluid, fibrin, neutrophils, eosinophils, 71 and mononuclear cells. There was perivascular and peribronchiolar infiltration with many lymphocytes and plasma cells and with few macrophages, neutrophils, and eosinophils. 71 There was mild epithelial degeneration and necrosis with associated inflammation in bronchi, trachea, and larynx. 72,122 Extrarespiratory tissues were generally free of histologic lesions. 7,27,72,122 However, suppurative tonsillitis, lymphocytic necrosis in lymphoid organs, hepatic necrosis, and renal tubular necrosis have been seen rarely. 72,122
Virus antigen expression occurred mainly in alveoli, bronchioles, and bronchi. In alveoli, it occurred in type I and type II pneumocytes, alveolar macrophages, neutrophils, and unidentified mononuclear cells, 27,71,72,122 as well as in endothelial cells. 7 The ratio of positive type II pneumocytes to positive type I pneumocytes was 3:1 or higher. 7 In the bronchioles, small bronchi, and large bronchi, influenza virus antigen was seen in ciliated epithelial cells and intraepithelial neutrophils, as well as in sloughed epithelial cells, macrophages, and neutrophils in the bronchial lumen. No influenza virus expression was seen in the trachea. 27,72,122 Outside the respiratory tract, influenza virus antigen expression was observed in cryptal epithelial cells and dendritic cells in the germinal centers of tonsils 72,122 and in unspecified cells in tracheobronchial and retropharyngeal lymph nodes, spleen, and leptomeninges. 7 No influenza virus expression was seen in other extrarespiratory organs. 27,72
The course of pulmonary disease has been followed from 12 hours postinoculation to 16 dpi. The first lesions were described at 1 dpi and consisted of necrotizing alveolitis and bronchiolitis. 7 The lesions were severe at 4 dpi, as described above. By 7 dpi, the severity was comparable but with relatively more lymphocytes and other mononuclear cells than neutrophils in the alveolar lumina and with regeneration of respiratory epithelium in alveoli (type II pneumocyte hyperplasia) and bronchioles. 27,72,122 The severity of pulmonary lesions at 10 dpi and 14 dpi was less than that at 7 dpi, with diffuse alveolar damage rarely observed (10 dpi) or not at all (14 dpi). 27 At 14 dpi, type II pneumocyte hyperplasia and interstitial fibrosis were prominent. 27 At 16 dpi, there was prominent peribronchiolar lymph follicle development and tissue regeneration. 29
The course of virus antigen expression over time parallels that of pulmonary disease. In one study, 29 virus expression was seen at the first time point, 12 hours postinoculation, in cells of the terminal and respiratory bronchioles. Virus expression expanded on 1 dpi to bronchi and alveoli and was maximal at 2 dpi, when it occurred in alveoli at the edge of lesions of alveolar edema. On the following days (3 to 16 dpi), the number of cells expressing viral antigen decreased. In another study, 27 virus antigen expression was seen only in scattered cells in the lung tissue at 10 dpi and was absent by 14 dpi. 27
Influenza in Other Experimental Animals
Less widely used models for human select agent influenza include guinea pig, domestic pig, and domestic cat. Although not a natural host of influenza virus infection, the guinea pig has been a model for respiratory transmission of human seasonal influenza. 83 Neither H5N1 HPAI nor 1918 H1N1 viruses cause severe clinical signs or mortality. 74,152 Historically, the first select agent influenza infections in guinea pigs may have occurred naturally, based on a report of guinea pigs dying with severe pneumonia in a vivarium that was spatially and chronologically associated with influenza in soldiers during the Spanish influenza pandemic. 75 Lesions with 1918 H1N1 and H5N1 HPAI viruses have been limited to the respiratory tract and are milder than those seen in mice or ferrets. 74,152
The domestic pig has been proposed and used as a model of human influenza because of cross transmission of human and swine influenza viruses between pigs and people under natural settings. 96 The pig respiratory epithelium has α-2,6- and α-2,3-linked sialic acid moiety receptors that can bind avian, swine, and human influenza viruses. 88 Pigs have been proposed as a mixing vessel for avian and human influenza virus genes for generation of pandemic viruses. 129 Pigs were naturally infected in 1918 with the H1N1 Spanish influenza virus, 67 and they have similar clinical signs and respiratory lesions as do humans infected by seasonal influenza viruses. Select agent influenza viruses have had limited studies in pig models. Intranasal inoculation with some H5N1 HPAI viruses failed to replicate or produce clinical disease, whereas other H5N1 HPAI viruses produced respiratory replication, mild reduction in weight gain, slight increase in body temperature, mild respiratory signs, mild bronchiolitis, and alveolitis and failed to transmit pig to pig. 17,32,56,103 However, the 1918 H1N1 virus produced mild respiratory signs, increased temperature, and bronchiolitis and alveolitis without the mortality seen in mice and ferret models. 126
The domestic cat has been used as a model for systemic disease from HPAI H5N1 influenza. Domestic cats reproducibly develop systemic disease after intratracheal inoculation or ingestion of infected meat 121 and so can be used to investigate the pathogenesis of influenza virus–induced lesions in extrarespiratory organs (Figs. 4–6). Furthermore, of all animal species studied, the attachment pattern of H5N1 HPAI virus most closely resembles that in the human respiratory tract. As in humans, H5N1 HPAI virus attaches predominantly to nonciliated cuboidal cells in the terminal bronchioles and to type II pneumocytes and alveolar macrophages in the alveoli. 154,155
The Way Forward
This review shows that there are well-established models among experimental animal species for influenza in humans. Still, there is room for improvement in different ways. First, it is important to better characterize the pathology of influenza in humans so that it is possible to determine whether the models are accurate reflections of the human situation. Because the many pathology studies of human influenza took place at the time of the pandemics in 1918, 1957, and 1968, they lacked the benefit of advanced immunologic and molecular biological techniques at our disposal today. Current cases of HPAI H5N1 and pH1N1 virus infection in humans provide opportunities for pathology studies on biopsies of influenza patients and on autopsies of fatal cases. In this context, veterinary and human pathologists should work together to develop a common terminology for the lesions and morphologic diagnoses of influenza viral pneumonia. Second, currently used animal models of influenza would benefit from better characterization of pathogenesis and pathology. There is a need for better understanding the temporal progression of pulmonary lesions—namely by time course experiments—so that the timing of sacrifice for pathologic examination is based on pathologic as well as virologic and clinical criteria. There is also a need for better integration of histopathologic, cytokine, and microarray data to gain a complete understanding of the pathogenesis of influenza virus infection. Third, research using animal models of influenza would profit from standardization of the most commonly used models to allow comparison of results from different experiments. Procedures to standardize include method of anesthesia, route of inoculation, viral dose of inoculum, volume of inoculum, day of sacrifice, and a semiquantitative grading system for scoring pulmonary lesions. Fourth, the exposure of experimental animals to aerosol as a route of inoculation needs to be developed to study select agent influenza viruses with regard to their threat as a biological weapon. Although aerosol exposure of experimental animals to influenza virus was commonly used in the past, 43,128 it is currently not used, probably because of disadvantages such as costly equipment (including its regular maintenance and testing) and the potential exposure of laboratory personnel to airborne infection. 91
These animal models may be applied for the development of 2 types of products. In the first place, there is a need for new antiviral drugs to supplement the 2 current options of neuraminidase inhibitors (oseltamivir, zanamivir) and M2 inhibitors (amantadine, rimantadine). These drugs suffer from 3 problems: antiviral resistance, limited antiviral efficacy in severe cases of influenza, and lack of parenteral use. 46 Increased understanding of the mechanisms of influenza virus replication has revealed potential new antiviral agents, including T-705 (a polymerase inhibitor) and DAS181 (an attachment inhibitor). These and other agents require testing in appropriate animal models to determine their safety and efficacy.
In the second place, there is a need for more efficacious vaccines against influenza virus infection. Problems include the need for high antigen dose, the limited production capacity, the long production time, the poor immunogenicity of adjuvants, and the limited cross-protective immunity (intrasubtypic and intersubtypic). 70 The use of reverse genetics to produce vaccine reassortant strains, the use of cell culture for mass vaccine production, and the development of more potent adjuvants will contribute to the more rapid availability and higher production of safe and effective vaccines. Again, suitable animal models are required for the evaluation of the efficacy and safety of novel influenza vaccines. 70
Footnotes
The authors declared no conflicts of interests with respect to the authorship or the publication of this article.
This work was supported by US Department of Agriculture Current Research Information System project No. 6612-32000-048-00D (M.P.J., D.E.S.), Dutch Ministry of Economic Affairs grant Impuls Veterinair Influenza Onderzoek Nederland, and VIRGO consortium, an Innovative Cluster approved by the Netherlands Genomics Initiative and partially funded by the Dutch Government (BSIK 03012; T.K., D.V.R.).
