Abstract
Protozoal meningoencephalitis is uncommon in raptors. An adult female bald eagle (Haliaeetus leucocephalus) was euthanized after several months of treatment for progressive neurologic signs. The predominant histologic lesion was lymphoplasmacytic and histiocytic meningoencephalitis involving the cerebrum and cerebellum. There was a marked segmental loss of granular cells and Purkinje cells, as well as segmental atrophy of the molecular layer in the cerebellum. Protozoal merozoites and schizonts were observed in the gray matter of the cerebellum. Ultrastructurally, the merozoites were classified as a species of Sarcocystis due to the lack of rhoptries. Immunohistochemistry of the agent revealed a positive reaction for Sarcocystis neurona, while sections were negative for Toxoplasma gondii and Neospora caninum. Sarcocystis sp. infection should be considered as a differential diagnosis in bald eagles with chronic neurologic disease.
An adult female bald eagle (Haliaeetus leucocephalus) was submitted to a veterinary clinic in the spring of 2000 with a fractured ulna related to a gunshot wound. The fracture was repaired surgically, but the bird was not releasable and was kept at a nature center in Missouri. The bird was kept in an aviary and fed a diet of poultry and rodents. Neurologic signs were first noted in early June 2004; the eagle leaned to the left, would lose balance, and developed limb weakness, repetitive head movements, and impaired vision. The eagle stayed on lower perches in the aviary. Whole-body radiographs and a complete blood count were within normal limits. A serum sample was negative for West Nile virus (WNV) nucleic acid by polymerase chain reaction (PCR a ). The eagle had received an intramuscular WNV vaccine b in 2002 and had received annual booster injections, most recently in May 2004. The intensity of the neurologic clinical signs was stable, but the bird's condition eventually deteriorated. Over the summer, the eagle became very unsteady with limited use of its right leg, and it held its left wing down for balance. The bird spent progressively less time on the perches and was eventually restricted to the ground. Multiple cutaneous ulcers were observed on the cranial left wing. In mid-September, bloodwork revealed a mildly elevated serum lead level (15 μg/dl; reference range, 0–10 μg/dl c ). Despite chelation therapy with calcium ethylenediamine tetraacetic acid, there was no improvement in clinical signs. Hematologic evaluation revealed evidence of elevated liver enzymes (aspartate aminotransferase = 887 IU/L) (reference range, 218 IU/L 2 [SD = 63]). A serum neutralization antibody test for WNV d was positive (1:40). Treatment during the neurologic episodes included anti-inflammatory agents (tepoxalin, dexamethasone, baby aspirin/acetylsalicylic acid, and celocoxib), antibiotics (enrofloxacin), vitamin B complex, and iron dextran. The bird was also treated with losartan potassium, an angiotensin II receptor antagonist that has been used off label with some anecdotal success in treating WNV infection in birds. The eagle had also received a number of homeopathic remedies, including belladonna, calcarea carbonica, natrum muriaticum, topical ointment (Calendula/Echinacea/Hypericum), and bioplasma (nutritional salt supplement). The clinical signs did not improve, and the bird was euthanized in October 2004.
At necropsy, the animal was in a fair nutritional state. Multiple, tan, fleshy, ulcerated, crusty nodules (0.5–2.5 cm in diameter) were present in the skin of the cranial left wing at the elbow, carpus, and interphalangeal joints. Approximately 25% of the pectoral muscle surface was mottled dark red and tan. Tissues were fixed in 10% neutral-buffered formalin, and the remaining unfixed carcass was submitted to the Minnesota Veterinary Diagnostic Laboratory. Sections of all fixed tissues were embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Selected sections were stained with Gram stain, Gomori methenamine silver, and acid-fast stain.
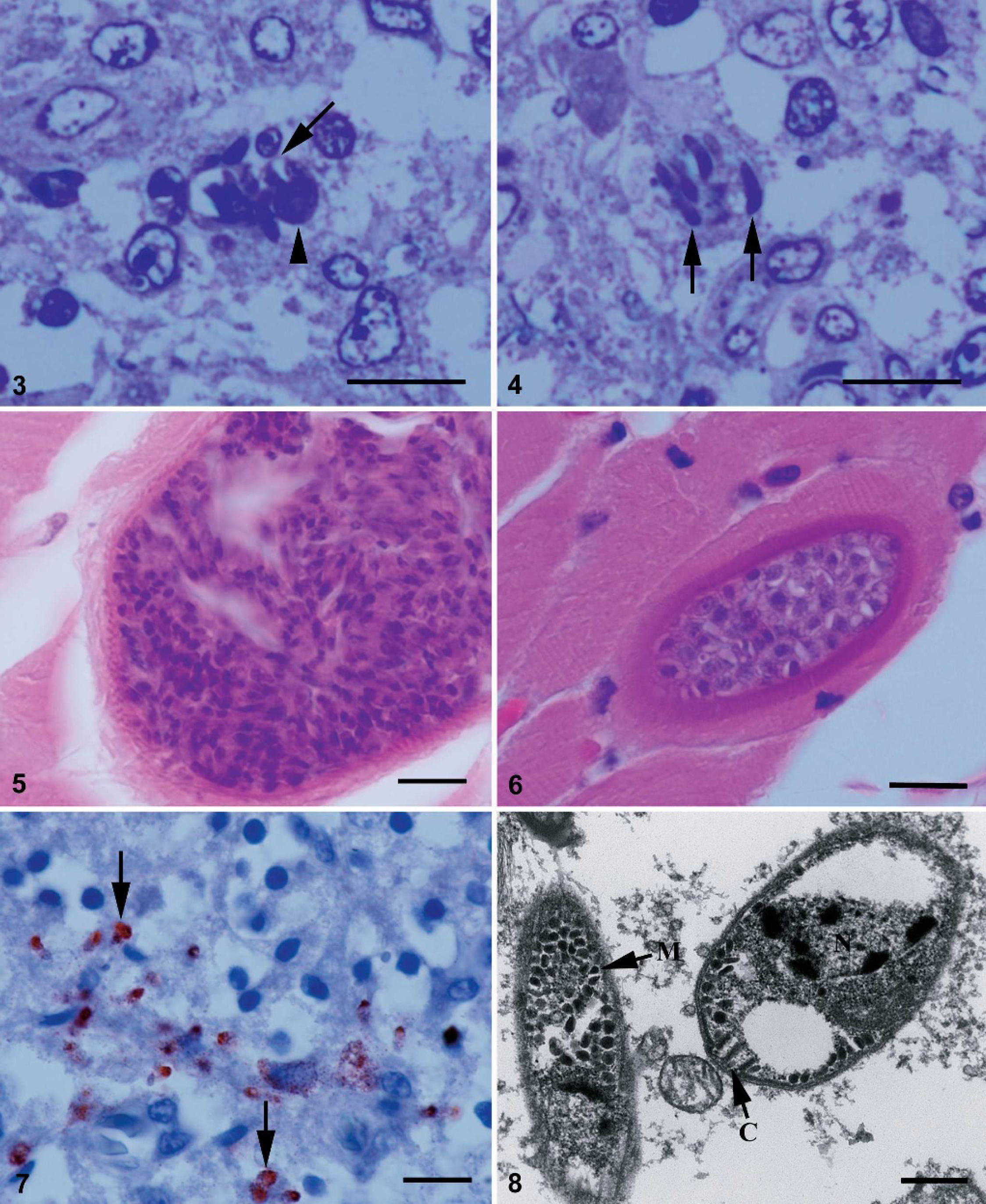
The main histologic lesion was in the central nervous system: moderate lymphoplasmacytic and histiocytic meningoencephalitis of the cerebrum and cerebellum. Multiple aggregates of lymphocytes and histiocytes expanded the meninges and were distributed randomly in both cerebral white and gray matter, in addition to numerous mononuclear vascular cuffs at small- to medium-caliber blood vessels. In the cerebellum, inflammatory infiltrates were scattered throughout nearly all folia. There was marked segmental loss of granular and Purkinje cells, segmental vacuolar change in the Purkinje cell layer, and atrophy of the molecular layer of multiple folia (Fig. 1). Protozoal organisms were scattered throughout the cerebellar parenchyma (Fig. 2); intracellular and extracellular merozoites and schizonts in 1-μm-thick sections stained with toluidine blue (Figs. 3, 4). Merozoites were 3 to 5 − 1 to 2 μm 2 with a centrally located vesicular nucleus. Schizonts were differentiated from host cells by the presence of large nucleoli. Merozoites budded from schizonts (Fig. 3) and were seen individually (Fig. 4). Glial nodules and lymphoplasmacytic perivascular infiltrates were present in the mesencephalon. The gray matter of the spinal cord was infiltrated multifocally by a few lymphocytes and plasma cells. In addition to the schizonts and merozoites observed in the sections of brain, 2 morphologic types of sarcocysts (types A and B) were found in ocular muscles. Type A sarcocysts were mature (approximately 30 μm in diameter) and contained tiny (5 − 1 μm) bradyzoites. Their sarcocyst wall was thin (< 1 μm) and contained small, spike-like villar protrusions (Fig. 5). Several type A sarcocysts were also observed in the myocardium. One immature type B sarcocyst containing only metrocytes was seen in a section of ocular muscle; the sarcocyst wall was 2 μm thick and appeared to be striated (Fig. 6). No inflammation was associated with encysted protozoa. There was a mild inflammatory infiltrate of lymphocytes and plasma cells in the pecten of the eye and a single focus of heterophilic myocarditis that did not appear to be associated with the Sarcocystis sp. Moderate heterophilic and ulcerative dermatitis at the left wing with abundant intralesional colonies of gram-positive cocci was present. Histologically, the pale streaks in the pectoral muscles corresponded to fibrosis, with myofibrillar atrophy and regeneration.
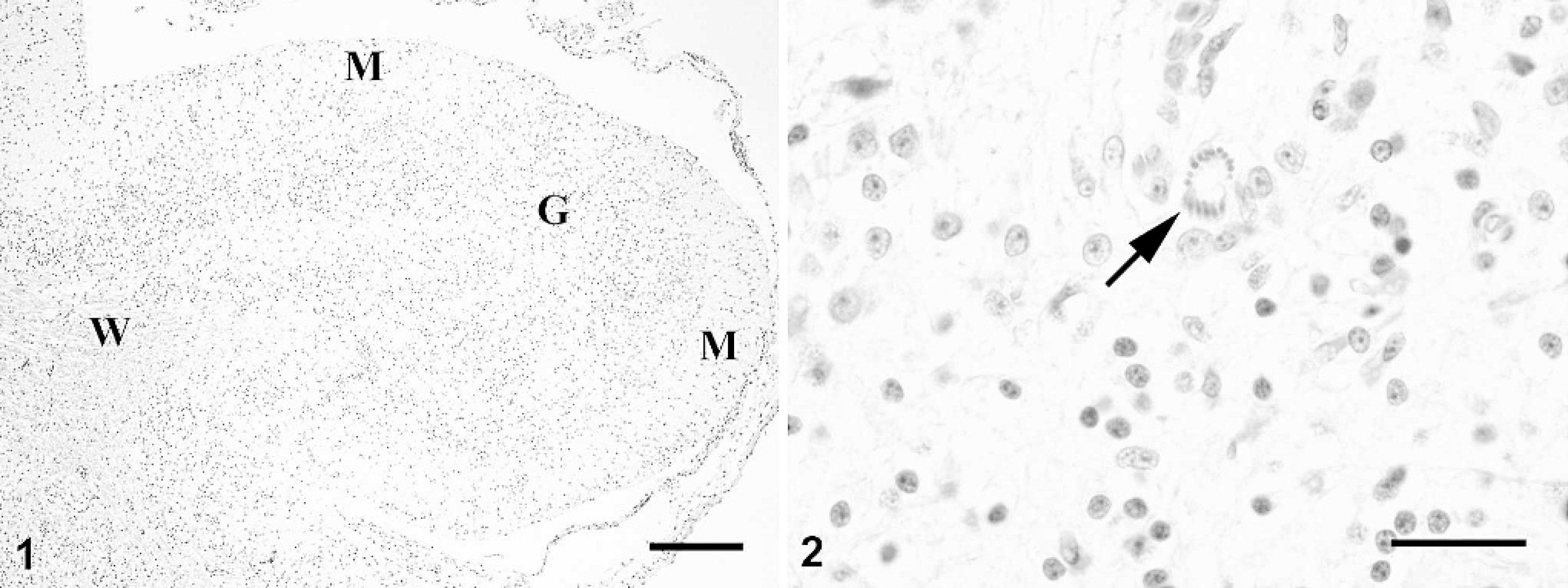
Cerebellum; bald eagle. The granular cell layer (G) of this cerebellar folium is severely depleted. The distinction between the cerebellar white matter (W) and gray matter is obscured. The molecular layer (M) is relatively unaffected. Hematoxylin and eosin. Bar = 200 μm.
Cerebellum; bald eagle. Protozoal schizont (arrow) in the cerebellar gray matter. Hematoxylin and eosin. Bar = 25 μm.
Selected sections of brain were subjected to immunohis-tochemistry (IHC) using antisera directed against Toxoplasma gondii antigen (rabbit polyclonal antibody, e 1:25 dilution), Neospora caninum antigen (goat polyclonal antibody/ 1:3000 dilution), and WNV envelope protein antigen (mouse monoclonal antibody, g 1:150 dilution). Formalin-fixed brain was submitted to the Animal Parasitic Diseases Laboratory, United States Department of Agriculture, Beltsville, Maryland, where IHC examination was repeated using T. gondii- and N. caninum-specific polyclonal rabbit antisera and antiserum to S. neurona as reported previously. 11,12
The organisms reacted with the S. neurona antiserum (Fig. 7). Neither T. gondii nor N. caninum antigen were detected in the cerebellar section by either laboratory. A structure resembling a WNV-positive dendrite in the molecular layer of the cerebellum was present in 1 of 4 examined brain sections; however, the reaction was considered to be equivocal. West Nile virus antigen was not detected in the cerebrum, spinal cord, eye, spleen, heart, skeletal muscle, ovary, adrenal gland, or spleen of the eagle.
A portion of embedded cerebellum was deparaffinized and processed for transmission electron microscopy. Ultrastructurally, protozoa were found in the neural parenchyma, and individual merozoites had a conoid, nucleus, and micronemes but no rhoptries (Fig. 8). The absence of rhoptries distinguishes Sarcocystis merozoites from tachyzoites of related protozoa.
Sarcocystis sp. nucleic acid-specific PCR using primers described previously failed to amplify the target DNA from the formalin-fixed brain tissue. 14 This may be attributed to the extended period of formalin fixation (>2 months) before PCR testing. Intestinal contents were negative for parasites by flotation analysis and direct examination. The hepatic lead concentration at necropsy was below the detection level (<1.0 ppm) using inductively coupled spectroscopy. The meningoencephalitis in this eagle was severe and likely caused by a Sarcocystis sp. infection based on structure and immunoreactivity of the parasite.
Sarcocystis sp. has a 2-host prey-predator life cycle. Carnivores are the definitive hosts for the parasite, and herbivores are the intermediate hosts. Carnivores become infected by ingesting herbivore tissue with cysts (sarcocysts). The parasite differentiates into male and female gamonts in the small intestine and produces oocysts (but the infection is not amplified). Oocysts sporulate in situ, and sporulated oocysts or sporocysts are passed in feces. Intermediate hosts become infected by ingesting sporocysts or oocysts. In the intermediate host, the parasite multiplies asexually (schizogony), generally in endothelial cells, and produces schizonts. Schizonts divide by endopolygeny, a process in which the nucleus divides into many nuclear lobes, and merozoites are released that eventually encyst in muscle sarcocysts. Sarcocystis in livestock is generally host specific for the intermediate hosts. 7 Although sarcocysts are common (90% of cattle are thought to be infected), they are rarely associated with clinical disease. 7 Acute clinical sarcocystosis is associated with schizogony, and it is rare to find schizonts in histologic sections of asymptomatic animals. It was not possible to determine the species of Sarcocystis in the bald eagle of the present report because there is no species-specific definitive test to diagnose sarcocystosis in birds. Until recently, most cases of acute sarcocystosis in birds were considered Sarcocystis falcatula infection; the bird is an intermediate host, and the North American opossum (Didelphis virginiana) is a definitive host. 3,4 Intermediate hosts for S. falcatula include cowbirds (Molothrus ater), sparrows (Passer domesticus), canaries (Serinus canarius), pigeons (Columba livia), and budgerigars (Melopsittacus undulalus).S. falcatula can be lethal to passerine species, particularly canaries and finches.

Cerebellum; bald eagle. Merozoites (arrow) budding from a residual body (arrowhead). Toluidine blue stain. Bar = 10 μm.

Cerebellum; bald eagle. Extracellular individual merozoites (arrows). Toluidine blue stain. Bar =10 μm.

Sarcocyst (type A) in ocular skeletal muscle of a bald eagle. This mature sarcocyst contains bradyzoites. The sarcocyst wall is thin and contains small, spike-like villar protrusions. Hematoxylin and eosin. Bar = 10 μm.

Sarcocyst (type B) in ocular skeletal muscle of a bald eagle. This immature sarcocyst contains only metrocytes. The sarcocyst wall is relatively thick and appears to be striated. Hematoxylin and eosin. Bar =10 μm.

Section of brain treated with polyclonal antiserum known to immunoreact with Sarcocystis neurona. The immunohistochemical reaction product is reddish-brown (arrows). Bar =10 μm.

Transmission electron micrograph of Sarcocystis sp. organisms in the brain. Note the conoid (C), nucleus (N), and micronemes (M) and the absence of rhoptries. The clear space adjacent to the nucleus (top right-hand corner) represents a shrinkage artifact. Bar = 500 nm.
The schizonts of the parasite in the bald eagle of the present report were distinct from schizonts of S. falcatula because the schizonts and merozoites were confined to the neural parenchyma. S. neurona is phylogenetically related to S. falcatula, and they have a common definitive host, the opossum, but S. neurona is not infective to passerines and S. falcatula is not infective to mammals. 9 S. neurona causes neurologic disease in many species of mammals, but there are no confirmed reports in birds. 8,13 There is some antigenic cross-reactivity by IHC between S. neurona and S. falcatula 11 and potentially other Sarcocystis sp. Polymerase chain reaction primers once considered species specific for S. neurona and S. falcatula are now known to react with other species of Sarcocystis transmitted by opossums. 6,10,14,16,21 Currently, avian sarcocystosis cannot be diagnosed definitively to the species level by DNA characterization using the available primers. 14
Muscular Sarcocystis sp. infection has been reported previously in 2 bald eagles from Alabama. 5,15 Crawley et al. found thin-walled sarcocysts in the heart and skeletal muscle. 5 The sarcocysts in the heart were immature and contained metrocytes, and septa were not seen. Sarcocysts in the skeletal muscle were mature with 2-μm-long bradyzoites. Lindsay and Blagburn found Sarcocystis-like bradyzoites in the muscle digest of another bald eagle. 15 Two morphologic types of sarcocysts occurred in the bald eagle of the present case report. The sarcocysts in the myocardium and skeletal muscle are likely an incidental finding. The 2 types of sarcocysts (A and B) observed in the myocardium and ocular muscles of the eagle may be developmental stages of the same protozoal species seen in the brain.
The species of Sarcocystis associated with encephalitis reported in other nonpasserine species, such as, golden eagle (Aquila chryaetos), 8 Northern goshawk (Accipiter gentiles atricapillus), 1 Northern gannet (Moms bossanus), 19 straw-necked ibis (Carphibis spinicollis), 12 wild turkey (Meleagris gallopavo), 22 and chickens (Gallus domesticus), 17 as well as the bald eagle described, remain to be determined. These reports originate from the Americas, and parasites were likely transmitted to the birds by consumption of feed or water contaminated with Sarcocystis sporocysts excreted by the opossum, D. virginiana.
The bald eagle described had been housed at a nature center at which approximately 1,000 opossums are received annually. Opossum fecal material may have been carried into the eagle exhibit accidentally by caretakers despite biosecurity measures. Rarely, opossum meat was fed to predators. Bald eagles did not receive opossum meat, but their food was prepared using the same cutting boards, providing a potential for the transmission of Sarcocystis organisms. Exposure of eagles housed in the aviary enclosure to wild opossums cannot be ruled out.
It is uncertain whether there was an underlying or superimposed WNV infection in the present case. Pectenitis and myocarditis are fairly common sequelae of WNV infection in the family Accipitridae, and antigen may be scant in raptors that die of WNV. 23 Bald eagles are susceptible to WNV infection and may die of the disease. 20 The eagle also had a positive neutralizing antibody titer for WNV (1:40), likely indicating exposure to the virus by natural infection rather than a vaccination titer. Repeated WNV vaccination failed to provoke seroconversion in red-tailed hawks. 18 The presence of a titer to WNV may not necessarily indicate that it caused disease in the bird. The dermatitis was likely due to repetitive traumatic injury to the wing; the bird used its wings to right itself, which resulted in injuries with secondary bacterial infection. The muscle fibrosis likely was due to recurrent intramuscular injections.
Despite being uncommon in raptors, neural sarcocystosis should be considered in the differential diagnosis for birds exhibiting neurologic clinical signs. In addition to conventional histology, molecular diagnostics, preferentially using fresh, unfixed material, may be helpful in the diagnosis of this disease.
Acknowledgements. We thank Jessica Mitchell, Shirley Casey, Andrea Schubert, and the staff at the Lakeside Nature Center (Kansas City, Missouri) for historical and technical assistance. We also thank Drs. Brock Exline (Hecker Animal Clinic, Kansas City, Missouri), Sandi Leonard (Peculiar, Missouri), and Julia Ponder (The Raptor Center, University of Minnesota, St. Paul, Minnesota) for clinical data. We are indebted to John Jenkins (Armed Forces Institute of Pathology, Washington, D.C.) for electron microscopy.
Footnotes
a.
Engene Biotechnologies, Inc., Carlsbad, CA.
b.
Fort Dodge, Overland Park, KS.
c.
Antech Diagnostics, Alsip, IL.
d.
Animal Health Diagnostic Laboratory, Cornell University, Ithaca, NY.
e.
Lab Vision Corp., Fremont, CA.
f.
Veterinary Medical Research and Development (VMRD), Inc., Pullman, WA.
g.
BioReliance Invitrogen Bioservices, Rockville, MD.
