Abstract
An experimental transmission study was designed to determine whether a causal relationship exists between a Ranavirus (BSTRV) isolated from a Burmese star tortoise that died and the lesions observed in that tortoise. A pilot study was performed with 3 box turtles (Terrapene ornata ornata) and 3 red-eared sliders (RESs; Trachemys scripta elegans) to assess their suitability in a larger study. Based on the outcome of this study, RESs were selected, and 2 groups of 4 RESs received either an oral (PO) or intramuscular (IM) inoculum containing105 50% Tissue Culture Infecting Dose (TCID50) of a BSTRV-infected cell lysate. One turtle each was mock inoculated PO or IM with the same volume of uninfected cell lysate. Three of four IM-inoculated RESs developed clinical signs (nasal and ocular discharge [3 of 3], oral plaques [1 of 3], conjunctivitis and hyphema [1 of 3] and extreme lethargy [3 of 3]). A Ranavirus was isolated from kidney homogenates of 3 euthanatized turtles; DNA sequences of a portion of the major capsid protein gene were amplified by polymerase chain reaction. Consistent histologic lesions were observed only in IM-inoculated turtles and included fibrinoid vasculitis centered on splenic ellipsoids, multifocal hepatic necrosis, and multicentric fibrin thrombi in a variety of locations, including hepatic sinusoids, glomerular capillary loops, and pulmonary capillaries. Virions compatible with Ranavirus were observed within necrotic cells of the spleen of 1 IM-inoculated turtle using transmission electron microscopy. This study fulfills Koch's postulates, confirming a causal relationship between BSTRV and the clinical and histologic changes in chelonians infected with this virus.
Viruses in the family Iridoviridae are large, double-stranded DNA viruses capable of infecting invertebrates and ectothermic vertebrates.43 Iridoviruses within the genus Ranavirus have been shown to be capable of infecting fish, amphibians, and reptiles,9 and have emerged as major pathogens of free-ranging amphibians worldwide.12,13,45 In a study of 64 amphibian morbidity and mortality events between 1996 and 2001, the most common cause of mortality events was infection with iridoviruses.18
Iridovirus infections in reptiles have been less well described than in amphibians and fish, with sporadic reports in chelonians (turtles and tortoises), snakes, and lizards.21,26,35 Infection of a tortoise was first reported in 1982 in a spur-tailed Mediterranean land tortoise (Testudo hermanni) that had necrotic foci in the liver, intestine, and spleen.19 Subsequently, iridoviruses were reported in other species of chelonians both in captivity and in the wild.8,14,34,42 Clinical signs associated with infections have included signs of upper respiratory tract disease, including respiratory distress and nasal discharge, as well as oral ulceration, cutaneous abscessation, anorexia, and lethargy.14,42 While there is circumstantial evidence that iridoviruses are primary pathogens in chelonians, Koch's postulates have never been fulfilled.
Ranaviruses were identified recently in tissues obtained from a variety of wild and captive chelonians that died in Georgia, Florida, Texas, New York, and Pennsylvania.25 Species affected were Burmese star tortoises (Geochelone platynota), gopher tortoises (Gopherus polyphemus), eastern box turtles (Terrapene carolina carolina), and Florida box turtles (Terrapene carolina bauri). Clinical signs in these cases included lethargy, anorexia, nasal discharge, conjunctivitis, severe subcutaneous cervical edema, and necrotizing pharyngitis-stomatitis. A ranavirus isolated from a Burmese star tortoise (G platynota) that died in an outdoor pen in a zoological collection from Georgia in 2003 was further characterized as either identical or closely related to Frog Virus 3.24 To determine whether or not a causal relationship exists between the isolated Ranavirus and the clinical signs and histologic lesions observed in these chelonians by attempting to fulfill Koch's postulates, we performed two experimental transmission studies using the Burmese star tortoise isolate that will hereafter be termed Burmese star tortoise Ranavirus (BSTRV). The first was a pilot study to determine the suitability of either western ornate box turtles (BTs; Terrapene ornata ornata) or red-eared sliders (RESs; Trachemys scripta elegans) as an experimental model for a subsequent larger transmission study. Here we report the findings of those studies.
Materials and Methods
Experimental animals and husbandry
Study 1 was a pilot study involving 3 adult western ornate BTs (Terrapene ornata ornata) and 3 adult RESs (Trachemys scripta elegans) that were purchased from a reptile supplier in Texas. Study 2 involved 10 adult RESs purchased from a reptile supplier in Louisiana. Both studies were performed under the approval of the Institutional Animal Care and Use Committee at the University of Florida. Turtles were housed individually in plastic containers in a centralized animal facility room maintained at approximately 25°C. For each species, appropriate husbandry that included commercially available foods was used. BTs were kept on land, with overhead heat lamps provided for basking at one end of the container (average temperature of 28°C). RESs were kept in water (temperatures averaging between 21.3°C and 25.6°C), with basking platforms provided under an overhead heat lamp (average basking temperature of 28°C). Fluorescent room lights were kept on a 12-hour light and 12-hour dark cycle.
Preinoculation sample collection
Upon arrival, each turtle was examined for the presence of any clinical signs of illness. Oral and cloacal swabs and urine were obtained from each turtle and tested for the presence of iridovirus using a polymerase chain reaction (PCR; see below). Blood samples were collected from each turtle into 2-ml tubes coated with lithium heparin and centrifuged at 4,500 × g for 5 minutes. Buffy coats were removed and also were tested for iridovirus using PCR. Plasma was removed and tested for anti-Ranavirus antibodies using an indirect enzyme-linked immunosorbent assay (ELISA; see below).
DNA preparation, PCR, and nucleotide sequencing
Oral and cloacal swabs were combined for each turtle into one 1.5-ml microcentrifuge tube, and 100 μl phosphate-buffered saline (PBS) was added. DNA was extracted from buffy coats, oral swabs, and cloacal swabs using the DNeasy kit (Qiagen, Valencia, CA), as were tissue samples later collected at necropsy. DNA from urine samples was extracted using the QIAmp Ultrasens virus kit (Qiagen). Sense primer (5′-GACTTGGCCACTTATCAC-3′) and antisense primer (5′-GTCTCTGGAGAAGAAGAA-3′) were used as previously described33 to amplify approximately 500 base pair segments of the Ranavirus major capsid protein gene. The PCR products were resolved in 1% agarose gels, and any bands were excised and purified using the QIAquick gel extraction kit (Qiagen). Products were sequenced in both directions directly using the Big-Dye Terminator Kit (PerkinElmer, Branchburg, NJ) and were analyzed on ABI 377 (PerkinElmer Life and Analytical Sciences, Waltham, MA) automated DNA sequencers at the University of Florida's Sequencing Center. The sequences were compared to known sequences in GenBank (National Center for Biotechnology Information, Bethesda, MD), EMBL (Cambridge, United Kingdom), and Data Bank of Japan (Mishima, Japan) databases using TBLASTX.2
ELISA
An indirect ELISA was used to determine the presence of anti-Ranavirus antibodies. The ELISA methodology was similar to that developed for use in identifying the presence of anti-tortoise herpesvirus antibodies in tortoises37 and anti–West Nile virus antibodies in alligators.22 The BSTRV isolate was used as the antigen in the assay. Each well of a 96-well high-protein binding microplate was coated overnight at 4°C with 50 μl of a 1:400 dilution of either an uninfected lysate from Terrapene heart cells (TH-1, ATCC-CCL 50; American Type Culture Collection, Rockville, MD) or TH-1 cell lysate from cells infected with BSTRV. Lysates were diluted in 0.01 M sodium phosphate buffer (pH 7.2) containing 0.15 NaCl and 0.02% NaN3 (PBS/A). Wells then were washed 4 times in ELISA wash buffer (PBS/A with 0.05% Tween-20). This washing process was repeated in between all of the following steps. Wells were blocked with 300 μl of 5% nonfat dry milk diluted in ELISA wash buffer (blocking buffer) for 1 hour at room temperature. All remaining steps were incubated for 1 hour at room temperature. Plasma samples were added in 50-μl volumes at a 1:100 dilution in blocking buffer. The secondary antibody used was a biotin-conjugated mouse anti-tortoise immunoglobulin (Ig) monoclonal antibody diluted to a final concentration of 0.5 μg/ml in blocking buffer. Alkaline phosphatase–conjugated streptavidin (Zymed Laboratories Inc., San Francisco, CA) then was applied to each well at 50 μl of a 1:5000 dilution in PBS/A. Next, 100 μl of a 1.0-mg/ml P-nitrophenyl phosphate prepared in 0.01 M sodium bicarbonate buffer containing 2 mM MgCl2 was added to each well, and the plates then were stored in the dark until being read. The optical density (OD) of each well was read at A405 using a StatFax 3200 microplate reader (Awareness Technology, Palm City, FL) after 30 minutes.
Each sample was done in triplicate: one time on wells coated with uninfected cell lysate and in duplicate on wells initially coated with infected cell lysate. The replicate values of the wells coated with BSTRV lysate were averaged and divided by the OD reading of the value of the plasma sample run on the uninfected cell lysate to subtract out any background binding that might be caused by cross-reactivity to the cells. Values greater than 2 were considered positive,22 suggesting previous exposure to the virus, and would preclude use of that turtle in the study. Plasma from a surviving penmate of the above Burmese star tortoise was used as a positive control.
Virus preparation
Terrapene heart cells (TH-1) were acquired from the American Type Culture Collection (ATCC-CCL 50; Rockville, MD) and grown to confluency in 225-cm2 tissue flasks (Costar, Corning, NY). Cells were cultured in Dulbecco modified Eagle medium (DMEM; Gibco, Carlsbad, CA) supplemented with 5% fetal bovine serum (Gibco), gentamicin (60 mg/l; Sigma, St. Louis, MO), penicillin G (120,000 U/l), streptomycin (120,000 U/l), and amphotericin B (300 μg/l; Sigma). Cells were inoculated with a fourth passage of BSTRV and incubated at 28°C in the presence of 5% CO2. When cytopathic effect (CPE) was observed, consisting of cell rounding and detachment from the flask in over 70% of cells, the flasks were scraped and contents transferred to 15-ml centrifuge tubes and clarified by slow-speed centrifugation at 4,500 × g for 30 minutes. The supernatant then was discarded and the cell pellet resuspended in 10 ml cell culture media. The preparation then was vortexed, frozen, and thawed 3 times to release virus from the cells into the supernatant. The preparation was again clarified by centrifuging for 30 minutes. The supernatant then was transferred to a new tube, and the cell pellet was discarded. The live virus in the media then was quantified by a 50% tissue culture infecting dose (TCID50) assay, diluted with media to create a concentration of 105 TCID50/ml, and frozen at −80°C until use.
Transmission studies
Study 1
Study 1 was designed as a pilot study to determine the suitability of either RESs or BTs as a model of Ranavirus infection for chelonians. A total of 3 RESs and 3 BTs were included in the study, and each turtle was allowed to acclimate for 2 weeks prior to infection. One of each species was assigned to one of three groups: 1) a mock-inoculated control group, with both turtles receiving 0.5 ml uninfected cell lysate orally (PO) and 0.5 ml by intramuscular injection (IM); 2) a PO-inoculated group, with both turtles receiving 1 ml infected cell lysate containing virus at 105 TCID50/ml by metal gavage feeding tube in the caudal esophagus; and 3) an IM-inoculated group, with both turtles receiving the same concentration of virus as the PO-inoculated group with 0.5 ml injected into both the left and right pectoral muscles. Turtles were observed daily after inoculation for the duration of the study. Oral swabs and buffy coats were collected 1 week postinoculation for evaluation by PCR for the presence of iridovirus. DNA extraction and PCR were performed as described previously for prescreening. Physical examinations were performed daily to assess the presence of the following clinical signs: lethargy, anorexia, cervical edema, palpebral or periocular edema, ocular discharge, nasal discharge, oral discharge, the presence of oral plaques, or any other abnormalities.
Turtles were euthanatized if clinical signs became severe or at 2 weeks postinoculation. For euthanasia, ketamine was administered IM at 100 mg/kg, followed by intravenous sodium pentobarbital. Once turtles were unresponsive to painful stimuli and showed no corneal reflex, they were decapitated, and a complete necropsy was performed. Portions of tongue, esophagus, stomach, small and large intestine, liver, kidney, and spleen were collected aseptically by flame-sterilizing tools between each organ, and were frozen at −80°C for virus isolation and/or DNA extraction and PCR. The following tissues were collected and fixed in 10% neutral phosphate-buffered formalin: tongue, esophagus, stomach, small and large intestine, liver, kidney, spleen, pancreas, heart, trachea, lung, brain, urinary bladder, thyroid gland, adrenal gland, bone, bone marrow, skin, skeletal muscle, nasal cavity, eye, and gonad. These tissues then were processed for histologic examination. They were embedded in paraffin, and 6-μm sections were stained with hematoxylin and eosin.
Virus isolation was performed on Terrapene heart cells seeded into 25-cm2 flasks (Costar) in the same media as described for virus prepartion and cultured to confluency. A small piece of kidney (approximately 50 mg) collected aseptically at necropsy was homogenized in separate 5-ml tissue grinders containing DMEM. The homogenate was passed through a 0.45-μm filter (Costar) onto a flask of cells. Cells were incubated at 28°C and observed daily. Flasks of cells were harvested when CPE was observed in over 70% of cells or at postinoculation day 10. DNA was extracted from cells using the DNeasy kit (Qiagen) protocol for animal cells. PCR and nucleotide sequencing were performed as previously described in the section titled “DNA preparation, PCR, and nucleotide sequencing” to confirm the presence of iridovirus DNA sequences.
Study 2
Based on the results of the pilot study, a second transmission study was designed that involved 10 RESs. Turtles were assigned to one of the following three groups: 1) control, 2) IM inoculated, or 3) PO inoculated. Turtles were numbered from 1 to 10 according to increasing weight. They then were blocked into two groups (1–5 and 6–10), and turtles within each block were randomized to: 1 control, 2 PO inoculated (turtle Nos. 1, 2, 7, and 10), and 2 IM inoculated (turtle Nos. 3, 5, 6, and 8). The control turtle in the lower weight block (turtle No. 4) was PO mock-inoculated with 1 ml uninfected cell lysate, whereas the control turtle in the higher weight block (turtle No. 9) received 1 ml IM, half in each pectoral muscle. Turtles were monitored daily for the presence of clinical signs consistent with iridovirus infection. Oral and cloacal swabs were taken three times a week throughout the 4-week study. Free catch urine samples were collected opportunistically at the same time periods. Turtles were euthanatized when clinical signs of infection became severe or at 4 weeks postinoculation.
Necropsies, tissue collections, and virus isolation were performed as described in “Study 1.” In addition, spleens from 1 IM-inoculated (No. 6) and 1 control (No. 9) turtle were collected in Trump solution (4% formaldehyde, 1% glutaraldehyde in a phosphate buffer; Electron Microscopy Sciences, Hatfield, PA) for transmission electron microscopy (TEM). Tissues were embedded in Spurr resin, and ultrathin sections were obtained and stained with lead citrate and uranyl acetate for TEM at the Electron Microscopy Core Laboratory, University of Florida.
Results
Experimental animals and preinoculation sampling
Upon arrival, all turtles were considered healthy. Using PCR, all oral swabs, cloacal swabs, urine, and buffy coats collected from turtles in studies 1 and 2 were negative for iridovirus DNA sequence. All plasma samples evaluated by ELISA were below the positive cutoff value, indicating that the turtles were seronegative for exposure to Ranavirus.
Transmission studies
Study 1
Mock- and PO-inoculated RESs and BTs showed no clinical signs of disease throughout the 2-week study. The IM-inoculated RES and BT both showed severe clinical signs. At 8 days postinoculation the IM-inoculated RES was basking continuously and started showing signs of lethargy and cutaneous erythema, and kept its palpebrae closed. It was found dead on the basking platform on the morning of 9 days postinoculation. At 8 days postinoculation, the IM-inoculated BT developed a thick, translucent ocular discharge (Fig. 1), at 10 days it became lethargic and anorexic, and at 12 days postinoculation it was euthanatized. The orally inoculated RES and BT remained normal.
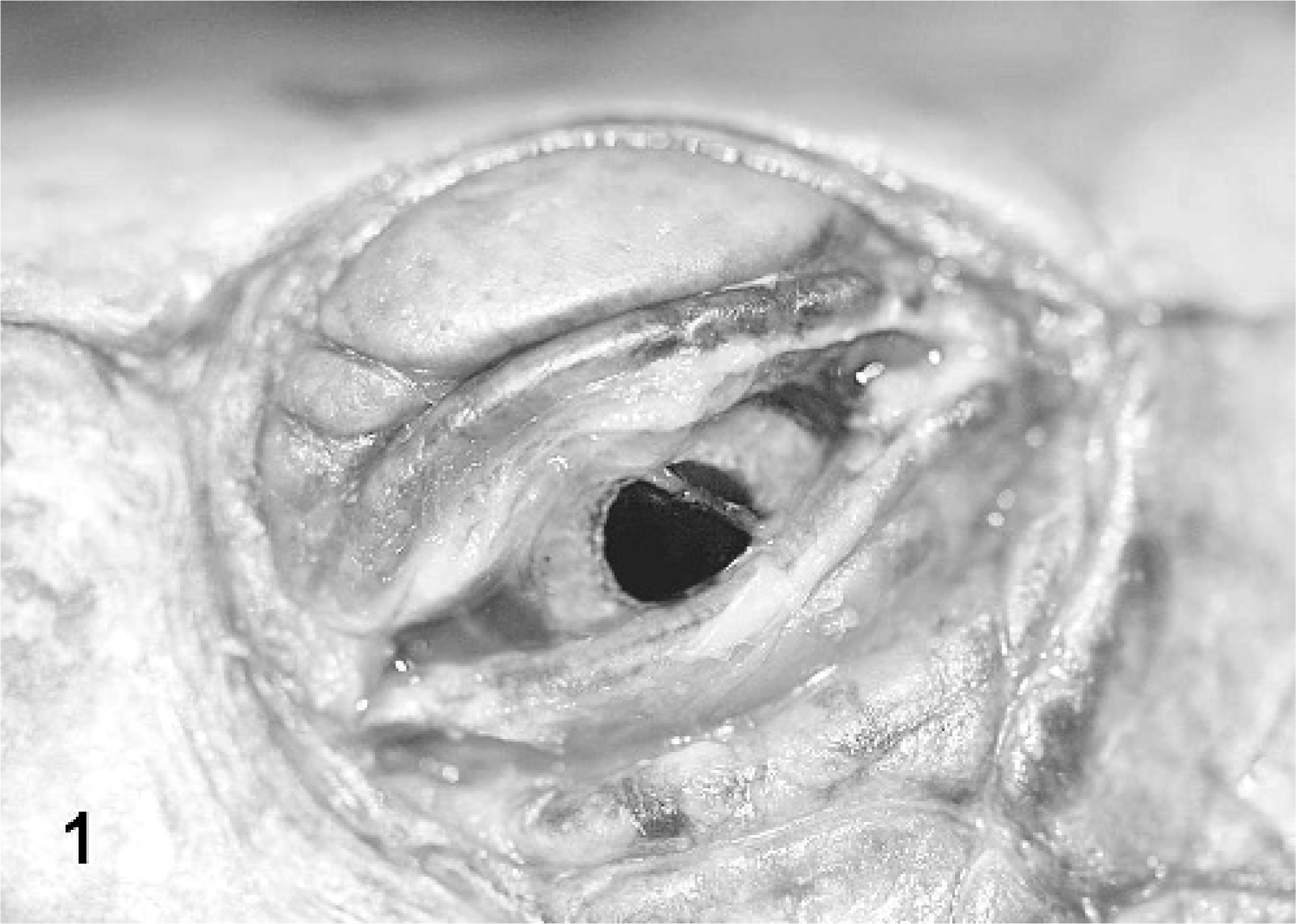
Western ornate box turtle (study 1, No. 3) intramuscularly inoculated with Ranavirus. Photograph taken 12 days postinoculation shows thick, translucent ocular discharge.
At 1 week postinoculation both IM-inoculated turtles in the pilot study were positive by PCR for iridovirus major capsid protein gene sequences using DNA extracted from oral swabs and buffy coats collected. Oral swabs and buffy coats collected on the PO-inoculated turtles and mock-inoculated control turtles were negative. PCR performed on DNA extracted from both IM-inoculated turtles was positive for all 8 tissues (Table 1). PCR was negative for all 8 tissues of both control turtles and both PO-inoculated turtles.
PCR results on tissues collected at necropsy from the pilot study BTs and RESs and RESs in the full transmission studies.



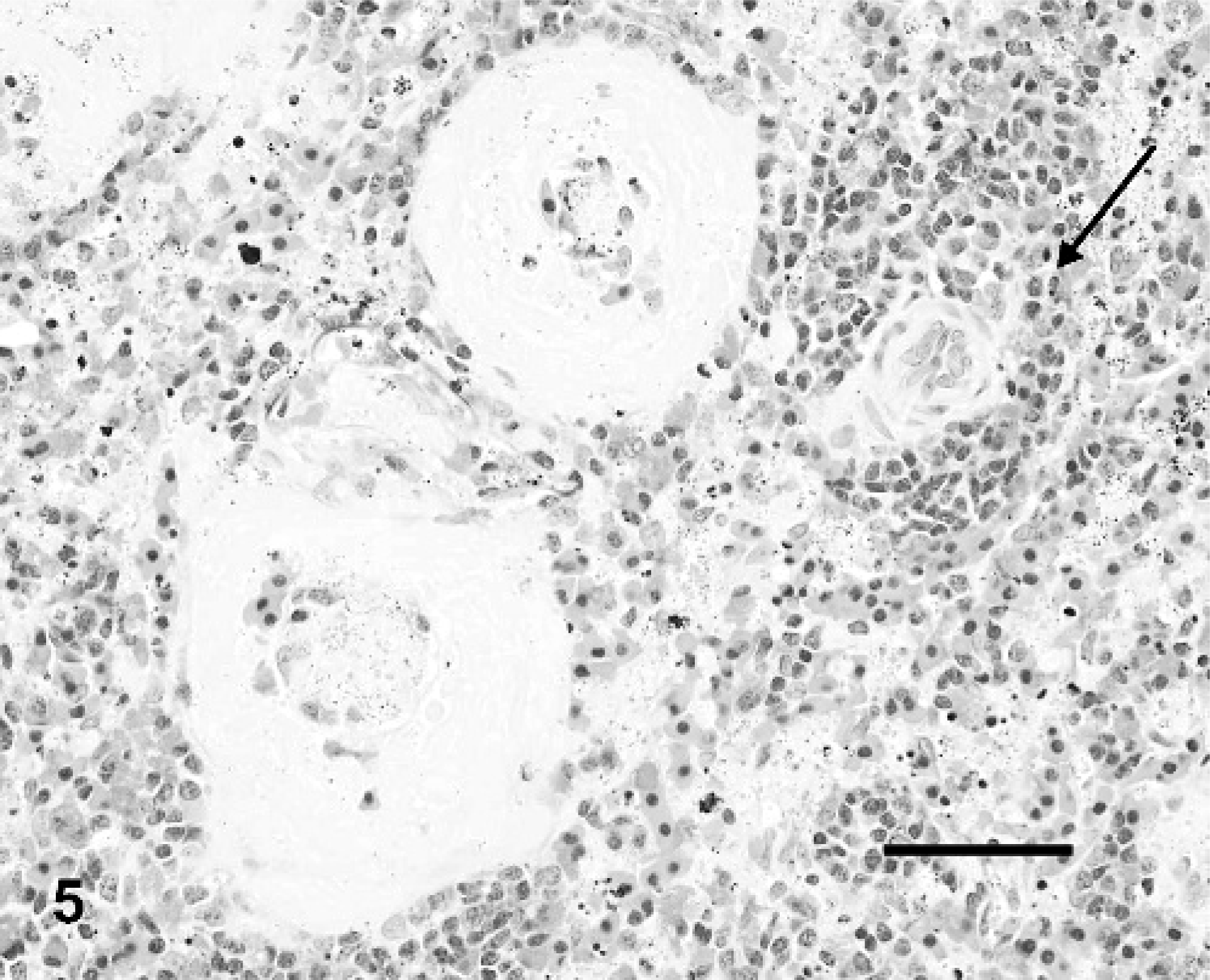
The most consistent and significant histologic lesions were observed only in the spleens (2 of 2) of the IM-inoculated BT and RES, and they were similar in both. Lesions were centered on the splenic ellipsoids or “sheathed capillaries” and will be described in detail for turtles in study 2 (below). Briefly, the walls of the ellipsoids were moderately to markedly expanded by homogenous to slightly fibrillar eosinophilic material (fibrin, fibrinoid vasculitis) with multifocal infiltrates of low numbers of heterophils. There were occasional luminal fibrin thrombi with admixed heterophils and karyorrhectic debris.
Kidney samples from both IM-inoculated turtles (P3 and P6) that were coated onto TH-1 cells demonstrated cytopathic effects of cell rounding and lysis within 2 days of incubation. Intracytoplasmic inclusion bodies were observed in infected cells, and PCR and nucleotide sequencing on DNA extracted from cells from each flask were positive for iridovirus. None of the cultures inoculated with tissues collected from PO-inoculated turtles or control turtles demonstrated any CPE up to 10 days postinoculation of cells, and each was negative by PCR for the presence of Ranavirus DNA.
Study 2
Similar to study 1, only IM-inoculated RESs showed clinical signs of disease and were euthanatized before the end of the 4-week study. Three of the four IM-inoculated turtles showing severe clinical signs were euthanized on 11, 13, and 23 days postinoculation. All 3 turtles became anorectic and extremely lethargic. Turtle 6, euthanatized 13 days postinoculation, developed oral plaques on the roof of the mouth and tip of the tongue (Fig. 2). Turtle 3, euthanatized 23 days postinoculation, exhibited exophthalmus, conjunctivitis, and hyphema (Fig. 3). All 3 turtles had clear serous ocular and nasal discharge. The fourth IM-inoculated turtle (No. 8) showed ocular discharge and subjectively basked more than the other turtles between 16 and 25 days postinoculation, but then recovered and showed no clinical signs at the termination of the study. It became anorectic 14 days postinoculation, and remained so throughout the study. Three of four PO-inoculated turtles also became anorectic after inoculation, and remained so throughout the study. No other signs of disease were noted in those turtles.

Red-eared slider (study 2, No. 3) intramuscularly inoculated with Ranavirus. Photograph taken 22 days postinoculation shows exophthalmus, conjunctivitis, and hyphema (arrow).
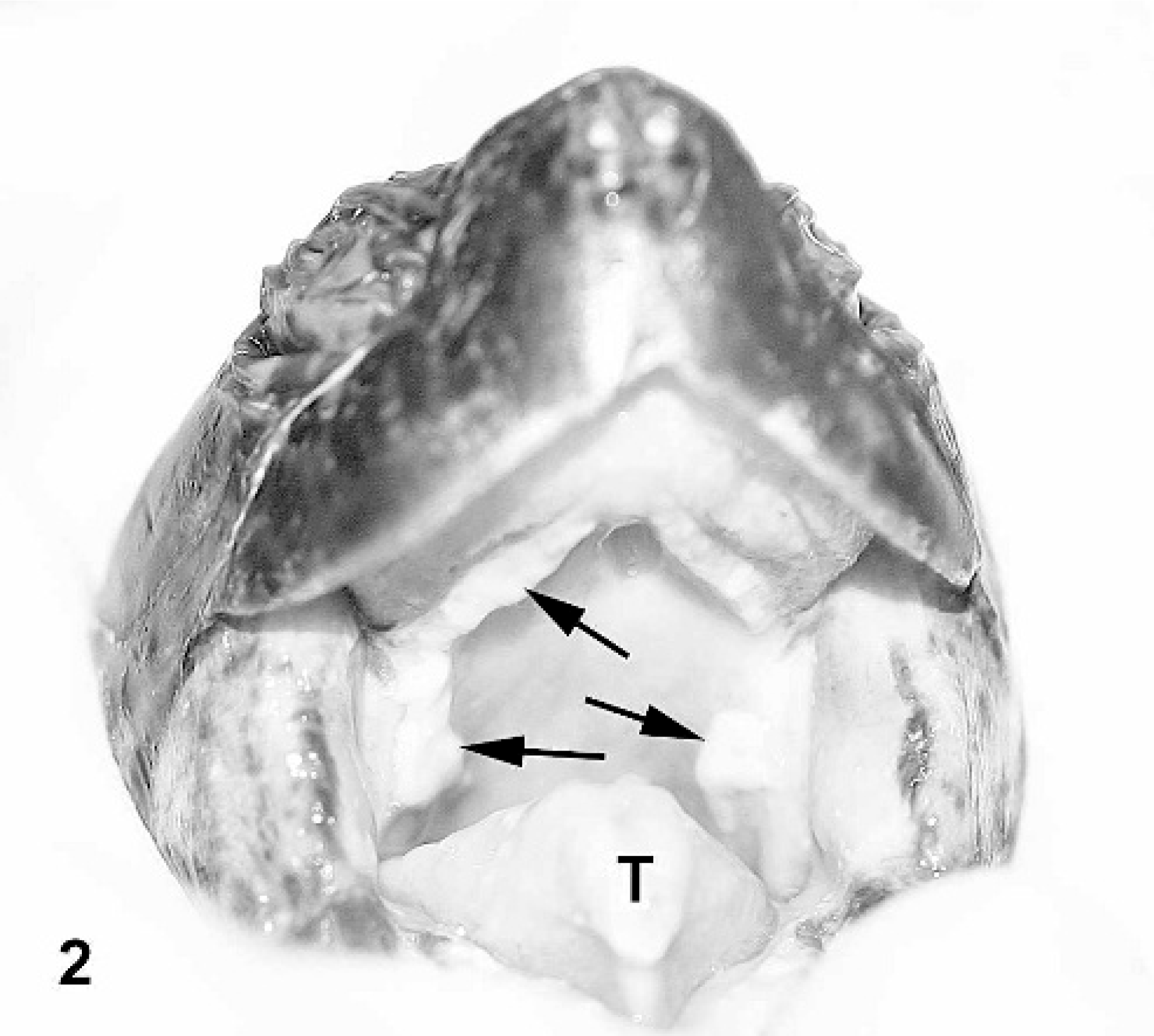
Red-eared slider (study 2, No. 6) intramuscularly inoculated with Ranavirus. Photograph taken 12 days postinoculation shows white caseous diphtheric plaques on the roof of the mouth and tip of the tongue (arrows and T).
Turtles mock inoculated were negative by PCR on all oral and cloacal swabs collected (Table 2). Three turtles PO inoculated (Nos. 2, 7, and 10) were positive by PCR on oral and/or cloacal swabs 2 days postinoculation but not in any subsequent samples. The fourth PO-inoculated turtle (No. 1) was not positive on any sample date. Oral and cloacal swabs from IM-inoculated turtles were positive by PCR for iridovirus at varying times throughout the study (Table 2). Turtle No. 8 (IM inoculated), which was euthanatized at the end of the study, was never positive on any oral swabs collected, but was positive on cloacal swab on 5 occasions, including the last two sampling dates, which were 23 and 26 days postinoculation. PCR on urine samples followed a similar pattern. Three of four IM-inoculated turtles were positive between 1 and 5 days prior to being euthanatized. One positive band from each turtle was sequenced to confirm the positive PCR results. All amplicons were of the expected size and each sequence shared 100% identity with the sequence of the original isolate. DNA extracted from 8 tissues from each turtle, including tongue, esophagus, stomach, small intestine, large intestine, kidney, spleen, and liver were positive by PCR on 3 of 4 IM-inoculated turtles (Nos. 3, 5, and 6) at necropsy (Table 1). Tissues from IM-inoculated turtle No. 8 and all PO-inoculated turtles and control mock-inoculated turtles were negative.
PCR results of oral and cloacal swabs taken on 11 different days postinoculation with an uninfected(control) or iridovirus-infected cell lysate throughout the 4-week full transmission study.
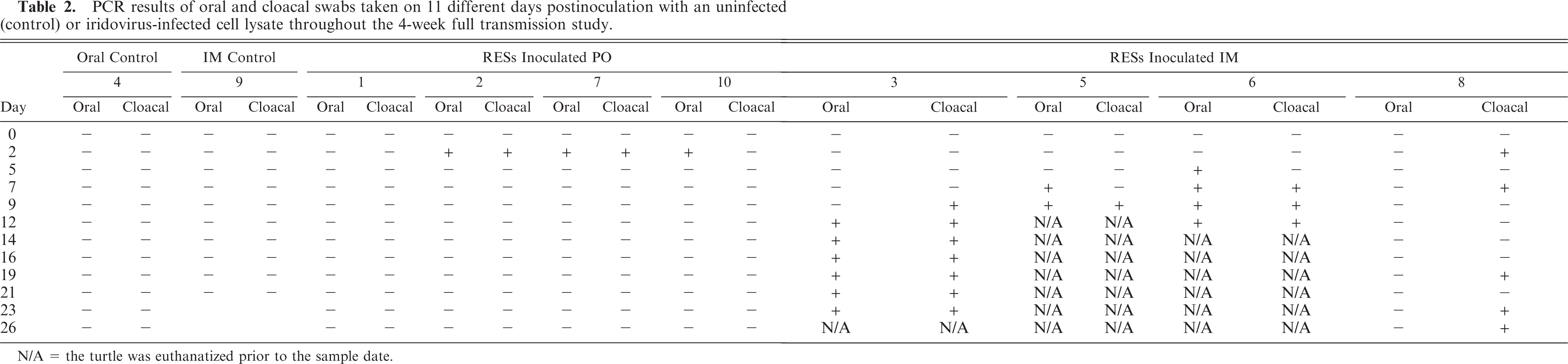
N/A = the turtle was euthanatized prior to the sample date.
At necropsy, gross changes were observed in several turtles. Turtle No. 3, which was IM inoculated, exhibited petechia in several organs, including the glottis, liver, pancreas, and fat. Congestion was observed in the stomach and on the surface of the bladder. The cecum and colon demonstrated a multifocal to coalescing area of hemorrhage. The gastrointestinal tracts of turtle Nos. 3, 5, and 6 were thickened and edematous. Turtle No. 6 also exhibited petechia on the surface of the pancreas and congestion in the stomach. No lesions were seen in PO-inoculated and control group turtles.
Significant histologic lesions were observed only in IM-inoculated turtles. Similar to the pilot study, the most consistent lesions were in the spleen (3 of 4 turtles) and centered on the splenic ellipsoids. Interpretation of changes was based on the normal histology of the spleen previously defined for RESs.28 The majority of the splenic white pulp surrounded ellipsoids (periellipsoidal lymphocyte sheath [PELS]) with lesser white pulp surrounding splenic arterioles (periarteriolar lymphocyte sheath [PALS]). Ellipsoids (Fig. 4) were characterized by plump, cuboidal, endothelial cells, a thick eosinophilic wall lacking smooth muscle (confirmed with Masson trichrome stain), and a lack of reticular fibers between lymphocytes of the PELS using Gordon and Sweet reticulin stain. In IM-inoculated turtles, the walls of the ellipsoids were moderately to markedly expanded by homogenous to slightly fibrillar eosinophilic material (fibrin, fibrinoid vasculitis; Fig. 5) with multifocal infiltrates of low numbers of heterophils and scattered free brown pigment granules (presumptively from disrupted melanomacrophage centers). There were occasional luminal fibrin thrombi with admixed heterophils and karyorrhectic debris (Fig. 5). There was mild to moderate lymphoid depletion and dispersion of lymphoid cells in the PELS, with relative sparing of lymphocytes in the PALS (Fig. 5). Replacing the PELS were combinations of karyorrhectic debris, heterophilic infiltrates, extravasated erythrocytes, and fibrin.
Spleen; Red-eared slider (study 2, No. 6). Turtle IM inoculated with Ranavirus. Two splenic ellipsoids and one splenic arteriole (arrow). The walls of the ellipsoids are expanded by fibrin, and there are luminal fibrin thrombi. Note the sparing of the splenic arteriole and associated lymphoid sheath [periarteriolar lymphoid sheath (PALS)]. HE. Bar = 50 μm.
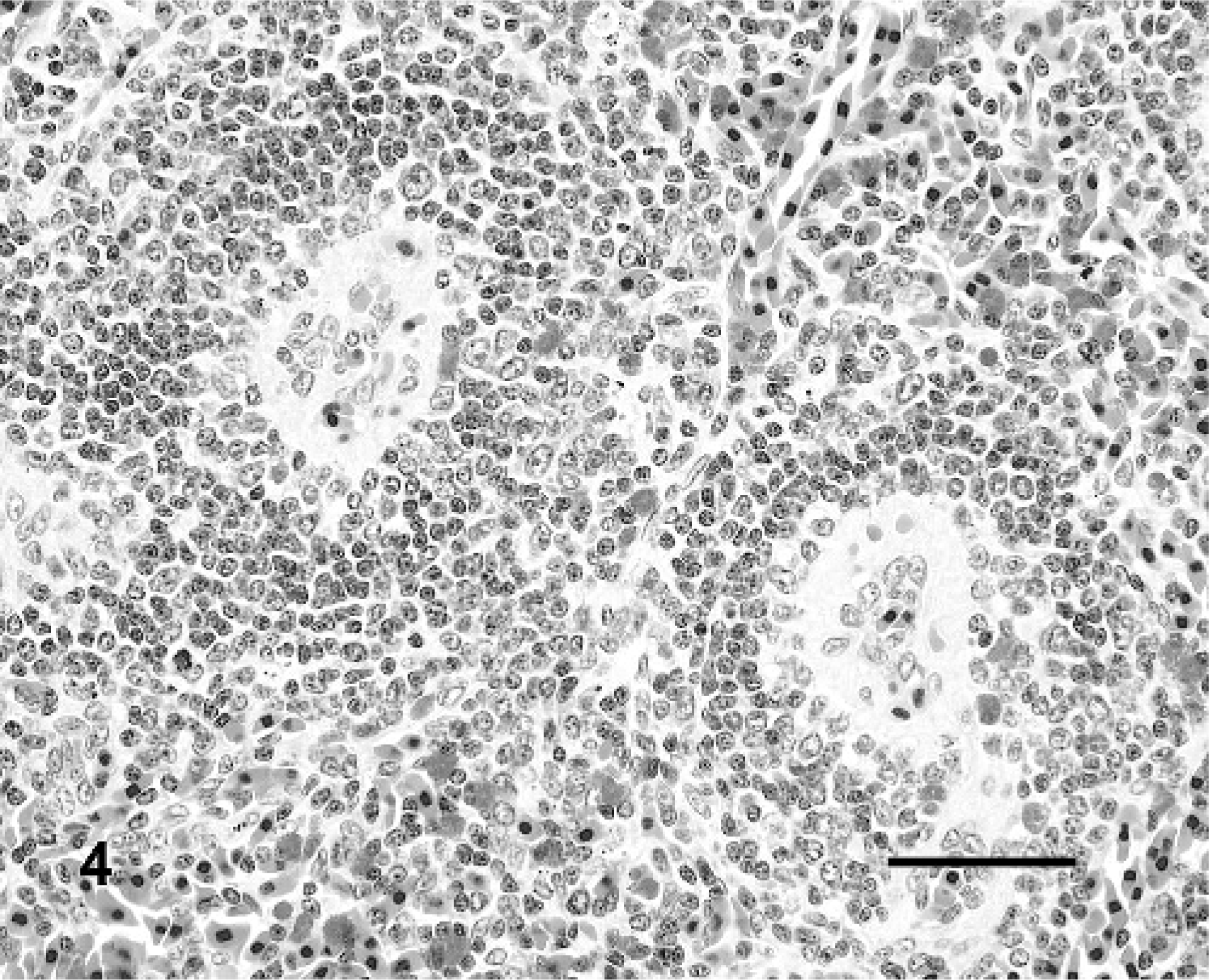
Spleen; Red-eared slider (study 2, No. 9). Mock-inoculated turtle. Two splenic ellipsoids and associated periellipsoidal lymphocyte sheathes (PELS). HE. Bar = 50 μm.
The livers of all turtles had mild to marked hepatocellular vacuolar change that was most pronounced in the mock-inoculated turtles (Fig. 6) and considerably less prominent in IM-inoculated turtles (depletion of hepatocellular lipid and glycogen; Fig. 7). Three (Nos. 3, 5, and 6) of four turtles had multifocal random dilatation of hepatic sinusoids by fibrin thrombi (Fig. 7) and variable single-cell necrosis of adjacent hepatocytes (Fig. 8). Rarely, hepatocytes had small intracytoplasmic basophilic inclusion bodies (Fig. 8). Admixed with fibrin thrombi and necrotic hepatocytes were small amounts of karyorrhectic debris, infiltrates of small numbers of heterophils and, for lesions occurring adjacent to melanomacrophage centers, small amounts of dispersed brown granular pigment. One turtle (No. 3) had moderate multifocal hemorrhage in association with liver lesions as well as moderate numbers of multifocal fibrin thrombi within small to medium portal venules and veins.
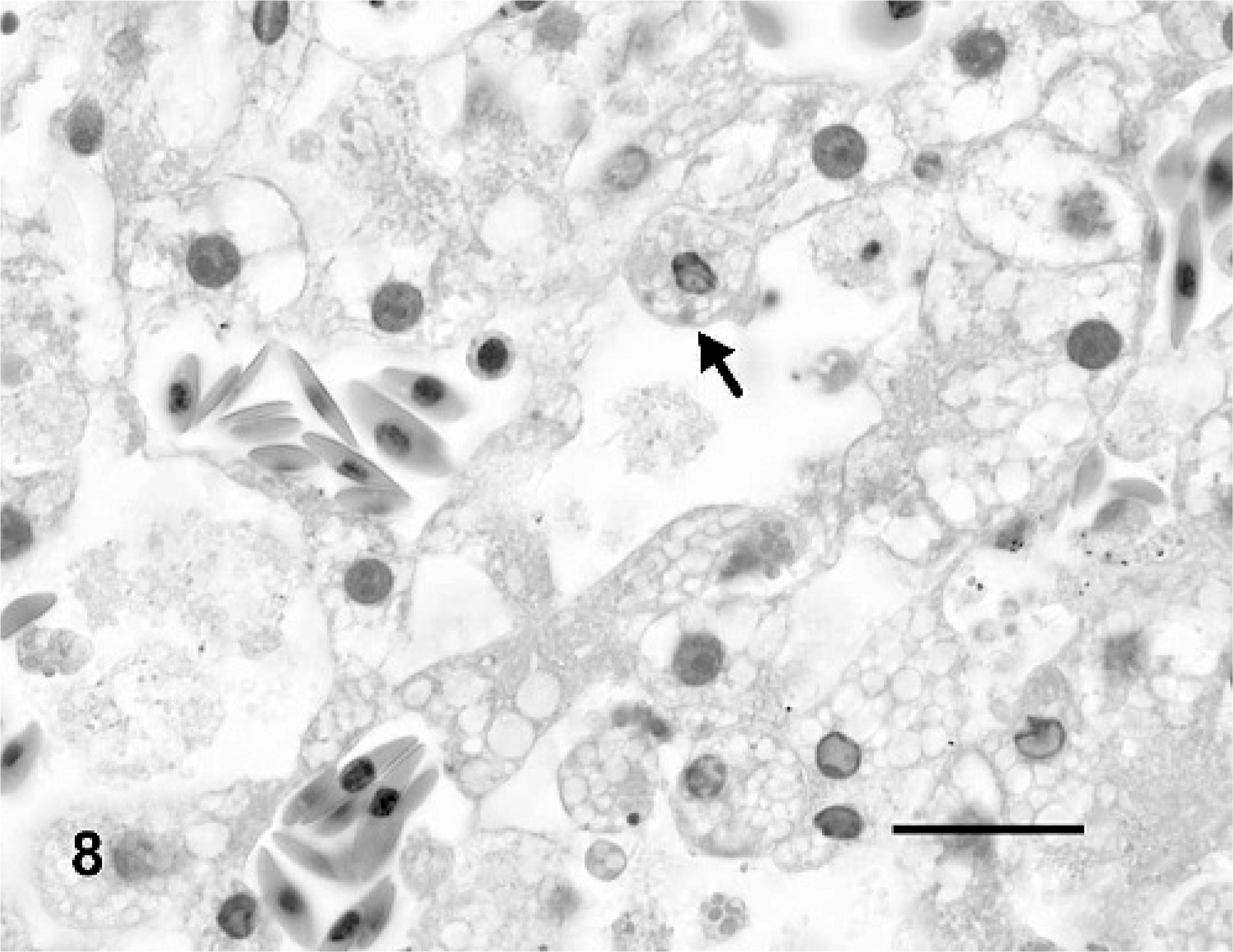
Liver; Red-eared slider (study 2, No. 6). Turtle intramuscularly inoculated with Ranavirus. There is single cell necrosis of hepatocytes with occasional intracytoplasmic basophilic inclusion bodies (arrow). HE. Bar = 20 μm.
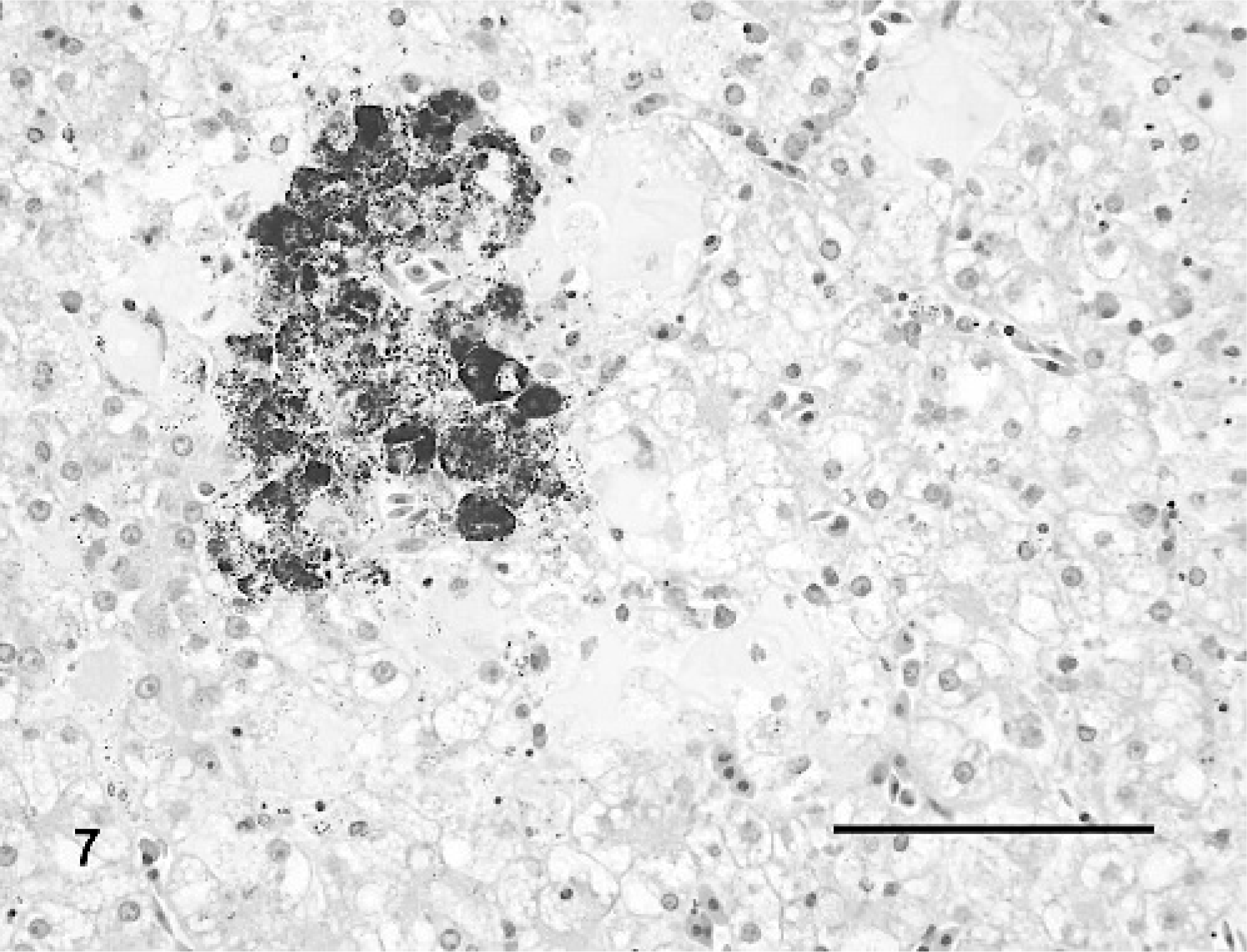
Liver; Red-eared slider (study 2, No. 6). Turtle intramuscularly inoculated with Ranavirus. There is depletion of normal hepatocellular lipid and glycogen. Sinusoids are multifocally expanded by fibrin thrombi with infiltrates of low numbers of heterophils. The melanomacrophage center is disrupted. HE. Bar = 100 μm.
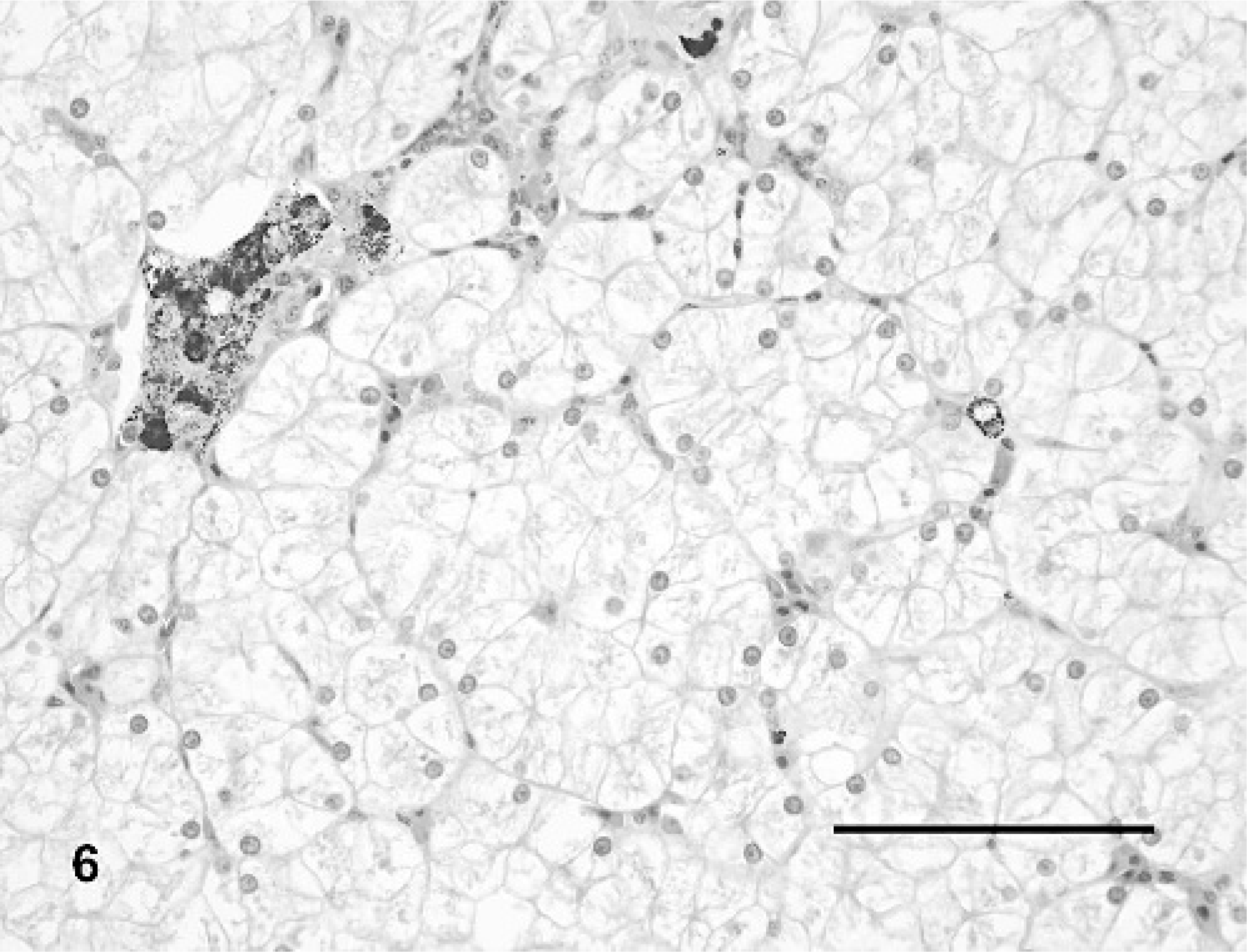
Liver; Red-eared slider (study 2, No. 9). Mock-inoculated turtle. There is marked diffuse vacuolation of hepatocytes. A melanomacrophage center is present in the upper left. HE. Bar = 100 μm.
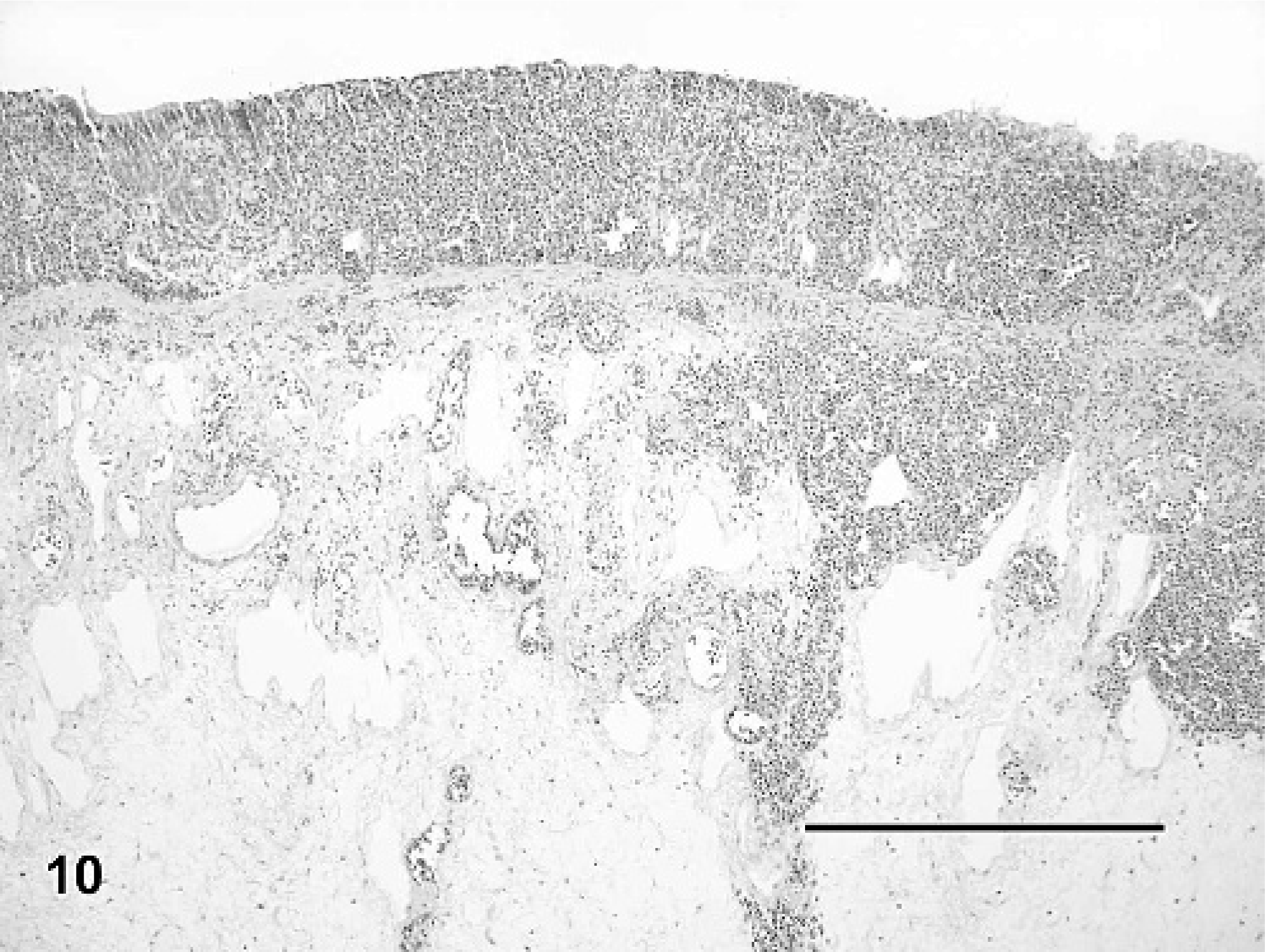
In addition to vascular changes in splenic ellipsoids, hepatic sinusoids, and portal blood vessels, fibrin thrombi also were observed in a variety of other tissues in IM-inoculated animals (Fig. 9). Thrombi were noted in gastric or intestinal lamina propria, submucosa, and serosa (Nos. 3, 5, and 6), glomerular capillaries (Nos. 3 and 6), esophagus (No. 3), pulmonary capillaries and veins (Nos. 3 and 6), meninges (Nos. 3 and 6), eye (No. 3), nasal mucosa (No. 3), and oral mucosa (No. 3). Lesions associated with thrombi included segmental marked colonic mucosal and submucosal hemorrhage (Figs. 10, 11), multifocal mild to moderate meningeal hemorrhage, mild to moderate heterophilic meningitis, and mild heterophilic interstitial pneumonia.
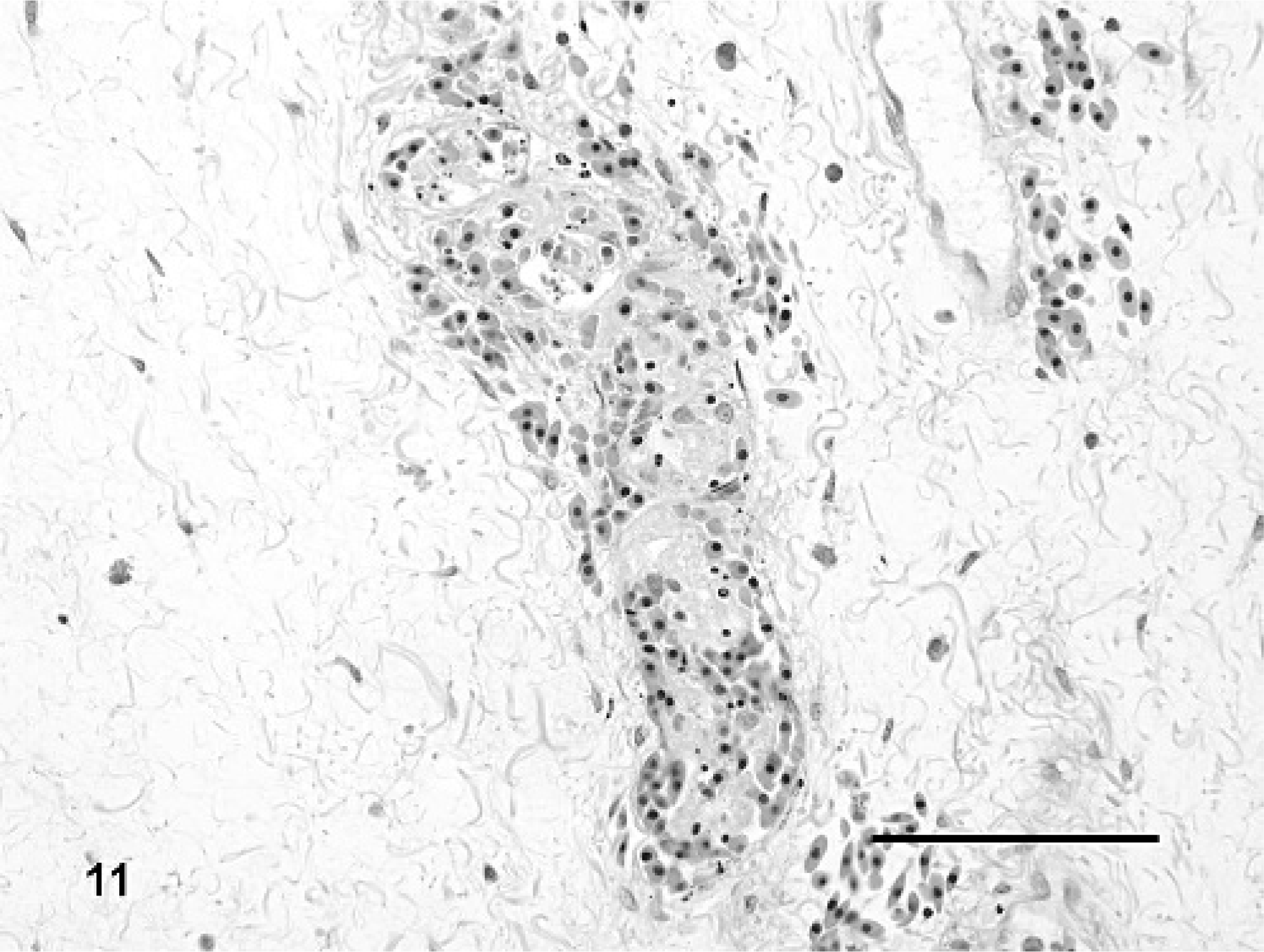
Colon, submucosal blood vessel; Red-eared slider (study 2, No. 3) intramuscularly inoculated with Ranavirus. There is a luminal fibrin thrombus with admixed karyorrhectic debris and perivascular edema and hemorrhage. HE. Bar = 100 μm.
Colon; Red-eared slider (study 2, No. 3) intramuscularly inoculated with Ranavirus. There is mucosal and superficial submucosal hemorrhage with marked submucosal edema. HE. Bar = 1.0 mm.
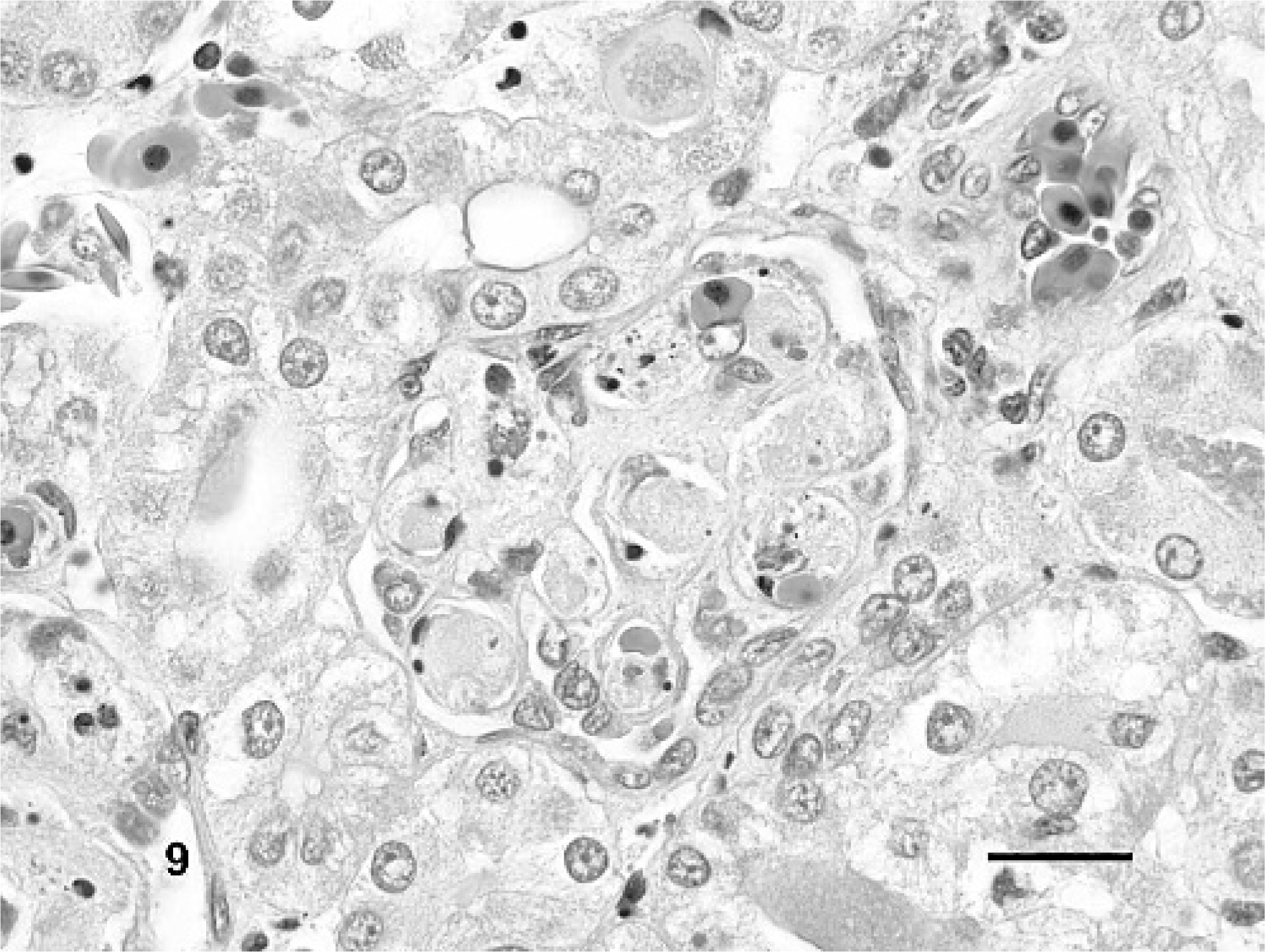
Kidney; Red-eared slider (study 2, No. 6). Turtle intramuscularly inoculated with Ranavirus. There are fibrin thrombi in glomerular capillary loops admixed with few heterophils and small amounts of karyorrhectic debris. HE. Bar = 20 μm.
A small number of IM-inoculated turtles also had multifocal mild to moderate ulcerative and heterophilic stomatitis and esophagitis (No. 6; Fig. 12), and multifocal mild to moderate heterophilic and necrotizing gastritis (Nos. 5 and 6). Turtles from all three groups had small granulomas in a wide variety of tissues that surrounded spirorchid-type trematode eggs and, rarely, adult trematodes were observed within mesenteric blood vessels. Granulomas in the gastric submucosa and serosa of a small number of control and virus-inoculated turtles contained an unidentified nematode. Trematodes and nematodes were interpreted as incidental findings.
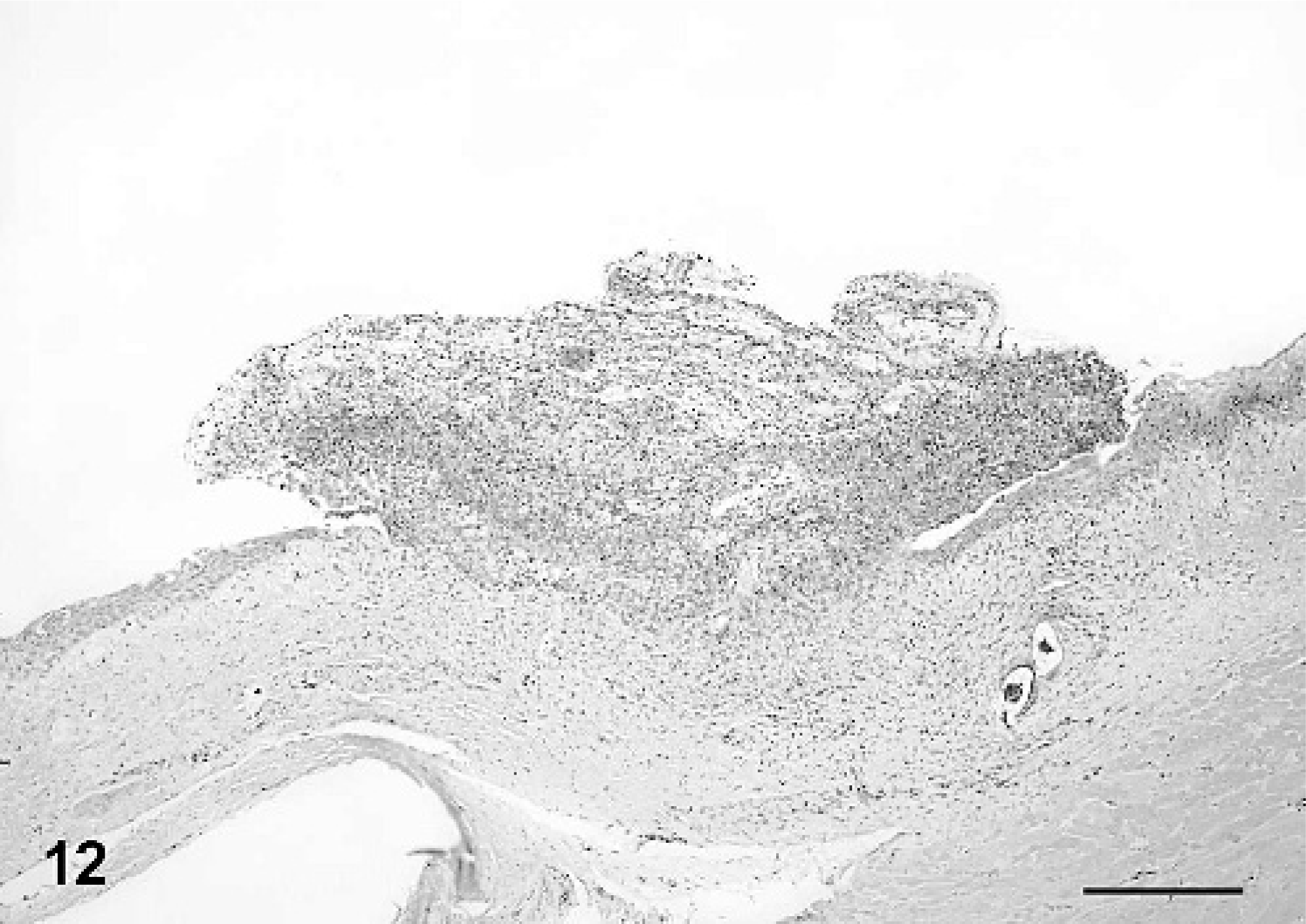
Oral mucosa; Red-eared slider (study 2, No. 6) intramuscularly inoculated with Ranavirus. There is focal mucosal ulceration and replacement by a mat of heterophils and fibrin. HE. Bar = 200 μm.
Similar to study 1, cytopathic effects consisting of cell rounding and lysis were seen in cultures of Terrapene heart cells that were coated with kidney homogenates from 3 of 4 IM-inoculated turtles (Nos. 3, 5, and 6). Using PCR and nucleotide sequencing, Ranavirus was identified in DNA extracted from cells from each flask. No CPE was seen in cells that received tissue homogenates from orally inoculated turtles, control turtles, and one of the IM-inoculated turtles (No. 8).
Transmission electron microscopy of splenic ellipsoids in 1 IM-inoculated turtle (No. 6) in the full transmission study demonstrated marked expansion of the vessel wall by a granular to fibrillar lightly electron-dense material that was consistent with fibrin. Admixed with fibrin and within remnants of the white pulp were scattered unidentified necrotic cells with intracytoplasmic arrays of icosahedral virions consistent in size (approximately 130 nm in diameter) and shape (Fig. 13) with iridoviruses.43 No virions were observed in cells associated with ellipsoids in the control turtle (No. 9).
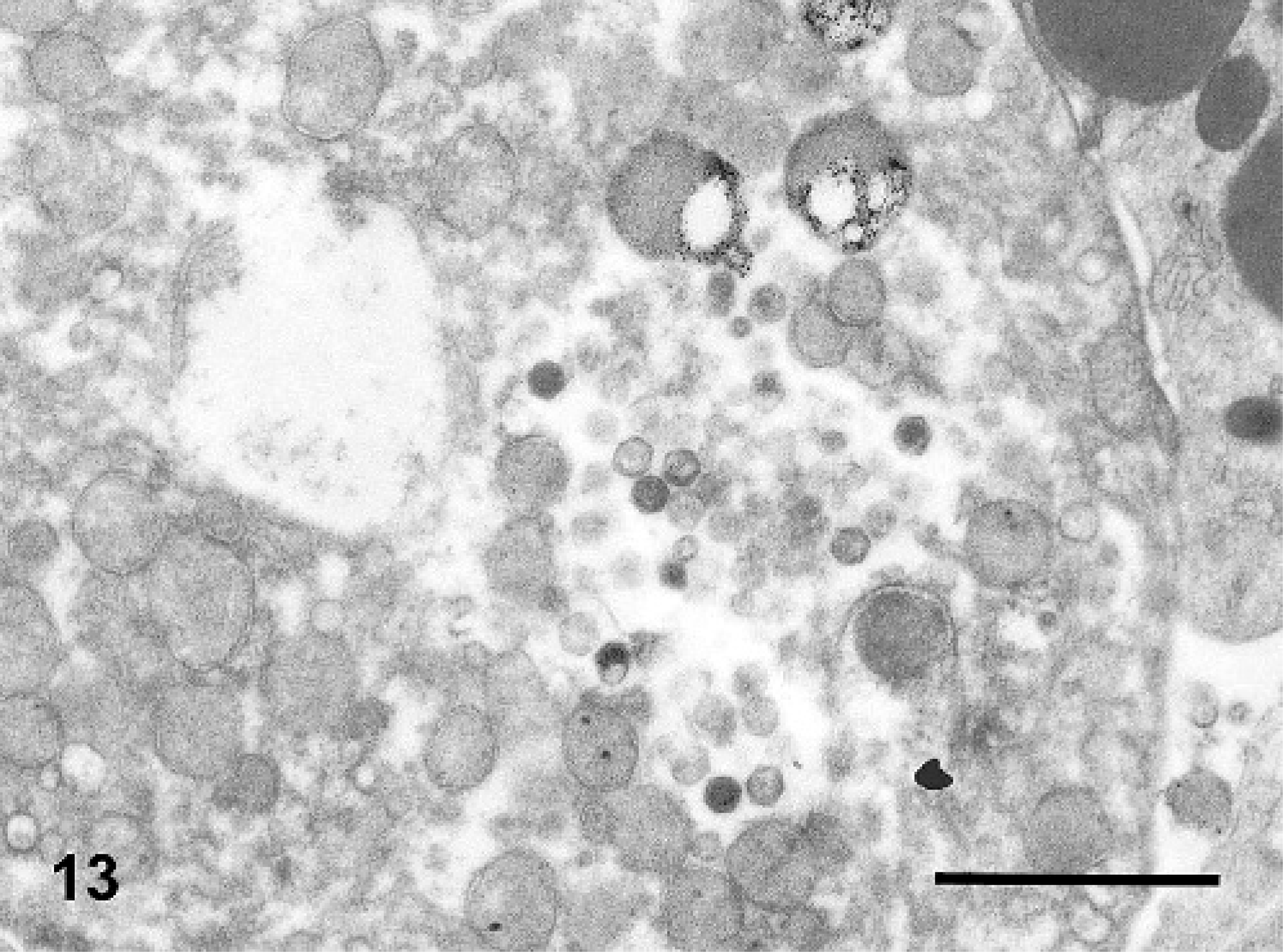
Spleen; Red-eared slider (study 2, No. 6). Turtle intramuscularly inoculated with Ranavirus. Transmission electron photomicrograph showing intracytoplasmic, icosahedral virions consistent with iridoviruses. Bar = 1000 nm.
Discussion
Henle-Koch's postulates provide a strict set of guidelines for proving a causal relationship between an infectious agent and a disease.17 Here we performed a transmission study using a Ranavirus isolate from a captive Burmese star tortoise that became ill and died in an attempt to determine whether a causal relationship exists between infection with this Ranavirus and the clinical and histologic changes observed in the Burmese star tortoise. Since it would be inappropriate to perform a challenge study in this critically endangered species, we decided to assess both BTs and RESs as a model for Ranavirus infection in chelonians. BTs were selected because Ranavirus infection has been identified in this species.14,24,33 We decided also to evaluate the suitability of RESs, since BT populations are declining throughout their range. RESs, however, are plentiful, since they are being raised commercially for the pet trade, which became a factor in their ultimate selection as an experimental animal in our studies. In addition, results of the pilot study showed that both species responded similarly when administered a Ranavirus isolate by two different routes. Both IM-inoculated turtles showed severe clinical signs and were euthanatized prior to the termination of the study at 2 weeks. Oral swabs from both were positive 1 week postinoculation, and histologic lesions were consistent between the two species. Both the PO-inoculated RES and BT did not develop any clinical signs or histologic lesions. Results of this study indicated that both RESs and BTs can serve as suitable models of Ranavirus infection for chelonians. More importantly, this study demonstrated the ability of this virus to infect and cause severe debilitating ranaviral disease in three different chelonid species, suggesting a broad host range for this virus, and thus the potential to cause significant disease and mortality in captive and free-ranging chelonians. Ranaviral infection associated with mortality in other species of chelonians has been previously described,14,24,35,42 suggesting that all species of chelonians should be considered susceptible to ranaviral disease.
Histologic lesions in turtles inoculated by IM injection in both studies consisted primarily of multicentric fibrinoid vasculitis and formation of fibrin thrombi in small blood vessels in numerous tissues, and in this regard resembled recent reports of Ranavirus infection in captive and free-ranging box turtles and tortoises.14,24 Lesions in blood vessels were consistent with observed Ranavirus infection of endothelial cells in a naturally infected captive Burmese star tortoise24 and with descriptions of apparent viral endotheliotropism in rainbow trout and redfin perch infected with the Ranavirus epizootic hematopoietic necrosis virus (EHNV).39 Involvement of endothelial cells also may be part of the pathogenesis of some amphibian Ranavirus infections, as suggested by multicentric hemorrhage and edema or observation of characteristic inclusion bodies within endothelial cells.3,12,15,44
The consistent involvement of the splenic ellipsoids (sheathed capillaries) provides a basis for the prominent necrotizing splenitis observed in some natural Ranavirus infections of chelonians.14,24 Similar lesions of the ellipsoids were observed in redfin perch, but not rainbow trout, experimentally infected with EHNV.39 A filtering function for splenic ellipsoids for particulate material and immune complexes has been documented in other species,41 and the lesions observed in chelonian Ranavirus infections could possibly be a consequence of antigen trapping in macrophages associated with the ellipsoid sheath during ranaviral viremia.
Necrotizing stomatitis, esophagitis, and/or pharyngitis are characteristic lesions in many naturally occurring chelonian Ranavirus infections.14,24,34,42 Clinical signs such as ocular and nasal discharge, conjunctivitis, and palpebral edema associated with Ranavirus infection (both naturally infected animals and the experimental animals in this report) overlap with those signs caused by infections of Mycoplasma agassizii 4 or tortoise herpesviruses.25,37 In particular, infection with tortoise herpesviruses 1 and 2 is associated with caseous or diphtheritic oral plaques that are grossly indistinguishable from oral lesions associated with Ranavirus infection. In the experimentally infected animals of this report, necrotizing stomatitis and esophagitis were observed in a single IM-inoculated RES (No. 6). Lesions were not observed in the nasal cavity of any animal examined. Oral lesions could be secondary to thrombus formation and infarction in small submucosal vessels or, alternatively, could be the result of viral infection and necrosis of oral epithelial cells. The observation of intracytoplasmic inclusion bodies consisting of Ranavirus in epithelial cells of some naturally occurring infections would appear to support the latter explanation. The reasons that oral lesions were not present in more of the experimentally inoculated turtles in this report is uncertain, but it is possible that epithelial infection is usually a late event that follows viremia, and hence was observed only in 1 turtle over the course of this study. Also, the virus may have been attenuated in cell culture, resulting in altered pathogenicity.
The intracytoplasmic basophilic inclusion bodies that are suggestive of iridovirus infection and prominent in many cases of Ranavirus infection in fish, amphibians, and some chelonians were not prominent in the experimentally inoculated turtles.3,15,34,39,42 This observation is consistent with recently reported naturally occurring chelonian Ranavirus infections, and indicates that inclusion bodies may be an inconsistent finding and should not be relied upon for use in formulating a histologic differential diagnosis.14,24 Virions consistent with ranaviruses were observed by TEM in cells within the spleen of an IM-inoculated RES, which suggests that TEM remains a useful diagnostic tool in chelonian Ranavirus infections even in the absence of visible inclusions on histologic section. Demonstration of intracytoplasmic virions in cells of an experimentally inoculated turtle is important because it shows that the virus is capable of entering and replicating within cells and that lesions were not induced by the presence of inoculated nonreplicating virus. Necrotizing liver lesions have been experimentally induced in mice and rats following injection of inactivated iridovirus virions or solubilized structural proteins.31 Similarly, the Ranavirus Frog Virus 3 can trigger apoptosis in tissue culture in the absence of viral gene expression.9 Future studies may better define the in vivo mechanism of cell death associated with iridovirus infections.
This study found that IM-inoculated turtles were more likely to become infected with Ranavirus, show clinical signs, and die compared with turtles that were orally inoculated. All 4 IM-inoculated turtles showed clinical signs, and 3 (75%) died as a result of infection, whereas no orally inoculated turtles showed any signs of disease or died. This could mean that turtles do not become exposed through ingestion of infected animals or water sources, as has been previously shown to occur in amphibians,23,38 or that abrasions naturally acquired from ingesting bones or other abrasive material may be necessary for virus to be introduced systemically. Another explanation for the inability to re-create disease in orally inoculated animals was that a natural exposure was not replicated in the laboratory setting. Viral dose administered was extrapolated from studies done with fish and amphibians,3,11,29,36 but requirements for infection of turtles may be higher or repeated exposure may be necessary. Experimental infections of salamanders with a Ranavirus showed that both dose and host characteristics influenced the virulence of infection.5 The infection dose was positively correlated with the mortality rate and inversely related to average survival times. Environmental temperatures also have been shown to significantly impact the percent mortality and time to death in salamanders experimentally inoculated with a Ranavirus,40 where salamanders inoculated at 18°C and 10°C were more likely to die than those exposed at 26°C. While water and room temperatures averaged between approximately 21°C and 25°C, basking areas were kept warmer at 28°C. Eliminating heat lamps over basking areas and lowering the room temperature might have kept turtles cooler, and altered the results in the orally inoculated group. Alternatively, other routes of transmission, such as vector-born transmission, may be required for turtles to become infected in the wild. Intracytoplasmic inclusion bodies were identified recently in the circulating leukocytes of an eastern box turtle infected with iridovirus.1 Ranaviruses are variably host specific, so virus may be able to survive in mosquitoes or other biting insects capable of transmitting virus from an infected animal to an uninfected one. This has been shown to be the case with insect iridoviruses, where parasites were experimentally shown capable of transmitting invertebrate iridescent virus from infected to uninfected larvae.30 It remains unknown whether natural outbreaks of iridovirus infections in any vertebrate species occur as a result of introduction of novel virus strains, recrudescence of latent or persistent infections in surviving populations as a result of stressors or other immunosuppressive causes, or viral persistence in the environment.43
Although the mechanism of transmission of iridoviruses in natural settings is unknown, it has been shown that sublethally infected amphibians can cause sporadic, recurrent disease outbreaks in amphibians.6 Experimentally and naturally infected tiger salamander larvae and metamorphs were able to maintain sublethal, transmissible infections for over 5 months. Apparently healthy infected dispersing metamorphs were returning to water bodies to breed, and it was speculated that these individuals were likely serving as a reservoir host for infecting newly hatched larvae, creating recurrent outbreaks of disease. The current study showed that turtles may also become asymptomatic carriers, although further studies might help to confirm this finding. Turtle No. 8, which was inoculated intramuscularly, showed transient signs of disease but then recovered. At necropsy, tissues collected from 8 different organs were negative for iridovirus on PCR. Kidney samples inoculated onto TH-1 cells showed no cytopathic effects. However, cloacal swabs collected 1, 4, and 8 days prior to necropsy were positive using PCR. If this was a result of slow elimination of the virus, all orally inoculated turtles should have had PCR-positive cloacal samples after 2 days postinoculation. However, all swabs taken after this time period were negative. While it is possible that this was a laboratory contaminant, all swabs were extracted and tested by PCR according to date collected, as opposed to all swabs being tested at the same time for each turtle. If there was contamination, we would not expect to see it on 5 occasions from 1 turtle, and not from any orally inoculated turtles on any date past 2 days postinoculation. Therefore, it seems more likely that virus shedding was occurring, and the site of virus replication was not detected. Some viruses show a predilection for specific cells, such as infectious bursal disease virus in chickens, where virus shows a predilection for the cells of the bursa of Fabricius, located in the cloaca.7 Further long-term studies would help to confirm whether Ranavirus persistence occurs, and immunohistochemical or in situ hybridization studies of tissues from infected turtles may help identify the tissue tropism for Ranavirus persistence. If turtles can serve as asymptomatic carriers, they may also serve as a reservoir host of virus for other turtles and other susceptible species. Interclass infections of iridovirus have been shown naturally and experimentally in sympatric species of fish and amphibians where both were capable of being infected with the same virus.32,36 In another study (data not shown), the isolate used in this experimental study was capable of infecting leopard frogs (Rana pipiens) that were injected intraperitoneally at the same dose. Thus, sublethally infected turtles, such as one of the turtles (No. 8) in our study, could serve as a reservoir host for amphibian populations in geographic locations where the species overlap.
In summary, the experimental inoculation of a Ranavirus in chelonians was investigated. Koch's postulates were fulfilled when IM inoculation of virus into naive turtles resulted in clinical and histologic changes consistent with those seen in natural infections, and when the same virus was subsequently recovered. Since oral inoculation failed to result in disease or mortality, the natural route of transmission in the wild remains unknown. The immune system of reptiles is temperature dependent,10 and perhaps by manipulating the environmental temperature and the temperature of the host, the susceptibility of turtles to infection can be altered. Another possibility that should be investigated is the transport and inoculation of Ranavirus into chelonians by arthropods. This study also suggests that sublethally infected turtles may serve as reservoir hosts of infection for other chelonians as well as amphibians. Ranaviruses are considered a global threat to amphibian populations based on the lack of host specificity, high virulence, and global distribution,13 and this study confirms that they should likewise be considered a threat to chelonian populations.
Footnotes
Acknowledegments
This work was funded by grant no. D04Z0-11 from the Morris Animal Foundation. We would like to thank Yvonne Cates for excellent histology support, and Kevin Chadbourne and April Childress for husbandry and laboratory support.
