Abstract
Lipid-rich carcinomas occurred in seven female dogs. Affected dogs were purebred (all but one), intact (all but one), and between 4 and 13 years of age. Five of them had a history of parity, one had pseudopregnancy, and none had received contraceptive steroids. The tumors were single (five cases) or multiple (two cases) well-circumscribed masses of different sizes (varying from 1 to 6 cm in diameter), composed of solid nests and cords of tumor cells separated by a moderate amount of stroma. The tumor cells contained either multiple and small or large and solitary vacuoles that pushed the nucleus to the periphery of the cell (signet-ring cell). A glandular epithelial immunophenotype (cytokeratins 5 and 8 and 8 and 18) was observed in the majority of tumor cells. All tumors lacked both estrogen and progesterone receptors, and five out of seven tumors gave rise to local recurrence and proximal or distant metastases or both.
Lipid-rich carcinoma is a mammary carcinoma characterized by cells with abundant vacuolated cytoplasm that contains a large amount of neutral lipid. 10 According to the two latest classifications of mammary tumors of the dog, this type of tumor is extremely rare in the species, but epidemiologic, clinical, and pathologic features are not fully characterized. 10,11 Here, we describe the clinicopathologic features of seven cases of lipid-rich carcinoma of the mammary gland of the dog. In addition, we have analyzed the immunophenotype and steroid hormone receptor content of the tumor cells by using different tumor markers and immunohistochemical techniques.
Clinicopathologic data of the seven dogs are given in Table 1. Tissue samples were fixed in formalin and processed routinely according to standard procedures. In addition, frozen material was taken from a recurrence in dog no. 2. Hematoxylin and eosin, periodic acid–Schiff, Congo red, and Oil Red O histochemical techniques as well as the avidin–biotin–peroxidase complex (ABC) immunohistochemical technique were performed. Technical data on specific antibodies and their immunoreactivity pattern in the normal mammary gland (internal positive control) are given in Table 2. Normal canine tissues known to contain the antigens sought were used as external positive controls as follows: canine skin for cytokeratins 5 and 8 (clone RCK-102; squamous epithelium), cytokeratins 8 and 18 (NCL-5D3; glandular epithelium), cytokeratin 14 (clone LL002; basal cells), actin (HHF 35; muscle), calponin (clone CALP; smooth muscle), and vimentin (dermal fibroblasts) antibodies; and canine uterus for estrogen and progesterone receptor (myometrial and endometrial—stromal and epithelial—cells) antibodies. Tissue sections under study treated with either normal rabbit serum (for polyclonal antibodies) or mouse ascites fluid (for monoclonal mouse antibodies) instead of the corresponding specific antibodies were run as negative controls. The immunohistochemical techniques of detection of estrogen and progesterone receptors have been detailed elsewhere. 9
Clinicopathological data of the seven dogs with lipid-rich carcinomas.
∗ Mammary glands numbered 1 (pectoral) to 5 (inguinal).
† L = left.
‡ R = right.
Grossly, single or multiple well-circumscribed masses of different sizes were observed affecting the mammary glands (Table 1). The nodules were firm and grayish or yellowish with small areas of hemorrhage and necrosis on the cut surface in dog nos. 1, 2, 3, and 7.
The antibodies used to analyze the immunophenotype and the hormone dependence of lipid-rich carcinomas of seven dogs, their technical features, and their immunoreactivity with epithelial and myoepithelial cells of the normal mammary gland using the avidin-biotin peroxidase complex technique are shown.

∗ Pronase = Protease E (Sigma Chem. Co.).
† HTAR = High-temperature antigen retrieval with 10 mM citrate buffer pH 6.0.
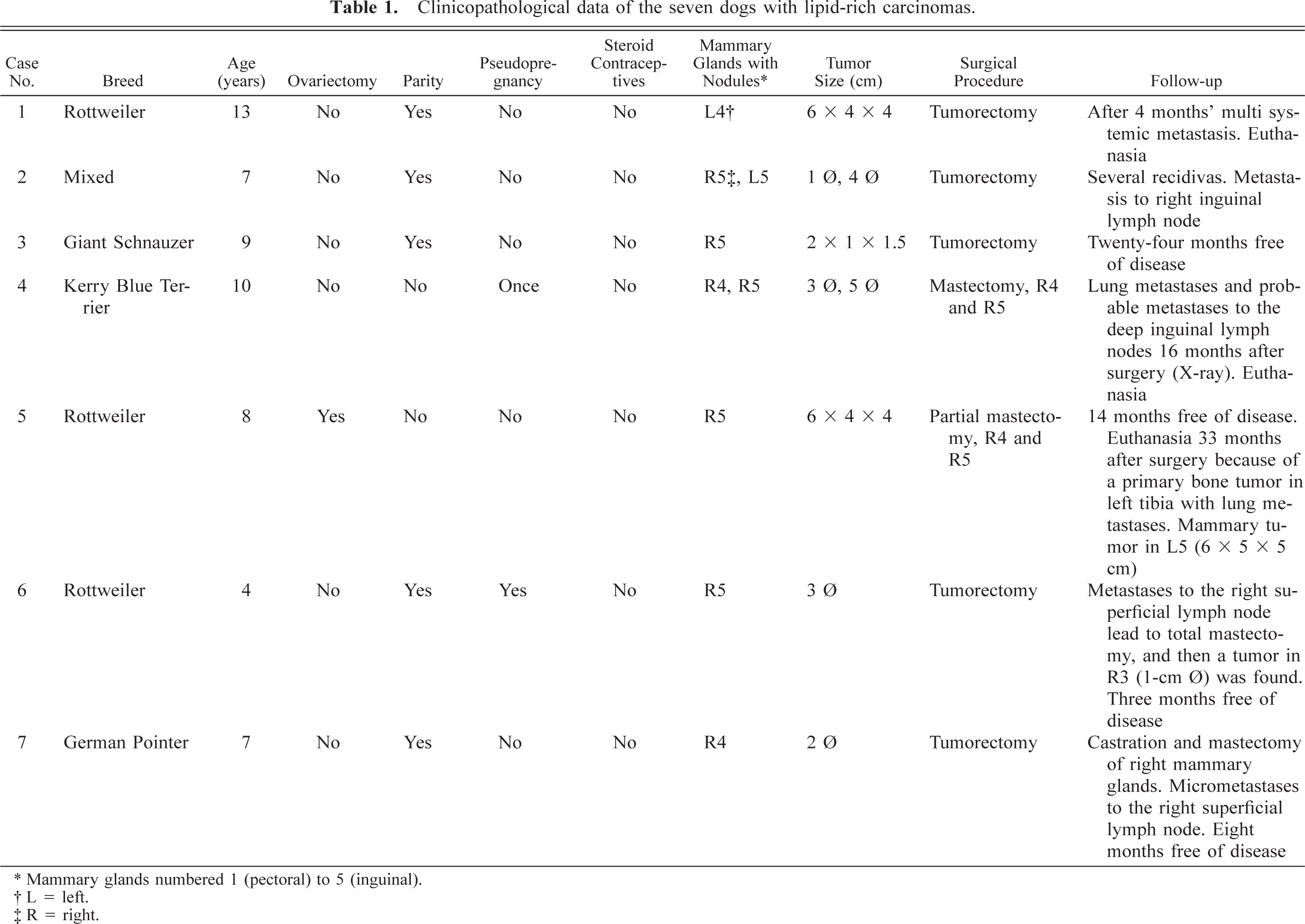
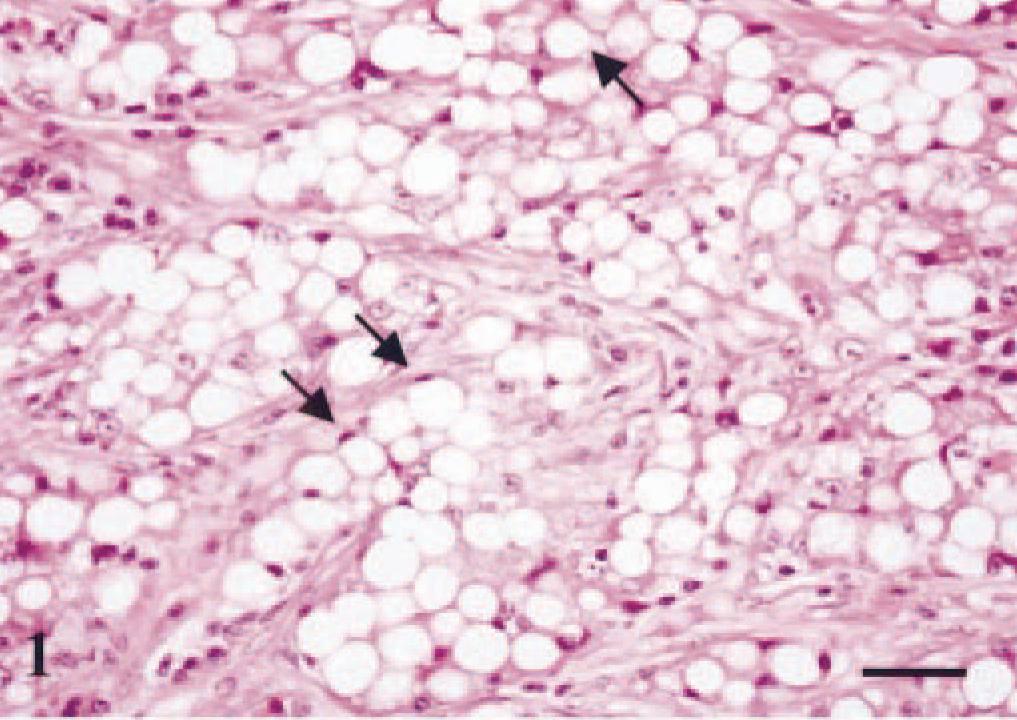
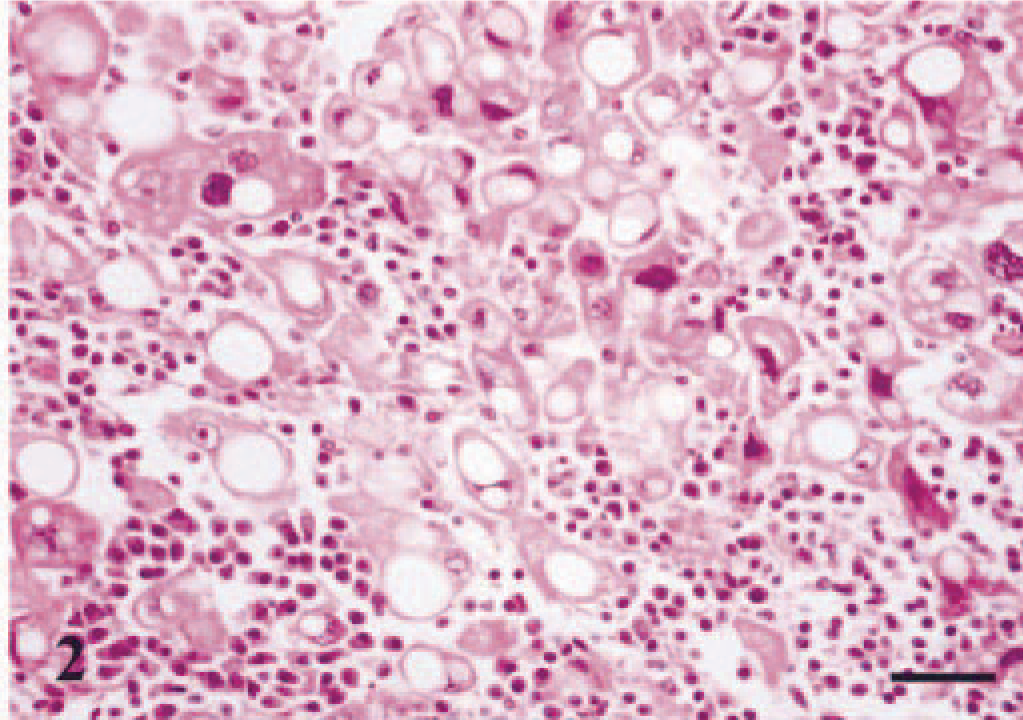
Histologic examination of the tissue specimens revealed mostly expansive neoplastic growths. Tumor cells were arranged in solid nests and cords separated by a moderate amount of stroma and had vacuolated cytoplasm and rounded or flattened nuclei (Fig. 1). Most frequently, there was a single vacuole pushing the nucleus to the periphery of the cell (signet-ring cell). Atypia was moderate in dog nos. 1, 2, 3, 5, 6, and 7, whereas tumor cells of dog no. 4 showed marked atypia and nuclear pleomorphism (Fig. 2). The mean number of mitosis in all cases was low (4 × 10 high-power fields). There were small areas of necrosis within the tumor, and several images of vascular invasion by emboli of tumor cells were observed in all but dog nos. 3 and 7 (Fig. 3a, b). A great amount of neutral lipid was identified in the cytoplasm of the tumor cells by Oil Red O stain on frozen material from a recurrence in dog no. 2 (Fig. 4) and on frozen material previously fixed in formalin from dog nos. 4, 5, and 6. No reaction with periodic acid–Schiff histochemical technique was observed within tumor cells. In some areas of dog nos. 1, 2, 4, and 6, an extracellular amorphous, pale eosinophilic material was seen in the basal membranes of some nests of tumor cells and in the stroma of the tumors. These deposits did not react with the Congo red histochemical stain. Lymphocytes, plasma cells, and eosinophils (Figs. 2, 5) diffusely infiltrated the tumor growths. At several sites of dog nos. 1, 2, 5, and 6, eosinophils constituted the main inflammatory cell and were intermixed with tumor cells.
Mammary gland; dog no. 5. Lipid-rich carcinoma. Tumor cells are arranged in solid nests separated by moderate amount of stroma. The cytoplasm contains either multiple and small or large and solitary vacuoles. In the latter, the nucleus is located at the periphery of the cell (signet-ring cell) (arrows). HE. Bar = 15 µm.
Mammary gland; dog no. 4. Lipid-rich carcinoma. Sheet of vacuolated tumor cells with high degree of atypia and nuclear pleomorphism diffusely infiltrated by lymphocytes and plasma cells. HE. Bar = 15 µm.
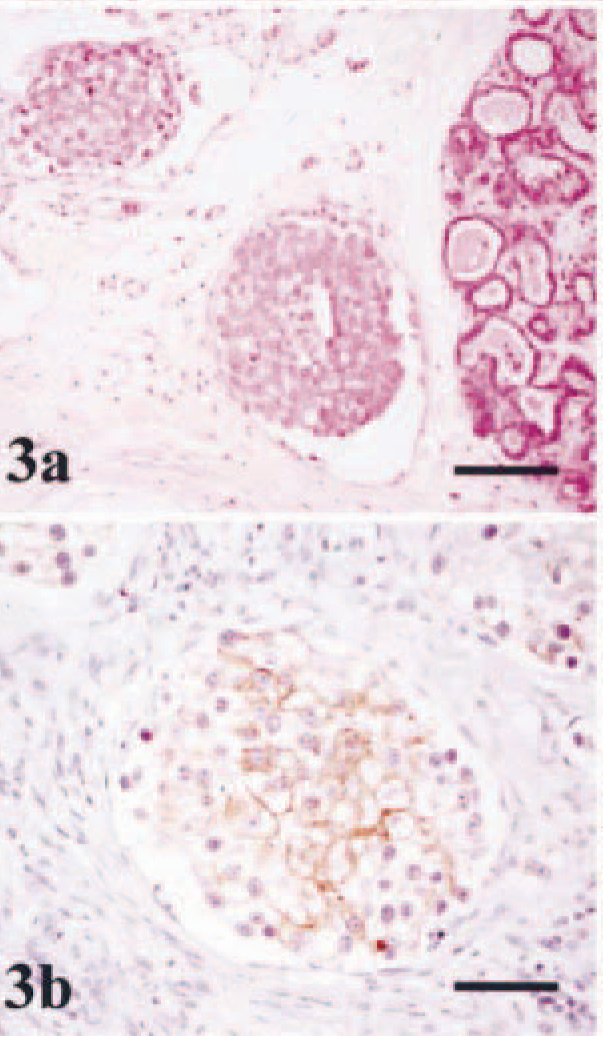
Mammary gland; dog no. 2. Lipid-rich carcinoma.
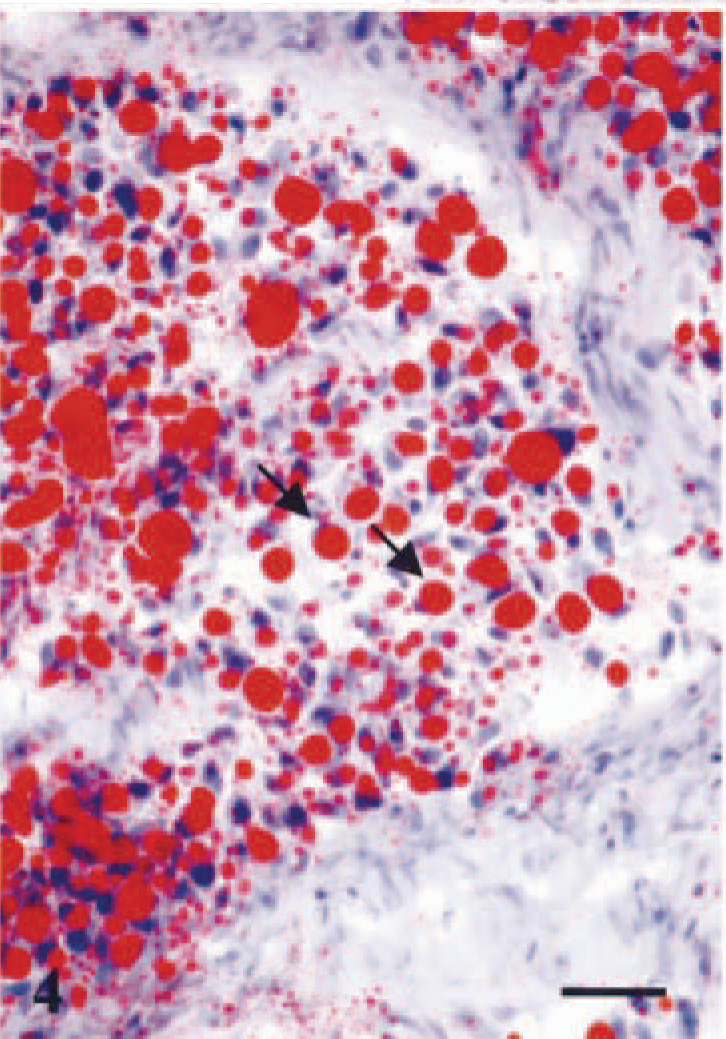
Mammary gland; dog no. 2. Lipid-rich carcinoma. The cytoplasm of tumor cells contains abundant large vacuoles of neutral lipid. In some of the cells the nucleus is located at the periphery (signet-ring cell) (arrows). Oil Red O staining. Bar = 20 µm.
At the time of the initial diagnoses, lipid-rich carcinoma was the only type of tumor observed in dog nos. 1, 2, 3, 4, 6, and 7. In dog no. 5, a mammary tumor (not further examined) was additionally found in the left fifth mammary gland. Tumor masses detected during postmortem examination of dog no. 1 (Table 1) in the sacrum–iliac lymph nodes, the vertebral bodies of T8, T10, and T11, the uterus, the lungs, the right adrenal gland, and the right kidney, as well as recurrent tumor and superficial iliac lymph node metastases of dog no. 2 (Table 1), had the same histologic features as the primary mammary tumor. Frozen sections taken from the latter revealed high amounts of neutral lipid within the cytoplasm of the tumor cells with the Oil Red O histochemical technique.
Immunohistochemically, the majority of tumor cells in dog nos. 1, 2, 4, 5, and 6, and at least one third of the cells in dog nos. 3 and 7, reacted with the antibody that recognizes cytokeratins 5 and 8 (RCK-102 antibody). The reaction was diffuse within the cytoplasm with membrane reinforcement (Fig. 5). In addition, a small number of cells had a faint intracytoplasmic reactivity with strong membrane reinforcement with the antibody that recognizes cytokeratins 8 and 18 (NCL-5D3 antibody) (Fig. 3b). Tumor cells did not react with cytokeratin 14, calponin, muscle actin, or vimentin antibodies. Estrogen receptor alpha and progesterone receptor were not detected in lipid-rich carcinomas, but adjacent ductal and acinar epithelial cells showed heterogeneous nuclear staining (not shown). Fusiform cells forming fascicles either surrounding (completely or incompletely) the neoplasm or within were identified as myofibroblasts because they reacted with calponin, muscle actin, and vimentin antibodies but did not express cytokeratin 14 (not shown).
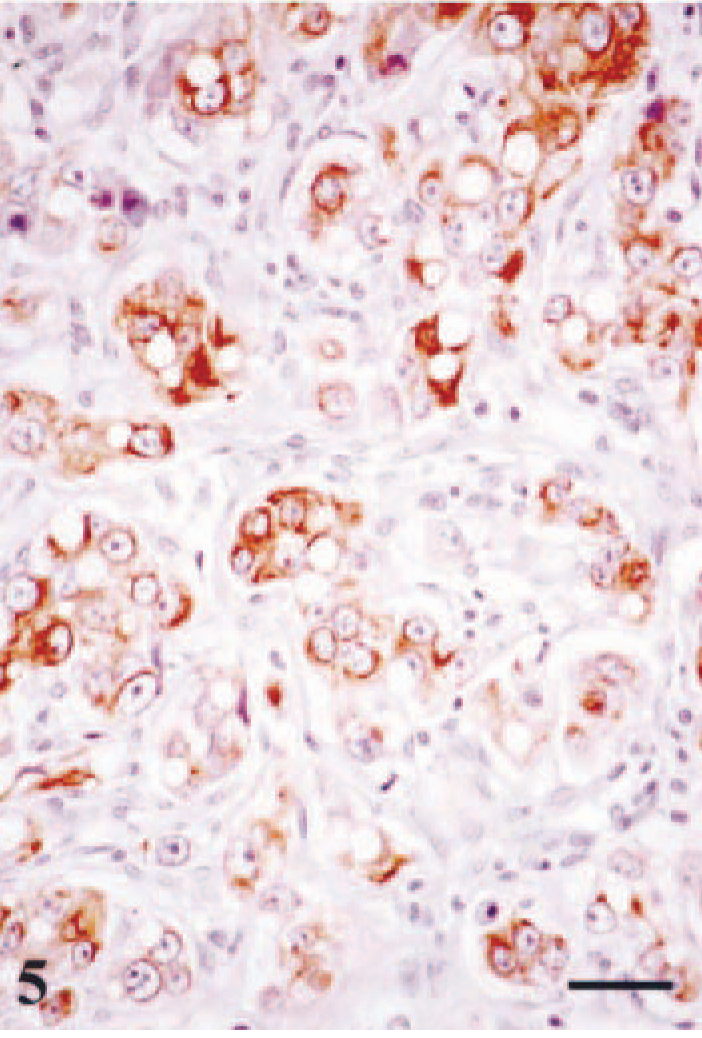
Mammary gland; dog no. 1. Lipid-rich carcinoma. The majority of cells show cytoplasmic immunoreactivity with cytokeratins 5 and 8 monoclonal antibody (RCK-102). The reaction is intracytoplasmic with occasional membrane reinforcement. A dense lymphoplasmacytic infiltrate is seen in the fibrous stroma. ABC immunohistochemical method counterstaining with Mayer's hematoxylin. Bar = 15 µm.
According to current morphologic descriptions of human and canine cases, the histologic appearance of the tumors was consistent with lipid-rich carcinomas. 10,11,13 However, it is difficult to diagnose carcinomas of this type, not only because there is little experience with them but also because a definite diagnosis can be made only if the presence of abundant lipid droplets in the tumor cells is proved by lipid staining or electron microscopy. 13 Therefore, the possibility exists that a number of carcinomas of this type had failed to be detected. 5 Lipid-rich carcinoma was first described in women by Aboumrad et al. 2 These authors thought that the lipid droplets were produced and secreted by the tumor cells instead of being the result of regressive degeneration because lipid droplets existed uniformly in metastasized lymph nodes and in tumor cells showing mitosis. We have observed that the tumor cells of metastasized growths in lymph nodes (dog nos. 1, 2, and 7) and bone marrow, lungs, endometrium, and medulla of adrenal gland (dog no. 1) contained lipid droplets. In addition, the lipid content of tumor cells was demonstrated in dog nos. 2, 4, 5, and 6 by Oil Red O stain. However, there is a controversy about the lipid-secreting nature of tumor cells on the basis of ultrastructural findings. 5,18
Immunohistochemically, the tumor cells had a glandular epithelial immunophenotype because they expressed high– and low–molecular weight cytokeratins exclusively. In humans, the expression of markers of epithelial glandular differentiation, including alpha lactalbumin, lactoferrin, and human milk fat globule membrane antigen (HMFG-2), also have been found. 15,18 An unusual variant of lipid-rich mammary carcinoma composed of a dual-cell population, where the more aggressive cell, which resembled an altered myoepithelial cell, was responsible for the lipid production, has also been described. 6 In our study, the tumor cells did not express the myoepithelial cell markers tested. 19
The presence of local eosinophilia has been described in canine mammary carcinoma 7 and in a number of human solid epithelial tumors and, rarely, in undifferentiated mesenchymal tumors. 8 Several theories have been proposed to explain this condition, e.g., that the tumor itself may produce a eosinophilic chemotactic factor 4,7 or an immune response, possibly to specific tumor types or to the by-products of tumor necrosis. 7
The usual form of presentation of canine lipid-rich carcinoma was that of a single large tumor mass affecting one mammary gland (dog nos. 1, 3, 5, 6, and 7) or two adjacent mammary glands (dog nos. 2 and 4). Six of seven animals were purebred, and three were Rottweilers. Age ranged between 4 and 13 years. Reproductive data indicated that six of seven dogs were intact females, five had previous pregnancies, two had pseudopregnancies, and none of the dogs had been given oral contraceptives. Postsurgery follow-up showed recurrence or metastases (or both) in four of the seven cases. Thus, dog no. 1 had recurrence, regional lymph node, and distant metastases confirmed by postmortem and microscopic examination 4 months after initial surgery. Dog no. 2 presented several recurrences in the months following the first surgery, and metastases to the right superficial inguinal lymph node were confirmed by microscopic examination. Dog no. 4 had lung metastases and probable metastases to the deep inguinal lymph nodes, as revealed by X-ray examination 16 months after surgery. Dog no. 6 had another primary tumor in R3 and metastases to the right superficial lymph node within 3 months of surgery, also confirmed by microscopic examination. Dog no. 7 had a micrometastasis to the right superficial lymph node but has been free of disease for 8 months. On the contrary, dog no. 5 was free of disease for 14 months but was euthanatized 33 months after surgery because of a primary bone tumor in the proximal metaphysis of the left tibia and lung metastases (X-ray), whereas dog no. 3 has been free of disease for 24 months.
Lipid-rich carcinoma is considered a rare variant of infiltrating duct carcinoma of the human breast, 13 the estimated frequency of which is low (0.2–1.7%). 2,5,12,13,17 There have been a number of reports on the occurrence of this type of tumor in Japanese women, 1,5,6,14,15,16 but race prevalence is not addressed in literature. In our study, three of seven dogs were Rottweiler, which points to a possible breed predisposition, as suggested in humans. As in the cases in this study, a wide age range has been observed from childhood 3 to the eighth decade of life. 5 Lipid-secreting carcinomas have aggressive clinical courses in women 2,12,14 and lack estrogen receptor alpha and progesterone receptor, 5,18 as in the cases in our study.
In conclusion, our results show that lipid-rich carcinomas of the mammary gland in dogs are aggressive, poorly differentiated neoplasm that lack estrogen and progesterone receptors and give rise to local recurrence and proximal and distal metastases.
Footnotes
Acknowledgements
This work was supported by grants PI1999/I55 (Consejería de Educación, Cultura y Deportes, Gobierno de Canarias, Spain) and CVI-805 (Plan Andaluz de Investigación, Consejería de Educación y Ciencia, Junta de Andalucía, Spain).
