Abstract
Melengestrol acetate (MGA), a potent synthetic progestin, has been used as a contraceptive in zoo felids since 1975. Mammary gland carcinomas have been linked to MGA treatment in zoo felids, but the histologic features of these tumors and steroid receptor expression have not been described. Zoo felid mammary tumors were requested from participating zoos from 1986 through 1998, and 31 mammary carcinomas from 28 MGA-treated and 3 untreated felids were received. The carcinomas were evaluated on the basis of histologic pattern, tumor grade, and occurrence of metastasis; then features of the tumors were compared to determine if carcinomas in MGA-treated felids differed from those that occur spontaneously. Estrogen- and progesterone-receptor expression was evaluated in 17 of the 31 carcinomas. Of the 31 tumors, 22 (70.9%) had multiple histologic patterns, 29 (93.5%) were high grade, and 28 (90.3%) had metastasized. Within tumors, the tubulopapillary pattern was most common (87.1%, n = 27); solid (61.3%, n = 19), cribriform (38.7%, n = 12), and comedone (25.8%, n = 8) patterns were less common; and the mucinous (3.2%, n = 1) pattern was rare. Both MGA-treated and untreated zoo felids had similar patterns and grades of mammary gland cancer as well as prevalence of metastasis. These results indicate that mammary carcinomas in zoo felids are high grade with a predominant tubulopapillary pattern and aggressive behavior. Five of 17 carcinomas expressed progesterone receptors, and 1 of 17 expressed estrogen receptors. Although more zoo felids with cancer had been exposed to MGA in this study, mammary carcinomas were similar in appearance and behavior in untreated and MGA-treated zoo felids. The association of MGA with the development of malignant mammary gland tumors should be considered when using this contraceptive in zoo felids.
Mammary gland tumors have been associated with the use of the synthetic progestins melengestrol acetate (MGA),12,18 megesterol acetate (MA),9,13,16,25,32 and medroxyprogesterone acetate (MPA)15,29 in both domestic and zoo felids. These drugs have been commonly used as a method of contraception,1,15,21,29,32,34 to treat dermatologic disease,9,16,29,32 and to modify behavior,21,32 although serious concerns regarding their side effects have been recognized. In domestic cats, MA and MPA treatment has been associated with fibroadenomatous mammary gland hyperplasia,9,13,16,24,32 while both benign and malignant tumors developed in domestic cats treated with MPA.15,29 In zoo felids, a close link between clinically aggressive mammary carcinomas and MGA treatment has been demonstrated.12 Studies in laboratory rodents have documented an increased potency, greater biologic activity, and increased tumorigenicity of MGA relative to other synthetic progestins and endogenous progesterone.10,23,31,40 Despite these concerns, MGA continues to be a common form of contraception in zoo felids.
Mammary growth is dependent on estrogens for simple ductular development and progesterone for complex ductular side-branching and lobuloalveolar growth.3,7,35,36 In most MGA-treated captive zoo felids, folliculogenesis and ovulation were not suppressed,23 despite MGA being a potent progestin. Therefore, mammary gland tissue in these cats was most likely simultaneously exposed to endogenous estrogen and progesterone, as well as a potent exogenous progestin.
The effects of estrogens and progestins on normal mammary gland depend on estrogen and progesterone receptors (ER and PR, respectively). Generally, estrogen up-regulates ER and subsequently up-regulates PR since PR expression is dependent on the presence of activated ER.6,41 Conversely, progesterone usually decreases both ER and PR,6 except in the mammary gland where persistence of PR is essential during pregnancy to delay lactogenesis.19 The presence or absence of ER and PR has been used for prognosis in animal and human mammary gland cancer.17 Lower ER and PR levels are associated with significantly decreased survival rates relative to higher receptor levels in canine mammary gland carcinomas.27 Low ER and PR content is associated with poor response to hormone therapy, decreased tumor-free intervals and survival, and increased tumor grade in humans.2,17,38,39 In domestic cats, lower numbers of ER or PR have been reported in malignant mammary tumors than in benign tumors,22,27,33 and in 1 study, PRs were present in a group of ER-negative feline mammary gland carcinomas. How MGA affects ER and PR in zoo felid mammary glands, whether steroid hormone receptors are expressed in mammary carcinomas from MGA-treated zoo felids, and whether receptor expression correlates with tumor behavior have not been investigated.
In a previous report, the American Zoo and Aquarium Association (AZA) Contraceptive Advisory Group (CAG) health surveillance program disclosed an association between MGA contraceptives and mammary cancer in zoo felids.12 Because the hormonal milieu in MGA-treated zoo felids differs from that in untreated felids, tumors in MGA-treated felids may differ histologically and behaviorally from those that develop spontaneously in cycling felids. The current study characterizes the histologic features and grades of all carcinomas received between 1986 and 1998 and compares these traits between MGA-treated and untreated zoo felids. This study also evaluates ER and PR expression in these cancers to determine whether expression correlates with biologic behavior.
Materials and Methods
Study animals
Mammary gland tumors from zoo felids were requested from participating zoos from 1986 through 1998 as part of the AZA CAG health surveillance program. Tumors were requested regardless of historic exposure of the felids to progestin contraceptives. Cases included in the study were limited to zoo felids with a malignant mammary gland tumor from which necropsy or biopsy tissue was available for review and for whom information on age, parity, contraceptive history, and presence of metastases was available. During this 12-year period, 45 mammary gland cancers from zoo felids were received. Thirty-one of the 45 felids (68.9%) met the inclusion criteria, including 28 of 31 MGA-treated (90.3%) and 3 of 31 untreated (9.7%) zoo felids. All 31 zoo felids included in the study were female.
The age range for MGA-treated zoo felids was 98–288 months (median age = 174 months; mean age = 171.5 ± 38.3 months) and, for untreated zoo felids, was 109–240 months (median age = 188 months; mean age = 179.0 ± 66.0 months). There was no significant difference in age between untreated and MGA-treated populations (P = .76). Five species from 25 institutions were represented in the population (Table 2), including 19 tigers (Panthera tigris), 5 jaguars (Panthera onca), 4 lions (Panthera leo), 2 jungle cats (Felis chaus), and a fishing cat (Felis viverrinus). The average length of MGA-treatment in zoo felids was 80.9 ± 35.7 months (range = 12–149 months). Parity information was available for 2 untreated and 24 MGA-treated zoo felids. One of 2 untreated zoo felids was parous prior to tumor development, and 14 (58.3%) MGA-treated zoo felids were parous prior to MGA treatment.
Criteria for determining individual morphologic scores for mammary gland carcinomas in zoo felids. Individual scores are subsequently used to determine overall tumor scores and tumor grade.

Morphologic patterns, overall tumor score, and tumor grade for mammary gland carcinomas in zoo felids.
MGA = melengestrol acetate; − = untreated; + = MGA-treated; C = comedone; CR = cribriform; M = mucinous; S = solid; TP = tubulopapillary.
Tumor patterns listed in each row are in order of prevalence.
Tumor morphology, tumor grade, and metastasis
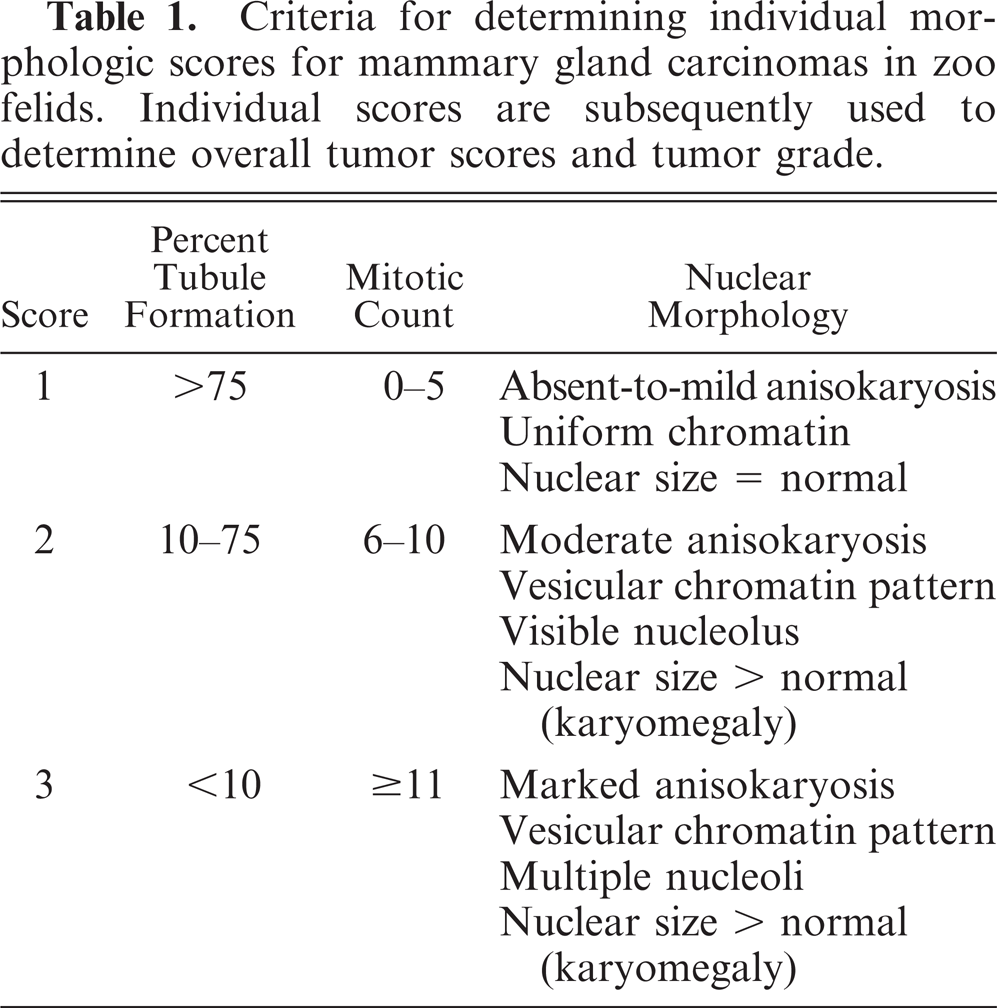
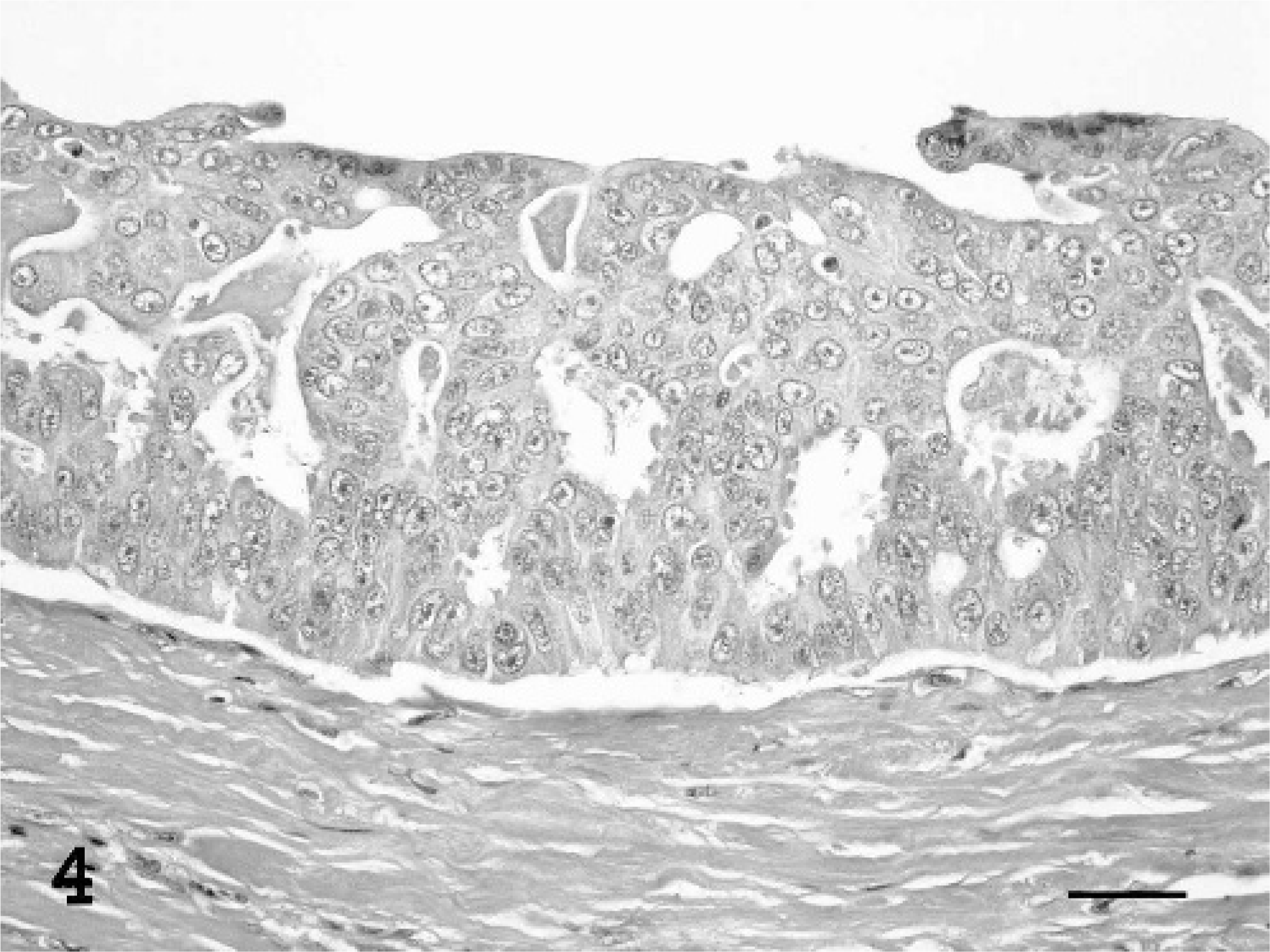
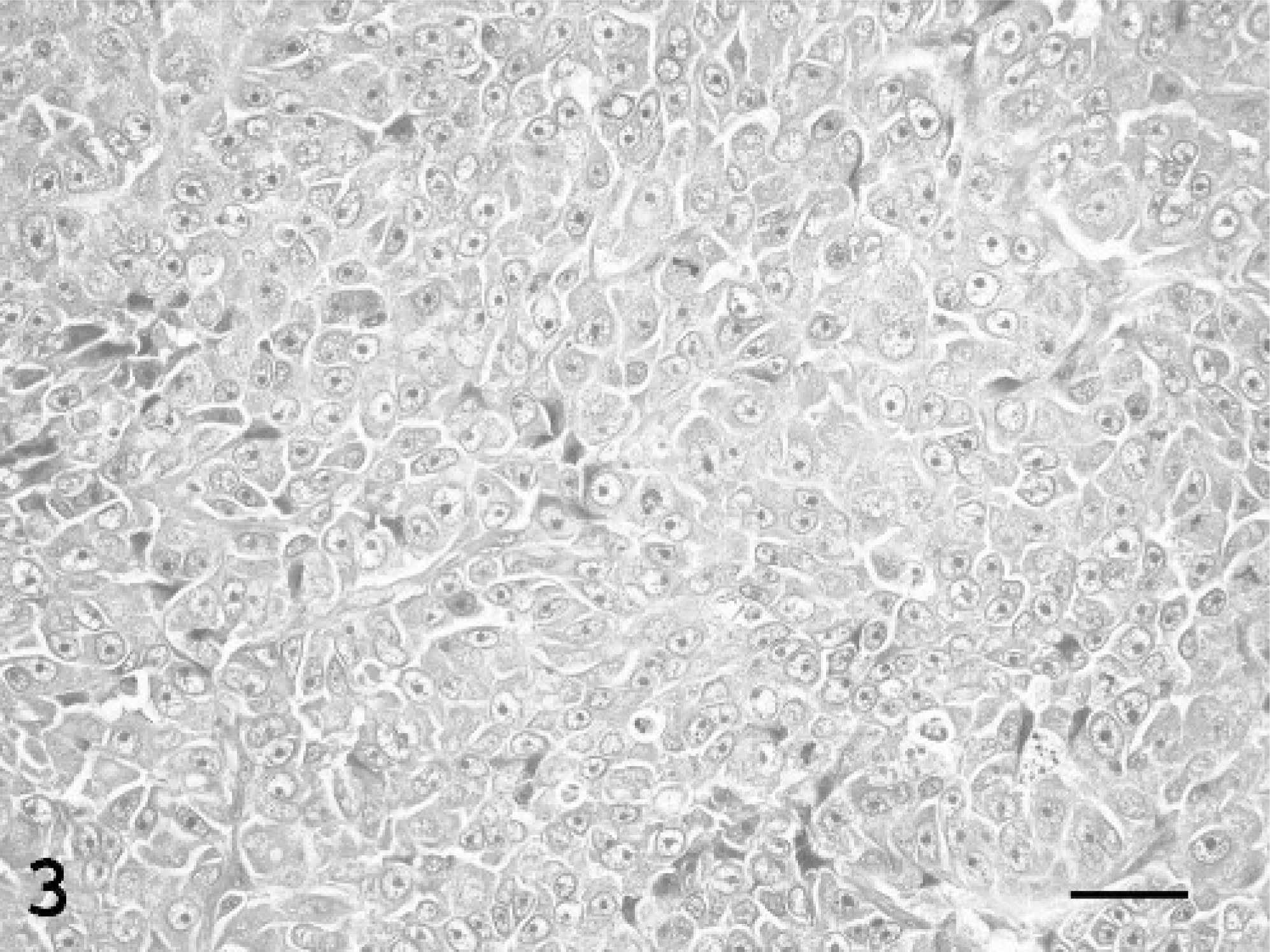
Histologic patterns were classified according to the World Health Organization criteria for mammary tumors of dogs and cats.30 Carcinomas were classified as tubulopapillary, solid, cribriform, or mucinous. Tubulopapillary carcinomas were characterized by the formation of varying sized tubules with (Fig. 1) or without papillary (Fig. 2) projections into the lumen. Carcinomas in which papillary fronds projected into cystic intralobular or lobar ducts were subclassified as papillary-cystic variants of tubulopapillary carcinomas. In solid carcinomas, tumor cells were arranged in solid sheets, cords, or nests (Fig. 3), with rare or no tubule formation and very little supporting stroma. Comedone carcinomas, a subclass of solid carcinoma, were composed of solid nodular tumor foci with central necrosis. Cribriform carcinomas were composed of anastomosing ribbons or cords of proliferating cells lining ductlike structures, resulting in the formation of multiple, small fenestrations creating a “sievelike” pattern (Fig. 4). Mucinous carcinomas consisted of individual or small clusters of neoplastic cells within a mucinous matrix.
Mammary gland/cribriform carcinoma; tiger, animal No. 17. The characteristic feature is the formation of cellular arcades or small, uniform fenestrations, creating a “sievelike” pattern. HE. Bar = 30 μm.
Mammary gland/solid carcinoma; jaguar, animal No. 25. Solid sheets of neoplastic epithelial cells are supported by scant stroma. HE. Bar = 30 μm.
Mammary gland/tubular carcinoma; fishing cat, animal No. 3. Round to irregularly shaped tubules of varying size and shape are lined by a single layer of neoplastic epithelium. HE. Bar = 30 μm.
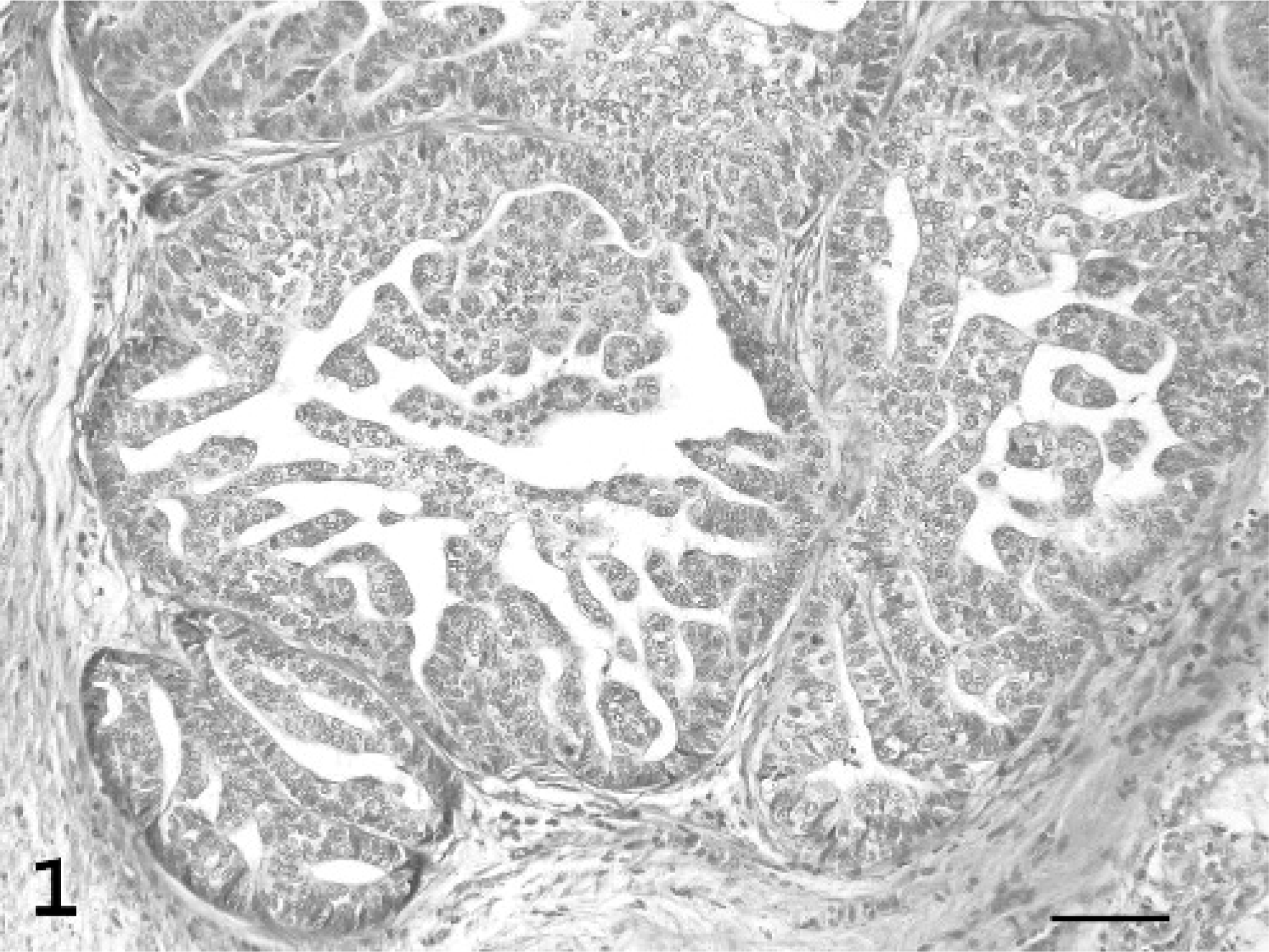
Mammary gland/tubulopapillary carcinoma; tiger, animal No. 7. Numerous, short papillary and tubular projections extend into the lumen from the duct wall. HE. Bar = 60 μm.
A single, representative, histologic section from each tumor was graded according to the method of Elston and Ellis,11,30 a grading scheme, originally developed in humans, that has been applied to felid mammary gland cancer.5,28,42 Percentage of tubule formation, mitotic count, and nuclear morphology were each assigned an individual score from 1 to 3 on the basis of the criteria listed in Table 1. Tubule formation score was based on the percentage (0–100%) of tumor tissue from the representative section in which clear tubule formation was present. It was defined as a circumferential arrangement of single cells around a central lumen. Mitotic count was based on the number of mitoses in 10 random 40× fields (field diameter = 0.45 mm; field area = 0.15 mm2) of tumor tissue.11 Mitoses were counted in random fields rather than at tumor margins11 because natural tumor margins were not available in all cases. Nuclear morphology score was based on subjective assessment of anisokaryosis and objective determination of karyomegaly. The latter was determined using an ocular micrometer. The diameter of 100 epithelial cell nuclei was measured in 1 normal tiger mammary gland and all tumors. The mean nuclear diameter size in each tumor was then compared with the mean nuclear diameter of normal tiger mammary gland epithelial cells using a 2-sample t-test. Tumors in which a difference in mean nuclear size relative to normal nuclear size was detected were categorized as having karyomegaly.
An overall tumor score (OTS) ranging from 3 to 9 was then calculated as the sum of the 3 individual scores (Table 2). On the basis of the OTS, each carcinoma was categorized as well differentiated, grade I (OTS = 3–5); moderately differentiated, grade II (OTS 6–7); or poorly differentiated, grade III (OTS = 8–9).
Immunohistochemistry
ER and PR immunohistochemistry was performed on 17 of the 31 zoo felids from whom formalin-fixed, paraffin-embedded tissue was available. Because receptor expression is regulated by the current hormonal environment, zoo felids were grouped as to whether they had never been treated with MGA (control, n = 2), were treated with MGA but not within 30 days of necropsy or biopsy (previous MGA, n = 10), or were being treated with MGA at the time of necropsy or biopsy (current MGA, n = 5). Routine immunohistochemical procedures were performed using a streptavidin biotin detection system (Biocare Medical, Walnut Creek, CA). To facilitate unmasking of receptor antigens, tissues were steam heated to 98°C in an ethylenediaminetetracetic acid buffer solution, pH 8 (LabVision Corp., Fremont, CA). Endogenous peroxidase activity was blocked by immersion in a 0.3% hydrogen peroxide/methanol solution (Sigma-Aldrich, St. Louis, MO) for 30 minutes, and endogenous proteins were blocked with a serum-free protein block for 10 minutes (Dako Corp., Carpinteria, CA). Tissues were then incubated with mouse monoclonal anti-ER (1:500, clone 1D5; Immunotech Inc., Westbrook, ME) or mouse monoclonal anti-PR (1:75, clone PR10A9; Immunotech Inc.) for 3 hours in a humidified chamber at room temperature. Antibody binding was labeled by incubating the tissues in commercially available biotinylated goat antimouse immunoglobulin G (IgG) (Biocare Medical). Slides were washed, streptavidin horseradish-peroxidase solution (Biocare Medical) was applied, labeled complexes were visualized using a 3-amino-9-ethylcarbazole kit (AEC kit, Zymed Laboratories, Inc., San Francisco, CA), and slides were counterstained with Mayer's hematoxylin (Sigma-Aldrich, Inc.). Feline uterus, oviduct, and normal mammary gland were used as positive controls. Negative controls were run concurrently using the same technique with the substitution of a mouse myeloma IgG1 isotype (Zymed Laboratories, Inc.) for the primary antibody.
Analyses
Cases were limited to the 12-year period when all available cases were requested from participating zoos. Because of the limited number of untreated animals, the number of felids in treated and untreated groups differed and a nonparametric test was used to account for the sample-size difference. Tumor grade (based on overall tumor score) was, therefore, compared between MGA-treated and untreated zoo felids, using a Wilcoxon-Mann-Whitney U Test, correcting for tied ranks.37
Results
The majority (70.9%) of mammary carcinomas had more than 1 histologic pattern (Table 2). Overall, the tubulopapillary pattern was most common (87.1%) followed by solid (61.3%), cribriform (38.7%), and comedone (25.8%) patterns. Mucinous (3.2%) patterns were rare. Patterns in MGA-treated animals were similar to those in untreated animals, with multiple morphologic patterns occurring in tumors from 2 of 3 untreated and 20 of 28 MGA-treated zoo felids (Table 2). The tubulopapillary pattern was present in tumors from all 3 untreated and 24 (86%) MGA-treated zoo felids. Cribriform (n = 2) and solid (n = 1) patterns occurred in carcinomas from the 3 untreated zoo felids. In the 28 MGA-treated felids, in addition to the prevalent tubulopapillary pattern, the solid pattern was common (64.3%); cribriform (35.7%) and comedone (28.6%) patterns were less common; and the mucinous (3.6%) pattern was rare.
Many tumors (58%) contained foci of inflammation. Inflammation was present in tumors from 2 of 3 untreated zoo felids and was predominantly neutrophilic in both animals. Tumors from 16 MGA-treated zoo felids contained inflammation. In these animals, inflammation was mixed, containing variable numbers of lymphocytes, plasma cells, and neutrophils.
Of the 31 tumors received during the study period that met the inclusion criteria, 28 (90.3%) were from zoo felids exposed to MGA. In affected felids, the majority (93.5%) of tumors were high grade and moderately or poorly differentiated, regardless of whether the animal had been exposed to MGA (Table 2). Moderately to poorly differentiated tumors occurred in all untreated and most (92.9%) MGA-treated zoo felids. A difference of overall tumor score between treated and untreated felids was not detected (P = .41); however, the small sample size limited the ability to detect a difference. The presence of metastasis also did not differ between groups, as all control and MGA-treated zoo felids for which metastasis information was available (n = 28) had evidence of metastasis at the time of tissue collection.
Five of 17 carcinomas were positive for PR by immunohistochemical staining, of which 3 were being treated with MGA at the time of biopsy or necropsy, 1 was previously treated, and 1 was a control. The carcinoma in 1 zoo felid being treated with MGA at the time of tissue collection was also positive for ER. The other 16 felids were negative for ER.
Discussion
This study demonstrated that mammary carcinomas in zoo felids have diverse histologic patterns and are predominantly of moderate-to-high grade. Despite the potency of MGA relative to endogenous progesterone,10,23,31,40 carcinomas arising in the MGA-treated felids have patterns, grades, and prevalence of metastases similar to those of spontaneous tumors. Although tigers may appear over-represented in this study population, MGA contraceptives have been used more extensively in this species over the last 2 decades than in other zoo felids (AZA CAG database). So the large number of affected tigers is likely an outcome of exposure rather than species predilection.
Multiple histologic patterns were seen in the majority of mammary carcinomas in both untreated and MGA-treated zoo felids in this study, although the tubulopapillary pattern was most common regardless of MGA treatment. Comparing patterns in mammary carcinomas of zoo felids with those mammary carcinomas arising spontaneously or in progestin-treated domestic cats was not possible because mammary cancer in domestic cats was usually designated by the predominant pattern, and whether multiple patterns occurred was not generally reported.5,8,13,14,26,28,33,42 Furthermore, tumor classification of mammary gland carcinomas in domestic cats has been inconsistently applied over the years, with tumors often either grouped as adenocarcinomas or subclassified by other patterns.
Most zoo felids in this study had high-grade mammary cancer based on tumor grading using the method of Elston and Ellis,11 and most had metastatic disease at the time of diagnosis, regardless of their history of progestin exposure. Mammary carcinomas in domestic cats have comparable aggressive features, high tumor grades (II or III), and evidence of metastasis at the time of diagnosis.5,28 These features are associated with decreased survival in domestic cat5,20,28 and human2,4,38,39 mammary cancer patients and are likely also important prognostic indicators in zoo felids. In domestic cats, the advanced status of these cancers at the time of diagnosis may be due in part to delayed owner observation.5,42 Failure of early detection is also a likely factor in zoo felids because diagnostic procedures such as physical examination and radiography are only performed opportunistically or at the yearly examination under anesthesia.12 However, it also remains possible that mammary cancers of high grade with high metastatic potential develop early in the course of carcinogenesis in both domestic and zoo felids. Although no differences were seen between untreated and MGA-treated zoo felids, untreated felids were under-represented in this study. The small number of submissions from untreated animals in this study likely reflects a low prevalence of mammary cancers in this group as a whole because submission was not biased by MGA exposure in this large, well-established health surveillance program.
The low proportion of mammary cancers with either PR- or ER-positive staining in zoo felids is similar to findings in domestic cat mammary carcinomas, which have fewer ER and PR receptors than normal mammary gland or benign mammary tumors.26,27,33 PR status does not appear to correspond to prognosis in zoo felids, as both PR-positive and -negative cancers were of high grade with metastasis.
In conclusion, mammary carcinomas in MGA-treated zoo felids were similar to those in untreated zoo felids and were morphologically complex with high-grade and frequent metastases. These histologic features corresponded to their aggressive clinical behavior noted previously.12 Although the tumors arising in MGA-treated zoo felids were not unique, their occurrence in animals receiving contraception appeared to be increased relative to untreated felids in this study, and this association should be considered when the benefits of contraception are being weighed against potential adverse effects.
Footnotes
Acknowledgements
This project was supported in part by the American Zoo and Aquarium Association Conservation Endowment Fund/Ralston Purina Big Cat Survival Funds and The Geraldine R. Dodge Foundation. The following zoological societies and veterinarians contributed mammary gland tissue to this study: Akron Zoological Park (G. Riggs), Baltimore Zoo (M. Cranfield), Baton Rouge Zoo (G. Pirie), Brandywine Zoo (L. Klein), Wildlife Conservation Society (T. McNamara), Buffalo Zoological Gardens (A. Prowton), Chaffee Zoological Gardens of Fresno (R. Gentzler, S. Lynch), Dallas Zoo (B. Raphael), Denver Zoo (D. Kenny), Glen Oaks Zoo (J. Peddie), Kansas City Zoo (W. Suedmeyer), Knoxville Zoological Gardens (P. Morris, E. Ramsey, D. Gillespie), Little Rock Zoo (M. Baeyens), Los Angeles Zoo (C. Stringfield), Memphis Zoo (E. Douglass), Reid Park Zoo (T. Miller), Riverbanks Zoological Park (B. Baker), Sacramento Zoo (N. Lamberski), San Antonio Zoo (M. Richardson), St. Louis Zoological Park (R. Junge), Sunset Zoo (J. Carpenter), and Toledo Zoo (T. Reichard). We thank Dr. D. Joly for his contributions to the statistical analysis.
