Abstract
Tularemia is a highly contagious infectious zoonosis, transmissible by inoculation, ingestion, or inhalation of the infectious agent Francisella tularensis. The disease is perpetuated by infected rodents, blood-sucking arthropods, and by contaminated water. Therefore, nonhuman primates housed outdoors may be at risk for exposure. An epizootic of F. tularensis occurred in an indoor/outdoor-housed group of cynomolgus monkeys (Macaca fascicularis) at the German Primate Center. Tularemia was diagnosed in 18 out of 35 animals within a period of 2 years. Six animals died with unspecific clinical symptoms; 12 animals developed seroconversion and were still alive. Pathologic findings were similar in all monkeys that died and resembled the clinical picture of the human disease, including an ulceroglandular syndrome with local lymphadenopathy, gingivostomatitis, and systemic spread, with manifestations such as subacute necrotizing hepatitis, granulomatous splenitis, and pneumonia. Tularemia was diagnosed by culture, real-time polymerase chain reaction, and ELISA techniques. This is the largest outbreak in nonhuman primates and the first report of tularemia in cynomolgus monkeys. An overview of the recent literature about tularemia in nonhuman primates is given.
Francisella tularensis is a zoonotic gram-negative bacterium, which causes the disease tularemia. It is widespread in North America as well as in parts of Europe and Asia. The organism is able to infect several different species of mammals and even some species of birds and reptiles. Rodents and lagomorphs are highly susceptible and are considered to be the main reservoir hosts in many areas of the world. Hematophagous arthropods like mosquitoes and ticks have been suggested as the main vectors.5 Transmission is often associated with an arthropod vector, but the infection can also be acquired orally, via the respiratory route, by bites of infected vertebrates, or from direct contact with infected tissue. Regardless of the route of infection, tularemia is a serious and often fatal disease. The course of infection depends on the virulence of the infectious strain, the portal of entry, the extent of systemic involvement, and the immune status of the host. Six classic forms of tularemia are described in human medicine. The predominant manifestations of human disease are the ulceroglandular, glandular, oculoglandular, pharyngeal, typhoidal, and the pneumonic form; however, overlapping of the different symptoms is observed frequently.13
Outbreaks of tularemia in nonhuman primates are fairly uncommon. There are only a few reports of naturally acquired tularemia in nonhuman primates.3,4,6,7,14,15 Most of the reported cases were observed among New World monkeys and lemurs in parts of the United States, where the disease is endemic.3 There are only 2 case reports on tularemia in great apes, including a lowland gorilla from Oklahoma City Zoo and Bornean orangutans in the Topeka Zoo.2,12 The situation in Europe is totally different. Only 2 cases of tularemia, 1 in a golden–headed lion tamarin (Leontopithecus chrysomelas) and another one in a common marmoset (Callithrix jacchus), were described in Switzerland.11,17
The clinical symptoms of the disease are nonspecific and resemble respiratory tract infections or gastroenteritis caused by other common bacterial or viral pathogens.11,17 Generally, infected animals developed a severe febrile disease.3 Often large lingual ulcers or abscesses in the tongue and severe submandibular lymphadenopathy were described as the most prominent findings.4,15
In most countries, human as well as animal disease shows seasonal patterns, especially in arthropod-borne tularemia. Contact with infected hares or rodents accounts for most cases of human disease in Western and Central Europe. In North America, human infections are mainly caused by tick bites or contact with rabbits. Wild ground squirrels, which play a dominant role in the epidemiology of tularemia in Canada, were identified as the source of infection in a Canadian zoological garden. Three black and red tamarins (Saguinus nigricollis) and 1 talapoin (Cercopithecus talapoin) died during an epizootic of tularemia. Ground squirrels were frequently seen around the building, and fleas were assumed to be the potential vector. F. tularensis was recovered from organs of 1 ground squirrel and from fleas infesting the squirrel. The veterinarian treating the monkeys was bitten in the hand by an infected animal and developed severe lymph node enlargement, malaise, fever, and diarrhea.15
In another case of a dead squirrel monkey housed by a private owner, epidemiologic investigation suggested that transmission of infection may have happened in the pet shop from which the monkey was purchased.6 Bornean orangutans (Pongo pygmaeus borneo) developed illness after numerous rabbits were found dead on grounds of Topeka Zoo, including the outdoor orangutan exhibit.12 Aside from these natural infections, pulmonary tularemia has been experimentally induced by inhalation of aerosols or nasal installation of infectious organisms in rhesus macaques (Macaca mulatta) and grivet monkeys (Cercopithecus aethiops).1,10
In Germany, tularemia in hares has not been reported since 1992, and notified human cases are in the range of 0–5 cases only per year. In the district of Göttingen, Lower Saxony, tularemia has never been reported before. The present study provides a detailed pathologic description of a tularemia epizootic outbreak among cynomolgus monkeys housed in an indoor-outdoor husbandry in this district of Germany.
Materials and Methods
Pathohistologic Examination
Organ samples from 6 cynomolgus monkeys were included in this study. The monkeys died in a time interval from late summer to early winter without previous signs of the disease. There was an equal sex ratio with 3 female and 3 male monkeys (Table 1). All animals were necropsied, and a complete spectrum of all tissue specimens was fixed in 10% neutral-buffered formalin as well as frozen in liquid nitrogen. Light microscopy was performed on paraffin-embedded tissue specimens. Sections of 3–4 μm were stained routinely with HE. Special stains including periodic acid–Schiff (PAS)-reaction, Grocott silver stain, Ziehl-Neelsen-stain, Giemsa and Gram's stains were used to evaluate the presence of infectious agents. Immunohistochemistry was performed on formalin-fixed, paraffin-embedded sections using a monoclonal antibody against F. tularensis antigen (Clon 11/1/6) in a dilution of 1:1,000 after heat pretreatment with citrate buffer (pH 6.0).8 Additional sections were stained with polyclonal rabbit antibodies (1:1,000 dilution) raised against F. tularensis (Live Vaccine Strain [LVS]) that recognize lipopolysaccharide (LPS). Biotinylated secondary antibodies, streptavidin, and the color-indicator alkaline phosphatase red detection kit (Cat. 760-031, Ventana, Tucson, AZ) were applied according to the supplier's instructions. Two negative controls, first, replacement of the primary antibody with sheep serum, and second, tissue sections of a control animal, were performed. For counterstaining, Mayer's hematoxylin was used.
Signalment, clinical signs of disease, and pathohistologic findings in cynomolgus monkeys with tularemia.
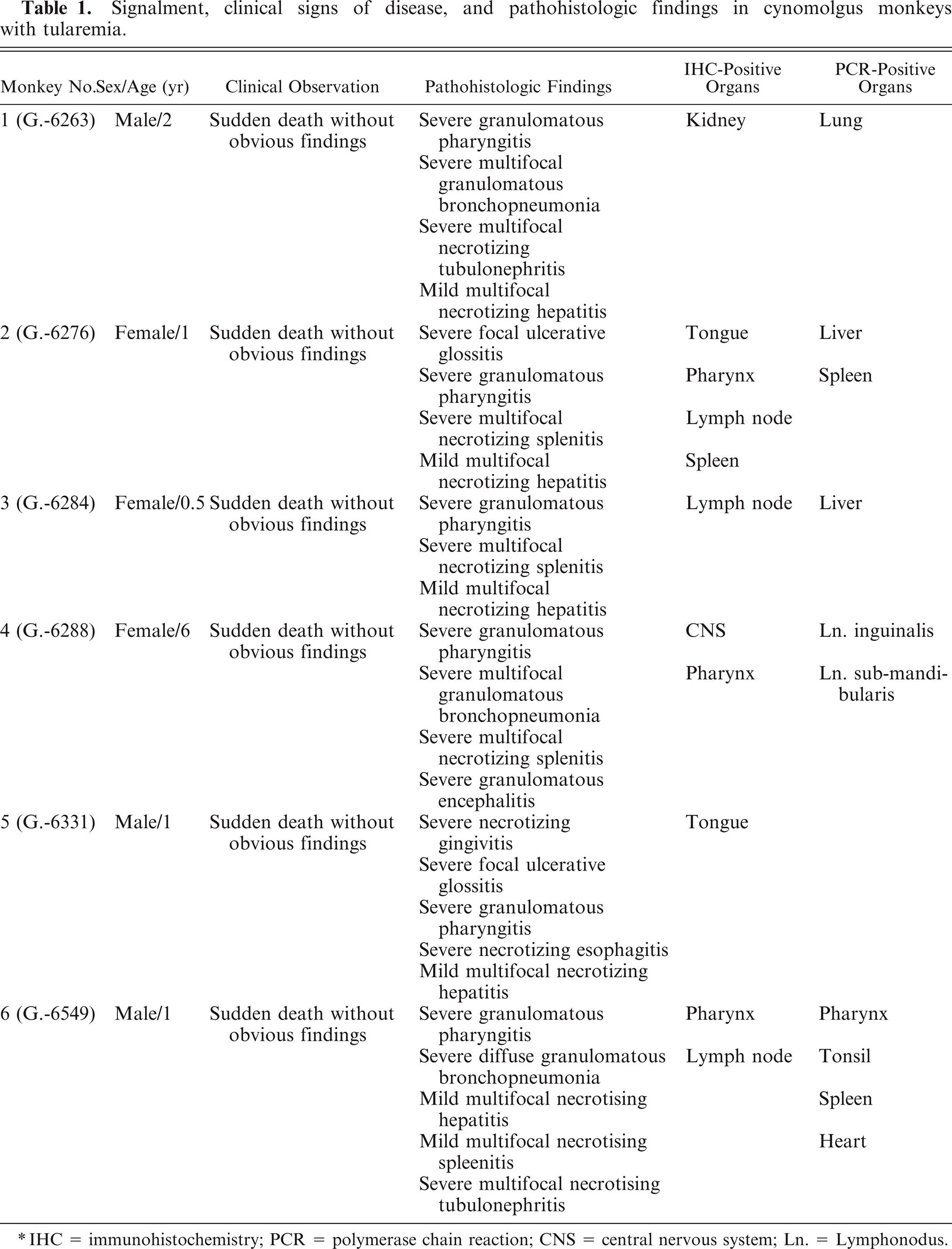
IHC = immunohistochemistry; PCR = polymerase chain reaction; CNS = central nervous system; Ln. = Lymphonodus.
Serology
Serum was collected from all monkeys living in the colony in 2002, 2004, and 2005 and analyzed for the presence of anti-F. tularensis antibodies using a competitive ELISA as described recently.18
Bacterial Isolation and Identification
Identification of the causative agent was achieved by culture, ELISA, and molecular techniques: Homogenized tissues from the spleen and liver of all animals were plated on conventional as well as enriched culture media suitable to support growth of Francisella species. Measures to control growth of contaminating bacteria were also implemented.16 For detection of F. tularensis–specific LPS, an antigen-capture ELISA was used.9 All specimens were also analyzed for the presence of F. tularensis sequences, employing 2 different real-time polymerase chain reaction (PCR) techniques.18,19
Results
Naturally acquired tularemia was diagnosed in 6 cynomolgus monkeys at the postmortem examination. The etiology was confirmed by histopathology, immunohistochemistry, culture, and PCR techniques. All animals that died were of young age, generally between 0.5 and 1 year old, with only 1 adult affected (Table 1).
In all cases, gross necropsy findings included marked splenomegaly (Fig. 1) and mild hepatomegaly, with numerous necrotic white foci up to 1 mm in diameter distributed throughout the parenchyma. The livers were friable with blunted margins. A severe multifocal subacute granulomatous bronchopneumonia was found in 3 animals (monkeys Nos. 1, 4, and 6) (Fig. 2). All animals developed severe inflammatory alterations of variable extent within the oral cavity and the pharyngeal region. The pharyngeal lymphoid tissue was enlarged and heavily inflamed and showed multifocal caseous abscess formations (Fig. 3). Oligofocal tongue ulcers were found in 2 animals (monkeys Nos. 2 and 5) (Fig. 3, inset). The described lesions in the oral cavity were accompanied by a severe gingivitis in monkey No. 5 (Fig. 4).
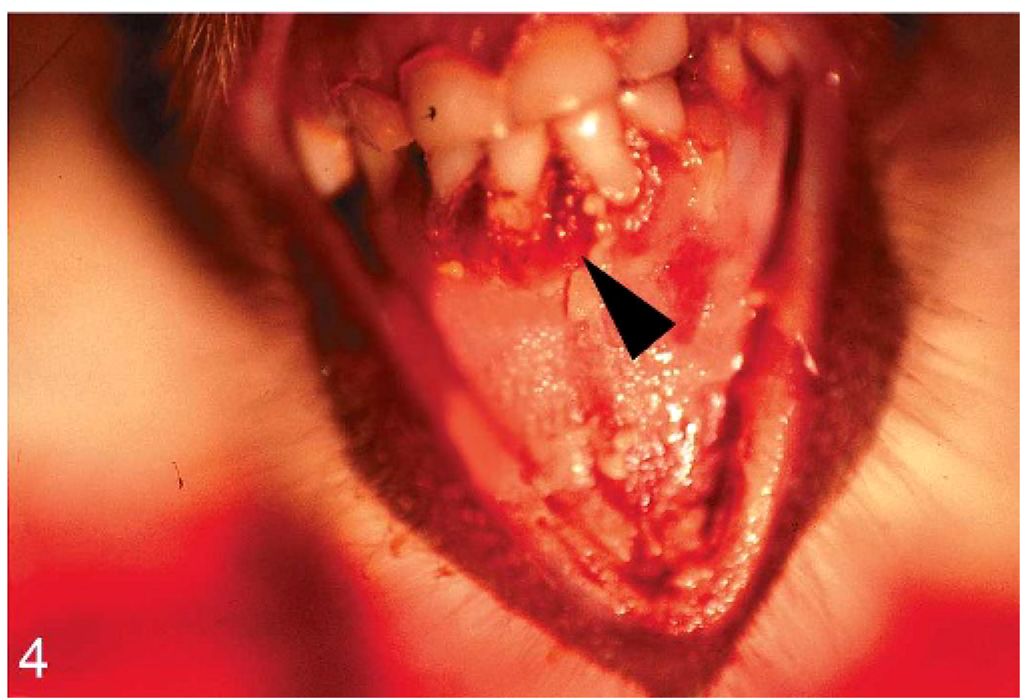
Oral cavity; cynomolgus monkey, monkey No. 5. Severe erosive-ulcerative gingivitis (arrow head) is seen.
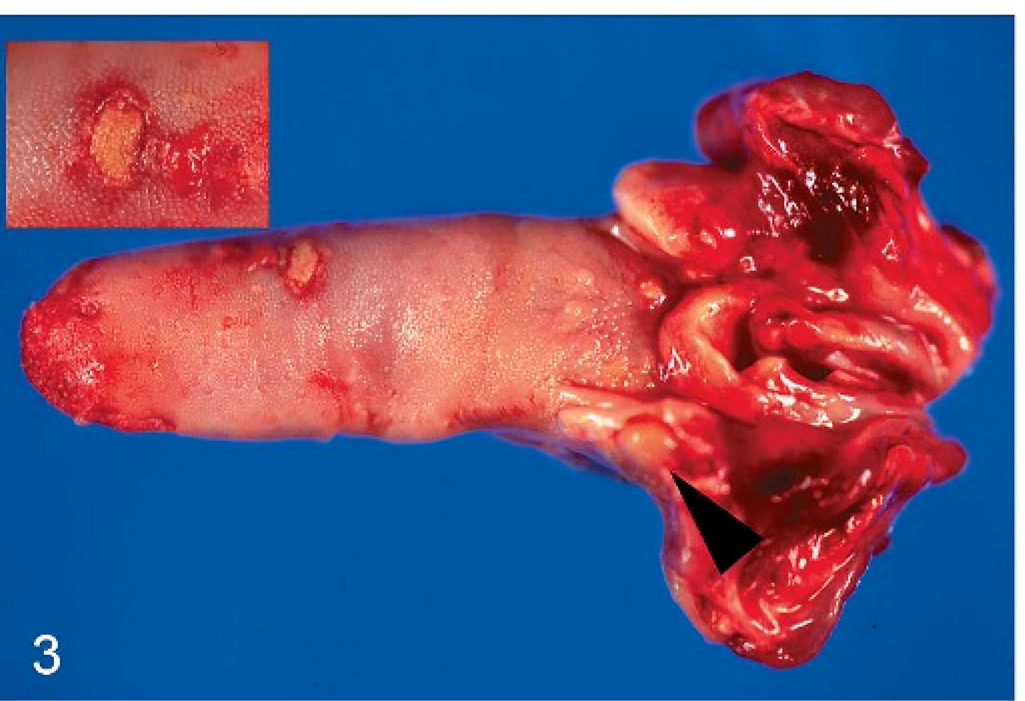
Tongue; cynomolgus monkey, monkey No. 2. Focal sharply demarcated tongue ulceration (inset) and granulomatous inflammation of the tonsils with abscess formation (arrow head) are seen.
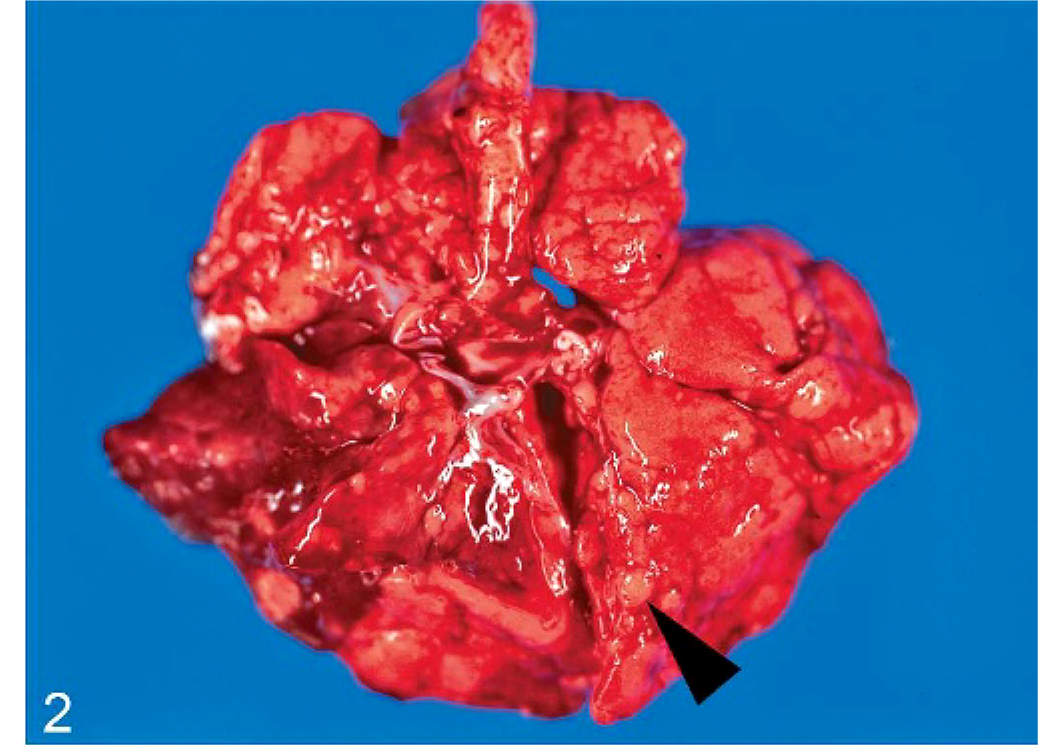
Lung; cynomolgus monkey, monkey No. 6. Severe granulomatous pneumonia has numerous granulomatous lesions (arrow head) distributed throughout the lung lobes.
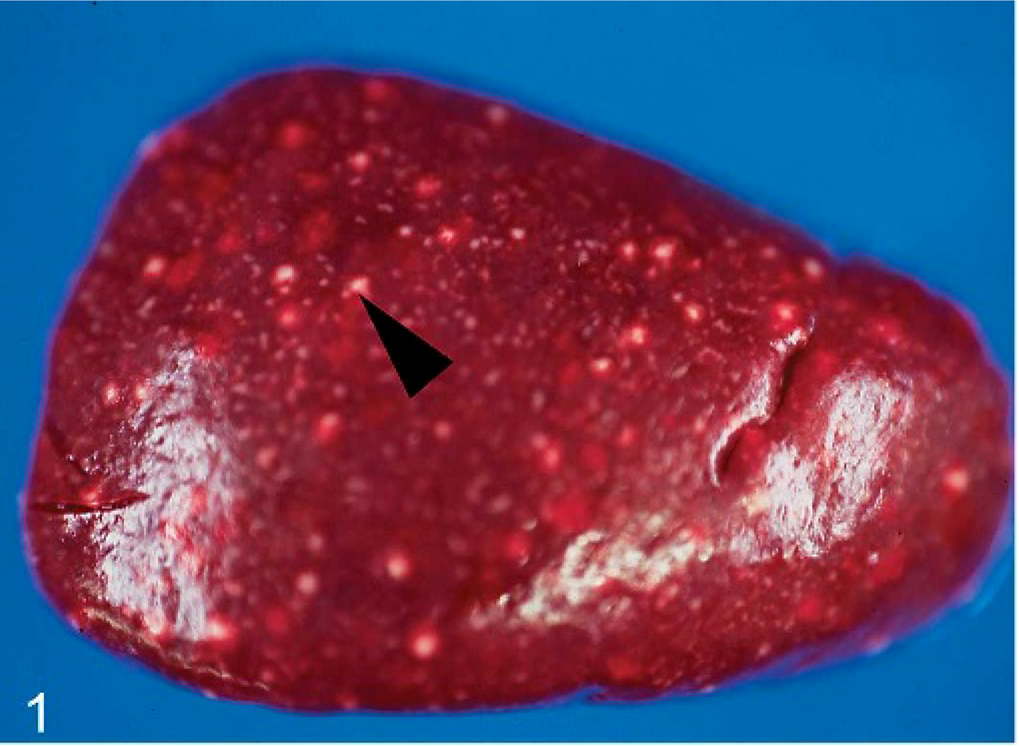
Spleen; cynomolgus monkey, monkey No. 2. Severe multifocal necrotizing splenitis has numerous necrotic lesions (arrow head) distributed throughout the parenchyma.
Histopathology
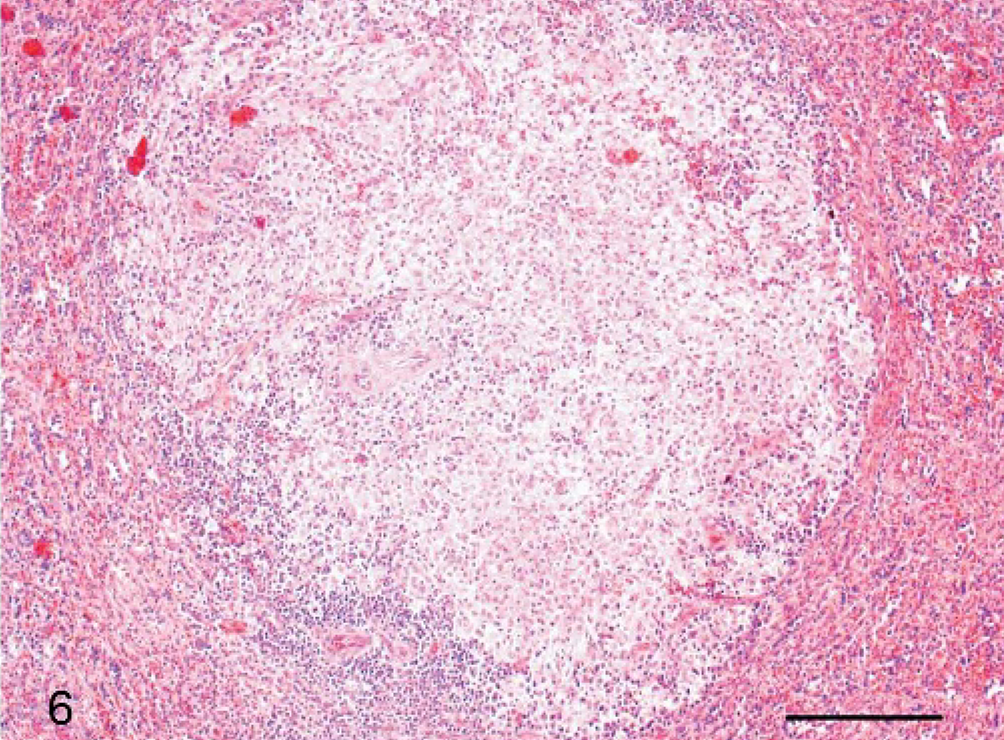
Histologically, the liver alterations were presented as moderate multifocal acute liver necrosis with regular or irregular outlines. The lesions consisted of amorphous cellular debris, necrosis of hepatocytes, and activated Kupffer cells and were infiltrated by some lymphocytes and neutrophils (Fig. 5). The spleens of all animals revealed a granulomatous inflammation in both white and red pulp (Fig. 6). The most common pulmonary histologic finding was a severe multifocal granulomatous pneumonia. Three of the 6 monkeys developed a severe multifocal granulomatous pneumonia, with characteristic granulomas diffusely distributed throughout the lung tissue but always found in close association with larger lung vessels (Fig. 7). The granulomas were sharply demarcated and consisted of central necrotic areas, hemorrhages, and mild infiltration of inflammatory cells (Fig. 7). They resembled the inflammatory reaction occurring in liver and spleen. Two of the 6 animals had severe multifocal pyogranulomatous tubulonephritis (monkeys Nos. 1 and 6) (Fig. 8).
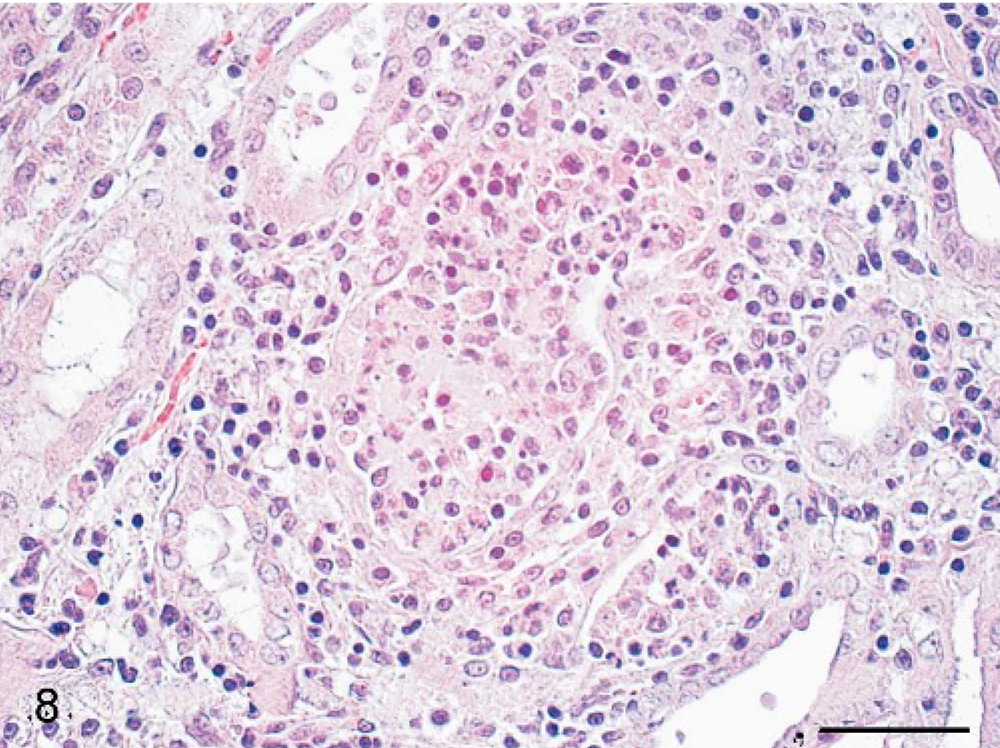
Kidney; cynomolgus monkey, monkey No. 1. Severe multifocal granulomatous tubulonephritis is seen. HE. Scale bar = 50 μm.
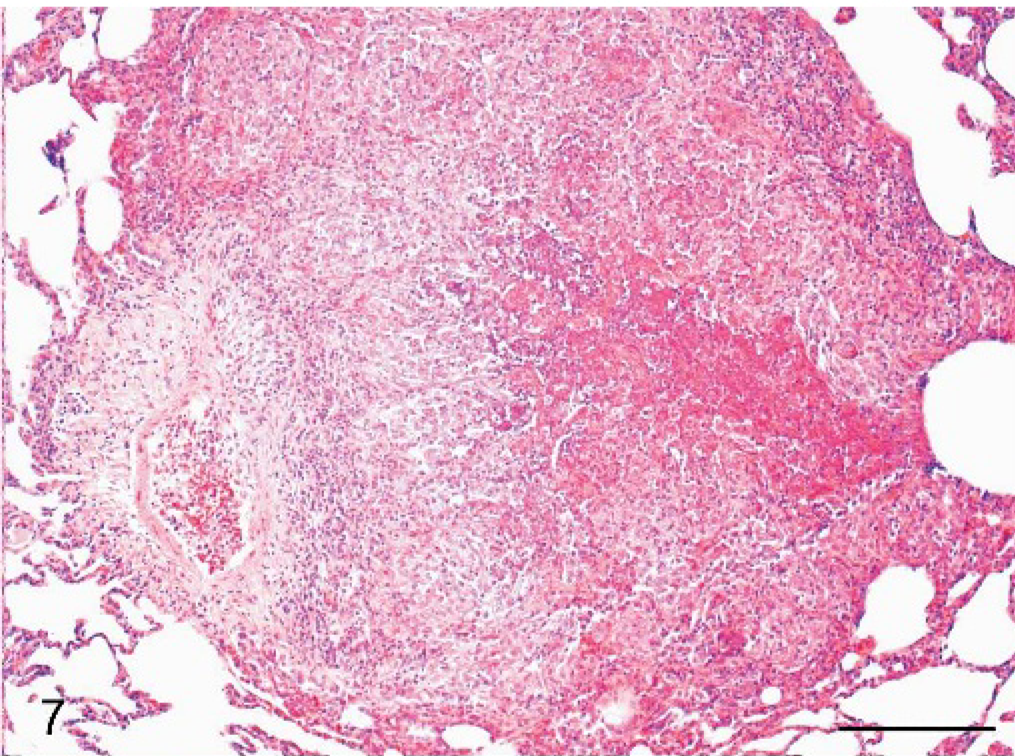
Lung; cynomolgus monkey, monkey No. 4. Sharply demarcated area of granulomatous lesions is within the lung parenchyma. HE. Scale bar = 200 μm.
Spleen; cynomolgus monkey, monkey No. 4. Granulomatous lesion is within the red and white spleen pulp. HE. Scale bar = 200 μm.
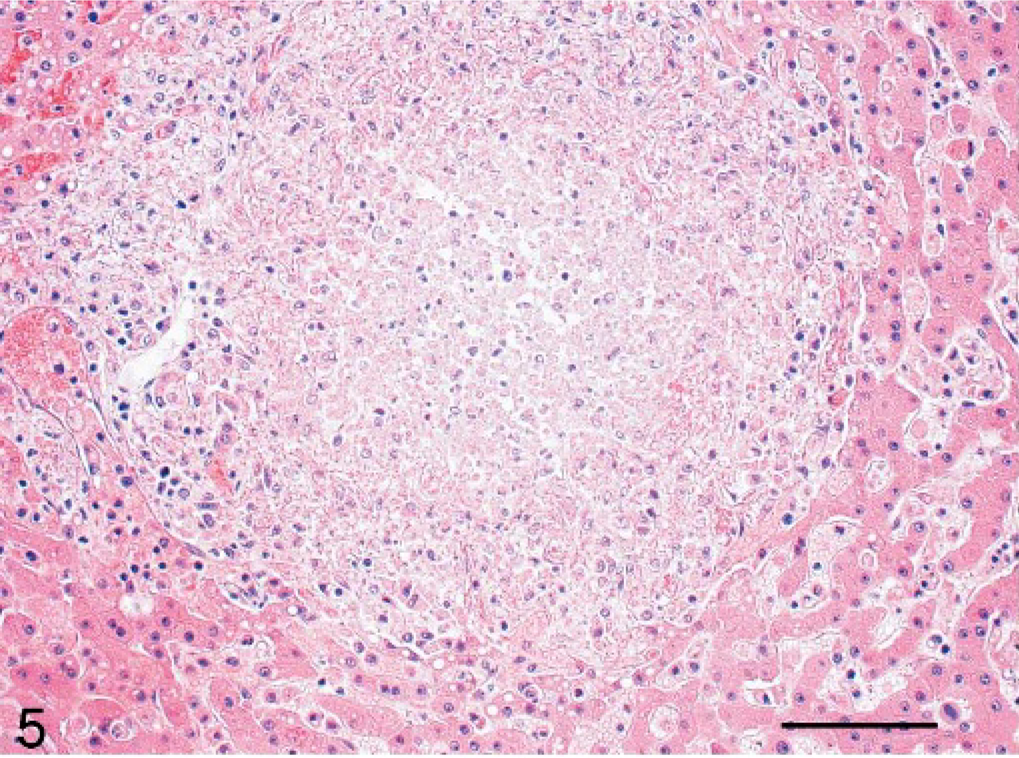
Liver; cynomolgus monkey, monkey No. 2. Sharply demarcated area of focal acute liver necrosis is seen. HE. Scale bar = 100 μm.
Pathohistologic findings of the lymph nodes were observed in all cases. The submandibular lymph nodes were grossly enlarged and showed signs of inflammation and necrosis. The lesions ranged from well-defined zones of acute inflammation and necrosis predominantly in the outer cortex to generalized necrosis that extended to the lymph node or developed into granulomatous inflammation.
More unusual histologic findings included suppurative leptomeningitis accompanied by granulomatous encephalitis (monkey No. 4) and necrotizing esophagitis (monkey No. 5), consisting of sharply demarcated areas of mucosal ulceration and necrotic debris overlying an ulcer area containing numerous neutrophilic cells and mixed bacterial overgrowth.
Special stains like Gram's, Giemsa, PAS-reaction, Grocott, and Ziehl-Neelsen failed to demonstrate microorganisms in any of the affected organs. The pathologic findings corresponded well to the histologic features in naturally acquired and experimentally induced tularemia in nonhuman primates. For this reason, immunohistochemistry was performed in all monkeys. Immunohistochemistry using a specific monoclonal anti-F. tularensis antibody confirmed the widespread distribution of Franciscella antigen within altered organ tissues (Figs. 9, 10).
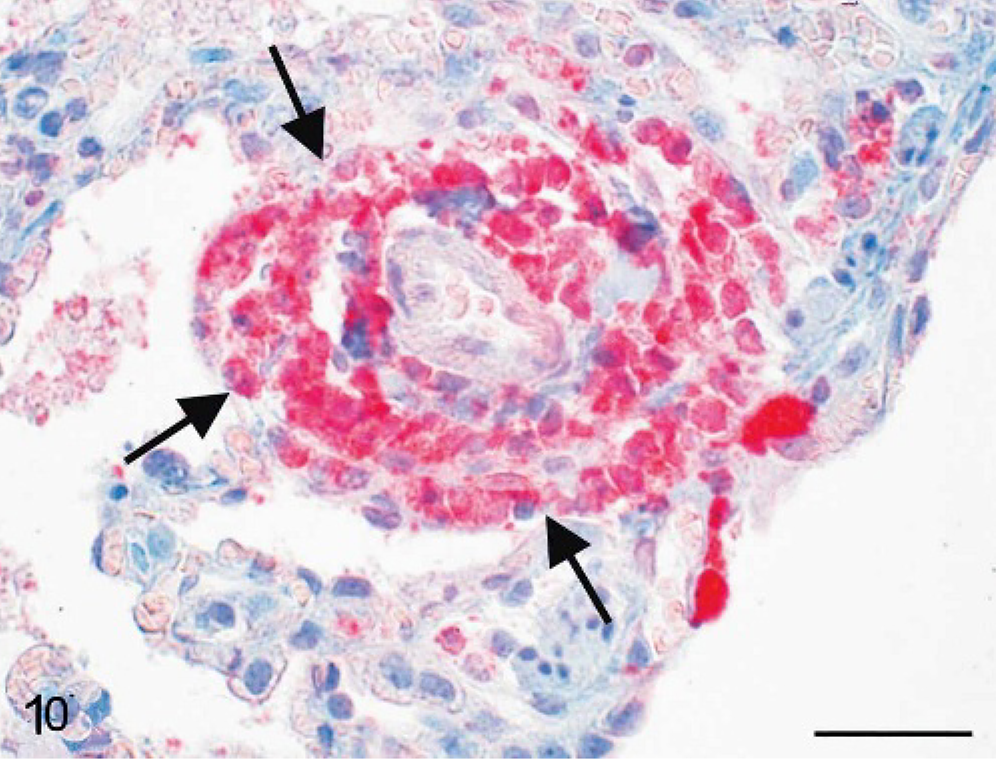
Lung; cynomolgus monkey, monkey No. 4. Francisella tularensis antigen is detectable within acute perivascular lung lesions (arrows). Immunohistochemistry, alkaline phosphatase anti-alkaline phosphatase method, monoclonal antibody clone 11/1/6, Mayer's hematoxylin counterstain. Scale bar = 50 μm.
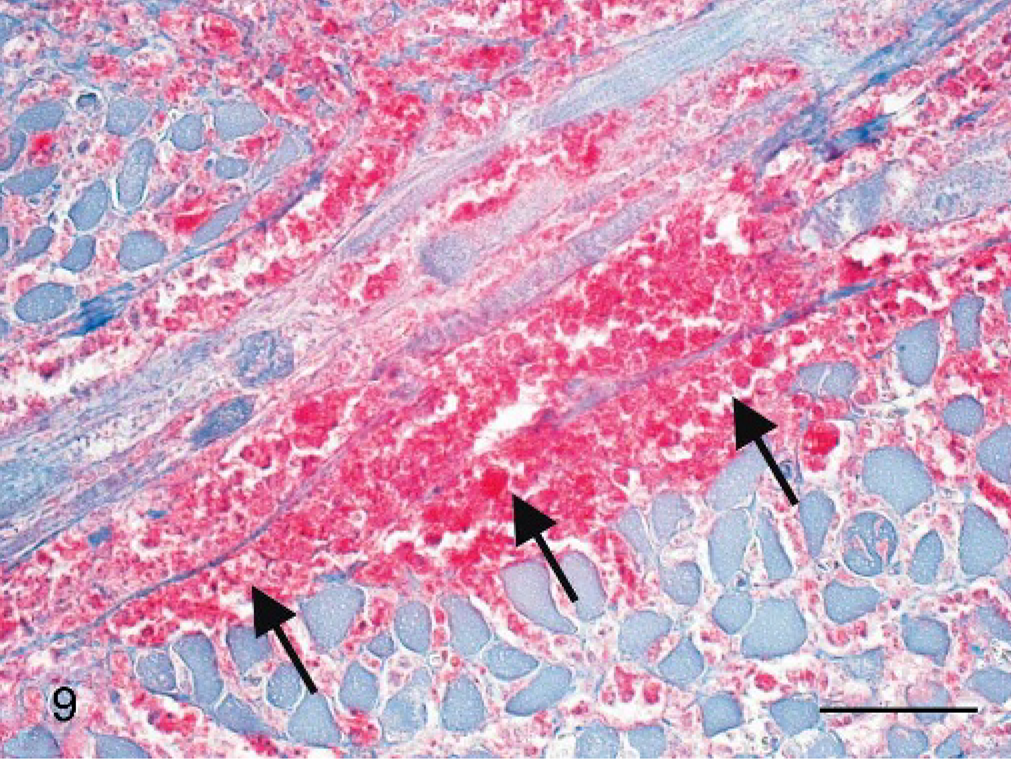
Pharynx; cynomolgus monkey, monkey No. 2. Francisella tularensis antigen is detectable within necrotic and granulomatous lesions between muscle bundles (arrows). Immunohistochemistry, alkaline phosphatase anti-alkaline phosphatase method, monoclonal antibody clone 11/1/6, Mayer's hematoxylin counterstain. Scale bar = 50 μm.
Interestingly, most bacterial antigen was distributed within altered oropharyngeal tissues (Fig. 9) or within regional lymph nodes. Most of the lesions within the spleen, the liver, and the lung were immunohistologically negative for Franciscella antigen. Only alterations within the peracute stage of disease contained bacterial antigen. Peracute perivascular lung lesions were most suitable for antigen detection (Fig. 10), whereas older granulomatous lesions showed no immunohistologic reaction.
Microbiology
Identification of the causative agent was achieved by culture, ELISA, and molecular techniques: In 4 out of 10 samples, F. tularensis LPS could be detected by antigen-capture ELISA, allowing the presumptive diagnosis of tularemia. Diagnosis in all animals was confirmed by real-time PCR, targeting the 16 SrDNA and tul4 genes.
Prolonged cultivation on cysteine heart agar revealed growth of F. tularensis from material of 4 out of 6 animals. Partial sequencing of the 16 SrRNA genes confirmed the presence of Francisella tularensis subsp. holarctica (Jellison type B).
Serology
Blood serum samples taken annually from all animals of the exposed group were investigated to estimate the possible risk of transmission and the epidemiologic spread within the group. Serologic diagnosis was performed by means of a competitive ELISA. Seroconversion occurred in 12 asymptomatic primates out of 35 cynomolgus monkeys tested.
Discussion
This is the first report of tularemia in a group of Old World monkeys in Germany. To our knowledge, this is the largest outbreak of tularemia in nonhuman primates affecting 18 animals in 1 group. Considering the very low prevalence of tularemia in animals, we believe that this finding may lead to the assumption that monkeys are highly susceptible to F. tularensis. There are 2 types of F. tularensis displaying large differences in virulence. Type A, F. tularensis tularensis, is the predominant form in the United States. The disease is lagomorph-associated, transmitted by the bite of an infected tick, and generally causes severe disease in humans. Type B, F. tularensis holartica, is primarily thought to be spread by rodents or water contamination and generally causes mild clinical disease in humans. In this epizootic of type B tularemia, a spectrum of different cases, ranging from asymptomatic seroconversion to severe illness with sudden death, was observed within 1 monkey group.
The pathologic findings in all cases corresponded to the macroscopic and histologic features in naturally acquired and experimentally induced tularemia in nonhuman primates.1,3,6,11,15,17 Most of the animals of the present epizootic developed alterations of the oropharyngeal mucosa. This location may be regarded as the portal of entry and primary manifestation site after ingestion of infected foodstuffs or of bacteria in tap water.
The cases illustrate that in addition to classic nodal and pulmonary involvement, tularemia regularly affects the liver and spleen and leads to hepatosplenomegalia and necrotic or granulomatous lesions within the target organs. The target organs in the cynomolgus monkey were the spleen, the liver, the lymph nodes, and the lungs. Characteristic lesions were foci of parenchymal cell necrosis, with macrophage activation and minimal response of other inflammatory cells.
A comparison with the findings of an experimental infection study showed that the caseous lesions within tonsils and lymph nodes closely resembled the lesions found in animals after 1 week of experimental infection.1 At this stage of infection, the nodular lesions of liver and lung took the appearance of granulomas.
The symptoms of tularemia are not pathognomonic, and the etiologic diagnosis requires the isolation of the causative agent or the detection of specific antigens or nucleotide sequences of F. tularensis.
Although tularemia is an uncommon disease in Germany, reservoirs of infection must exist in free-ranging animal populations. Type B F. tularensis is generally assumed to be associated with rodents. A murine tularemia epizootic was the most likely source of infection for the nonhuman primates. The source of infection of this epizootic is still under investigation. In an attempt to identify the source of infection, rodents were collected from the primate facility and the surrounding area. The indigenous rodent population appeared to be unusually high. Starting several weeks prior to the epizootic, many dead mice had been found in the facility. The most probable route of infection is assumed to be via ingestion of infected mice or ticks or through food contaminated by infected mice feces or urine. An alimentary route of infection is likely because main gross findings were located in the oropharyngeal tissue. Since cynomolgus monkeys are omnivores, eating insects and fruit, a possible source of infection could be infected insects like flies or ticks. However, mice are a potential vector for the disease. Monkeys catch and eat mice if they are kept in outdoor facilities. For this reason, appropriate steps to decrease possible contact between rodents, and primates and primate diets were implemented.
The cases illustrate the occurrence of zoonotic diseases in primate husbandries, where close contact with wild rodents or birds is common. Although this outbreak was caused by the less virulent subspecies of F. tularensis, 6 of 35 exposed animals died with signs of a subacute-to-chronic infection. Peracute tularemia characterized by disseminated intravascular coagulopathy and abortion was not observed during this outbreak. Interestingly, the age of the animals that died was generally 0.5–1 year, with only 1 adult affected, leading to the assumption that young animals are more sensitive to the disease than older animals who seroconverted.
Due to their close taxonomic relationship to man, nonhuman primates are of special concern as potential sources of zoonotic disease. Zoonotic infections are a constant hazard to both animal keepers and animal health personnel. The highly infectious nature of the organism involved demands strict precautions in handling nonhuman primates.
Footnotes
ACKNOWLEDGEMENTS
We thank Dr. Holger Scholz for his technical support regarding the 16 SrDNA sequencing. We are grateful to W. Henkel, K. Kaiser-Jarry, N. Knöchelmann, E. Lischka, and H. Zuri for their excellent technical assistance.
