Abstract
Selenosis, or selenium toxicosis, occurs in wildlife and livestock, usually because of excessive intake of selenium via selenium-containing plants. Although it is known that wild slider turtles can accumulate large amounts of selenium, little is known about how selenium exposure may affect these reptiles. In this study, the authors report histopathologic changes in yellow-bellied sliders (Trachemys scripta scripta) caused by experimental exposure to selenomethionine. Microscopic changes in kidney and claw tissue were most significant and resembled those reported in birds. Turtles in the selenium treatment groups had acute tubular degeneration and regeneration in the kidney, with hyaline droplets in the high-dose animals, and changes in the claws ranging from epidermal hyperplasia with disorganization and intercellular edema to ulceration, and accumulation of seroheterophilic exudate between the epidermis and cornified layer. Although selenium burdens in this study are comparable with values found in wild slider turtles, more data are needed to determine if similar histopathologic abnormalities arise in wild animals exposed to high levels of selenium.
Selenosis, or selenium toxicosis, is usually the result of excess intake of seleniferous forages but can also occur following parenteral administration. It is characterized by teratogenesis, morbidity, and mortality in a variety of vertebrate species. 8,12 Previous studies have shown that livestock and aquatic bird species are sensitive to excess amounts of selenium. When exposed to toxic levels of selenium, birds had microscopic changes in the liver, kidneys, and keratinized tissues (eg, claws). 8 Feather loss, liver necrosis, and mild nephrosis were all common in American coots, pied-billed grebes, and mallards exposed to excess amounts of selenium. 8,13 Selenosis in livestock causes a condition called “alkali disease,” which is characterized by lack of energy, anemia, sloughing of hooves, damage to other keratinized tissues, and lameness. 15
Although the effects of selenosis have been described in livestock and waterfowl, the effects of this condition in reptiles are poorly documented. In fact, aside from an exposure experiment with American alligators (Alligator mississippiensis), there are no reports of selenosis in reptiles. 3 However, previous research shows that slider turtles in selenium-contaminated environments can accumulate large amounts of selenium and other inorganic contaminants. 9 Inorganic forms of selenium, such as selenium salts found in coal ash wastes, can be transformed into organic forms (eg, selenomethionine) by bacteria and plants, which then can be transferred through the food web. 18 Many species of reptiles are also long-lived (eg, testudines and crocodilians), which suggests that their exposure could be much longer relative to other vertebrate species. Coal combustion residues, which are typically rich in selenium, cause histologic changes in liver tissue of southern banded watersnakes (Nerodia fasciata fasciata), 6 and these findings suggest that chronic exposure to coal combustion contaminants could lead to deleterious effects (such as negative impacts on reproduction or survival) in watersnakes.
Yellow-bellied sliders (Trachemys scripta scripta) are geographically widespread, have well-characterized natural and life histories, and can represent the greatest biomass of turtles within aquatic ecosystems in the southeastern United States. 2,6 In the summer of 2015, we exposed slider turtles to selenium to examine its impact on their hematology, immune response, and metabolism. 10 Because we observed signs indicative of selenosis, we collected samples for histologic analysis to determine if selenium exposure caused changes in turtle tissues. Overall, the objective of this study was to determine if histologic changes occur in tissues from yellow-bellied slider turtles associated with experimentally induced selenosis.
Seventy slider turtles (weight range, 154–320 g; plastron length range, 84–111 mm) were obtained from Concordia Turtle Farms and randomly placed into 1 of 3 treatment groups. Sliders were dosed weekly via oral gavage (∼300 μL) with 0 mg/kg (n = 24), 15 mg/kg (n = 23), or 30 mg/kg (n = 23) seleno-
In addition to tissues being harvested for selenium analysis, liver, kidney, skeletal muscle, and claws, from a subset of each treatment group (0 mg/kg, n = 6; 15 mg/kg, n = 5; 30 mg/kg, n = 7), were collected in 10% buffered formalin for histologic analysis. Claws were decalcified in a 14% neutral ethylenediaminetetraacetic acid solution. Formalin-fixed tissues were processed routinely and embedded in paraffin, and 4-μm sections were stained with hematoxylin and eosin. Additional sections were stained with a modified Brown and Brenn gram stain and Grocott’s methenamine silver stain for bacteria and fungus, respectively. For each tissue, severity of change was subjectively scored from 0 to 3, where 0 = none, 1 = mild, 2 = moderate, and 3 = severe change. The “grouped” method of masking tissues for histopathologic examination was used. 7 Samples were coded by group, and the pathologist knew the study design. A series of Fisher exact tests of independence (α = 0.05) were used to compare proportions of individuals exhibiting histologic changes in claw, liver, kidney, and muscle tissues among treatment groups.
In our primary experiment, slider turtles treated with the highest dose of SeMet exhibited multiple signs of selenosis, including negative hematologic effects (eg, reduced lymphocyte counts, increased heterophil counts, and hemolytic anemia), sloughing of claws, and mortality. 10 Selenium-treated turtles from which tissues were collected for histopathology all had tissue selenium levels that surpassed “level of concern” thresholds established for avian species. 10,14 Average tissue selenium concentrations from turtles in the present histology study were highest in the kidney (0 mg/kg, 4.37 ± 0.38 mg/kg; 15 mg/kg, 136.31 ± 7.96 mg/kg; 30 mg/kg, 237.71 ± 15.38 mg/kg), followed by the liver (0 mg/kg, 1.81 ± 0.26 mg/kg; 15 mg/kg, 30.21 ± 4.07 mg/kg; 30 mg/kg, 75.29 ± 15.10 mg/kg) and muscle (0 mg/kg, 1.95 ± 0.19 mg/kg; 15 mg/kg, 29.28 ± 1.53 mg/kg; 30 mg/kg, 41.15 ± 1.81 mg/kg). 10 All 4 tissues examined had changes that were either not seen in the controls (claw and kidney) or were more severe than in the controls (muscle and liver) (Table 1).
Histologic Lesions in Yellow-Bellied Slider Turtles After Administration of 0, 15, or 30 mg/kg Seleno-
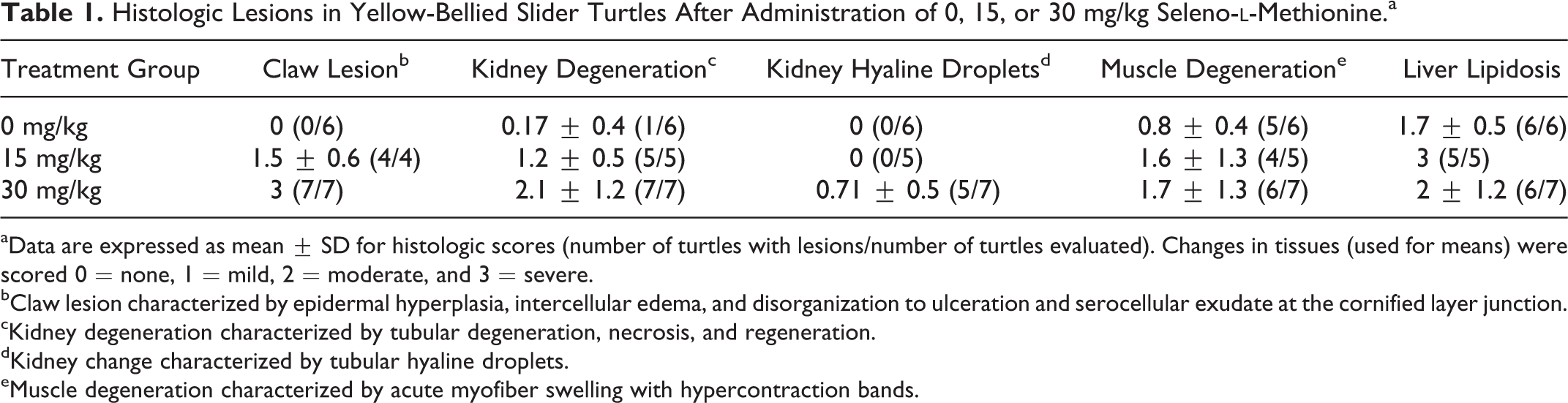
aData are expressed as mean ± SD for histologic scores (number of turtles with lesions/number of turtles evaluated). Changes in tissues (used for means) were scored 0 = none, 1 = mild, 2 = moderate, and 3 = severe.
bClaw lesion characterized by epidermal hyperplasia, intercellular edema, and disorganization to ulceration and serocellular exudate at the cornified layer junction.
cKidney degeneration characterized by tubular degeneration, necrosis, and regeneration.
dKidney change characterized by tubular hyaline droplets.
eMuscle degeneration characterized by acute myofiber swelling with hypercontraction bands.
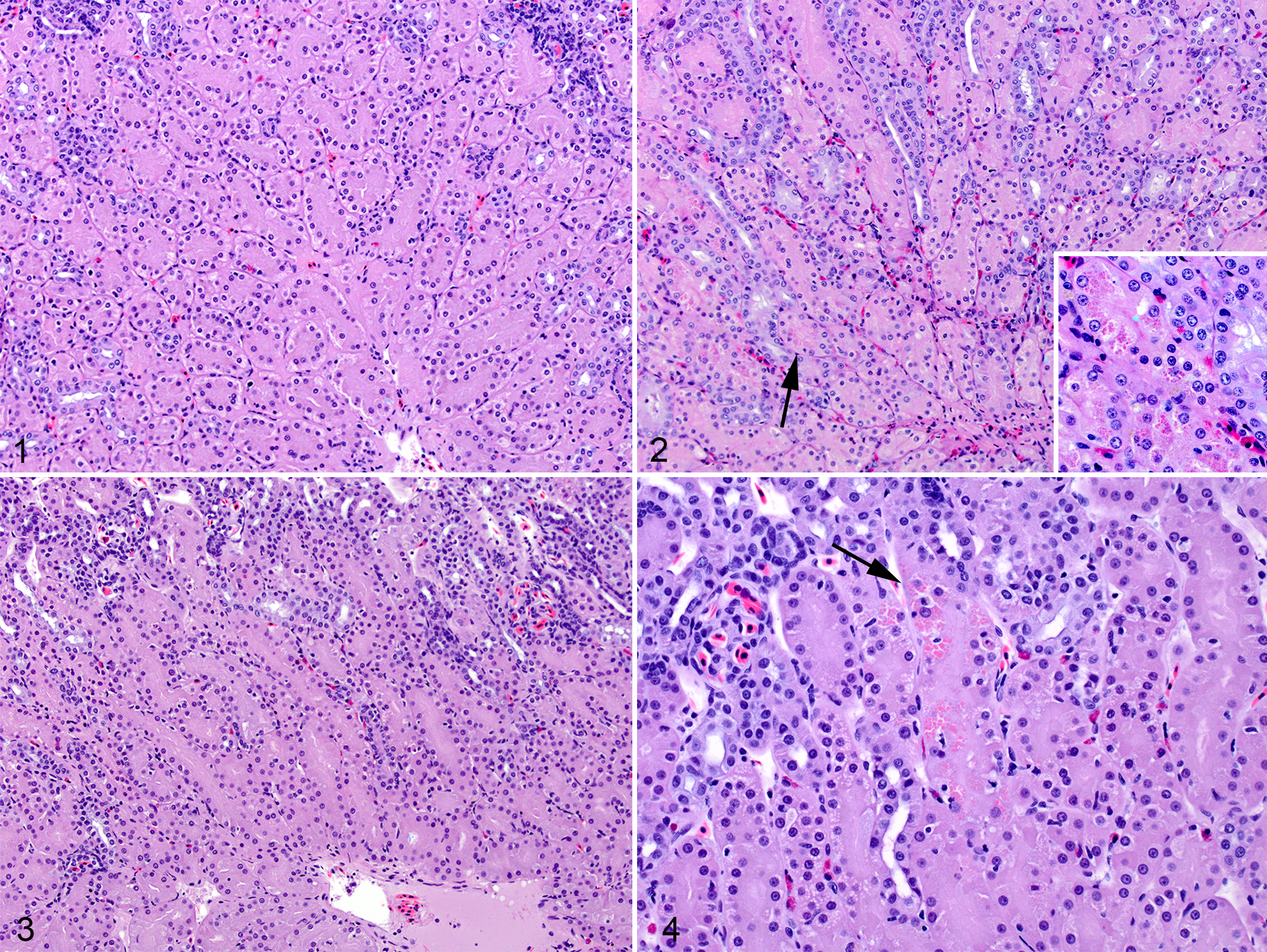
In kidney (Figs. 1–4), there was tubular degeneration, necrosis, and regeneration in both selenium-dosed groups. The histologic scores were higher in the selenium-dosed group relative to the control group (P = .0008). Abnormalities were more severe in the high-dose group: although both low-dose and high-dose scores ranged from 1 to 3, more high-dose turtle kidneys were scored 3 (Suppl. Table S1). Histologic scores in controls were all 0. Hyaline droplets were seen in the tubular epithelium in 5 of 7 of the high-dose turtles with scores of 2 or 3, but not in the control or low-dose groups (P = .003).
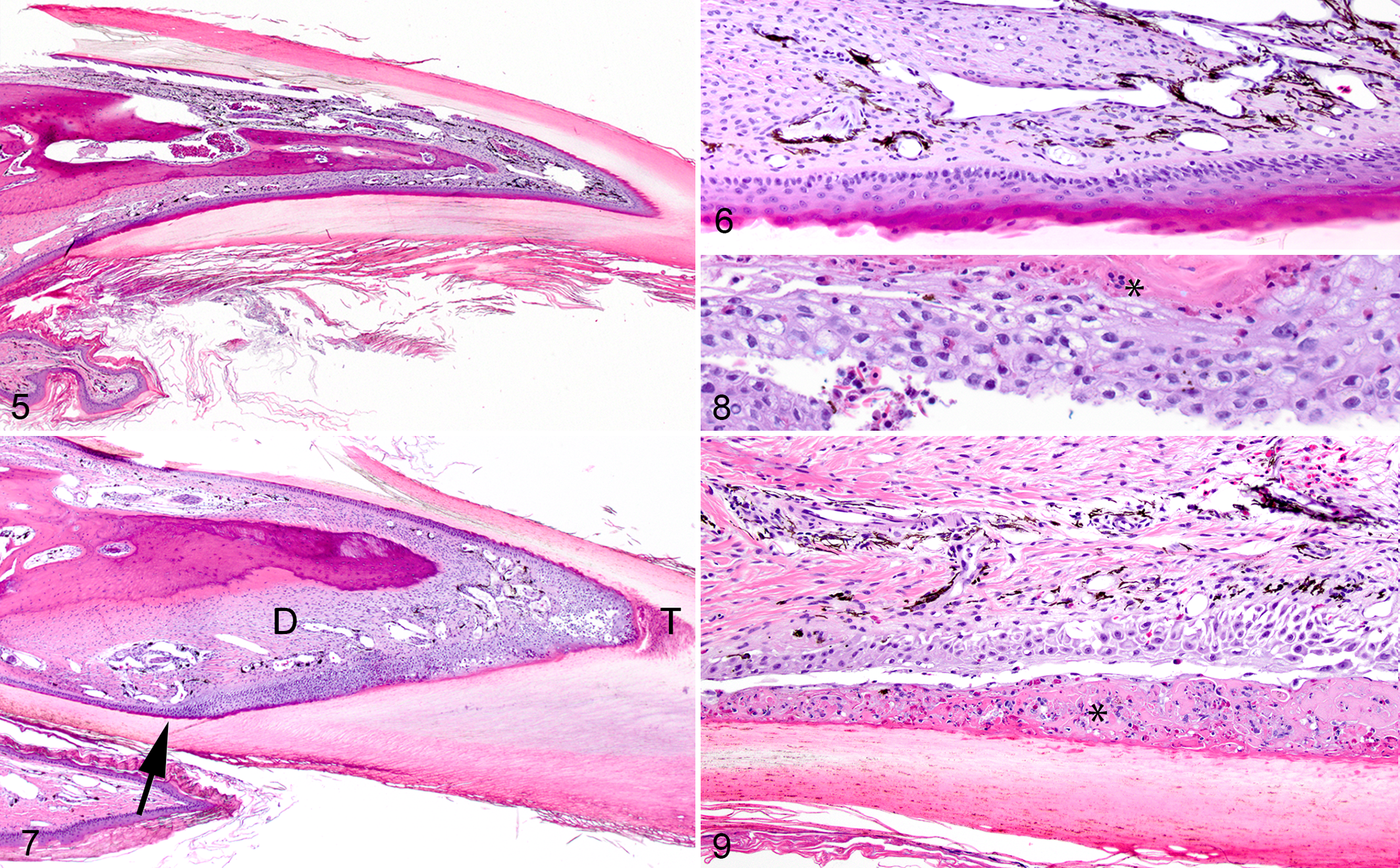
Claws from control group turtles had no changes (Figs. 5 and 6), while claws from turtles of both selenium treatment groups had similar changes (P = 8.08 × 10–5), but these were more severe in the high-dose group (low-dose score range, 1–3; high-dose scores were all 3). Changes in the epidermis were most severe over the claw tip and along the palmar/plantar surface and included epidermal hyperplasia with disorganization, vacuolar degeneration, intercellular edema, necrosis, and ulceration (Figs. 7–9). Inflammatory cells and proteinaceous material separated the epidermis or dermis from the stratum corneum. The dermis was mildly edematous and thickened by fibrosis with a mild lymphocytic infiltration. Infectious agents were not identified on gram and Grocott’s methenamine silver stains.
Hepatic lipidosis was present in all turtles except 1 in the high-dose group (P = 1.00 for all comparisons). In this individual, hepatocytes were small with glassy cytoplasm consistent with atrophy, probably indicating emaciation. Lipidosis was most severe in the low- and high-dose groups (control score range, 1–2; low-dose score range, 2–3; high-dose score range, 1–3, excluding the turtle that did not have hepatic lipidosis).
In the skeletal (pectoralis) muscle of all groups, most turtles had rare to many myofibers with acute degenerative changes characterized by hyalinization and hypercontraction bands (P = 1.00 for all comparisons). This was most apparent in the high-dose group (score range, 1–3), followed by the low-dose group (score range, 0–3) and control group (score range, 0–1).
Similar to these turtles, changes in keratinized structures such as hooves, claws, and beaks are a major sign of selenosis in many species of mammals and birds. 15,16 In humans, in whom there has been an increase in cases of selenosis due to misformulated dietary supplements, nail loss (onycholysis and onychoptosis) is also a common finding. 13 The microscopic lesions in these turtles were similar to the more severe lesions described in experimental selenosis of mallard ducks, in which there was epidermal necrosis and accumulation of cellular and proteinaceous material that separated the cornified layer from the dermis. 16 Those birds also had similar changes in the maxillary beak. It is also possible these turtles had beak lesions, but unfortunately the beaks were not examined.
Renal lesions, although mild, were the other prominent finding of selenosis in these turtles. High levels of orally administered selenium were associated with tubular damage whose severity varied in a dose-dependent manner. There were also hyaline droplets in the tubular epithelium of the high-dose group, suggesting proteinuria. 4 Lesions similar to those described in these turtles have been reported in mallards with experimental selenosis, 8 and renal calcinosis has been attributed to selenium toxicosis in trout. 11
Acute degenerative changes in skeletal muscle were present in both control and treated groups, so they were probably associated with stress and handling at the time of euthanasia. However, changes were more pronounced in the treated groups, suggesting that exposure to high levels of selenium might make muscles more prone to exertional rhabdomyolysis.
Factors other than high selenium levels, such as a high-energy diet or an inadequate photoperiod, were likely the primary cause of hepatic lipidosis in these turtles, as it was observed in both control and treated groups. However, lipidosis was more severe in the treated groups, suggesting that high levels of selenium may also have had an adverse effect on the liver or on energy metabolism. Watersnakes fed a contaminated diet that contained selenium as well as other trace elements had hepatic changes. 5 Although changes were described as fibrosis, this could have been due to the extended length of time the snakes received the contaminated diet. Mallards with experimentally induced selenosis also had liver lesions, although different in character to the lipidosis described here. 8
Selenosis in slider turtles was associated with changes in tissues that are similar to those reported in avian species. The most notable changes in this study were found in kidney and claw tissues, with the high-dose group exhibiting the most severe changes. Selenium blood and liver content in our sliders from this study are comparable with levels found in wild sliders in coal-contaminated environments. 10 However, more research is needed to determine if exposure in the wild results in similar histopathologic abnormalities in slider turtles.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0300985817750454 - Experimentally Induced Selenosis in Yellow-Bellied Slider Turtles (Trachemys scripta scripta)
Supplemental Material, DS1_VET_10.1177_0300985817750454 for Experimentally Induced Selenosis in Yellow-Bellied Slider Turtles (Trachemys scripta scripta) by David L. Haskins, Elizabeth W. Howerth, and Tracey D. Tuberville in Veterinary Pathology
Footnotes
Acknowledgments
We would like to thank Caitlin Kupar, Matt Hamilton, Katrina Woods, Naya Eady, Jarad Cochran, Amanda Jones, and Sam Dean for their help with animal husbandry, selenium administration, turtle dissections, and tissue preparation. This research was funded by the US Department of Energy under award number DE-FC09-07SR22506 to the University of Georgia Research Foundation and by Savannah River Nuclear Solutions – Area Completions Project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
