Abstract
Juvenile eastern water dragons (Intellagama lesueurii lesueurii) are highly susceptible to infection with Bohle iridovirus (BIV), a species of ranavirus first isolated from ornate burrowing frogs in Townsville, Australia. To investigate the progression of BIV infection in eastern water dragons, 11 captive-bred juveniles were orally inoculated with a dose of 104.33 TCID50 and euthanized at 3, 6, 8, 10, 12, and 14 days postinfection (dpi). Viral DNA was detected via polymerase chain reaction (PCR) in the liver, kidney, and cloacal swabs at 3 dpi. Mild lymphocytic infiltration was observed in the submucosa and mucosa of the tongue and liver at 3 dpi. Immunohistochemistry (IHC) first identified viral antigen in foci of splenic necrosis and in hepatocytes with intracytoplasmic inclusion or rare single-cell necrosis at 6 dpi. By 14 dpi, positive IHC labeling was found in association with lesions in multiple tissues. Selected tissues from an individual euthanized at 14 dpi were probed using in situ hybridization (ISH). The ISH labeling matched the location and pattern detected by IHC. The progression of BIV infection in eastern water dragons, based on lesion severity and virus detection, appears to start in the spleen, followed by the liver, then other organs such as the kidney, pancreas, oral mucosa, and skin. The early detection of ranaviral DNA in cloacal swabs and liver and kidney tissue samples suggests these to be a reliable source of diagnostic samples in the early stage of disease before the appearance of clinical signs, as well as throughout the infection.
Ranaviruses infect a wide range of ectothermic vertebrates worldwide and are considered emerging pathogens of significant ecological importance in amphibians and fish. 28 Lately, ranaviruses have been detected at an increasing frequency in reptiles and are associated with mortality events in captive lizards belonging to the Agamidae, Anguidade, and Iguanidae families. 8,33,34
Several studies have documented the infection potential of individual ranaviral isolates to hosts in different classes of lower vertebrates. 7,9,10 Studies exploring the effects of temperature, dose, and exposure route have been conducted in amphibians and turtles using frog virus 3 (FV3), 2,17 in amphibians using Ambystoma tigrinum virus (ATV), 12 and in fish, amphibians, and reptiles using Bohle iridovirus (BIV). 4,5,24,27,32 Several species of fish, amphibians, and turtles shown to be susceptible to BIV have an overlapping geographic distribution with eastern water dragons (Intellagama lesueurii lesueurii). 11 This semiaquatic squamate is a strong swimmer often found in large numbers along freshwater rivers and creeks on the east coast of Australia. 11 Eastern water dragons (EWDs) are also common in the pet trade, and although viruses (eg, adenovirus and ranavirus) are commonly identified in captive lizards and other ectotherms held in collections all over the world, 15,34 natural ranaviral infection has not been reported in captive or wild EWD populations to date. Experimental exposure of EWDs to BIV, a ranavirus originally isolated from ornate burrowing frogs (Limnodynastes ornatus) in Townsville, Australia, showed that the lizards were highly susceptible to infection via intramuscular and oral exposure and developed histologic lesions of necrosis, hemorrhage, and inflammation in the pancreas, liver, spleen, kidney, and gastrointestinal tract. 24 However, the progression and effect of BIV in juvenile EWDs are not understood. Here we describe the pathogenesis of experimental BIV infection in juvenile EWDs via time-course sampling of individual animals to identify the progression of infection from inoculation to euthanasia.
Materials and Methods
Eleven captive-bred EWDs were purchased from a commercial breeder and kept under permit (Scientific Purposes Permit #WISP15053914) from the Queensland Department of Environment and Heritage Protection at the College of Public Health, Medical and Veterinary Sciences, James Cook University (JCU). Husbandry and experimental and sampling procedures were carried out with approval from the JCU Animal Ethics Committee (Ethics Approval #A2277). At the start of the trial, dragons were 10.5 months old and weighed 11.9 to 18.1 g (mean, 14.5 g). Sex was determined only after euthanasia, during necropsy examination (5 females, 4 males, 2 unknown). The data analyzed in this study are available upon request to the authors.
Husbandry of Experimental Dragons
Dragons were housed individually for 20 weeks in 5-L plastic vivariums with a small plastic pipe hide and a water dish. After acclimation, 13 dragons were randomly assigned to either infection groups (n = 11) or served as negative controls (n = 2) and were moved to larger 20-L group tanks (2–4 dragons per tank). Dragons were uniquely identified using a nontoxic marker (Duramark; Staples, Stuttgart, Germany) on the dorsal skin and were permitted a further 14-day acclimation period within these groups prior to inoculation. Negative control (NC) dragons were housed in a quarantine room and the BIV-infected dragons in the adjoining infection room. Room conditions were controlled with a 12-hour dark and 12-hour light cycle, and temperatures were recorded twice daily (average, 28.8°C; range, 26.3°C–30.1°C).
The dragons were fed a diet of small crickets 3 times weekly, where 2 of the 3 feeds were dusted with Multical Dust supplement (Vetafarm, New South Wales, Australia). Weight was recorded weekly to ascertain steady growth.
BIV Viral Stock
The viral strain used in this study was the 1992 BIV isolate from ornate burrowing frogs that was isolated at JCU 32 and subsequently sequenced 19 (GenBank accession number KX185156). This strain was propagated at 25°C in fathead minnow (FHM) cells, in Dulbecco’s modified eagle medium (DMEM; Thermo Fisher Scientific, NY, USA), supplemented with 100× antibiotic-antimycotic (Thermo Fisher Scientific, NY, USA) and 10% fetal bovine serum (Bovogen Biologicals, Melbourne, Australia). Titration of viral stock was performed in a 96-well tissue culture plate following the standard methods to determine the tissue culture infection dose (TCID50). 30
Animal Inoculation and Sampling
Time from infection to euthanasia (end point) in this experimental infection study was predetermined and ranged from 3 to 14 days postinfection (dpi). This was based on a previous infection study of BIV in juvenile EWDs, where the onset of clinical signs and death via oral exposure occurred at 8 to 11 dpi and 9 to 14 dpi, respectively. 24 The same method of exposure and viral titer was used in this study due to its effectiveness at causing acute disease, and the predetermined end points allowed us to cover the early stages of infection and euthanize the dragons at a predefined humane end point. To reduce the number of dragons needed for this study, tissues collected from 3 dragons involved in a previous study, 24 infected and housed in the same conditions as those in the present study, were included in the analysis. These 3 dragons were euthanized at corresponding time points (1 at 12 dpi and 2 at 14 dpi), received an equivalent oral dose of BIV, and were housed in similar conditions.
After the 14-day acclimation period, dragons in the infection treatments were orally inoculated with 100 μl (104.33 TCID50) BIV using a syringe placed at the back of the oral cavity. NC dragons received 100 μl orally of phosphate-buffered saline. Dragons were examined twice daily for any behavioral change (eg, decreased activity) and clinical signs previously described 24 (ie, distended abdomen, loss of appetite, decreased activity, loss of startle and rollover reflexes, loss of equilibrium, and focal areas of skin ulceration or pustules). Consumption of food and changes in appetite were recorded and water was refreshed daily. A cloacal swab was collected at –1, 3, 6, 8, 10, 12, and 14 dpi; stored at –80°C in 1 ml DMEM (Thermo Fisher Scientific, United States) supplemented with 100× antibiotic-antimycotic (Thermo Fisher Scientific, United States), and later tested for ranaviral DNA by polymerase chain reaction (PCR).
The dragons were euthanized at predetermined end points: 3, 6, 8, 10, 12, and 14 dpi, to record the time-course progression of infection. Two dragons were euthanized at each of the following time points: 3, 6, 8, 10, and 12 dpi; 1 dragon was euthanized at 14 dpi. The dragon to be euthanized at each time point was determined by random selection prior to commencing the experiment. The 2 NC dragons were euthanized at the end of the trial (14 days). Euthanasia was a 2-stage method using tricaine methanesulfonate (MS222). 14 Briefly, lizards were injected intracoelomically (IC) with 250 mg/kg 1% MS222 (stage 1). Once it was confirmed that the dragon had lost its toe-pinch reflex, lizards were injected IC with 0.4 to 0.5 ml 50% (v/v) MS222 (stage 2). Immediately after euthanasia, a dorsal and ventral photograph was taken, and any lesions or abnormalities were documented. During necropsy, small tissue samples were aseptically obtained from the liver and kidney, frozen at –80°C, and later used for PCR and viral isolation.
Although no testing was performed prior to inoculation with BIV, we are confident of the naive status of all dragons since (1) all dragons originated from the same captive breeding facility where no outbreaks have ever been detected, and (2) the dragons kept as uninfected (controls) were all negative for ranavirosis based on histopathology and PCR results.
Histopathology
A cross section of the proximal limb for normal skin and skin samples with gross lesions, lung, liver, pancreas, spleen, kidney, digestive tract, heart, tongue, brain, adrenal gland, reproductive organs, and bone marrow were preserved in 10% neutral buffered formalin. Tissues were processed routinely for histological examination and stained with hematoxylin-eosin. 6 Sections that included bone (toes, feet, legs, spine, and head) were trimmed and placed in Gooding and Stewart’s decalcifying fluid for 24 hours prior to processing. 6 The histopathology and immunohistochemistry (IHC) results from 3 orally exposed dragons from a previous study of BIV in juvenile EWDs were included. 24
Immunohistochemistry and In Situ Hybridization
Formalin-fixed, paraffin-embedded tissues from individuals euthanized at 3, 6, 8, 10, 12, and 14 dpi were immunolabeled for Ranavirus sp antigen using a polyclonal rabbit antibody against a member of the Ranavirus genus: epizootic hemorrhagic necrosis virus (EHNV). 29 Anti-EHNV antibodies are known to cross-react with other viruses in the genus. 3,20 In an automated staining system (BOND-MAX; Leica Microsystems, Buffalo Grove, IL), sections were labeled using a previously validated protocol for affinity-purified rabbit anti-EHNV (lot M708, OIE Reference Laboratory for EHN Virus, University of Sydney) IHC staining. Briefly, slides were dewaxed with Bond Dewax Solution (cat. AR9222; Leica Microsystems). A Heat Epitope Retrieval with Bond Epitope Retrieval Solution 1 was applied for 30 minutes (cat. AR9961; Leica Microsystems). Rabbit anti-EHNV antibody diluted at 1:3000 was applied to slides for 15 minutes. A polymer (secondary antibody, anti-rabbit poly-HRP-IgG; cat. DS9390; Leica Microsystems) was applied for 10 minutes. Leica Bond Polymer Refine Red Detection was applied for 15 minutes (cat. DS9390; Leica Microsystems). Finally, the tissue was counterstained with hematoxylin applied for 5 minutes (cat. DS9390; Leica Microsystems). Control slides were processed with the above method but omitting the primary antibody. A tissue was considered positive if strong and distinct staining with anti-EHNV (Ranavirus sp) antibody was present in the cytoplasm of 1 or more cells and background staining was either absent or clearly distinct from true specific staining. Diffuse pale staining of a specific type of tissue or cell (eg, stomach glandular epithelium) was considered background artifactual staining.
Selected tissues (skin, bone marrow, liver, pancreas, stomach, intestine, and spleen) from an individual euthanized at 14 dpi were labeled with an in situ hybridization probe (ACDBio RNAscope Probe- V-FV3-orf90 R, cat. 439991) for Frog Virus 3 (GenBank KF646249.1), following the manufacturer’s specifications (Advanced Cell Diagnostics, Newark, CA) and compared with the immunolabeling using anti-EHNV antibody.
Viral Isolation
Sections of the liver and kidney aseptically collected at necropsy were homogenized with 1 ml DMEM supplemented with 100× Antibiotic-Antimycotic (Thermo Fisher Scientific, NY, USA) and subjected to 3 freeze/thaw cycles at –20°C before clarification by centrifugation at 13 523 g for 5 minutes. Tenfold serial dilutions (1–10–5) were prepared from each sample, and a total of 50 μl was added in duplicate to 80% confluent monolayers of FHM cells in a 96-well tissue culture plate (SARSTEDT, Nümbrecht, Germany). The plates were incubated at 25°C and checked for cytopathic effect daily for 1 week. Supernatant from each sample was preserved at –20°C for further analysis.
Polymerase Chain Reaction
Total DNA was extracted from a 200-μl aliquot of the media that the cloacal swab sample was stored in and from small portions of the homogenized liver and kidney samples using a spin-column DNA purification procedure (Bioline ISOLATE II Genomic DNA Kit, animal tissue protocol; Bioline, Luckenwalde, Germany). Viral isolation supernatant was prepared using the same protocol following the preparation instructions for “cultured cells.” A single-round of PCR targeting the major capsid protein (MCP) region of the EHNV genome was performed using primers (forward primer, 5′- GACTGACCAACGCCAGCCTTAACG-3′; reverse primer, 5′-GCGGTGGTGTACCCAGAGTTGTCG-3′) designed by Jaramillo et al. 21 The reaction mixture was as follows: 1× GoTaq qPCR Mastermix (Promega, Madison, WI), 0.8 μM of forward and reverse primer, 2 μl of template DNA (≈80 ng), and nuclease-free water in a 20-μl reaction. Thermocycling parameters were as follows: 95°C for 2 minutes, then 40 cycles of [95°C for 5 seconds, 58°C for 10 seconds and 72°C for 15 seconds] with a final extension at 95°C for 2 minutes. All PCR tests were run with positive (BIV DNA) and negative controls (no template control).
Results
Gross Pathology
Lesions were detected at necropsy in the skin of dragons euthanized at 10, 12, and 14 dpi (Fig. 2). The skin of the distal limbs, particularly the digits, had multifocal swelling and small ulcerations, sometimes associated with keratin layer retention (dyskeratosis). The spleens of dragons euthanized at 3 dpi were diffusely congested. The spleens of dragons euthanized at 6, 8, 10, 12, and 14 dpi had multifocal white pinpoint foci of necrosis (Fig. 1). A few petechial hemorrhages were observed in dragons euthanized at 6, 8, 10, 12, and 14 dpi. Multifocal ecchymotic hemorrhages on the serosal surface of the intestines and stomach were present in dragons euthanized at 10 and 12 dpi. One individual euthanized at 8 dpi had petechial hemorrhages in the tongue.
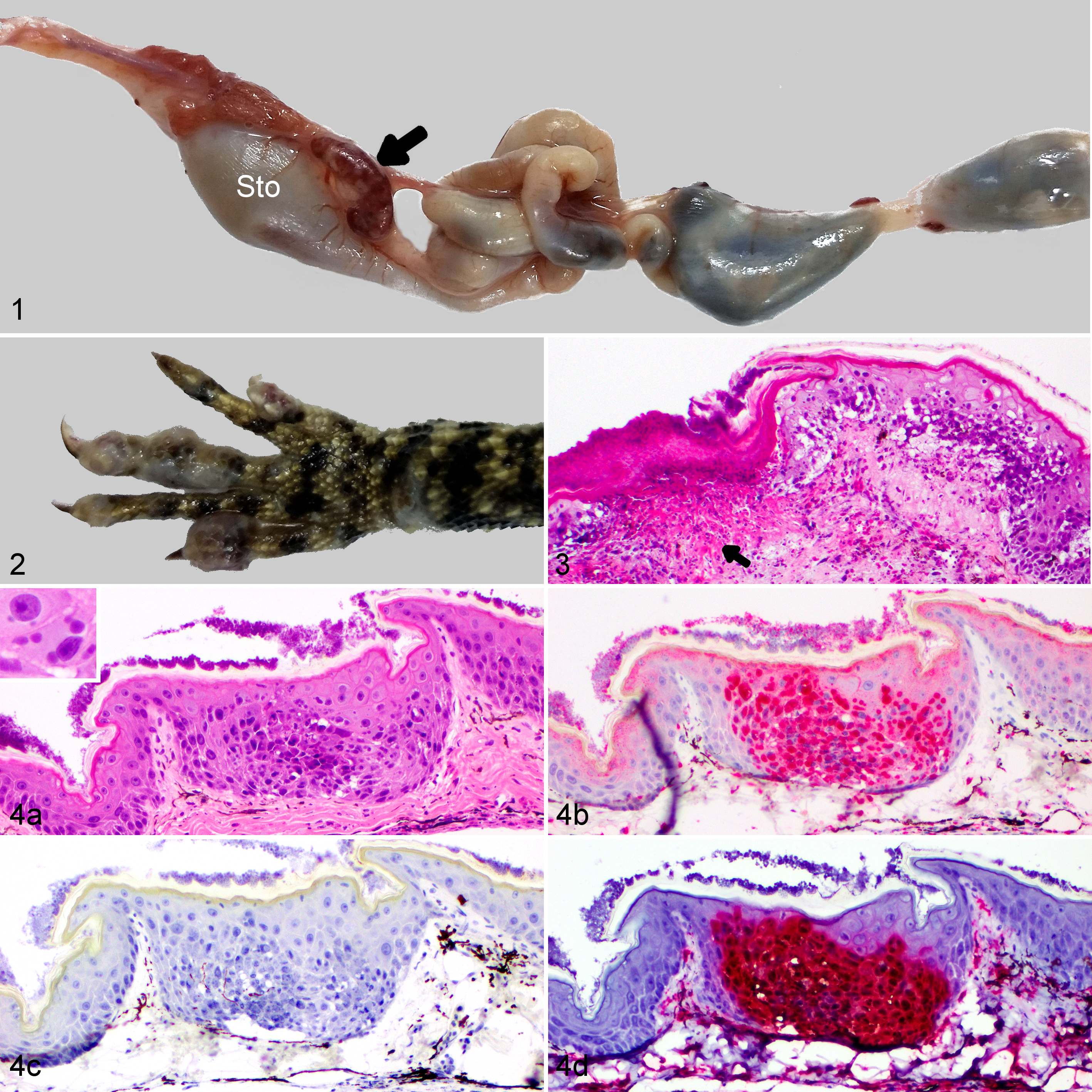
Bohle iridovirus experimental infection (104.33 TCID50 orally), eastern water dragon.
Histopathology
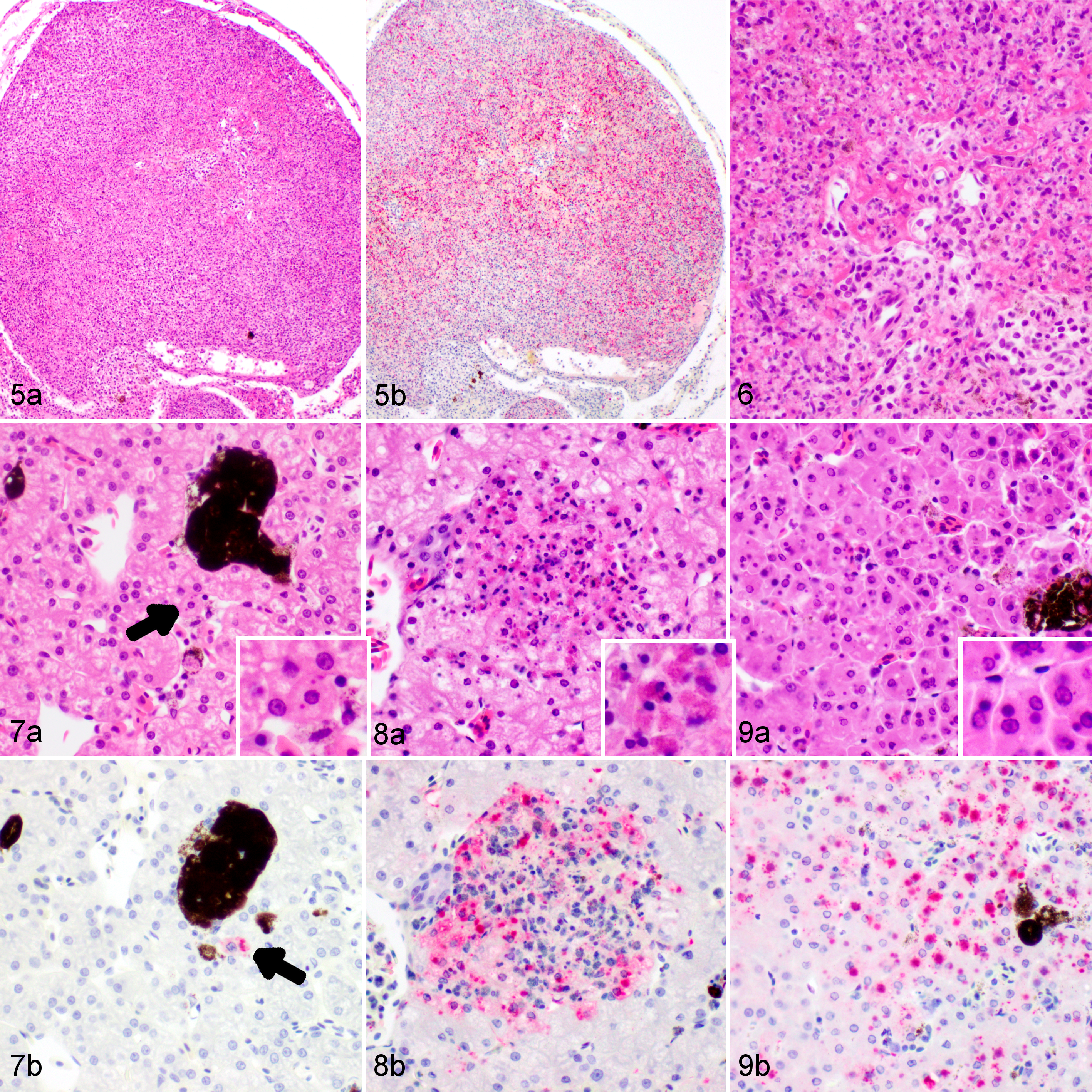
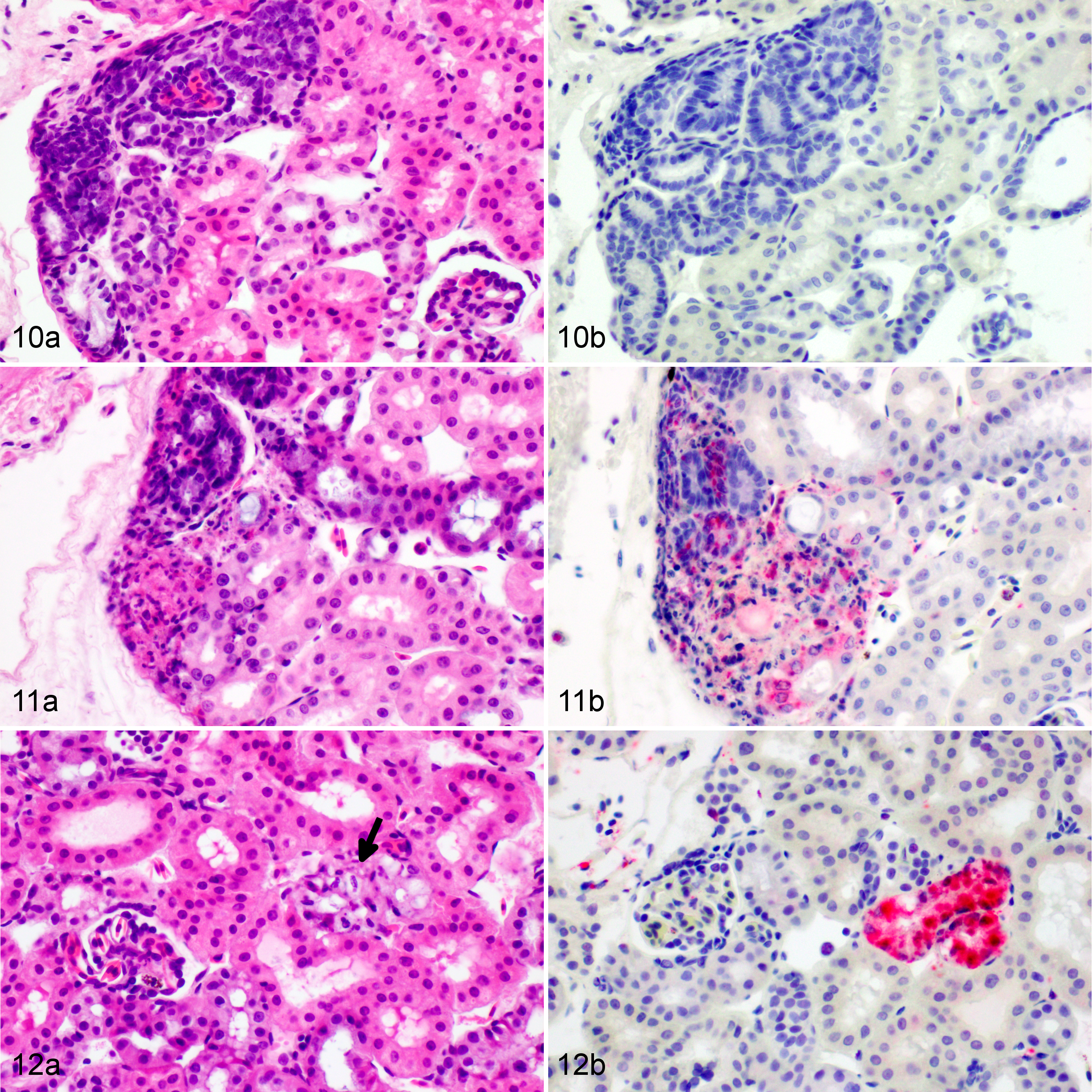
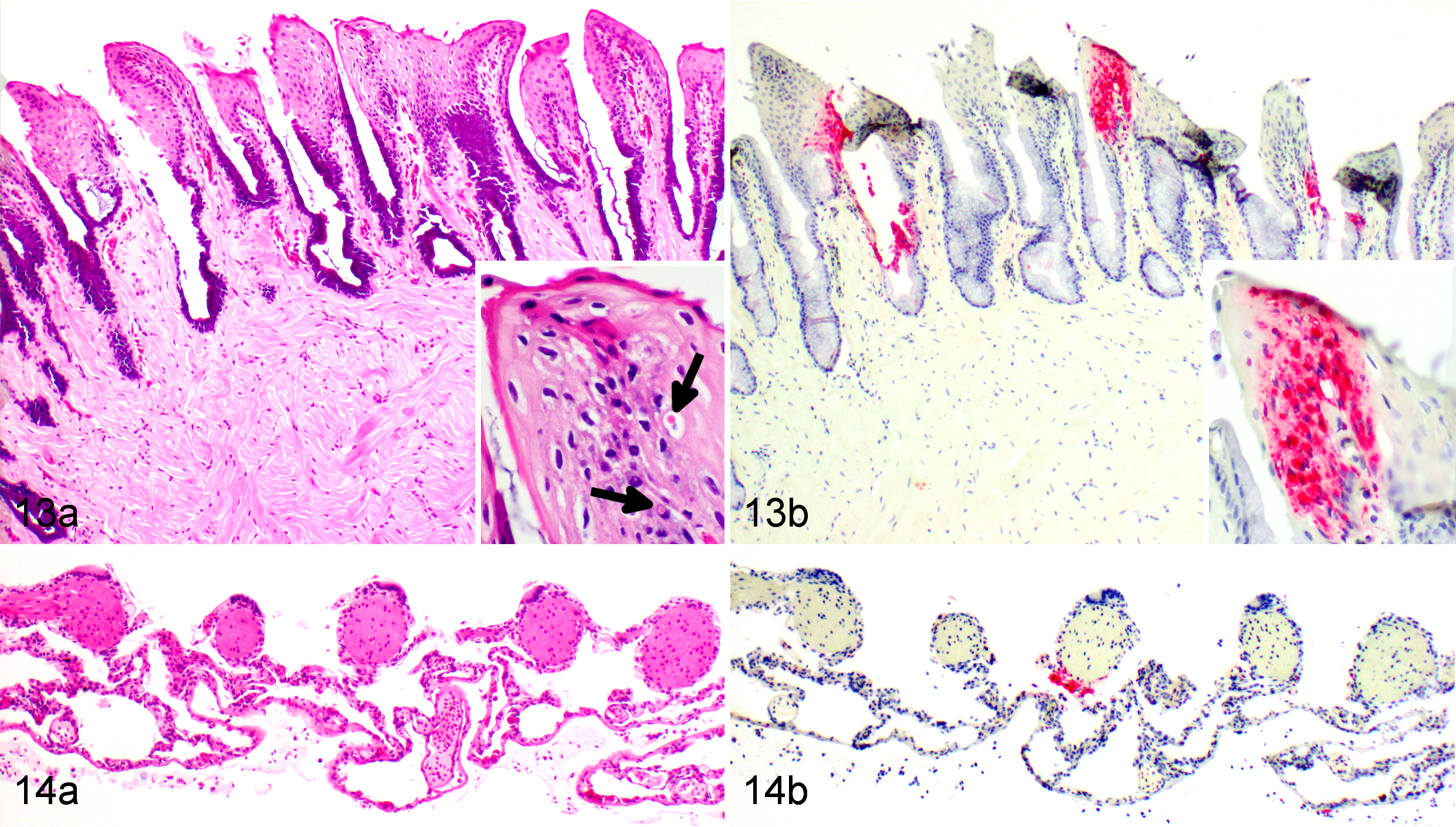
Histopathological changes are listed in Table 1. At 3 dpi, multifocal infiltration of lymphocytes was observed in the mucosa and submucosa of the tongue and in the liver. The liver had small foci of melanomacrophage aggregations and several small focal areas of lymphocyte accumulation. Necrosis was not observed in any tissue. Dragons euthanized at 6 dpi had moderate splenic necrosis (Figs. 5–6) and no lesions in any other viscera except for possible rare intracytoplasmic inclusions in hepatocytes (Fig. 7). At 8 dpi, there was multifocal to diffuse necrosis in the spleen with infiltration of macrophages. Dragons euthanized at 10 dpi had severe splenic necrosis, focal lymphocyte and granulocyte infiltration in the pancreatic connective tissue, and inflammation around the pancreatic duct and the submucosa of the duodenum adjacent to the pancreas. Variably sized basophilic intracytoplasmic inclusion bodies were observed in the hepatocytes and heterophils of the liver, which had mild necrosis and congestion (Fig. 8). Mild interstitial and tubular necrosis of the kidney was present (Fig. 11). Ballooning degeneration of epidermis with necrosis was associated with the skin lesions of 1 dragon (Fig. 3).
Histopathological Findings in Juvenile Eastern Water Dragons Infected Orally With 104.33 TCID50 of Bohle Iridovirus (Ranavirus sp) and Euthanized at Different Days Postinfection (dpi).a
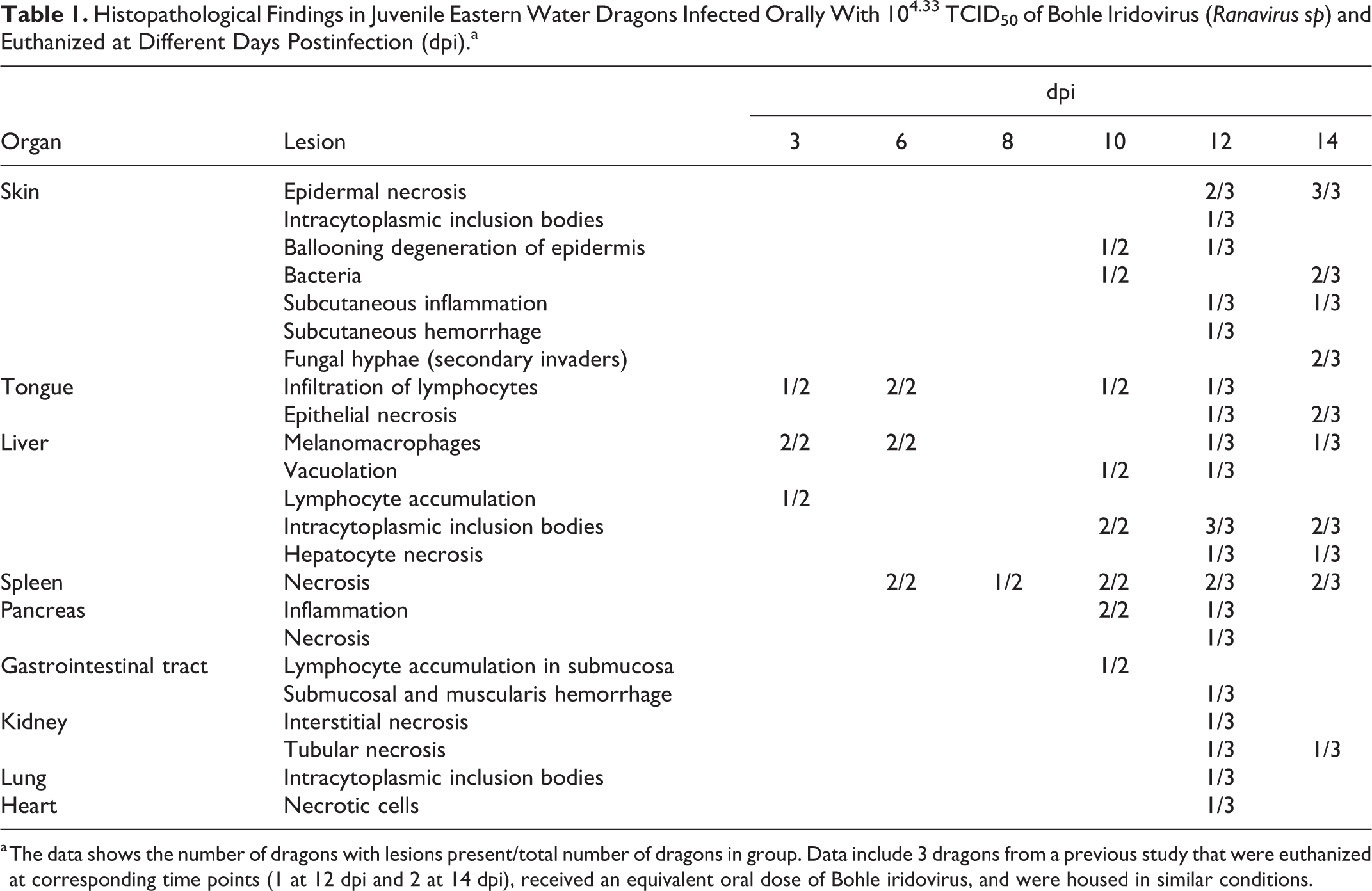
a The data shows the number of dragons with lesions present/total number of dragons in group. Data include 3 dragons from a previous study that were euthanized at corresponding time points (1 at 12 dpi and 2 at 14 dpi), received an equivalent oral dose of Bohle iridovirus, and were housed in similar conditions.
Bohle iridovirus infection, eastern water dragon.
Bohle iridovirus infection, kidney, eastern water dragon.
Dragons euthanized at 12 dpi had moderate to severe necrosis in the kidney, tongue, gastrointestinal tract, spleen, pancreas, and liver. The necrosis and hemorrhage in the spleen extended out through the splenic capsule and into the pancreas in 1 dragon. Intracytoplasmic inclusion bodies were observed within hepatocytes in the liver and in a consolidated section of the lung adjacent to the trachea. Multifocal subcutaneous hemorrhage and necrosis, as well as severe extensive multifocal subcutaneous necrosis with ballooning degeneration in epidermis, were observed in association with toe and leg lesions in 2 of 3 dragons euthanized at 12 dpi.
Dragons euthanized at 14 dpi had marked multifocal to coalescing necrosis in the liver, spleen, and kidney (Figs. 9, and 12). Mild to moderate necrosis of the tongue epithelium with small intracytoplasmic inclusions was present (Fig. 14). Scattered melanomacrophages were prominent in the liver, heart, and lung. Intracytoplasmic inclusion bodies were frequent in hepatocytes throughout the liver (Fig. 9). Multifocal epidermal necrotizing dermatitis and inflammation in the dermis and underlying connective tissue were observed in association with skin lesions (Fig. 4). In some areas, these lesions also had fungal hyphae extending into the keratin layer and bacteria in the superficial keratin, considered secondary invaders. No histopathological changes were observed in the brain of BIV-infected dragons or in any of the tissues of NC dragons.
Bohle iridovirus infection, eastern water dragon.
Immunohistochemistry and In Situ Hybridization
No immunolabeling was present in any tissue at 3 dpi (Table 2). Positive labeling with anti-EHNV antibody (Ranavirus sp) was first present in the spleen and liver at 6 dpi. The splenic staining was intense, generalized, and associated with necrosis, whereas the liver staining was sparse, often in individual or small clusters of cells, most of which showed mild degeneration or only small intracytoplasmic inclusion bodies. Rare hepatocytes stained positively at 6 dpi, usually in areas were small intracytoplasmic inclusions could be found but necrosis and inflammation were absent (Fig. 7). None of the other tissues showed any immunolabeling (eg, Fig. 10). Positive immunolabeling at 8 dpi was found only in the spleen, also in association with necrosis. At 10 dpi, positive immunolabeling in the spleen was associated with marked necrosis; immunolabeling was associated with mild to moderate necrosis in the liver (Fig. 8), skin, adrenal gland, kidney (Fig. 11), and pancreas. Positive immunolabeling without lesions was present in the bone marrow, lung (Fig. 14), and the endothelium and mesothelium of the stomach. At 12 dpi, immunolabeling associated with moderate to severe necrosis was present in the spleen, liver, skin, kidney, pancreas, lung, tongue, and gastric and intestinal mucosa; positive staining restricted to the endothelium was present in the bone marrow; positive staining restricted to the mesothelium was present in the ovary. At 14 dpi, all tissues examined were positive (Figs. 4, 9, and 13) except for the heart, which had included clusters of positive melanomacrophages in 1 of 2 EWDs euthanized at 12 dpi but was consistently negative in all other individuals at all other times. Brain and spinal cord were consistently negative on dragons euthanized at 6, 10, and 12 dpi. No brain tissue was available for testing from individuals euthanized at 14 dpi.
Immunohistochemical Labeling 29 of Selected Tissues From Juvenile Eastern Water Dragons Orally Infected With 104.33 TCID50 Bohle Iridovirus (Ranavirus sp) and Euthanized at Different Days Postinfection (dpi).
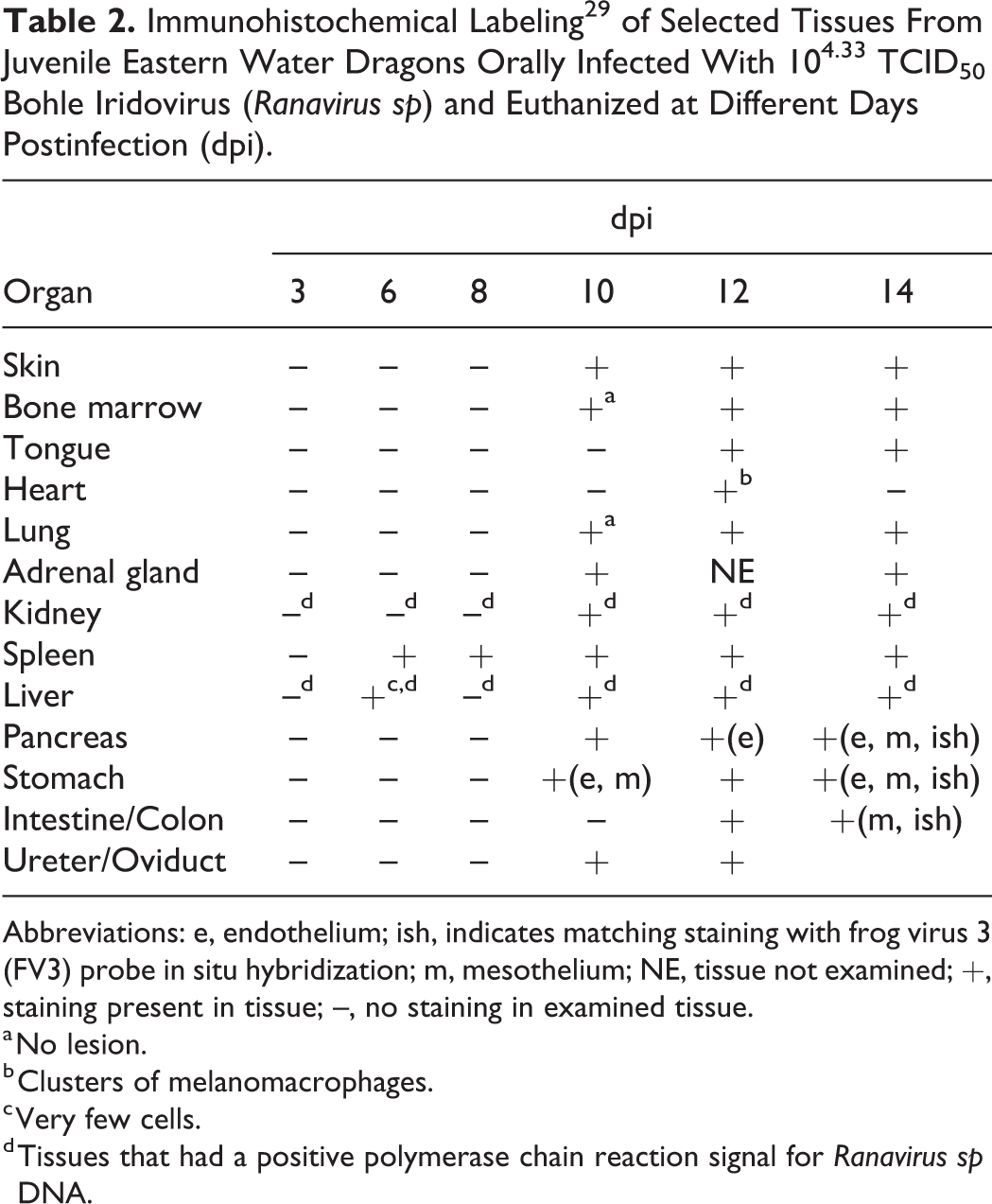
Abbreviations: e, endothelium; ish, indicates matching staining with frog virus 3 (FV3) probe in situ hybridization; m, mesothelium; NE, tissue not examined; +, staining present in tissue; –, no staining in examined tissue.
a No lesion.
b Clusters of melanomacrophages.
c Very few cells.
d Tissues that had a positive polymerase chain reaction signal for Ranavirus sp DNA.
Artifactual background staining was consistently present in the corneal (keratinized) layer of the epidermis, variably present in the glandular epithelium of the stomach and mucosal epithelium of the intestine. This artifactual staining was almost as intense as true immunolabeling in the skin but was present diffusely, was absent when the primary antibody was omitted from the protocol (Fig. 4c), and was found in a NC dragon known to be negative for BIV. Background staining was much paler than specific staining in the gastric and intestinal mucosa and thus easy to classify as artifact. Background staining was absent in all negative control slides (ie, those produced omitting the primary antibody).
The FV3 in situ hybridization (ISH) probe labeled skin (Fig. 4d), bone marrow, liver, pancreas, stomach, intestine, and spleen in the 1 individual tested, which was euthanized at 14 dpi. The ISH labeling matched the location and pattern detected by IHC. The background nonspecific IHC staining noted in the epidermis (Fig. 4b), gastric glandular epithelium, and intestinal mucosa did not correspond to the ISH staining, confirming it was an artifact and did not result from the presence of virus.
Viral Isolation
Virus was isolated from every liver and kidney sample from all experimentally infected EWDs at all time points tested. Cytopathic effect was observed in cell cultures after 96 hours of incubation and confirmed to be due to BIV by PCR on tissue culture supernatant.
Polymerase Chain Reaction
PCR for ranaviral DNA was positive for every sample (liver, kidney, and cloacal swab) from dragons euthanized at 3, 6, 8, 10, 12, and 14 dpi (Table 3). All positive PCR results corresponded to a positive IHC staining except for the liver from dragons euthanized at 3 and 8 dpi and the kidney from dragons euthanized at 3, 6, and 8 dpi (Table 2). All tissues from both control dragons (n = 2) were PCR negative for ranaviral DNA. Cloacal swabs collected prior to infection (day -1) from all experimental dragons were negative for ranaviral DNA.
PCR Testing for Ranaviral DNA in the Tissues of Juvenile Eastern Water Dragons Euthanized at Different Days Postinfection (dpi) After Oral Inoculation With 104.33 TCID50 Bohle Iridovirus (Ranavirus sp).a
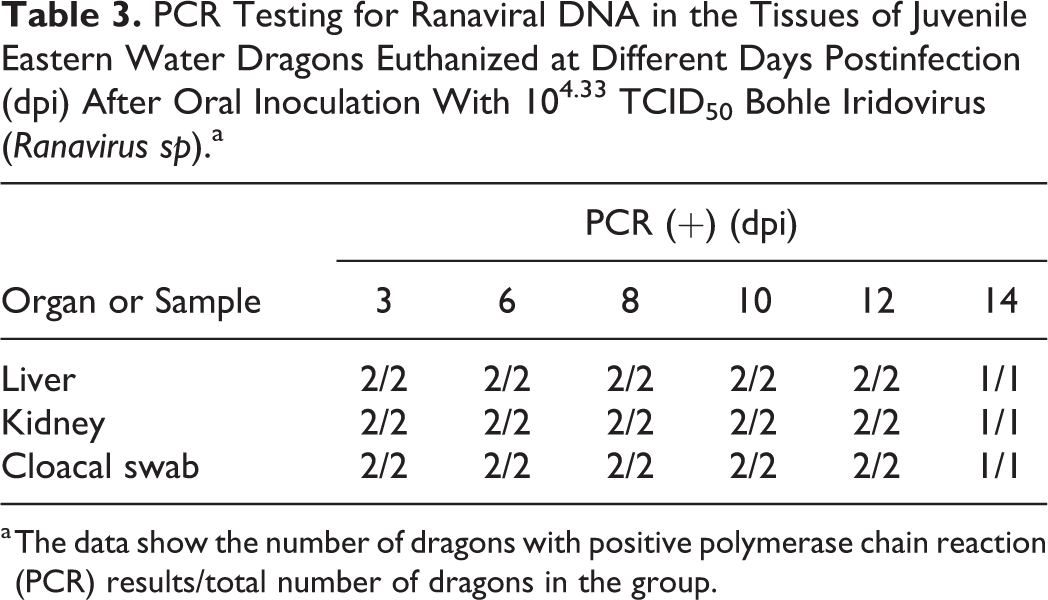
a The data show the number of dragons with positive polymerase chain reaction (PCR) results/total number of dragons in the group.
Discussion
Detailed examination of samples from BIV-inoculated juvenile EWDs revealed that viral infection and inflammation of visceral organs were well under way 4 days prior to onset of clinical signs (eg, skin lesions). Lesions caused by BIV infection in juvenile EWDs in this study were consistent with what we have recently described in fatally infected EWDs, 24 where multiple organs were affected. Maclaine et al 24 reported variably sized basophilic intracytoplasmic inclusion bodies near focal areas of necrosis in the liver, splenic necrosis, and multifocal renal interstitial and tubular necrosis in the terminal phase of infection. In the current study, lymphocyte accumulation was observed in the tongue and liver 72 hours after inoculation (3 dpi) but was not associated with positive IHC staining. Necrosis was not evident until 6 dpi, where it was observed in the spleen while inclusion bodies and early single-cell necrosis were found in the liver of infected dragons. Our findings suggest that BIV infection in juvenile EWDs induces cell death in the spleen between 3 dpi and 6 dpi, as evident by the positive IHC staining that was associated with mild necrosis, and, to a lesser extent, in the liver where IHC staining was often in individual cells showing early degeneration or inclusion bodies at 6 dpi. In samples collected at 10 dpi, the damage to kidney, liver, and spleen was more severe, with positive IHC staining associated with mild to moderate necrosis in the liver, and evident in the skin, adrenal gland, kidney, and pancreas. Positive IHC staining without lesions was observed in the lung, bone marrow, and endothelium and mesothelium of the stomach, indicating presence of virus in these areas, either in phagocytic or possibly antigen-presenting cells. All examined tissues from dragons euthanized on 14 dpi had positive IHC staining. The progression of infection in EWDs appears to start in the spleen, followed by the liver, then the other organs such as the kidney and pancreas, and subsequently oral mucosa and skin.
Immunolabeling with anti-EHNV antibodies was effective in demonstrating BIV presence in various tissues. However, caution should be exercised when interpreting IHC staining of sections of skin where artifactual background staining of the corneal layers was apparent, something that seems to be unavoidable in this species.
Inclusion bodies have previously been described in multiple organs of ranavirus-infected fish, tortoises, turtles, and amphibians 1,13,16,17,22,25,29,35 and, up until now, only in the liver of lizards. 8,24 While inclusion bodies are commonly associated with ranaviral infections, 8,17,18 they are not consistently reported. 2,5,34 This could be due to low numbers or sporadic occurrence of the viral inclusions, individual variation in host or strain, the viral load, duration of exposure, or a combination of these factors. In our study, variably sized intracytoplasmic inclusion bodies were observed in the liver of EWDs euthanized at 6, 10, 12, and 14 dpi but were not consistently found in all dragons euthanized at these time points. In addition, basophilic inclusion bodies were observed in a consolidated area of lung adjacent to the trachea and in association with skin lesions in dragons euthanized 10, 12, and 14 dpi (Figs. 3–4). An apparent delayed appearance of inclusion bodies in infected animals was also described in FV3-infected wood frogs (Rana sylvatica) euthanized at 9 and 14 dpi but was not a consistent finding across all individuals within those groups and not recorded in animals euthanized prior to this. 16 Although this is the first report of inclusion bodies in lungs of ranaviral-infected dragons, there are reports of inclusion bodies in keratinocytes adjacent to areas of necrosis in skin lesions of BIV-like infected boreal toads (Anaxyrus boreas boreas), in circulating leukocytes of a FV3-infected eastern box turtle (Terrapene carolina carolina), and in the cytoplasm of skin epidermal cells of FV3-infected wood frogs euthanized 14 dpi. 1,13,16
To identify the progression of BIV infection in EWDs at different time points, we collected cloacal swabs and tissue samples for PCR, virus isolation, histopathology, and IHC. In our study, Ranavirus sp DNA was detected in all cloacal swabs, as well as liver and kidney tissue samples collected at the first time point (3 dpi) in the early stages of infection, which contrasts to an FV3 pathogenesis study in wood frogs (Rana sylvatica) where DNA was not detected in multiple tissues until shortly before death. 16 Although the detection of ranaviral DNA in cloacal swabs could arguably be passage of the original inoculum from the mouth to the cloaca over 3 days postexposure, the early detection by PCR in liver and kidney samples was accompanied by an inflammatory response in the organs as visualized by histology and by virus isolation from these organs. The results from the IHC staining from both the wood frog study 16 and the study reported here did not appear to be as sensitive as PCR, and positive immunolabeling of organs was more consistently identified later in the trials than PCR-positive results for the same organs. Alternatively, the positive virus isolation and PCR result from kidney and liver could reflect the presence of virus in blood cells (viremia), as it disseminated systemically from its site of entry in the digestive tract. The presence of nonspecific staining of the keratin layer and the absence of IHC staining in positive PCR tissues could be addressed by a more specific and sensitive technique such as ISH 16 (as seen in Fig. 4b,d).
Several factors may influence the outcome of the exposure to infection in a given host. Although infection caused acute and fatal disease affecting multiple organs in this study, it could have a different outcome with other doses, routes of infection, or environmental conditions. Environmental temperature has been shown to affect the disease progression and survival in ranavirus-infected fish, amphibians, and turtles 2,23,31 but has not been explored in ranavirus-infected lizards. This experimental infection was conducted using male and female juvenile EWDs during spring when the air temperature was within the preferred body temperature range of this species 36 and resembled temperatures in the wild and therefore reflective of the seasonal conditions. Dose-dependent studies have shown that the viral load affects the severity and type of lesions, as well as the chance of survival. 17 Dragons in this study were infected with a high dose of BIV that caused an acute infection with a sudden onset. The dose used in this study was equivalent to the dose used by Maclaine et al, 24 where it caused high mortality in EWDs exposed orally, intramuscularly, and via cohabitation. This may not reflect natural BIV infection where the infective dose is likely less and the infection is possibly protracted. Because the dragons were housed communally in small groups, we cannot rule out the possibility that reinfection from contact with shedding cohorts could have exacerbated the severity of lesions or modified the course of infection. However, cohabitation with orally infected lizards under similar experimental conditions showed a delay of 9.5 days for appearance of clinical signs, indicating that orally infected lizards were shedding virus around 9.5 dpi, and in this case, the histology, PCR, and viral isolation of animals housed together indicated that they were initially infected on 0 dpi with the massive oral dose they received.
The effect of different Ranavirus sp isolates and how they interact with different reptilian hosts has not been fully explored, but there appear to be similarities in the clinical signs and histopathological changes observed in lizards that were naturally infected with ranavirus. Skin lesions observed in dragons infected with BIV in this study were most commonly ulcerative and pustular and located on the distal limbs and digits, in contrast to brown-crusted skin lesions observed on the ventral abdominal surface and dorsum of ranavirus-infected Asian glass lizards (Dopasia gracilis). 33 In the current study, bacteria in the superficial keratin and fungal hyphae extending into the keratin layer in association with lesions were considered secondary invaders and have previously been reported in association with skin lesions of lizards infected with other Ranavirus sp. 24,33,34 Skin lesions are believed to be a result of the ranaviral infection, and an intersegment breach in the skin barrier allows for entry of other microorganisms such as bacteria and fungi. Liver necrosis and basophilic intracytoplasmic inclusion bodies in the liver observed in infected EWDs were similar to those observed in green striped tree dragons 8 but with the absence of bacterial colonies that were observed in a leaf tail gecko (Uroplatus fimbriatus). 26 Mild interstitial and tubular necrosis was observed in the kidney of infected EWDs in contrast to the vacuolar tubulonephrosis of the distal renal tubules reported in green striped dragons. 8 Another difference between lesions in the kidney of frogs infected with FV3 16 is the relatively late appearance of lesions in the kidney of our dragons infected with BIV. Although this could be associated with different affinities in each species of ranavirus, it may simply reflect different anatomical features between amphibians and reptiles. Lesions in amphibian kidneys are often associated, at least partially, with their interstitial hematopoietic tissues. The kidneys of our water dragons, like those of most reptiles, did not appear to contain hematopoietic tissues and thus lacked the tissue targeted by ranaviruses in amphibians.
Previous reports of ranavirus infection in lizards are from captive lizards that presented with skin lesions or were investigated following high mortalities or sudden death. 8,26,33,34 As with most case reports or case series, ranavirus was found to be the cause at postmortem examination, but not always considered by clinicians upon presentation. Our findings show that clinical signs of ranavirus infection in water dragons, such as inappetence and skin lesions, are nonspecific and appear in the late stages of infection. Further hampering the clinical (antemortem) diagnosis of ranavirus in skin lesions of lizards are secondary fungal or bacterial infections. We recommend that veterinarians consider ranavirus as a differential diagnosis in fatal outbreaks of skin lesions in lizards.
Maclaine et al 24 reported that naive EWDs can be infected by direct contact with BIV-infected EWDs, although it is not understood whether the transmission is caused by ingestion of infected excreta or water, or contact with skin lesions. Viral shedding from skin lesions, from gastrointestinal mucosa or from infected renal tubular cells, may lead to transmission by direct contact with skin or by ingestion or contact with infected excreta or water, respectively. In this study, ranaviral DNA was detected in cloacal swabs at all time points, as well as in all kidney and liver samples.
Here we report the progression and effect of BIV in experimentally infected juvenile EWDs. Ranavirosis became clinically evident at 7 dpi. Virus-associated histologic lesions were not observed at 3 dpi but were observed at 6 dpi and subsequent times. BIV may travel by blood within macrophages and other white blood cells, entering endothelial cells and then passing into tissue from either apoptotic endothelial cells or with migrating macrophages. The early detection of ranaviral DNA in cloacal swabs and liver and kidney tissue samples suggests these are a reliable source of diagnostic samples in the early stage of disease before the appearance of clinical signs, as well as throughout the infection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by an Australian Government Research Training Program Scholarship. We thank Robin T. Pasiak and Melissa Fadden, Cornell University, for validating and performing the IHC and ISH staining, respectively, and Wytamma Wirth, James Cook University, for his support. The BIV isolate and control DNA was kindly provided by Professor Richard Whittington, Dr Paul Hick, and Alison Tweedie, Sydney University.
