Abstract
In breast cancer of women, the estrogen receptor–α (ERα) and progesterone receptor (PR) status has prognostic and therapeutic significance. The aim of this study was (1) to characterize by immunohistochemistry the expression of ERα and PR in nonneoplastic and neoplastic mammary gland tissue of pet rabbits and (2) to correlate the ERα/PR status and histological features. All 124 rabbits included in this study had a mammary tumor; in addition, 2 rabbits had lobular hyperplasia and 25 had multiple cysts. Of the 124 neoplasms, 119 (96%) were carcinoma, 2 (2%) were carcinoma in situ, and 3 (2%) were adenoma. ERα or PR or both were detected in 2 of 2 carcinomas in situ, 3 of 3 adenomas, 19 of 25 cysts, and 2 of 2 lesions of lobular hyperplasia. Most carcinomas (75/119, 63%) were negative for both ERα and PR; 22 of 119 carcinomas (18%) were double-immunopositive. The ERα and PR expression was not influenced by histotype or histological tumor grade. In carcinomas, there was a statistically significant correlation between increased mitotic count and reduced expression of ERα and PR, and the mitotic count was higher in double-immunonegative carcinomas (75/119). The findings suggest that in rabbit mammary carcinomas, proliferative activity is mainly influenced by factors other than estrogen and progesterone and provides the basis for future investigations into the prognostic significance of the ERα and PR status of mammary tumors.
Keywords
Neoplastic diseases are important causes of morbidity and mortality in pet rabbits. Compared to uterine carcinoma, 1,30,31 mammary gland neoplasms are less frequently diagnosed, and most are carcinomas. 4,28
So far, predictive markers for the biological behavior of mammary carcinomas in pet or laboratory rabbits have not been identified.
Estrogen and progesterone control the postnatal development of the mammary gland; 16,17 they contribute to mammary tumors in women 8,17 and most likely also in bitches or queens. 12 In humans, the estrogen receptor–α (ERα) and progesterone receptor (PR) status of breast carcinomas is routinely analyzed through standardized procedures and used as a prognostic indicator to decide the most appropriate treatment. 10,14 In bitches, malignant mammary tumors have a lower ERα and PR expression than benign neoplasms, hyperplastic/dysplastic lesions, and unaltered mammary tissue. 9,19,21,23,27 A consensus on the determination of ERα and PR in canine mammary carcinomas has been published. 24 Immunohistochemistry (IHC), however, is often not routinely included in the diagnostic workup of these tumors, 24 and its prognostic value remains controversial. 9,12,19
To our knowledge, there is no comprehensive study into the expression of ERα and PR in rabbit mammary gland tumors. The objective of this study was to characterize the ERα and PR expression in nonneoplastic and neoplastic mammary gland tissue of 124 pet rabbits. Results of this investigation will add to the basic knowledge on pathophysiological features of normal, hyperplastic/dysplastic, and neoplastic mammary gland tissue of pet rabbits. Furthermore, they will provide the basis for future investigations into the prognostic significance of the ERα and PR status in mammary tumors in this species.
Materials and Methods
This retrospective study (2007–2015) investigated mammary tumors, mammary tissue with multiple cysts and/or lobular hyperplasia, and adjacent normal mammary tissue of 124 pet rabbits by histopathology and immunohistochemistry for the detection of ERα and PR. The investigated cases were selected from the archives of 2 diagnostic laboratories. They represented all proliferative mammary gland lesions in pet rabbits regardless of the reported sex that were submitted from 2007 to 2014 to Laboklin (Bad Kissingen, Germany) and all cases of such lesions that were submitted in 2015 to the Institute of Veterinary Pathology of the University of Leipzig (Leipzig, Germany).
All mammary samples were surgical specimens that had been excised in veterinary practices and immediately fixed in 10% buffered formalin for 24 to 72 hours. After embedding in paraffin wax, tissues were routinely processed, sectioned at 3 μm, and stained with hematoxylin and eosin (HE). From 3 of these cases (rabbit Nos. 39, 23, and 94), tissue samples stored in formalin were available. These were used for cryostat sections containing unaltered mammary parenchyma and neoplastic tissue that were stained with Sudan red.
Histopathology
The classification of mammary neoplasia, hyperplasia, and dysplasia, as well as the further histopathological characterization of the mammary gland carcinomas, was performed as previously described. 4,28 The histotype of the mammary carcinomas was determined according to the predominant growth pattern. 12,22 In regard to all carcinomas (n = 119), a scoring system was applied to evaluate the tumor invasion into the stroma and the adjacent tissue, the presence of necrosis within the tumor, and the secretory activity of the tumor. 28
The latter was determined separately for the amount of proteinaceous secretion and the presence of vacuolated neoplastic cells. The degree of tissue invasion (mild, moderate, or marked) was evaluated for the tumor stroma and the peritumoral nonneoplastic tissue. 28 Necrosis and secretory activity were determined as minimal, mild, moderate, or marked (ie, if <10%, 10%–39%, 40%–69%, or ≥70% of the tumor area was involved, respectively). 28 The number of mitotic figures (MFs) in 10 high-power fields (HPFs) was counted using the 40× objective of the Olympus CH30 microscope (diameter of the 400× field: 159 μm2; Olympus, Hamburg, Germany). 20
The 119 mammary carcinomas were graded according to the modified method of Elston and Ellis. 11,29 This method scored the following 3 parameters: the percentage of tumor area with a tubular growth pattern (score 1: >75%; score 2: 10%–75%; score 3: <10%), the degree of nuclear pleomorphism (score 1: mild; score 2: moderate; score 3: marked; Suppl. Figs. S1–S3), and the mitotic count in neoplastic cells under consideration of the field diameter of the microscope (MFs in 10 HPFs: score 1: ≤9 MFs; score 2: 10–19; score 3: ≥20). 10 Individual scores were added to give a total differentiation score ranging from 3 to 9 that determined the histological tumor grade: grade I (well differentiated: scores 3–5), grade II (moderately differentiated: scores 6–7), and grade III (poorly differentiated: scores 8–9). 11
Immunohistochemistry
Immunostaining for the detection of ERα and PR was applied to tissue samples of all rabbits (n = 124). The peroxidase antiperoxidase (PAP) method with 3,3′-diaminobenzidine tetrahydrochloride (DAB) as chromogen was applied. Immunohistochemistry was performed manually by the use of disposable immunostaining chambers (Shandon coverplate; Thermo Scientific Fisher, Waltham, MA). The following primary antibodies were used: mouse anti–human ERα (1:20, clone 6F11, citrate buffer pretreatment; Novocastra Laboratories, Newcastle upon Tyne, UK) and mouse anti–human PR (1:100, clones 16 and SAN 27, citrate buffer pretreatment; Novocastra Laboratories). Immunostaining for pan-cytokeratin (1:50, citrate buffer pretreatment, clone AE1/AE3; DAKO, Glostrup, Denmark) was applied to confirm the general immunoreactivity of the investigated tissue samples. In detail, tissue sections were dewaxed and rehydrated. To block the endogenous peroxidase, sections were treated with 3% H2O2 in methanol for 30 minutes at room temperature (RT). For antigen retrieval, sections were heated at 95°C in citrate buffer (10 mM, pH 6) for 25 minutes and remained 17.5 minutes in the buffer. Subsequently, sections were incubated with the primary antibodies or a nonrelated isotype-matched antibody (negative control) overnight at 4°C. 26 After washing in Tris-buffered saline (TBS), rat anti–mouse immunoglobulin G (1:100; Dianova, Hamburg, Germany) was applied for 30 minutes at RT. The sections were washed in TBS before applying the mouse-PAP-complex (1:100; Dianova) for 30 minutes at RT. Antibodies and the PAP-complex were diluted in TBS containing 1% bovine serum albumin. Rabbit uterus was used as positive control (Fig. 1). 1,30
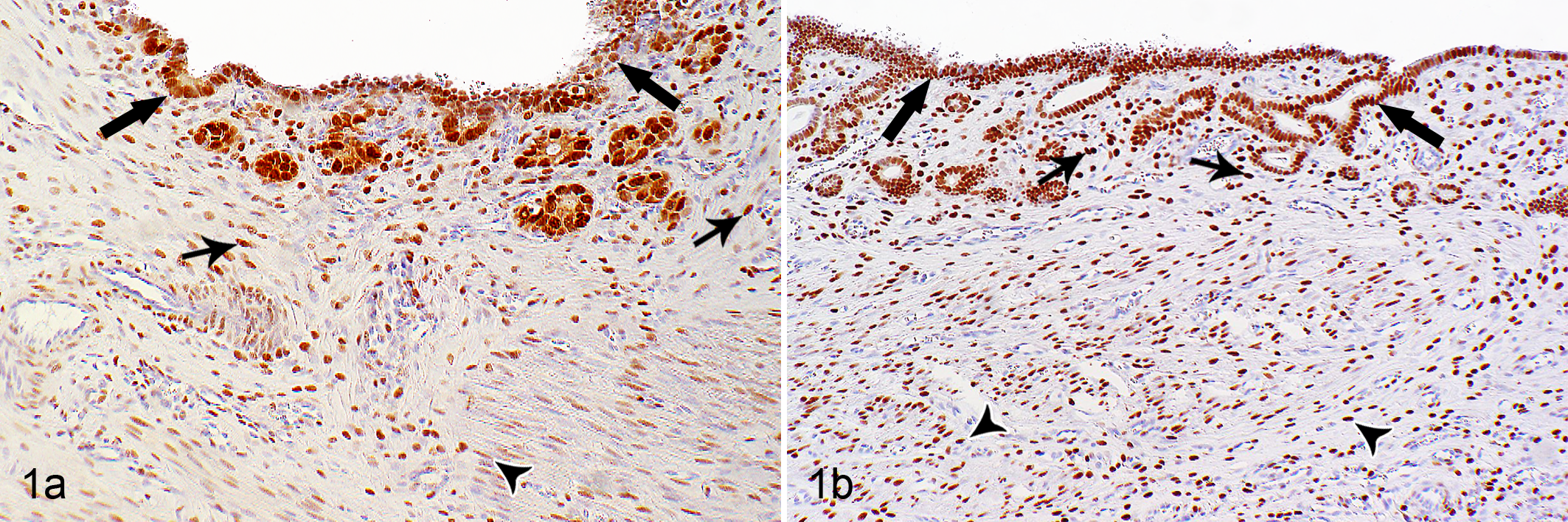
Normal uterus, rabbit. (a) Strong nuclear immunostaining for estrogen receptor–α and (b) progesterone receptor in luminal and glandular epithelial cells (thick arrows), stromal cells (thin arrows), and smooth muscle cells (arrowheads).
In regard to the nonneoplastic and neoplastic mammary lesions, numbers of ERα- and PR-immunopositive cells were quantified by image analysis using the Olympus DP12 camera and the Soft Imaging System 5 (Olympus). For each nonneoplastic and neoplastic mammary lesion, digital images were taken from 3 representative areas (S Plan Apo 40× objective, Olympus BH2 microscope) that did not contain foci of tissue necrosis. In these areas, all neoplastic epithelial cells were evaluated for their ERα or PR immunostaining (using the count tool of Adobe Photoshop CS5.1; Adobe, San Jose, CA) and the percentages of immunopositive and immunonegative cells were calculated; the numbers of examined neoplastic epithelial cells per tumor varied between 298 and 1538 cells depending on the size and cellular density of the tumor. The intensity of the ERα and PR immunostaining was further classified as mild, moderate, and marked. These data on percentage and intensity were used to score the ERα and PR immunolabeling. This was performed by the use of 2 different scores: the immunoreactive score (IRS) 2 and the H-score. 10 The IRS was calculated as [(weakly positive cells) + (moderately positive cells × 5) + (strongly positive cells × 10) / 100]. 2 The H-score was calculated as (weakly positive cells) + (moderately positive cells × 2) + (strongly positive cells × 3). 10
Statistical Analysis
The statistical analysis was carried out using R-Studio software (version 0.97.248; R version 3.0.0; RStudio, Boston, MA). The linear mixed-effects model was implemented by regression analyses and single-stratum analysis of variance. Furthermore, analysis of variance (ANOVA) was used to determine the difference between the different fitted model objects. In this study, the fitted model objects were defined as follows: the sex of the rabbits (female, female spayed), the 3 categories of the tubular growth pattern (<10%, 10%–75%, >75%), the histologic tumor grade of the carcinomas (grade I, II, or III), and the total differentiation score (3–9) of the carcinomas. For these investigations, Tukey’s test was used as post hoc test. For the investigation of a nonlinear relationship between ERα and the mitotic count as well as PR and the mitotic count, the Spearman’s rank correlation test (Spearman’s ρ) was applied. To determine the statistical differences between the 2 groups of ERα- and PR-positive (double-positive) carcinomas and ERα- and PR-negative (double-negative) carcinomas, the Mann-Whitney-Wilcoxon test was used.
Results with P < .05 were considered statistically significant.
Results
For 59 of 124 pet rabbits, more detailed information about the breed was available: dwarf (n = 35), dwarf lop (n = 7), Lionhead (n = 7), lop (n = 6), Angora (n = 1), Rex (n = 1), Teutoburger (n = 1), and Netherland dwarf (n = 1). Of 112 rabbits of known sex, 88 (79%) were female, 24 (21%) female spayed, and none male. Of the 101 rabbits of known age, the ages ranged from 1.5 to 10 years (mean, 5.3 years). Per clinical history, 6 of 124 rabbits (5%) also had a uterine neoplasm: carcinoma (n = 4), leiomyoma (n = 1), or leiomyosarcoma (n = 1). Follow-up clinical data were available for 15 rabbits with a mammary carcinoma (rabbit Nos. 103–106, 108, 110–119). Of these, 6 developed 1 (rabbit Nos. 103, 110, 111, 114) to several (rabbit Nos. 111, 118) additional mammary masses, and 3 (rabbit Nos. 103, 108, 111) showed nodular masses in the lungs that were diagnosed by radiology. Histological examination was only performed on the mammary mass of rabbit No. 117 and revealed a mammary adenoma.
All 124 rabbits included in this study had a mammary tumor; in addition, 2 rabbits had lobular hyperplasia and 25 had multiple cysts in the adjacent mammary parenchyma. Most of the 124 tumors were carcinomas (n = 119, 96%), whereas carcinoma in situ (n = 2, 1.6%) and adenoma (n = 3, 2.4%) were rare. Notably, no mesenchymal tumor was diagnosed.
In 28 of 124 rabbits (23%), the concurrent presence of 2 or more mammary lesions was observed. Multiple mammary cysts were detected in 25 of 124 rabbits (20%) with a mammary carcinoma. In addition, the concurrent presence of carcinoma and adenoma (n = 1), adenoma and lobular hyperplasia (n = 1), and carcinoma, adenoma, and lobular hyperplasia (n = 1) was observed. Five animals with mammary carcinoma and 1 with cystic adenoma also had a reported uterine neoplasm.
The histopathological and immunohistochemical findings of the mammary proliferative lesions are provided in Tables 1 and 2 and Supplemental Tables S1 to S5.
Mitotic Counts in Proliferative Mammary Lesions of 124 Pet Rabbits.
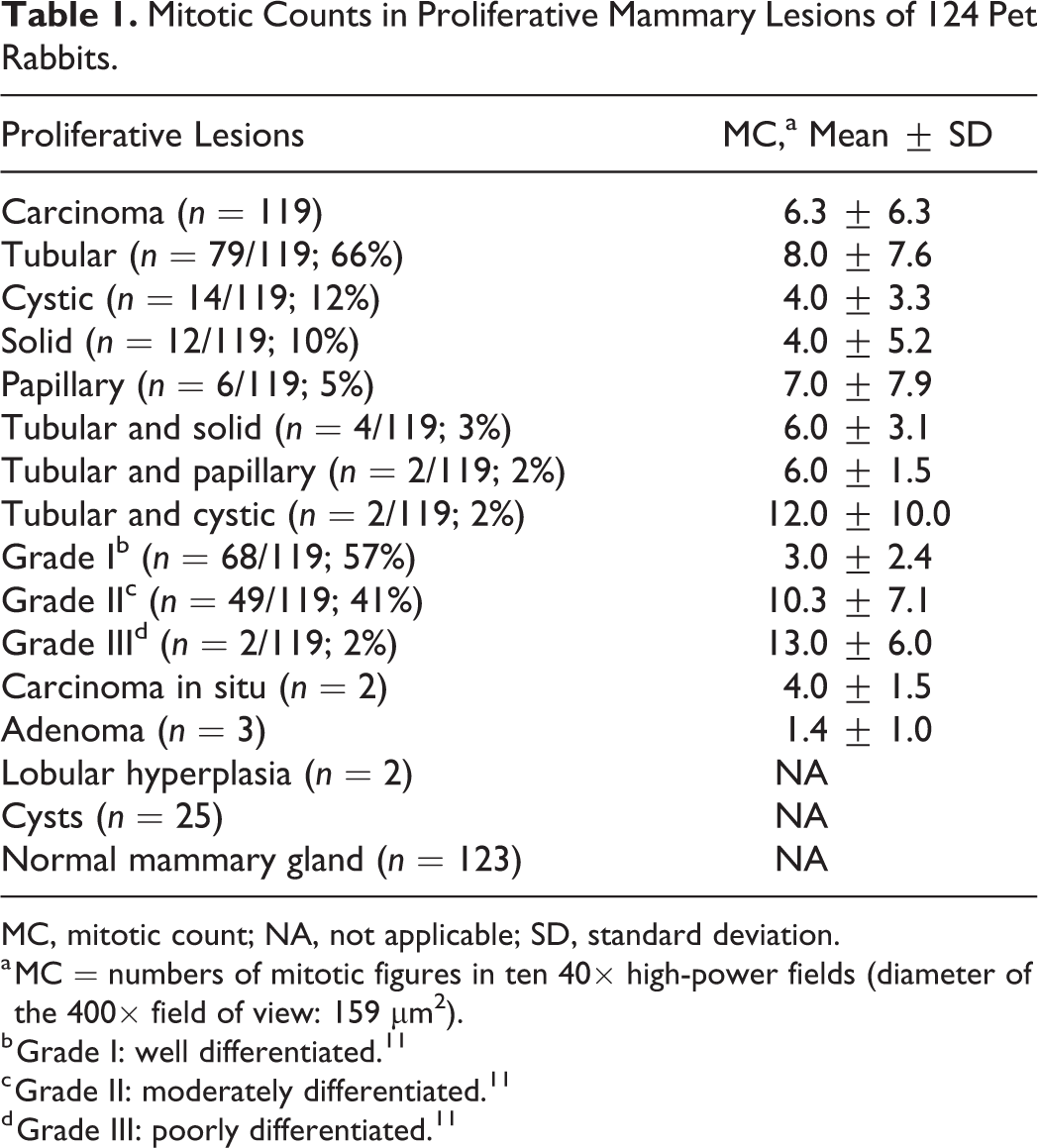
MC, mitotic count; NA, not applicable; SD, standard deviation.
a MC = numbers of mitotic figures in ten 40× high-power fields (diameter of the 400× field of view: 159 μm2).
b Grade I: well differentiated. 11
c Grade II: moderately differentiated. 11
d Grade III: poorly differentiated. 11
Histological Grades of Mammary Carcinomas and Their Histotypes in 119 Pet Rabbits.
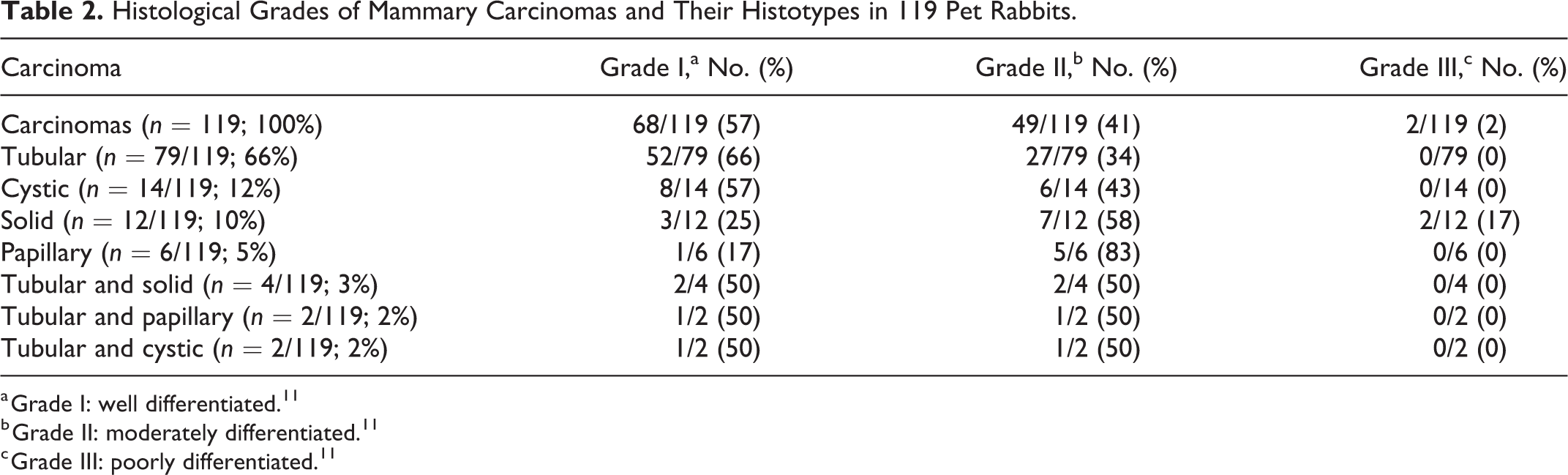
a Grade I: well differentiated. 11
b Grade II: moderately differentiated. 11
c Grade III: poorly differentiated. 11
In the positive control (rabbit uterus), a positive immunostaining for ERα and PR was located within the nuclei of luminal and glandular epithelial cells, endometrial stromal cells, and smooth muscle cells of the tunica media (Fig. 1).
Normal Mammary Tissue
In 123 of 124 samples examined, the sections also contained adjacent normal mammary tissue. Frequent findings were secretory activity of the glandular tissue with the detection of proteinaceous secretion (104/123, 85%) and clear vacuoles within alveolar epithelial cells (65/123, 53%). The latter were confirmed as lipid droplets in Sudan red–stained cryostat sections. Positive ERα expression was observed within normal mammary tissue in 62 cases (50%) and positive PR expression in 80 cases (65%). At least 1 of these receptors was expressed in 92 cases (75%). Coexpression of both receptors was detected in 50 cases (41%), and 31 cases (25%) did not express either receptor (Fig. 2; Suppl. Table S1). Mammary tissue samples with a coexpression of ERα and PR (50/123) had a slightly higher secretory activity than those with an absence of ERα and PR (31/123). In regard to the former, 84% (42/50) of the cases contained proteinaceous secretion and 52% (26/50) also contained lipid droplets, whereas 14% (7/50) completely lacked evidence of secretory activity (absence of proteinaceous material and lipid droplets). In the latter, 74% (23/31) showed secretory activity, that is, the presence of proteinaceous secretion (23/31, 74%) and lipid droplets (16/31, 52%).
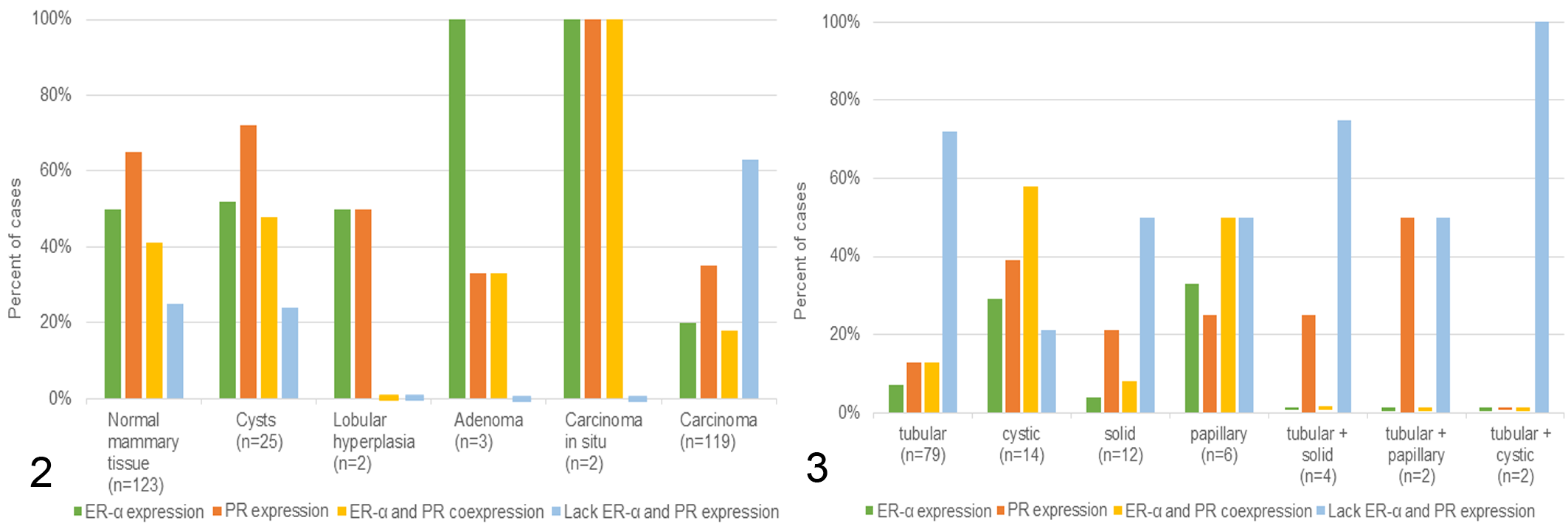
Immunostaining for estrogen receptor–α (ERα) and progesterone receptor (PR) in normal mammary gland, cysts, mammary hyperplasia, and tumors of 124 rabbits. Most cases with normal mammary tissue, cysts, lobular hyperplasia, adenoma, and carcinoma in situ are immunopositive for ERα and/or PR. In comparison, most mammary carcinomas are immunonegative for both hormone receptors.
Mammary Cysts
The mammary gland tissue of 25 of 124 rabbits (20%) contained multiple cysts. The age of these rabbits ranged from 1.5 to 10 years (mean, 5 years); the age of 4 rabbits was unknown. Mammary cysts involved the cystic dilation of tubuloalveolar structures and contained various amounts of intraluminal proteinaceous secretory material. They displayed PR immunolabeling in 17 cases (68%); of these, 10 (40%) showed concurrent expression of ERα (Figs. 2, 4; Suppl. Table S1).
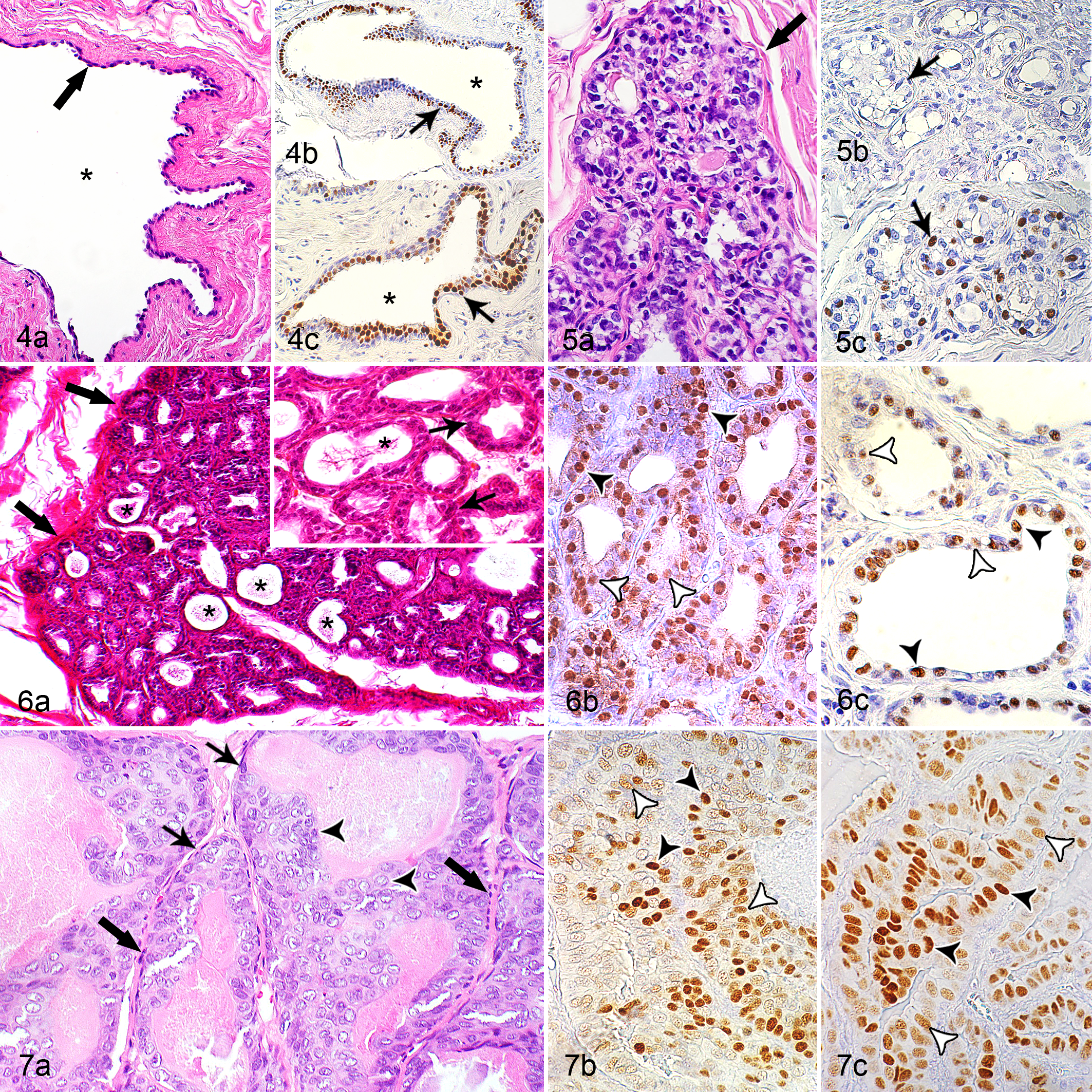
Cyst, mammary gland, rabbit No. 119. (a) The cyst (asterisk) is formed by dilation of an extralobular duct and is lined by attenuated epithelial cells (arrow). Hematoxylin and eosin (HE). (b, c) Nuclei of the lining epithelium have strong immunolabelling for estrogen receptor-α (ERα: arrow, panel b) and for progesterone receptor (PR: arrow, panel c).
The percentage of ERα-immunopositive cells ranged from 15% to 91%, and the percentage of PR-immunopositive cells varied between 7% and 88%. A mild staining intensity was most common for ERα and PR; the percentage of positive cells with a mild staining intensity was 20.1 ± 24.0 and 16.4 ± 14.7 (mean ± standard deviation), respectively. A marked staining intensity was the least frequently observed (ie, 0.4% ± 1.0% and 4.0% ± 5.9% of ERα- and PR-immunopositive cells, respectively). For ERα-positive cysts, the IRS ranged from 0.1 to 2.3 and the H-scores from 14.9 to 124.5. PR-positive cysts had IRS ranging from 0.2 to 3.7 and H-scores ranging from 9.3 to 147.5 (Suppl. Tables S3 and S4).
Lobular Mammary Hyperplasia
The mammary tissue of 2 of 124 rabbits (1.6%) contained lesions of lobular hyperplasia. The does with lobular hyperplasia were 5 and 7 years of age. The lesions of lobular hyperplasia displayed secretory activity characterized by the presence of secretory material within hyperplastic alveoli and small ducts. One case was positive for ERα (21.3% positive cells) and negative for PR; of ERα-positive cells, 11.6% showed a mild staining intensity and 2.5% had a marked staining intensity. The other case was positive for PR (31.9% positive cells) and negative for ERα; of PR-positive cells, 14.2% displayed a mild staining intensity, whereas 3.8% had a marked staining intensity (Figs. 2, 5; Suppl. Table S1). For ERα, the IRS was 0.3, and the H-score was 32.5 (Suppl. Table S3). For PR, the IRS was 1.2 and the H-score was 48.4 (Suppl. Table S4).
Mammary Adenoma
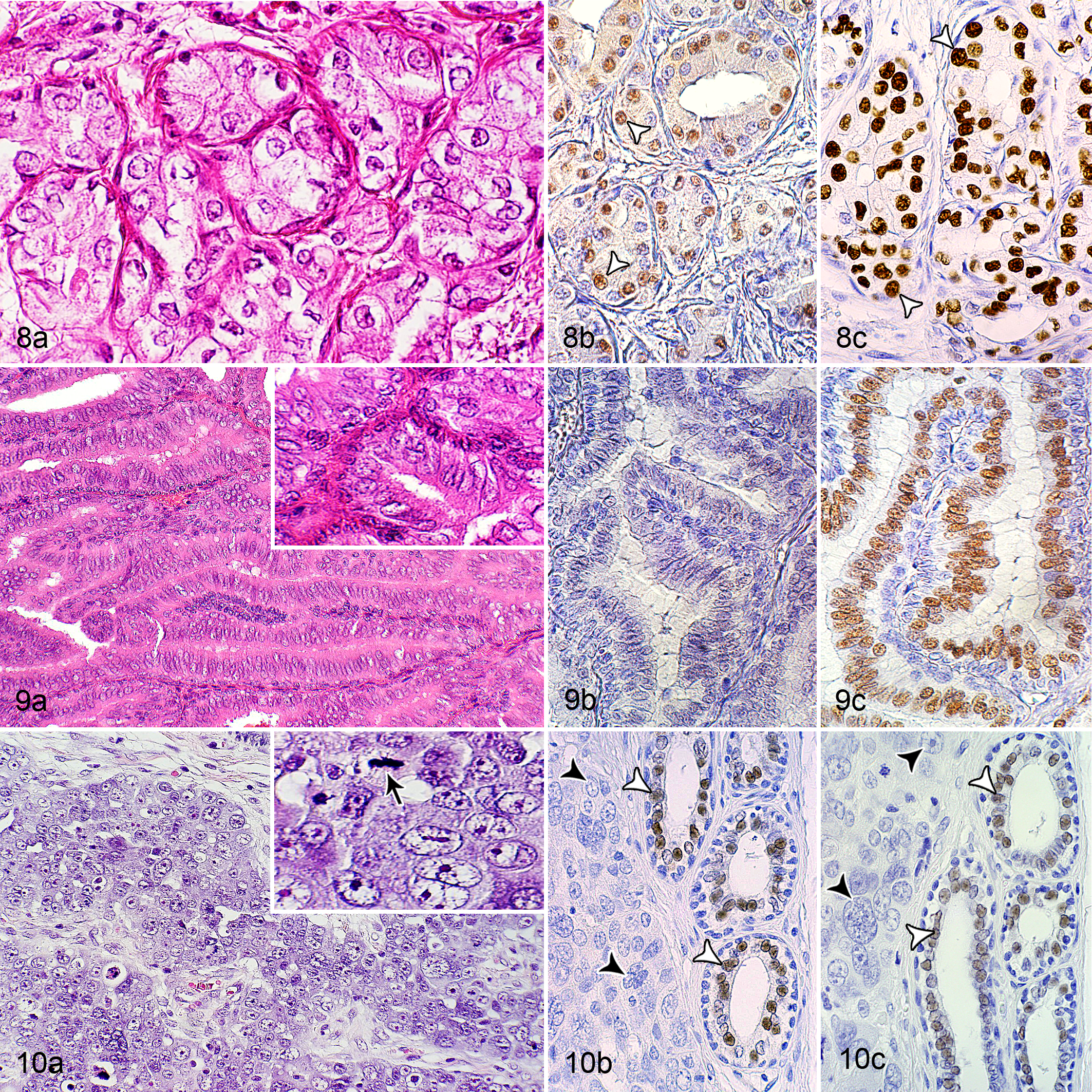
Three rabbits had a mammary adenoma; these were 4, 7, and 8 years of age. One of these tumors was a cystic adenoma with a moderate secretory activity and moderate numbers of vacuolated neoplastic cells. The other 2 were tubular adenomas with only mild secretory activity and small numbers of vacuolated tumor cells. The cystic adenoma and 1 tubular adenoma had 2 mitotic figures in 10 HPFs. No mitoses were detected in the neoplastic cells of the other tubular adenoma (Table 1). All adenomas were positive for ERα, whereas only the cystic adenoma was PR positive (Suppl. Table S1; Fig. 6). These tumors had 35%, 82%, and 91% of ERα-immunopositive cells; 2 showed mainly a strong ERα staining and 1 had predominantly a moderate ERα labeling. The IRS ranged from 2.7 to 3.7 and the H-scores from 102.6 to 156.0 (Suppl. Tables S3 and S5). The PR-positive adenoma contained 57% PR-immunopositive cells; most of these had a moderate staining intensity. The IRS was 2.3 and the H-score was 99.1 (Suppl. Tables S4 and S5).
Mammary Carcinoma In Situ
The 2 rabbits with carcinoma in situ were 5 years of age. These tumors had a tubular histotype, had mild secretory activity, and contained small numbers of vacuolated neoplastic cells (Suppl. Table S2). In contrast to adenomas, their neoplastic cells often had vesicular nuclei and showed mild anisocytosis and anisokaryosis, and there were 2 and 5 mitotic figures in 10 HPFs (Table 1; Suppl. Tables S1–S2; Fig. 7). Both tumors had immunolabeling for both ERα and PR. The IRS values were 0.3 and 1.0 for ERα and 2.5 and 2.2 for PR, respectively. The H-scores were 28.1 and 73.9 for ERα and 100.9 and 93.5 for PR, respectively (Figs. 2, 7; Suppl. Tables S2–S5).
Mammary Carcinoma
Carcinomas were diagnosed in 119 rabbits. Affected animals were between 1.5 and 10 years of age, including 2 cases involving 1.5-year-old rabbits with mammary carcinoma and no additional mammary lesions.
Histopathological findings
Tubular carcinoma was the most frequent histotype (79/119, 66%), followed by cystic (14/119, 12%), solid (12/119, 10%), papillary (6/119, 5%), tubular and solid (4/119, 3%), tubular and papillary (2/119, 2%), and tubular and cystic (2/119, 2%) histotypes (Fig. 3; Suppl. Table S2). Although these predominant growth patterns were used to determine the histotypes, most tumors displayed the additional presence of 1 or more growth patterns (114/119, 96%; Suppl. Table S2).
All carcinomas contained proteinaceous material, and neoplastic cells had 1 to several cytoplasmic empty vacuoles with distinct margins indicative of lipid droplets (confirmed by Sudan red stain in 3 tumors; Suppl. Fig. S4).
Invasion into the stroma and/or adjacent tissue was mostly mild (stroma: 51/119, 43%; peritumoral tissue: 54/115, 47%) or moderate (stroma: 62/119, 52%; peritumoral tissue: 47/115, 41%) and less frequently marked (stroma: 3/119, 3%; peritumoral tissue: 9/115, 8%; Suppl. Table S2).
The number of mitotic figures in 10 HPFs markedly differed between individual tumors and between histotypes ranging from 0 to 32 (Table 1; Suppl. Tables S1–S2).
Across all histotypes of carcinomas, well-differentiated tumors (grade I) were more frequently observed (68/119, 57%) than moderately differentiated (grade II: 49/119, 41%) or poorly differentiated tumors (grade III: 2/119, 2%) (Figs. 8–10; Table 2; Suppl. Table S2). Most carcinomas with a tubular (52/79, 66%) and cystic histotype (8/14, 57%) were grade I, whereas most solid (7/12, 58%) and papillary carcinomas (5/6, 83%) were grade II (Table 2; Suppl. Table S2).
Carcinoma, mammary gland, rabbit.
Intravascular and intralymphatic tumor cell emboli were detected in 9 of 119 (8%) and 5 of 119 cases (4%), respectively (Suppl. Table S2).
Immunohistochemical findings
Immunoreaction for ERα was observed in 24 of 119 tumors (20%) and immunoreaction for PR in 42 of 119 (35%) tumors. Only 22 of 119 (18%) showed coexpression of ERα and PR, and 75 of 119 (63%) were negative for both receptors (Figs. 2, 8–10; Suppl. Table S5).
The number of mitoses per 10 HPFs was 2.8 ± 3.2 (mean ± standard deviation) in double-immunopositive tumors, 7.4 ± 6.3 in double-immunonegative tumors, 2.5 ± 2.5 in ERα-positive PR-negative tumors, and 6.2 ± 7.2 in ERα-negative PR-positive tumors. All carcinomas with ≥14 mitoses per 10 HPFs were immunonegative for ERα, and all carcinomas with ≥31 mitoses per 10 HPFs were immunonegative for PR.
Of the 9 carcinomas with intravascular tumor cell emboli, 7 (rabbit Nos. 8, 10, 22, 45, 50, 61, 109) were immunonegative for ERα, and 4 of these (rabbit Nos. 8, 50, 61, 109) were also immunonegative for PR.
The percentage of ERα-immunopositive cells within the tumors ranged from 12% to 91% and those with PR immunostaining from 7% to 95%. The staining intensity for ERα and PR was exclusively mild in 29% (35/119) and 14% (17/119) of the carcinomas, respectively. However, in most carcinomas (ERα: 84/119, 71%; PR: 102/119, 86%), immunolabeled nuclei varied in their degree (mild, moderate, or marked) of ERα or PR immunostaining. For ERα-positive carcinomas, the IRS ranged from 0.2 to 2.3 and the H-scores from 14.9 to 124.6. PR-positive carcinomas had IRS ranging from 0.2 to 3.8 and H-scores ranging from 9.3 to 162.2 (Suppl. Table S5).
Most carcinomas with a tubular histotype (56/79, 71%) and 8 of 12 (67%) carcinomas with a solid histotype were negative for ERα and PR, whereas those with a cystic histotype were mainly positive (8/14, 58%) for both receptors. Carcinomas with a papillary and tubular histotype were either double positive (1/2, 50%) or double negative (1/2, 50%; Fig. 3; Suppl. Table S1). The hormone receptor status of grade I, II, and III carcinomas is also provided in Supplemental Tables S1, S3, and S4. Notably, most grade I and grade II tumors (62% and 69%, respectively) were immunonegative for ERα and PR. Both grade III tumors were negative for ERα and PR (Suppl. Table S1).
The carcinoma and the adjacent normal mammary tissue displayed an opposite immunoreaction (positive or negative) for ERα in 30 of 119 (25%) cases and for PR in 40 of 119 cases (33%). In all but 1 of these cases, the adjacent normal mammary tissue was immunopositive for the respective steroid receptor(s), whereas the carcinoma was immunonegative. In only 1 case, the carcinoma showed a positive ERα immunostaining, whereas the adjacent normal mammary tissue was negative for the presence of this receptor. In 30 of 119 (25%) carcinomas with a lack of immunostaining for ERα and PR, the adjacent normal mammary tissue was also immunonegative for these receptors (Suppl. Table S5). The general immunoreactivity of these tissue samples was confirmed by detecting cytokeratin immunostaining in nonneoplastic and neoplastic mammary epithelial cells (Suppl. Figs. S5 and S6).
Statistical Analysis
Female (n = 87) and female-spayed (n = 22) rabbits showed no statistically significant difference in the total differentiation scores (3–9) of the mammary carcinomas. There was also no significant association between the age of the rabbit (which was known for 97 rabbits) and the area of the tubular growth pattern within a particular carcinoma (<10%, 10%–75%, or >75%), the total differentiation score (3–9) of the carcinoma, or the histological tumor grade (grade I, II, or III).
The 2 scores (IRS, H-score) for the evaluation of the expression of ERα and PR were significantly correlated (P < .001; ρ = 1, Figs. 11, 12). There was a significant correlation between increased mitotic count and reduced IRS or H-score for ERα and PR (ie, carcinomas with a higher mitotic count had a reduced expression of ERα and PR) (Suppl. Table S6). For a better illustration of this correlation, rabbit mammary carcinomas were subdivided into 2 categories: those with a lack of ERα and/or PR expression (IRS and H-score = 0) and those with a positive immunostaining for ERα and/or PR (IRS and H-score >0). ERα- and/or PR-immunonegative carcinomas had significantly higher mitotic counts than ERα- and/or PR-immunopositive carcinomas (Figs. 13–16; Suppl. Table S7).
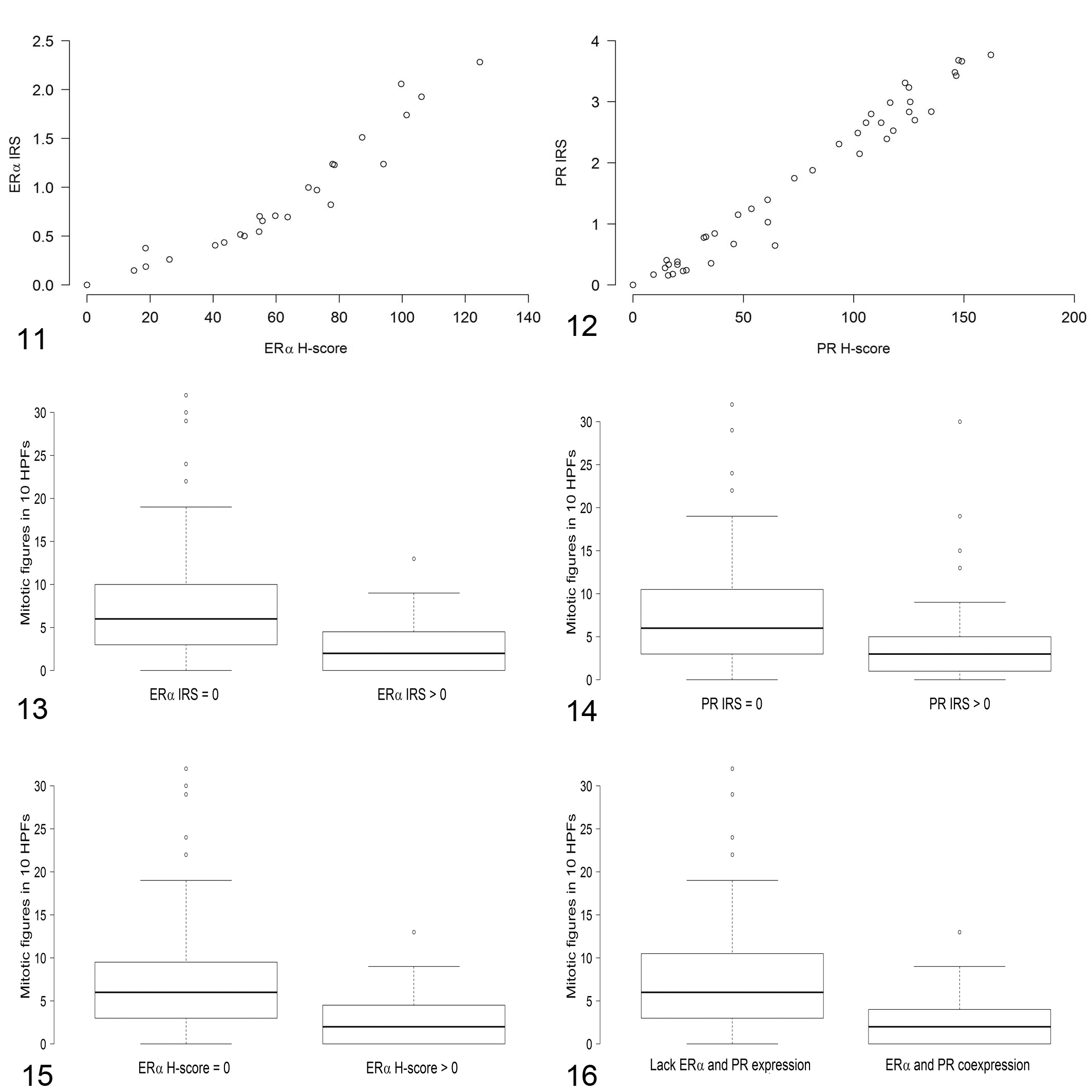
Comparison of the immunoreactive score (IRS) and the H-score for estrogen receptor–α (ERα, Fig. 11) and progesterone receptor (PR, Fig. 12) immunoreactivity in 119 rabbit mammary carcinomas. The IRS and H-scores are significantly correlated (P = .001) for ERα and for PR.
There was no significant relationship between the IRS or H-scores for ERα and PR and the total differentiation scores of the tumors or the histological tumor grade (grade I, II, or III; Suppl. Table S6). A negative statistical correlation (P < .001), however, existed between the percentage of tubular growth and the IRS or H-scores for ERα and PR (Suppl. Table S6).
Discussion
This study characterized mammary tumors and other proliferative lesions of 124 pet rabbits by histopathology and immunohistochemistry for the detection of ERα and PR. In agreement with previous investigations, 4,28 most rabbits with mammary tumors were female. However, the findings of this study cannot properly address whether early spaying reduces the incidence of mammary tumors in rabbits, since the time and reason for spaying of the rabbits are unknown.
As already reported, 4,28 the mammary tumors, other proliferative lesions, and the normal mammary tissue often contained epithelial cells with cytoplasmic vacuoles that were identified as lipid droplets. The lipid droplets likely represent histological features of secretory activity, since the mammary secretion of does is lipid rich 7 , and proteinaceous material together with intracellular lipid droplets also has been described in secretory mammary neoplasms of mice and rats. 18
This study confirmed the previous finding that carcinoma is the most frequently diagnosed mammary lesion of rabbits. 4,28
Two separate mechanisms for the development of mammary carcinomas in laboratory rabbits were proposed: progression from nonneoplastic mammary cystic lesions and de novo development. 13 In the present study, the concurrent presence of mammary cysts and a carcinoma in 20% (25/124) of the rabbits, as well cystic areas in 21% (25/119) of carcinomas, may support the suggested multistep tumor development. 13 The mammary cysts and the cystic tumor areas, however, might have also developed secondary to tissue compression by the neoplasm. This could impair the release of secretory material and might result in its retention within tubuloalveolar structures followed by their cystic distension.
The rabbit mammary carcinomas were mainly well differentiated or moderately differentiated (ie, histological grade I or II according to Elston and Ellis 11 ). In humans, the histological grade according to Elston and Ellis 11 and the hormone receptor status have prognostic value. 10,25 These 2 parameters, however, do not necessarily correlate, although grade I tumors most commonly are ERα positive. 25 Histological grade is an independent prognostic indicator that predicts the biological behavior (ie, likelihood to recur and metastasize) of a breast carcinoma regardless of its hormone receptor status. 10,28 In contrast, the ERα/PR status is not an independent prognostic factor but instead predicts the response of the tumor to an endocrine therapy and thus assists in selection of optimal therapy. 10,25
To our knowledge, there are no comprehensive studies of the expression of ERα and PR receptors in healthy and diseased mammary gland of rabbits. In 30 of 119 cases, the normal mammary tissue was immunonegative for ERα and PR. Using a hormone receptor binding assay, it has been shown that levels of ERα and PR in mammary gland tissue differed between estrus virgins that did not tolerate the buck and those accepting the males, and both receptors markedly declined during early pregnancy. 15 Moreover, PR levels were very low during early lactation and undetectable at day 10 of lactation. 15 This indicates that, in does, the expression of ER and PR in mammary tissue may change depending on the respective hormonal influences. Thus, in the present study, the negative immunostaining of normal mammary tissue for ERα and/or PR immunolabeling suggests the downregulation of hormone receptors.
Previously, the immunohistochemical expression of ERα together with HER2 and cytokeratins 5 and 6 had been investigated on a limited number of mammary lesions of pet rabbits (3 hyperplastic glands, 2 adenomas, 10 carcinomas), and 1 case of mammary gland hyperplasia showed a negative immunostaining for ERα. 3 Similarly, the present study also contained a case of lobular hyperplasia with a lack of ERα immunostaining.
There was a high percentage (75/119, 63%) of rabbit mammary carcinomas that completely lacked expression of both steroid hormone receptors. This indicates that their tumor growth is independent of exposure to estrogen and/or progesterone.
A false-negative immunostaining for ERα and PR was ruled out by detecting ERα and PR immunostaining in the positive controls and by confirming the general immunoreactivity of the double-immunonegative tissue by the use of an antibody for cytokeratin.
A scoring system for the quantification of an immunoreaction should include the percentage of immunopositive cells as well as their staining intensity. 10,24 For analysis of the ERα and PR expression in breast cancer of women, the H-score and the Quick score are mainly used, and both have prognostic utility. 10 The Quick score includes the percentage of immunopositive cells as well as their average staining intensity. 10,24 In contrast, the H-score represents a summation of the percentages of tumor cells with mild, moderate, and marked immunoreactivity. 10,24 In the equine endometrium, cyclic changes in ERα and PR expression were quantified by the use of the IRS. 2 Similar to the H-score, the IRS is based on the percentages of tumor cells with mild, moderate, or marked immunoreaction. 2
For the evaluation of the pet rabbit carcinomas, the IRS 2 and the H-score 10 appeared to be most suitable, since the staining intensities for ERα and PR in the rabbit carcinomas were often heterogeneous. Thus, in the present study, the possible application of these scores for quantification of ERα and PR expression in rabbit mammary carcinomas was compared. The IRS and H-score applied in the present study statistically significantly correlate.
In the present study, there was no correlation of histotype or histological grade with the ERα/PR status of the carcinomas. However, a significant correlation existed between a higher mitotic count and a lower expression of ERα and PR that was determined by the calculation of the IRS and the H-scores.
The standardized diagnosis of the ERα and PR status in mammary carcinomas of women is important for the selection of the appropriate treatment. The lack of steroid receptors renders double-negative tumors unresponsive to hormonal treatment. 14 Hormone receptor antagonists such as tamoxifen are a standard therapy for ERα-positive tumors (ie, carcinomas with ERα expression in >1% of the tumor cells) in women. 14 In bitches, tamoxifen therapy is not recommended as its estrogen-like effects on the reproductive organs predispose to pyometra and incontinence. 5 In comparison, an antiestrogenic effect of tamoxifen has been observed in uterine tissue of rabbits. 6 Thus, tamoxifen might be of therapeutic value for ERα-positive rabbit mammary carcinomas.
In conclusion, carcinomas were the most frequently diagnosed mammary proliferative lesions in pet rabbits. Despite the prevalence of the tubular histotype and good differentiation, most of them showed negative immunostaining for ERα and/or PR. The latter is expected to render them insensitive to hormonal treatment. The statistically significant negative correlation between a higher mitotic count and a lack of immunostaining for ERα and/or PR indicates that proliferative activity in rabbit mammary carcinomas is influenced by factors other than estrogen or progesterone.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985818788611 - Estrogen Receptor–α and Progesterone Receptor Expression in Mammary Proliferative Lesions of Female Pet Rabbits
Supplemental Material, DS1_VET_10.1177_0300985818788611 for Estrogen Receptor–α and Progesterone Receptor Expression in Mammary Proliferative Lesions of Female Pet Rabbits by Sophie Degner, Heinz-Adolf Schoon, Claudia Laik-Schandelmaier, Heike Aupperle-Lellbach, and Sandra Schöniger in Veterinary Pathology
Footnotes
Acknowledgements
We thank Maritta Wipplinger for the immunostaining and Mathias Baudis and Patrick Scheibe for the statistical advice.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
