Abstract
The immunohistochemically determined estrogen receptor (ER) α (ERα) and progesterone receptor (PR) status, as well as recognized, well-accepted prognostic indicators and host factors were prospectively analyzed in 84 cases of primary canine mammary carcinoma for their effect on disease-free period (recurrence free, metastasis free, or combined) (DFP) after an observation period of 18 months. The presence of one or both receptors, as well as tumor size, lymph node status, histologic grading, intravascular growth, and necrosis, were of prognostic value for DFP. In multivariate analysis, only tumor size and histologic grading proved to be independent prognosticators. None of the host factors analyzed were of prognostic value for DFP. ERα, PR, or both were detected in 173 out of 228 tumors: 70 ERα and PR; 5 ERα only; 98 PR only. Statistically significant differences regarding the presence of one or both receptors were observed between benign and malignant tumors and between complex, mixed, and simple histologic subtypes of benign and malignant tumors. In the group of malignant tumors (
Mammary tumors are the most common type of tumors in intact female dogs. According to the histologic diagnosis, between 41 and 53% of the mammary tumors that occur in the bitch are considered malignant. 34, 50 However, histologic evidence of malignancy does not invariably imply a malignant clinical course. 50 Therefore, reliable prognostic factors are of great importance for estimating the individual risk of unfavorable clinical outcome. There are some recognized, well-accepted prognostic factors of malignant mammary tumors of the dog, such as tumor size, lymph node status, distant metastasis, histologic type, histologic malignancy grade, and degree of nuclear differentiation. 34, 50 There are some other proven or controversial host and tumor prognostic factors, and the number of new ones is steadily increasing. 26, 51, 62 However, the clinical experience is still limited, and reliable results of prospective studies are not always available. Prognostic power, feasibility, economy, reproducibility, and, if possible, executability without highly sophisticated technical equipment should determine the introduction of new prognostic factors into clinical routine.
Steroid receptors have proven their usefulness in characterizing subgroups with different prognosis among female dogs with mammary cancer. 25, 31, 39, 42, 52 However, the majority of available data are based on the measurement of steroid receptors using tumor cytosols. 25, 31, 42, 52 Because these techniques require special conditions of storage and assay of tissue samples and are expensive, their use has not been introduced in clinical practice. With the development of monoclonal antibodies against nuclear estrogen and progestin receptors, 17, 45 the immunohistochemical (IHC) measurement has become increasingly important. Among other advantages over tumor cytosol–based assays, IHC methods of study of estrogen and progestin receptors work on routine, formalin-fixed, paraffin-embedded tissue, 14, 16, 23, 24, 39, 56 but the literature gives only scarce pertinent data on the prognostic relevance. 39 We therefore investigated the predictiveness for recurrence and metastasis of the immunohistochemically determined estrogen receptor (ER) α (ERα) and progesterone receptor (PR) in a series of 84 bitches with malignant mammary tumors within a framework of approved prognosticators. Univariate and multivariate statistical studies were used. In addition, the relationship between hormone receptor expression and host and tumor factors was also studied using all 228 tumors harvested from the female dogs of our series.
Materials and Methods
Animals
Samples were obtained by excisional biopsy (mastectomy or nodulectomy) from 100 female dogs admitted to veterinary clinics for clinical evaluation and treatment of mammary tumors. These animals had not undergone chemotherapy or radiotherapy, had one malignant tumor, at least, diagnosed by histopathology, and were clinically and radiologically free from distant metastases. Twenty-six dogs had enlarged and excised regional lymph nodes. Data retrieved from the owners included age, ovariectomy status, contraceptive use, parity, and pseudopregnancy. Data collected on physical examination were number of tumors, chain location, and affected gland(s). Follow-up physical and radiologic examinations were conducted at regular intervals as recommended by the clinician. Retrieved data included presence or absence of relapse (a new palpable tumor mass at the place of nodulectomy or mastectomy) and presence or absence of pulmonary metastases. Death was recorded in one animal with relapse, five animals with pulmonary metastasis, and 16 without any of those events. The later 16 dogs were excluded from the follow-up study because neither the exact date of death nor the necropsy confirmation of the cause of the death was available, but their tumors were included in the histopathologic and IHC studies.
Histology
Total number of tumor tissue samples available was 228 because 37 dogs had multiple tumors. Tumor size (largest diameter) was determined by the pathologist on the gross specimen and classified as T1 (3 cm), T2 (3–5 cm), or T3 (> 5 cm). 40, 50 Tissues were fixed in 10% formalin in phosphate-buffered saline pH 7.6 (PBS) for 24–48 hours, embedded in paraffin wax, and stained with hematoxylin and eosin (HE). Histologic classification followed the WHO classification, 34 and grading was done according to the method of Lagadic and Estrada (1990). 22 Sections for the histologic evaluation of the lymph nodes were available in 26 dogs. The presence of intravascular growth, necrosis, and lymphocyte infiltrate in the vicinity of the tumor were recorded. Histologic evaluation was executed by two investigators.
IHC assays
IHC assays were performed on 3 μm sections of formalin-fixed, paraffin-embedded tissue samples. The monoclonal mouse anti-human ERα clone 1D5 (Dakocytomation S.A., Sant Just Desvern, Barcelona, Spain), diluted 1 : 50, was used with an streptavidin–biotin–peroxidase complex technique (LSAB Peroxidase Universal kit, Dakocytomation S.A). Dewaxed and rehydrated sections were subjected to high-temperature antigen retrieval by incubation with 0.01 M citrate buffer, pH 6.0, at 95 C for 3 minutes in a stainless steel pressure cooker. The monoclonal mouse anti-human PR clone PRA109 (Immunotech, Marseille, France), diluted 1 : 500, was used with an avidin–biotin–peroxidase complex (ABC) technique (Vectastain, ABC kit Elite, Vector Corporation, Burlingame, CA). Dewaxed and rehydrated sections were subjected to high-temperature antigen retrieval by incubation with 0.01 M citrate buffer, pH 6.0, at 95 C for 25 minutes in a water bath. All further IHC staining procedures were according to the instructions of the test kits. Tissue sections were counterstained with Mayer's hematoxylin.
Tissue sections of formalin-fixed, paraffin-embedded canine uterus and human breast carcinoma were run as positive controls in every assay, whereas those of canine liver were used as negative controls in the standardization of the techniques. The substitution of the specific primary antibodies by PBS in tissue sections of the dogs under study was used as negative control in every assay.
Classification of staining data was executed semiquantitatively by examining the entire tumor present in the tissue section(s) using an immunoperoxidase score. 1 The percentage of tumor cells with positive staining (proportion score, PS) was graded from 0 to 3 points by estimation. Zero points means less than 5% staining, 1 point represents 5–19%, 2 points 20–60%, and 3 points indicate that more than 60% of the tumor nuclei show positive staining. Staining intensity (intensity score, IS) estimated the average staining intensity of positive tumor cells and was scored as 0 = negative, 1 = slight staining, 2 = moderate staining, 3 = strong staining. In case of heterogeneous staining intensity within the tumor, the most predominant intensity was scored. The PS and IS are added to obtain a total score (TS) (range 0–6). ERα and PR were considered positive when PS was ≥ 1 and TS ≥ 2. Two blinded investigators determined the IHC receptors. Interobserver reproducibility was 94.6%.
Statistics
The disease-free period (DFP) was defined as the time from operation to the first recurrence of tumor, to the first lung metastasis, or combined. The DFP was grouped as follows: up to 6 months after surgery; between 6 and 12 months after surgery; more than 12 and less than 18 months after surgery; and 18 or more months after surgery.
Univariate analysis
The influence of presumptive prognostic tumor and host factors was tested in univariate analysis applying the Pearson's chi-square test with Yate's correction for 2 × 2 tables. All statistical tests were two-sided. Data were grouped as follows: hormone receptor expression (yes, not); tumor size (< 3 cm, ≥ 3 cm); lymph node status (with and without histologically proven metastasis); histologic grading (I, II, and III); histologic subtype (simple, complex, and mixed); histologic growth pattern of simple tumors (solid, tubulo-papillary); intravascular growth (yes, not); necrosis (yes, not); lymphocyte cell infiltrate (yes, not); age (≥ 8 years, > 8 years); ovariectomy (yes, not; spayed before 5 years of age, spayed after 5 years of age); contraceptive use (yes, not); parity (yes, not); pseudopregnancy (yes, not); number of tumors (single, multiple), chain location (right, left), and affected gland(s) (1, 2, 3 versus 4, 5). A
The Pearson's chi-square test with Yate's correction for 2 × 2 tables was also applied to study potential differences in host and pathologic variables on the basis of the expression of hormone receptors. A
Multivariate analysis
A categorical dependent variable model (SPSS 11.5 statistical package, SPSS Inc., Chicago, IL) was used to investigate the independent effect of all factors that proved to be significant in the univariate analysis. Logit models are similar to linear regression models but are adapted for dichotomized dependent variables. Estimation was made on the basis of maximum likelihood using the optimization procedure of Newton-Raphson (ML-binary logit). Stepwise forward selection with default inclusion (
Results
Follow-up
Eighteen dogs developed recurrences, four distant metastases, and one developed both recurrence and distant metastasis. Twenty-two dogs died during the follow-up period: 1 animal with relapse, 5 animals with pulmonary metastasis, and 16 without any of those events. The latter 16 dogs were excluded from the follow-up study because neither the exact date of death nor the necropsy confirmation of the cause of the death were available. Total number of dogs evaluated in the prognostic study was 84, and the DFP was classified as follows: up to 6 months, 13 dogs (15%) (8 had relapse only, 4 had metastasis only, 1 had both of them); between 6 and 12 months, eight dogs (9%) (7 had relapse, one had metastasis and died); more than 12 and less than 18 months, two dogs (3%) (both had relapse); and 18 or more months, 61 dogs (73%).
Epidemiologic and clinical data of the 84 dogs
Mean age of the animals was 9.3 years (range 4 to 16 year of age). Fourteen percent were spayed (9% before 5 years of age, 91% after 5 years of age), 1% had received hormonal treatment to control estrus, 75% were nulliparous, and 28% had had signs of pseudopregnancy. Fifty-six percent of dogs had single tumors, half in the right and half in the left mammary chains. The fourth and fifth glands were affected in 62% of the cases. In dogs with multiple tumors (
Histologic study
Histologic investigation yielded 155 malignant and 73 benign tumors of simple (
Forty-seven dogs had single malignant tumors, and 37 dogs had multiple, benign or malignant (or both) tumors. In the latter, the tumor selected to study the influence of morphologic features on the DFP was that with the most severe pathologic features. Total number of malignant tumors used for the prognostic study was 84. They were classified as simple (
IHC study
In the canine mammary gland, ERα staining was observed in the nucleus of normal and tumoral epithelial cells, whereas normal stromal and myoepithelial cells and spindle cells, stellate cells, cartilaginous cells, and bone cells in complex and mixed tumors were ERα negative (Figs. 1, 2, 3a). PR staining was observed in the nucleus of normal and tumoral epithelial cells also (Figs. 3b, 4–7). In addition, elongated immunoreactive nuclei located in the vicinity of epithelial cells were also found in normal and tumorous tissue (Fig. 4). Cytoplasmic staining was sometimes found in spindle and stellate cells of complex and mixed tumors (Fig. 4). The latter staining was very heterogeneous from tumor to tumor and within the same tumor (Figs. 4, 5). Normal or benign-appearing ERα- and PR-positive nuclei were frequently intermingled with atypical negative nuclei (Fig. 7). These cases were scored negative. In the positive controls, localization of ERα and PR was restricted to the nucleus of cells in all cases of positivity (surface and glandular epithelial cells and stromal fibroblasts of the endometrium, smooth muscle cells of the myometrium, and human breast carcinoma cells). In the negative controls, all nuclei were negative.
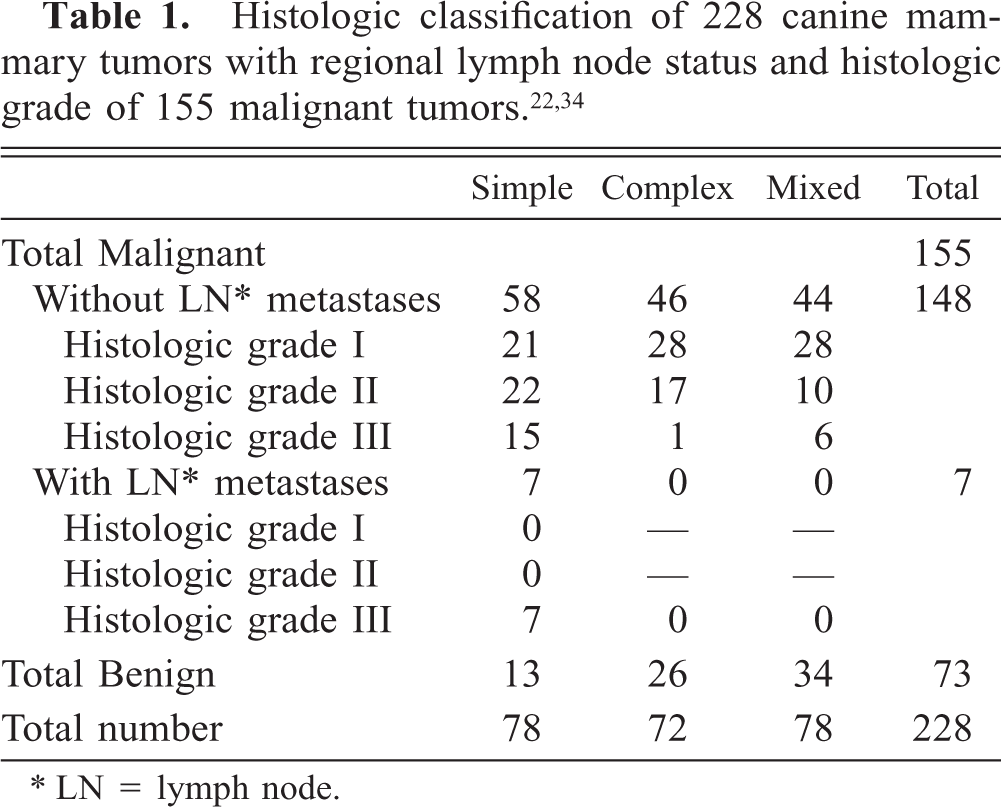
Mammary gland; dog No. 7. Benign mixed tumor. Immunoreactive products to ERα antibody are observed in the nuclei of epithelial cells (white arrows). Staining intensity is moderate. Spindle cells in their surroundings are negative (black arrows). LSAB IHC method counterstaining with Mayer's hematoxylin. Bar = 25 mm.
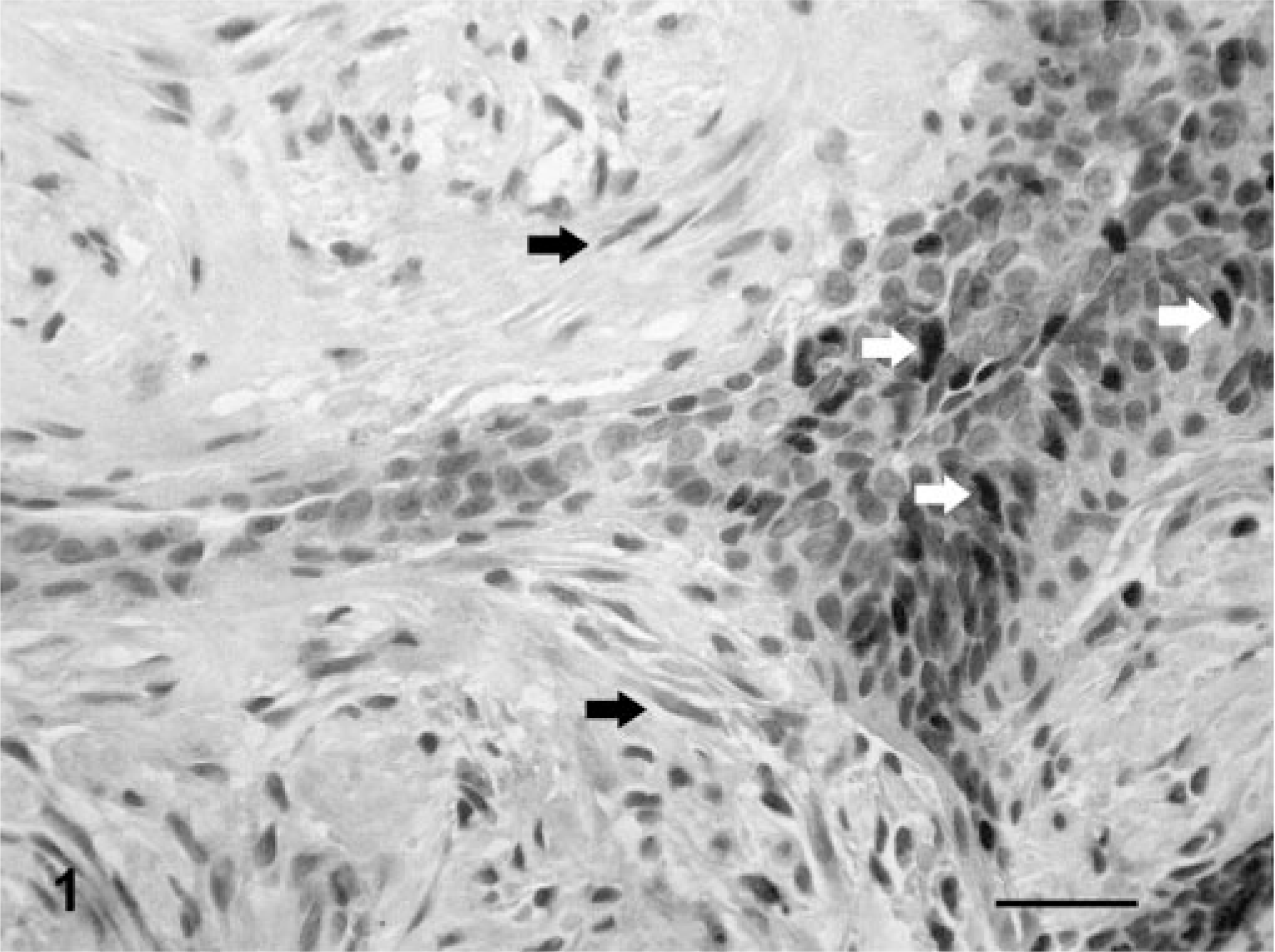
Mammary gland; dog No. 26. Simple solid carcinoma. Immunoreactive products to ERα antibody are observed in some atypical nuclei (arrows). Staining intensity is moderate. LSAB IHC method counterstaining with Mayer's hematoxylin. Bar = 15 mm.
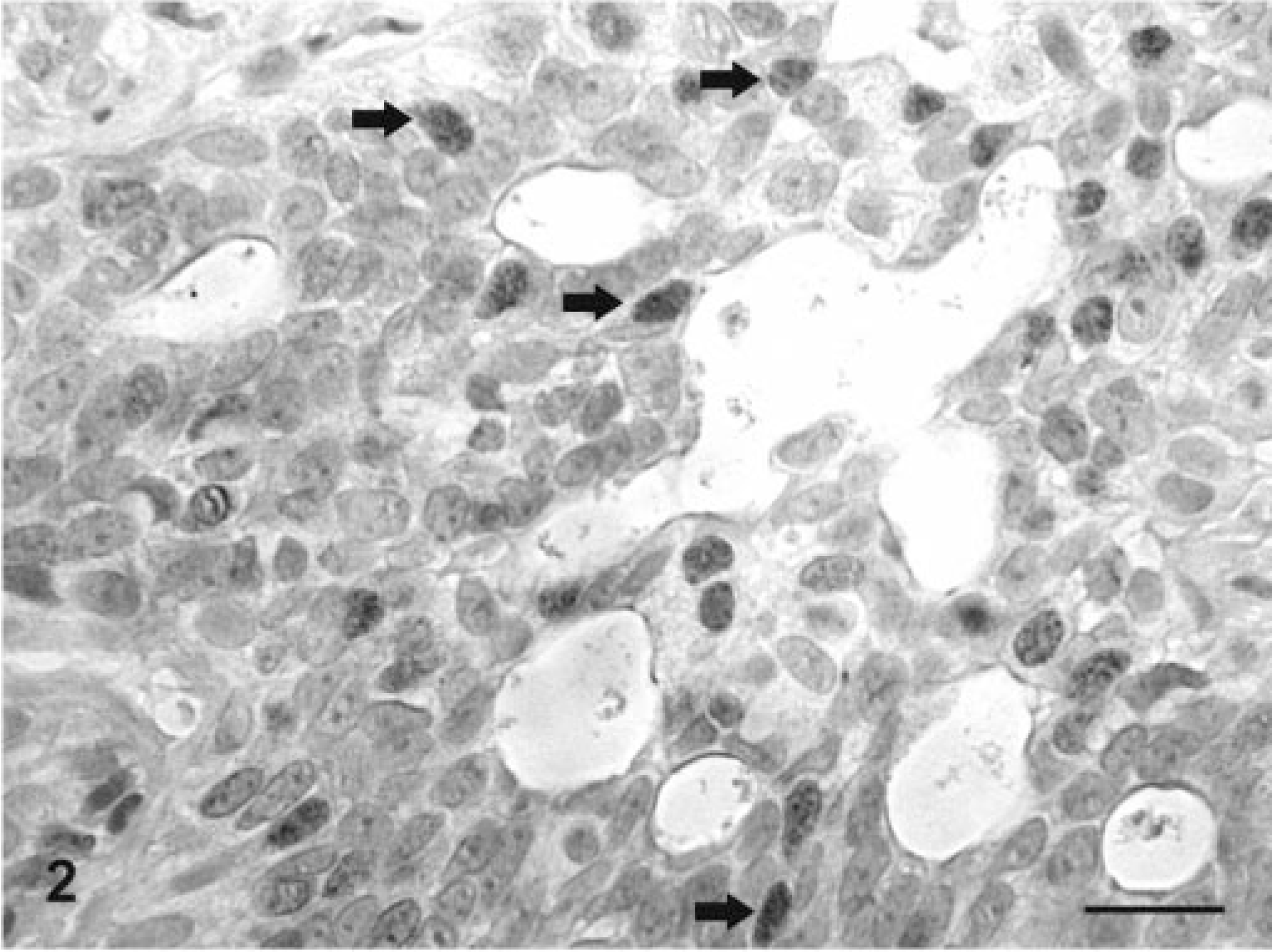
Mammary gland; dog No. 73. Carcinoma in benign tumor. ERα (
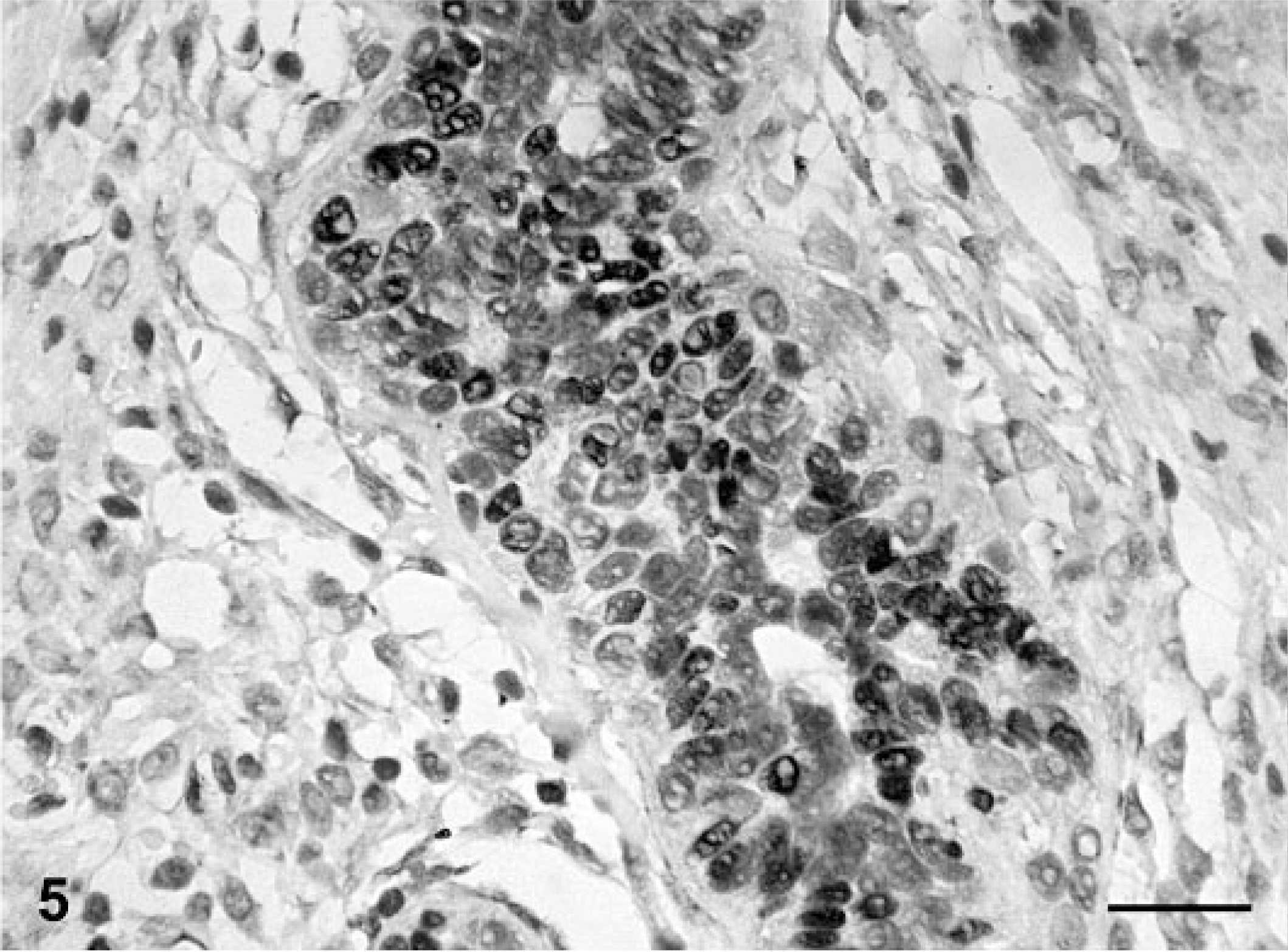
Mammary gland; dog No. 10. Complex adenoma. PR positivity is seen in the nuclei of epithelial tumor cells (thick arrows), in the cytoplasm of numerous myoepithelial tumor cells (thin arrows) and in isolated, elongated nuclei in the vicinity of epithelial cells (short arrows). ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 25 mm.
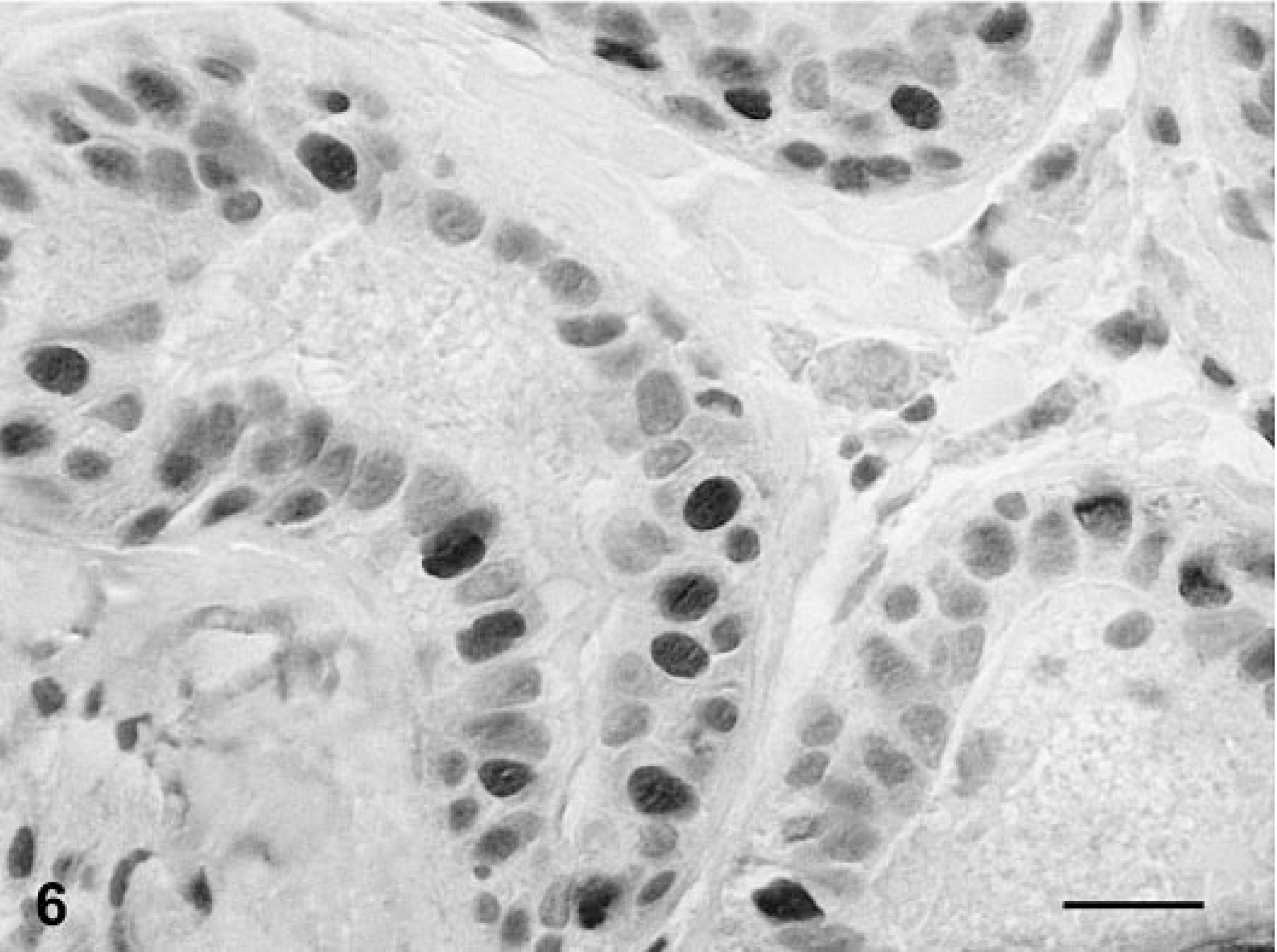
Mammary gland; dog No. 12. Complex carcinoma. Immunoreactive products to PR antibody are seen in the nuclei of epithelial tumor cells, whereas no staining is observed in the spindle cell component of the tumor. ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 15 mm.
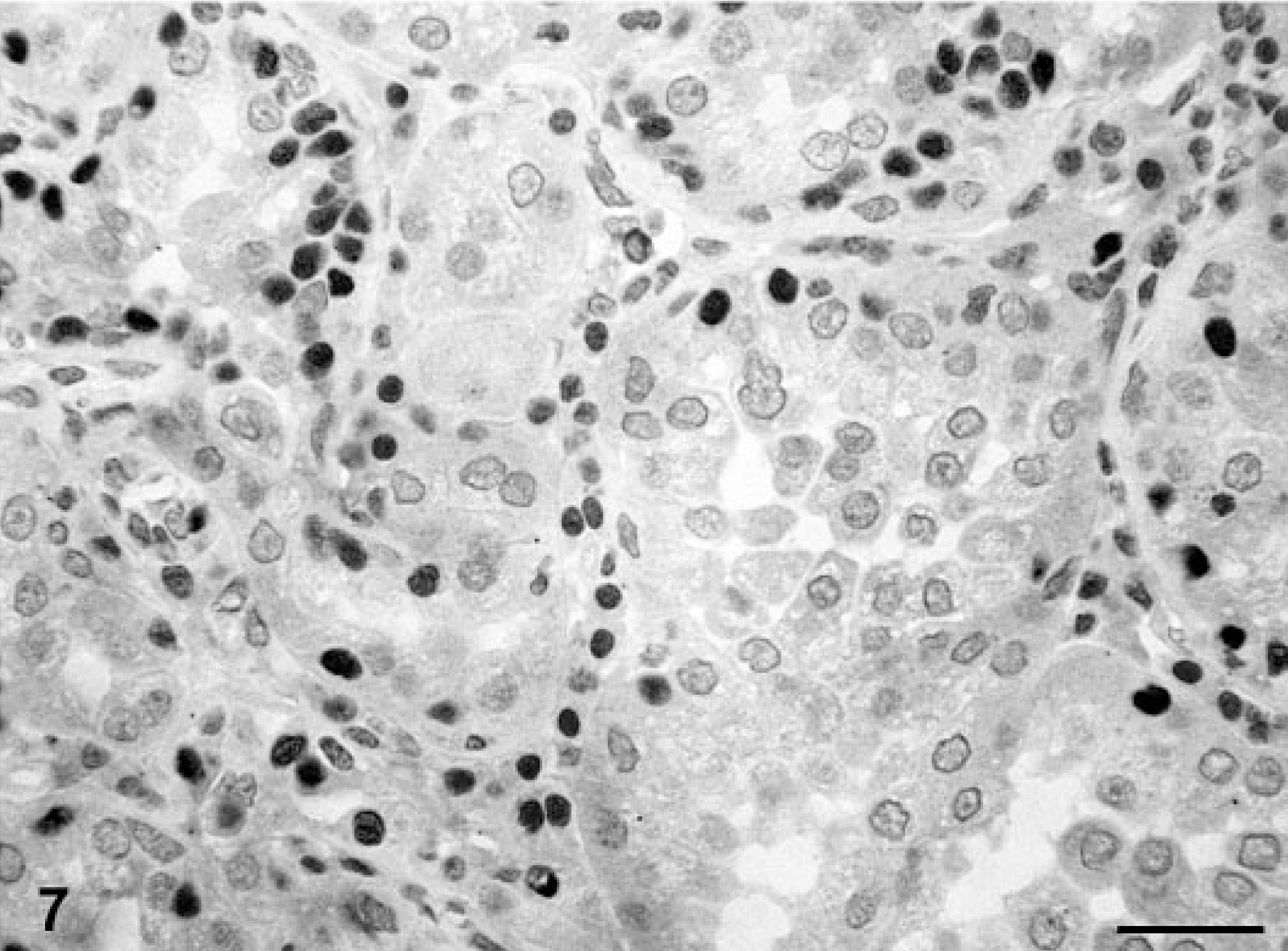
Mammary gland; dog No. 17. Simple tubular carcinoma. Immunoreactive products to PR antibody are observed in some atypical nuclei. Staining intensity is strong. ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 15 mm.
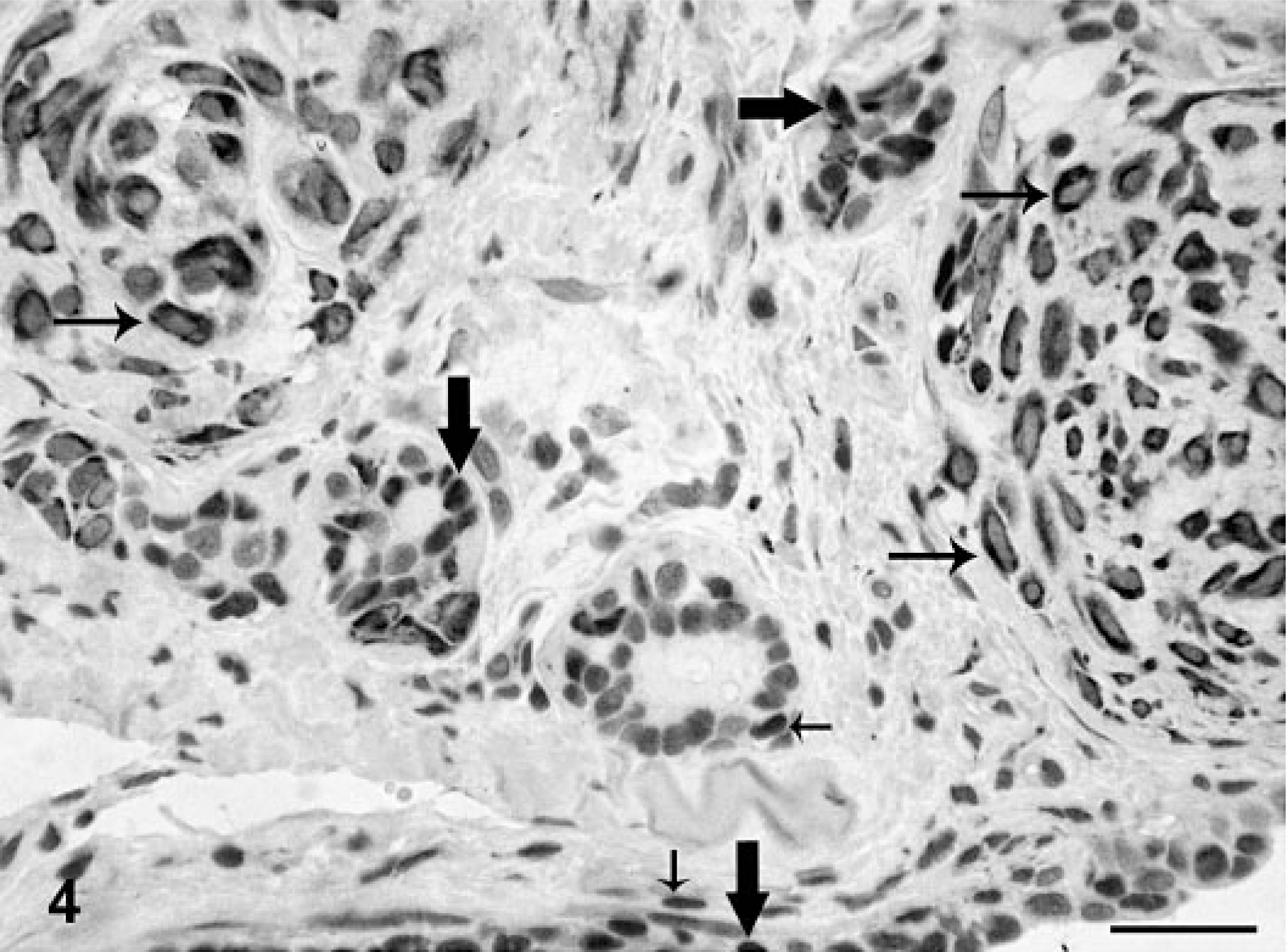
Mammary gland; dog No. 43. Simple solid carcinoma. Immunoreactive products to PR antibody are observed in benign-appearing nuclei, whereas atypical nuclei are unreactive. ABC IHC method counterstaining with Mayer's hematoxylin. Bar = 15 mm.
In the whole series of mammary tumors analyzed (
Combined expression of ERα and PR in 228 canine mammary tumors according to histologic classification. 34
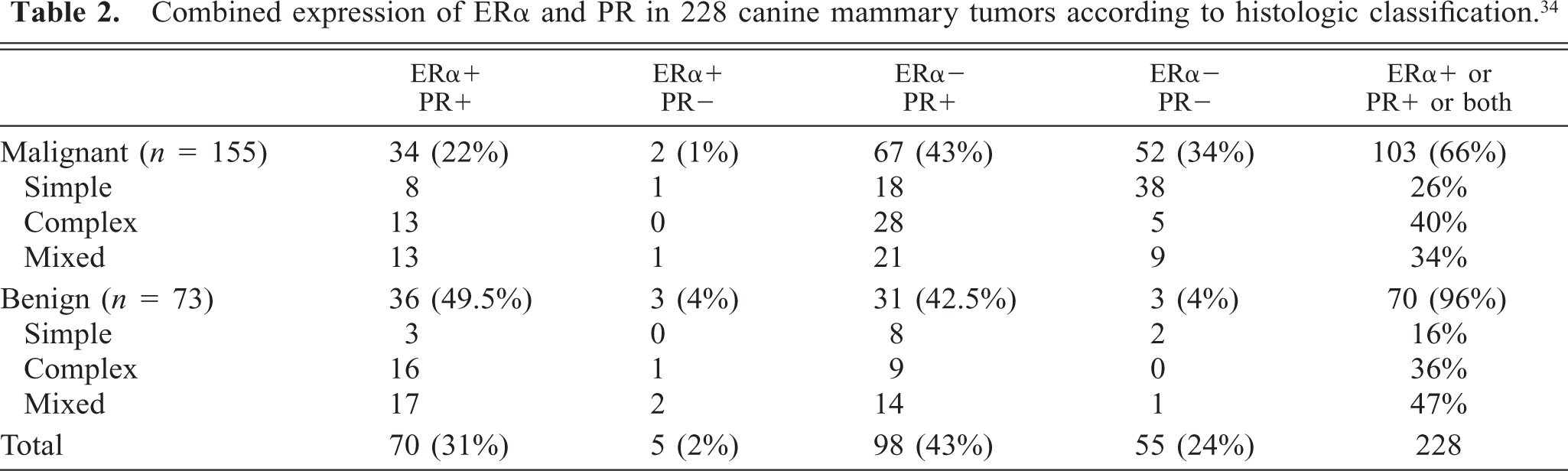
In the group of malignant tumors (
There was no statistically significant difference in hormone receptor expression (one or both receptors) between the different groups of animals regarding spaying status, parity status, hormonal treatments to control estrus, pseudopregnancy, age of the animals, and number or location of tumors. The study of individual associations between ERα expression, on the one hand, and PR expression, on the other, with clinical and epidemiologic variables, revealed that ERα expression was more frequent in the group of younger animals (≤ 8 years-old,
Univariate analysis
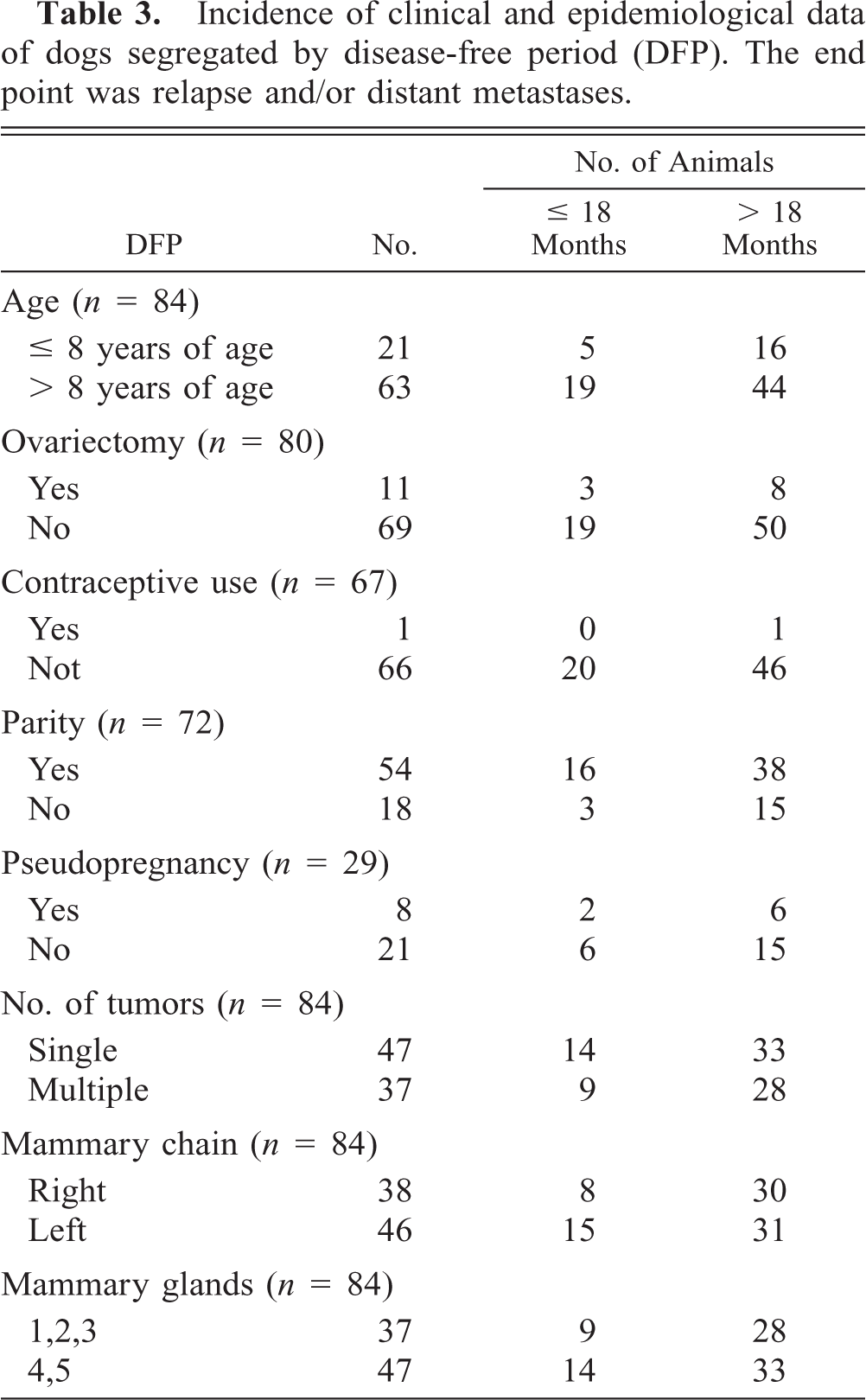
Incidence of host and tumor factors segregated by DFP are shown in Tables 3, 4, respectively. Tumor size (
Incidence of clinical and epidemiological data of dogs segregated by disease-free period (DFP). The end point was relapse and/or distant metastases.
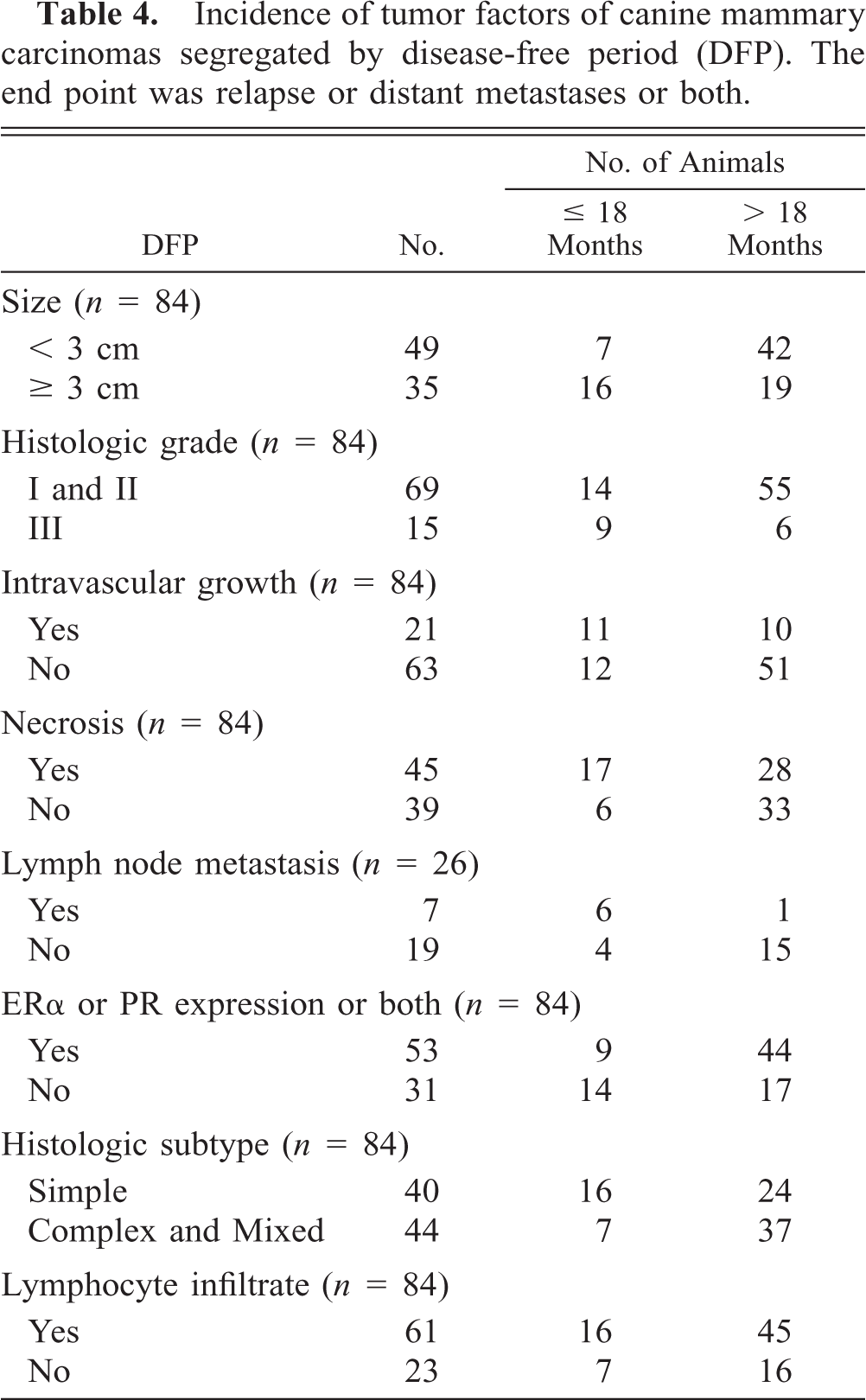
Incidence of tumor factors of canine mammary carcinomas segregated by disease-free period (DFP). The end point was relapse or distant metastases or both.
Univariate and multivariate analysis of the prognostic factors under study. The end point was relapse or distant metastases or both.∗
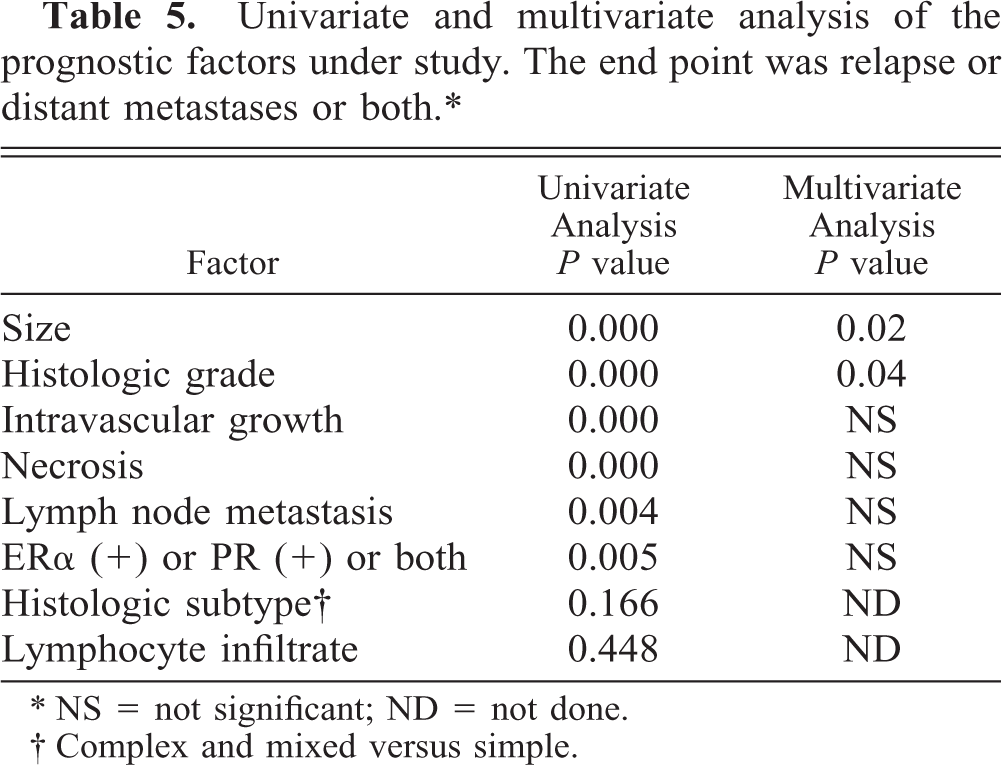
∗ NS = not significant; ND = not done.
† Complex and mixed versus simple.
Multivariate analyses
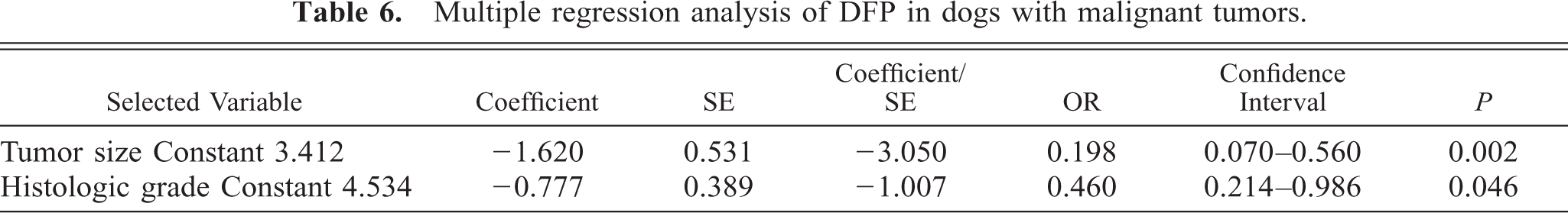
To evaluate the joint effect of tumor size, lymph node status, histologic grade of malignancy, hormone receptor expression, intravascular growth, and necrosis (independent variables) on the follow-up variable DFP (dependent variable), multivariate analyses were done. The results are based on data from 84 animals.
Tumor size (
Multiple regression analysis of DFP in dogs with malignant tumors.
Discussion
In the present series of bitches with malignant mammary tumors, tumor size, 4, 9, 18, 21, 35, 44, 46, 55, 60 histologic grading, 15, 22, 44 intravascular growth, 11, 15, 21, 22, 43, 51, 60 and necrosis were the most significant prognosticators in the univariate statistical study, followed by lymphnode status 12, 15, 18, 21, 35, 44, 60 and steroid receptor status. 25, 31, 39, 42, 52 However, to analyze the possibility these prognosticators were related between each other, a multivariate stepwise analysis was performed in order to identify independent prognostic factors. Multivariate stepwise analysis confirmed the independent prognostic value of tumor size and histologic grading for disease-free survival 15 and was in agreement with the independent prognostic value of these two variables when overall survival was used as the end point. 18, 22, 35, 46, 55 The prognostic power of the other factors was not retained in the multivariate analysis after tumor size and histologic grading had been introduced into the model, which means that their association with a longer DFP was dependent on tumor size and histologic grade.
Intravascular growth was defined as the presence of neoplastic emboli in vessels on HE-stained tissue sections. As in previous univariate studies using either overall survival
11,
43,
60
or disease-free survival as the end point,
15,
21,
22
intravascular growth had prognostic value. However, other authors failed to find such a relationship
35
and it also dropped on multivariate analysis.
60
Sarli et al.
51
analyzed intravascular growth as part of a staging system, which included stromal invasion also, and found it to be an independent prognosticator. These differences may be because of the difficulty that sometimes arises when trying to differentiate lymphatic and, less often, blood vessels from artifacts on HE-stained tissue sections. The presence of ischemic necrosis is considered to be an indirect measurement of the growth rate of malignant tumors. Nowadays, there are more accurate (and expensive) methods of evaluation of this feature, such as the expression of cell proliferation antigens.
44,
51
The prognostic value of the presence of necrotic foci was not retained in the multivariate analysis most probably because of its close relationship with tumor size. In the univariate analysis, lymph node involvement indicated poorer disease-free survival. Thus, six out of seven dogs with histologically proven lymph node metastasis at first presentation developed recurrences (
Previous studies based on the measurement of ER and progestin receptor in tumor cytosols demonstrated the prognostic value of hormone receptor content which is currently attributed to this characteristic of canine mammary tumors. 33, 34, 50 Those studies used univariate statistical analysis with overall survival as the end point. 25, 31, 42, 52 The independent prognostic value of immunohistochemically determined ER using the DFP as end point has been demonstrated, 39 but the prognostic value of immunohistochemically determined combined expression of ERα and PR has not been analyzed previously. In contrast to the results of Nieto et al., 39 steroid receptor status was not an independent prognosticator for DFP in the multivariate analysis, conceivably because of the significant correlation between lower tumor size and histologic grading, on the one hand, and steroid receptor positivity, on the other. Nonetheless, more studies regarding the prognostic value of the immunohistochemically determined steroid receptor status in canine mammary cancer are needed before a conclusion can be drawn. Thus, initially, ERα-positive human breast tumors were associated with an improved prognosis, but studies with long-term follow-up have suggested that ERα-positive tumors, despite having a slower growth rate, do not have a lower metastatic potential. 61
Lymphoid cellular reactions are assumed to indicate morphologic evidence of an antitumor immune response and have been associated with lower recurrence rate,
15
but they are not yet considered a recognized, well-accepted prognosticator in canine mammary cancer.
33
The histologic classification of malignant tumors according to the participation of different cell types in simple, complex, and mixed
34
is based on studies indicating a range of increasing malignancy from complex carcinoma to simple carcinoma to sarcoma,
9,
35,
43
the later being the most malignant type.
18
Using the WHO classification,
34
we found no differences in DFP between simple (one-cell type) and complex and mixed (more than one-cell type) tumors. One reason may be the heterogeneity of the group of complex and mixed tumors. Thus, it included 11 grade I, 14 grade II, and 1 grade III complex carcinomas, whereas 10 out of 18 “mixed carcinoma” had histologic grading II (
Another objective of this study was to study the relationship between hormone receptor expression and host and tumor factors using all 228 tumors harvested from the female dogs of this series. Immunohisto-chemically determined ERα, PR, or both were detected in 76% of 228 canine mammary tumors, a figure which is within the range of 65–92% reported using tumor cytosol–based biochemical methods of measurement of steroid receptor content. 3, 6, 30, 42, 47 The combined expression of ERα and PR in canine mammary cancer has not been evaluated with IHC methods of analysis but our findings confirm previous data of immunohistochemically determined ERα 14, 16, 39, 56 and PR. 14, 24 Localization of ERα and PR was restricted to the nucleus of epithelial, but not myoepithelial or mesenchymal tumor cells, as already reported in previous IHC studies of the canine, 14, 16, 23, 24 feline, 28 and human species. 17, 19, 20, 41, 45, 57 The cytoplasmic staining observed in spindle and stellate cells of complex and mixed mammary tumors has been observed by others using the same PR antibody and has been attributed to disturbances in nuclear transport of the receptor. 23 The staining of some fusiform nuclei in normal and tumoral feline mammary gland has been observed using the same PR antibody. 28 A recent study suggests that some of these cells may be of myoepithelial origin considering their IHC characteristics. 10
Statistically significant differences regarding the presence of one or both receptors were observed between benign and malignant tumors as well as between complex-mixed and simple histologic subtypes of benign and malignant tumors. The presence of one or both receptors was more frequent in benign tumors 3, 6, 14, 39, 42, 47, 48, 52 as well as in complex and mixed histologic subtypes of benign and malignant tumors. The direct relationship between the histologic subtypes complex and mixed and the presence of one or both receptors has been already reported in the single IHC study that has evaluated this parameter. 14 However, previous biochemical studies showed that ERα or PR content (or both) were similar in simple, complex, and mixed histologic subtypes of both benign and malignant mammary tumors of the dog. 8, 30, 31, 42, 47, 52 It is difficult to compare IHC and biochemical studies concerning this issue because of two reasons. First, IHC methods allow both to ensure that the tissue section contains tumor, avoiding false-positive results due to steroid receptors in adjacent or trapped normal tissue, and relate receptor content to morphology. This permits knowledge of intratumor heterogeneity, which is particularly important in canine mammary tumors. Second, great differences exist in histologic classifications used in earlier studies.
In the group of malignant tumors (
No association was found between the IHC expression of one or both receptors and age of the animals, spaying and parity status, hormonal treatments to control estrus, pseudopregnancy, and number or location of tumors. There is no unanimity concerning this issue in literature. Thus, either lack 7, 42, 47, 48, 52 or presence 3, 39 of relationship between age and hormone receptor (ERα, PR, or both) have been reported. In this study, ERα expression, irrespective of the PR status, was more frequent in bitches less than 8 years of age. 3, 39 The relationship between spaying and the presence of hormone receptors in mammary carcinoma has been seldom evaluated and either no relationship 52 or an indirect relationship 39 have been reported. Similarly, either absence 31, 39, 47 or presence 6, 43 of a direct relationship between hormone receptors and clinical sign of pseudopregnancy have been reported. Also, different findings were reported concerning the hormone receptor content of mammary carcinoma regarding multiplicity of tumors. 6, 31, 42, 47 On the contrary, parity status, hormonal treatments to control estrus, and location of tumors have not been shown to correlate with hormone receptor content 6, 7, 31, 39, 42, 47 or expression. 39
One of the disadvantages of IHC methods of study of ERs is that all receptors demonstrated may not be functional because a defect in the ERα functional cascade may exist at transcriptional, translational, and posttranslational levels.
2
A method of estimating ERα function is through detection of PR because PR synthesis is considered to be dependent on the presence of estradiol and a normal functioning ERα.
20,
29
We analyzed the combined expression of ERα and PR, and differences were found between benign and malignant tumors. Thus, the most frequent group of hormone receptors–positive benign tumors were the ERα-positive and PR-positive group.
31,
42,
47
However, in contrast with previous data, the ERα-negative and PR-positive group was the most frequent hormone receptor–positive group among malignant tumors, being twice as frequent as the ERα-positive and PR-positive group (22%).
25,
30,
42,
47,
52
Canine mammary cancer with PR-only ranges from 8
30,
42,
47
to 22%,
52
as in human breast cancer (7–17%).
19,
29,
41,
57
The two main groups of malignant tumors, the ERα-positive and PR-positive group (
The unexpected and high prevalence of the PR-only expression group in malignant tumors could be because of technical reasons, such as the use of different antibodies between the different studies and the low sensitivity of our IHC method of detection of ERα. Fifty percent of benign and 22% of malignant mammary tumors expressed ERα. These figures are in line with previous IHC studies in which 55% of benign 39 and 24% of malignant mammary tumors 16 expressed ERα. However, lower (10%), 14 higher (59%), 56 and much higher figures (87.5%) 39 have been reported in malignant tumors. Negative staining was not likely to be attributed to an experimental artifact because ERα immunoreactivity was seen in nonmalignant epithelial nuclei present in the same specimen. Variability in staining can occur for a number of technical reasons. These include the method of antigen retrieval, concentration of primary antibody, and sensitivity of the detection system. 2 Our results are similar to those of Graham et al., which obtained a very high concordance rate of ERα status between ligand binding and IHC assays. 16 A high concordance rate between ligand binding and IHC assays is one of the requirements of IHC quality assurance schemes currently available for the measurement of steroid hormone receptor in human breast cancer. 2 The possibility also exists that the IHC method of detection of PR is giving false positives. However, technical controls were adequate and previous IHC studies have shown similar data among benign 14, 24 and malignant 14 canine mammary tumors. Nonetheless, assessment of staining, particularly regarding the correct cutoff point to distinguish hormone receptor “positive” from hormone receptor “negative” tumors, is a difficult area in veterinary studies because it is not based on correlation studies between positive tumors and positive response to hormone therapy as in humans. 1, 2 We, as well as others, 39 used a human scoring system based on both proportion of stained cells and intensity of staining. Our results suggest that the functional status of ER cascade is kept in half of the benign and one third of the malignant hormone receptor–positive tumors. Factors that could explain why some tumors contain PR and no detectable ERα include nuclear processing of ERα, which decreases the ERα content by about 70%. 52 Thus, if ERα were in low concentrations initially, this type of loss or inactivation may result in ERα at a concentration too low to be detected by routine biochemical or IHC methods. A transient increase in serum estradiol associated with proestrus in intact dogs (86% of the animals in this study) may stimulate cells containing ERα to begin PR synthesis, followed by subsequent ERα processing. On the other hand, experimental evidence suggests that some false-negative ERα values might result from splice variants that lack the hormone-binding domain (and are not detected by ligand binding and some IHC assays) but are translated and still retain some functional activities. 13 Furthermore, there may be aberrant ERα forms, which are unable to transfer into cell nucleus and be observed by ERα antibodies. 20 The possibility exists that the functional estrogen cascade is maintained in the ERα-negative and PR-positive group through the expression of the second ER (ERß). 27 Finally, the expression of PR-only may reflect involvement of other pathologic or normal mechanisms in the regulation of cell proliferation in that particular subset of tumors. Thus, it has been suggested that progestin-induced and locally produced growth hormone acts as an intermediate in the progestin-stimulated development of canine mammary tumors. 36 A building body of evidence, from cell culture, animal studies, and, most recently, several major clinical studies involving hormone replacement therapy, strongly supports the notion that progestins generally stimulate breast cancer. 37 These data indicate that progestin-related pathways may provide effective targets for breast cancer therapy. Although ERα-positive canine mammary tumor cell lines have been shown to respond to the selective ER modulator tamoxifen, 53 adjuvant endocrine therapy is not currently used on the basis of ERα expression in canine mammary carcinoma. Treatment of bitches with tamoxifen has been reported to produce estrogen-like side effects and is not advised for dogs. 50 The high prevalence of PR expression in canine mammary carcinoma opens the possibility to explore both the use of PR antagonists to control the progression of the disease after surgery in bitches and the value of the dog as a natural model for neoadjuvant hormone therapy in women with breast cancer.
Footnotes
Acknowledgements
This work has been supported by grants AGL2003-06289 (Dirección General de Investigación, Ministerio de Ciencia y Tecnología, Spain) and CVI-287 (Plan Andaluz de Investigación, Consejería de Educación y Ciencia, Junta de Andalucía, Spain).