Abstract
Mucinous carcinoma of the mammary gland is a rare tumor characterized by excessive mucin production. In human and canine pathology, the diagnosis of mucinous carcinoma is based on the demonstration of an epithelial phenotype of mucus-producing cells and periodic acid-Schiff (PAS)-diastase positivity of the mucin. The histologic and immunohistologic characteristics of feline mucinous mammary carcinoma were examined. Of 656 cases of feline mammary neoplasms and dysplasias, 3.2% were found to be mucin-producing tumors. Cytokeratin 19 (16 cases positive, 4 heterogenous, and 1 negative) and vimentin (15 cases positive, 2 heterogenous, and 4 negative) expression were examined, and the mucin produced was alcian blue positive. PAS-diastase staining was variable (38.1%). Based on these findings, mucinous mammary carcinoma in the cat varies significantly from the human and canine varieties and alcian blue is the prominent stain in the diagnosis of feline mucinous carcinoma.
Introduction
Mammary mucinous carcinomas are tumors of the mammary gland characterized by abundant mucin production. In the human, mucinous carcinoma is to be considered pure when no other histotype than the mucinous is present and nonpure when mucinous is associated with other histotypes of carcinoma. 14 These are rarely reported in the feline veterinary literature. Previous case reports have indicated the following characteristics: 1 case showed extensive mucin production and adenoid cystic pattern; 4 cases were anti-RCK-102 antibody positive (specificity for human keratin 5 + 8), 3 of 4 were positive to anti-NCL-5D3 antibody (specificity for human keratin 8 + 18 + 19), 3 of 4 were also positive to vimentin and actin. 6, 9 Misdorp et al. 12 defined mucinous carcinoma as “characterized by abundant mucin production, and these rare tumors occur in the dog and the cat and may be of simple or complex type.” Misdorp 10 asserted that the predominant feature is the presence of large amounts of mucinous material that stains positive to periodic acid–schiff (PAS), with and without diastase, and alcian blue stains.
In the case of human and canine mucinous carcinoma, a diagnosis is considered definitive after the demonstration of an epithelial phenotype, mucus production, and PAS-diastase positivity. 1– 4, 11 Histologically, 3 subtypes have been reported in dogs and humans: mucinous (colloid) carcinoma, signet-ring-cell carcinoma, and secretory carcinoma. 1, 2, 7 In human medicine, its origin from neuroendocrine cells is still debated. In the dog, only a few cases proved to be of neuroendocrine origin. 1
To date, little is known about the morphology and immunohistochemical features of this type of tumor in the cat. The aim of the present paper was to study the histomorphologic features of mucinous carcinomas in the cat and to find a diagnostic confirmation by histochemical and immunohistochemical stains.
Materials and Methods
Six hundred and fifty-six feline mammary glands were examined from the files of the Department of Veterinary Public Health and Animal Pathology and consisted in completely excised samples. Of these, 21 cases (3.2%) of mucinous carcinomas were identified. Histologic classification was carried out on hematoxylin and eosin (HE) stained slides according to Misdorp et al. 12 The tumors were then classified according to Toikkanen and Kujari. 14 Breed, age, and survival data were obtained from referring clinicians. Survival data were based on a 1-year postoperative period.
The histochemical features of the mucus were assessed by means of PAS, PAS-diastase, alcian blue pH 2.5 and pH 1.0, and Grimelius stains applied to serial 5-μm sections. As positive controls for mucin, feline intestinal and bronchial mucosa were used and the pancreas (islet cells) were used for Grimelius stain.
The sections were dewaxed in toluene and rehydrated. Endogenous peroxidase was blocked by immersion for 30 minutes in methanol that contained 3% hydrogen peroxide. The sections were then rinsed in Tris buffer, immersed in citrate buffer (2.1 g citric acid monohydrate/liter distilled water), pH 6.0, heated for four 5-minute periods in a microwave oven at 750 W, and allowed to cool at room temperature (approximately 20 minutes.). The primary antibodies (Dako, Glostrup, Denmark) used consisted of monoclonal antibodies against cytokeratin 19 (clone BA 17, diluted 1 in 40), anticytokeratin (clone AE1/AE3, diluted 1 in 50), antismooth muscle α-actin (clone 1 A4, diluted 1 in 100) and anti-vimentin (clone V9, diluted 1 in 40), and polyclonal antibody against chromogranin A (diluted 1 in 100). All antibodies were incubated with the sections overnight at 4°C and were followed by a commercial streptavidin-biotin-peroxidase kit (LSAB kit, Dako). Sections of normal feline mammary gland, muscular intestinal wall, tendon, and pancreas (pancreatic islets) were used as positive controls for anticytokeratin 19, antismooth muscle actin, antivimentin, and antichromogranin A antibodies, respectively. For negative controls, an unrelated antibody of the same isotype and a similar protein concentration were used instead of the primary antibody.
Results
The breed, age, and histologic classification of cases are displayed in Table 1. The majority of cases (16/21) were Domestic Shorthair cats. The average age was 12.14 years.
Breed, age, morphology, histochemistry and immunohistochemical results, and survival of the 21 cases of mucinous carcinoma.
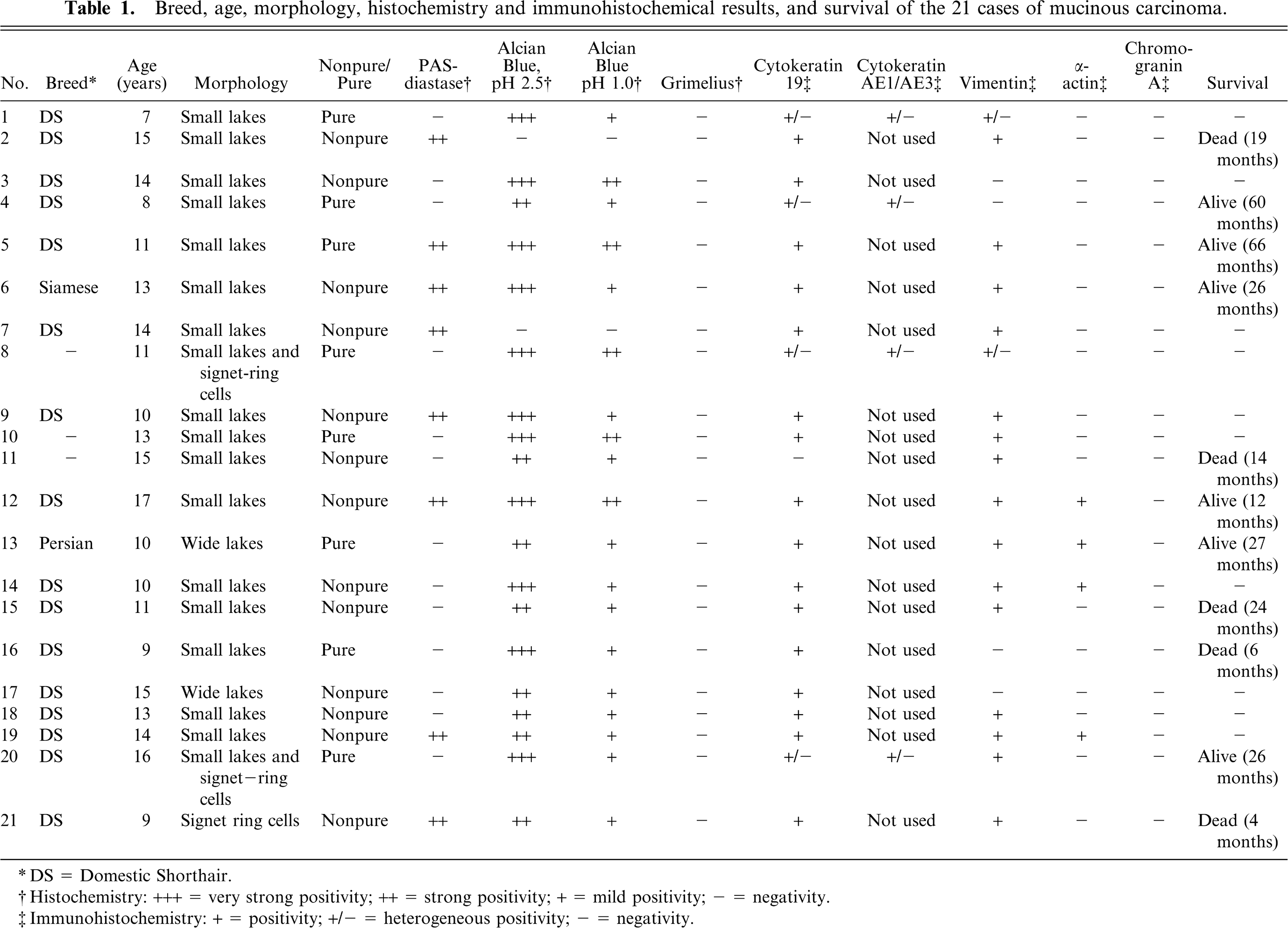
∗ DS = Domestic Shorthair.
†Histochemistry: +++ = very strong positivity; ++ = strong positivity; + = mild positivity; − = negativity.
‡Immunohistochemistry: + = positivity; +/− = heterogeneous positivity; − = negativity.
Histology
With routine HE staining, the most striking morphologic feature was the presence of mucus, which was basophilic to amphophilic when extracellular, or slightly eosinophilic when intracellular. Two subtypes of feline mucinous carcinomas were noted: pure mucinous (8/21) and nonpure (13/21), in which mucinous carcinoma coexisted with a different type of carcinoma. In most cases (18/21), the mucus was extracellular and formed irregularly confluent small lakes lined by neoplastic cells (Fig. 1); of the 18 cases, 2 showed the presence of occasional signet-ring–shaped cells (Fig. 2). Two of the remaining 3 cases were characterized by wide lakes of extracellular mucin (Fig. 3), with few cellular elements forming papillae, and one had a signet-ring-cell intracellular pattern, in which the nucleus was eccentric. Necrosis was present in the center of the nodules in 17 cases.
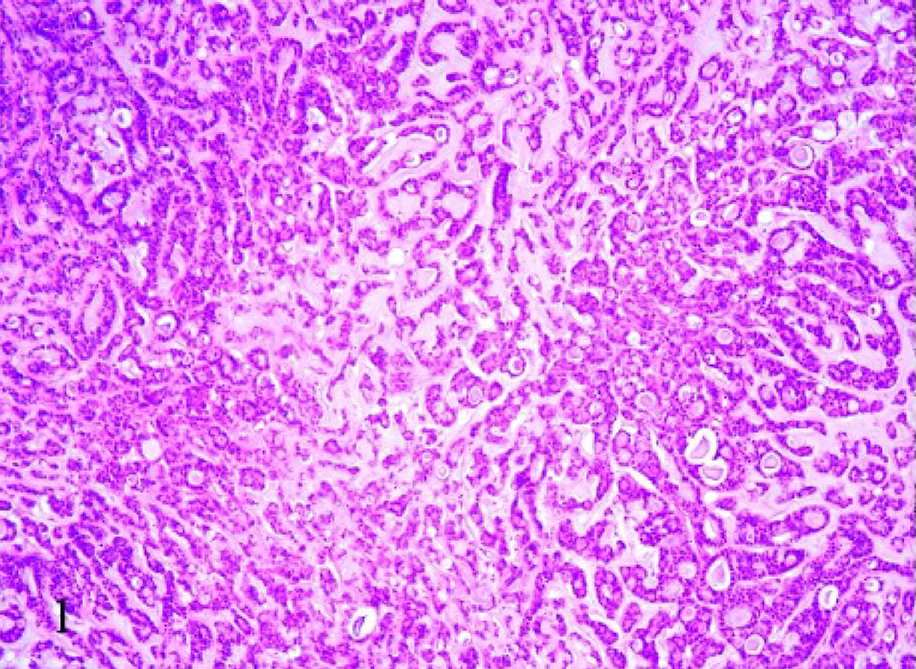
Mammary tissue; cat. Mucinous carcinoma with extracellular mucin. Pattern with confluent small lakes lined by neoplastic cells. HE, 10×.
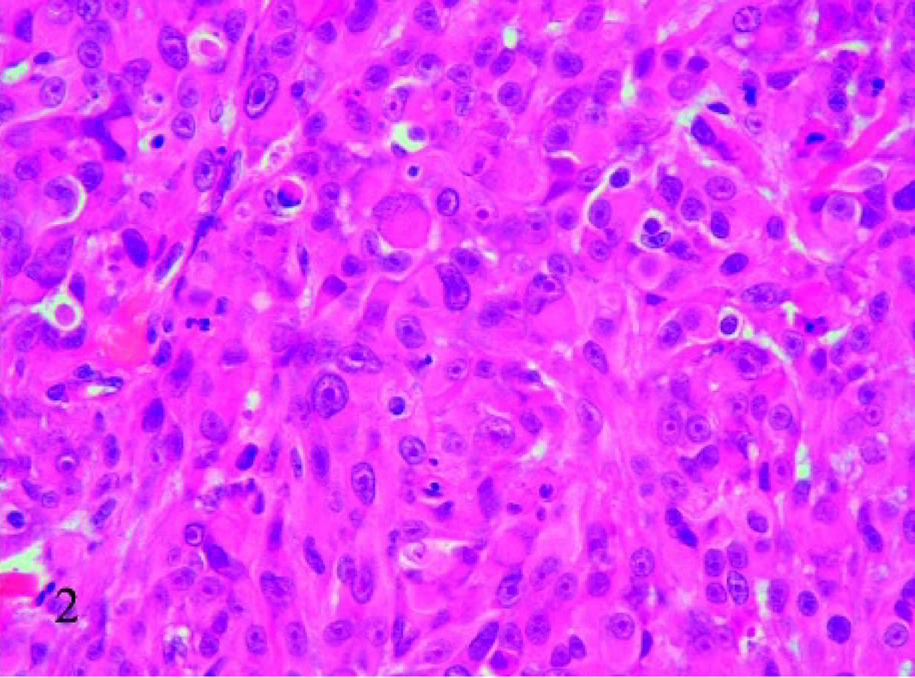
Mammary tissue; cat. Mucinous carcinoma with signet-ring cells. The nucleus of some cells exhibits an eccentric location because of the presence of intracellular mucin. HE, 40×.
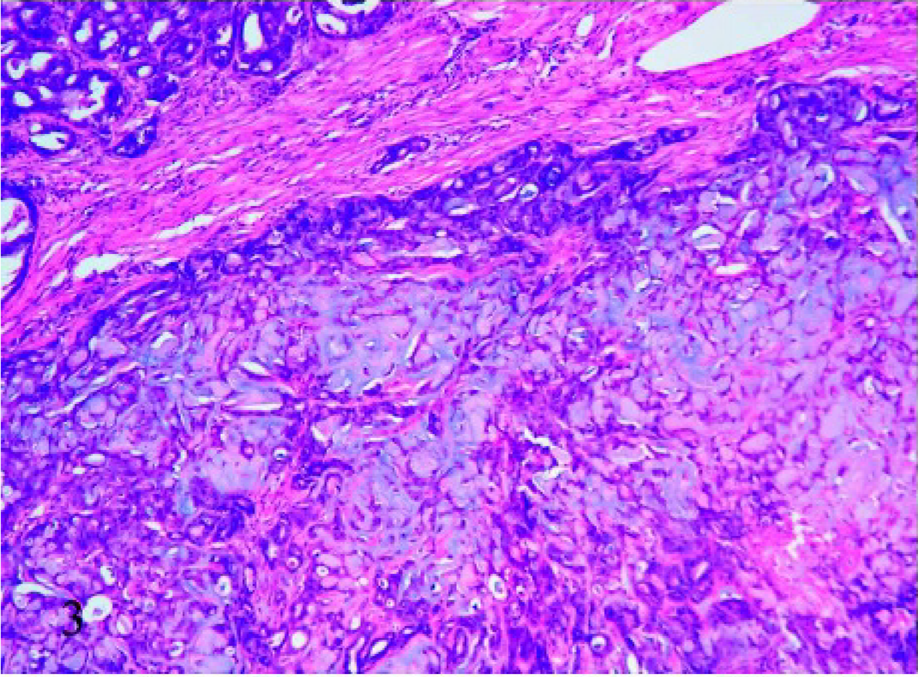
Mammary tissue; cat. Mucinous carcinoma characterized by dispersed cells in wide mucin lakes. HE, 10×.
Mucin characterization
Of 21 cases of mucinous mammary gland tumors, 8 were PAS-diastase positive (Fig. 4) and 13 were negative. Regarding alcian blue stain, the reaction was stronger at pH 2.5 (Fig. 5) than 1.0 in 19 cases; in particular, the PAS-diastase negative cases showed strong positivity to alcian blue at pH 2.5, and the 2 cases negative to alcian blue were strong positive to PAS-diastase.
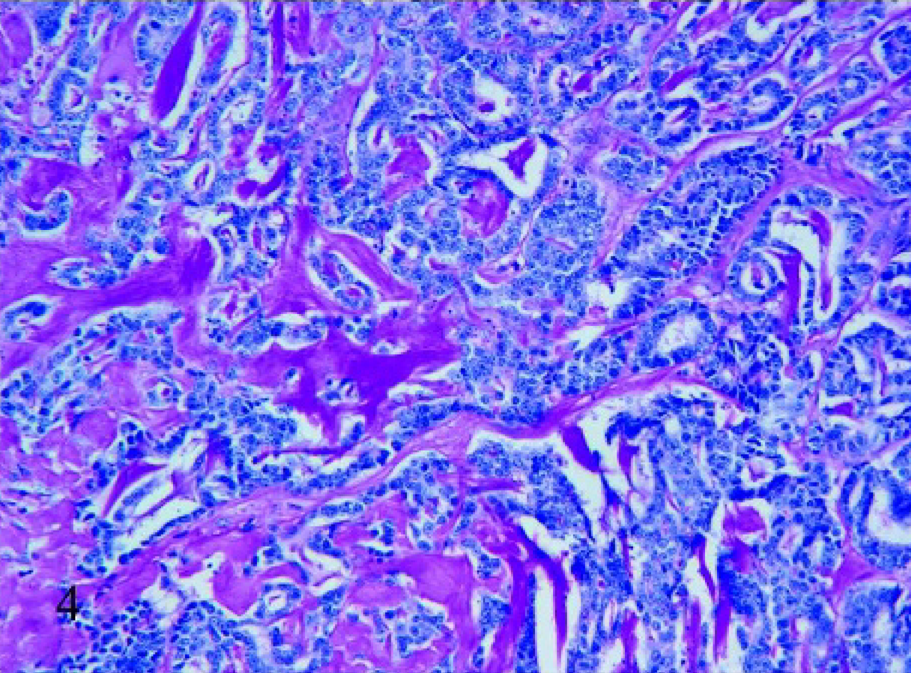
Mammary tissue; cat. Mucinous carcinoma with a small lakes pattern. Positive PAS-diastase staining of the mucin, 20×.
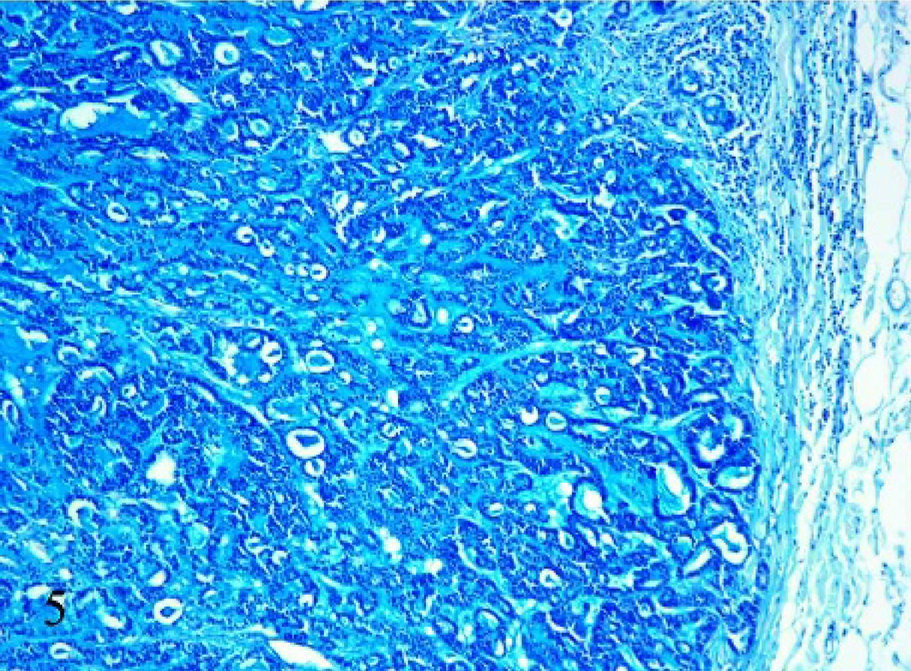
Mammary tissue; cat. Mucinous carcinoma with a small lakes pattern. Positive reaction to alcian blue pH 2.5. 10×.
Cell phenotype
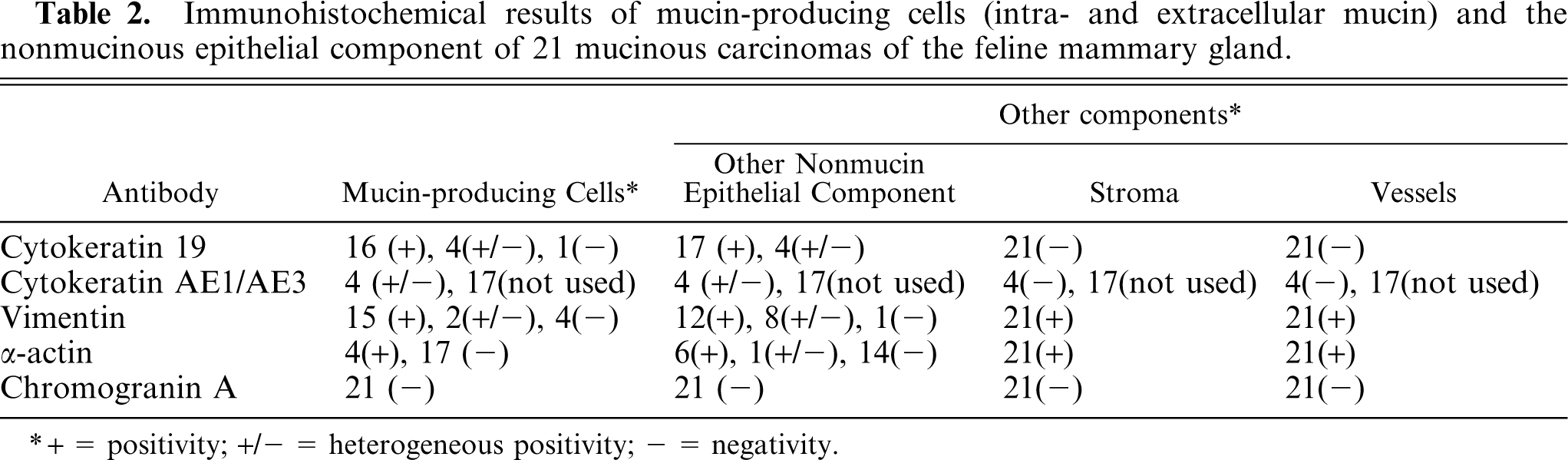
The positivity to cytokeratin 19 confirmed the diagnosis of malignant tumors with an epithelial phenotype in all cases. The histochemical and immunohistochemical results are summarized in Tables 1 and 2.
Immunohistochemical results of mucin-producing cells (intra- and extracellular mucin) and the nonmucinous epithelial component of 21 mucinous carcinomas of the feline mammary gland.
∗ + = positivity; +/− = heterogeneous positivity; − = negativity.
Mucin-producing cells (intra- and extracellular mucin)
Mucin-producing cells showed an epithelial phenotype (i.e., cytokeratin 19 expression in the cytoplasm) in 16 of 21 cases (Fig. 6). In 4 cases, heterogenous (areas of the same tumor stain either positive or negative) cytokeratin 19 expression was confirmed by the same result with anticytokeratin AE1-AE3 antibody. One case was negative to cytokeratin 19. However, in all cases, reduction of cytokeratin 19 expression paralleled the increase in extracellular mucus collection. In 15 samples, cells were strongly vimentin positive (Fig. 7), 2 cases were heterogenous, and 4 were negative. Thirteen cases with strong positivity to vimentin were also cytokeratin 19 positive. Three tumors were negative or heterogenous to both vimentin and cytokeratin 19. The α-actin monoclonal antibody produced positive results in 4 cases (Fig. 8) in which the cells were also positive for vimentin and cytokeratin 19. All cases were Grimelius and chromogranin A negative.
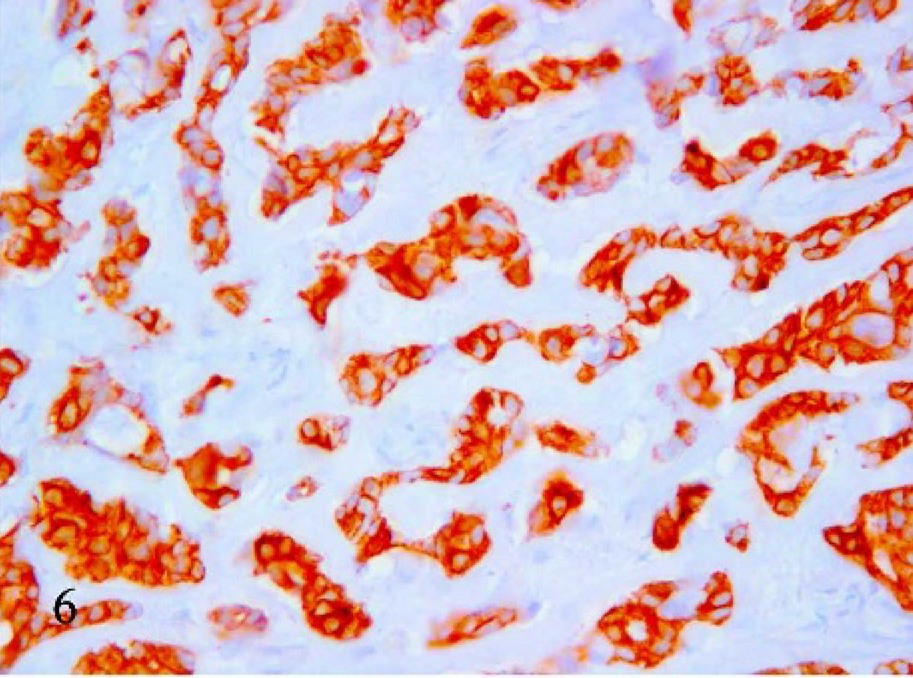
Mammary tissue; cat. Mucinous carcinoma. Cytokeratin 19 immunohistochemical positivity of the mucus-producing cells. 40×.
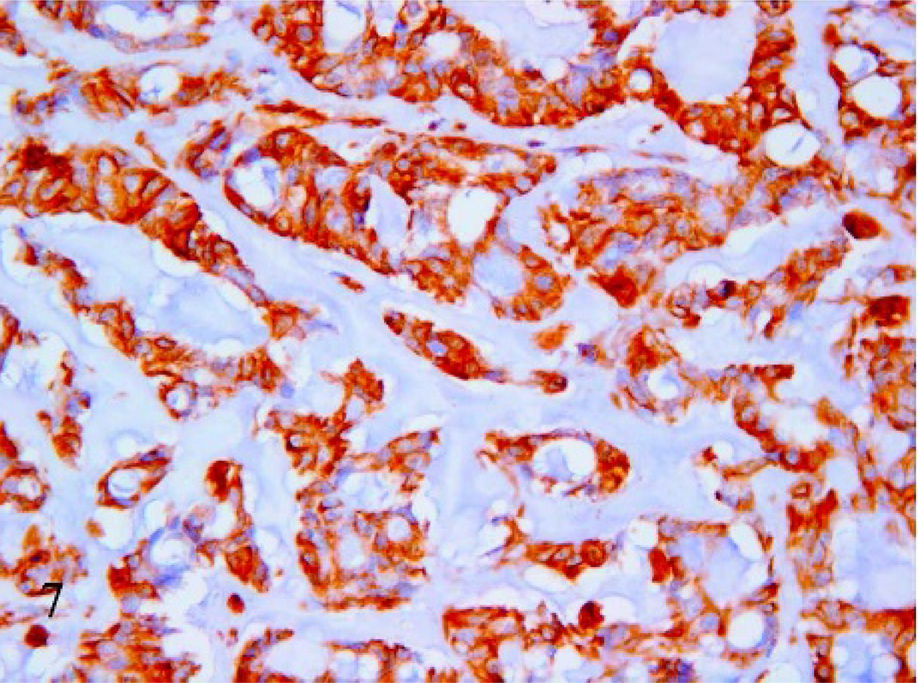
Mammary tissue; cat. Mucinous carcinoma. Immunohistochemical reaction to vimentin. Positive labeling of the mucus-producing cells. 40×.
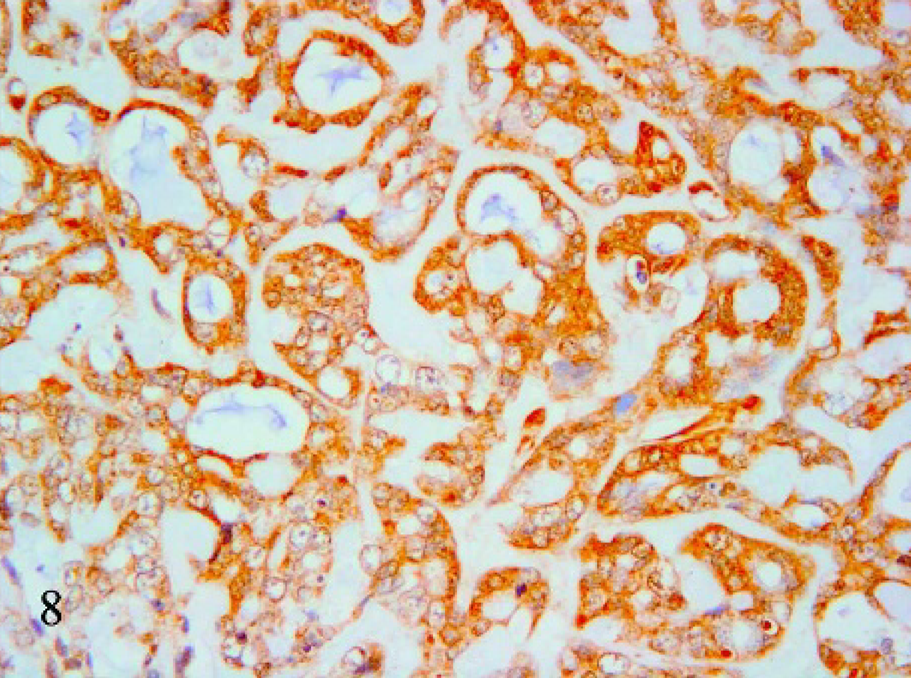
Mammary tissue; cat. Mucinous carcinoma. α-actin immunohistochemical positivity of the mucin-producing cells. 40×.
Nonmucinous epithelial component
In all cases, nonmucin-producing epithelial components were present, as well as the other carcinomatous counterparts in nonpure forms and also dysplastic lesions at the periphery of the tumors. These epithelial/carcinomatous cells were positive to cytokeratin 19 in 17 of 21 cases, the remaining 4 cases showed heterogenous positivity. The carcinomatous nonmucinous counterparts in nonpure tumors were positive to vimentin in 12 of 21 cases, 8 cases were heterogenous, and 1 was negative. In 11 cases, the cells were positive to both cytokeratin 19 and vimentin and, in 2 cases, heterogenous result to both antibodies. In 6 of 21 cases, the cells were α-actin positive, 1 case was heterogenous, and the remaining were negative. All the 6 α-actin positive cases were also cytokeratin 19 and vimentin positive. All nonmucinous epithelial cells were chromogranin A negative.
Stroma
Fibroblasts of the stroma were negative to cytokeratin 19, AE1/AE3, and chromogranin A when used; stromal cells were always positive for vimentin and α-actin antibodies.
Vessels
The vessel cells (endothelial and myocytes of the vascular wall) showed the same immunoreactivity as the stroma; they were negative to cytokeratin 19, AE1/AE3, and chromogranin A antibodies, and positive to vimentin and α-actin antibodies.
Discussion
The percentage of mucinous carcinoma (3.2%) assessed in this study on feline mammary tumors was similar to the data reported in humans (1.0–6.0%) but higher than in the dog (0.3%). 1, 8 The prevalent breed was Domestic Shorthair cats, and the average age was 12.14 years. The morphology of tumors was predominantly of extracellular mucus-forming small lakes, which, by confluence, gave origin to wide mucin lakes. Nonpure forms were more numerous than pure forms.
Mucin varied from that characterized in bronchial and intestinal positive controls in which PAS-diastase and alcian blue at pH 2.5 positivity and alcian blue at pH 1.0 negativity predominated. On the contrary, in feline mammary mucinous carcinoma, more than 50% of the cases were PAS-diastase negative (because of the lack of neutral mucopolysaccharides). In addition, mucinous carcinomas maintained their alcian blue positivity (stronger at pH 2.5 than 1.0), which is indicative of the presence of acid mucopolysaccharides (more carboxylated than sulphated). 15, 16 Only 2 cases negative to alcian blue were PAS-diastase positive. Therefore, on the basis of the results obtained, alcian blue is the prominent stain in the diagnosis of feline mucinous carcinoma. In all cases, the tumors had an epithelial pattern, based on cellular morphology, besides cytokeratin 19 positivity of mucin-immersed cells or nonmucinous cells of mucinous carcinoma.
The use of cytokeratin AE1/AE3 (clone AE1 immunoreact with cytokeratin 10, 13, 14, 15, 16, and 19; clone AE3 with cytokeratin 1, 2, 3, 4, 5, 6, 7, and 8) did not produce any useful results with respect to cytokeratin 19 alone, which suggests only glandular epithelium. The only case with mucin-immersed cells negative to cytokeratin 19 had the same vimentin-positive and actin-negative cells; but the other nonmucin epithelial cells were positive to cytokeratin 19, which confirmed a true cytokeratin 19 negativity in the mucinous counterpart.
In 15 cases, the mucin-producing cells were vimentin positive, and 13 of these were also cytokeratin 19 positive. A true double pattern (epithelial/mesenchymal) was not demonstrable given the ability of feline mammary carcinomas to express vimentin, as well as cytokeratin. 9 Similarly, epithelial cells of human-breast carcinoma can express vimentin (18% of the cases reported by Niveditha and Bajaj; 13 13% reported by Martin de las Mulas et al. 9 ). In feline mammary tumors, Martin de las Mulas et al. 9 reported 76% of cases with luminal vimentin-positive epithelial-like cells, in our study, 81% (15 positive and 2 inconstant). Vimentin immunoreactivity in epithelial tumors may be attributed to an aberrant expression or reversion to a more primitive or embryonic cell type and may evidence rapidly proliferating cells, associated with a poor prognosis. 5
Four of 5 cats that died expressed vimentin. Vimentin expression in human breast carcinomas is an indicator of malignancy; whereas the study cohort available in this study is too small to draw a definite conclusion, it could be that vimentin similarly indicates malignancy. 13 It is well known that feline mammary tumors express vimentin and that most of them are malignant. 9
Because epithelial cells of feline mammary carcinoma express vimentin, a panel of antibodies (including actin) was used to be tested to discriminate for mesenchymal cells. 9 Actin expression was lower than vimentin in the mucin-producing cells of feline mucinous carcinomas (4 positive and 17 negative); however, it was not a discriminant of mesenchymal cells, because all 4 positive cases were also cytokeratin 19 and vimentin positive. It is likely that this filament, as well as vimentin, is easily expressed by epithelial cells of the feline mammary gland.
In conclusion, in terms of histochemical features, mucinous mammary carcinoma in the cat is not always rich in neutral mucin, but acid mucin is almost always present. The mucus-producing cells showed an epithelial pattern both for the morphology and for its positivity to cytokeratin 19 in most cases, even though many of them coexpress vimentin and some actin as well. The neuroendocrine origin of these tumors was not demonstrated in the cases included in this study. Based on these findings, mucinous mammary carcinoma in the cat varies significantly from the human and canine varieties, and alcian blue is the prominent stain in the diagnosis of feline mucinous carcinoma.
